ABSTRACT
Introduction and objectives: Drug-eluting stents (DES) represent the standard-of-care for treatment of de novo coronary artery lesions. However, permanent vessel metallic scaffolding is associated with short- and long-term complications. Drug-coated balloon (DCB) angioplasty, a scaffold-free alternative, may address these issues. This study aimed to compare the safety and efficacy profile of DCB vs DES for the treatment of de novo coronary lesions.
Methods: Electronic databases were interrogated for randomized controlled trials (RCT) comparing DCB with DES for the treatment of de novo lesions. The primary endpoint was target lesion revascularization, while secondary endpoints included target vessel revascularization, myocardial infarction, target vessel myocardial infarction, vessel thrombosis, cardiac death, all-cause mortality, major adverse cardiovascular events, and major bleeding. Effect estimates and 95% confidence intervals (95%CI) were calculated using random-effects models. Angiographic outcomes included binary restenosis, late lumen loss, minimal luminal diameter, net luminal gain, and diameter stenosis.
Results: A total of 13 RCT including 7776 patients were included. Sirolimus-coated DCB was tested in 1 RCT, while others assessed paclitaxel-coated DCB. Both groups showed comparable target lesion revascularization rates (RR, 1.24; 95%CI, 0.79-1.95; mean follow-up, 22.9 months) and similar results for all other ischemic endpoints. Major bleeding was significantly lower with DCB angioplasty vs DES (RR, 0.65; 95%CI, 0.43-0.96), an effect mainly observed in trials adopting shorter durations of dual antiplatelet therapy in the DCB group. These findings were consistent across multiple sensitivity and subgroup analyses. At angiographic follow-up (mean, ~8.4 months), DCB was associated with lower late lumen loss, whereas DES achieved lower diameter stenosis and greater minimal luminal diameter and net luminal gain, with no significant difference in binary restenosis.
Conclusions: DCB angioplasty appears as a viable therapeutic strategy for the treatment of de novo coronary lesions in contemporary clinical practice. However, large-scale randomized trials are warranted to confirm its long-term safety, efficacy, and optimal patient selection (PROSPERO: CRD42024587941).
Keywords: Drug coated balloon. Drug eluting stent. De novo coronary lesion. Percutaneous coronary intervention.
RESUMEN
Introducción y objetivos: Los stents farmacoactivos (SFA) representan el estándar de tratamiento para las lesiones coronarias de novo, aunque el implante permanente de una malla metálica puede asociarse a complicaciones a corto y largo plazo. La angioplastia con balón farmacoactivo (BFA), estrategia «sin andamiaje», podría superar estas limitaciones. El objetivo fue comparar la eficacia y la seguridad del BFA frente al SFA en lesiones coronarias de novo.
Métodos: Se realizaron búsquedas sistemáticas para identificar ensayos clínicos aleatorizados que compararan BFA y SFA en lesiones de novo. El objetivo primario fue la revascularización de la lesión diana; los objetivos secundarios incluyeron la revascularización del vaso diana, el infarto de miocardio, el infarto de miocardio del vaso diana, la trombosis, la muerte por causa cardiaca, la muerte por cualquier causa, los eventos cardiovasculares adversos mayores y el sangrado mayor. Las estimaciones del efecto y los intervalos de confianza del 95% (IC95%) se calcularon mediante modelos de efectos aleatorios. Los desenlaces angiográficos incluyeron reestenosis binaria, pérdida luminal tardía, diámetro luminal mínimo, ganancia luminal neta y estenosis del diámetro.
Resultados: Se incluyeron 13 ensayos clínicos aleatorizados (7.776 pacientes). Un estudio evaluó BFA con sirolimus y los restantes BFA con paclitaxel. No hubo diferencias significativas en la revascularización de la lesión diana (RR = 1,24; IC95%, 0,79-1,95; seguimiento medio 22,9 meses) ni en otros desenlaces isquémicos. El sangrado mayor fue menor con BFA (RR = 0,65; IC95%, 0,43-0,96), principalmente en los ensayos con menor duración de la doble antiagregación plaquetaria. En el seguimiento angiográfico (~8,4 meses), el BFA mostró menor pérdida luminal tardía, mientras que con el SFA se observaron menor estenosis del diámetro y mayores diámetros luminales mínimos y ganancia luminal neta, sin diferencias en la reestenosis binaria. Los resultados fueron consistentes en los análisis de sensibilidad y de subgrupos.
Conclusiones: El BFA parece una alternativa viable en las lesiones coronarias de novo, aunque se requieren ensayos de mayor tamaño para confirmar su seguridad, eficacia y adecuada selección de los pacientes a largo plazo (PROSPERO: CRD42024587941).
Palabras clave: Balón farmacoactivo. Stent farmacoactivo. Lesión coronaria de novo. Intervención coronaria percutánea.
Abbreviations
DCB: drug-coated balloon. DES: drug-eluting stent. MACE: major adverse cardiovascular events. PCI: percutaneous coronary intervention. RCT: randomized clinical trial.
INTRODUCTION
Percutaneous coronary intervention (PCI) with drug-eluting stents (DES) is the current standard of care for coronary revascularization in patients with coronary artery disease (CAD).1 Despite substantial advances in stent technology, the permanent presence of metal scaffolding presents a persistent hazard of stent-related adverse events (e.g.,, stent failure).2 These events occur in ~2% of patients per year and persist over time, thereby limiting long-term outcomes.3,4 Drug-coated balloons (DCB) have emerged as an attractive “leave-nothing-behind” alternative when an optimal angiographic result is achieved,5,6 delivering antiproliferative drugs directly into the arterial wall without leaving a permanent implant, potentially promoting arterial healing, preserving vessel anatomy, and avoiding stent-related complications.7,8 DCB have demonstrated efficacy in the treatment of in-stent restenosis,9,10 an accumulating evidence supports their use in selected settings, such as small-vessel disease (SVD) and bifurcation lesions.11-13 However, their role in de novo coronary lesions remains controversial. Randomized controlled trials (RCT) evaluating DCB have often been limited by small sample sizes, heterogeneous populations, variable procedural protocols, and reliance on surrogate angiographic endpoints.14 Notably, the 2 largest RCT published to date have yielded conflicting results, leaving the comparative efficacy of DCB vs DES unresolved.15-16
We conducted a systematic review and pairwise meta-analysis of RCT to assess the efficacy and safety profile of DCB vs DES for the treatment of de novo coronary lesions.
METHODS
This meta-analysis was prospectively registered in PROSPERO (CRD4202458794) and followed the PRISMA and Cochrane Collaboration guidelines (tables 1S and S2).17 Ethical approval was waived given the nature of the study.
