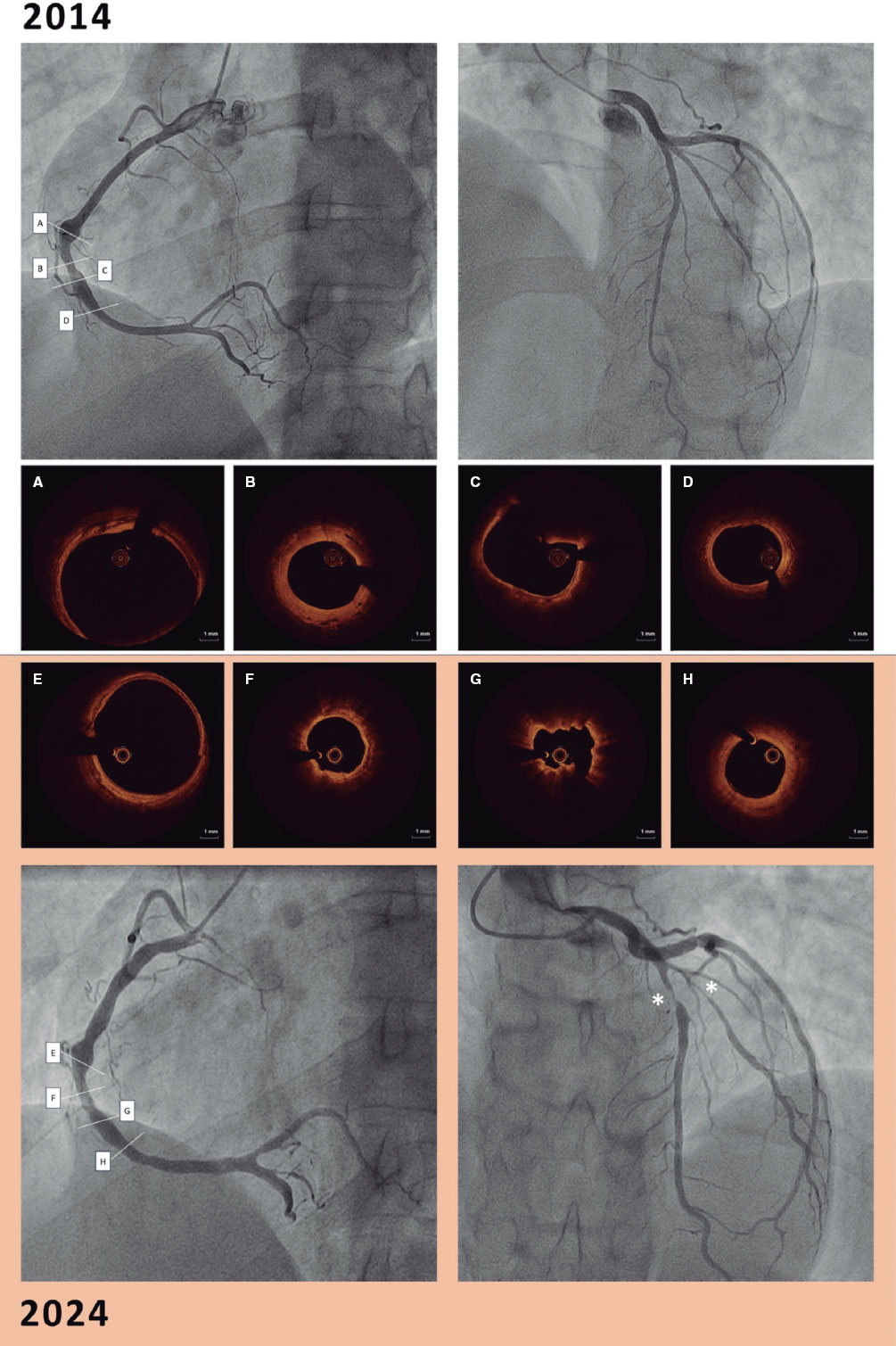
Original article
Optical coherence tomography for the diagnosis and management of stent thrombosis
Tomografía de coherencia óptica en el diagnóstico y el tratamiento de la trombosis del stent
aServicio de Cardiología, Hospital Universitario Central de Asturias, Oviedo, Asturias, Spain
bServicio de Cardiología, Hospital Universitario de La Princesa, Universidad Autónoma de Madrid, IIS-IP, Madrid, Spain cCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain dServicio de Cardiología, Hospital Universitario de Cabueñes, Gijón, Spain
ABSTRACT
Introduction and objectives: The ROLLER COASTR-EPIC22 trial randomized 171 patients with moderate-to-severe calcified coronary lesions to undergo percutaneous coronary intervention with rotational atherectomy, intravascular lithotripsy, or excimer laser. This trial and its 1-year follow-up demonstrated no significant differences among the 3 arms in terms of procedural success, complications and clinical outcomes. This subanalysis aimed to describe age-related outcomes among patients undergoing percutaneous coronary intervention with plaque-modification techniques.
Methods: Of 171 patients (118 < 75 years; 53 ≥ 75 years), we compared final stent expansion by optical coherence tomography using an intention-to-treat analysis. Secondary endpoints included minimum stent area, angiographic, procedural, and clinical success, in-hospital complications, 1-year all-cause mortality, and major adverse cardiovascular events (MACE), including cardiac death, target vessel myocardial infarction, target lesion revascularization, and stent thrombosis.
Results: Baseline characteristics and treatment allocation were similar among groups. Final stent expansion was 85 ± 17.1 % in patients < 75 vs 84 ± 18.9 % in those ≥ 75 (P = .76). Minimum stent area, procedural success, and in-hospital complications were comparable. At 1 year, the ≥ 75 cohort had significantly higher MACE (11.3% vs 1.7%; P = .01) and all-cause mortality (9.4% vs 0.9%; P = .01).
Conclusions: Elderly patients achieved similar procedural outcomes as younger patients, but higher rates of MACE and all-cause mortality at 1-year follow-up.
Keywords: Rotational atherectomy. Intravascular lithotripsy. Excimer laser coronary angioplasty. Coronary calcification. Optical coherence tomography. Elderly patients.
RESUMEN
Introducción y objetivos: En el ensayo ROLLER COASTR-EPIC22 se aleatorizó a 171 pacientes con lesiones coronarias con calcificación moderada a grave para ser tratados con intervención coronaria percutánea mediante aterectomía rotacional, litotricia intravascular o láser de excímeros (ELCA). El ensayo fundamental y su seguimiento a 1 año demostraron que no había diferencias significativas entre los 3 grupos en cuanto a éxito del procedimiento, complicaciones y eventos clínicos. Este subanálisis tuvo como objetivo describir los resultados estratificados por edad en pacientes sometidos a intervención coronaria percutánea con técnicas de modificación de placa.
Métodos: En un total de 171 pacientes (118 < 75 años y 53 ≥ 75 años) se comparó la expansión final del stent mediante tomografía de coherencia óptica utilizando un análisis por intención de tratar. Los objetivos secundarios fueron el área mínima del stent, el éxito angiográfico, de procedimiento y clínico, las complicaciones intrahospitalarias, la mortalidad por cualquier causa al año y los eventos cardiovasculares adversos mayores (MACE), incluida la muerte de causa cardiovascular, el infarto de miocardio del vaso tratado, la revascularización de la lesión tratada y la trombosis del stent.
Resultados: Las características basales y la asignación del tratamiento fueron similares entre los grupos. La expansión final del stent fue del 85 ± 17,1% en los pacientes < 75 años y del 84 ± 18,9% en aquellos ≥ 75 años (p = 0,76). Al año, la cohorte ≥ 75 años presentó una tasa de MACE significativamente más alta (11,3 frente a 1,7%; p = 0,01), así como de mortalidad por todas las causas (9,4 frente a 0,85%; p = 0,01).
Conclusiones: Los pacientes ancianos lograron resultados procedimentales similares a los más jóvenes, pero con tasas más altas de MACE y de mortalidad por cualquier causa en el seguimiento a 1 año.
Palabras clave: Aterectomía rotacional. Litotricia intravascular. Aterectomía coronaria con láser de excímeros. Calcificación coronaria. Tomografía de coherencia óptica. Pacientes ancianos.
Abbreviations
ELCA: excimer laser coronary atherectomy. IVL: intravascular lithotripsy. MACE: major adverse cardiovascular events. OCT: optical coherence tomography. PCI: percutaneous coronary intervention. PMT: plaque modification technique.
INTRODUCTION
Coronary artery calcification constitutes a major challenge during percutaneous coronary intervention (PCI), as it reduces procedural success and increases the risk of complications. It limits optimal stent expansion, which predicts restenosis, stent thrombosis, and the need of target lesion revascularization.1-3 Several plaque modification techniques (PMT) have been developed to improve clinical outcomes in this setting. Rotational atherectomy has long been considered the standard approach for managing resistant calcified lesions. However, the development of newer technologies such as intravascular lithotripsy (IVL), together with increasing interest in excimer laser coronary atherectomy (ELCA), has expanded the range of available therapeutic options.4-7
The ROLLER COASTR-EPIC22 trial was the first randomized trial comparing rotational atherectomy, IVL, and ELCA in patients with moderate-to-severe calcified coronary lesions. Procedural success, safety and 1-year clinical outcomes were similar across all techniques.8
Elderly patients (≥ 75 years), who often present with increased comorbidity and complex coronary anatomy, remain underrepresented in clinical trials and may experience higher procedural risk and higher long-term mortality.9 In this post-hoc analysis of the ROLLER COASTR-EPIC22 trial, we assessed the impact of age on outcomes of PCI for moderately to severely calcified coronary stenosis treated with contemporary plaque-modification techniques. Specifically, we compared stent expansion, procedural success, in-hospital complications and 1-year clinical outcomes between patients < 75 and ≥ 75 years of age. Exploratory analyses of individual PMT within age strata were performed but were not powered for formal between-technique comparisons.
METHODS
Study design and population
ROLLER COASTR-EPIC22 trial was a multicenter randomized trial (NCT04181268) conducted at 8 high-volume centers in Spain.10 Complete inclusion and exclusion criteria, as well as the randomization process and procedural details, have been previously described.11 Briefly, eligible patients had moderate-to-severe calcification estimated by coronary angiography, and culprit lesions of ST-segment elevation acute coronary syndromes were excluded. All patients gave therir prior written informed consent before participation. Included patients were randomized 1:1:1 to rotational atherectomy, IVL, or ELCA from July 2019 through December 2023.10,11
Stratification and endpoints
This study is a post-hoc age-stratified analysis. Patients were stratified by age in 2 groups: < 75 years (n = 118) and ≥ 75 years (n = 53). Comparisons within age subgroups are exploratory and should be viewed as descriptive rather than confirmatory.
The primary endpoint was stent expansion assessed by optical coherence tomography (OCT), calculated as minimal stent area divided by the mean reference lumen area, analyzed according to the intention-to-treat principle.11
The secondary endpoints included angiographic success (final Thrombolysis in Myocardial Infarction grade-3 flow, assessed at both < 20% and < 30% residual stenosis), procedural success (angiographic success without major perioperative complications), clinical success (procedural success without in-hospital major adverse cardiovascular events [MACE]), device success (defined as achievement of adequate plaque modification with the initial device without the need for a second advanced PMT), minimal stent area by OCT, and in-hospital complications.11
Major perioperative complications were defined as death, perforation, flow-limiting dissection, abrupt vessel closure, or stent thrombosis. MACE was defined as the composite of cardiac death, target vessel myocardial infarction, target lesion revascularization, and definite/probable stent thrombosis.11
Procedures and imaging
Procedures have been previously described and followed latest European guideline recommendations.10 Pre- and post-PMT OCT imaging were recommended to characterize the lesion. Predilation with low-profile balloons was permitted to allow OCT catheter crossing prior to PMT. After plaque modification, drug-eluting stent implantation and optimization were performed, followed by the final mandatory OCT. OCT image acquisition and stent optimization protocols have been previously described.11
Rotational atherectomy, IVL and ELCA were performed according to current recommendations and device instructions, as previously described.6,12,13
Statistical analysis
The principal analyses were conducted in the intention-to-treat population. An additional analysis was performed in an as-treated fashion, based on the last PMT used. Continuous variables are expressed as mean ± standard deviation if normally distributed or median [IQR] otherwise, and categorical variables as counts and percentages. Between-group comparisons used the chi-square test or Fisher’s exact test for categorical variables and Student t test or Mann–Whitney U test for continuous variables. Noninferiority of IVL and ELCA vs rotational atherectomy for stent expansion was tested with a 1-sided t test against a prespecified noninferiority margin of −7%. This margin was adopted from the pivotal ROLLER COASTR-EPIC22 trial and was defined a priori by the steering committee as the smallest absolute difference in stent expansion considered clinically relevant, based on previous studies.11,14,15 Statistical significance was set at P < .05.10,11
Because this was an exploratory, post-hoc age-stratified analysis with a relatively small sample size in the elderly subgroup, no formal adjustment for multiple comparisons was applied. Given the large number of comparisons (2 age groups, 3 PMT and several procedural and clinical endpoints), conservative corrections such as Bonferroni would have markedly reduced statistical power and increased the risk of type II error. Instead, all effect estimates, 95% confidence intervals and exact P values are reported to allow readers to interpret the results with appropriate caution, and all subgroup findings are considered hypothesis-generating rather than confirmatory.
Given the low number of events, survival was assessed with Kaplan–Meier curves and log-rank tests. All statistical analyses were conducted using R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Patient allocation and baseline characteristics
A total of 171 patients were enrolled, of whom 118 (69%) were younger than 75 years (mean age, 66.8 ± 6.4 years; 79.5% male) and 53 (31%) were aged ≥ 75 years (mean age, 79.7 ± 3.7 years; 73.6% male). In patients < 75 years (n = 118), age was 66.8 ± 6.4 years (median, 68; IQR 64–71; range, 44–74), whereas in those ≥ 75 years (n = 53) age was 79.7 ± 3.7 years (median, 79; IQR 77–82; range, 75–89) (table S1 and figure S1).
Baseline clinical characteristics were broadly comparable between age groups, including rates of diabetes, hypertension, and prior revascularization. However, older patients had a lower prevalence of active smoking (15.1% vs 33.6%; P = .02), lower body mass index (27.0 ± 3.8 vs 28.5 ± 4.3 kg/m²; P = .04), and lower haemoglobin (12.8 ± 1.8 vs 13.5 ± 2.0 g/L; P = .04). No significant differences were observed in left ventricular ejection fraction, basal creatinine or clinical presentation, although elderly patients showed a trend towards a more frequent presentation as acute coronary syndrome (table 1).
Table 1. Baseline clinical characteristics
| Variable | < 75 years (n = 118) | ≥ 75 years (n = 53) | P |
|---|---|---|---|
| Age | 66.8 ± 6.4 | 79.7 ± 3.7 | < .01 |
| Male | 93 (79.5) | 39 (73.6) | .51 |
| BMI (kg/m2) | 28.5 ± 4.3 | 27.0 ± 3.8 | .04 |
| Current smoker | 39 (33.1) | 8 (15.1) | .02 |
| Diabetes mellitus | 57 (48.3) | 27 (50.9) | 92 |
| Hypertension | 93 (78.8) | 43 (81.1) | .97 |
| Dyslipidemia | 86 (72.9) | 40 (75.5) | .93 |
| Prior PCI | 40 (33.9) | 16 (30.2) | .73 |
| Prior CABG | 4 (3.4) | 2 (3.8) | 1 |
| LVEF | 54.8 ± 10.9 | 54.9 ± 10.7 | .96 |
| Serum creatinine (mg/dL) | 1.2 ± 1.2 | 1.2 ± 1.0 | .95 |
| Hemoglobin (g/L) | 13.5 ± 2.0 | 12.8 ± 1.8 | .04 |
| Clinical presentation | .08 | ||
| CCS | 77 (65.3) | 29 (54.7) | |
| Unstable angina | 12 (10.2) | 10 (18.8) | |
| NSTEMI | 18 (15.2) | 12 (22.6) | |
| STEMI | 8 (6.8) | 0 (0) | |
|
BMI, body mass index; CABG, coronary artery bypass graft; CCS, chronic coronary syndrome; LVEF, left ventricular ejection fraction; NSTEMI, non-ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction. Data are expressed as No. (%) or mean ± standard deviation. All angiographic parameters were measured at a central core laboratory blinded to the treatment arm. |
|||
Angiographic and procedural data
Target vessels and lesion characteristics, including severity and length of calcification, were comparable across age strata and treatment arms. Most procedures were performed via radial access (> 80%), and treatment allocation was well balanced across age strata. Procedural duration, sheath size, and use of ad hoc PCI did not differ by age. Angiographic and procedural details are summarized in table S2.
OCT findings
The optional pre-plaque modification OCT was performed in 39 of 118 patients < 75 years (33.1%) and in 20 of 53 patients ≥ 75 years (37.7%), with no significant difference between age groups (P = .67).
Final OCT runs were obtained in more than 75% of patients. OCT parameters of calcification, assessed by an independent central core laboratory, consistently demonstrated a heavy calcium burden across all cases, with no significant differences between age groups or treatment modalities. OCT findings are presented in table S3.
Primary and secondary endpoints
No significant differences in terms of final stent expansion, minimal stent area, or final residual stenosis were observed between groups (table 2, figure S2). Angiographic, procedural, and clinical success rates exceeded 90% (considering < 30% residual percent diameter stenosis) in both age groups and did not differ by technique (table 2, figure 1).
Table 2. Procedural outcomes and complications
| Procedural outcomes | < 75 years (n = 118) | ≥ 75 years (n = 53) | P |
|---|---|---|---|
| Success considering < 30% residual percent diameter stenosis | |||
| Angiographic | 112 (94.9%) | 49 (92.5%) | .78 |
| Procedural | 108 (91.5%) | 49 (92.5%) | 1.00 |
| Clinical | 108 (91.5%) | 48 (90.6%) | 1.00 |
| Success considering < 20% residual percent diameter stenosis | |||
| Angiographic | 87 (73.7) | 42 (79.2) | .56 |
| Procedural | 83 (70.3) | 42 (79.2) | .30 |
| Clinical | 83 (70.3) | 41 (77.4) | .44 |
| Final TIMI grade flow | .63 | ||
| 0 | 1 (0.8) | 0 | |
| 1 | 0 | 0 | |
| 2 | 1 (0.8) | 0 | |
| 3 | 116 (98.3) | 53 (100) | |
| Final diameter diameter stenosis (%) | 14.7 ± 11.5 | 12.8 ± 10.9 | .30 |
| Final SE (%) | 85.0 ± 17.1 | 84.0 ± 18.9 | .76 |
| Severe procedural complications | 4 (3.4) | 0 (0) | .42 |
| Death | 0 (0) | 0 (0) | – |
| Perforation | 4 (3.4) | 0 (0) | .42 |
| Flow-limiting dissection | 0 (0) | 0 (0) | – |
| Abrupt vessel closure | 0 (0) | 0 (0) | – |
| Stent thrombosis | 0 (0) | 0 (0) | – |
| Dissection | |||
| A | 3 (2.5) | 1 (1-9) | 1.00 |
| B | 0 (0) | 2 (3.8) | .18 |
| D | 1 (0.8) | 1 (1.9) | 1.00 |
| Slow flow/No-reflow | 1 (0.8) | 1 (1.9) | 1.00 |
| Side branch occlusion | 0 (0) | 1 (1.9) | .68 |
| Complications during admission | 0 (0) | 2 (3.8) | .18 |
| Mortality | 0 (0) | 1 (1.9) | .68 |
| Cardiac death | 0 (0) | 1 (1.9) | .68 |
| Target lesion revascularization | 0 (0) | 0 (0) | – |
| Stent thrombosis | 0 (0) | 0 (0) | – |
| Stroke | 0 (0) | 0 (0) | – |
| BARC grade ≥ 3b bleeding | 0 (0) | 1 (1.9) | .68 |
|
BARC, Bleeding Academic Research Consortium; SE, stent expansion; TIMI, Thrombolysis in Myocardial Infarction. Data are expressed as No. (%) or mean ± standard deviation. Severe procedural complications were defined as death, perforation, flow-limiting dissection, abrupt vessel closure or stent thrombosis. |
|||
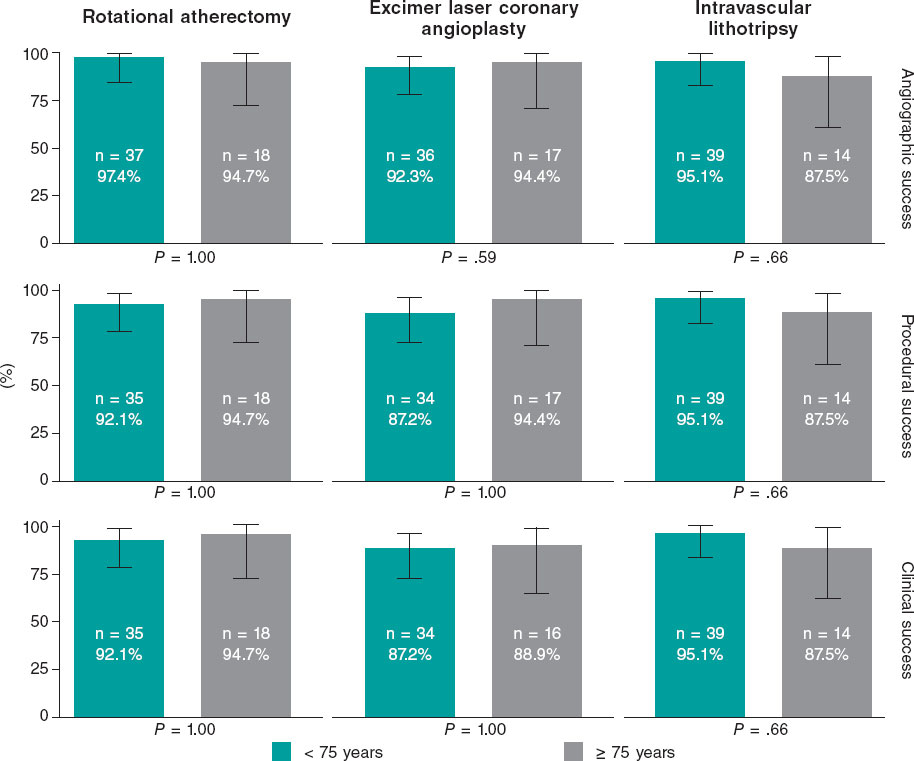
Figure 1. Angiographic, procedural, and clinical success of each technique. Angiographic success defined as final Thrombolysis in Myocardial Infarction grade-3 flow with < 30% residual percent diameter stenosis. Procedural success was considered angiographic success without major perioperative complications. Clinical success was defined as procedural success without in-hospital major adverse cardiovascular events.
Among patients ≥ 75 years, mean stent expansion values were broadly similar across the 3 PMT, and no statistically significant between-technique differences were detected; however, 95%CI, were wide because of the small sample size in each subgroup (table S3). Exploratory analyses of mean differences in stent expansion between techniques yielded results that were directionally consistent with the overall ROLLER COASTR-EPIC22 trial but were clearly underpowered in this age-stratified cohort and should be interpreted as hypothesis-generating only (figure 2 and table S4).
