ABSTRACT
Introduction and objectives: Calcified coronary nodules (CN) are associated with suboptimal outcomes following percutaneous coronary intervention. The aim of this study is to compare the safety and efficacy profile of orbital atherectomy (OA) and intravascular lithotripsy (IVL) in the management of CN.
Methods: The ORBIT-SHOCK pilot study is a randomized, multicenter clinical trial designed to enroll 50 patients with coronary CN identified by optical coherence tomography and deemed suitable for percutaneous coronary intervention. Patients will be randomly assigned in a 1:1 ratio to receive either OA or IVL, with stratified randomization based on the type of CN (eruptive or noneruptive) to ensure balanced distribution across treatment groups. The primary endpoint is stent expansion at the site of the CN, assessed by optical coherence tomography, while secondary endpoints will include procedural success, strategy success, stent apposition, and the rate of adverse clinical events such as target lesion failure, target vessel failure, and major adverse cardiovascular events at the 12-month follow-up.
Conclusions: The ORBIT-SHOCK pilot trial is designed as the first randomized clinical trial to compare the safety and efficacy of OA and IVL in the management of CN. (ClinicalTrials.gov: NCT06736665).
Keywords: Coronary calcification. Calcified nodules. Orbital atherectomy. Intravascular lithotripsy. Percutaneous coronary intervention. Complex PCI.
RESUMEN
Introducción y objetivos: Los nódulos coronarios calcificados (NC) se asocian con resultados subóptimos tras la intervención coronaria percutánea. El objetivo de este estudio es comparar la eficacia y la seguridad de la aterectomía orbital (AO) y la litotricia intravascular (LIV) en el tratamiento de los NC.
Métodos: El estudio piloto ORBIT-SHOCK es un ensayo clínico aleatorizado y multicéntrico que incluirá 50 pacientes con NC identificados mediante tomografía de coherencia óptica y candidatos a intervención coronaria percutánea. Los pacientes se asignarán aleatoriamente en una proporción 1:1 para recibir AO o LIV, con una aleatorización estratificada basada en el tipo de NC (eruptivo o no eruptivo) para garantizar una distribución equilibrada entre los grupos de tratamiento. El objetivo primario del estudio es la expansión del stent en el sitio del NC evaluada por tomografía, mientras que los objetivos secundarios son el éxito del procedimiento, el éxito de la estrategia, la aposición del stent y la incidencia de eventos clínicos adversos, como el fallo de la lesión diana, el fallo del vaso diana y los eventos cardiovasculares mayores a los 12 meses de seguimiento.
Conclusiones: El ensayo piloto ORBIT-SHOCK será el primer estudio aleatorizado para comparar la eficacia y la seguridad de la AO y la LIV en el tratamiento de los NC. (ClinicalTrials.gov: NCT06736665).
Palabras clave: Calcificación coronaria. Nódulos calcificados. Aterectomía orbitacional. Litotricia intracoronaria. Intervención coronaria percutánea. ICP compleja.
Abbreviations
CN: calcified nodule. IVL: intravascular lithotripsy. OA: orbital atherectomy. OCT: optical coherence tomography. PCI: percutaneous coronary intervention.
INTRODUCTION
Calcified nodules (CN) represent a significant challenge during percutaneous coronary intervention (PCI), as their mechanical resistance limits effective lesion preparation and adequate stent expansion, resulting in suboptimal angiographic outcomes and a higher risk of adverse clinical events.1-7
Intracoronary optical coherence tomography (OCT) is highly effective for the detection and characterization of coronary calcium, offering superior sensitivity and specificity than angiography. Robust evidence demonstrates that OCT-guided PCI improves both procedural and clinical outcomes, supporting guideline recommendations for its use in complex coronary lesions.8-11
Several dedicated devices and techniques have been developed to facilitate calcium modification and optimize lesion preparation. Among these, intravascular lithotripsy (IVL) has emerged as a widely adopted strategy for the treatment of CN because of its ease of use, low complication rates, and favorable outcomes in recent studies.2,3,7,12
Alternatively, orbital atherectomy (OA) has been shown to be an effective strategy for the treatment of severely calcified and non- dilatable coronary lesions. However, evidence regarding its performance in CN remains limited and is largely derived from small observational studies and case reports.1,12-23
Despite major advances in imaging and plaque-modification technologies, the optimal management strategy for CN remains uncertain.
METHODS
Population and study design
The ORBIT-SHOCK pilot study is a multicenter, prospective, investigator-initiated randomized pilot study, sponsored by the Spanish Society of Cardiology. It will include patients diagnosed with atherosclerotic disease with evidence of CN identified by OCT, causing significant angiographic stenosis eligible for revascularization through PCI.