Eligibility criteria
Studies were eligible if they: a) enrolled patients undergoing PCI for de novo coronary artery lesions of any vessel size; b) randomized participants to DCB angioplasty or DES implantation; and c) reported at least one outcome of interest. Exclusion criteria included: a) absence of a second- or third-generation DES as the primary comparator; and b) evaluation of DCB as part of a hybrid or combined strategy with concomitant stent implantation. No language restrictions were applied.
Search strategy, study selection, data extraction, and qualitative assessment
Three electronic databases (PubMed, Web of Science, and Scopus) were searched from inception to 29 October 2025 (table S1). Additional sources included society websites, conference proceedings, and reference lists of eligible studies. Two reviewers (R. Rinaldi and K. Bujak) independently screened titles and abstracts, assessed full texts for final inclusion, and extracted trial- and arm-level data into standardized spreadsheets. Disagreements were resolved by consensus. When multiple reports referred to the same trial, the one with the longest follow-up was selected. Study quality was assessed using the Cochrane Risk of Bias 2 tool.18 Publication bias was evaluated using funnel plots and Egger’s test, with adjustment using the trim-and-fill method for endpoints suspected of publication bias.
Study endpoints
The prespecified primary endpoint was trial-defined target lesion revascularization at the longest follow-up. When target lesion revascularization was not reported, target vessel revascularization was used as the closest available reported revascularization endpoint. Secondary endpoints included target vessel revascularization, myocardial infarction (MI), target-vessel MI, vessel thrombosis, cardiac and all-cause mortality, major adverse cardiovascular events (MACE), and major bleeding. When multiple definitions of the same endpoint were reported within a trial, we extracted the definition most closely aligned with standardized Academic Research Consortium criteria (eg, clinically driven target lesion revascularization).19,20 Angiographic endpoints included binary restenosis, late lumen loss (postoperative minus follow-up minimal luminal diameter), minimal luminal diameter, diameter stenosis, and net luminal gain (follow-up minus preoperative minimal luminal diameter).
Statistical analysis
Analyses were performed on an intention-to-treat (ITT) basis. Pairwise meta-analyses were conducted using a frequentist random- effects models (DerSimonian-Laird estimator) to account for anticipated biological variability. Binary outcomes were expressed as risk ratios (RR) with 95% confidence interval (95%CI), while continuous outcomes were reported as standardized mean differences (SMD). A continuity correction of 0.5 was applied when 0 events occurred in either study group. Heterogeneity was quantified using the Cochran Q test and I² statistic, interpreted as low (< 25%), moderate (25-50%), or high (> 50%). Baujat plots were used to identify influential studies. Statistical significance was defined as two-sided P < .05.
Prespecified subgroup analyses were performed by stratifying the trials according to study-level characteristics, including: a) drug type (paclitaxel vs sirolimus); b) lesion characteristics (SVD < 3.0 mm; large vessels; bifurcation; ST-segment elevation myocardial infarction culprit lesion); c) dual antiplatelet therapy (DAPT) duration (shorter in DCB group vs equal duration in both groups). Meta- regression analyses were conducted to assess the impact of confounding variables, including a) mean study-level reference vessel diameter; b) follow-up duration; and c) year of publication. Sensi- tivity analyses were performed for both the primary and secondary outcomes using alternative modeling approaches (eg, incidence rate ratios; trim and fill analysis, Bayesian random-effects meta-analysis with weakly informative priors), and by excluding studies: a) at high risk of bias or without ITT analysis; b) using paclitaxel-eluting stents as comparators; c) using ultrasound-guided DCB; d) reporting target vessel revascularization instead of target lesion revascularization or non-Academic Research Consortium primary endpoint definitions (eg, definitions other than clinically driven target lesion revascularization); e) using DCB devices other than most commonly tested one in included trials (SeQuent Please); or f) identified as principal contributors to heterogeneity. All analyses were conducted using R version 4.2.0 (R Foundation for Statistical Computing, Austria).
RESULTS
Study selection, baseline, and angiographic characteristics
The study selection flowchart is reported in figure S1. After screening, 13 RCT met the inclusion criteria and were included in the quantitative synthesis.11,12,15,16,21-29 Among these, 12 trials had already been published,11,12,15,21-29 and one was available only as a conference proceeding.16 Key trials characteristics are summarized in table 1. A total of 5 studies focused exclusively on SVD,11,12,25,27,28 4 on de novo lesions of any vessel size,15,16,23,29 3 on ST-segment elevation myocardial infarction,26,30,31 and 1 on bifurcation lesions.24 One trial evaluated a sirolimus-coated balloon,16 while the remaining 12 tested paclitaxel-coated balloons.11,12,15,21-29 The weighted mean clinical follow-up was 22.9 months, and the weighted mean angiographic follow-up (reported in 10 studies), 8.4 months. Available endpoints and definitions across included trials are reported in tables S2 and S3, respectively. Overall, 7776 patients were included (3896 DCB vs 3880 DES), of whom 46.0% presented with acute coronary syndrome. The weighted mean reference vessel diameter was 2.95 mm. The weighted mean minimum anticipated DAPT duration was 5.0 months in the DCB group and 5.9 months in the DES group (table 1). Baseline clinical, angiographic, and procedural characteristics are presented in tables S4 and S5. The risk of bias was high in 3 studies (imprecise reporting or deviation from the ITT analysis) and moderate in remaining studies due to the open-label design (figure S2). Publication bias was detected only for late lumen loss and minimal luminal diameter endpoints (figure S3).
