ABSTRACT
Background and objectives: Percutaneous coronary intervention for chronic total occlusion (CTO PCI) is a prolonged and technically demanding procedure often associated with patient anxiety and discomfort. We evaluated whether intraoperative audiovisual distraction via virtual reality (VR) goggles reduces procedural anxiety vs usual care.
Methods: The ReViCTO trial was a prospective, single-center, randomized, open-label study enrolling 59 patients undergoing elective CTO PCI. Participants were randomized to receive intraoperative audiovisual distraction via VR goggles (n = 31) or usual care (n = 28). The primary endpoint was maximum patient-reported procedural anxiety assessed immediately after theprocedure using a visual analogue scale (VAS; range, 0-10). Secondary endpoints included procedural pain, intraoperative sedative/analgesic requirements, and patient satisfaction.
Results: Baseline clinical characteristics were similar between groups. There was no significant difference in the primary endpoint of maximum procedural anxiety between the VR and control groups (mean VAS, 3.23 ± 2.78 vs 3.75 ± 2.77; mean difference, –0.52; P = .472). Similarly, no significant differences were observed regarding maximum procedural pain (P = .964) or the use and dosage of intraoperative morphine or midazolam. The intervention was safe, and 80.6% of patients in the VR group reported willingness to use the device during a future procedure.
Conclusions: Among patients undergoing elective CTO PCI, the use of immersive VR was feasible and well-tolerated but did not significantly reduce patient-reported peak anxiety, pain, or intraoperative pharmacologic requirements vs contemporary usual care.
[ClinicalTrials.gov: NCT05458999].
Keywords: Virtual reality. Percutaneous coronary intervention. Chronic total occlusion. Anxiety. Pain.
RESUMEN
Introducción y objetivos: La intervención coronaria percutánea (ICP) para las oclusiones coronarias totales crónicas (OTC) es un procedimiento prolongado y técnicamente exigente, a menudo asociado a ansiedad y malestar del paciente. Se evalúa si la distracción audiovisual durante el procedimiento mediante gafas de realidad virtual (RV) reduce la ansiedad procedimental en comparación con la atención habitual.
Métodos: El ensayo ReViCTO es un estudio prospectivo, unicéntrico, aleatorizado y abierto que incluyó 59 pacientes sometidos a ICP electiva de OTC. Los participantes se asignaron al azar para recibir distracción audiovisual durante el procedimiento con gafas de RV (n = 31) o atención habitual (n = 28). El objetivo principal fue la ansiedad máxima percibida por el paciente durante el procedimiento, evaluada inmediatamente después con una escala visual analógica (EVA; rango 0-10). Los objetivos secundarios fueron el dolor procedimental, las necesidades de sedación o analgesia durante el procedimiento, y la satisfacción del paciente.
Resultados: Las características clínicas basales fueron comparables entre los grupos. No hubo diferencias significativas en el objetivo principal de ansiedad máxima entre el grupo de RV y el grupo control (EVA media 3,23 ± 2,78 frente a 3,75 ± 2,77; diferencia media −0,52; p = 0,472). Tampoco se encontraron diferencias significativas en el dolor máximo (p = 0,964) ni en el uso y la dosis de morfina o midazolam durante el procedimiento. La intervención fue segura y el 80,6% de los pacientes del grupo de RV manifestaron que estarían dispuestos a utilizar el dispositivo en una intervención futura.
Conclusiones: En los pacientes sometidos a ICP electiva de OTC, el uso de RV inmersiva, en comparación con la atención habitual contemporánea, fue factible y bien tolerado, pero no redujo de manera significativa la ansiedad máxima ni el dolor percibidos por el paciente, y tampoco los requerimientos farmacológicos durante el procedimiento.
[ClinicalTrials.gov: NCT05458999].
Palabras clave: Realidad virtual. Intervención coronaria percutánea. Oclusión total crónica. Ansiedad. Dolor.
Abbreviations
CTO: chronic total occlusion. PCI: percutaneous coronary intervention. VASa: visual analogue scale for anxiety. VASp: visual analogue scale for pain. VR: virtual reality.
INTRODUCTION
Chronic total occlusions (CTO) are common among patients undergoing coronary angiography and represent one of the most technically demanding scenarios for percutaneous coronary intervention (PCI). Although contemporary CTO PCI programs achieve high success rates,1 these procedures frequently require prolonged fluoroscopy time, dual arterial access,2,3 and sustained patient immobility. In addition, ischemia-related chest discomfort may occur during complex procedural strategies.4,5
Anxiety is common in patients undergoing coronary procedures in the cath lab and may contribute to procedural discomfort and the need for pharmacologic sedation or analgesia.6-8 In routine practice, premedication and intraoperative administration of benzodiazepines or opioids are often used to mitigate anxiety and pain; however, their benefits are modest, and pharmacologic strategies and their use varies across centers.9 CTO PCI may be particularly associate with anxiety due to its typical duration, access strategy, and potential for procedural chest pain.
Virtual reality (VR) is an immersive audiovisual distraction strategy that can reduce procedural pain and anxiety across clinical settings. A systematic review demonstrated that VR-based distraction is effective for pain reduction in multiple procedural contexts.10
In interventional cardiology, early evidence suggests feasibility and potential benefit of VR during procedures performed under conscious sedation, including transcatheter aortic valve implantation and atrial fibrillation ablation.11,12 However, there are no randomized data evaluating VR during CTO PCI, a setting in which nonpharmacologic anxiolysis could be particularly valuable.
The ReViCTO trial was designed to test whether VR use during elective CTO PCI reduces the maximum level of patient-reported procedural anxiety vs usual care. Secondary endpoints included procedural pain, the use and dose of intraoperative anxiolytic or analgesic drugs, and patient satisfaction with the VR intervention. Figure 1 summarizes the main findings.