A CN, as identified by OCT, is defined as a localized protruding mass within the coronary artery wall that appears as a signal-poor region with sharply delineated borders. An eruptive CN is characterized by an accumulation of small calcium fragments that protrude through and disrupt the overlying fibrous cap, typically associated with small amount of thrombus. In contrast, a noneruptive CN is defined as an accumulation of small calcium fragments covered by a smooth intact fibrous cap without overlying thrombus.1
Inclusion and exclusion criteria are detailed in table 1. The selection of eligible patients will be conducted by the investigators at each participant center. Following confirmation of the presence of a CN and verification of compliance with the inclusion criteria, and absence of exclusion criteria, patients will be randomized in a 1:1 ratio to 1 of the 2 lesion preparation strategies under study: OA or IVL (figure 1). Randomization will be stratified according to the type of CN (eruptive or noneruptive) to ensure a balanced distribution across treatment groups. Randomization occurs immediately after OCT confirmation of the presence of CN and before any plaque-modification strategy has been started.
Table 1. Inclusion and exclusion criteria
| Inclusion criteria |
|---|
| Patients aged ≥ 18 years |
| Atherosclerotic disease with calcified nodules identified by OCT in a native vessel eligible for percutaneous coronary revascularization |
| Clinical presentation with chronic coronary syndrome or non-ST-segment elevation acute coronary syndrome. Nonculprit lesions suitable for revascularization during a staged procedure after ST-segment elevation myocardial infarction (STEMI) will also be eligible for inclusion |
| Distal vessel reference diameters ≥ 2.5 mm and ≤ 4.0 mm |
| Exclusion criteria |
| Culprit lesions in ST-segment elevation acute coronary syndrome |
| Left main disease |
| In-stent restenosis lesions |
| Critical stenoses in which the OCT catheter cannot be advanced across the lesion despite predilation with a balloon ≤ 2 mm in diameter |
| Lesion involving a bifurcation with a side branch diameter ≥ 2 mm |
| Cardiogenic shock |
| Patients requiring cardiac surgery or percutaneous valve intervention within 3 months before or after angioplasty |
| Pregnancy |
| Life expectancy < 1 year |
| Contraindication to appropriate antiplatelet therapy after revascularization |
| Coronary artery disease with an indication for surgical revascularization |
| Advanced chronic kidney disease* or anatomical characteristics that preclude the use of optical coherence tomography |
| Inability to obtain informed consent |
| Allergy to eggs or soy, contraindicating the use of OA |
|
* Evaluated based on the investigator’s clinical judgment regarding the safety and potential risks associated with the contrast volume required for OCT-guided PCI. OA, orbital atherectomy; OCT, optical coherence tomography; STEMI, ST-segment elevation myocardial infarction. |
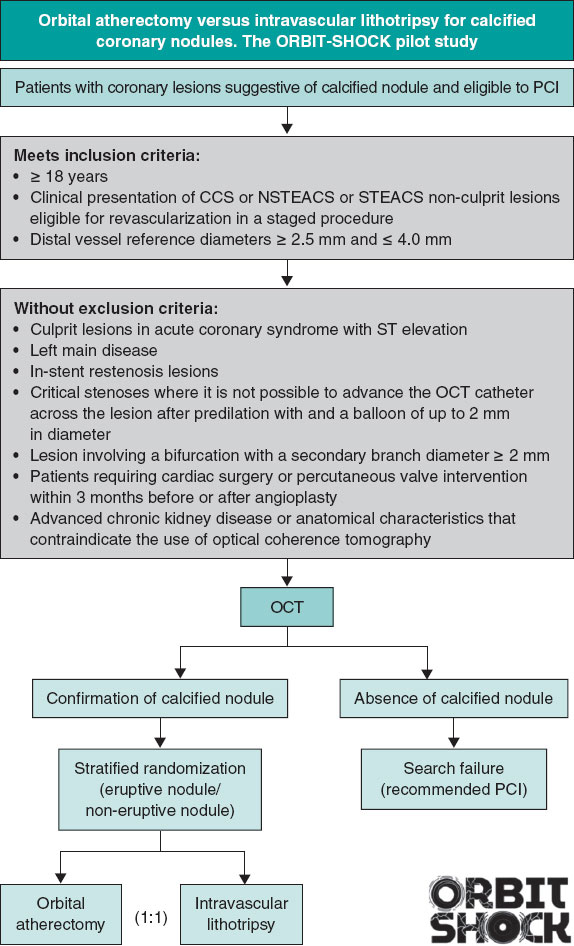
Figure 1. Central illustration. ORBIT-SHOCK study design and flow chart. Eligible patients will be selected by investigators at each participant center. After confirmation of the presence of a CN and verification of compliance with the inclusion criteria, and exclusion of any exclusion criteria, patients will be randomized in a 1:1 ratio to 1 of the 2 lesion preparation strategies under investigation: OA or IVL. Randomization will be stratified according to the type of CN (eruptive or noneruptive) to ensure a balanced distribution across treatment groups. CN, calcified nodule; IVL, intravascular lithotripsy; NSTEACS, non-ST-segment elevation acute coronary syndrome; STEACS, ST-segment elevation acute coronary syndrome; OA, orbital atherectomy; OCT, optical coherence tomography; PCI, percutaneous coronary intervention.