Table 1. Key baseline characteristics across included randomized controlled trials
| Study, year | Sample size | Primary endpoint | DCB type | DES type | Type of de novo target lesion | DAPT | Follow-up, month* |
|---|---|---|---|---|---|---|---|
| SELUTION DeNovo, 202516 | 3323 | Composite of cardiac death, TVMI, and clinically driven target lesion revascularization | SELUTION SLR (sirolimus) | DES according to local practice | RVD ≥ 2.0 and ≤ 5.0 mm | 6 (CCS), 12 (ACS) / 6 (CCS), 12 (ACS) | Clinical: 12 |
| REC-CAGEFREE I, 202515 | 2272 | Composite of CV death, TVMI, and clinically driven and protocol driven target lesion revascularization | Swide (paclitaxel) | DP-SES (Firebird 2) | Noncomplex coronary artery disease (irrespective of RVD) | 1 / 1 (A+C or A+T followed by A or C monotherapy; the decision on DAPT > 1 month was left to the treating physician, and the overall DAPT duration was shorter in the DCB group on average) | Clinical: 36 |
| Dissolve SVD, 202427 | 247 | In-segment percent diameter stenosis | Dissolve (paclitaxel) | DP-ZES (Resolute) | RVD ≥ 2.25 and ≤ 2.75 mm | 3 / 6 (A+C or A+T followed by A monotherapy) | Angiographic: 9 Clinical: 12 |
| REVELATION, 202422 | 120 | Functional assessment using FFR | Pantera Lux (paclitaxel) | DP-SES (Orsiro); DP-EES (Xience) | – RVD: 2.5-4 mm – No severe calcification – Percent diameter stenosis < 50% after thrombus aspiration and predilatation. | 9 / 9 (A+T or A+P) | Angiographic: 9 Clinical: 60 |
| PICCOLETTO-II, 202328 | 232 | In-lesion late lumen loss | Elutax SV/Emperor (paclitaxel) | DP-EES (Xience) | RVD ≥ 2.00 and ≤ 2.75 mm, no LM or ostial lesions, no CTO, no severe calcification or tortuosity, lesion length < 25 mm | 1 / 6 | Angiographic: 6 Clinical: 36 |
| Ke et al., 202324 | 60 | 1-year late lumen loss and cumulative MACE (composite of CV death, TVMI, target lesion thrombosis, or target lesion revascularization/target vessel revascularization. | SeQuent Please (paclitaxel) | DP-ZES (Resolute); DP-EES (Xience); DP-SES (Firebird 2 and Excel) | True BL (Medina 1,1,1; 0,1,1; 1,0,1) and SB ≥ 2.25 mm without severe calcification or tortuosity | 12 / 12 (A+C or A+T followed by A, C or T monotherapy) | Angiographic and clinical: 12 |
| Kawai et al., 202225 | 42 | Endothelial function adjacent to the treated segment by a vasomotion test | SeQuent Please (paclitaxel) | DP-EES (Synergy) | RVD ≥ 2.0 and ≤ 3.0 mm, no LM or ostial lesion, no severe calcification | 3 / 6 (A+C) | Angiographic: 8 |
| Wang et al., 202221 | 184 | In-segment late lumen loss | Vasoguard (paclitaxel) | BP-SES (Cordimax) | RVD ≥ 2.0 and ≤ 4.0 mm, percent diameter stenosis ≥ 50% and successful predilatation | 3 / 12 (A+C) | Angiographic: 9 Clinical: 12 |
| Yu et al., 202229 | 170 | Late lumen loss | SeQuent Please (paclitaxel) | DP-ZES (Resolute); DP-EES (Synergy); BP-DES (Rapamycin, Firehawk) | RVD ≥ 2.25 and ≤ 4.0 mm and lesion length ≤ 30 mm | 1-3 (CCS), 6-12 (ACS) / 12 (A+C or A+T) | Angiographic: 9 Clinical: 12 |
| BASKET-SMALL 2, 202012 | 758 | MACE (composite of CV death, non-fatal MI, and target vessel revascularization), all-cause mortality, probable or definite stent thrombosis, and major bleeding | SeQuent Please (paclitaxel) | DP-EES (Xience); DP-DES (Paclitaxel, Taxus Element) | RVD ≥ 2.0 to < 3.0 mm and successful predilatation | 1 (CCS), 12 (ACS) / 6 (CCS), 12 (ACS) (A+C or A+T or A+P) | Clinical: 36 |
| RESTORE SVD China, 202011 | 230 | In-segment percent diameter stenosis | Restore (paclitaxel) | DP-ZES (Resolute) | RVD ≥ 2.25 and ≤ 2.75 mm | 6 / 6 (A+C or A+T) | Angiographic: 9 Clinical: 24 |
| Gobic´ et al., 201726 | 75 | MACE (composite of CV death, reinfarction, target lesion revascularization and stent thrombosis) and late lumen loss | SeQuent Please (paclitaxel) | BP-DES (Sirolimus, Biomime) | RVD ≥ 2.5 and ≤ 4.0 mm, without severe tortuosity | 12 / 12 (A+C) | Angiographic and clinical: 6 |
| Nishiyama et al., 201623 | 60 | Target lesion revascularization and late lumen loss | SeQuent Please (paclitaxel) | DP-EES (Xience) | Significant stenosis, successful predilatation, and lesion length ≤ 25 mm | 8 / 8 (A+C) | Angiographic and clinical: 8 |
|
A, aspirin; ACS, acute coronary syndrome; ARC, Academic Research Consortium; BL, bifurcation lesions; BP, biodegradable polymer; CABG, coronary artery bypass graft; CCS, chronic coronary syndrome; CS, cardiogenic shock; CTO, chronic total coronary occlusion; CV, cardiovascular; DAPT, dual antiplatelet therapy; DES, drug eluting stent; DoCE, device-oriented composite endpoint; DP, durable polymer; DS, diameter stenosis; EES, sirolimus eluting stent; eGFR, estimated glomerular filtration rate; FFR, fractional flow reserve; ISR, in-stent restenosis; IVUS, intravascular ultrasound; LM, left main; LVEF, left ventricular ejection fraction; MACE, major adverse cardiovascular events; NSTEMI, non-ST-segment elevation myocardial infarction; P, prasugrel; PCI, percutaneous coronary intervention; pPCI, primary percutaneous coronary intervention; RVD, reference vessel diameter; SB, side branch; SES, sirolimus eluting stent; STEMI, ST-segment elevation myocardial infarction; T, ticagrelor; TVMI, target vessel myocardial infarction; VHD, valvular heart disease; ZES, zotarolimus eluting stent. * Follow-up refers to the maximum available. |
|||||||
Primary endpoint
No statistically significant difference was observed between DCB and DES for target lesion revascularization (RR, 1.24; 95%CI, 0.79-1.95; figure 1), with high heterogeneity (I² = 54.8%).