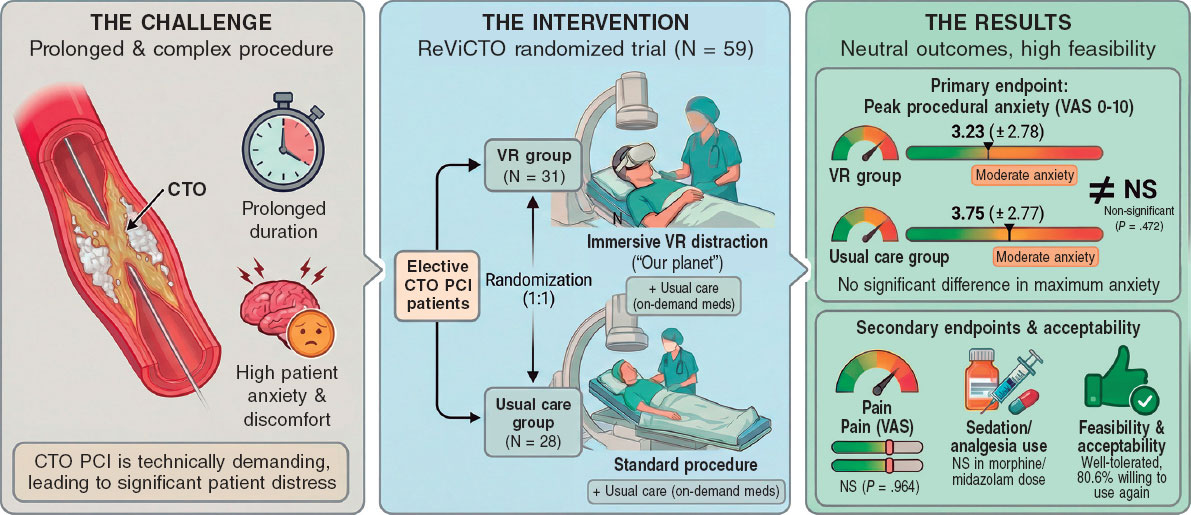
Figure 1. Central illustration. ReViCTO trial overview and main findings. Elective CTO PCI is a prolonged, technically complex procedure frequently associated with patient anxiety and discomfort. In the ReViCTO randomized trial (N = 59), patients were assigned to immersive audiovisual distraction using VR goggles (n = 31) or usual care (n = 28). VR use was feasible and well tolerated but did not significantly reduce peak procedural anxiety (VAS 0–10) or pain or intraoperative sedative or analgesic requirements vs usual care; 80.6% of VR patients reported willingness to use VR again in a future intervention. CTO, chronic total occlusion; PCI, percutaneous coronary intervention; VAS, visual analogue scale; VR, virtual reality.
METHODS
Trial design and oversight
The ReViCTO trial is an investigator-initiated, single-center, randomized, controlled, open-label, superiority trial with 2 parallel groups comparing immersive VR goggles vs usual care during elective CTO PCI. The study received no external funding. The rationale and full trial design have been published previously.13
The trial was conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Good Clinical Practice guidelines. The protocol was approved by the Clinical Research Ethics Committee of Hospital Clínico Universitario de València on 28 February, 2022, and all patients gave their prior written informed consent. The trial was registered at ClinicalTrials.gov (NCT05458999).
Participants
Adults (age > 18 years) scheduled for elective CTO PCI at Hospital Clínico Universitario de València were screened for eligibility. Key exclusion criteria were any condition precluding VR use, such as significant visual impairment, dementia, language barrier (inability to communicate fluently in Spanish or English), or other circumstances preventing safe use of the headset. Eligibility criteria were prespecified in the protocol publication.13
Randomization and trial procedures
Patients were randomized in a 1:1 ratio to the VR group or the control group using computer-generated permuted blocks to reduce the risk of imbalance in group sizes in a small trial.14 Allocation concealment was implemented via a web-based application that generated a unique trial identification number and assigned treatment arm after enrollment, preventing post-randomization modification or deletion. Due to the nature of the intervention, no blinding was applied to participants, operators, or outcome assessors.
CTO PCI was performed according to contemporary clinical practice by an experienced CTO team. Pharmacologic management of anxiety and pain was not protocolized and was left to the discretion of the primary operator in both groups. Specifically, morphine chloride and/or midazolam could be administered on demand during the procedure according to observed or reported anxiety or pain, in accordance with the trial protocol.
Interventions
Virtual reality group
A commercially available head-mounted display (Oculus Quest 2, Meta Platforms, United States) was used. Participants viewed the documentary series Our Planet via a video-streaming application (Netflix) in a virtual “theater” environment, starting with episode 1 with sequential autoplay thereafter, as specified in the protocol. The headset was applied before arterial puncture and was maintained throughout the intervention unless the patient requested removal or a serious complication occurred. Patient status was monitored at regular intervals during the procedure.
Control group
Participants assigned to control underwent CTO PCI under usual care without VR goggles.
Outcomes
The prespecified primary endpoint was the maximum level of anxiety perceived by the patient during the procedure, assessed immediately after the procedure and before leaving the cath lab using a visual analogue scale for anxiety (VASa). Secondary endpoints included maximum patient-perceived pain during the procedure (VASp), the use and total dose of intraoperative anxiolytic or analgesic drugs (midazolam and morphine chloride), procedure-related nausea or dizziness, and patient satisfaction with the VR intervention, including willingness to use VR again. Endpoint definitions and timing of assessment followed the protocol publication.
For analysis, VAS scores were treated as numeric ratings ranging from 0 (none) to 10 (worst), consistent with the trial questionnaires and the observed data range. Baseline angina-related health status was measured using the Seattle Angina Questionnaire (SAQ) to contextualize symptom burden.15
Data collection and management
Demographic characteristics, medical history, comorbidities, baseline symptom status (including SAQ), and procedural characteristics (arterial access strategy, procedure duration, fluoroscopy time, and radiation dose metrics) were collected from institutional electronic health records and procedure reports, supplemented by direct patient interview when necessary. Immediately after completion of CTO PCI, a trained study nurse administered the postoperative questionnaire and recorded VAS anxiety and pain, as well as nausea, dizziness, and satisfaction items. Intraoperative administration of morphine and midazolam and their total doses were recorded contemporaneously by the study nurse.