Endpoints
All endpoints are detailed in table 2 and defined in table S1. The primary endpoint is the percentage of stent expansion at the CN site, assessed by OCT and defined as the ratio between the minimum stent area (MSA) at the CN site and the reference area at the CN site:
Table 2. Study endpoints
| Primary endpoints |
|---|
| Percentage of stent expansion at the CN site: assessed by OCT and defined as the ratio between the minimum stent area at the CN site and the reference lumen area at the CN site. |
| Secondary endpoints |
| Procedural success: defined as achieving a stent expansion of ≥ 80% with TIMI grade-3 flow, in the absence of stent loss, coronary perforation, or intraoperative mortality |
| Strategy success: defined as procedural success without need for crossover to an alternative treatment |
| Minimum stent area |
| Minimum stent area at the CN site |
| Significant stent malapposition at the CN site: measured using OCT, defined as a stent strut detachment ≥ 0.4 mm from the underlying vessel wall with a longitudinal extension ≥ 1 mm |
| Degree of CN debulking, defined as the reduction in CN size (mm2), measured after plaque modification and prior to stenting |
| Evidence of fracture at the CN site, defined as a new disruption or discontinuity observed on OCT after plaque modification and prior to stenting |
| Degree of stent ellipticity at the CN site: measured using OCT at the end of the procedure, calculated as the ratio of the maximum luminal diameter to the minimum luminal diameter of the stent at the CN site |
| Residual luminal protrusion of the CN after stenting, measured by OCT in mm2 |
| Target lesion failure at 12 months, defined as the composite of clinically driven target lesion revascularization, myocardial infarction or cardiac death related to the target lesion |
| Target lesion revascularization at 12 months, defined as a repeat percutaneous coronary intervention of the target lesion or bypass surgery of the target vessel performed for restenosis or other complication of the target lesion. |
| Major adverse cardiovascular events at 12 months: defined as a composite of cardiac death, non-fatal target lesion myocardial infarction, unplanned target lesion revascularization or stent thrombosis. |
|
CN, calcified nodule; OA, orbital atherectomy; OCT, optical coherence tomography; TIMI, Thrombolysis in Myocardial Infarction. |
Stent expansion at the CN site (%) = (MSA at the CN site / reference lumen area at the CN site) x 100
The stent reference area at the CN site will be manually calculated by CoreLab specialists using co-registered pre- and post-stent images. Additionally, stent expansion will also be assessed using the Aptivue automated software (Abbott, United States). Specific methodological details regarding the selection of reference cross- sections and algorithms used to calculate stent expansion are described in the methods section of the supplementary data.
Secondary endpoints include procedural and strategy success, and mechanistic OCT-derived parameters related to calcium modification, including nodule debulking volume and calcium fracture. Additional OCT metrics of final stent optimization will also be assessed, including minimum stent area, stent ellipticity, significant malapposition, and residual nodule protrusion. Finally, clinical outcomes including target lesion failure, target lesion revascularization, and major adverse cardiovascular events will be assessed at the 12-month follow-up.
Study devices and procedures
The recommendations for the use of OA and IVL have been established according to the SCASI Expert Consensus Statement.24 Only CE-marked devices locally approved and commercially available will be used in the study.
The study procedures and their sequence are illustrated in figure 2.
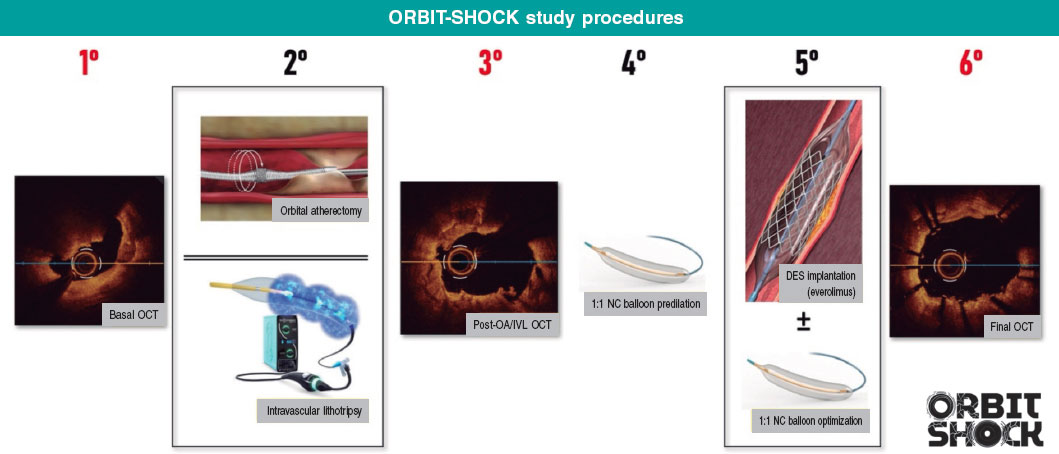
Figure 2. Study procedures. Red numbers indicate OCT acquisitions. 1. The presence of a CN must be confirmed by optical coherence tomography before enrolling a patient with suggestive angiographic findings. OCT imaging before any intervention on the target lesion is mandatory. Predilation with a 1.5 – 2.0 mm balloon is permitted if the OCT catheter cannot cross the lesion. 2. Lesion preparation with OA or IVL according to randomization. 3. Obtaining OCT images after the use of OA or IVL is mandatory, provided it is feasible and the patient’s clinical condition permits it. 4. Prior to stenting, dilation with a 1:1 NC balloon dilation is mandatory. 5. Stenting (use of a next-generation everolimus-eluting stent with CE marking is recommended). Stent optimization through post-dilation with an NC balloon based on OCT images is mandatory. 6. Final OCT images after stenting and optimization must be obtained to document the final procedural result. CN, calcified nodule; DES, drug-eluting stents; IVL, intravascular lithotripsy; NC, noncompliant; OA, orbital atherectomy; OCT, optical coherence tomography.