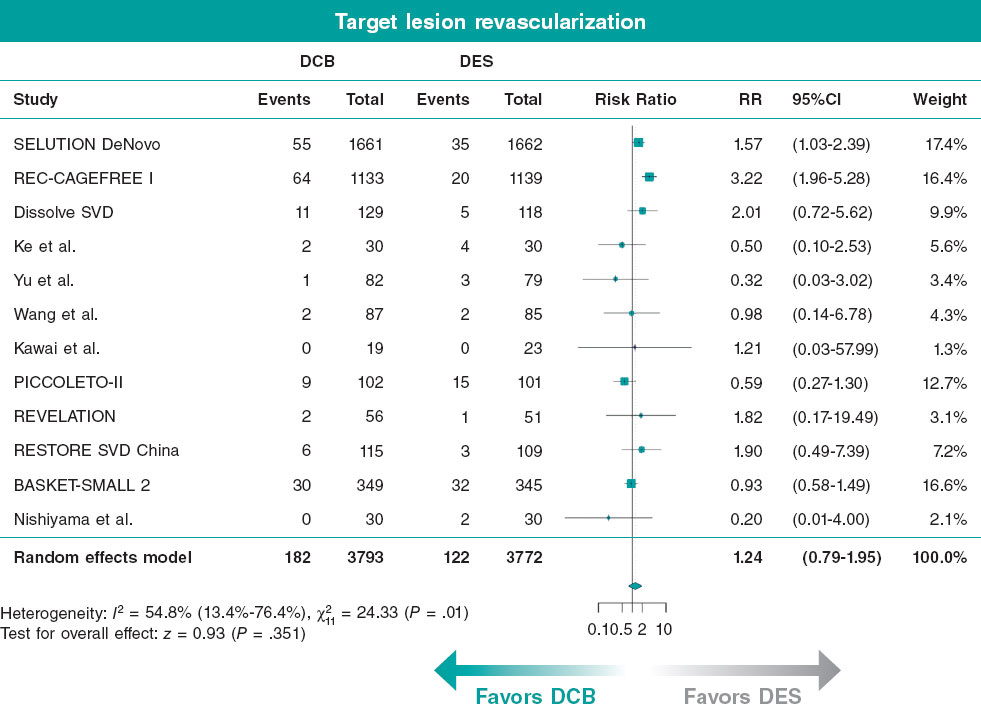
Figure 1. Results of meta-analysis for the primary endpoint. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; RR, risk ratio. The bibliographical references cited in this figure correspond to: Spaulding,16 Gao et al.,15 Liu et al.,27 Ke et al.,24 Yu et al.,29 Wang et al.,21 Kawai et al.,25 Cortese et al.,28 Niehe et al.,22 Tian et al.,11 Jeger et al.,12 and Nishiyama et al.23.
Secondary endpoints
No statistically significant differences were observed between DCB angioplasty and DES implantation for target vessel revascularization (RR, 1.39; 95%CI, 0.91-2.13), MI (RR, 0.92; 95%CI, 0.71-1.19), target-vessel MI (RR, 1.09; 95%CI, 0.78-1.51), cardiac death (RR, 1.05; 95%CI, 0.75-1.48), all-cause mortality (RR, 1.05; 95%CI, 0.82-1.36), or MACE (RR, 1.05; 95%CI, 0.78-1.41) (figure 2 and figure S4A-G). DCB angioplasty was associated with a significantly lower risk of major bleeding vs DES implantation (RR, 0.65; 95%CI, 0.43-0.96), primarily in studies adopting shorter DAPT regimens for DCB-treated patients (figure 2 and figure S4H). However, the interaction test between treatment effect and DAPT duration was not statistically significant (figure S5I). Heterogeneity was low to moderate across endpoints, except for target vessel revascularization, which showed high heterogeneity (I2 = 55.2%).
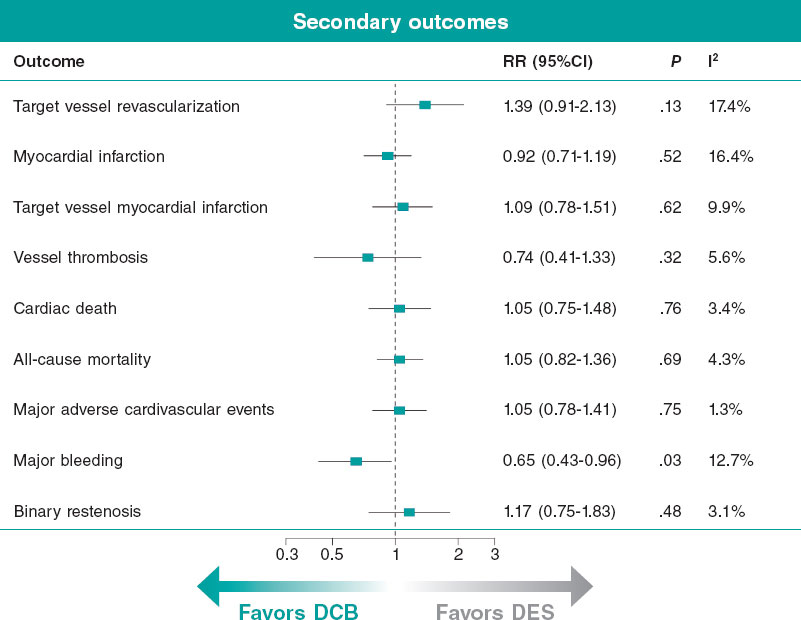
Figure 2. Forest plot presenting pooled effect estimates for the secondary clinical outcomes and binary restenosis. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; RR, risk ratio.
Angiographic endpoints
At angiographic follow-up, binary restenosis did not differ between groups (RR, 1.17; 95%CI, 0.75-1.83) (figure 2 and figure S4I). DES implantation was associated with significantly higher late lumen loss (SMD, −0.41; 95%CI, −0.61 to −0.21), minimal luminal diameter (SMD, -0.50, 95%CI −0.64 to −0.37) and net luminal gain (SMD, −0.61; 95%CI, −0.77 to −0.44) vs DCB angioplasty, whereas DCB angioplasty was associated with greater diameter stenosis vs DES (SMD, 0.32; 95%CI, 0.20 to −0.44) (figure 3; figure S6). Heterogeneity was low for most angiographic outcomes, except for late lumen loss, for which heterogeneity was high.
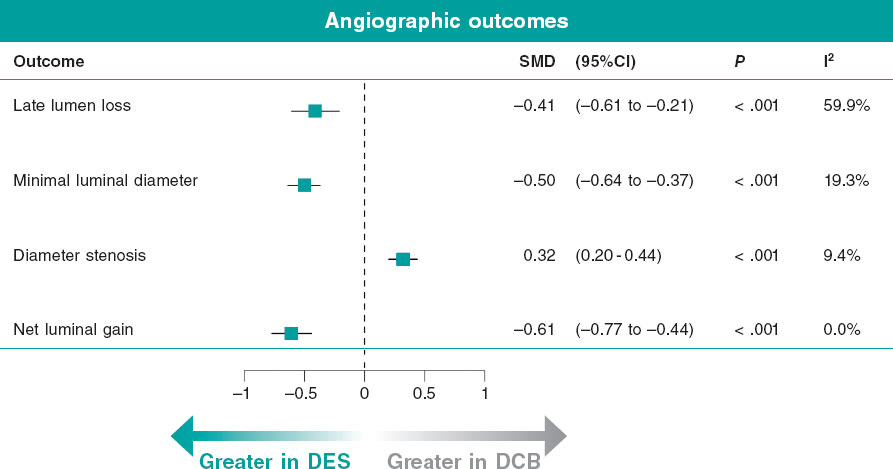
Figure 3. Forest plot presenting pooled effect estimates for angiographic outcomes. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; SMD, standardized mean differences.
Sensitivity and subgroup analyses
Subgroup analyses by DAPT duration showed no significant differences between DCB and DES for primary or secondary ischemic endpoints (figure S5A-H). Subgroup analyses stratified by clinical indication and DCB drug type (paclitaxel- vs sirolimus-coated balloons) also showed no significant interactions (figures S7,S8).