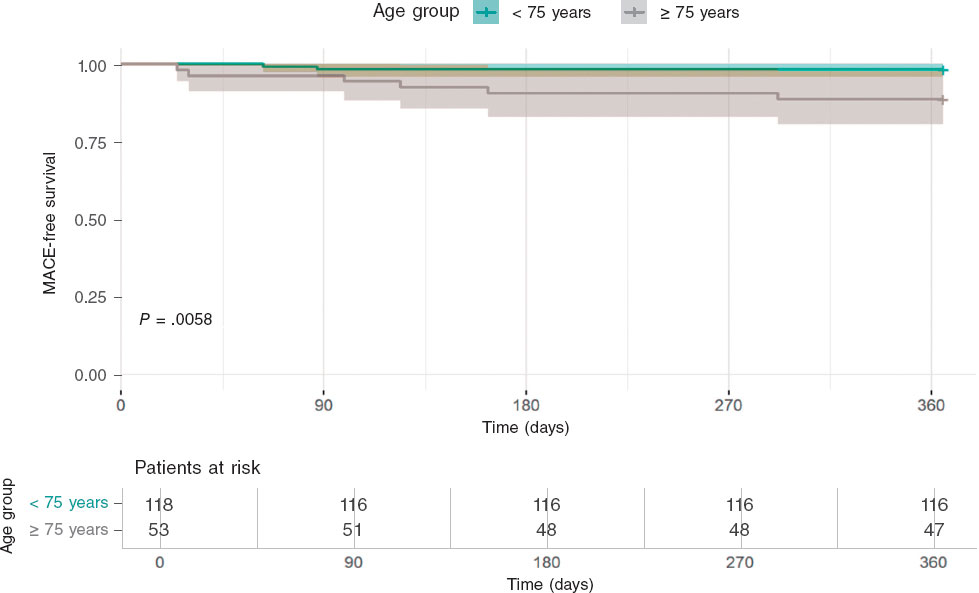
Figure 2. One-year MACE-free survival. Kaplan-Meier curves showing 1-year freedom from MACE in the intention to-treat population. MACE was defined as the composide endpoing of cardiac death, target vessel myocardial infarction, target lesion revascularization, stent thrombosis. MACE, major adverse cardiovascular events.
Overall, device success rate was 88.3% overall and was similar by age: < 75 (89.7%) vs ≥ 75 years (85.7%); P = .76. The causes of crossover differed by device: as in the pivotal trial, in the rotational atherectomy arm most crossovers were prompted by inadequate lesion dilation after atherectomy, whereas in the IVL and ELCA arms the primary driver was inability to cross the lesion with the dedicated balloon or laser catheter (figure S3 and table S5). In-hospital complication rates (perforations, bleeding, death) were low and balanced by age (table 2).
In 1-year follow-up (table 3, figure 2), patients aged ≥ 75 years had a markedly higher rate of MACE than those < 75 years (11.3% vs 1.7%; P = .01). All-cause mortality was also greater in the older cohort (9.4% vs 0.9%; P = .01).
Table 3. One-year event rates
| Event | < 75 years | ≥ 75 years | P |
|---|---|---|---|
| MACE | 2 (1.7%) | 6 (11.3%) | .01 |
| All-cause mortality | 1 (0.9%) | 5 (9.4%) | .01 |
| Cardiac death | 0 (0.00%) | 2 (3.8%) | .10 |
| TVMI | 0 (0.00%) | 2 (3.8%) | .10 |
| TLR | 1 (0.9%) | 2 (3.8%) | .23 |
| TVR | 2 (1.7%) | 4 (7.6%) | .08 |
| ST | 0 (0.00%) | 2 (3.8%) | .10 |
|
MACE, major adverse cardiovascular events; ST, stent thrombosis; TLR, target lesion revascularization; TVMI, target vessel myocardial infarction; TVR, target vessel revascularization. Data are expressed as No. (%). MACE was defined as a composite endpoint of cardiac death, target vessel acute myocardial infarction, target lesion revascularization, and stent thrombosis. P values have been calculated as log-rank p-values from Kaplan-Meier comparisons. |
|||
Although less frequent, each individual component of the composite endpoint was numerically higher in patients ≥ 75 years without reaching statistical significance: cardiac death (11.3% vs 0%; P = .1), target vessel myocardial infarction (3.8% vs 0%; P = .1), target lesion revascularization (3.8% vs 0.8%; P = .23), target vessel revascularization (7.5% vs 1.7%; P = .08) and stent thrombosis (3.8% vs 0%; P = .10).
In a multivariable logistic regression including age ≥ 75 years, anemia, acute coronary syndrome presentation and renal dysfunction, age ≥ 75 years remained the strongest predictor of 1-year endpoints. For MACE, age ≥ 75 years was associated with an adjusted OR, 7.4 (95%CI, 1.5–53.9; P = .02), whereas none of the other covariates showed a clear independent association with the composite endpoint. For all-cause mortality, the adjusted OR for age ≥ 75 years was 10.7 (95%CI, 1.5–218.7; P = .04) and none of the other covariates demonstrated a clear independent effect. These findings are presented in table S6 and figure S4.
DISCUSSION
In this post-hoc age-stratified analysis of the ROLLER COASTR-EPIC22 trial and its 1-year follow-up, we observed that the overall safety and efficacy of PMT appeared consistent across age groups. However, at 1 year older patients experienced significantly higher rates of MACE (11.3% vs 1.7%; P = .01) and all-cause mortality (9.4% vs 0.9%; P = .01) despite comparable in-hospital complication rates.
After adjustment for anemia, acute coronary syndrome presentation, and renal dysfunction, age ≥ 75 years remained associated with higher adjusted odds of both 1-year MACE and all-cause mortality. None of the adjustment covariates demonstrated a clear independent effect, suggesting that unmeasured factors closely related to aging, such as frailty, are likely to account for much of the excess risk observed in older patients.
Randomized evidence directly comparing contemporary PMT in calcified coronary disease remains scarce. The ROLLER COASTR-EPIC22 trial was the first trial to randomize patients among rotational atherectomy, IVL, and ELCA.10 Previous device-specific studies and registries have demonstrated the feasibility of each technique, but none had evaluated all 3 within a single randomized framework.16-18
Coronary calcium is independently associated with long-term mortality risk, and age is not only a key predictor of coronary calcification but also an independent predictor of MACE, regardless of calcium burden.19 Nevertheless, elderly patients are underrepresented in PMT studies, leaving limited evidence to guide the choice of the optimal PMT strategy. Regarding rotational atherectomy, there is little evidence in elderly patients, but retrospective data suggests similar success and in-hospital complication rates compared with younger individuals.20 Similarly, the BENELUX-IVL prospective registry demonstrated the safety profile and technical success of IVL in patients aged ≥ 80 years.21 ELCA, however, has not been specifically studied in the elderly population.
Selecting elderly patients for revascularization requires balancing benefits and risks. Comorbidities (anemia, chronic kidney disease, acute coronary syndrome) and limitations in physiological reserve increase complexity and may reduce revascularization uptake in this population.22,23 Our findings support that chronological age alone should not preclude revascularization, as PMT appeared effective and safe when performed by experienced operators, regardless of age. These results, however, should be interpreted cautiously given the limited sample size and exploratory, post-hoc nature of the age-stratified analysis. The importance of operator experience in PMT for complex scenarios has been highlighted by the CRATER trial program, which evaluated rotational atherectomy in patients with chronic kidney disease and severely calcified lesions and reported high procedural success but also substantial perioperative and long-term event rates, largely reflecting the extreme baseline risk of this population.24,25
Notably, our analysis suggests no excess perioperative complications in older adults, although the study was not powered for subgroup comparisons and 95%CI, were wide, particularly in the elderly ELCA subgroup. We advocate for meticulous patient selection, based on preoperative risks and comorbidities; however, PMT should be used when necessary for adequate lesion preparation to maximize stent expansion and avoid suboptimal implantation, a known driver of adverse clinical events.
In our cohort, elderly patients achieved similar procedural success, stent expansion and in-hospital safety outcomes vs younger patients yet experienced a markedly higher rate of 1-year MACE and all- cause mortality. This pattern is consistent with the e-ULTIMASTER and MOSCA FRAIL trials, in which frailty, rather than age per se, emerged as the main determinant of adverse outcomes in older adults undergoing PCI.26,27 Taken together, these data support that revascularization decisions in elderly patients should not rely exclusively on chronological age, but should incorporate assessment of frailty, functional capacity and comorbidity to individualize the benefit–risk balance of an invasive strategy. The lack of formal frailty indices in our study therefore represents an important limitation. Future trials in elderly patients undergoing complex PCI should prospectively integrate standardized frailty tools to better guide patient selection and tailor treatment.
In the subset of patients with available baseline OCT, we did not observe relevant differences in calcium burden or morphology nor in angiographic severity or extent of disease, between patients < 75 and ≥ 75 years. This suggests that the underlying calcific process is biologically similar across age groups, even if it becomes clinically manifest at different ages, and is consistent with the comparable postoperative stent expansion and minimal stent area observed in both strata. Nevertheless, despite similar stent optimization, the ≥ 75-year group showed a numerically higher rate of definite/probable stent thrombosis and cardiovascular death at 1 year (3.8%), which is clinically meaningful in this context. As underexpansion alone is unlikely to fully explain these events, age-related factors such as greater comorbidity and frailty, a prothrombotic milieu and a reduced myocardial reserve, may have contributed to the excess risk observed in older patients, even after apparently adequate angiographic and OCT-guided results.
Contemporary randomized PMT trials (such as ROTAXUS, PREPARE-CALC, ISAR-CALC 2 or ECLIPSE ) enrolled patients with a mean age around 70 years, but none of them reported age-specific outcomes for those ≥ 75 years, limiting its applicability to older populations.28-32 Our analysis contributes to fill this knowledge gap suggesting procedural success and safety of PMT in the elderly. However, due to the nonprespecified nature of our analysis, these results should be considered hypothesis-generating. Future randomized studies should integrate objective frailty and functional measures and include sufficient follow-up to assess both procedural and long-term outcomes in this growing population.
Study limitations
This study has several limitations to consider. First, it was a post-hoc analysis and the findings should be regarded as hypothesis- generating rather than definitive. Second, because age stratification was not prespecified in the trial design, the study was not statistically powered for subgroup comparisons based on age, and the relatively small number of patients aged ≥ 75 years may limit the generalizability of the results.
Third, frailty status and functional capacity –both known to strongly influence outcomes in elderly patients– were not assessed, which limits the clinical granularity of the findings.
Fourth, the study was not powered to detect differences in clinical events and results should be interpreted cautiously due to the limited number of events.
Fifth, posttreatment OCT images were not available in slightly more than 20% of patients, with a higher proportion of missing studies in the ≥ 75-year group. This pattern of missing data may introduce selection bias, as patients without analyzable postoperative OCT could differ systematically from those with complete imaging, potentially limiting the internal validity and generalizability of the stent expansion findings, particularly in the elderly subgroup.
Sixth, randomization was based on angiographic calcification alone, without prior evaluation of lesion crossability or balloon dilatability, which could have impacted the need for secondary PMT. Indeed, some patients required a second PMT, complicating the interpretation of the intention-to-treat analysis, although complementary as-treated and per-protocol analyses were performed to mitigate this limitation. Additionally, not all patients underwent complete pre- and post-PCI OCT, which may have influenced both device selection and the assessment of stent expansion metrics.
Seventh, the 1-year follow-up provides only mid-term outcome data and does not address long-term endpoints such as stent durability or the need for target lesion revascularization. Lastly, the trial was conducted in high-volume centres by experienced operators, which may limit the applicability of these results to lower-volume settings or institutions with less experience in complex PCI and PMT.
CONCLUSIONS
In this age-stratified analysis of the ROLLER COASTR-EPIC22 trial, elderly patients undergoing PCI for calcified coronary lesions achieved similar procedural success, stent expansion, and in-hospital safety outcomes as younger patients. However, patients aged ≥ 75 years experienced significantly higher rates of MACE and all-cause mortality at 1 year (figure 3).
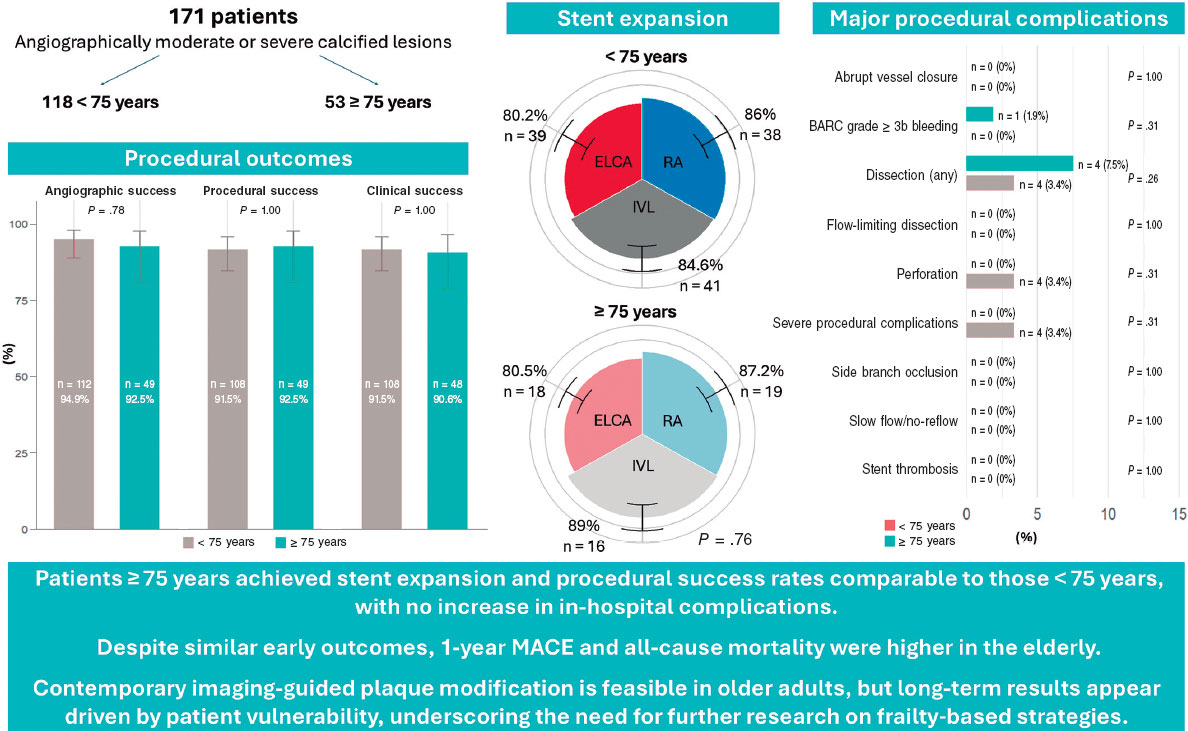
Figure 3. Central illustration. Senior-ROLLER: age-related outcomes of plaque modification in coronary stenosis. Procedural success, stent expansion, and major procedural complications were similar in patients < 75 and those ≥ 75 years; however, the 1-year MACE and all-cause mortality rates were higher in the elderly, which highlights the need for careful patient selection and individualized treatment strategies. BARC, Bleeding Academic Research Consortium; ELCA, excimer laser coronary atherectomy; IVL, intravascular lithotripsy; MACE, major adverse cardiovascular events; RA, rotational atherectomy.
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
FUNDING
The ROLLER COASTR-EPIC22 trial was supported by an unrestricted grant from Fundación EPIC.
ETHICAL CONSIDERATIONS
This is a subanalysis of the ROLLER COASTR-EPIC22 trial (NCT04181268), which was approved by the institutional ethics committee at each participant center and conducted in full compliance with the principles outlined in the Declaration of Helsinki. The authors confirm that written informed consent was obtained from all participants before enrolment.
Sex and gender aspects were considered according to the Sex and Gender Equity in Research (SAGER) guidelines.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Artificial intelligence was not used in this manuscript.
AUTHORS’ CONTRIBUTIONS
J. Vila-García and M. Basile contributed equally to the manuscript and share first authorship. J. Vila-García and M. Basile contributed equally to several phases of the study, including conception and design, and acquisition, analysis and interpretation of the data. In addition, J. Vila-García contributed to the drafting of the original manuscript and to the writing and revision of the final version, while M. Basile was primarily responsible for the critical review of the article. A. Gómez-Menchero, J. Caballero-Borrego, B. Rivero- Santana, G. Galeote, I.J. Amat-Santos, S. Jiménez-Valero, G. Miñana, S. Ojeda, A. Gonzálvez-García, D. Tébar-Márquez, S. Camacho- Freire, J. Zubiaur, R. Ocaranza-Sánchez and A. Domínguez participated in the acquisition of the data and in the review and editing of the final text. A. Jurado-Román and R. Moreno participated in the conception and design of the study, in the acquisition, analysis and interpretation of the data, in the drafting, review, editing and approval of the final manuscript, and in the overall supervision of the project. A. Jurado-Román was responsible for funding and administration. All authors approved the final version of the manuscript.
CONFLICTS OF INTEREST
S. Ojeda and R. Moreno are associate editors of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. A. Jurado-Román is a proctor for Abbott, Boston Scientific, World Medica, and Philips; has received consulting fees from Boston Scientific and Philips; and has received speaker fees from Abbott, Boston Scientific, Shockwave Medical, Philips, and World Medica. S. Ojeda has received consulting fees from Medtronic and Edwards Lifesciences; and has received speaker fees from Abbott, Boston Scientific, Philips, and World Medical. All other authors report no relationships relevant to the contents of this article to disclose.
ACKNOWLEDGMENTS
We thank the ROLLER COASTR-EPIC22 trial investigators, coordinators, and participating patients for their dedication.
WHAT IS KNOWN ABOUT THE TOPIC?
- Contemporary PMT (rotational atherectomy, IVL, and ELCA) have demonstrated efficacy in managing heavily calcified coronary lesions across all age groups. However, elderly patients remain significantly underrepresented in randomized controlled trials of interventional cardiology, limiting the evidence base for clinical decision-making in this growing population. The ROLLER COASTR-EPIC22 Trial trial was the first randomized study to directly compare all 3 main PMT in a single cohort, establishing their procedural safety and efficacy profile in younger patients. Prior data suggest that age alone may not be an absolute contraindication to contemporary interventional strategies; however, age-stratified outcomes for elderly patients specifically remain sparse in the literature.
WHAT DOES THIS STUDY ADD?
- This analysis provides the first randomized age-stratified evaluation of PMT. Our findings demonstrate that procedural success and stent expansion are comparable between elderly and younger cohorts, with no excess in-hospital complications in older adults. Despite similar early procedural outcomes, elderly patients experienced significantly higher rates of MACE and all-cause mortality at 1-year follow-up, highlighting the importance of patient vulnerability and frailty over chronological age alone. These results suggest that age should not be an exclusion criterion for contemporary PMT when clinically indicated, but rather that individualized risk assessment and targeted follow-up strategies are essential for optimizing outcomes in elderly populations undergoing complex percutaneous coronary intervention.
REFERENCES
1. Généreux P, Madhavan MV, Mintz GS, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes: Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trials. J Am Coll Cardiol. 2014;63:1845-1854.
2. Ma W, Wang Q, Wang B, et al. Novel predictors of stent under-expansion regarding calcified coronary lesions assessed by optical coherence tomography. Catheter Cardiovasc Interv. 2022;99(S1):1473-1481.
3. Ng P, Maehara A, Kirtane AJ, et al. Management of Coronary Stent Underexpansion. J Am Coll Cardiol. 2025;85:625-644.
4. Shah M, Najam O, Bhindi R, Silva KD. Calcium Modification Techniques in Complex Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2021;14:E009870.
5. Barbato E, Gallinoro E, Abdel-Wahab M, et al. Management strategies for heavily calcified coronary stenoses: an EAPCI clinical consensus statement in collaboration with the EURO4C-PCR group. Eur Heart J. 2023;44:4340-4356.
6. Barbato E, CarriéD, Dardas P, et al. European expert consensus on rotational atherectomy. EuroIntervention. 2015;11:30-36.
7. Cobarro L, Jurado-Román A, Tébar-Márquez D, et al. Excimer laser coronary atherectomy in severely calcified lesions: time to bust the myth. REC Interv Cardiol. 2023;6:33-40.
8. Basile M, Gómez-Menchero A, Rivero-Santana B, et al. Rotational Atherectomy, Lithotripsy, or Laser for Calcified Coronary Stenosis: One-Year Outcomes From the ROLLER COASTER-EPIC22 Trial. Catheter Cardiovasc Interv. 2025;106:702-710.
9. Kuna C, Bradaric C, Koch T, et al. Age-related ten-year outcomes after percutaneous coronary intervention of in-stent restenosis. Int J Cardiol. 2025;428:133109.
10. Jurado-Román A, Gómez-Menchero A, Rivero-Santana B, et al. Rotational Atherectomy, Lithotripsy, or Laser for Calcified Coronary Stenosis: The ROLLER COASTR-EPIC22 Trial. JACC Cardiovasc Interv. 2025;18:606-618.
11. Jurado-Román A, Gómez-Menchero A, Amat-Santos IJ, et al. Design of the ROLLERCOASTR trial: rotational atherectomy, lithotripsy or laser for the management of calcified coronary stenosis. REC Interv Cardiol. 2023;5:279-286.
12. Rawlins J, Din JN, Talwar S, O'Kane P. Coronary Intervention with the Excimer Laser: Review of the Technology and Outcome Data. Interv Cardiol Rev. 2016;11:27.
13. Riley RF, Patel MP, Abbott JD, et al. SCAI Expert Consensus Statement on the Management of Calcified Coronary Lesions. J Soc Cardiovasc Angiogr Interv. 2024;3:101259.
14. de Waha S, Allali A, Büttner HJ, et al. Rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: Two-year clinical outcome of the randomized ROTAXUS trial. Catheter Cardiovasc Interv. 2016;87:691-700.
15. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of Stent Expansion Guided by Optical Coherence Tomography Vs Intravascular Ultrasound: The ILUMIEN II Study (Observational Study of Optical Coherence Tomography [OCT] in Patients Undergoing Fractional Flow Reserve [FFR] and Percutaneous Coronary Intervention). JACC Cardiovasc Interv. 2015;8:1704-1714.
16. Jurado-Román A, Gonzálvez A, Galeote G, Jiménez-Valero S, Moreno R. RotaTripsy: Combination of Rotational Atherectomy and Intravascular Lithotripsy for the Treatment of Severely Calcified Lesions. JACC Cardiovasc Interv. 2019;12:e127-e129.
17. Hill JM, Kereiakes DJ, Shlofmitz RA, et al. Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Artery Disease. J Am Coll Cardiol. 2020;76:2635-2646.
18. Shibui T, Tsuchiyama T, Masuda S, Nagamine S. Excimer laser coronary atherectomy prior to paclitaxel-coated balloon angioplasty for de novo coronary artery lesions. Lasers Med Sci. 2021;36:111-117.
19. McClelland RL, Chung H, Detrano R, Post W, Kronmal RA. Distribution of coronary artery calcium by race, gender, and age: Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2006;113:30-37.
20. Sharma V, Abdul F, Haider ST, et al. Rotablation in the Very Elderly –Safer than We Think?Cardiovasc Revasc Med. 2021;22:36-41.
21. Oliveri F, García PV, Oort MJH van, et al. Intravascular lithotripsy for the treatment of calcified coronary lesions in individuals of advanced age: a post-hoc analysis of the multicentre, prospective BENELUX-IVL study. eClinicalMedicine. 2025;85:103342.