Optical coherence tomography
The presence of a CN must be confirmed by OCT before including a patient with suggestive angiographic images.
OCT imaging must be obtained before any intervention on the target lesion. Limited predilation with a 1.5–2.0 mm balloon is permitted if the OCT catheter cannot cross the lesion. This minimal predilation is intended solely to facilitate catheter delivery while minimizing structural modification of the nodule and preserving the accuracy of the baseline morphological assessment. OCT acquisition immediately after OA/IVL and after stenting (final result) is mandatory, provided it is technically feasible and the patient’s clinical condition allows it. Although not mandatory, performing an OCT run after 1:1 balloon predilation and immediately prior to stenting is highly recommended to confirm adequate plaque preparation and obtain accurate measurements of the reference vessel diameter and required stent length. Stent optimization through post-dilation with a noncompliant (NC) balloon based on OCT images is mandatory. In such cases, a repeat OCT acquisition must be performed to document the final procedural result.
Orbital atherectomy
A smooth forward and backward motion at a slow advancement speed (approximately 1.0 mm/s) is required to optimize ablation efficiency. For vessels with a reference diameter ≥ 3.0 mm, the use of high rotational speed (120 000 rpm) is strongly recommended. For vessels < 3.0 mm, low rotational speed (80 000 rpm) should be used initially. Operators should aim for the maximum feasible number of passes (typically > 5) to maximize lumen gain, ensuring that individual treatment runs do not exceed 30 seconds to prevent thermal injury. The total treatment time with a single crown should not exceed 5 minutes. Before stenting, dilation with a 1:1 NC balloon is mandatory.
Intravascular lithotripsy
The IVL balloon should be sized 1:1 to the reference vessel diameter to ensure efficient energy transfer. Pulses: delivery of the maximum number of pulses (up to the catheter limit) is strongly recommended to ensure adequate modification of the CN. Appropriate rest periods between therapy cycles are required to allow for distal washout and prevent hemodynamic compromise. Before stenting, dilation with a 1:1 NC balloon is mandatory.
Lesion stenting
To ensure greater treatment homogeneity, the use of a next-generation everolimus-eluting stent with CE marking is recommended. If this is not feasible, the use of another next-generation stent with CE marking is permitted.
Crossover between techniques and the management of special situations are detailed in the procedures section of the supplementary data.
Data collection
A clinical evaluation will be performed and data will be collected at 3 predefined time points: at patient enrollment, at hospital discharge, and at 12 months after PCI (table 3). All data will be recorded in an electronic case report form (eCRF).
Table 3. Study visits and data collection
| Study procedure | Baseline | Hospital discharge | 1-year follow-up (± 30 days) |
|---|---|---|---|
| Patient screening (inclusion/exclusion criteria) | X | ||
| Patient informed consent | X | ||
| Randomization | X | ||
| Patient characteristics | |||
| Demographics | X | ||
| Health history | X | ||
| Physical examination | X | X | X |
| Angina status (CCS class) | X | X | X |
| Medication | X | X | X |
| ECG | X | X | X |
| Echocardiography | X | ||
| Laboratory data | X | X | X |
| Vital status | X | X | X |
| Indication for PCI | X | ||
| PCI procedural variables | |||
| Angiography data | X | ||
| OCT imaging data (measured by a external core laboratory) | X | ||
| Technical variables | X | ||
| Endpoints | X | X | |
| Adverse events | X | X | |
|
CCS, Canadian Cardiovascular Society angina grading system; ECG, electrocardiogram; OCT, optical coherence tomography; PCI, percutaneous coronary intervention. |
|||
OCT images will be analyzed by a central core laboratory, which will remain blinded to the lesion modification technique employed. The core laboratory will be responsible for providing accurate and standardized interpretations, thereby ensuring unbiased results regardless of the treatment strategy applied.
Statistical considerations
Sample size
The limited number of studies specifically addressing CN, together with the heterogeneity in patient selection criteria and methods used to calculate stent expansion, makes it challenging to estimate the sample size required to ensure adequate statistical power. To address this limitation, a pilot study has been designed with 50 patients (25 per group) to evaluate and compare the outcomes of OA and IVL in this clinical setting.