Meta-regression analyses showed consistent treatment effects across reference vessel diameter and follow-up duration (figure S9A,B). Meta-regression according to publication year identified an increasing risk of target lesion revascularization for DCB in more recent trials (figure S9C).
Sensitivity analyses confirmed the results of the primary analyses (figures S10-S15). After excluding the trial using paclitaxel-eluting stent in one-third of control patients, DCB was associated with a significantly higher risk of target vessel revascularization, with no other significant differences observed (figure S16). Bayesian sensitivity analyses were largely consistent with the frequentist results across endpoints. For major bleeding, the treatment effect remained directionally consistent but did not reach statistical significance (figure S17).
Baujat plots identified REC-CAGEFREE I trial15 as the principal source of heterogeneity for target lesion revascularization and target vessel revascularization, and the study by Gobic´ et al.26 for late lumen loss (figure S18). Excluding these studies yielded consistent pooled estimates with low heterogeneity (figure S19).
DISCUSSION
The main findings of our meta-analysis can be summarized as follows: a) DCB angioplasty for de novo lesions showed a comparable risk of target lesion revascularization to DES at a mean follow-up of 2 years; b) no increased risk of MI, target vessel revascularization, cardiac or all-cause mortality was observed with DCB vs DES; c) DCB angioplasty was associated with a significantly lower risk of major bleeding vs DES, although the interaction according to DAPT duration was not statistically significant; and d) from an angiographic perspective, DCB was associated with lower late lumen loss, whereas DES achieved larger minimal luminal diameter and net luminal gain, reflecting the mechanical scaffolding effect (figure 4).
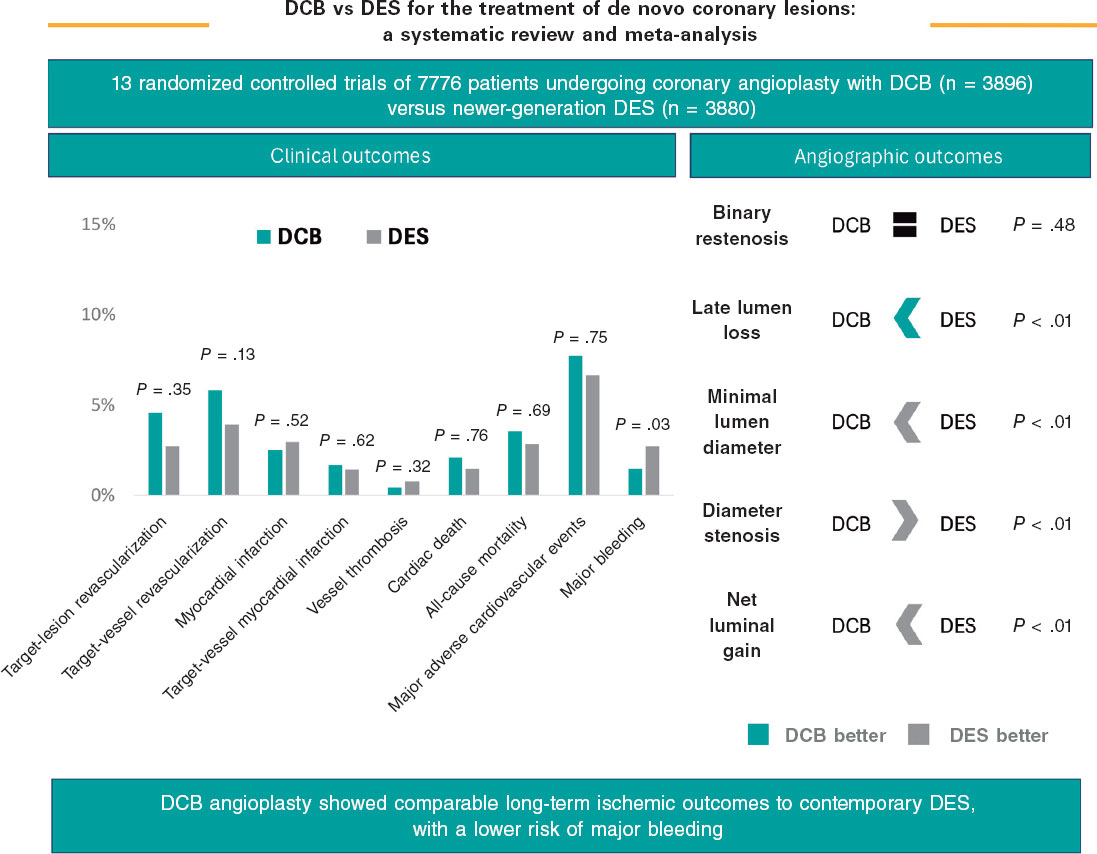
Figure 4. Central illustration. Main results of the study. DCB, drug-coated balloon; DES, drug-eluting stent.