22. Tisminetzky M, Miozzo R, Gore JM, et al. Trends in the magnitude of chronic conditions in patients hospitalized with a first acute myocardial infarction. J Multimorb Comorbidity. 2021;11:2633556521999570.
23. Nanna MG, Sutton NR, Kochar A, et al. Assessment and Management of Older Adults Undergoing PCI, Part 1: A JACC: Advances Expert Panel. JACC Adv. 2023;2:100389.
24. Galeote G, Zubiaur J, Jurado?Román A, et al. Coronary Rotational Atherectomy Elective Vs Bailout in Patients With Severely Calcified Lesions and Chronic Renal Failure (CRATER) Trial. Catheter Cardiovasc Interv. 2025;106:1702-1712.
25. Zubiaur J, Galeote G, Jurado-Román A, et al. Coronary Rotational ATherectomy Elective Vs Bailout in Patients With Severely Calcified Lesions and Chronic Renal Failure: Long-Term Outcomes of CRATER Trial. Catheter Cardiovasc Interv. 2026;107:431-438.
26. Saada M, Kobo O, Kauer F, et al. Prognosis of PCI in the Older Adult Population: Outcomes From the Multicenter Prospective e-ULTIMASTER Registry. J Soc Cardiovasc Angiogr Interv. 2022;1:100442.
27. Sanchis J, Bueno H, Miñana G, et al. Effect of Routine Invasive vs Conservative Strategy in Older Adults With Frailty and Non–ST-Segment Elevation Acute Myocardial Infarction: A Randomized Clinical Trial. JAMA Intern Med. 2023;183:407-415.
28. Abdel-Wahab M, Richardt G, Büttner HJ, et al. High-speed rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: The randomized ROTAXUS (Rotational Atherectomy Prior to Taxus Stent Treatment for Complex Native Coronary Artery Disease) trial. JACC Cardiovasc Interv. 2013;6:10-19.
29. Mankerious N, Richardt G, Allali A, et al. Lower revascularization rates after high-speed rotational atherectomy compared to modified balloons in calcified coronary lesions: 5-year outcomes of the randomized PREPARE-CALC trial. Clin Res Cardiol. 2024;113:1051-1059.
30. Rheude T, Rai H, Richardt G, et al. Super high-pressure balloon vs scoring balloon to prepare severely calcified coronary lesions: the ISAR-CALC randomised trial: Balloon-based techniques for calcific CAD. EuroIntervention. 2021;17:481.
31. Abdel-Wahab M, Toelg R, Byrne RA, et al. High-speed rotational atherectomy vs modified balloons prior to drug-eluting stent implantation in severely calcified coronary lesions: The randomized prepare-CALC trial. Circ Cardiovasc Interv. 2018;11:e007415.
32. Kirtane AJ, Généreux P, Lewis B, et al. Orbital atherectomy vs balloon angioplasty before drug-eluting stent implantation in severely calcified lesions eligible for both treatment strategies (ECLIPSE): a multicentre, open-label, randomised trial. Lancet. 2025;405:1240-1251.
ABSTRACT
Background and objectives: Percutaneous coronary intervention for chronic total occlusion (CTO PCI) is a prolonged and technically demanding procedure often associated with patient anxiety and discomfort. We evaluated whether intraoperative audiovisual distraction via virtual reality (VR) goggles reduces procedural anxiety vs usual care.
Methods: The ReViCTO trial was a prospective, single-center, randomized, open-label study enrolling 59 patients undergoing elective CTO PCI. Participants were randomized to receive intraoperative audiovisual distraction via VR goggles (n = 31) or usual care (n = 28). The primary endpoint was maximum patient-reported procedural anxiety assessed immediately after theprocedure using a visual analogue scale (VAS; range, 0-10). Secondary endpoints included procedural pain, intraoperative sedative/analgesic requirements, and patient satisfaction.
Results: Baseline clinical characteristics were similar between groups. There was no significant difference in the primary endpoint of maximum procedural anxiety between the VR and control groups (mean VAS, 3.23 ± 2.78 vs 3.75 ± 2.77; mean difference, –0.52; P = .472). Similarly, no significant differences were observed regarding maximum procedural pain (P = .964) or the use and dosage of intraoperative morphine or midazolam. The intervention was safe, and 80.6% of patients in the VR group reported willingness to use the device during a future procedure.
Conclusions: Among patients undergoing elective CTO PCI, the use of immersive VR was feasible and well-tolerated but did not significantly reduce patient-reported peak anxiety, pain, or intraoperative pharmacologic requirements vs contemporary usual care.
[ClinicalTrials.gov: NCT05458999].
Keywords: Virtual reality. Percutaneous coronary intervention. Chronic total occlusion. Anxiety. Pain.
RESUMEN
Introducción y objetivos: La intervención coronaria percutánea (ICP) para las oclusiones coronarias totales crónicas (OTC) es un procedimiento prolongado y técnicamente exigente, a menudo asociado a ansiedad y malestar del paciente. Se evalúa si la distracción audiovisual durante el procedimiento mediante gafas de realidad virtual (RV) reduce la ansiedad procedimental en comparación con la atención habitual.
Métodos: El ensayo ReViCTO es un estudio prospectivo, unicéntrico, aleatorizado y abierto que incluyó 59 pacientes sometidos a ICP electiva de OTC. Los participantes se asignaron al azar para recibir distracción audiovisual durante el procedimiento con gafas de RV (n = 31) o atención habitual (n = 28). El objetivo principal fue la ansiedad máxima percibida por el paciente durante el procedimiento, evaluada inmediatamente después con una escala visual analógica (EVA; rango 0-10). Los objetivos secundarios fueron el dolor procedimental, las necesidades de sedación o analgesia durante el procedimiento, y la satisfacción del paciente.
Resultados: Las características clínicas basales fueron comparables entre los grupos. No hubo diferencias significativas en el objetivo principal de ansiedad máxima entre el grupo de RV y el grupo control (EVA media 3,23 ± 2,78 frente a 3,75 ± 2,77; diferencia media −0,52; p = 0,472). Tampoco se encontraron diferencias significativas en el dolor máximo (p = 0,964) ni en el uso y la dosis de morfina o midazolam durante el procedimiento. La intervención fue segura y el 80,6% de los pacientes del grupo de RV manifestaron que estarían dispuestos a utilizar el dispositivo en una intervención futura.
Conclusiones: En los pacientes sometidos a ICP electiva de OTC, el uso de RV inmersiva, en comparación con la atención habitual contemporánea, fue factible y bien tolerado, pero no redujo de manera significativa la ansiedad máxima ni el dolor percibidos por el paciente, y tampoco los requerimientos farmacológicos durante el procedimiento.
[ClinicalTrials.gov: NCT05458999].
Palabras clave: Realidad virtual. Intervención coronaria percutánea. Oclusión total crónica. Ansiedad. Dolor.
Abbreviations
CTO: chronic total occlusion. PCI: percutaneous coronary intervention. VASa: visual analogue scale for anxiety. VASp: visual analogue scale for pain. VR: virtual reality.
INTRODUCTION
Chronic total occlusions (CTO) are common among patients undergoing coronary angiography and represent one of the most technically demanding scenarios for percutaneous coronary intervention (PCI). Although contemporary CTO PCI programs achieve high success rates,1 these procedures frequently require prolonged fluoroscopy time, dual arterial access,2,3 and sustained patient immobility. In addition, ischemia-related chest discomfort may occur during complex procedural strategies.4,5
Anxiety is common in patients undergoing coronary procedures in the cath lab and may contribute to procedural discomfort and the need for pharmacologic sedation or analgesia.6-8 In routine practice, premedication and intraoperative administration of benzodiazepines or opioids are often used to mitigate anxiety and pain; however, their benefits are modest, and pharmacologic strategies and their use varies across centers.9 CTO PCI may be particularly associate with anxiety due to its typical duration, access strategy, and potential for procedural chest pain.
Virtual reality (VR) is an immersive audiovisual distraction strategy that can reduce procedural pain and anxiety across clinical settings. A systematic review demonstrated that VR-based distraction is effective for pain reduction in multiple procedural contexts.10
In interventional cardiology, early evidence suggests feasibility and potential benefit of VR during procedures performed under conscious sedation, including transcatheter aortic valve implantation and atrial fibrillation ablation.11,12 However, there are no randomized data evaluating VR during CTO PCI, a setting in which nonpharmacologic anxiolysis could be particularly valuable.
The ReViCTO trial was designed to test whether VR use during elective CTO PCI reduces the maximum level of patient-reported procedural anxiety vs usual care. Secondary endpoints included procedural pain, the use and dose of intraoperative anxiolytic or analgesic drugs, and patient satisfaction with the VR intervention. Figure 1 summarizes the main findings.
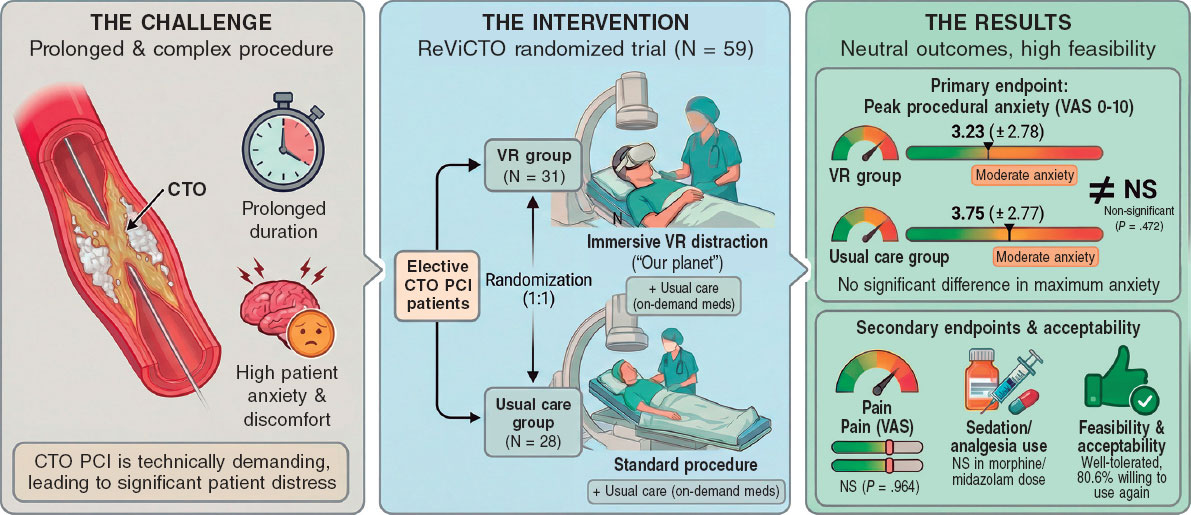
Figure 1. Central illustration. ReViCTO trial overview and main findings. Elective CTO PCI is a prolonged, technically complex procedure frequently associated with patient anxiety and discomfort. In the ReViCTO randomized trial (N = 59), patients were assigned to immersive audiovisual distraction using VR goggles (n = 31) or usual care (n = 28). VR use was feasible and well tolerated but did not significantly reduce peak procedural anxiety (VAS 0–10) or pain or intraoperative sedative or analgesic requirements vs usual care; 80.6% of VR patients reported willingness to use VR again in a future intervention. CTO, chronic total occlusion; PCI, percutaneous coronary intervention; VAS, visual analogue scale; VR, virtual reality.
METHODS
Trial design and oversight
The ReViCTO trial is an investigator-initiated, single-center, randomized, controlled, open-label, superiority trial with 2 parallel groups comparing immersive VR goggles vs usual care during elective CTO PCI. The study received no external funding. The rationale and full trial design have been published previously.13
The trial was conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Good Clinical Practice guidelines. The protocol was approved by the Clinical Research Ethics Committee of Hospital Clínico Universitario de València on 28 February, 2022, and all patients gave their prior written informed consent. The trial was registered at ClinicalTrials.gov (NCT05458999).
Participants
Adults (age > 18 years) scheduled for elective CTO PCI at Hospital Clínico Universitario de València were screened for eligibility. Key exclusion criteria were any condition precluding VR use, such as significant visual impairment, dementia, language barrier (inability to communicate fluently in Spanish or English), or other circumstances preventing safe use of the headset. Eligibility criteria were prespecified in the protocol publication.13
Randomization and trial procedures
Patients were randomized in a 1:1 ratio to the VR group or the control group using computer-generated permuted blocks to reduce the risk of imbalance in group sizes in a small trial.14 Allocation concealment was implemented via a web-based application that generated a unique trial identification number and assigned treatment arm after enrollment, preventing post-randomization modification or deletion. Due to the nature of the intervention, no blinding was applied to participants, operators, or outcome assessors.
CTO PCI was performed according to contemporary clinical practice by an experienced CTO team. Pharmacologic management of anxiety and pain was not protocolized and was left to the discretion of the primary operator in both groups. Specifically, morphine chloride and/or midazolam could be administered on demand during the procedure according to observed or reported anxiety or pain, in accordance with the trial protocol.
Interventions
Virtual reality group
A commercially available head-mounted display (Oculus Quest 2, Meta Platforms, United States) was used. Participants viewed the documentary series Our Planet via a video-streaming application (Netflix) in a virtual “theater” environment, starting with episode 1 with sequential autoplay thereafter, as specified in the protocol. The headset was applied before arterial puncture and was maintained throughout the intervention unless the patient requested removal or a serious complication occurred. Patient status was monitored at regular intervals during the procedure.
Control group
Participants assigned to control underwent CTO PCI under usual care without VR goggles.
Outcomes
The prespecified primary endpoint was the maximum level of anxiety perceived by the patient during the procedure, assessed immediately after the procedure and before leaving the cath lab using a visual analogue scale for anxiety (VASa). Secondary endpoints included maximum patient-perceived pain during the procedure (VASp), the use and total dose of intraoperative anxiolytic or analgesic drugs (midazolam and morphine chloride), procedure-related nausea or dizziness, and patient satisfaction with the VR intervention, including willingness to use VR again. Endpoint definitions and timing of assessment followed the protocol publication.
For analysis, VAS scores were treated as numeric ratings ranging from 0 (none) to 10 (worst), consistent with the trial questionnaires and the observed data range. Baseline angina-related health status was measured using the Seattle Angina Questionnaire (SAQ) to contextualize symptom burden.15
Data collection and management
Demographic characteristics, medical history, comorbidities, baseline symptom status (including SAQ), and procedural characteristics (arterial access strategy, procedure duration, fluoroscopy time, and radiation dose metrics) were collected from institutional electronic health records and procedure reports, supplemented by direct patient interview when necessary. Immediately after completion of CTO PCI, a trained study nurse administered the postoperative questionnaire and recorded VAS anxiety and pain, as well as nausea, dizziness, and satisfaction items. Intraoperative administration of morphine and midazolam and their total doses were recorded contemporaneously by the study nurse.
Data were entered into a dedicated electronic database incorporating range checks for numeric variables and duplicate checks for hospital identifiers and stored on a restricted-access workstation as described in the protocol.
Sample size estimation
The target sample size (58 patients, 29 per group) was calculated based on the primary endpoint, assuming a common standard deviation of 2.7 points for VAS anxiety and aiming to detect an absolute between-group difference of at least 2 points with a 2-sided alpha of 0.05 and 80% power. The standard deviation assumption was informed by prior cath lab anxiety studies.9,16
Statistical analysis
Continuous variables are expressed as mean (standard deviation, SD) or median (interquartile range, IQR) as appropriate (for the primary endpoint and Japan Chronic Total Occlusion (J-CTO) score, both measures are reported to allow comparison with sample size assumptions and to account for non-normal distribution). Categorical variables as counts and percentages. Between-group comparisons were performed using the Student t test for normally distributed continuous variables and the Mann–Whitney U test otherwise; categorical variables were compared using Fisher’s exact test or the chi-square test, as appropriate. Post hoc, we performed an analysis of covariance (ANCOVA) model with VASa as the dependent variable and treatment group as the main effect, adjusting for baseline anxiety. As a sensitivity analysis, we additionally adjusted for intraoperative opioid and benzodiazepine administration. Finally, to address the randomization imbalance in angiographic complexity, a multivariable linear regression analysis was conducted with maximum procedural anxiety as the dependent variable, adjusting for baseline anxiety and the J-CTO score. All tests were 2-sided, with a significance threshold of P < .05. Analyses were performed using R (V. 4.3.2, R Foundation for Statistical Computing, Austria). Reporting followed CONSORT guidelines for trial conduct and prespecified analyses (checklist in the supplementary data).17
RESULTS
Patients
Between 1 March 2022 and 23 October 2025, a total of 119 scheduled CTO PCI were performed. Fifteen patients declined the use of the headset, and in 44 cases the VR device was not available. One patient was excluded due to visual impairment (blindness). Overall, 59 patients were randomized to VR goggles (n = 31) or usual care (n = 28), as shown in figure 2.
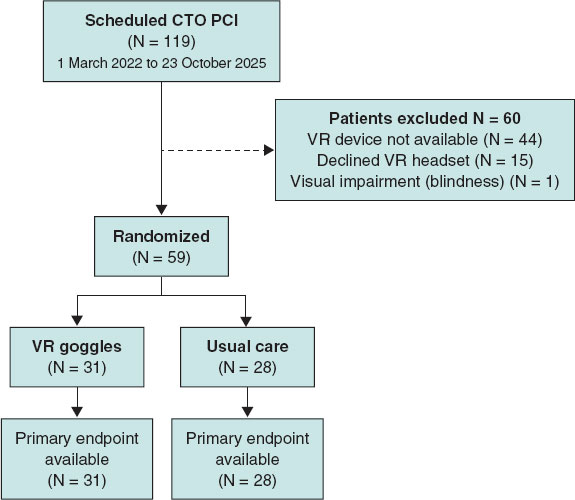
Figure 2. Study flow diagram. CTO PCI, percutaneous coronary intervention for chronic total occlusion; VR, virtual reality.
Enrolment was prospective; however, randomization was contingent on operational availability of the VR system. Early in the study, a single trained investigator was responsible for device preparation and operation, so randomization could only occur when this investigator was present in the cath lab (eg, during vacations or other clinical/research commitments). From June 2023 onward, a second investigator was trained, increasing coverage. In addition, the VR system was out of service for approximately 2 months due to changes in the required internet infrastructure. Importantly, nonrandomization during these periods was driven by logistical constraints rather than patient characteristics.
Baseline characteristics
Baseline clinical characteristics were broadly similar between groups. Median age was 67 [60;70] years in the VR group and 60 [58;66] years in the control group, and most participants were men (29 [93.5%] vs 26 [92.9%]). The prevalence of hypertension (51.6% vs 60.7%) and diabetes (41.9% vs 46.4%) was comparable; dyslipidemia was numerically less frequent in the VR group (58.1% vs 82.1%). Baseline anxiety assessed before entering the cath lab was moderate and did not differ significantly between groups (VAS anxiety: mean ± SD, 3.35 ± 3.10 vs 4.21 ± 3.05; P = .289; median [IQR], 2 [0; 6] vs 5 [2; 6]). Baseline pain was minimal in both groups (VAS pain baseline: 0.06 ± 0.36 vs 0.00 ± 0.00; P = .346; median [IQR], 0 [0 to 0] in both). Baseline angina-related health status was similar. The SAQ score was 52.4 [36.3; 64.3] in the VR group and 50.0 [44.0; 64.3] in the control group (P = .475), as shown in table 1.
Angiographic complexity differed between groups. The J-CTO score was higher in the VR group (mean ± SD, 3.10 ± 1.11; median [IQR], 3 [2; 4]) than in the control group (2.32 ± 1.36; median [IQR], 2 [1; 3]) (Student t test P = .019; Mann-Whitney U test P = .040).
Procedural characteristics and clinical outcomes
The distribution of procedural approach (antegrade, retrograde, or hybrid) was similar between groups (P = .826). Procedural duration, fluoroscopy time, radiation dose-area product, and contrast volume were not significantly different between groups, although radiation exposure tended to be higher in the VR group.
Technical success was achieved in 25 of 31 patients (80.6%) in the VR and 25 of 28 patients (89.3%) in the usual care group (P = .477). Procedural complications occurred in 4 of 31 patients (12.9%) in the VR group and 4 of 28 patients (14.3%) in the control group (P = 1.00) (table 1).