Data were entered into a dedicated electronic database incorporating range checks for numeric variables and duplicate checks for hospital identifiers and stored on a restricted-access workstation as described in the protocol.
Sample size estimation
The target sample size (58 patients, 29 per group) was calculated based on the primary endpoint, assuming a common standard deviation of 2.7 points for VAS anxiety and aiming to detect an absolute between-group difference of at least 2 points with a 2-sided alpha of 0.05 and 80% power. The standard deviation assumption was informed by prior cath lab anxiety studies.9,16
Statistical analysis
Continuous variables are expressed as mean (standard deviation, SD) or median (interquartile range, IQR) as appropriate (for the primary endpoint and Japan Chronic Total Occlusion (J-CTO) score, both measures are reported to allow comparison with sample size assumptions and to account for non-normal distribution). Categorical variables as counts and percentages. Between-group comparisons were performed using the Student t test for normally distributed continuous variables and the Mann–Whitney U test otherwise; categorical variables were compared using Fisher’s exact test or the chi-square test, as appropriate. Post hoc, we performed an analysis of covariance (ANCOVA) model with VASa as the dependent variable and treatment group as the main effect, adjusting for baseline anxiety. As a sensitivity analysis, we additionally adjusted for intraoperative opioid and benzodiazepine administration. Finally, to address the randomization imbalance in angiographic complexity, a multivariable linear regression analysis was conducted with maximum procedural anxiety as the dependent variable, adjusting for baseline anxiety and the J-CTO score. All tests were 2-sided, with a significance threshold of P < .05. Analyses were performed using R (V. 4.3.2, R Foundation for Statistical Computing, Austria). Reporting followed CONSORT guidelines for trial conduct and prespecified analyses (checklist in the supplementary data).17
RESULTS
Patients
Between 1 March 2022 and 23 October 2025, a total of 119 scheduled CTO PCI were performed. Fifteen patients declined the use of the headset, and in 44 cases the VR device was not available. One patient was excluded due to visual impairment (blindness). Overall, 59 patients were randomized to VR goggles (n = 31) or usual care (n = 28), as shown in figure 2.
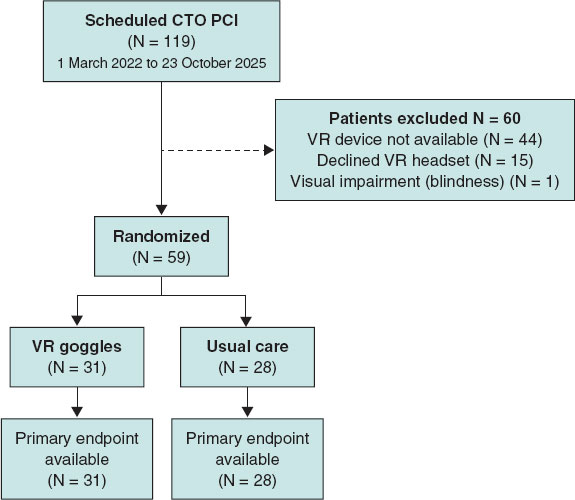
Figure 2. Study flow diagram. CTO PCI, percutaneous coronary intervention for chronic total occlusion; VR, virtual reality.
Enrolment was prospective; however, randomization was contingent on operational availability of the VR system. Early in the study, a single trained investigator was responsible for device preparation and operation, so randomization could only occur when this investigator was present in the cath lab (eg, during vacations or other clinical/research commitments). From June 2023 onward, a second investigator was trained, increasing coverage. In addition, the VR system was out of service for approximately 2 months due to changes in the required internet infrastructure. Importantly, nonrandomization during these periods was driven by logistical constraints rather than patient characteristics.
Baseline characteristics
Baseline clinical characteristics were broadly similar between groups. Median age was 67 [60;70] years in the VR group and 60 [58;66] years in the control group, and most participants were men (29 [93.5%] vs 26 [92.9%]). The prevalence of hypertension (51.6% vs 60.7%) and diabetes (41.9% vs 46.4%) was comparable; dyslipidemia was numerically less frequent in the VR group (58.1% vs 82.1%). Baseline anxiety assessed before entering the cath lab was moderate and did not differ significantly between groups (VAS anxiety: mean ± SD, 3.35 ± 3.10 vs 4.21 ± 3.05; P = .289; median [IQR], 2 [0; 6] vs 5 [2; 6]). Baseline pain was minimal in both groups (VAS pain baseline: 0.06 ± 0.36 vs 0.00 ± 0.00; P = .346; median [IQR], 0 [0 to 0] in both). Baseline angina-related health status was similar. The SAQ score was 52.4 [36.3; 64.3] in the VR group and 50.0 [44.0; 64.3] in the control group (P = .475), as shown in table 1.
Angiographic complexity differed between groups. The J-CTO score was higher in the VR group (mean ± SD, 3.10 ± 1.11; median [IQR], 3 [2; 4]) than in the control group (2.32 ± 1.36; median [IQR], 2 [1; 3]) (Student t test P = .019; Mann-Whitney U test P = .040).
Procedural characteristics and clinical outcomes
The distribution of procedural approach (antegrade, retrograde, or hybrid) was similar between groups (P = .826). Procedural duration, fluoroscopy time, radiation dose-area product, and contrast volume were not significantly different between groups, although radiation exposure tended to be higher in the VR group.
Technical success was achieved in 25 of 31 patients (80.6%) in the VR and 25 of 28 patients (89.3%) in the usual care group (P = .477). Procedural complications occurred in 4 of 31 patients (12.9%) in the VR group and 4 of 28 patients (14.3%) in the control group (P = 1.00) (table 1).