In recent years, the “leave-nothing-behind” concept has gained increasing attention as a strategy to reduce stent-related adverse events and promote physiological vessel healing.32 However, evidence for DCB use in de novo CAD remains limited and heterogeneous.14 Previous meta-analyses generally reported comparable outcomes between DCB and DES, but were frequently restricted to specific subgroups, such as SVD,33,34 large vessels,35,36 or acute MI,37 or included observational data or early-generation DES comparators.38,39 The recent ANDROMEDA individual-patient-data meta-analysis suggested a possible reduction in MACE with DCB in SVD, although its interpretation was limited by the inclusion of only a few RCT and the use of early-generation stents as the comparator group.40
In contrast, our meta-analysis, incorporating 2 large recent RCT,15,16 provides the most contemporary and comprehensive evidence to date. By encompassing a broad spectrum of vessel sizes, lesion complexities, and clinical presentations, it demonstrates the consistent efficacy of DCB angioplasty across diverse clinical settings. Compared with the recent meta-analysis by Niu et al.,41 the present analysis incorporates nearly twice the number of randomized clinical trials and > 4 times the number of patients, including 2 large contemporary trials not previously analysed. In addition, our study focuses exclusively on newer-generation DES and includes bleeding outcomes, dual antiplatelet therapy stratification, meta-regression, and Bayesian sensitivity analyses. Rates of target lesion revascularization were equivalent between DCB and new-generation DES, both at early and extended follow-up, confirming the sustained durability of the DCB strategy over time. These findings suggest that the absence of a permanent metallic scaffold does not compromise vessel patency at 2-year follow-up or predispose to late restenosis, thereby supporting the role of DCB as a valid and reliable alternative to DES for the treatment of de novo coronary lesions.32
Furthermore, our findings help to contextualize the results of the REC-CAGEFREE I trial, which failed to demonstrate the non-inferiority of DCB vs DES in non-complex, de novo coronary lesions.15 Indeed, REC-CAGEFREE I represented the main source of heterogeneity in our analysis, and its exclusion yielded results consistent with the overall pooled effect, markedly reducing heterogeneity. Several methodological aspects may explain this discrepancy. First, unlike most previous RCT that focused on specific subgroups (eg, SVD or ST-egment elevation myocardial infarction), REC-CAGEFREE I adopted an all-comer design and used a device-oriented composite endpoint as its primary outcome. Second, extensive lesion preparation with cutting or scoring balloons in > 60% of cases may have contributed to improved outcomes in the DES group by facilitating stent expansion and reducing restenosis, while also enhancing drug delivery and procedural success in the DCB group, thereby potentially attenuating differences between treatment strategies.42,43 Third, approximately 10% of patients in the DCB group required bailout stenting for significant dissections or residual percent diameter stenosis > 30%, yet were still analysed within the DCB group in both ITT and per-protocol analyses. This methodological choice may have biased results against DCB because these patients typically have higher risks of repeat revascularization due to complex lesions, suboptimal angioplasty results, or residual dissections.44 Importantly, it was not specified whether adverse events occurred in patients treated exclusively with DCB or in those receiving bailout DES, potentially conflating outcomes driven by stented segments. Conversely, in trials such as SELUTION De Novo, where provisional stenting was incorporated within the DCB strategy, the inclusion of DES in the experimental group may have diluted potential treatment differences under ITT analysis, thereby limiting the ability to detect superiority of systematic stent implantation. Finally, the open-label design could have introduced surveillance bias, with a potentially lower threshold for ischemia testing and repeat angiography in the DCB group. Notably, event curves began to diverge around 100 days, coinciding with the transition from telephone to in-person follow-up.44
Furthermore, nearly all trials included in this meta-analysis evaluated paclitaxel-coated DCB, except for the SELUTION DeNovo trial,16 which tested a sirolimus-coated balloon in a large, all-comer population. The consistent findings across studies, irrespective of the antiproliferative agent used, suggest a class effect of DCB in terms of safety and efficacy when compared with contemporary DES. Nonetheless, further head-to-head studies directly comparing paclitaxel- and sirolimus-coated balloons are warranted.
The angiographic findings provide mechanistic insights into the clinical results. While binary restenosis rates were similar, DES achieved lower diameter stenosis and greater minimal luminal diameter and net luminal gain, consistent with its mechanical scaffolding properties. In contrast, DCB demonstrated lower late lumen loss. However, the smaller immediate postoperative lumen with DCB angioplasty may partially account for the lower late lumen loss observed at follow-up. Nevertheless, this difference may also reflect the absence of a permanent metallic implant, potentially more physiological vessel remodelling, and differences in local drug delivery dynamics and antiproliferative efficacy at the treated segment.45 Thus, DES may provide an immediate luminal advantage, as reflected by the greater net luminal gain, which represents a more integrative angiographic parameter less influenced by differences in acute luminal gain. This advantage may be particularly beneficial in complex, calcified, or large-vessel disease. Conversely, DCB may be preferable when vessel compliance and healing are prioritized, such as in SVD, bifurcations, or in younger patients in whom long-term metal avoidance may be advantageous.46 These complementary characteristics suggest an individualized approach that balances the need for mechanical support with the benefits of a “leave-nothing-behind” strategy.
Finally, we observed a lower incidence rate of major bleeding with DCB angioplasty vs DES implantation. This difference was primarily observed in those studies adopting shorter DAPT regimens for DCB-treated patients, suggesting that abbreviated antiplatelet strategies may be safely feasible with DCB and may make this approach a valuable option for patients at high bleeding risk.46-48 However, these findings derive from subgroup analyses stratified according to DAPT regimens specified in the trial protocols rather than the actual DAPT durations received by patients and should therefore be interpreted with caution. Ongoing trials such as the DEBATE (NCT04814212) and the PICCOLETTO IV (NCT06535568) are expected to provide further insights on this issue, particularly in anticoagulated or high-bleeding-risk populations.
Given differences in costs and lack of clinical superiority of DCB vs DES at either short- or long-term follow-up, the value of DCB angioplasty will ultimately depend on identifying specific patient populations who may derive the greatest benefit from a “leave-nothing-behind” strategy, as well as on the results of longer-term studies. Importantly, our findings should be interpreted as evidence of comparable efficacy rather than superiority, and do not support the routine replacement of DES in unselected patients.
Limitations
The present meta-analysis has several limitations. First, a certain degree of heterogeneity was observed across the included randomized trials, largely driven by the REC-CAGEFREE I trial. Furthermore, all included randomized trials enrolled patients in whom both treatment strategies were considered technically feasible and clinically appropriate. Therefore, the study populations reflect selected procedural scenarios rather than unselected, real-world practice. Second, as this was a study-level meta-analysis, our analyses were limited to aggregate endpoints reported in the original publications. Definitions of major bleeding varied substantially across trials, and this heterogeneity may limit the interpretability and comparability of the observed bleeding signal. Third, an analysis stratified by clinical presentation was not feasible because several RCT either did not report event rates according to presentation or excluded one of these subgroups entirely. Fourth, the open-label design of all included studies may have introduced performance or ascertainment bias. Fifth, although nearly all DCB used in the included studies were paclitaxel-coated, DES comparators consisted of a mixture of “limus”-eluting and paclitaxel-eluting stents, which could have influenced relative efficacy estimates in favor of certain DES platforms. Sixth, potential publication bias was detected for angiographic endpoints, possibly reflecting selective reporting of angiographic follow-up data. Although trim-and-fill analyses yielded estimates consistent with the primary results, these surrogate endpoints should be interpreted with caution. Furthermore, as with any meta-analysis based predominantly on published data, the possibility of publication bias cannot be entirely excluded. Seventh, data from the conference proceedings reporting the results of the recent SELUTION De Novo trial,16 whose full-text publication is not yet available, were included in the quantitative synthesis to incorporate all available evidence. Finally, differences in follow-up duration across studies may have limited the ability to capture late adverse events or long-term efficacy trends, highlighting the need for extended follow-up data from ongoing large-scale RCT.
CONCLUSIONS
This meta-analysis indicates that DCB angioplasty may achieve clinical outcomes comparable to those of newer-generation DES for treatment of de novo coronary lesions, without evidence of ischemic superiority. A reduction in major bleeding was observed in studies adopting shorter dual antiplatelet therapy regimens, although this finding should be interpreted with caution. Overall, these findings may support the integration of DCB into routine daily clinical practice as a viable therapeutic strategy in selected clinical scenarios. However, large-scale randomized trials are still warranted to confirm their long- term safety and efficacy profile, as well as their potential superiority over DES.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
Ethical approval was waived given the nature of the study.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
None declared.