Table 1. Baseline characteristics, lesion and procedural characteristics
| Characteristic | VR goggles (n = 31) | Usual care (n = 28) | P |
|---|---|---|---|
| Age, years | 67 [60; 70] | 60 [58; 66] | .191 |
| Male sex | 29/31 (93.5%) | 26/28 (92.9%) | 1.000 |
| Hypertension | 16/31 (51.6%) | 17/28 (60.7%) | .482 |
| Dyslipidemia | 18/31 (58.1%) | 23/28 (82.1%) | .045 |
| Diabetes mellitus | 13/31 (41.9%) | 13/28 (46.4%) | .728 |
| Chronic kidney disease | 2/31 (6.5%) | 3/28 (10.7%) | .661 |
| Peripheral arterial disease | 5/31 (16.1%) | 4/28 (14.3%) | 1.000 |
| Current smoker | 5/31 (16.1%) | 10/28 (35.7%) | .084 |
| Former smoker | 15/31 (48.4%) | 6/28 (21.4%) | .031 |
| Previous myocardial infarction | 16/31 (51.6%) | 15/28 (53.6%) | .880 |
| Previous PCI | 19/31 (61.3%) | 17/28 (60.7%) | .966 |
| Previous CABG | 4/31 (12.9%) | 0/28 (0.0%) | .118 |
| Previous CTO-PCI attempt | 5/31 (16.1%) | 4/28 (14.3%) | 1.000 |
| Number of previous CTO attempts | 0 [0; 1] | 0 [0; 1] | .917 |
| NYHA class | 1 [1; 2] | 1 [1; 2] | .742 |
| Left ventricular ejection fraction, % | 50.4 ± 15.1 | 51.6 ± 14.0 | .742 |
| Hemoglobin, g/dL | 14.30 ± 1.79 | 14.33 ± 1.81 | .955 |
| Creatinine, mg/dL | 0.94 [0.80; 1.10] | 0.92 [0.81; 1.10] | .911 |
| NT-proBNP, pg/mL | 432 [186; 1406] | 390 [213; 1002] | .765 |
| Seattle Angina Questionnaire summary score (0-100) | 52.4 [36.3; 64.3] | 50.0 [44.0; 64.3] | .475 |
| Baseline anxiety (VAS 0-10) (mean) | 3.35 ± 3.10 | 4.21 ± 3.05 | .289 |
| Baseline anxiety (VAS 0-10) (IQR) | 2 [0; 6] | 5 [2; 6] | .289 |
| Baseline pain (VAS 0-10) (mean) | 0.06 ± 0.36 | 0.00 ± 0.00 | .346 |
| Baseline pain (VAS 0-10) (IQR) | 0 [0; 0] | 0 [0; 0] | .346 |
| Familiarity with technology: no | 4/31 (12.9%) | 2/28 (7.1%) | .389 |
| Familiarity with technology: cell phone | 10/31 (32.3%) | 11/28 (39.3%) | |
| Familiarity with technology: cell phone + personal computer | 15/31 (48.4%) | 15/28 (53.6%) | |
| Familiarity with technology: missing | 2/31 (6.5%) | 0/28 (0.0%) | |
| Previous VR experience: no | 25/31 (80.6%) | 24/28 (85.7%) | .103 |
| Previous VR experience: yes | 4/31 (12.9%) | 3/28 (10.7%) | |
| Previous VR experience: unknown | 0/31 (0.0%) | 1/28 (3.6%) | |
| Previous VR experience: missing | 2/31 (6.5%) | 0/28 (0.0%) | |
| Belief that video could relax: no | 7/31 (22.6%) | 1/28 (3.6%) | .103 |
| Belief that video could relax: yes | 23/31 (74.2%) | 26/28 (92.9%) | |
| Belief that video could relax: unknown | 1/31 (3.2%) | 1/28 (3.6%) | |
| SYNTAX score | 12 [9; 21] | 13 [11; 18] | .451 |
| J-CTO score (median) | 3 [2; 4] | 2 [1; 3] | .040 |
| J-CTO score (mean) | 3.10 ± 1.11 | 2.32 ± 1.36 | .019 |
| In-stent CTO | 1/30 (3.3%) | 3/28 (10.7%) | .344 |
| Occlusion length, mm | 30 [20; 40] | 30 [15; 40] | .209 |
| Blunt proximal cap | 21/31 (67.7%) | 22/28 (78.6%) | .350 |
| Moderate/severe calcification | 26/31 (83.9%) | 15/28 (53.6%) | .012 |
| Bending > 45° | 17/31 (54.8%) | 7/28 (25.0%) | .020 |
| Collateral pattern: ipsilateral | 4/31 (12.9%) | 2/28 (7.1%) | .346 |
| Collateral pattern: contralateral | 20/31 (64.5%) | 15/28 (53.6%) | |
| Collateral pattern: ipsi- and contralateral | 7/31 (22.6%) | 11/28 (39.3%) | |
| Approach: antegrade | 25/31 (80.6%) | 22/28 (78.6%) | .826 |
| Approach: retrograde | 4/31 (12.9%) | 3/28 (10.7%) | |
| Approach: hybrid | 2/31 (6.5%) | 3/28 (10.7%) | |
| Any femoral access used | 4/31 (12.9%) | 4/28 (14.3%) | 1.000 |
| Procedure duration, min | 123 ± 38 | 107 ± 55 | .216 |
| Fluoroscopy time, min | 48 [35; 63] | 40 [20; 67] | .370 |
| Dose-area product, Gy · cm2 | 218 [183; 321] | 178 [103; 281] | .052 |
| Contrast volume, mL | 145 [106; 220] | 150 [112; 198] | .967 |
| No. of stents | 2 [1; 3] | 1 [1; 2] | .030 |
| Total stent length, mm | 80 ± 40 | 52 ± 28 | .006 |
| Technical success | 25/31 (80.6%) | 25/28 (89.3%) | .477 |
| Procedural complications | 4/31 (12.9%) | 4/28 (14.3%) | 1.000 |
|
Data are expressed as mean (SD), median [interquartile range], or n/N (%). P values were calculated using the Student t test for normally distributed variables, the Mann–Whitney U test for non-normally distributed variables, and the chi-square or Fisher exact test for categorical variables, according to the prespecified analysis plan. NT-proBNP available in 13 VR and 12 control patients; SAQ available in 30 VR and 27 control patients; NYHA available in 30 VR and 28 control patients. For baseline VAS anxiety and pain, both mean ± SD and median [Q1; Q3] are reported. CABG, coronary artery bypass grafting; CTO, chronic total occlusion; J-CTO, Japanese Chronic Total Occlusion score; NT-proBNP, N-terminal pro-B-type natriuretic peptide; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; SAQ, Seattle Angina Questionnaire; SD, standard deviation; SYNTAX, Synergy Between PCI With TAXUS and Cardiac Surgery score; VAS, visual analogue scale; VR, virtual reality. |
|||
Primary endpoint
The primary endpoint (maximum procedural anxiety; [VASa]) and key secondary endpoints (maximum procedural pain [VASp] and intraoperative drug administration) were available for all randomized participants.
Maximum procedural anxiety did not differ significantly between groups. Mean VASa was 3.23 ± 2.78 with VR and 3.75 ± 2.77 with usual care (mean difference, −0.52 points; 95% confidence interval (95%CI), −1.97 to 0.92; Student t test P = .472). Median [IQR] values were 4 [0; 5] and 4 [2; 6], respectively. Because the VASa distribution was skewed, results were consistent using the Mann–Whitney U test (P = .379) (figure 3, table 2 and table S1).
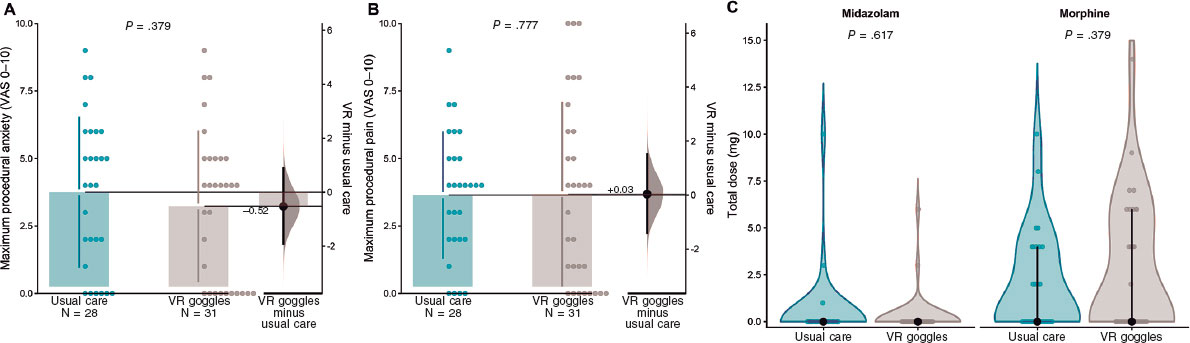
Figure 3. Patient-reported procedural anxiety and pain, and intraoperative drug use. A: maximum procedural anxiety assessed immediately after the procedure using a visual analogue scale for anxiety (VASa; 0–10). B: maximum procedural pain assessed using a visual analogue scale for pain (VASp; 0–10). C: total intraoperative dose (mg) of midazolam and morphine by treatment group; zero values indicate no drug administration. In panels A and B, dots represent individual patients; the right-side estimation plot shows the mean between-group difference (VR minus usual care) with its 95% confidence interval. P values correspond to prespecified between-group comparisons. VAS, visual analogue scale; VR, virtual reality.
Table 2. Endpoints and safety
| Variable | VR goggles | Usual care | P |
|---|---|---|---|
| Maximum procedural anxiety* | 3.23 ± 2.78 | 3.75 ± 2.77 | .472 |
| Maximum procedural anxiety* | 4 [0; 5] | 4 [2 to 6] | .379 |
| Maximum procedural pain* | 3.68 ± 3.39 | 3.64 ± 2.33 | .964 |
| Maximum procedural pain* | 4 [1; 6] | 4 [2;5] | .777 |
| Morphine administered | 14/31 (45.2) | 13/28 (46.4) | 1.000 |
| Total morphine dose, mg | 2.74 ± 3.61 | 2.00 ± 2.71 | .379 |
| Midazolam administered | 2/31 (6.5) | 3/28 (10.7) | .661 |
| Total midazolam dose, mg | 0.29 ± 1.19 | 0.50 ± 1.95 | .617 |
| Nausea during procedure | 1/31 (3.2) | 0/28 (0.0) | 1.000 |
| Dizziness during procedure | 1/31 (3.2) | 0/28 (0.0) | 1.000 |
|
Values are mean ± SD, median [Q1; Q3], or n/N (%). For visual analogue scale (VAS) anxiety and pain, both mean ± SD and median [Q1; Q3] are reported due to skewness of the distributions. P values for rows reporting mean ± SD are from Student’s t test; P values for rows reporting median [Q1; Q3] are from Mann–Whitney U tests. Categorical variables were compared using Fisher’s exact test. Total morphine and midazolam doses (mg) include zeros for patients not receiving the drug. SD, standard deviation; VR, virtual reality. * VAS 0-10. |
|||
In a post hoc ANCOVA adjusting for baseline anxiety, VR was not associated with lower peak procedural anxiety (adjusted mean difference, −0.21; 95%CI, −1.62-1.20; P = .773). Results remained consistent after additional adjustment for intraoperative morphine and midazolam dose (table S1 and table S2).
Given the significantly higher angiographic complexity in the VR group, a sensitivity analysis was conducted using multivariable linear regression. After adjusting for J-CTO score and baseline anxiety, the effect of VR on procedural anxiety remained non-significant (adjusted coefficient -0.40; 95%CI, -1.81 to 1.02; P = .576). In this model, procedural complexity was not independently associated with patient-reported anxiety (P = .370); see table S1.
Secondary endpoints
VASp was similar in the 2 groups: 3.68 ± 3.39 with VR and 3.64 ± 2.33 with usual care (mean difference, 0.03; 95%CI, −1.50-1.57; P = .964). Median [IQR] values were 4 [1; 6] and 4 [2; 5], respectively (P = .777).
Intraoperative pharmacologic treatment did not differ between groups. Morphine was administered to 14 of 31 patients (45.2%) in the VR group and 13 of 28 (46.4%) in the control group (P = 1.00). Total morphine dose (including zero valus for nonuse) was 2.74 ± 3.61 mg and 2.00 ± 2.71 mg, respectively (P = .379). Midazolam was administered to 2 of 31 patients (6.5%) in the VR group and 3 of 28 (10.7%) in the control group (P = .661), with total midazolam doses of 0.29 ± 1.19 mg and 0.50 ± 1.95 mg, respectively (P = .617) (figure 3 and table 2).
Safety and acceptability
Symptoms potentially attributable to VR were uncommon. Nausea occurred in 1 patient (3.2%) in the VR group and in no patients in the control group (P = 1.00). Dizziness occurred in 1 patient (3.2%) in the VR group and in no patients in the control group (P = 1.00).
Among patients assigned to VR, 25 of 31 (80.6%) reported that they would be willing to use a relaxation video again during a future intervention; 5 (16.1%) would not, and 1 (3.2%) was uncertain.
DISCUSSION
We conducted a randomized trial to evaluate whether VR-based audiovisual distraction during elective CTO PCI can improve the patient experience. Several key findings emerge. First, VR did not meaningfully reduce patient-reported peak procedural anxiety, and pain outcomes were similarly neutral, arguing against a clinically relevant anxiolytic or analgesic effect with the intervention as delivered. Second, VR did not reduce the use of intraoperative opioid or benzodiazepine requirements, suggesting limited incremental benefit beyond contemporary usual care in the cath lab. Third, VR implementation appeared feasible and was not associated with signals of procedural harm, with overall procedural outcomes remaining comparable despite an imbalance toward greater angiographic complexity in the VR group. Finally, acceptability was generally high among patients assigned to VR, supporting its role as a patient-centered option for selected individuals even when average effects on anxiety are modest.
Interpreting the neutral result in the context of prior interventional cardiology VR trials
Several factors may explain the absence of a measurable anxiolytic effect in ReViCTO. First, peak procedural anxiety levels were relatively low, leaving limiting the potential for improvement and increasing the likelihood of a floor effect. A similar pattern has been observed in contemporary minimally sedated structural interventions, in which overall anxiety burden is modest and VR has not consistently produced detectable differences on global assessments.18
Second, anxiety and pain were assessed immediately after the procedure as a single recalled measure of peak intensity. Although pragmatic and consistent with the study protocol, this approach relies on retrospective recall and may underestimate brief, procedure-specific peaks of discomfort that are particularly relevant in CTO PCI, such as arterial puncture, prolonged immobility, complex device manipulation, or episodes of ischemic chest pain.19 Consequently, any benefit limited to these discrete high-stress intervals may have been diluted when summarized as a single postprocedural peak value.
Third, the VR content in the ReViCTO consisted of passive content (documentary viewing). Across procedural settings, the magnitude of VR’s effect appears to depend on how strongly the experience captures attention and induces relaxation. Interventions incorporating guided relaxation, hypnosis-based modules, or interactive elements have more consistently demonstrated larger and more reproducible effects than passive viewing alone.20-22
In the context of the broader interventional cardiology literature, the neutral effect of VR on peak intraoperative anxiety in ReViCTO is therefore less surprising. Existing cath lab and structural heart studies suggest that observable benefit depends on when VR is delivered, which patients are targeted, particularly those with higher baseline anxiety, and how outcomes are measured. In transcatheter aortic valve implantation (TAVI), early randomized studies reported lower VAS-based procedural anxiety, supporting feasibility and a potential anxiolytic effect.11 In contrast, a larger minimalistic TAVI randomized study found improvements in state anxiety (State-Trait Anxiety Inventory, state scale [STAI-S]) and perceived procedure duration, yet less consistent effects on VAS-based anxiety or pain, underscoring the influence of measurement instruments on signal detection.23 Evidence from coronary procedures is similarly nuanced. In the VR InCard program, which targeted patients with elevated preoperative anxiety and incorporated structured preprocedural sessions, the primary analysis was neutral but adjusted analyses suggested modest reductions, with heterogeneity by clinical context.24,25 Similarly, a randomized trial conducted before coronary angiography reported anxiety reduction using questionnaire-based assessments rather than a single recalled postoperative peak rating.26 Collectively, these data support the interpretation that VR may be most effective when targeted to patients with higher baseline anxiety, delivered during anticipatory or discrete high-stress phases, and evaluated using instruments sensitive to global state anxiety, rather than relying solely on a single recalled peak score.22,27
Pharmacologic co-intervention and post hoc adjustment
A key pragmatic feature of ReViCTO was that background anxiolysis and analgesia were not protocolized, reflecting real-world practice in the cath lab where conscious sedation and intraoperative comfort measures are typically individualized. This approach is consistent with contemporary cath lab randomized studies evaluating patient comfort under usual care conditions.28,29 In the final dataset, intraoperative morphine and midazolam use were similar, and dose distributions did not differ materially. To align with current analytic recommendations, we performed a post hoc ANCOVA adjusting peak anxiety for baseline anxiety, an approach that can improve precision when baseline values are prognostic.30 Baseline anxiety was strongly associated with peak anxiety, while VR assignment was not (adjusted VR effect approximately -0.32 VAS points; P = .64). Adding opioid and benzodiazepine doses did not materially change the VR estimate, supporting that the neutral primary result is unlikely to be explained by baseline imbalance or differential pharmacologic rescue. Baseline anxiety was higher in the usual care group; adjusted analyses yielded consistent findings.
Feasibility, tolerability, and patient-center implementation
From a feasibility standpoint, VR use in CTO PCI appeared safe and compatible with the cath lab environment, with nausea and dizziness reported rarely and no signal suggesting increased procedural instability. The practical challenge was tolerability during long procedures: early discontinuation of the headset occurred in 9 VR patients, which is consistent with broader evidence that fully immersive head-mounted displays can trigger discomfort or cybersickness in a minority of users, particularly with extended exposure.31 Implementation studies further indicate that device-related factors—such as physical interference, usability barriers, and the need for individual tailoring—can influence both uptake and sustained use, supporting a selective rather than universal deployment strategy.32 These observations support a patient-center approach: VR may be most useful when offered to patients with higher baseline anxiety, a clear preference for audiovisual distraction, or anticipated prolonged immobility, and when focused around discrete high-stress phases. Preoperative counseling remains essential with VR serving as a complementary, rather than substitutive, strategy.
Limitations
This trial has limitations that should be considered. First, it was a single-center, open-label study and subjective outcomes may be susceptible to reporting buas; however, randomization, standardized outcome collection, and consistently neutral findings across patient-reported outcomes and medication use make a major bias-driven effect unlikely. Second, recruitment was not consecutive and 12.6% of eligible patients declined participation, which may limit generalizability; nonetheless, this also reflects real-world acceptability of a wearable intervention, and the decliner rate is transparently reported. Third, anxiety and pain were assessed as a single postoperative peak rating rather than repeated measures during predefined high-stress phases; this approach was identical in both groups, and recalled peak experience remains clinically relevant for satisfaction and willingness to undergo future procedures. Fourth, anxiolysis and analgesia were not protocolized and were operator-directed, potentially attenuating any incremental benefit of VR. Nevertheless, this pragmatic design improves applicability and bailout medication was systematically recorded. Fifth, VR content was passive and exposure time likely varied due to early discontinuation; the intention-to-treat analysis therefore estimates the real-world effect of offering VR. Finally, although angiographic complexity (J-CTO score) was higher in the VR group, multivariable analyses indicated this imbalance did not explain the neutral anxiety results and complexity was not a significant predictor of distress in this cohort. Benzodiazepine amnesia may attenuate recall; midazolam use was low and balanced.
CONCLUSIONS
In this randomized trial of patients undergoing elective CTO PCI, although immersive VR was feasible and well tolerated, it did not reduce patient-reported peak intraoperative anxiety or pain, or the need for intraoperative morphine or midazolam vs usual care.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The protocol was approved by the Clinical Research Ethics Committee of Hospital Clínico Universitario de València (28 February, 2022), and all participants gave their prior written informed consent. Sex distribution is reported, and reporting adhered to SAGER guidelines to mitigate potential sex- and gender-related bias.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Generative artificial intelligence was used to support language editing and formatting of the revised manuscript. No artificial intelligence tools were used to analyze study data. All content was reviewed and approved by the authors, who take full responsibility for the manuscript.
AUTHORS’ CONTRIBUTIONS
A. Fernández-Cisnal and B. Silla conceived the study and designed the trial. A. Fernández-Cisnal, B. Silla, C.E. Vergara-Uzcategui, J.M. Ramón, E. Valero, and C. Romero Menor contributed to patient inclusion and data acquisition. A. Fernández-Cisnal performed the statistical analyses and drafted the manuscript. B. Silla, C.E. Vergara- Uzcategui, J.M. Ramón, E. Valero, C. Romero Menor, J. Núñez, V. Bodí, and G. Miñana critically revised the manuscript for important intellectual content. All authors approved the final version and agreed to be accountable for all aspects of the work.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- CTO PCI is a long, technically complex cath lab procedure that can cause anxiety and discomfort for patients.
- Patient anxiety during coronary procedures may worsen perceived discomfort and can increase the need for intraoperative sedatives or analgesics, although pharmacological strategies show modest and variable benefits in routine practice.
- VR provides immersive audiovisual distraction and has been shown to reduce pain and anxiety in several procedural and clinical settings.
- Early studies in interventional cardiology suggest VR can be feasible during procedures performed under conscious sedation, but evidence is mixed and randomized data in CTO PCI were lacking.
WHAT DOES THIS STUDY ADD?
- In the prospective, single-center randomized ReViCTO trial with 59 patients undergoing elective CTO PCI, intraoperative VR goggles were feasible, safe, and generally well tolerated.
- VR did not significantly reduce patient-reported peak procedural anxiety vs usual care and similarly showed no reduction in peak pain.
- VR did not decrease the use or total dose of intraoperative morphine or midazolam, suggesting limited incremental benefit over contemporary usual care.
- Despite a higher angiographic complexity in the VR group, procedural outcomes and complication rates were comparable, and most VR patients reported they would consider using the device again in future interventions.
REFERENCES
1. Patel VG, Brayton KM, Tamayo A, et al. Angiographic Success and Procedural Complications in Patients Undergoing Percutaneous Coronary Chronic Total Occlusion Interventions. JACC Cardiovasc Interv. 2013;6:128-136.
2. Brilakis ES, Grantham JA, Rinfret S, et al. A Percutaneous Treatment Algorithm for Crossing Coronary Chronic Total Occlusions. JACC Cardiovasc Interv. 2012;5:367-379.
3. Christopoulos G, Karmpaliotis D, Alaswad K, et al. Application and outcomes of a hybrid approach to chronic total occlusion percutaneous coronary intervention in a contemporary multicenter US registry. Int J Cardiol. 2015;198:222-228.
4. Fefer P, Knudtson ML, Cheema AN, et al. Current Perspectives on Coronary Chronic Total Occlusions. J Am Coll Cardiol. 2012;59:991-997.
5. Tajti P, Burke MN, Karmpaliotis D, et al. Update in the Percutaneous Management of Coronary Chronic Total Occlusions. JACC Cardiovasc Interv. 2018;11:615-625.
6. Delewi R, Vlastra W, Rohling WJ, et al. Anxiety levels of patients undergoing coronary procedures in the catheterization laboratory. Int J Cardiol. 2017;228:926-930.
7. Trotter R, Gallagher R, Donoghue J. Anxiety in patients undergoing percutaneous coronary interventions. Heart Lung. 2011;40:185-192.
8. Astin F, Jones K, Thompson DR. Prevalence and patterns of anxiety and depression in patients undergoing elective percutaneous transluminal coronary angioplasty. Heart Lung. 2005;34:393-401.
9. Vlastra W, Delewi R, Rohling WJ, et al. Premedication to reduce anxiety in patients undergoing coronary angiography and percutaneous coronary intervention. Open Heart. 2018;5:000833.
10. Teh JJ, Pascoe DJ, Hafeji S, et al. Efficacy of virtual reality for pain relief in medical procedures:a systematic review and meta-analysis. BMC Med. 2024;22:64.
11. Bruno RR, Lin Y, Wolff G, et al. Virtual reality-assisted conscious sedation during transcatheter aortic valve implantation:a randomised pilot study. EuroIntervention. 2020;16:1014-1020.
12. Roxburgh T, Li A, Guenancia C, et al. Virtual Reality for Sedation During Atrial Fibrillation Ablation in Clinical Practice:Observational Study. J Med Internet Res. 2021;23:26349.
13. Fernández-Cisnal A, Silla B, María Ramón J, et al. Efficacy of virtual reality reducing anxiety during CTO revascularization:the ReViCTO trial design. REC Interv Cardiol. 2023;5:203-209.
14. Schulz KF, Grimes DA. Unequal group sizes in randomised trials:guarding against guessing. Lancet. 2002;359:966-970.
15. Spertus JA, Winder JA, Dewhurst TA, et al. Development and evaluation of the Seattle Angina questionnaire:A new functional status measure for coronary artery disease. J Am Coll Cardiol. 1995;25:333-341.
16. Delewi R, Vlastra W, Rohling WJ, et al. Anxiety levels of patients undergoing coronary procedures in the catheterization laboratory. Int J Cardiol. 2017;228:926-930.
17. Chan AW, Boutron I, Hopewell S, et al. SPIRIT 2025 statement:updated guideline for protocols of randomised trials. BMJ. 2025;389:081477.
18. Chatterjee S, Ooms J, De Ronde M, et al. Anxiety during transcatheter aortic valve replacement under local anesthesia - the ART-VR trial. Cardiovasc Revasc Med. 2025;79:71-77.
19. Brilakis ES, Banerjee S, Karmpaliotis D, et al. Procedural Outcomes of Chronic Total Occlusion Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2015;8:245-253.
20. Bekelis K, Calnan D, Simmons N, MacKenzie TA, Kakoulides G. Effect of an Immersive Preoperative Virtual Reality Experience on Patient Reported Outcomes:A Randomized Controlled Trial. Ann Surg. 2017;265:1068-1073.
21. Gullo G, Rotzinger DC, Colin A, et al. Virtually Augmented Self-Hypnosis in Peripheral Vascular Intervention:A Randomized Controlled Trial. Cardiovasc Intervent Radiol. 2023;46:786-793.