The determination of this sample size was primarily driven by feasibility and pragmatic considerations to ensure the successful completion of the pilot study. Despite this pragmatic approach, the planned sample of 50 patients provides an estimated statistical power (0.8) to detect a clinically relevant difference of 12% in stent expansion under an exploratory superiority framework, assuming an expected standard deviation of ± 15%.
Descriptive statistics and hypothesis testing methodology
The normality of continuous variables will be assessed using the Kolmogorov-Smirnov test. Comparisons of normally distributed quantitative variables between treatment groups will be performed using the Student t test. For ordinal variables or continuous variables that do not follow a normal distribution, the non-parametric Mann-Whitney U test will be used. Qualitative variables wil be compared using the chi-square test or Fisher’s exact test. P values < .05 will be considered statistically significant.
Datasets
The intention-to-treat (ITT) analysis set will include all randomized patients, regardless of the treatment ultimately received.
The per-protocol (PP) analysis will include patients who received the treatment as assigned at randomization and met all inclusion criteria. Patients with major protocol deviations, including those who underwent crossover, will be excluded from this analysis.
The as-treated analysis set will include patients analyzed according to the treatment they actually received, rather than the treatment to which they were originally randomized.
Primary endpoint analysis
The primary endpoint of stent expansion at the CN site will be analyzed using an ITT approach, with results being expressed as mean and SD or median and IQR if the data deviate from normal distribution. In addition to the ITT analysis, the primary endpoint will also be assessed in the PP and as-treated sets.
Secondary endpoint analysis
The analysis of procedure-related secondary endpoints will be conducted initially in the ITT set, followed by assessments in the PP and as treated sets. The rate of clinical endpoints (target lesion revascularization, target lesion failure, major adverse cardiovascular events) will be compared between the 2 treatment groups using Cox proportional hazards regression, and event-free survival curves will be estimated using the Kaplan-Meier method. Differences in the primary endpoint between groups will be evaluated using adjusted hazard ratios with 95% confidence intervals and P values derived from Cox regression.
Hierarchical analysis using the win ratio method
Given that the trial includes multiple secondary endpoints that may influence the clinical course, an exploratory efficacy analysis comparing the 2 strategies will be performed using a hierarchical win-ratio method.
The first outcome in the hierarchy will be stent expansion at the CN. A “win” for one group will be defined as stent expansion at least 10 percentage points greater than that observed in the comparator patient. For this analysis, percent expansion will be truncated at a maximum of 100% prior to comparison. In the event of a tie, the procedure will proceed to the next predefined hierarchical endpoint, sequentially assessing procedural/strategy success, rate of major adverse cardiovascular events, stent malapposition at the CN, stent ellipticity, CN protrusion into the stent lumen, and minimum stent lumen area.
The overall win ratio will be estimated as the ratio of the total number of wins between the 2 treatment groups. Statistical significance will be assessed using the Finkelstein–Schoenfeld method with a 2-sided P < .05.
Analysis of predictors of adverse events
Predictors of suboptimal angioplasty outcomes will be evaluated using logistic regression models. Predictors of adverse clinical events during follow-up will be assessed using Cox proportional hazards regression models.
Adjusted analyses
Multivariable regression models will be used adjusted for CN type (eruptive vs noneruptive) and for any baseline or procedural differences observed between the groups.
Predefined subgroup analyses
Predefined subgroup analyses will include CN type (eruptive vs noneruptive), sex, vessel size (≥ 3.0 mm vs < 3.0 mm), and calcium length (≥ 15 mm vs < 15 mm).
Interim analysis
Once enrollment of all patients has been completed, an interim analysis will be performed. This analysis will focus on the primary endpoint (stent expansion at the CN site, assessed by OCT) as well as procedure-related secondary endpoints. The statistical analysis will be performed using STATA 18 (StataCorp, LLC, United States).
Ethical considerations
The ORBIT-SHOCK trial is conducted in full compliance with the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the central Clinical Research Ethics Committee of Hospital Universitario Ramón y Cajal (Madrid, Spain) and subsequently by the ethics committees of all other participant centers. All patients provide written informed consent before enrollment and randomization. Furthermore, the study was designed and is reported in accordance with the SAGER guidelines. Measures have been implemented to promote equitable participant inclusion and to minimize sex and gender bias. Sex-disaggregated data will be collected and analyzed to assess potential differences in procedural and clinical outcomes between men and women. The ORBIT-SHOCK pilot study is registered at ClinicalTrials.gov with the identifier NCT06736665.
DISCUSSION
Coronary calcification in the form of CN is consistently associated with worse outcomes due to the difficulty if achieving adequate calcium fracture and vessel expansion before stenting.
IVL has emerged as a commonly used strategy for the treatment of CN. However, although stent expansion > 100% is achieved in many patients, nodule fractures are observed in only a minority of cases, and nodule deformation occurs in less than half. As a result, stents expansion is often asymmetric, and residual nodule protrusion into the stent lumen is present in most patients. These findings could potentially increase the risk of future adverse events during follow-up.