AUTHORS’ CONTRIBUTIONS
R. Rinaldi, K. Bujak, G. Occhipinti, and C. Laudani contributed to conceptualization and design of the study, methodology, investigation, data curation, formal analysis, and participated in the drafting and review of the manuscript. S. Brugaletta contributed to the conceptualization and design of the study, methodology, investigation, data curation, project administration, supervision, and drafting, including critical review and editing of the manuscript. J. Sanz Sánchez, R.A. Montone, E. Nicolini, T. Piva, M. Gąsior, F. Ottani, F. Crea, S. Eccleshall, and M. Sabaté, contributed to drafting, review and editing of the manuscript. All authors have reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
CONFLICTS OF INTEREST
F. Crea reports receiving speaker fees from Amgen, AstraZeneca, Abbott, Menarini, Chiesi, and Daiichi Sankyo. M. Sabaté reports receiving consultant fees from Abbott Vascular and iVascular. Other authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- DCB represent a “leave-nothing-behind” strategy designed to avoid long-term stent-related complications and promote natural vessel healing.
- Previous evidence supported the use of DCB mainly for in-stent restenosis and SVD, while data on de novo coronary lesions remain limited and heterogeneous.
- Prior meta-analyses were often constrained by small sample sizes, selective populations, and comparisons with outdated DES.
WHAT DOES THIS STUDY ADD?
- This comprehensive meta-analysis of 13 randomized trials demonstrates a comparable long-term safety and efficacy profile between DCB angioplasty and contemporary DES for de novo coronary lesions.
- DCB angioplasty was associated with a lower risk of major bleeding, particularly in studies using shorter regimens of dual antiplatelet therapy, without compromising ischemic outcomes.
- Findings support DCB as a valid alternative to DES in selected patients, reinforcing the potential of a “leave- nothing-behind” revascularization strategy in modern PCI practice.
REFERENCES
1. Stefanini GG, Byrne RA, Windecker S, Kastrati A. State of the art:coronary artery stents - past, present and future. EuroIntervention. 2017;13:706-716.
2. Serruys PW, Garcia-Garcia HM, Onuma Y. From metallic cages to transient bioresorbable scaffolds: change in paradigm of coronary revascularization in the upcoming decade?Eur Heart J. 2012;33:16-25.
3. Madhavan M V., Kirtane AJ, Redfors B, et al. Stent-Related Adverse Events >1 Year After Percutaneous Coronary Intervention. J Am Coll Cardiol. 2020;75:590-604.
4. Rodriguez F, Harrington RA. Management of Antithrombotic Therapy after Acute Coronary Syndromes. N Engl J Med. 2021;384:452-460.
5. Jeger R V., Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease: Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13:1391-1402.
6. Yerasi C, Case BC, Forrestal BJ, et al. Drug-Coated Balloon for De Novo Coronary Artery Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:1061-1073.
7. Camaj A, Leone PP, Colombo A, et al. Drug-Coated Balloons for the Treatment of Coronary Artery Disease: A Review. JAMA Cardiol. 2025;10:189-198.
8. Colombo A, Leone PP. Redefining the way to perform percutaneous coronary intervention: a view in search of evidence. Eur Heart J. 2023;44:4321-4323.
9. Giacoppo D, Alfonso F, Xu B, et al. Drug-Coated Balloon Angioplasty Versus Drug-Eluting Stent Implantation in Patients With Coronary Stent Restenosis. J Am Coll Cardiol. 2020;75:2664-2678.
10. Giacoppo D, Alfonso F, Xu B, et al. Paclitaxel-coated balloon angioplasty vs drug-eluting stenting for the treatment of coronary in-stent restenosis: a comprehensive, collaborative, individual patient data meta-analysis of 10 randomized clinical trials (DAEDALUS study). Eur Heart J. 2020;41:3715-3728.
11. Tian J, Tang Y da, Qiao S, et al. Two-year follow-up of a randomized multicenter study comparing a drug-coated balloon with a drug-eluting stent in native small coronary vessels: The RESTORE Small Vessel Disease China trial. Catheter Cardiovasc Interv. 2020;95 Suppl 1:587-597.
12. Jeger R V., Farah A, Ohlow MA, et al. Long-term efficacy and safety of drug-coated balloons versus drug-eluting stents for small coronary artery disease (BASKET-SMALL 2): 3-year follow-up of a randomised, non-inferiority trial. Lancet. 2020;396:1504-1510.
13. Fezzi S, Scheller B, Rissanen TT, et al. Drug-coated balloons for coronary bifurcation lesions. EuroIntervention. 2025;21:e1177-e1197.
14. O'Callaghan D, Rai H, Giacoppo D, et al. Drug Coated Balloons Versus Drug-Eluting Stents in Patients With De Novo Coronary Artery Disease. Catheter Cardiovasc Interv. 2025;106:1843-1853.
15. Gao C, He X, Ouyang F, et al. Drug-coated balloon angioplasty with rescue stenting versus intended stenting for the treatment of patients with de novo coronary artery lesions (REC-CAGEFREE I):an open-label, randomised, non-inferiority trial. Lancet. 2024;404:1040-1050.
16. Spaulding CM. One-Year Results of the SELUTION DeNovo Trial Comparing a Strategy of PCI with a Sirolimus-eluting Balloon and Provisional Stenting Versus Systematic DES Implantation to Treat DeNovo Coronary Lesions |tctmd.com. Available at:https://www.tctmd.com/slide/one-year-results-selution-denovo-trial-comparing-strategy-pci-sirolimus-eluting-balloon-and. Accessed 5 Nov 2025.
17. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement:an updated guideline for reporting systematic reviews. BMJ. 2021;372.
18. Sterne JAC, Savovic´J, Page MJ, et al. RoB 2:a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366.
19. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials: The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
20. Fezzi S, Scheller B, Cortese B, et al. Definitions and standardized endpoints for the use of drug-coated balloon in coronary artery disease: consensus document of the Drug Coated Balloon Academic Research Consortium. Eur Heart J. 2025;46:2498-2519.
21. Wang Z, Yin Y, Li J, et al. New Ultrasound-Controlled Paclitaxel Releasing Balloon vs Asymmetric Drug-Eluting Stent in Primary ST-Segment Elevation Myocardial Infarction - A Prospective Randomized Trial. Circ J. 2022;86:642-650.
22. Niehe SR, Vos NS, Van Der Schaaf RJ, et al. 5-Year Clinical Outcomes of Paclitaxel-Coated Balloon Angioplasty vs DES in Acute MI: The REVELATION Trial. JACC Cardiovasc Interv. 2024;17:1185-1186.