22. El Mathari S, Hoekman A, Kharbanda RK, et al. Virtual Reality for Pain and Anxiety Management in Cardiac Surgery and Interventional Cardiology. JACC:Advances. 2024;3:100814.
23. Lind A, Ahsan M, Totzeck M, et al. Virtual reality-assisted distraction during transcatheter aortic valve implantation under local anaesthesia:A randomised study. Int J Cardiol. 2023;387:131130.
24. Groenveld TD, Breunissen EHW, Bonnes JL, et al. Virtual Reality to Reduce Preprocedural Anxiety During Invasive Coronary Angiography. JACC:Advances. 2025;4:101976.
25. Breunissen EHW, Groenveld TD, Garms L, Bonnes JL, Van Goor H, Damman P. Virtual reality to reduce periprocedural anxiety during invasive coronary angiography:rationale and design of the VR InCard trial. Open Heart. 2024;11:002628.
26. Keshvari M, Yeganeh MR, Paryad E, Roushan ZA, Pouralizadeh M. The effect of virtual reality distraction on reducing patients'anxiety before coronary angiography:a randomized clinical trial study. Egypt Heart J. 2021;73:98.
27. Micheluzzi V, Burrai F, Casula M, et al. Effectiveness of virtual reality on pain and anxiety in patients undergoing cardiac procedures:A systematic review and meta-analysis of randomized controlled trials. Curr Probl Cardiol. 2024;49:102532.
28. Boukantar M, Chiaroni PM, Gallet R, et al. A Randomized Controlled Trial of Nonfasting vs Fasting Before Interventional Coronary Procedures. JACC Cardiovasc Interv. 2024;17:1200-1210.
29. Ferreira D, Hardy J, Meere W, et al. Fasting >no fasting prior to catheterization laboratory procedures:the SCOFF trial. Eur Heart J. 2024;45:4990-4998.
30. Tackney MS, Morris T, White I, Leyrat C, Diaz-Ordaz K, Williamson E. A comparison of covariate adjustment approaches under model misspecification in individually randomized trials. Trials. 2023;24:14.
31. Cossio S, Chiappinotto S, Dentice S, et al. Cybersickness and discomfort from head-mounted displays delivering fully immersive virtual reality:A systematic review. Nurse Educ Pract. 2025;85:104376.
32. Kouijzer MMTE, Kip H, Bouman YHA, Kelders SM. Implementation of virtual reality in healthcare:a scoping review on the implementation process of virtual reality in various healthcare settings. Implement Sci Commun. 2023;4:67.
ABSTRACT
Introduction and objectives: Calcified coronary nodules (CN) are associated with suboptimal outcomes following percutaneous coronary intervention. The aim of this study is to compare the safety and efficacy profile of orbital atherectomy (OA) and intravascular lithotripsy (IVL) in the management of CN.
Methods: The ORBIT-SHOCK pilot study is a randomized, multicenter clinical trial designed to enroll 50 patients with coronary CN identified by optical coherence tomography and deemed suitable for percutaneous coronary intervention. Patients will be randomly assigned in a 1:1 ratio to receive either OA or IVL, with stratified randomization based on the type of CN (eruptive or noneruptive) to ensure balanced distribution across treatment groups. The primary endpoint is stent expansion at the site of the CN, assessed by optical coherence tomography, while secondary endpoints will include procedural success, strategy success, stent apposition, and the rate of adverse clinical events such as target lesion failure, target vessel failure, and major adverse cardiovascular events at the 12-month follow-up.
Conclusions: The ORBIT-SHOCK pilot trial is designed as the first randomized clinical trial to compare the safety and efficacy of OA and IVL in the management of CN. (ClinicalTrials.gov: NCT06736665).
Keywords: Coronary calcification. Calcified nodules. Orbital atherectomy. Intravascular lithotripsy. Percutaneous coronary intervention. Complex PCI.
RESUMEN
Introducción y objetivos: Los nódulos coronarios calcificados (NC) se asocian con resultados subóptimos tras la intervención coronaria percutánea. El objetivo de este estudio es comparar la eficacia y la seguridad de la aterectomía orbital (AO) y la litotricia intravascular (LIV) en el tratamiento de los NC.
Métodos: El estudio piloto ORBIT-SHOCK es un ensayo clínico aleatorizado y multicéntrico que incluirá 50 pacientes con NC identificados mediante tomografía de coherencia óptica y candidatos a intervención coronaria percutánea. Los pacientes se asignarán aleatoriamente en una proporción 1:1 para recibir AO o LIV, con una aleatorización estratificada basada en el tipo de NC (eruptivo o no eruptivo) para garantizar una distribución equilibrada entre los grupos de tratamiento. El objetivo primario del estudio es la expansión del stent en el sitio del NC evaluada por tomografía, mientras que los objetivos secundarios son el éxito del procedimiento, el éxito de la estrategia, la aposición del stent y la incidencia de eventos clínicos adversos, como el fallo de la lesión diana, el fallo del vaso diana y los eventos cardiovasculares mayores a los 12 meses de seguimiento.
Conclusiones: El ensayo piloto ORBIT-SHOCK será el primer estudio aleatorizado para comparar la eficacia y la seguridad de la AO y la LIV en el tratamiento de los NC. (ClinicalTrials.gov: NCT06736665).
Palabras clave: Calcificación coronaria. Nódulos calcificados. Aterectomía orbitacional. Litotricia intracoronaria. Intervención coronaria percutánea. ICP compleja.
Abbreviations
CN: calcified nodule. IVL: intravascular lithotripsy. OA: orbital atherectomy. OCT: optical coherence tomography. PCI: percutaneous coronary intervention.
INTRODUCTION
Calcified nodules (CN) represent a significant challenge during percutaneous coronary intervention (PCI), as their mechanical resistance limits effective lesion preparation and adequate stent expansion, resulting in suboptimal angiographic outcomes and a higher risk of adverse clinical events.1-7
Intracoronary optical coherence tomography (OCT) is highly effective for the detection and characterization of coronary calcium, offering superior sensitivity and specificity than angiography. Robust evidence demonstrates that OCT-guided PCI improves both procedural and clinical outcomes, supporting guideline recommendations for its use in complex coronary lesions.8-11
Several dedicated devices and techniques have been developed to facilitate calcium modification and optimize lesion preparation. Among these, intravascular lithotripsy (IVL) has emerged as a widely adopted strategy for the treatment of CN because of its ease of use, low complication rates, and favorable outcomes in recent studies.2,3,7,12
Alternatively, orbital atherectomy (OA) has been shown to be an effective strategy for the treatment of severely calcified and non- dilatable coronary lesions. However, evidence regarding its performance in CN remains limited and is largely derived from small observational studies and case reports.1,12-23
Despite major advances in imaging and plaque-modification technologies, the optimal management strategy for CN remains uncertain.
METHODS
Population and study design
The ORBIT-SHOCK pilot study is a multicenter, prospective, investigator-initiated randomized pilot study, sponsored by the Spanish Society of Cardiology. It will include patients diagnosed with atherosclerotic disease with evidence of CN identified by OCT, causing significant angiographic stenosis eligible for revascularization through PCI.
A CN, as identified by OCT, is defined as a localized protruding mass within the coronary artery wall that appears as a signal-poor region with sharply delineated borders. An eruptive CN is characterized by an accumulation of small calcium fragments that protrude through and disrupt the overlying fibrous cap, typically associated with small amount of thrombus. In contrast, a noneruptive CN is defined as an accumulation of small calcium fragments covered by a smooth intact fibrous cap without overlying thrombus.1
Inclusion and exclusion criteria are detailed in table 1. The selection of eligible patients will be conducted by the investigators at each participant center. Following confirmation of the presence of a CN and verification of compliance with the inclusion criteria, and absence of exclusion criteria, patients will be randomized in a 1:1 ratio to 1 of the 2 lesion preparation strategies under study: OA or IVL (figure 1). Randomization will be stratified according to the type of CN (eruptive or noneruptive) to ensure a balanced distribution across treatment groups. Randomization occurs immediately after OCT confirmation of the presence of CN and before any plaque-modification strategy has been started.
Table 1. Inclusion and exclusion criteria
| Inclusion criteria |
|---|
| Patients aged ≥ 18 years |
| Atherosclerotic disease with calcified nodules identified by OCT in a native vessel eligible for percutaneous coronary revascularization |
| Clinical presentation with chronic coronary syndrome or non-ST-segment elevation acute coronary syndrome. Nonculprit lesions suitable for revascularization during a staged procedure after ST-segment elevation myocardial infarction (STEMI) will also be eligible for inclusion |
| Distal vessel reference diameters ≥ 2.5 mm and ≤ 4.0 mm |
| Exclusion criteria |
| Culprit lesions in ST-segment elevation acute coronary syndrome |
| Left main disease |
| In-stent restenosis lesions |
| Critical stenoses in which the OCT catheter cannot be advanced across the lesion despite predilation with a balloon ≤ 2 mm in diameter |
| Lesion involving a bifurcation with a side branch diameter ≥ 2 mm |
| Cardiogenic shock |
| Patients requiring cardiac surgery or percutaneous valve intervention within 3 months before or after angioplasty |
| Pregnancy |
| Life expectancy < 1 year |
| Contraindication to appropriate antiplatelet therapy after revascularization |
| Coronary artery disease with an indication for surgical revascularization |
| Advanced chronic kidney disease* or anatomical characteristics that preclude the use of optical coherence tomography |
| Inability to obtain informed consent |
| Allergy to eggs or soy, contraindicating the use of OA |
|
* Evaluated based on the investigator’s clinical judgment regarding the safety and potential risks associated with the contrast volume required for OCT-guided PCI. OA, orbital atherectomy; OCT, optical coherence tomography; STEMI, ST-segment elevation myocardial infarction. |
Figure 1. Central illustration. ORBIT-SHOCK study design and flow chart. Eligible patients will be selected by investigators at each participant center. After confirmation of the presence of a CN and verification of compliance with the inclusion criteria, and exclusion of any exclusion criteria, patients will be randomized in a 1:1 ratio to 1 of the 2 lesion preparation strategies under investigation: OA or IVL. Randomization will be stratified according to the type of CN (eruptive or noneruptive) to ensure a balanced distribution across treatment groups. CN, calcified nodule; IVL, intravascular lithotripsy; NSTEACS, non-ST-segment elevation acute coronary syndrome; STEACS, ST-segment elevation acute coronary syndrome; OA, orbital atherectomy; OCT, optical coherence tomography; PCI, percutaneous coronary intervention.
Endpoints
All endpoints are detailed in table 2 and defined in table S1. The primary endpoint is the percentage of stent expansion at the CN site, assessed by OCT and defined as the ratio between the minimum stent area (MSA) at the CN site and the reference area at the CN site:
Table 2. Study endpoints
| Primary endpoints |
|---|
| Percentage of stent expansion at the CN site: assessed by OCT and defined as the ratio between the minimum stent area at the CN site and the reference lumen area at the CN site. |
| Secondary endpoints |
| Procedural success: defined as achieving a stent expansion of ≥ 80% with TIMI grade-3 flow, in the absence of stent loss, coronary perforation, or intraoperative mortality |
| Strategy success: defined as procedural success without need for crossover to an alternative treatment |
| Minimum stent area |
| Minimum stent area at the CN site |
| Significant stent malapposition at the CN site: measured using OCT, defined as a stent strut detachment ≥ 0.4 mm from the underlying vessel wall with a longitudinal extension ≥ 1 mm |
| Degree of CN debulking, defined as the reduction in CN size (mm2), measured after plaque modification and prior to stenting |
| Evidence of fracture at the CN site, defined as a new disruption or discontinuity observed on OCT after plaque modification and prior to stenting |
| Degree of stent ellipticity at the CN site: measured using OCT at the end of the procedure, calculated as the ratio of the maximum luminal diameter to the minimum luminal diameter of the stent at the CN site |
| Residual luminal protrusion of the CN after stenting, measured by OCT in mm2 |
| Target lesion failure at 12 months, defined as the composite of clinically driven target lesion revascularization, myocardial infarction or cardiac death related to the target lesion |
| Target lesion revascularization at 12 months, defined as a repeat percutaneous coronary intervention of the target lesion or bypass surgery of the target vessel performed for restenosis or other complication of the target lesion. |
| Major adverse cardiovascular events at 12 months: defined as a composite of cardiac death, non-fatal target lesion myocardial infarction, unplanned target lesion revascularization or stent thrombosis. |
|
CN, calcified nodule; OA, orbital atherectomy; OCT, optical coherence tomography; TIMI, Thrombolysis in Myocardial Infarction. |
Stent expansion at the CN site (%) = (MSA at the CN site / reference lumen area at the CN site) x 100
The stent reference area at the CN site will be manually calculated by CoreLab specialists using co-registered pre- and post-stent images. Additionally, stent expansion will also be assessed using the Aptivue automated software (Abbott, United States). Specific methodological details regarding the selection of reference cross- sections and algorithms used to calculate stent expansion are described in the methods section of the supplementary data.
Secondary endpoints include procedural and strategy success, and mechanistic OCT-derived parameters related to calcium modification, including nodule debulking volume and calcium fracture. Additional OCT metrics of final stent optimization will also be assessed, including minimum stent area, stent ellipticity, significant malapposition, and residual nodule protrusion. Finally, clinical outcomes including target lesion failure, target lesion revascularization, and major adverse cardiovascular events will be assessed at the 12-month follow-up.
Study devices and procedures
The recommendations for the use of OA and IVL have been established according to the SCASI Expert Consensus Statement.24 Only CE-marked devices locally approved and commercially available will be used in the study.
The study procedures and their sequence are illustrated in figure 2.
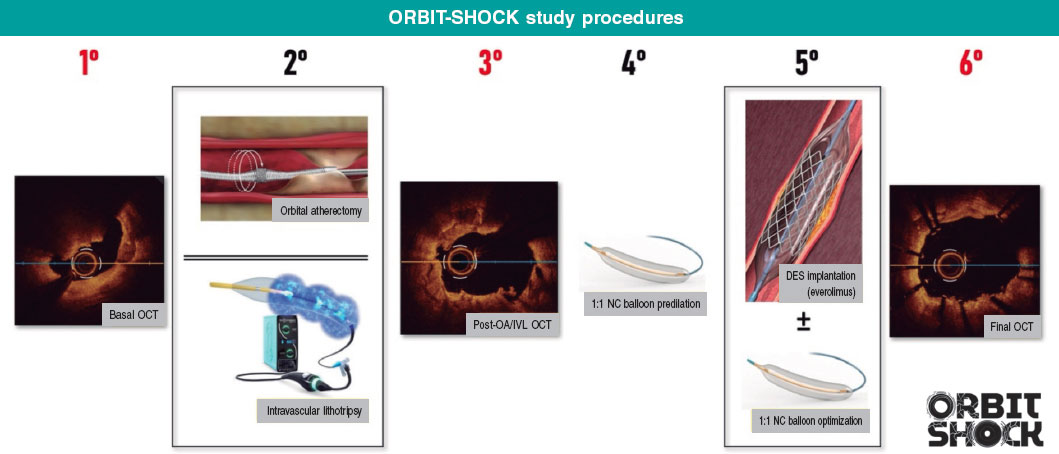
Figure 2. Study procedures. Red numbers indicate OCT acquisitions. 1. The presence of a CN must be confirmed by optical coherence tomography before enrolling a patient with suggestive angiographic findings. OCT imaging before any intervention on the target lesion is mandatory. Predilation with a 1.5 – 2.0 mm balloon is permitted if the OCT catheter cannot cross the lesion. 2. Lesion preparation with OA or IVL according to randomization. 3. Obtaining OCT images after the use of OA or IVL is mandatory, provided it is feasible and the patient’s clinical condition permits it. 4. Prior to stenting, dilation with a 1:1 NC balloon dilation is mandatory. 5. Stenting (use of a next-generation everolimus-eluting stent with CE marking is recommended). Stent optimization through post-dilation with an NC balloon based on OCT images is mandatory. 6. Final OCT images after stenting and optimization must be obtained to document the final procedural result. CN, calcified nodule; DES, drug-eluting stents; IVL, intravascular lithotripsy; NC, noncompliant; OA, orbital atherectomy; OCT, optical coherence tomography.
Optical coherence tomography
The presence of a CN must be confirmed by OCT before including a patient with suggestive angiographic images.
OCT imaging must be obtained before any intervention on the target lesion. Limited predilation with a 1.5–2.0 mm balloon is permitted if the OCT catheter cannot cross the lesion. This minimal predilation is intended solely to facilitate catheter delivery while minimizing structural modification of the nodule and preserving the accuracy of the baseline morphological assessment. OCT acquisition immediately after OA/IVL and after stenting (final result) is mandatory, provided it is technically feasible and the patient’s clinical condition allows it. Although not mandatory, performing an OCT run after 1:1 balloon predilation and immediately prior to stenting is highly recommended to confirm adequate plaque preparation and obtain accurate measurements of the reference vessel diameter and required stent length. Stent optimization through post-dilation with a noncompliant (NC) balloon based on OCT images is mandatory. In such cases, a repeat OCT acquisition must be performed to document the final procedural result.
Orbital atherectomy
A smooth forward and backward motion at a slow advancement speed (approximately 1.0 mm/s) is required to optimize ablation efficiency. For vessels with a reference diameter ≥ 3.0 mm, the use of high rotational speed (120 000 rpm) is strongly recommended. For vessels < 3.0 mm, low rotational speed (80 000 rpm) should be used initially. Operators should aim for the maximum feasible number of passes (typically > 5) to maximize lumen gain, ensuring that individual treatment runs do not exceed 30 seconds to prevent thermal injury. The total treatment time with a single crown should not exceed 5 minutes. Before stenting, dilation with a 1:1 NC balloon is mandatory.
Intravascular lithotripsy
The IVL balloon should be sized 1:1 to the reference vessel diameter to ensure efficient energy transfer. Pulses: delivery of the maximum number of pulses (up to the catheter limit) is strongly recommended to ensure adequate modification of the CN. Appropriate rest periods between therapy cycles are required to allow for distal washout and prevent hemodynamic compromise. Before stenting, dilation with a 1:1 NC balloon is mandatory.
Lesion stenting
To ensure greater treatment homogeneity, the use of a next-generation everolimus-eluting stent with CE marking is recommended. If this is not feasible, the use of another next-generation stent with CE marking is permitted.
Crossover between techniques and the management of special situations are detailed in the procedures section of the supplementary data.
Data collection
A clinical evaluation will be performed and data will be collected at 3 predefined time points: at patient enrollment, at hospital discharge, and at 12 months after PCI (table 3). All data will be recorded in an electronic case report form (eCRF).
Table 3. Study visits and data collection
| Study procedure | Baseline | Hospital discharge | 1-year follow-up (± 30 days) |
|---|---|---|---|
| Patient screening (inclusion/exclusion criteria) | X | ||
| Patient informed consent | X | ||
| Randomization | X | ||
| Patient characteristics | |||
| Demographics | X | ||
| Health history | X | ||
| Physical examination | X | X | X |
| Angina status (CCS class) | X | X | X |
| Medication | X | X | X |
| ECG | X | X | X |
| Echocardiography | X | ||
| Laboratory data | X | X | X |
| Vital status | X | X | X |
| Indication for PCI | X | ||
| PCI procedural variables | |||
| Angiography data | X | ||
| OCT imaging data (measured by a external core laboratory) | X | ||
| Technical variables | X | ||
| Endpoints | X | X | |
| Adverse events | X | X | |
|
CCS, Canadian Cardiovascular Society angina grading system; ECG, electrocardiogram; OCT, optical coherence tomography; PCI, percutaneous coronary intervention. |
|||
OCT images will be analyzed by a central core laboratory, which will remain blinded to the lesion modification technique employed. The core laboratory will be responsible for providing accurate and standardized interpretations, thereby ensuring unbiased results regardless of the treatment strategy applied.
Statistical considerations
Sample size
The limited number of studies specifically addressing CN, together with the heterogeneity in patient selection criteria and methods used to calculate stent expansion, makes it challenging to estimate the sample size required to ensure adequate statistical power. To address this limitation, a pilot study has been designed with 50 patients (25 per group) to evaluate and compare the outcomes of OA and IVL in this clinical setting.
The determination of this sample size was primarily driven by feasibility and pragmatic considerations to ensure the successful completion of the pilot study. Despite this pragmatic approach, the planned sample of 50 patients provides an estimated statistical power (0.8) to detect a clinically relevant difference of 12% in stent expansion under an exploratory superiority framework, assuming an expected standard deviation of ± 15%.
Descriptive statistics and hypothesis testing methodology
The normality of continuous variables will be assessed using the Kolmogorov-Smirnov test. Comparisons of normally distributed quantitative variables between treatment groups will be performed using the Student t test. For ordinal variables or continuous variables that do not follow a normal distribution, the non-parametric Mann-Whitney U test will be used. Qualitative variables wil be compared using the chi-square test or Fisher’s exact test. P values < .05 will be considered statistically significant.
Datasets
The intention-to-treat (ITT) analysis set will include all randomized patients, regardless of the treatment ultimately received.
The per-protocol (PP) analysis will include patients who received the treatment as assigned at randomization and met all inclusion criteria. Patients with major protocol deviations, including those who underwent crossover, will be excluded from this analysis.
The as-treated analysis set will include patients analyzed according to the treatment they actually received, rather than the treatment to which they were originally randomized.
Primary endpoint analysis
The primary endpoint of stent expansion at the CN site will be analyzed using an ITT approach, with results being expressed as mean and SD or median and IQR if the data deviate from normal distribution. In addition to the ITT analysis, the primary endpoint will also be assessed in the PP and as-treated sets.
Secondary endpoint analysis
The analysis of procedure-related secondary endpoints will be conducted initially in the ITT set, followed by assessments in the PP and as treated sets. The rate of clinical endpoints (target lesion revascularization, target lesion failure, major adverse cardiovascular events) will be compared between the 2 treatment groups using Cox proportional hazards regression, and event-free survival curves will be estimated using the Kaplan-Meier method. Differences in the primary endpoint between groups will be evaluated using adjusted hazard ratios with 95% confidence intervals and P values derived from Cox regression.
Hierarchical analysis using the win ratio method
Given that the trial includes multiple secondary endpoints that may influence the clinical course, an exploratory efficacy analysis comparing the 2 strategies will be performed using a hierarchical win-ratio method.
The first outcome in the hierarchy will be stent expansion at the CN. A “win” for one group will be defined as stent expansion at least 10 percentage points greater than that observed in the comparator patient. For this analysis, percent expansion will be truncated at a maximum of 100% prior to comparison. In the event of a tie, the procedure will proceed to the next predefined hierarchical endpoint, sequentially assessing procedural/strategy success, rate of major adverse cardiovascular events, stent malapposition at the CN, stent ellipticity, CN protrusion into the stent lumen, and minimum stent lumen area.
The overall win ratio will be estimated as the ratio of the total number of wins between the 2 treatment groups. Statistical significance will be assessed using the Finkelstein–Schoenfeld method with a 2-sided P < .05.
Analysis of predictors of adverse events
Predictors of suboptimal angioplasty outcomes will be evaluated using logistic regression models. Predictors of adverse clinical events during follow-up will be assessed using Cox proportional hazards regression models.
Adjusted analyses
Multivariable regression models will be used adjusted for CN type (eruptive vs noneruptive) and for any baseline or procedural differences observed between the groups.