Table 1. Baseline characteristics, lesion and procedural characteristics
| Characteristic | VR goggles (n = 31) | Usual care (n = 28) | P |
|---|---|---|---|
| Age, years | 67 [60; 70] | 60 [58; 66] | .191 |
| Male sex | 29/31 (93.5%) | 26/28 (92.9%) | 1.000 |
| Hypertension | 16/31 (51.6%) | 17/28 (60.7%) | .482 |
| Dyslipidemia | 18/31 (58.1%) | 23/28 (82.1%) | .045 |
| Diabetes mellitus | 13/31 (41.9%) | 13/28 (46.4%) | .728 |
| Chronic kidney disease | 2/31 (6.5%) | 3/28 (10.7%) | .661 |
| Peripheral arterial disease | 5/31 (16.1%) | 4/28 (14.3%) | 1.000 |
| Current smoker | 5/31 (16.1%) | 10/28 (35.7%) | .084 |
| Former smoker | 15/31 (48.4%) | 6/28 (21.4%) | .031 |
| Previous myocardial infarction | 16/31 (51.6%) | 15/28 (53.6%) | .880 |
| Previous PCI | 19/31 (61.3%) | 17/28 (60.7%) | .966 |
| Previous CABG | 4/31 (12.9%) | 0/28 (0.0%) | .118 |
| Previous CTO-PCI attempt | 5/31 (16.1%) | 4/28 (14.3%) | 1.000 |
| Number of previous CTO attempts | 0 [0; 1] | 0 [0; 1] | .917 |
| NYHA class | 1 [1; 2] | 1 [1; 2] | .742 |
| Left ventricular ejection fraction, % | 50.4 ± 15.1 | 51.6 ± 14.0 | .742 |
| Hemoglobin, g/dL | 14.30 ± 1.79 | 14.33 ± 1.81 | .955 |
| Creatinine, mg/dL | 0.94 [0.80; 1.10] | 0.92 [0.81; 1.10] | .911 |
| NT-proBNP, pg/mL | 432 [186; 1406] | 390 [213; 1002] | .765 |
| Seattle Angina Questionnaire summary score (0-100) | 52.4 [36.3; 64.3] | 50.0 [44.0; 64.3] | .475 |
| Baseline anxiety (VAS 0-10) (mean) | 3.35 ± 3.10 | 4.21 ± 3.05 | .289 |
| Baseline anxiety (VAS 0-10) (IQR) | 2 [0; 6] | 5 [2; 6] | .289 |
| Baseline pain (VAS 0-10) (mean) | 0.06 ± 0.36 | 0.00 ± 0.00 | .346 |
| Baseline pain (VAS 0-10) (IQR) | 0 [0; 0] | 0 [0; 0] | .346 |
| Familiarity with technology: no | 4/31 (12.9%) | 2/28 (7.1%) | .389 |
| Familiarity with technology: cell phone | 10/31 (32.3%) | 11/28 (39.3%) | |
| Familiarity with technology: cell phone + personal computer | 15/31 (48.4%) | 15/28 (53.6%) | |
| Familiarity with technology: missing | 2/31 (6.5%) | 0/28 (0.0%) | |
| Previous VR experience: no | 25/31 (80.6%) | 24/28 (85.7%) | .103 |
| Previous VR experience: yes | 4/31 (12.9%) | 3/28 (10.7%) | |
| Previous VR experience: unknown | 0/31 (0.0%) | 1/28 (3.6%) | |
| Previous VR experience: missing | 2/31 (6.5%) | 0/28 (0.0%) | |
| Belief that video could relax: no | 7/31 (22.6%) | 1/28 (3.6%) | .103 |
| Belief that video could relax: yes | 23/31 (74.2%) | 26/28 (92.9%) | |
| Belief that video could relax: unknown | 1/31 (3.2%) | 1/28 (3.6%) | |
| SYNTAX score | 12 [9; 21] | 13 [11; 18] | .451 |
| J-CTO score (median) | 3 [2; 4] | 2 [1; 3] | .040 |
| J-CTO score (mean) | 3.10 ± 1.11 | 2.32 ± 1.36 | .019 |
| In-stent CTO | 1/30 (3.3%) | 3/28 (10.7%) | .344 |
| Occlusion length, mm | 30 [20; 40] | 30 [15; 40] | .209 |
| Blunt proximal cap | 21/31 (67.7%) | 22/28 (78.6%) | .350 |
| Moderate/severe calcification | 26/31 (83.9%) | 15/28 (53.6%) | .012 |
| Bending > 45° | 17/31 (54.8%) | 7/28 (25.0%) | .020 |
| Collateral pattern: ipsilateral | 4/31 (12.9%) | 2/28 (7.1%) | .346 |
| Collateral pattern: contralateral | 20/31 (64.5%) | 15/28 (53.6%) | |
| Collateral pattern: ipsi- and contralateral | 7/31 (22.6%) | 11/28 (39.3%) | |
| Approach: antegrade | 25/31 (80.6%) | 22/28 (78.6%) | .826 |
| Approach: retrograde | 4/31 (12.9%) | 3/28 (10.7%) | |
| Approach: hybrid | 2/31 (6.5%) | 3/28 (10.7%) | |
| Any femoral access used | 4/31 (12.9%) | 4/28 (14.3%) | 1.000 |
| Procedure duration, min | 123 ± 38 | 107 ± 55 | .216 |
| Fluoroscopy time, min | 48 [35; 63] | 40 [20; 67] | .370 |
| Dose-area product, Gy · cm2 | 218 [183; 321] | 178 [103; 281] | .052 |
| Contrast volume, mL | 145 [106; 220] | 150 [112; 198] | .967 |
| No. of stents | 2 [1; 3] | 1 [1; 2] | .030 |
| Total stent length, mm | 80 ± 40 | 52 ± 28 | .006 |
| Technical success | 25/31 (80.6%) | 25/28 (89.3%) | .477 |
| Procedural complications | 4/31 (12.9%) | 4/28 (14.3%) | 1.000 |
|
Data are expressed as mean (SD), median [interquartile range], or n/N (%). P values were calculated using the Student t test for normally distributed variables, the Mann–Whitney U test for non-normally distributed variables, and the chi-square or Fisher exact test for categorical variables, according to the prespecified analysis plan. NT-proBNP available in 13 VR and 12 control patients; SAQ available in 30 VR and 27 control patients; NYHA available in 30 VR and 28 control patients. For baseline VAS anxiety and pain, both mean ± SD and median [Q1; Q3] are reported. CABG, coronary artery bypass grafting; CTO, chronic total occlusion; J-CTO, Japanese Chronic Total Occlusion score; NT-proBNP, N-terminal pro-B-type natriuretic peptide; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; SAQ, Seattle Angina Questionnaire; SD, standard deviation; SYNTAX, Synergy Between PCI With TAXUS and Cardiac Surgery score; VAS, visual analogue scale; VR, virtual reality. |
|||
Primary endpoint
The primary endpoint (maximum procedural anxiety; [VASa]) and key secondary endpoints (maximum procedural pain [VASp] and intraoperative drug administration) were available for all randomized participants.