The mechanism of action of OA may provide particular advantages for the treatment of CN. First, the rotational movement of the crown enables debulking of the nodule, while the orbital movement allows for circumferential modification of the vessel wall without being constrained by the guidewire position within the vessel.
Despite the theoretical advantages of its mechanism of action for treating CN, a recent observational study specifically comparing OA and IVL in these lesions did not demonstrate superior outcomes for OA.23 The study reported similar efficacy between the 2 techniques in terms of stent expansion (90.0% for OA vs 92.5% for IVL) and minimum lumen area (5.6 mm2 vs 5.5 mm2), as well as comparables rates of target lesion failure at 2 years (12.0% for OA vs 9.8% for IVL). Although the authors performed a propensity score-matched analysis, the sample size remained limited. In addition, given the retrospective design of the data, the presence of unmeasured confounding and significant differences in baseline lesion complexity are highly likely. In real-world clinical practice, atherectomy devices are preferentially used for uncrossable or more extensively calcified lesions, inherently introducing selection bias.
Moreover, in that study, the median number of OA runs was only 4 (IQR, 3-5) for a median calcium length of 25 mm, and the rotational speeds were not reported. This limited number of passes may be insufficient to achieve adequate lesion preparation and debulking of a CN, which often requires a higher number of runs and high rotational speeds.24 Only a randomized controlled design can ensure a homogeneous and balanced cohort, allowing an unbiased comparison of both strategies. Accordingly, the ORBIT-SHOCK pilot study will be the first randomized trial designed to directly compare OA and IVL for the treatment of CN, with all procedures performed under a strict, OCT-guided protocol and mandatory stent optimization.
Sample size determination and rationale for a randomized pilot study
Several studies have evaluated stent expansion after angioplasty using intracoronary imaging modalities. In the ILUMIEN II study, the minimum stent expansion in the OCT-guided group (calculated using the mean of the proximal and distal reference lumen areas) was 72.8% (IQR, 63.3 - 81.3).25 This corresponds to an estimated standard deviation (SD) of 13.3%, assuming a normal distribution. In the ILUMIEN IV trial, minimum stent expansion in the OCT-guided group was 80.8 ± 16.8%.9
Previous retrospective registries evaluating IVL or OA have reported wide SDs in stent expansion, ranging from 16.5% to > 27%. 1,3,4,17,20 A recent observational study comparing IVL and OA specifically for the treatment of CN reported SDs for stent expansion ranging from 13.0% (in eruptive nodules treated with IVL) to 25.9% (in noneruptive nodules treated with OA)12. This marked variability may be largely attributed to the heterogeneity of treated lesions, small sample sizes, heterogeneous definitions of the reference lumen area, or the lack of standardized imaging protocols. In contrast, the ORBIT-SHOCK pilot trial incorporates a strict OCT-guided PCI protocol with mandatory stent optimization. Under these controlled conditions, operator-dependent is expected to be substantially reduced. Consequently, an estimated SD of ± 15%, consistent with monitored trials such as ILUMIEN IV, was consiered the most appropriate assumption.
Regarding the clinically meaningful effect size, several pivotal trials, such as ULTIMATE,26 ILUMIEN III,27 and OPINION,28 have used a stent expansion threshold > 90% as the standard for optimal stent deployment, a target associated with significantly lower rates of target lesion failure during follow-up. Furthermore, the DOCTORS trial demonstrated that stent expansion < 79.4% predicts a fractional flow reserve < 0.90, suggesting a sub-optimal hemodynamic result. Accordingly, and acknowledging that achieving > 90% expansion may not be feasible in a significant proportion of complex lesions, the EAPCI expert consensus document proposes > 80% expansion as a pragmatic target for routine clinical practice.29 A 12% difference in stent expansion may therefore have important clinical implications, potentially shifting outcomes from an optimized result to a range associated with less functional outcomes.
Given the limited and heterogeneous data currently available, this pilot study was pragmatically sized to assess feasibility while maintaining sufficient exploratory power to detect a clinically relevant 12% difference in stent expansion. These findings will provide the essential methodological foundation and variance estimates necessary of future large-scale confirmatory trials.
Study limitations
Focusing on patients with CN (which account for approximately 20%–40% of severely calcified lesions) and the potential excluding of individuals with impaired renal function due to the contrast requirements of OCT may limit the eligible study population. These strict inclusion criteria could therefore result in a slower recruitment rate than initially anticipated. Nevertheless, participation of 6 centers, with an estimated enrollment rate of 1 patient per center per month, is expected to allow completion of patient recruitment within the planned timeframe.
The distinct behavior of eruptive and noneruptive CN, both in terms of stent expansion and the occurrence of adverse events during follow-up, could introduce bias if the distribution of these 2 subtypes is not balanced between treatment groups. To mitigate this risk, stratified randomization has been implemented to ensure a balanced allocation of both CN types across the treatment arm.