Predefined subgroup analyses
Predefined subgroup analyses will include CN type (eruptive vs noneruptive), sex, vessel size (≥ 3.0 mm vs < 3.0 mm), and calcium length (≥ 15 mm vs < 15 mm).
Interim analysis
Once enrollment of all patients has been completed, an interim analysis will be performed. This analysis will focus on the primary endpoint (stent expansion at the CN site, assessed by OCT) as well as procedure-related secondary endpoints. The statistical analysis will be performed using STATA 18 (StataCorp, LLC, United States).
Ethical considerations
The ORBIT-SHOCK trial is conducted in full compliance with the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the central Clinical Research Ethics Committee of Hospital Universitario Ramón y Cajal (Madrid, Spain) and subsequently by the ethics committees of all other participant centers. All patients provide written informed consent before enrollment and randomization. Furthermore, the study was designed and is reported in accordance with the SAGER guidelines. Measures have been implemented to promote equitable participant inclusion and to minimize sex and gender bias. Sex-disaggregated data will be collected and analyzed to assess potential differences in procedural and clinical outcomes between men and women. The ORBIT-SHOCK pilot study is registered at ClinicalTrials.gov with the identifier NCT06736665.
DISCUSSION
Coronary calcification in the form of CN is consistently associated with worse outcomes due to the difficulty if achieving adequate calcium fracture and vessel expansion before stenting.
IVL has emerged as a commonly used strategy for the treatment of CN. However, although stent expansion > 100% is achieved in many patients, nodule fractures are observed in only a minority of cases, and nodule deformation occurs in less than half. As a result, stents expansion is often asymmetric, and residual nodule protrusion into the stent lumen is present in most patients. These findings could potentially increase the risk of future adverse events during follow-up.
The mechanism of action of OA may provide particular advantages for the treatment of CN. First, the rotational movement of the crown enables debulking of the nodule, while the orbital movement allows for circumferential modification of the vessel wall without being constrained by the guidewire position within the vessel.
Despite the theoretical advantages of its mechanism of action for treating CN, a recent observational study specifically comparing OA and IVL in these lesions did not demonstrate superior outcomes for OA.23 The study reported similar efficacy between the 2 techniques in terms of stent expansion (90.0% for OA vs 92.5% for IVL) and minimum lumen area (5.6 mm2 vs 5.5 mm2), as well as comparables rates of target lesion failure at 2 years (12.0% for OA vs 9.8% for IVL). Although the authors performed a propensity score-matched analysis, the sample size remained limited. In addition, given the retrospective design of the data, the presence of unmeasured confounding and significant differences in baseline lesion complexity are highly likely. In real-world clinical practice, atherectomy devices are preferentially used for uncrossable or more extensively calcified lesions, inherently introducing selection bias.
Moreover, in that study, the median number of OA runs was only 4 (IQR, 3-5) for a median calcium length of 25 mm, and the rotational speeds were not reported. This limited number of passes may be insufficient to achieve adequate lesion preparation and debulking of a CN, which often requires a higher number of runs and high rotational speeds.24 Only a randomized controlled design can ensure a homogeneous and balanced cohort, allowing an unbiased comparison of both strategies. Accordingly, the ORBIT-SHOCK pilot study will be the first randomized trial designed to directly compare OA and IVL for the treatment of CN, with all procedures performed under a strict, OCT-guided protocol and mandatory stent optimization.
Sample size determination and rationale for a randomized pilot study
Several studies have evaluated stent expansion after angioplasty using intracoronary imaging modalities. In the ILUMIEN II study, the minimum stent expansion in the OCT-guided group (calculated using the mean of the proximal and distal reference lumen areas) was 72.8% (IQR, 63.3 - 81.3).25 This corresponds to an estimated standard deviation (SD) of 13.3%, assuming a normal distribution. In the ILUMIEN IV trial, minimum stent expansion in the OCT-guided group was 80.8 ± 16.8%.9
Previous retrospective registries evaluating IVL or OA have reported wide SDs in stent expansion, ranging from 16.5% to > 27%. 1,3,4,17,20 A recent observational study comparing IVL and OA specifically for the treatment of CN reported SDs for stent expansion ranging from 13.0% (in eruptive nodules treated with IVL) to 25.9% (in noneruptive nodules treated with OA)12. This marked variability may be largely attributed to the heterogeneity of treated lesions, small sample sizes, heterogeneous definitions of the reference lumen area, or the lack of standardized imaging protocols. In contrast, the ORBIT-SHOCK pilot trial incorporates a strict OCT-guided PCI protocol with mandatory stent optimization. Under these controlled conditions, operator-dependent is expected to be substantially reduced. Consequently, an estimated SD of ± 15%, consistent with monitored trials such as ILUMIEN IV, was consiered the most appropriate assumption.
Regarding the clinically meaningful effect size, several pivotal trials, such as ULTIMATE,26 ILUMIEN III,27 and OPINION,28 have used a stent expansion threshold > 90% as the standard for optimal stent deployment, a target associated with significantly lower rates of target lesion failure during follow-up. Furthermore, the DOCTORS trial demonstrated that stent expansion < 79.4% predicts a fractional flow reserve < 0.90, suggesting a sub-optimal hemodynamic result. Accordingly, and acknowledging that achieving > 90% expansion may not be feasible in a significant proportion of complex lesions, the EAPCI expert consensus document proposes > 80% expansion as a pragmatic target for routine clinical practice.29 A 12% difference in stent expansion may therefore have important clinical implications, potentially shifting outcomes from an optimized result to a range associated with less functional outcomes.
Given the limited and heterogeneous data currently available, this pilot study was pragmatically sized to assess feasibility while maintaining sufficient exploratory power to detect a clinically relevant 12% difference in stent expansion. These findings will provide the essential methodological foundation and variance estimates necessary of future large-scale confirmatory trials.
Study limitations
Focusing on patients with CN (which account for approximately 20%–40% of severely calcified lesions) and the potential excluding of individuals with impaired renal function due to the contrast requirements of OCT may limit the eligible study population. These strict inclusion criteria could therefore result in a slower recruitment rate than initially anticipated. Nevertheless, participation of 6 centers, with an estimated enrollment rate of 1 patient per center per month, is expected to allow completion of patient recruitment within the planned timeframe.
The distinct behavior of eruptive and noneruptive CN, both in terms of stent expansion and the occurrence of adverse events during follow-up, could introduce bias if the distribution of these 2 subtypes is not balanced between treatment groups. To mitigate this risk, stratified randomization has been implemented to ensure a balanced allocation of both CN types across the treatment arm.
CN rarely occur in isolation and are frequently associated with extensive background coronary calcification. To address this, the study protocol specifically recommends applying the randomized technique to treat all concomitant calcified lesions within the target segment. This may include performing multiple OA runs or using more than 1 IVL balloon when necessary, with the explicit objective of achieving optimal lesion preparation and final results throughout the entire lesion length, not only at the nodule site. Although the primary endpoint focuses on stent expansion at the CN site, broader secondary clinical outcomes may be influenced by the overall calcific burden of the treated segment. Nevertheless, the randomized design of the study is expected to evenly distribute this anatomical complexity between the 2 treatment arms.
Finally, as a pilot clinical trial with a sample size determined primarily by feasibility, this study is not statistical powered to detect small differences between OA and IVL. However, the exploratory superiority framework is design to detect a clinically relevant effect size, such as a 12% difference in stent expansion. The fundamental purpose of this pilot study is to establish the methodological foundation and generate the preliminary data required to design future, adequately powered confirmatory trials.
CONCLUSIONS
The ORBIT-SHOCK pilot study will be the first randomized trial to allow a direct comparison of the outcomes of OA and IVL in the management of calcified CN, a common and complex clinical scenario that requires robust evidence from randomized clinical trials.
FUNDING
The study was funded by an unrestricted institutional research grant from Abbott (United States).
ETHICAL CONSIDERATIONS
The ORBIT-SHOCK trial is conducted in full compliance with the principles outlined in the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the central Clinical Research Ethics Committee of Hospital Universitario Ramón y Cajal (Madrid, Spain) and subsequently by the ethics committees of all other participant centers. All patients gave their written informed consent before enrollment and randomization. Furthermore, the study was designed and is being reported following the SAGER guidelines. We have implemented measures to ensure an equitable inclusion of participants and avoid sex/gender bias. Sex-disaggregated data will be collected and analyzed to evaluate potential differences in procedural and clinical outcomes between men and women.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the preparation of this manuscript.
AUTHORS’ CONTRIBUTIONS
L.M. Domínguez-Rodríguez: conceptualization, original draft, review, and editing. J. Bayón Lorenzo, I. Amat Santos, J. Martín Moreiras, J. Valencia Martín, M. Pan, L. Salido Tahoces, A. Pardo Sanz, J.L. Mestre-Barceló, A. Larrea-Iñarra, J.M. Ruiz-Nodar, A. Diego Nieto, C. González-Juanatey: drafting, review, and editing. J.L. Zamorano, and Á. Sánchez-Recalde: conceptualization, drafting, review, and editing. All authors have reviewed and approved the final version of this manuscript.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- CN are associated with suboptimal outcomes following percutaneous coronary intervention.
- IVL has become a widely adopted strategy for the treatment of CN due to its ease of use, low complication rates, and favorable outcomes reported in recent studies.
- OA has been shown to be an effective strategy for the treatment of severely calcified and nondilatable coronary lesions. However, evidence regarding its performance specifically in CN remains limited and is largely derived from small observational studies and case reports.
- Despite major advances in imaging and plaque-modification technologies, the optimal management strategy for CN has yet to be clearly defined.
WHAT DOES THIS STUDY ADD?
- The ORBIT-SHOCK pilot trial is designed as the first randomized clinical trial to compare the safety and efficacy profile of OA and IVL in the treatment of CN.
REFERENCES
1. Sato T, Matsumura M, Yamamoto K, et al. Impact of Eruptive vs Noneruptive Calcified Nodule Morphology on Acute and Long-Term Outcomes After Stenting. JACC Cardiovasc Interv. 2023;16:1024-1035.
2. Brott BC. The Calcified Nodule Paradox. JACC Cardiovasc Interv. 2023;16: 1036-1038.
3. McInerney A, Travieso A, Baza AJ, et al. Impact of coronary calcium morphology on intravascular lithotripsy. EuroIntervention. 2024;20:656-668.
4. Ali ZA, Kereiakes DJ, Hill JM, et al. Impact of Calcium Eccentricity on the Safety and Effectiveness of Coronary Intravascular Lithotripsy:Pooled Analysis From the Disrupt CAD Studies. Circ Cardiovasc Interv. 2023;16: E012898.
5. Prati F, Gatto L, Fabbiocchi F, et al. Clinical outcomes of calcified nodules detected by optical coherence tomography:a sub-analysis of the CLIMA study. EuroIntervention. 2020;16:380-386.
6. Akasaka T, Kubo T. OCT-derived coronary calcified nodules as a predictor of high-risk patients. EuroIntervention. 2020;16:361-363.
7. Ali ZA, Kereiakes D, Hill J, et al. Safety and Effectiveness of Coronary Intravascular Lithotripsy for Treatment of Calcified Nodules. JACC Cardiovasc Interv. 2023;16:1122-1124.
8. Lee JM, Choi KH, Song Y Bin, et al. Intravascular Imaging–Guided or Angiography-Guided Complex PCI. New Engl J Med. 2023;388:1668-1679.
9. Ali ZA, Landmesser U, Maehara A, et al. Optical Coherence Tomography–Guided versus Angiography-Guided PCI. New Engl J Med. 2023;389: 1466-1476.
10. Holm NR, Andreasen LN, Neghabat O, et al. OCT or Angiography Guidance for PCI in Complex Bifurcation Lesions. New Engl J Med. 2023; 389:1477-1487.
11. Vrints C, Andreotti F, Koskinas KC, et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J. 2024:3415-3537.
12. Dakroub AH, Shin D, Singh M, et al. Procedural and Clinical Outcomes After Orbital Atherectomy Versus Intravascular Lithotripsy in Patients With Calcified Nodules. Circ Cardiovasc Interv. 2025;18:015254.
13. Parikh K, Chandra P, Choksi N, Khanna P, Chambers J. Safety and feasibility of orbital atherectomy for the treatment of calcified coronary lesions:The ORBIT i Trial. Catheter Cardiovasc Interv. 2013;81:1134-1139.
14. Chambers JW, Feldman RL, Himmelstein SI, et al. Pivotal trial to evaluate the safety and efficacy of the orbital atherectomy system in treating de Novo, severely calcified coronary lesions (ORBIT II). JACC Cardiovasc Interv. 2014;7:510-518.
15. Florek K, Bartoszewska E, Biegała S, et al. Rotational Atherectomy, Orbital Atherectomy, and Intravascular Lithotripsy Comparison for Calcified Coronary Lesions. J Clin Med. 2023;12:7246.
16. Faria D, Vinhas H, Bispo J, et al. Initial experience with orbital atherectomy in a non-surgical center in Portugal. Rev Port Cardiol. 2024;43:645-728.
17. Okamoto N, Egami Y, Nohara H, et al. Direct Comparison of Rotational vs Orbital Atherectomy for Calcified Lesions Guided by Optical Coherence Tomography. JACC Cardiovasc Interv. 2023;16:2125-2136.
18. Yamamoto MH, Maehara A, Kim SS, et al. Effect of orbital atherectomy in calcified coronary artery lesions as assessed by optical coherence tomography. Catheter Cardiovasc Interv. 2019;93:1211-1218.
19. Yamamoto MH, Maehara A, Karimi Galougahi K, et al. Mechanisms of Orbital Versus Rotational Atherectomy Plaque Modification in Severely Calcified Lesions Assessed by Optical Coherence Tomography. JACC Cardiovasc Interv. 2017;10:2584-2586.
20. Okamoto N, Ueda H, Bhatheja S, et al. Procedural and one-year outcomes of patients treated with orbital and rotational atherectomy with mechanistic insights from optical coherence tomography. EuroIntervention. 2019;14:1760-1767.
21. Rola P, Włodarczak S, Barycki M, et al. Safety and Efficacy of Orbital Atherectomy in the All-Comer Population:Mid-Term Results of the Lower Silesian Orbital Atherectomy Registry (LOAR). J Clin Med. 2023;12:5842.
22. Chiang CSM, Alan Chan KC, Lee M, Chan KT. Orbital-Tripsy:Novel Combination of Orbital-Atherectomy and Intravascular-Lithotripsy, in Calcified Coronaries After Failed Intravascular-Lithotripsy. JACC Case Rep. 2020;2:2437-2444.
23. Shin D, Dakroub A, Singh M, et al. Debulking Effect of Orbital Atherectomy for Calcified Nodule Assessed by Optical Coherence Tomography. Circ Cardiovasc Interv. 2024;17:014145.
24. Riley RF, Patel MP, Abbott JD, et al. SCAI Expert Consensus Statement on the Management of Calcified Coronary Lesions. J Soc Cardiovasc Angiogr Interv. 2024;3:101259.
25. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of stent expansion guided by optical coherence tomography versus intravascular ultrasound:The ILUMIEN II study (Observational Study of Optical Coherence Tomography [OCT] in Patients Undergoing Fractional Flow Reserve [FFR] and Percutaneous Cor. JACC Cardiovasc Interv. 2015;8:1704-1714.
26. Zhang J, Gao X, Kan J, et al. Intravascular Ultrasound Versus Angiography-Guided Drug-Eluting Stent Implantation:The ULTIMATE Trial. J Am Coll Cardiol. 2018;72:3126-3137.
27. Ali ZA, Galougahi KK, Maehara A, et al. Outcomes of optical coherence tomography compared with intravascular ultrasound and with angiography to guide coronary stent implantation:One-year results from the ILUMIEN III:OPTIMIZE PCI trial. EuroIntervention. 2021;16:1085-1091.
28. Kubo T, Shinke T, Okamura T, et al. Optical frequency domain imaging vs intravascular ultrasound in percutaneous coronary intervention (OPINION trial):One-year angiographic and clinical results. Eur Heart J. 2017;38:3139-3147.
29. Räber L, Mintz GS, Koskinas KC, et al. Clinical use of intracoronary imaging. Part 1:Guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention. 2018;14:656-677.
ABSTRACT
Introduction and objectives: Drug-eluting stents (DES) represent the standard-of-care for treatment of de novo coronary artery lesions. However, permanent vessel metallic scaffolding is associated with short- and long-term complications. Drug-coated balloon (DCB) angioplasty, a scaffold-free alternative, may address these issues. This study aimed to compare the safety and efficacy profile of DCB vs DES for the treatment of de novo coronary lesions.
Methods: Electronic databases were interrogated for randomized controlled trials (RCT) comparing DCB with DES for the treatment of de novo lesions. The primary endpoint was target lesion revascularization, while secondary endpoints included target vessel revascularization, myocardial infarction, target vessel myocardial infarction, vessel thrombosis, cardiac death, all-cause mortality, major adverse cardiovascular events, and major bleeding. Effect estimates and 95% confidence intervals (95%CI) were calculated using random-effects models. Angiographic outcomes included binary restenosis, late lumen loss, minimal luminal diameter, net luminal gain, and diameter stenosis.
Results: A total of 13 RCT including 7776 patients were included. Sirolimus-coated DCB was tested in 1 RCT, while others assessed paclitaxel-coated DCB. Both groups showed comparable target lesion revascularization rates (RR, 1.24; 95%CI, 0.79-1.95; mean follow-up, 22.9 months) and similar results for all other ischemic endpoints. Major bleeding was significantly lower with DCB angioplasty vs DES (RR, 0.65; 95%CI, 0.43-0.96), an effect mainly observed in trials adopting shorter durations of dual antiplatelet therapy in the DCB group. These findings were consistent across multiple sensitivity and subgroup analyses. At angiographic follow-up (mean, ~8.4 months), DCB was associated with lower late lumen loss, whereas DES achieved lower diameter stenosis and greater minimal luminal diameter and net luminal gain, with no significant difference in binary restenosis.
Conclusions: DCB angioplasty appears as a viable therapeutic strategy for the treatment of de novo coronary lesions in contemporary clinical practice. However, large-scale randomized trials are warranted to confirm its long-term safety, efficacy, and optimal patient selection (PROSPERO: CRD42024587941).
Keywords: Drug coated balloon. Drug eluting stent. De novo coronary lesion. Percutaneous coronary intervention.
RESUMEN
Introducción y objetivos: Los stents farmacoactivos (SFA) representan el estándar de tratamiento para las lesiones coronarias de novo, aunque el implante permanente de una malla metálica puede asociarse a complicaciones a corto y largo plazo. La angioplastia con balón farmacoactivo (BFA), estrategia «sin andamiaje», podría superar estas limitaciones. El objetivo fue comparar la eficacia y la seguridad del BFA frente al SFA en lesiones coronarias de novo.
Métodos: Se realizaron búsquedas sistemáticas para identificar ensayos clínicos aleatorizados que compararan BFA y SFA en lesiones de novo. El objetivo primario fue la revascularización de la lesión diana; los objetivos secundarios incluyeron la revascularización del vaso diana, el infarto de miocardio, el infarto de miocardio del vaso diana, la trombosis, la muerte por causa cardiaca, la muerte por cualquier causa, los eventos cardiovasculares adversos mayores y el sangrado mayor. Las estimaciones del efecto y los intervalos de confianza del 95% (IC95%) se calcularon mediante modelos de efectos aleatorios. Los desenlaces angiográficos incluyeron reestenosis binaria, pérdida luminal tardía, diámetro luminal mínimo, ganancia luminal neta y estenosis del diámetro.
Resultados: Se incluyeron 13 ensayos clínicos aleatorizados (7.776 pacientes). Un estudio evaluó BFA con sirolimus y los restantes BFA con paclitaxel. No hubo diferencias significativas en la revascularización de la lesión diana (RR = 1,24; IC95%, 0,79-1,95; seguimiento medio 22,9 meses) ni en otros desenlaces isquémicos. El sangrado mayor fue menor con BFA (RR = 0,65; IC95%, 0,43-0,96), principalmente en los ensayos con menor duración de la doble antiagregación plaquetaria. En el seguimiento angiográfico (~8,4 meses), el BFA mostró menor pérdida luminal tardía, mientras que con el SFA se observaron menor estenosis del diámetro y mayores diámetros luminales mínimos y ganancia luminal neta, sin diferencias en la reestenosis binaria. Los resultados fueron consistentes en los análisis de sensibilidad y de subgrupos.
Conclusiones: El BFA parece una alternativa viable en las lesiones coronarias de novo, aunque se requieren ensayos de mayor tamaño para confirmar su seguridad, eficacia y adecuada selección de los pacientes a largo plazo (PROSPERO: CRD42024587941).
Palabras clave: Balón farmacoactivo. Stent farmacoactivo. Lesión coronaria de novo. Intervención coronaria percutánea.
Abbreviations
DCB: drug-coated balloon. DES: drug-eluting stent. MACE: major adverse cardiovascular events. PCI: percutaneous coronary intervention. RCT: randomized clinical trial.
INTRODUCTION
Percutaneous coronary intervention (PCI) with drug-eluting stents (DES) is the current standard of care for coronary revascularization in patients with coronary artery disease (CAD).1 Despite substantial advances in stent technology, the permanent presence of metal scaffolding presents a persistent hazard of stent-related adverse events (e.g.,, stent failure).2 These events occur in ~2% of patients per year and persist over time, thereby limiting long-term outcomes.3,4 Drug-coated balloons (DCB) have emerged as an attractive “leave-nothing-behind” alternative when an optimal angiographic result is achieved,5,6 delivering antiproliferative drugs directly into the arterial wall without leaving a permanent implant, potentially promoting arterial healing, preserving vessel anatomy, and avoiding stent-related complications.7,8 DCB have demonstrated efficacy in the treatment of in-stent restenosis,9,10 an accumulating evidence supports their use in selected settings, such as small-vessel disease (SVD) and bifurcation lesions.11-13 However, their role in de novo coronary lesions remains controversial. Randomized controlled trials (RCT) evaluating DCB have often been limited by small sample sizes, heterogeneous populations, variable procedural protocols, and reliance on surrogate angiographic endpoints.14 Notably, the 2 largest RCT published to date have yielded conflicting results, leaving the comparative efficacy of DCB vs DES unresolved.15-16
We conducted a systematic review and pairwise meta-analysis of RCT to assess the efficacy and safety profile of DCB vs DES for the treatment of de novo coronary lesions.
METHODS
This meta-analysis was prospectively registered in PROSPERO (CRD4202458794) and followed the PRISMA and Cochrane Collaboration guidelines (tables 1S and S2).17 Ethical approval was waived given the nature of the study.
Eligibility criteria
Studies were eligible if they: a) enrolled patients undergoing PCI for de novo coronary artery lesions of any vessel size; b) randomized participants to DCB angioplasty or DES implantation; and c) reported at least one outcome of interest. Exclusion criteria included: a) absence of a second- or third-generation DES as the primary comparator; and b) evaluation of DCB as part of a hybrid or combined strategy with concomitant stent implantation. No language restrictions were applied.
Search strategy, study selection, data extraction, and qualitative assessment
Three electronic databases (PubMed, Web of Science, and Scopus) were searched from inception to 29 October 2025 (table S1). Additional sources included society websites, conference proceedings, and reference lists of eligible studies. Two reviewers (R. Rinaldi and K. Bujak) independently screened titles and abstracts, assessed full texts for final inclusion, and extracted trial- and arm-level data into standardized spreadsheets. Disagreements were resolved by consensus. When multiple reports referred to the same trial, the one with the longest follow-up was selected. Study quality was assessed using the Cochrane Risk of Bias 2 tool.18 Publication bias was evaluated using funnel plots and Egger’s test, with adjustment using the trim-and-fill method for endpoints suspected of publication bias.