Maximum procedural anxiety did not differ significantly between groups. Mean VASa was 3.23 ± 2.78 with VR and 3.75 ± 2.77 with usual care (mean difference, −0.52 points; 95% confidence interval (95%CI), −1.97 to 0.92; Student t test P = .472). Median [IQR] values were 4 [0; 5] and 4 [2; 6], respectively. Because the VASa distribution was skewed, results were consistent using the Mann–Whitney U test (P = .379) (figure 3, table 2 and table S1).
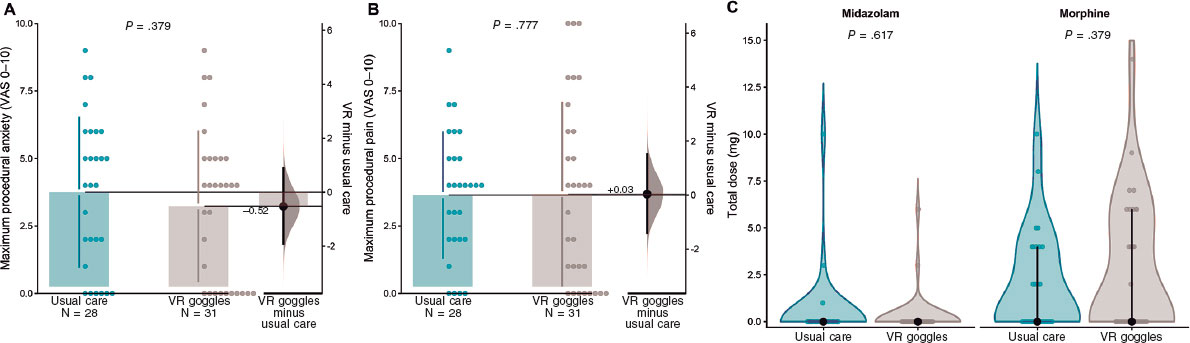
Figure 3. Patient-reported procedural anxiety and pain, and intraoperative drug use. A: maximum procedural anxiety assessed immediately after the procedure using a visual analogue scale for anxiety (VASa; 0–10). B: maximum procedural pain assessed using a visual analogue scale for pain (VASp; 0–10). C: total intraoperative dose (mg) of midazolam and morphine by treatment group; zero values indicate no drug administration. In panels A and B, dots represent individual patients; the right-side estimation plot shows the mean between-group difference (VR minus usual care) with its 95% confidence interval. P values correspond to prespecified between-group comparisons. VAS, visual analogue scale; VR, virtual reality.
Table 2. Endpoints and safety
| Variable | VR goggles | Usual care | P |
|---|---|---|---|
| Maximum procedural anxiety* | 3.23 ± 2.78 | 3.75 ± 2.77 | .472 |
| Maximum procedural anxiety* | 4 [0; 5] | 4 [2 to 6] | .379 |
| Maximum procedural pain* | 3.68 ± 3.39 | 3.64 ± 2.33 | .964 |
| Maximum procedural pain* | 4 [1; 6] | 4 [2;5] | .777 |
| Morphine administered | 14/31 (45.2) | 13/28 (46.4) | 1.000 |
| Total morphine dose, mg | 2.74 ± 3.61 | 2.00 ± 2.71 | .379 |
| Midazolam administered | 2/31 (6.5) | 3/28 (10.7) | .661 |
| Total midazolam dose, mg | 0.29 ± 1.19 | 0.50 ± 1.95 | .617 |
| Nausea during procedure | 1/31 (3.2) | 0/28 (0.0) | 1.000 |
| Dizziness during procedure | 1/31 (3.2) | 0/28 (0.0) | 1.000 |
|
Values are mean ± SD, median [Q1; Q3], or n/N (%). For visual analogue scale (VAS) anxiety and pain, both mean ± SD and median [Q1; Q3] are reported due to skewness of the distributions. P values for rows reporting mean ± SD are from Student’s t test; P values for rows reporting median [Q1; Q3] are from Mann–Whitney U tests. Categorical variables were compared using Fisher’s exact test. Total morphine and midazolam doses (mg) include zeros for patients not receiving the drug. SD, standard deviation; VR, virtual reality. * VAS 0-10. |
|||
In a post hoc ANCOVA adjusting for baseline anxiety, VR was not associated with lower peak procedural anxiety (adjusted mean difference, −0.21; 95%CI, −1.62-1.20; P = .773). Results remained consistent after additional adjustment for intraoperative morphine and midazolam dose (table S1 and table S2).
Given the significantly higher angiographic complexity in the VR group, a sensitivity analysis was conducted using multivariable linear regression. After adjusting for J-CTO score and baseline anxiety, the effect of VR on procedural anxiety remained non-significant (adjusted coefficient -0.40; 95%CI, -1.81 to 1.02; P = .576). In this model, procedural complexity was not independently associated with patient-reported anxiety (P = .370); see table S1.