CN rarely occur in isolation and are frequently associated with extensive background coronary calcification. To address this, the study protocol specifically recommends applying the randomized technique to treat all concomitant calcified lesions within the target segment. This may include performing multiple OA runs or using more than 1 IVL balloon when necessary, with the explicit objective of achieving optimal lesion preparation and final results throughout the entire lesion length, not only at the nodule site. Although the primary endpoint focuses on stent expansion at the CN site, broader secondary clinical outcomes may be influenced by the overall calcific burden of the treated segment. Nevertheless, the randomized design of the study is expected to evenly distribute this anatomical complexity between the 2 treatment arms.
Finally, as a pilot clinical trial with a sample size determined primarily by feasibility, this study is not statistical powered to detect small differences between OA and IVL. However, the exploratory superiority framework is design to detect a clinically relevant effect size, such as a 12% difference in stent expansion. The fundamental purpose of this pilot study is to establish the methodological foundation and generate the preliminary data required to design future, adequately powered confirmatory trials.
CONCLUSIONS
The ORBIT-SHOCK pilot study will be the first randomized trial to allow a direct comparison of the outcomes of OA and IVL in the management of calcified CN, a common and complex clinical scenario that requires robust evidence from randomized clinical trials.
FUNDING
The study was funded by an unrestricted institutional research grant from Abbott (United States).
ETHICAL CONSIDERATIONS
The ORBIT-SHOCK trial is conducted in full compliance with the principles outlined in the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the central Clinical Research Ethics Committee of Hospital Universitario Ramón y Cajal (Madrid, Spain) and subsequently by the ethics committees of all other participant centers. All patients gave their written informed consent before enrollment and randomization. Furthermore, the study was designed and is being reported following the SAGER guidelines. We have implemented measures to ensure an equitable inclusion of participants and avoid sex/gender bias. Sex-disaggregated data will be collected and analyzed to evaluate potential differences in procedural and clinical outcomes between men and women.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the preparation of this manuscript.
AUTHORS’ CONTRIBUTIONS
L.M. Domínguez-Rodríguez: conceptualization, original draft, review, and editing. J. Bayón Lorenzo, I. Amat Santos, J. Martín Moreiras, J. Valencia Martín, M. Pan, L. Salido Tahoces, A. Pardo Sanz, J.L. Mestre-Barceló, A. Larrea-Iñarra, J.M. Ruiz-Nodar, A. Diego Nieto, C. González-Juanatey: drafting, review, and editing. J.L. Zamorano, and Á. Sánchez-Recalde: conceptualization, drafting, review, and editing. All authors have reviewed and approved the final version of this manuscript.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- CN are associated with suboptimal outcomes following percutaneous coronary intervention.
- IVL has become a widely adopted strategy for the treatment of CN due to its ease of use, low complication rates, and favorable outcomes reported in recent studies.
- OA has been shown to be an effective strategy for the treatment of severely calcified and nondilatable coronary lesions. However, evidence regarding its performance specifically in CN remains limited and is largely derived from small observational studies and case reports.
- Despite major advances in imaging and plaque-modification technologies, the optimal management strategy for CN has yet to be clearly defined.
WHAT DOES THIS STUDY ADD?
- The ORBIT-SHOCK pilot trial is designed as the first randomized clinical trial to compare the safety and efficacy profile of OA and IVL in the treatment of CN.
REFERENCES
1. Sato T, Matsumura M, Yamamoto K, et al. Impact of Eruptive vs Noneruptive Calcified Nodule Morphology on Acute and Long-Term Outcomes After Stenting. JACC Cardiovasc Interv. 2023;16:1024-1035.
2. Brott BC. The Calcified Nodule Paradox. JACC Cardiovasc Interv. 2023;16: 1036-1038.
3. McInerney A, Travieso A, Baza AJ, et al. Impact of coronary calcium morphology on intravascular lithotripsy. EuroIntervention. 2024;20:656-668.
4. Ali ZA, Kereiakes DJ, Hill JM, et al. Impact of Calcium Eccentricity on the Safety and Effectiveness of Coronary Intravascular Lithotripsy:Pooled Analysis From the Disrupt CAD Studies. Circ Cardiovasc Interv. 2023;16: E012898.
5. Prati F, Gatto L, Fabbiocchi F, et al. Clinical outcomes of calcified nodules detected by optical coherence tomography:a sub-analysis of the CLIMA study. EuroIntervention. 2020;16:380-386.
6. Akasaka T, Kubo T. OCT-derived coronary calcified nodules as a predictor of high-risk patients. EuroIntervention. 2020;16:361-363.
7. Ali ZA, Kereiakes D, Hill J, et al. Safety and Effectiveness of Coronary Intravascular Lithotripsy for Treatment of Calcified Nodules. JACC Cardiovasc Interv. 2023;16:1122-1124.
8. Lee JM, Choi KH, Song Y Bin, et al. Intravascular Imaging–Guided or Angiography-Guided Complex PCI. New Engl J Med. 2023;388:1668-1679.