Study endpoints
The prespecified primary endpoint was trial-defined target lesion revascularization at the longest follow-up. When target lesion revascularization was not reported, target vessel revascularization was used as the closest available reported revascularization endpoint. Secondary endpoints included target vessel revascularization, myocardial infarction (MI), target-vessel MI, vessel thrombosis, cardiac and all-cause mortality, major adverse cardiovascular events (MACE), and major bleeding. When multiple definitions of the same endpoint were reported within a trial, we extracted the definition most closely aligned with standardized Academic Research Consortium criteria (eg, clinically driven target lesion revascularization).19,20 Angiographic endpoints included binary restenosis, late lumen loss (postoperative minus follow-up minimal luminal diameter), minimal luminal diameter, diameter stenosis, and net luminal gain (follow-up minus preoperative minimal luminal diameter).
Statistical analysis
Analyses were performed on an intention-to-treat (ITT) basis. Pairwise meta-analyses were conducted using a frequentist random- effects models (DerSimonian-Laird estimator) to account for anticipated biological variability. Binary outcomes were expressed as risk ratios (RR) with 95% confidence interval (95%CI), while continuous outcomes were reported as standardized mean differences (SMD). A continuity correction of 0.5 was applied when 0 events occurred in either study group. Heterogeneity was quantified using the Cochran Q test and I² statistic, interpreted as low (< 25%), moderate (25-50%), or high (> 50%). Baujat plots were used to identify influential studies. Statistical significance was defined as two-sided P < .05.
Prespecified subgroup analyses were performed by stratifying the trials according to study-level characteristics, including: a) drug type (paclitaxel vs sirolimus); b) lesion characteristics (SVD < 3.0 mm; large vessels; bifurcation; ST-segment elevation myocardial infarction culprit lesion); c) dual antiplatelet therapy (DAPT) duration (shorter in DCB group vs equal duration in both groups). Meta- regression analyses were conducted to assess the impact of confounding variables, including a) mean study-level reference vessel diameter; b) follow-up duration; and c) year of publication. Sensi- tivity analyses were performed for both the primary and secondary outcomes using alternative modeling approaches (eg, incidence rate ratios; trim and fill analysis, Bayesian random-effects meta-analysis with weakly informative priors), and by excluding studies: a) at high risk of bias or without ITT analysis; b) using paclitaxel-eluting stents as comparators; c) using ultrasound-guided DCB; d) reporting target vessel revascularization instead of target lesion revascularization or non-Academic Research Consortium primary endpoint definitions (eg, definitions other than clinically driven target lesion revascularization); e) using DCB devices other than most commonly tested one in included trials (SeQuent Please); or f) identified as principal contributors to heterogeneity. All analyses were conducted using R version 4.2.0 (R Foundation for Statistical Computing, Austria).
RESULTS
Study selection, baseline, and angiographic characteristics
The study selection flowchart is reported in figure S1. After screening, 13 RCT met the inclusion criteria and were included in the quantitative synthesis.11,12,15,16,21-29 Among these, 12 trials had already been published,11,12,15,21-29 and one was available only as a conference proceeding.16 Key trials characteristics are summarized in table 1. A total of 5 studies focused exclusively on SVD,11,12,25,27,28 4 on de novo lesions of any vessel size,15,16,23,29 3 on ST-segment elevation myocardial infarction,26,30,31 and 1 on bifurcation lesions.24 One trial evaluated a sirolimus-coated balloon,16 while the remaining 12 tested paclitaxel-coated balloons.11,12,15,21-29 The weighted mean clinical follow-up was 22.9 months, and the weighted mean angiographic follow-up (reported in 10 studies), 8.4 months. Available endpoints and definitions across included trials are reported in tables S2 and S3, respectively. Overall, 7776 patients were included (3896 DCB vs 3880 DES), of whom 46.0% presented with acute coronary syndrome. The weighted mean reference vessel diameter was 2.95 mm. The weighted mean minimum anticipated DAPT duration was 5.0 months in the DCB group and 5.9 months in the DES group (table 1). Baseline clinical, angiographic, and procedural characteristics are presented in tables S4 and S5. The risk of bias was high in 3 studies (imprecise reporting or deviation from the ITT analysis) and moderate in remaining studies due to the open-label design (figure S2). Publication bias was detected only for late lumen loss and minimal luminal diameter endpoints (figure S3).
Table 1. Key baseline characteristics across included randomized controlled trials
| Study, year | Sample size | Primary endpoint | DCB type | DES type | Type of de novo target lesion | DAPT | Follow-up, month* |
|---|---|---|---|---|---|---|---|
| SELUTION DeNovo, 202516 | 3323 | Composite of cardiac death, TVMI, and clinically driven target lesion revascularization | SELUTION SLR (sirolimus) | DES according to local practice | RVD ≥ 2.0 and ≤ 5.0 mm | 6 (CCS), 12 (ACS) / 6 (CCS), 12 (ACS) | Clinical: 12 |
| REC-CAGEFREE I, 202515 | 2272 | Composite of CV death, TVMI, and clinically driven and protocol driven target lesion revascularization | Swide (paclitaxel) | DP-SES (Firebird 2) | Noncomplex coronary artery disease (irrespective of RVD) | 1 / 1 (A+C or A+T followed by A or C monotherapy; the decision on DAPT > 1 month was left to the treating physician, and the overall DAPT duration was shorter in the DCB group on average) | Clinical: 36 |
| Dissolve SVD, 202427 | 247 | In-segment percent diameter stenosis | Dissolve (paclitaxel) | DP-ZES (Resolute) | RVD ≥ 2.25 and ≤ 2.75 mm | 3 / 6 (A+C or A+T followed by A monotherapy) | Angiographic: 9 Clinical: 12 |
| REVELATION, 202422 | 120 | Functional assessment using FFR | Pantera Lux (paclitaxel) | DP-SES (Orsiro); DP-EES (Xience) | – RVD: 2.5-4 mm – No severe calcification – Percent diameter stenosis < 50% after thrombus aspiration and predilatation. | 9 / 9 (A+T or A+P) | Angiographic: 9 Clinical: 60 |
| PICCOLETTO-II, 202328 | 232 | In-lesion late lumen loss | Elutax SV/Emperor (paclitaxel) | DP-EES (Xience) | RVD ≥ 2.00 and ≤ 2.75 mm, no LM or ostial lesions, no CTO, no severe calcification or tortuosity, lesion length < 25 mm | 1 / 6 | Angiographic: 6 Clinical: 36 |
| Ke et al., 202324 | 60 | 1-year late lumen loss and cumulative MACE (composite of CV death, TVMI, target lesion thrombosis, or target lesion revascularization/target vessel revascularization. | SeQuent Please (paclitaxel) | DP-ZES (Resolute); DP-EES (Xience); DP-SES (Firebird 2 and Excel) | True BL (Medina 1,1,1; 0,1,1; 1,0,1) and SB ≥ 2.25 mm without severe calcification or tortuosity | 12 / 12 (A+C or A+T followed by A, C or T monotherapy) | Angiographic and clinical: 12 |
| Kawai et al., 202225 | 42 | Endothelial function adjacent to the treated segment by a vasomotion test | SeQuent Please (paclitaxel) | DP-EES (Synergy) | RVD ≥ 2.0 and ≤ 3.0 mm, no LM or ostial lesion, no severe calcification | 3 / 6 (A+C) | Angiographic: 8 |
| Wang et al., 202221 | 184 | In-segment late lumen loss | Vasoguard (paclitaxel) | BP-SES (Cordimax) | RVD ≥ 2.0 and ≤ 4.0 mm, percent diameter stenosis ≥ 50% and successful predilatation | 3 / 12 (A+C) | Angiographic: 9 Clinical: 12 |
| Yu et al., 202229 | 170 | Late lumen loss | SeQuent Please (paclitaxel) | DP-ZES (Resolute); DP-EES (Synergy); BP-DES (Rapamycin, Firehawk) | RVD ≥ 2.25 and ≤ 4.0 mm and lesion length ≤ 30 mm | 1-3 (CCS), 6-12 (ACS) / 12 (A+C or A+T) | Angiographic: 9 Clinical: 12 |
| BASKET-SMALL 2, 202012 | 758 | MACE (composite of CV death, non-fatal MI, and target vessel revascularization), all-cause mortality, probable or definite stent thrombosis, and major bleeding | SeQuent Please (paclitaxel) | DP-EES (Xience); DP-DES (Paclitaxel, Taxus Element) | RVD ≥ 2.0 to < 3.0 mm and successful predilatation | 1 (CCS), 12 (ACS) / 6 (CCS), 12 (ACS) (A+C or A+T or A+P) | Clinical: 36 |
| RESTORE SVD China, 202011 | 230 | In-segment percent diameter stenosis | Restore (paclitaxel) | DP-ZES (Resolute) | RVD ≥ 2.25 and ≤ 2.75 mm | 6 / 6 (A+C or A+T) | Angiographic: 9 Clinical: 24 |
| Gobic´ et al., 201726 | 75 | MACE (composite of CV death, reinfarction, target lesion revascularization and stent thrombosis) and late lumen loss | SeQuent Please (paclitaxel) | BP-DES (Sirolimus, Biomime) | RVD ≥ 2.5 and ≤ 4.0 mm, without severe tortuosity | 12 / 12 (A+C) | Angiographic and clinical: 6 |
| Nishiyama et al., 201623 | 60 | Target lesion revascularization and late lumen loss | SeQuent Please (paclitaxel) | DP-EES (Xience) | Significant stenosis, successful predilatation, and lesion length ≤ 25 mm | 8 / 8 (A+C) | Angiographic and clinical: 8 |
|
A, aspirin; ACS, acute coronary syndrome; ARC, Academic Research Consortium; BL, bifurcation lesions; BP, biodegradable polymer; CABG, coronary artery bypass graft; CCS, chronic coronary syndrome; CS, cardiogenic shock; CTO, chronic total coronary occlusion; CV, cardiovascular; DAPT, dual antiplatelet therapy; DES, drug eluting stent; DoCE, device-oriented composite endpoint; DP, durable polymer; DS, diameter stenosis; EES, sirolimus eluting stent; eGFR, estimated glomerular filtration rate; FFR, fractional flow reserve; ISR, in-stent restenosis; IVUS, intravascular ultrasound; LM, left main; LVEF, left ventricular ejection fraction; MACE, major adverse cardiovascular events; NSTEMI, non-ST-segment elevation myocardial infarction; P, prasugrel; PCI, percutaneous coronary intervention; pPCI, primary percutaneous coronary intervention; RVD, reference vessel diameter; SB, side branch; SES, sirolimus eluting stent; STEMI, ST-segment elevation myocardial infarction; T, ticagrelor; TVMI, target vessel myocardial infarction; VHD, valvular heart disease; ZES, zotarolimus eluting stent. * Follow-up refers to the maximum available. |
|||||||
Primary endpoint
No statistically significant difference was observed between DCB and DES for target lesion revascularization (RR, 1.24; 95%CI, 0.79-1.95; figure 1), with high heterogeneity (I² = 54.8%).
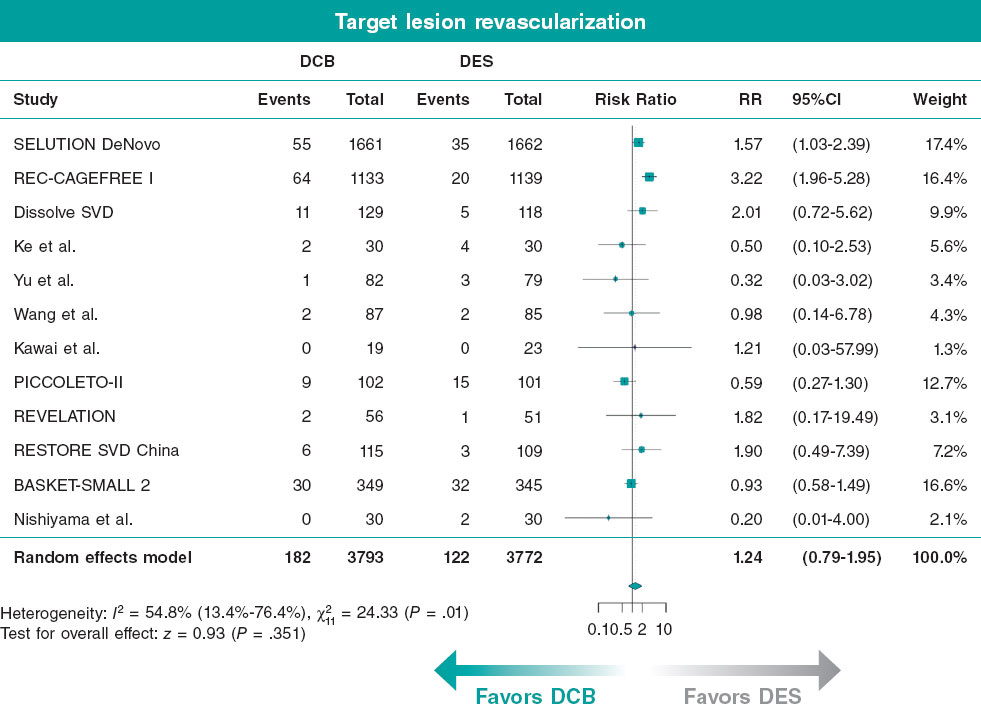
Figure 1. Results of meta-analysis for the primary endpoint. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; RR, risk ratio. The bibliographical references cited in this figure correspond to: Spaulding,16 Gao et al.,15 Liu et al.,27 Ke et al.,24 Yu et al.,29 Wang et al.,21 Kawai et al.,25 Cortese et al.,28 Niehe et al.,22 Tian et al.,11 Jeger et al.,12 and Nishiyama et al.23.
Secondary endpoints
No statistically significant differences were observed between DCB angioplasty and DES implantation for target vessel revascularization (RR, 1.39; 95%CI, 0.91-2.13), MI (RR, 0.92; 95%CI, 0.71-1.19), target-vessel MI (RR, 1.09; 95%CI, 0.78-1.51), cardiac death (RR, 1.05; 95%CI, 0.75-1.48), all-cause mortality (RR, 1.05; 95%CI, 0.82-1.36), or MACE (RR, 1.05; 95%CI, 0.78-1.41) (figure 2 and figure S4A-G). DCB angioplasty was associated with a significantly lower risk of major bleeding vs DES implantation (RR, 0.65; 95%CI, 0.43-0.96), primarily in studies adopting shorter DAPT regimens for DCB-treated patients (figure 2 and figure S4H). However, the interaction test between treatment effect and DAPT duration was not statistically significant (figure S5I). Heterogeneity was low to moderate across endpoints, except for target vessel revascularization, which showed high heterogeneity (I2 = 55.2%).
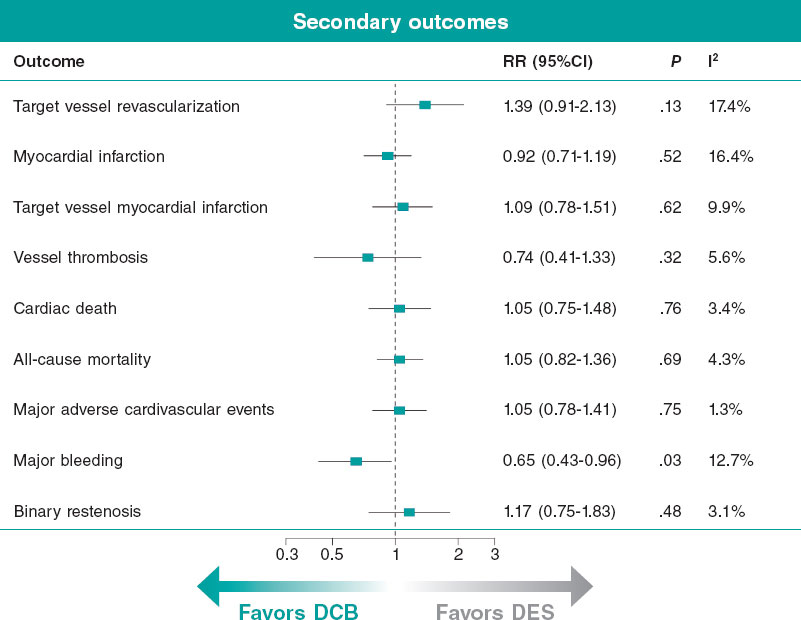
Figure 2. Forest plot presenting pooled effect estimates for the secondary clinical outcomes and binary restenosis. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; RR, risk ratio.
Angiographic endpoints
At angiographic follow-up, binary restenosis did not differ between groups (RR, 1.17; 95%CI, 0.75-1.83) (figure 2 and figure S4I). DES implantation was associated with significantly higher late lumen loss (SMD, −0.41; 95%CI, −0.61 to −0.21), minimal luminal diameter (SMD, -0.50, 95%CI −0.64 to −0.37) and net luminal gain (SMD, −0.61; 95%CI, −0.77 to −0.44) vs DCB angioplasty, whereas DCB angioplasty was associated with greater diameter stenosis vs DES (SMD, 0.32; 95%CI, 0.20 to −0.44) (figure 3; figure S6). Heterogeneity was low for most angiographic outcomes, except for late lumen loss, for which heterogeneity was high.
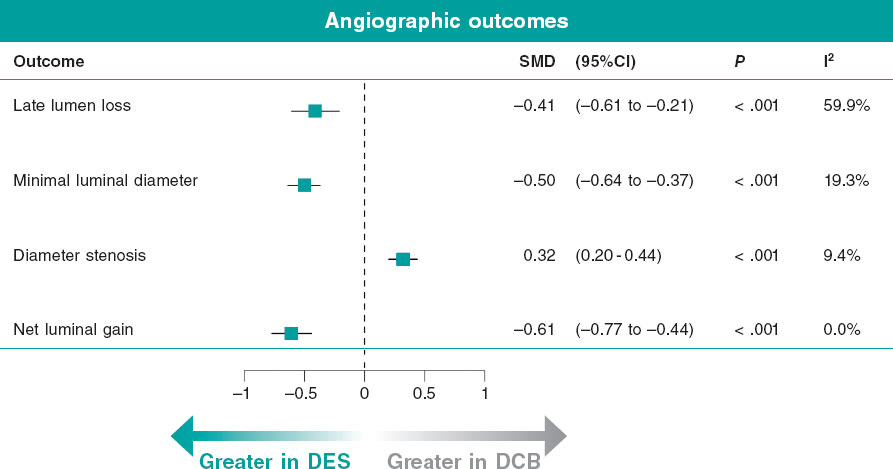
Figure 3. Forest plot presenting pooled effect estimates for angiographic outcomes. 95%CI, 95% confidence interval; DCB, drug-coated balloon; DES, drug-eluting stent; SMD, standardized mean differences.
Sensitivity and subgroup analyses
Subgroup analyses by DAPT duration showed no significant differences between DCB and DES for primary or secondary ischemic endpoints (figure S5A-H). Subgroup analyses stratified by clinical indication and DCB drug type (paclitaxel- vs sirolimus-coated balloons) also showed no significant interactions (figures S7,S8).
Meta-regression analyses showed consistent treatment effects across reference vessel diameter and follow-up duration (figure S9A,B). Meta-regression according to publication year identified an increasing risk of target lesion revascularization for DCB in more recent trials (figure S9C).
Sensitivity analyses confirmed the results of the primary analyses (figures S10-S15). After excluding the trial using paclitaxel-eluting stent in one-third of control patients, DCB was associated with a significantly higher risk of target vessel revascularization, with no other significant differences observed (figure S16). Bayesian sensitivity analyses were largely consistent with the frequentist results across endpoints. For major bleeding, the treatment effect remained directionally consistent but did not reach statistical significance (figure S17).
Baujat plots identified REC-CAGEFREE I trial15 as the principal source of heterogeneity for target lesion revascularization and target vessel revascularization, and the study by Gobic´ et al.26 for late lumen loss (figure S18). Excluding these studies yielded consistent pooled estimates with low heterogeneity (figure S19).
DISCUSSION
The main findings of our meta-analysis can be summarized as follows: a) DCB angioplasty for de novo lesions showed a comparable risk of target lesion revascularization to DES at a mean follow-up of 2 years; b) no increased risk of MI, target vessel revascularization, cardiac or all-cause mortality was observed with DCB vs DES; c) DCB angioplasty was associated with a significantly lower risk of major bleeding vs DES, although the interaction according to DAPT duration was not statistically significant; and d) from an angiographic perspective, DCB was associated with lower late lumen loss, whereas DES achieved larger minimal luminal diameter and net luminal gain, reflecting the mechanical scaffolding effect (figure 4).
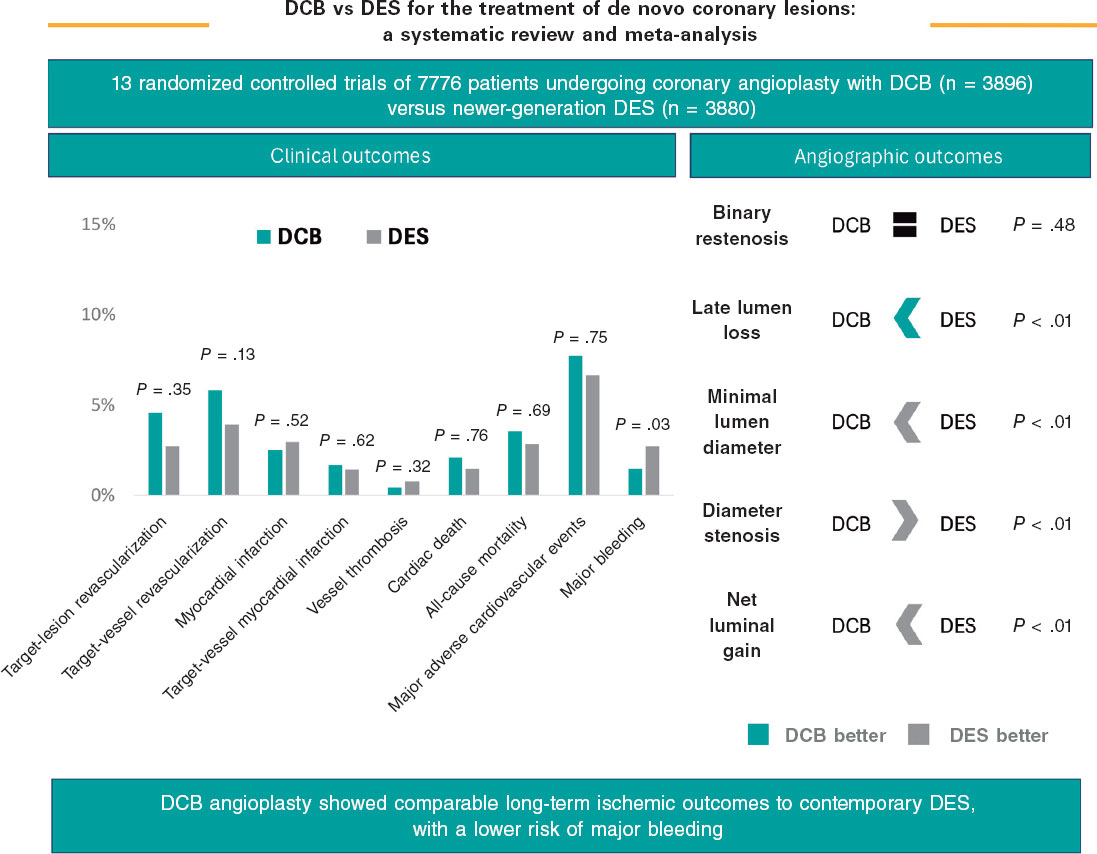
Figure 4. Central illustration. Main results of the study. DCB, drug-coated balloon; DES, drug-eluting stent.