Secondary endpoints
VASp was similar in the 2 groups: 3.68 ± 3.39 with VR and 3.64 ± 2.33 with usual care (mean difference, 0.03; 95%CI, −1.50-1.57; P = .964). Median [IQR] values were 4 [1; 6] and 4 [2; 5], respectively (P = .777).
Intraoperative pharmacologic treatment did not differ between groups. Morphine was administered to 14 of 31 patients (45.2%) in the VR group and 13 of 28 (46.4%) in the control group (P = 1.00). Total morphine dose (including zero valus for nonuse) was 2.74 ± 3.61 mg and 2.00 ± 2.71 mg, respectively (P = .379). Midazolam was administered to 2 of 31 patients (6.5%) in the VR group and 3 of 28 (10.7%) in the control group (P = .661), with total midazolam doses of 0.29 ± 1.19 mg and 0.50 ± 1.95 mg, respectively (P = .617) (figure 3 and table 2).
Safety and acceptability
Symptoms potentially attributable to VR were uncommon. Nausea occurred in 1 patient (3.2%) in the VR group and in no patients in the control group (P = 1.00). Dizziness occurred in 1 patient (3.2%) in the VR group and in no patients in the control group (P = 1.00).
Among patients assigned to VR, 25 of 31 (80.6%) reported that they would be willing to use a relaxation video again during a future intervention; 5 (16.1%) would not, and 1 (3.2%) was uncertain.
DISCUSSION
We conducted a randomized trial to evaluate whether VR-based audiovisual distraction during elective CTO PCI can improve the patient experience. Several key findings emerge. First, VR did not meaningfully reduce patient-reported peak procedural anxiety, and pain outcomes were similarly neutral, arguing against a clinically relevant anxiolytic or analgesic effect with the intervention as delivered. Second, VR did not reduce the use of intraoperative opioid or benzodiazepine requirements, suggesting limited incremental benefit beyond contemporary usual care in the cath lab. Third, VR implementation appeared feasible and was not associated with signals of procedural harm, with overall procedural outcomes remaining comparable despite an imbalance toward greater angiographic complexity in the VR group. Finally, acceptability was generally high among patients assigned to VR, supporting its role as a patient-centered option for selected individuals even when average effects on anxiety are modest.
Interpreting the neutral result in the context of prior interventional cardiology VR trials
Several factors may explain the absence of a measurable anxiolytic effect in ReViCTO. First, peak procedural anxiety levels were relatively low, leaving limiting the potential for improvement and increasing the likelihood of a floor effect. A similar pattern has been observed in contemporary minimally sedated structural interventions, in which overall anxiety burden is modest and VR has not consistently produced detectable differences on global assessments.18
Second, anxiety and pain were assessed immediately after the procedure as a single recalled measure of peak intensity. Although pragmatic and consistent with the study protocol, this approach relies on retrospective recall and may underestimate brief, procedure-specific peaks of discomfort that are particularly relevant in CTO PCI, such as arterial puncture, prolonged immobility, complex device manipulation, or episodes of ischemic chest pain.19 Consequently, any benefit limited to these discrete high-stress intervals may have been diluted when summarized as a single postprocedural peak value.
Third, the VR content in the ReViCTO consisted of passive content (documentary viewing). Across procedural settings, the magnitude of VR’s effect appears to depend on how strongly the experience captures attention and induces relaxation. Interventions incorporating guided relaxation, hypnosis-based modules, or interactive elements have more consistently demonstrated larger and more reproducible effects than passive viewing alone.20-22
In the context of the broader interventional cardiology literature, the neutral effect of VR on peak intraoperative anxiety in ReViCTO is therefore less surprising. Existing cath lab and structural heart studies suggest that observable benefit depends on when VR is delivered, which patients are targeted, particularly those with higher baseline anxiety, and how outcomes are measured. In transcatheter aortic valve implantation (TAVI), early randomized studies reported lower VAS-based procedural anxiety, supporting feasibility and a potential anxiolytic effect.11 In contrast, a larger minimalistic TAVI randomized study found improvements in state anxiety (State-Trait Anxiety Inventory, state scale [STAI-S]) and perceived procedure duration, yet less consistent effects on VAS-based anxiety or pain, underscoring the influence of measurement instruments on signal detection.23 Evidence from coronary procedures is similarly nuanced. In the VR InCard program, which targeted patients with elevated preoperative anxiety and incorporated structured preprocedural sessions, the primary analysis was neutral but adjusted analyses suggested modest reductions, with heterogeneity by clinical context.24,25 Similarly, a randomized trial conducted before coronary angiography reported anxiety reduction using questionnaire-based assessments rather than a single recalled postoperative peak rating.26 Collectively, these data support the interpretation that VR may be most effective when targeted to patients with higher baseline anxiety, delivered during anticipatory or discrete high-stress phases, and evaluated using instruments sensitive to global state anxiety, rather than relying solely on a single recalled peak score.22,27
Pharmacologic co-intervention and post hoc adjustment
A key pragmatic feature of ReViCTO was that background anxiolysis and analgesia were not protocolized, reflecting real-world practice in the cath lab where conscious sedation and intraoperative comfort measures are typically individualized. This approach is consistent with contemporary cath lab randomized studies evaluating patient comfort under usual care conditions.28,29 In the final dataset, intraoperative morphine and midazolam use were similar, and dose distributions did not differ materially. To align with current analytic recommendations, we performed a post hoc ANCOVA adjusting peak anxiety for baseline anxiety, an approach that can improve precision when baseline values are prognostic.30 Baseline anxiety was strongly associated with peak anxiety, while VR assignment was not (adjusted VR effect approximately -0.32 VAS points; P = .64). Adding opioid and benzodiazepine doses did not materially change the VR estimate, supporting that the neutral primary result is unlikely to be explained by baseline imbalance or differential pharmacologic rescue. Baseline anxiety was higher in the usual care group; adjusted analyses yielded consistent findings.