9. Ali ZA, Landmesser U, Maehara A, et al. Optical Coherence Tomography–Guided versus Angiography-Guided PCI. New Engl J Med. 2023;389: 1466-1476.
10. Holm NR, Andreasen LN, Neghabat O, et al. OCT or Angiography Guidance for PCI in Complex Bifurcation Lesions. New Engl J Med. 2023; 389:1477-1487.
11. Vrints C, Andreotti F, Koskinas KC, et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J. 2024:3415-3537.
12. Dakroub AH, Shin D, Singh M, et al. Procedural and Clinical Outcomes After Orbital Atherectomy Versus Intravascular Lithotripsy in Patients With Calcified Nodules. Circ Cardiovasc Interv. 2025;18:015254.
13. Parikh K, Chandra P, Choksi N, Khanna P, Chambers J. Safety and feasibility of orbital atherectomy for the treatment of calcified coronary lesions:The ORBIT i Trial. Catheter Cardiovasc Interv. 2013;81:1134-1139.
14. Chambers JW, Feldman RL, Himmelstein SI, et al. Pivotal trial to evaluate the safety and efficacy of the orbital atherectomy system in treating de Novo, severely calcified coronary lesions (ORBIT II). JACC Cardiovasc Interv. 2014;7:510-518.
15. Florek K, Bartoszewska E, Biegała S, et al. Rotational Atherectomy, Orbital Atherectomy, and Intravascular Lithotripsy Comparison for Calcified Coronary Lesions. J Clin Med. 2023;12:7246.
16. Faria D, Vinhas H, Bispo J, et al. Initial experience with orbital atherectomy in a non-surgical center in Portugal. Rev Port Cardiol. 2024;43:645-728.
17. Okamoto N, Egami Y, Nohara H, et al. Direct Comparison of Rotational vs Orbital Atherectomy for Calcified Lesions Guided by Optical Coherence Tomography. JACC Cardiovasc Interv. 2023;16:2125-2136.
18. Yamamoto MH, Maehara A, Kim SS, et al. Effect of orbital atherectomy in calcified coronary artery lesions as assessed by optical coherence tomography. Catheter Cardiovasc Interv. 2019;93:1211-1218.
19. Yamamoto MH, Maehara A, Karimi Galougahi K, et al. Mechanisms of Orbital Versus Rotational Atherectomy Plaque Modification in Severely Calcified Lesions Assessed by Optical Coherence Tomography. JACC Cardiovasc Interv. 2017;10:2584-2586.
20. Okamoto N, Ueda H, Bhatheja S, et al. Procedural and one-year outcomes of patients treated with orbital and rotational atherectomy with mechanistic insights from optical coherence tomography. EuroIntervention. 2019;14:1760-1767.
21. Rola P, Włodarczak S, Barycki M, et al. Safety and Efficacy of Orbital Atherectomy in the All-Comer Population:Mid-Term Results of the Lower Silesian Orbital Atherectomy Registry (LOAR). J Clin Med. 2023;12:5842.
22. Chiang CSM, Alan Chan KC, Lee M, Chan KT. Orbital-Tripsy:Novel Combination of Orbital-Atherectomy and Intravascular-Lithotripsy, in Calcified Coronaries After Failed Intravascular-Lithotripsy. JACC Case Rep. 2020;2:2437-2444.
23. Shin D, Dakroub A, Singh M, et al. Debulking Effect of Orbital Atherectomy for Calcified Nodule Assessed by Optical Coherence Tomography. Circ Cardiovasc Interv. 2024;17:014145.
24. Riley RF, Patel MP, Abbott JD, et al. SCAI Expert Consensus Statement on the Management of Calcified Coronary Lesions. J Soc Cardiovasc Angiogr Interv. 2024;3:101259.
25. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of stent expansion guided by optical coherence tomography versus intravascular ultrasound:The ILUMIEN II study (Observational Study of Optical Coherence Tomography [OCT] in Patients Undergoing Fractional Flow Reserve [FFR] and Percutaneous Cor. JACC Cardiovasc Interv. 2015;8:1704-1714.
26. Zhang J, Gao X, Kan J, et al. Intravascular Ultrasound Versus Angiography-Guided Drug-Eluting Stent Implantation:The ULTIMATE Trial. J Am Coll Cardiol. 2018;72:3126-3137.
27. Ali ZA, Galougahi KK, Maehara A, et al. Outcomes of optical coherence tomography compared with intravascular ultrasound and with angiography to guide coronary stent implantation:One-year results from the ILUMIEN III:OPTIMIZE PCI trial. EuroIntervention. 2021;16:1085-1091.
28. Kubo T, Shinke T, Okamura T, et al. Optical frequency domain imaging vs intravascular ultrasound in percutaneous coronary intervention (OPINION trial):One-year angiographic and clinical results. Eur Heart J. 2017;38:3139-3147.
29. Räber L, Mintz GS, Koskinas KC, et al. Clinical use of intracoronary imaging. Part 1:Guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention. 2018;14:656-677.