23. Nishiyama N, Komatsu T, Kuroyanagi T, et al. Clinical value of drug-coated balloon angioplasty for de novo lesions in patients with coronary artery disease. Int J Cardiol. 2016;222:113-118.
24. Ke D, He X, Chen C, et al. Randomized Pilot Study to Compare DCB-Based versus DST-Based Strategies for the Treatment of True or Complex Coronary Bifurcation Lesions. Rev Cardiovasc Med. 2023;24:99.
25. Kawai T, Watanabe T, Yamada T, et al. Coronary vasomotion after treatment with drug-coated balloons or drug-eluting stents: a prospective, open-label, single-centre randomised trial. EuroIntervention. 2022;18:E140-E148.
26. Gobic´D, Tomulic´V, Lulic´D, et al. Drug-Coated Balloon Versus Drug-Eluting Stent in Primary Percutaneous Coronary Intervention: A Feasibility Study. Am J Med Sci. 2017;354:553-560.
27. Liu S, Zhou Y, Shen Z, et al. Comparison of Drug-Coated Balloon and Drug-Eluting Stent for the Treatment of Small Vessel Disease (from the Dissolve SVD Randomized Trial). Am J Cardiol. 2024;211:29-39.
28. Cortese B, Testa G, Rivero F, Erriquez A, Alfonso F. Long-Term Outcome of Drug-Coated Balloon vs Drug-Eluting Stent for Small Coronary Vessels: PICCOLETO-II 3-Year Follow-Up. JACC Cardiovasc Interv. 2023;16:1054-1061.
29. Yu X, Wang X, Ji F, et al. A Non-inferiority, Randomized Clinical Trial Comparing Paclitaxel-Coated Balloon Versus New-Generation Drug-Eluting Stents on Angiographic Outcomes for Coronary De Novo Lesions. Cardiovasc Drugs Ther. 2022;36:655-664.
30. Niehe SR, Vos NS, Van Der Schaaf RJ, et al. 5-Year Clinical Outcomes of Paclitaxel-Coated Balloon Angioplasty vs DES in Acute MI: The REVELATION Trial. JACC Cardiovasc Interv. 2024;17:1185-1186.
31. Wang Z, Yin Y, Li J, et al. New Ultrasound-Controlled Paclitaxel Releasing Balloon vs Asy mmetric Drug-Eluting Stent in Primary ST-Segment Elevation Myocardial Infarction —A Prospective Randomized Trial —. Circ J. 2022;86:642-650.
32. Korjian S, McCarthy KJ, Larnard EA, et al. Drug-Coated Balloons in the Management of Coronary Artery Disease. Circ Cardiovasc Interv. 2024;17:E013302.
33. Sanz Sánchez J, Chiarito M, Cortese B, et al. Drug-Coated balloons vs drug-eluting stents for the treatment of small coronary artery disease: A meta-analysis of randomized trials. Catheter Cardiovasc Interv. 2021;98:66-75.
34. Dobromir Angheluta A, Levett JY, Zolotarova T, et al. A Meta-Analysis of 3-Year Outcomes of Drug-Coated Balloons Versus Drug-Eluting Stents for Small-Vessel Coronary Artery Disease. JACC Adv.2024;3:101204.
35. Sun B, Zhang XT, Chen RR. Comparison of Efficacy and Safety Between Drug-Coated Balloons Versus Drug-Eluting Stents in the Treatment of De Novo Coronary Lesions in Large Vessels: A Study-Level Meta-Analysis of Randomized Control Trials. Cardiovasc Drugs Ther. 2024;38:1375-1384.
36. Jiang JL, Huang QJ, Chen MH. Efficacy and safety of drug-coated balloon for de novo lesions of large coronary arteries: Systematic review and meta-analysis of randomized controlled trials. Heliyon. 2024;10:e25264.
37. Abdelaziz A, Hafez A, Atta K, et al. Drug-coated balloons versus drug-eluting stents in patients with acute myocardial infarction undergoing percutaneous coronary intervention: an updated meta-analysis with trial sequential analysis. BMC Cardiovasc Disord. 2023;23:605.
38. Caminiti R, Vizzari G, Ielasi A, et al. Drug-coated balloon versus drug-eluting stent for treating de novo large vessel coronary artery disease: a systematic review and meta-analysis of 13 studies involving 2888 patients. Clin Res Cardiol. 2025;114:978-990.
39. Cui K, Lyu S, Song X, et al. Drug-eluting balloon versus bare-mental stent and drug-eluting stent for de novo coronary artery disease: A systematic review and meta-analysis of 14 randomized controlled trials. PLoS One. 2017;12:e0176365.
40. Fezzi S, Giacoppo D, Fahrni G, et al. Individual patient data meta-analysis of paclitaxel-coated balloons vs drug-eluting stents for small-vessel coronary artery disease: the ANDROMEDA study. Eur Heart J. 2025;46:1586-1599.
41. Niu J, Wang K, Wang W, et al. Drug-Coated Balloons Versus Drug-Eluting Stents for the Treatment of De Novo Coronary Artery Disease: A Meta-Analysis of Randomized Controlled Trials. Rev Cardiovasc Med. 2024;25:446.
42. Muramatsu T, Kozuma K, Tanabe K, et al. Clinical expert consensus document on drug-coated balloon for coronary artery disease from the Japanese Association of Cardiovascular Intervention and Therapeutics. Cardiovasc Interv Ther. 2023;38:166-176.
43. Cuculi F, Bossard M, Zasada W, et al. Performing percutaneous coronary interventions with predilatation using non-compliant balloons at high-pressure versus conventional semi-compliant balloons: insights from two randomised studies using optical coherence tomography. Open Heart. 2020;7:e001204.
44. Lamelas P, Belardi J, Whitlock R, Stone GW. Limitations of Repeat Revascularization as an Outcome Measure: JACC Review Topic of the Week. J Am Coll Cardiol. 2019;74:3164-3173.
45. Giacoppo D, Saucedo J, Scheller B. Coronary Drug-Coated Balloons for De Novo and In-Stent Restenosis Indications. J Soc Cardiovasc Angiogr Interv.2023;2:100625.
46. Pasterkamp G, De Kleijn DPV, Borst C. Arterial remodeling in atherosclerosis, restenosis and after alteration of blood flow: potential mechanisms and clinical implications. Cardiovasc Res. 2000;45:843-852.
47. Cortese B, Serruys PW. Single-Antiplatelet Treatment After Coronary Angioplasty With Drug-Coated Balloon. J Am Heart Assoc. 2023;12:e028413.
48. Cortese B, Silva Orrego P, Agostoni P, et al. Effect of Drug-Coated Balloons in Native Coronary Artery Disease Left With a Dissection. JACC Cardiovasc Interv. 2015;8:2003-2009.