Feasibility, tolerability, and patient-center implementation
From a feasibility standpoint, VR use in CTO PCI appeared safe and compatible with the cath lab environment, with nausea and dizziness reported rarely and no signal suggesting increased procedural instability. The practical challenge was tolerability during long procedures: early discontinuation of the headset occurred in 9 VR patients, which is consistent with broader evidence that fully immersive head-mounted displays can trigger discomfort or cybersickness in a minority of users, particularly with extended exposure.31 Implementation studies further indicate that device-related factors—such as physical interference, usability barriers, and the need for individual tailoring—can influence both uptake and sustained use, supporting a selective rather than universal deployment strategy.32 These observations support a patient-center approach: VR may be most useful when offered to patients with higher baseline anxiety, a clear preference for audiovisual distraction, or anticipated prolonged immobility, and when focused around discrete high-stress phases. Preoperative counseling remains essential with VR serving as a complementary, rather than substitutive, strategy.
Limitations
This trial has limitations that should be considered. First, it was a single-center, open-label study and subjective outcomes may be susceptible to reporting buas; however, randomization, standardized outcome collection, and consistently neutral findings across patient-reported outcomes and medication use make a major bias-driven effect unlikely. Second, recruitment was not consecutive and 12.6% of eligible patients declined participation, which may limit generalizability; nonetheless, this also reflects real-world acceptability of a wearable intervention, and the decliner rate is transparently reported. Third, anxiety and pain were assessed as a single postoperative peak rating rather than repeated measures during predefined high-stress phases; this approach was identical in both groups, and recalled peak experience remains clinically relevant for satisfaction and willingness to undergo future procedures. Fourth, anxiolysis and analgesia were not protocolized and were operator-directed, potentially attenuating any incremental benefit of VR. Nevertheless, this pragmatic design improves applicability and bailout medication was systematically recorded. Fifth, VR content was passive and exposure time likely varied due to early discontinuation; the intention-to-treat analysis therefore estimates the real-world effect of offering VR. Finally, although angiographic complexity (J-CTO score) was higher in the VR group, multivariable analyses indicated this imbalance did not explain the neutral anxiety results and complexity was not a significant predictor of distress in this cohort. Benzodiazepine amnesia may attenuate recall; midazolam use was low and balanced.
CONCLUSIONS
In this randomized trial of patients undergoing elective CTO PCI, although immersive VR was feasible and well tolerated, it did not reduce patient-reported peak intraoperative anxiety or pain, or the need for intraoperative morphine or midazolam vs usual care.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The protocol was approved by the Clinical Research Ethics Committee of Hospital Clínico Universitario de València (28 February, 2022), and all participants gave their prior written informed consent. Sex distribution is reported, and reporting adhered to SAGER guidelines to mitigate potential sex- and gender-related bias.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Generative artificial intelligence was used to support language editing and formatting of the revised manuscript. No artificial intelligence tools were used to analyze study data. All content was reviewed and approved by the authors, who take full responsibility for the manuscript.
AUTHORS’ CONTRIBUTIONS
A. Fernández-Cisnal and B. Silla conceived the study and designed the trial. A. Fernández-Cisnal, B. Silla, C.E. Vergara-Uzcategui, J.M. Ramón, E. Valero, and C. Romero Menor contributed to patient inclusion and data acquisition. A. Fernández-Cisnal performed the statistical analyses and drafted the manuscript. B. Silla, C.E. Vergara- Uzcategui, J.M. Ramón, E. Valero, C. Romero Menor, J. Núñez, V. Bodí, and G. Miñana critically revised the manuscript for important intellectual content. All authors approved the final version and agreed to be accountable for all aspects of the work.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- CTO PCI is a long, technically complex cath lab procedure that can cause anxiety and discomfort for patients.
- Patient anxiety during coronary procedures may worsen perceived discomfort and can increase the need for intraoperative sedatives or analgesics, although pharmacological strategies show modest and variable benefits in routine practice.
- VR provides immersive audiovisual distraction and has been shown to reduce pain and anxiety in several procedural and clinical settings.
- Early studies in interventional cardiology suggest VR can be feasible during procedures performed under conscious sedation, but evidence is mixed and randomized data in CTO PCI were lacking.
WHAT DOES THIS STUDY ADD?
- In the prospective, single-center randomized ReViCTO trial with 59 patients undergoing elective CTO PCI, intraoperative VR goggles were feasible, safe, and generally well tolerated.
- VR did not significantly reduce patient-reported peak procedural anxiety vs usual care and similarly showed no reduction in peak pain.
- VR did not decrease the use or total dose of intraoperative morphine or midazolam, suggesting limited incremental benefit over contemporary usual care.
- Despite a higher angiographic complexity in the VR group, procedural outcomes and complication rates were comparable, and most VR patients reported they would consider using the device again in future interventions.
REFERENCES
1. Patel VG, Brayton KM, Tamayo A, et al. Angiographic Success and Procedural Complications in Patients Undergoing Percutaneous Coronary Chronic Total Occlusion Interventions. JACC Cardiovasc Interv. 2013;6:128-136.
2. Brilakis ES, Grantham JA, Rinfret S, et al. A Percutaneous Treatment Algorithm for Crossing Coronary Chronic Total Occlusions. JACC Cardiovasc Interv. 2012;5:367-379.
3. Christopoulos G, Karmpaliotis D, Alaswad K, et al. Application and outcomes of a hybrid approach to chronic total occlusion percutaneous coronary intervention in a contemporary multicenter US registry. Int J Cardiol. 2015;198:222-228.
4. Fefer P, Knudtson ML, Cheema AN, et al. Current Perspectives on Coronary Chronic Total Occlusions. J Am Coll Cardiol. 2012;59:991-997.
5. Tajti P, Burke MN, Karmpaliotis D, et al. Update in the Percutaneous Management of Coronary Chronic Total Occlusions. JACC Cardiovasc Interv. 2018;11:615-625.
6. Delewi R, Vlastra W, Rohling WJ, et al. Anxiety levels of patients undergoing coronary procedures in the catheterization laboratory. Int J Cardiol. 2017;228:926-930.
7. Trotter R, Gallagher R, Donoghue J. Anxiety in patients undergoing percutaneous coronary interventions. Heart Lung. 2011;40:185-192.
8. Astin F, Jones K, Thompson DR. Prevalence and patterns of anxiety and depression in patients undergoing elective percutaneous transluminal coronary angioplasty. Heart Lung. 2005;34:393-401.
9. Vlastra W, Delewi R, Rohling WJ, et al. Premedication to reduce anxiety in patients undergoing coronary angiography and percutaneous coronary intervention. Open Heart. 2018;5:000833.
10. Teh JJ, Pascoe DJ, Hafeji S, et al. Efficacy of virtual reality for pain relief in medical procedures:a systematic review and meta-analysis. BMC Med. 2024;22:64.
11. Bruno RR, Lin Y, Wolff G, et al. Virtual reality-assisted conscious sedation during transcatheter aortic valve implantation:a randomised pilot study. EuroIntervention. 2020;16:1014-1020.
12. Roxburgh T, Li A, Guenancia C, et al. Virtual Reality for Sedation During Atrial Fibrillation Ablation in Clinical Practice:Observational Study. J Med Internet Res. 2021;23:26349.
13. Fernández-Cisnal A, Silla B, María Ramón J, et al. Efficacy of virtual reality reducing anxiety during CTO revascularization:the ReViCTO trial design. REC Interv Cardiol. 2023;5:203-209.
14. Schulz KF, Grimes DA. Unequal group sizes in randomised trials:guarding against guessing. Lancet. 2002;359:966-970.
15. Spertus JA, Winder JA, Dewhurst TA, et al. Development and evaluation of the Seattle Angina questionnaire:A new functional status measure for coronary artery disease. J Am Coll Cardiol. 1995;25:333-341.
16. Delewi R, Vlastra W, Rohling WJ, et al. Anxiety levels of patients undergoing coronary procedures in the catheterization laboratory. Int J Cardiol. 2017;228:926-930.
17. Chan AW, Boutron I, Hopewell S, et al. SPIRIT 2025 statement:updated guideline for protocols of randomised trials. BMJ. 2025;389:081477.
18. Chatterjee S, Ooms J, De Ronde M, et al. Anxiety during transcatheter aortic valve replacement under local anesthesia - the ART-VR trial. Cardiovasc Revasc Med. 2025;79:71-77.
19. Brilakis ES, Banerjee S, Karmpaliotis D, et al. Procedural Outcomes of Chronic Total Occlusion Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2015;8:245-253.
20. Bekelis K, Calnan D, Simmons N, MacKenzie TA, Kakoulides G. Effect of an Immersive Preoperative Virtual Reality Experience on Patient Reported Outcomes:A Randomized Controlled Trial. Ann Surg. 2017;265:1068-1073.
21. Gullo G, Rotzinger DC, Colin A, et al. Virtually Augmented Self-Hypnosis in Peripheral Vascular Intervention:A Randomized Controlled Trial. Cardiovasc Intervent Radiol. 2023;46:786-793.
22. El Mathari S, Hoekman A, Kharbanda RK, et al. Virtual Reality for Pain and Anxiety Management in Cardiac Surgery and Interventional Cardiology. JACC:Advances. 2024;3:100814.
23. Lind A, Ahsan M, Totzeck M, et al. Virtual reality-assisted distraction during transcatheter aortic valve implantation under local anaesthesia:A randomised study. Int J Cardiol. 2023;387:131130.
24. Groenveld TD, Breunissen EHW, Bonnes JL, et al. Virtual Reality to Reduce Preprocedural Anxiety During Invasive Coronary Angiography. JACC:Advances. 2025;4:101976.
25. Breunissen EHW, Groenveld TD, Garms L, Bonnes JL, Van Goor H, Damman P. Virtual reality to reduce periprocedural anxiety during invasive coronary angiography:rationale and design of the VR InCard trial. Open Heart. 2024;11:002628.
26. Keshvari M, Yeganeh MR, Paryad E, Roushan ZA, Pouralizadeh M. The effect of virtual reality distraction on reducing patients'anxiety before coronary angiography:a randomized clinical trial study. Egypt Heart J. 2021;73:98.
27. Micheluzzi V, Burrai F, Casula M, et al. Effectiveness of virtual reality on pain and anxiety in patients undergoing cardiac procedures:A systematic review and meta-analysis of randomized controlled trials. Curr Probl Cardiol. 2024;49:102532.
28. Boukantar M, Chiaroni PM, Gallet R, et al. A Randomized Controlled Trial of Nonfasting vs Fasting Before Interventional Coronary Procedures. JACC Cardiovasc Interv. 2024;17:1200-1210.
29. Ferreira D, Hardy J, Meere W, et al. Fasting >no fasting prior to catheterization laboratory procedures:the SCOFF trial. Eur Heart J. 2024;45:4990-4998.
30. Tackney MS, Morris T, White I, Leyrat C, Diaz-Ordaz K, Williamson E. A comparison of covariate adjustment approaches under model misspecification in individually randomized trials. Trials. 2023;24:14.
31. Cossio S, Chiappinotto S, Dentice S, et al. Cybersickness and discomfort from head-mounted displays delivering fully immersive virtual reality:A systematic review. Nurse Educ Pract. 2025;85:104376.
32. Kouijzer MMTE, Kip H, Bouman YHA, Kelders SM. Implementation of virtual reality in healthcare:a scoping review on the implementation process of virtual reality in various healthcare settings. Implement Sci Commun. 2023;4:67.