In recent years, the “leave-nothing-behind” concept has gained increasing attention as a strategy to reduce stent-related adverse events and promote physiological vessel healing.32 However, evidence for DCB use in de novo CAD remains limited and heterogeneous.14 Previous meta-analyses generally reported comparable outcomes between DCB and DES, but were frequently restricted to specific subgroups, such as SVD,33,34 large vessels,35,36 or acute MI,37 or included observational data or early-generation DES comparators.38,39 The recent ANDROMEDA individual-patient-data meta-analysis suggested a possible reduction in MACE with DCB in SVD, although its interpretation was limited by the inclusion of only a few RCT and the use of early-generation stents as the comparator group.40
In contrast, our meta-analysis, incorporating 2 large recent RCT,15,16 provides the most contemporary and comprehensive evidence to date. By encompassing a broad spectrum of vessel sizes, lesion complexities, and clinical presentations, it demonstrates the consistent efficacy of DCB angioplasty across diverse clinical settings. Compared with the recent meta-analysis by Niu et al.,41 the present analysis incorporates nearly twice the number of randomized clinical trials and > 4 times the number of patients, including 2 large contemporary trials not previously analysed. In addition, our study focuses exclusively on newer-generation DES and includes bleeding outcomes, dual antiplatelet therapy stratification, meta-regression, and Bayesian sensitivity analyses. Rates of target lesion revascularization were equivalent between DCB and new-generation DES, both at early and extended follow-up, confirming the sustained durability of the DCB strategy over time. These findings suggest that the absence of a permanent metallic scaffold does not compromise vessel patency at 2-year follow-up or predispose to late restenosis, thereby supporting the role of DCB as a valid and reliable alternative to DES for the treatment of de novo coronary lesions.32
Furthermore, our findings help to contextualize the results of the REC-CAGEFREE I trial, which failed to demonstrate the non-inferiority of DCB vs DES in non-complex, de novo coronary lesions.15 Indeed, REC-CAGEFREE I represented the main source of heterogeneity in our analysis, and its exclusion yielded results consistent with the overall pooled effect, markedly reducing heterogeneity. Several methodological aspects may explain this discrepancy. First, unlike most previous RCT that focused on specific subgroups (eg, SVD or ST-egment elevation myocardial infarction), REC-CAGEFREE I adopted an all-comer design and used a device-oriented composite endpoint as its primary outcome. Second, extensive lesion preparation with cutting or scoring balloons in > 60% of cases may have contributed to improved outcomes in the DES group by facilitating stent expansion and reducing restenosis, while also enhancing drug delivery and procedural success in the DCB group, thereby potentially attenuating differences between treatment strategies.42,43 Third, approximately 10% of patients in the DCB group required bailout stenting for significant dissections or residual percent diameter stenosis > 30%, yet were still analysed within the DCB group in both ITT and per-protocol analyses. This methodological choice may have biased results against DCB because these patients typically have higher risks of repeat revascularization due to complex lesions, suboptimal angioplasty results, or residual dissections.44 Importantly, it was not specified whether adverse events occurred in patients treated exclusively with DCB or in those receiving bailout DES, potentially conflating outcomes driven by stented segments. Conversely, in trials such as SELUTION De Novo, where provisional stenting was incorporated within the DCB strategy, the inclusion of DES in the experimental group may have diluted potential treatment differences under ITT analysis, thereby limiting the ability to detect superiority of systematic stent implantation. Finally, the open-label design could have introduced surveillance bias, with a potentially lower threshold for ischemia testing and repeat angiography in the DCB group. Notably, event curves began to diverge around 100 days, coinciding with the transition from telephone to in-person follow-up.44
Furthermore, nearly all trials included in this meta-analysis evaluated paclitaxel-coated DCB, except for the SELUTION DeNovo trial,16 which tested a sirolimus-coated balloon in a large, all-comer population. The consistent findings across studies, irrespective of the antiproliferative agent used, suggest a class effect of DCB in terms of safety and efficacy when compared with contemporary DES. Nonetheless, further head-to-head studies directly comparing paclitaxel- and sirolimus-coated balloons are warranted.
The angiographic findings provide mechanistic insights into the clinical results. While binary restenosis rates were similar, DES achieved lower diameter stenosis and greater minimal luminal diameter and net luminal gain, consistent with its mechanical scaffolding properties. In contrast, DCB demonstrated lower late lumen loss. However, the smaller immediate postoperative lumen with DCB angioplasty may partially account for the lower late lumen loss observed at follow-up. Nevertheless, this difference may also reflect the absence of a permanent metallic implant, potentially more physiological vessel remodelling, and differences in local drug delivery dynamics and antiproliferative efficacy at the treated segment.45 Thus, DES may provide an immediate luminal advantage, as reflected by the greater net luminal gain, which represents a more integrative angiographic parameter less influenced by differences in acute luminal gain. This advantage may be particularly beneficial in complex, calcified, or large-vessel disease. Conversely, DCB may be preferable when vessel compliance and healing are prioritized, such as in SVD, bifurcations, or in younger patients in whom long-term metal avoidance may be advantageous.46 These complementary characteristics suggest an individualized approach that balances the need for mechanical support with the benefits of a “leave-nothing-behind” strategy.
Finally, we observed a lower incidence rate of major bleeding with DCB angioplasty vs DES implantation. This difference was primarily observed in those studies adopting shorter DAPT regimens for DCB-treated patients, suggesting that abbreviated antiplatelet strategies may be safely feasible with DCB and may make this approach a valuable option for patients at high bleeding risk.46-48 However, these findings derive from subgroup analyses stratified according to DAPT regimens specified in the trial protocols rather than the actual DAPT durations received by patients and should therefore be interpreted with caution. Ongoing trials such as the DEBATE (NCT04814212) and the PICCOLETTO IV (NCT06535568) are expected to provide further insights on this issue, particularly in anticoagulated or high-bleeding-risk populations.
Given differences in costs and lack of clinical superiority of DCB vs DES at either short- or long-term follow-up, the value of DCB angioplasty will ultimately depend on identifying specific patient populations who may derive the greatest benefit from a “leave-nothing-behind” strategy, as well as on the results of longer-term studies. Importantly, our findings should be interpreted as evidence of comparable efficacy rather than superiority, and do not support the routine replacement of DES in unselected patients.
Limitations
The present meta-analysis has several limitations. First, a certain degree of heterogeneity was observed across the included randomized trials, largely driven by the REC-CAGEFREE I trial. Furthermore, all included randomized trials enrolled patients in whom both treatment strategies were considered technically feasible and clinically appropriate. Therefore, the study populations reflect selected procedural scenarios rather than unselected, real-world practice. Second, as this was a study-level meta-analysis, our analyses were limited to aggregate endpoints reported in the original publications. Definitions of major bleeding varied substantially across trials, and this heterogeneity may limit the interpretability and comparability of the observed bleeding signal. Third, an analysis stratified by clinical presentation was not feasible because several RCT either did not report event rates according to presentation or excluded one of these subgroups entirely. Fourth, the open-label design of all included studies may have introduced performance or ascertainment bias. Fifth, although nearly all DCB used in the included studies were paclitaxel-coated, DES comparators consisted of a mixture of “limus”-eluting and paclitaxel-eluting stents, which could have influenced relative efficacy estimates in favor of certain DES platforms. Sixth, potential publication bias was detected for angiographic endpoints, possibly reflecting selective reporting of angiographic follow-up data. Although trim-and-fill analyses yielded estimates consistent with the primary results, these surrogate endpoints should be interpreted with caution. Furthermore, as with any meta-analysis based predominantly on published data, the possibility of publication bias cannot be entirely excluded. Seventh, data from the conference proceedings reporting the results of the recent SELUTION De Novo trial,16 whose full-text publication is not yet available, were included in the quantitative synthesis to incorporate all available evidence. Finally, differences in follow-up duration across studies may have limited the ability to capture late adverse events or long-term efficacy trends, highlighting the need for extended follow-up data from ongoing large-scale RCT.
CONCLUSIONS
This meta-analysis indicates that DCB angioplasty may achieve clinical outcomes comparable to those of newer-generation DES for treatment of de novo coronary lesions, without evidence of ischemic superiority. A reduction in major bleeding was observed in studies adopting shorter dual antiplatelet therapy regimens, although this finding should be interpreted with caution. Overall, these findings may support the integration of DCB into routine daily clinical practice as a viable therapeutic strategy in selected clinical scenarios. However, large-scale randomized trials are still warranted to confirm their long- term safety and efficacy profile, as well as their potential superiority over DES.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
Ethical approval was waived given the nature of the study.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
None declared.
AUTHORS’ CONTRIBUTIONS
R. Rinaldi, K. Bujak, G. Occhipinti, and C. Laudani contributed to conceptualization and design of the study, methodology, investigation, data curation, formal analysis, and participated in the drafting and review of the manuscript. S. Brugaletta contributed to the conceptualization and design of the study, methodology, investigation, data curation, project administration, supervision, and drafting, including critical review and editing of the manuscript. J. Sanz Sánchez, R.A. Montone, E. Nicolini, T. Piva, M. Gąsior, F. Ottani, F. Crea, S. Eccleshall, and M. Sabaté, contributed to drafting, review and editing of the manuscript. All authors have reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
CONFLICTS OF INTEREST
F. Crea reports receiving speaker fees from Amgen, AstraZeneca, Abbott, Menarini, Chiesi, and Daiichi Sankyo. M. Sabaté reports receiving consultant fees from Abbott Vascular and iVascular. Other authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- DCB represent a “leave-nothing-behind” strategy designed to avoid long-term stent-related complications and promote natural vessel healing.
- Previous evidence supported the use of DCB mainly for in-stent restenosis and SVD, while data on de novo coronary lesions remain limited and heterogeneous.
- Prior meta-analyses were often constrained by small sample sizes, selective populations, and comparisons with outdated DES.
WHAT DOES THIS STUDY ADD?
- This comprehensive meta-analysis of 13 randomized trials demonstrates a comparable long-term safety and efficacy profile between DCB angioplasty and contemporary DES for de novo coronary lesions.
- DCB angioplasty was associated with a lower risk of major bleeding, particularly in studies using shorter regimens of dual antiplatelet therapy, without compromising ischemic outcomes.
- Findings support DCB as a valid alternative to DES in selected patients, reinforcing the potential of a “leave- nothing-behind” revascularization strategy in modern PCI practice.
REFERENCES
1. Stefanini GG, Byrne RA, Windecker S, Kastrati A. State of the art:coronary artery stents - past, present and future. EuroIntervention. 2017;13:706-716.
2. Serruys PW, Garcia-Garcia HM, Onuma Y. From metallic cages to transient bioresorbable scaffolds: change in paradigm of coronary revascularization in the upcoming decade?Eur Heart J. 2012;33:16-25.
3. Madhavan M V., Kirtane AJ, Redfors B, et al. Stent-Related Adverse Events >1 Year After Percutaneous Coronary Intervention. J Am Coll Cardiol. 2020;75:590-604.
4. Rodriguez F, Harrington RA. Management of Antithrombotic Therapy after Acute Coronary Syndromes. N Engl J Med. 2021;384:452-460.
5. Jeger R V., Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease: Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13:1391-1402.
6. Yerasi C, Case BC, Forrestal BJ, et al. Drug-Coated Balloon for De Novo Coronary Artery Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:1061-1073.
7. Camaj A, Leone PP, Colombo A, et al. Drug-Coated Balloons for the Treatment of Coronary Artery Disease: A Review. JAMA Cardiol. 2025;10:189-198.
8. Colombo A, Leone PP. Redefining the way to perform percutaneous coronary intervention: a view in search of evidence. Eur Heart J. 2023;44:4321-4323.
9. Giacoppo D, Alfonso F, Xu B, et al. Drug-Coated Balloon Angioplasty Versus Drug-Eluting Stent Implantation in Patients With Coronary Stent Restenosis. J Am Coll Cardiol. 2020;75:2664-2678.
10. Giacoppo D, Alfonso F, Xu B, et al. Paclitaxel-coated balloon angioplasty vs drug-eluting stenting for the treatment of coronary in-stent restenosis: a comprehensive, collaborative, individual patient data meta-analysis of 10 randomized clinical trials (DAEDALUS study). Eur Heart J. 2020;41:3715-3728.
11. Tian J, Tang Y da, Qiao S, et al. Two-year follow-up of a randomized multicenter study comparing a drug-coated balloon with a drug-eluting stent in native small coronary vessels: The RESTORE Small Vessel Disease China trial. Catheter Cardiovasc Interv. 2020;95 Suppl 1:587-597.
12. Jeger R V., Farah A, Ohlow MA, et al. Long-term efficacy and safety of drug-coated balloons versus drug-eluting stents for small coronary artery disease (BASKET-SMALL 2): 3-year follow-up of a randomised, non-inferiority trial. Lancet. 2020;396:1504-1510.
13. Fezzi S, Scheller B, Rissanen TT, et al. Drug-coated balloons for coronary bifurcation lesions. EuroIntervention. 2025;21:e1177-e1197.
14. O'Callaghan D, Rai H, Giacoppo D, et al. Drug Coated Balloons Versus Drug-Eluting Stents in Patients With De Novo Coronary Artery Disease. Catheter Cardiovasc Interv. 2025;106:1843-1853.
15. Gao C, He X, Ouyang F, et al. Drug-coated balloon angioplasty with rescue stenting versus intended stenting for the treatment of patients with de novo coronary artery lesions (REC-CAGEFREE I):an open-label, randomised, non-inferiority trial. Lancet. 2024;404:1040-1050.
16. Spaulding CM. One-Year Results of the SELUTION DeNovo Trial Comparing a Strategy of PCI with a Sirolimus-eluting Balloon and Provisional Stenting Versus Systematic DES Implantation to Treat DeNovo Coronary Lesions |tctmd.com. Available at:https://www.tctmd.com/slide/one-year-results-selution-denovo-trial-comparing-strategy-pci-sirolimus-eluting-balloon-and. Accessed 5 Nov 2025.
17. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement:an updated guideline for reporting systematic reviews. BMJ. 2021;372.
18. Sterne JAC, Savovic´J, Page MJ, et al. RoB 2:a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366.
19. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials: The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
20. Fezzi S, Scheller B, Cortese B, et al. Definitions and standardized endpoints for the use of drug-coated balloon in coronary artery disease: consensus document of the Drug Coated Balloon Academic Research Consortium. Eur Heart J. 2025;46:2498-2519.
21. Wang Z, Yin Y, Li J, et al. New Ultrasound-Controlled Paclitaxel Releasing Balloon vs Asymmetric Drug-Eluting Stent in Primary ST-Segment Elevation Myocardial Infarction - A Prospective Randomized Trial. Circ J. 2022;86:642-650.
22. Niehe SR, Vos NS, Van Der Schaaf RJ, et al. 5-Year Clinical Outcomes of Paclitaxel-Coated Balloon Angioplasty vs DES in Acute MI: The REVELATION Trial. JACC Cardiovasc Interv. 2024;17:1185-1186.
23. Nishiyama N, Komatsu T, Kuroyanagi T, et al. Clinical value of drug-coated balloon angioplasty for de novo lesions in patients with coronary artery disease. Int J Cardiol. 2016;222:113-118.
24. Ke D, He X, Chen C, et al. Randomized Pilot Study to Compare DCB-Based versus DST-Based Strategies for the Treatment of True or Complex Coronary Bifurcation Lesions. Rev Cardiovasc Med. 2023;24:99.
25. Kawai T, Watanabe T, Yamada T, et al. Coronary vasomotion after treatment with drug-coated balloons or drug-eluting stents: a prospective, open-label, single-centre randomised trial. EuroIntervention. 2022;18:E140-E148.
26. Gobic´D, Tomulic´V, Lulic´D, et al. Drug-Coated Balloon Versus Drug-Eluting Stent in Primary Percutaneous Coronary Intervention: A Feasibility Study. Am J Med Sci. 2017;354:553-560.
27. Liu S, Zhou Y, Shen Z, et al. Comparison of Drug-Coated Balloon and Drug-Eluting Stent for the Treatment of Small Vessel Disease (from the Dissolve SVD Randomized Trial). Am J Cardiol. 2024;211:29-39.
28. Cortese B, Testa G, Rivero F, Erriquez A, Alfonso F. Long-Term Outcome of Drug-Coated Balloon vs Drug-Eluting Stent for Small Coronary Vessels: PICCOLETO-II 3-Year Follow-Up. JACC Cardiovasc Interv. 2023;16:1054-1061.
29. Yu X, Wang X, Ji F, et al. A Non-inferiority, Randomized Clinical Trial Comparing Paclitaxel-Coated Balloon Versus New-Generation Drug-Eluting Stents on Angiographic Outcomes for Coronary De Novo Lesions. Cardiovasc Drugs Ther. 2022;36:655-664.
30. Niehe SR, Vos NS, Van Der Schaaf RJ, et al. 5-Year Clinical Outcomes of Paclitaxel-Coated Balloon Angioplasty vs DES in Acute MI: The REVELATION Trial. JACC Cardiovasc Interv. 2024;17:1185-1186.
31. Wang Z, Yin Y, Li J, et al. New Ultrasound-Controlled Paclitaxel Releasing Balloon vs Asy mmetric Drug-Eluting Stent in Primary ST-Segment Elevation Myocardial Infarction —A Prospective Randomized Trial —. Circ J. 2022;86:642-650.
32. Korjian S, McCarthy KJ, Larnard EA, et al. Drug-Coated Balloons in the Management of Coronary Artery Disease. Circ Cardiovasc Interv. 2024;17:E013302.
33. Sanz Sánchez J, Chiarito M, Cortese B, et al. Drug-Coated balloons vs drug-eluting stents for the treatment of small coronary artery disease: A meta-analysis of randomized trials. Catheter Cardiovasc Interv. 2021;98:66-75.
34. Dobromir Angheluta A, Levett JY, Zolotarova T, et al. A Meta-Analysis of 3-Year Outcomes of Drug-Coated Balloons Versus Drug-Eluting Stents for Small-Vessel Coronary Artery Disease. JACC Adv.2024;3:101204.
35. Sun B, Zhang XT, Chen RR. Comparison of Efficacy and Safety Between Drug-Coated Balloons Versus Drug-Eluting Stents in the Treatment of De Novo Coronary Lesions in Large Vessels: A Study-Level Meta-Analysis of Randomized Control Trials. Cardiovasc Drugs Ther. 2024;38:1375-1384.
36. Jiang JL, Huang QJ, Chen MH. Efficacy and safety of drug-coated balloon for de novo lesions of large coronary arteries: Systematic review and meta-analysis of randomized controlled trials. Heliyon. 2024;10:e25264.
37. Abdelaziz A, Hafez A, Atta K, et al. Drug-coated balloons versus drug-eluting stents in patients with acute myocardial infarction undergoing percutaneous coronary intervention: an updated meta-analysis with trial sequential analysis. BMC Cardiovasc Disord. 2023;23:605.
38. Caminiti R, Vizzari G, Ielasi A, et al. Drug-coated balloon versus drug-eluting stent for treating de novo large vessel coronary artery disease: a systematic review and meta-analysis of 13 studies involving 2888 patients. Clin Res Cardiol. 2025;114:978-990.
39. Cui K, Lyu S, Song X, et al. Drug-eluting balloon versus bare-mental stent and drug-eluting stent for de novo coronary artery disease: A systematic review and meta-analysis of 14 randomized controlled trials. PLoS One. 2017;12:e0176365.
40. Fezzi S, Giacoppo D, Fahrni G, et al. Individual patient data meta-analysis of paclitaxel-coated balloons vs drug-eluting stents for small-vessel coronary artery disease: the ANDROMEDA study. Eur Heart J. 2025;46:1586-1599.
41. Niu J, Wang K, Wang W, et al. Drug-Coated Balloons Versus Drug-Eluting Stents for the Treatment of De Novo Coronary Artery Disease: A Meta-Analysis of Randomized Controlled Trials. Rev Cardiovasc Med. 2024;25:446.
42. Muramatsu T, Kozuma K, Tanabe K, et al. Clinical expert consensus document on drug-coated balloon for coronary artery disease from the Japanese Association of Cardiovascular Intervention and Therapeutics. Cardiovasc Interv Ther. 2023;38:166-176.
43. Cuculi F, Bossard M, Zasada W, et al. Performing percutaneous coronary interventions with predilatation using non-compliant balloons at high-pressure versus conventional semi-compliant balloons: insights from two randomised studies using optical coherence tomography. Open Heart. 2020;7:e001204.
44. Lamelas P, Belardi J, Whitlock R, Stone GW. Limitations of Repeat Revascularization as an Outcome Measure: JACC Review Topic of the Week. J Am Coll Cardiol. 2019;74:3164-3173.
45. Giacoppo D, Saucedo J, Scheller B. Coronary Drug-Coated Balloons for De Novo and In-Stent Restenosis Indications. J Soc Cardiovasc Angiogr Interv.2023;2:100625.
46. Pasterkamp G, De Kleijn DPV, Borst C. Arterial remodeling in atherosclerosis, restenosis and after alteration of blood flow: potential mechanisms and clinical implications. Cardiovasc Res. 2000;45:843-852.
47. Cortese B, Serruys PW. Single-Antiplatelet Treatment After Coronary Angioplasty With Drug-Coated Balloon. J Am Heart Assoc. 2023;12:e028413.
48. Cortese B, Silva Orrego P, Agostoni P, et al. Effect of Drug-Coated Balloons in Native Coronary Artery Disease Left With a Dissection. JACC Cardiovasc Interv. 2015;8:2003-2009.
ABSTRACT
Introduction and objectives: Drug-coated balloons (DCB) are emerging as a valid alternative for the treatment of coronary bifurcation lesions, particularly in the side branch (SB). Among bifurcation lesions, the left anterior descending (LAD)-diagonal location is the most frequently treated. The aim of this study was to evaluate the long-term effectiveness of a DCB in the SB compared with a conventional strategy in this setting.
Methods: This case-control study compared LAD-diagonal lesions treated in which the SB was treated with a DCB vs a conventional strategy consisting of provisional stenting or a 2-stent technique. The mean follow-up was 3 years. The incidence rates of death, acute myocardial infarction, target lesion revascularization, target vessel revascularization, and the composite of major adverse cardiovascular events were recorded.
Results: A total of 86 patients with LAD-diagonal bifurcation lesions treated with a DCB in the SB were included and compared with a cohort of 88 patients who underwent conventional SB management. The mean follow-up was 1008 days (range, 5-2411 days). In the control group, the strategy consisted of stenting of the main branch only in 76.2% of cases, SB stenting only in 6.8%, and stenting of both branches in 17%. The comparative rate (control vs DCB) of adverse events was as follows: cardiac death, 1.1% vs 2.3% (non-significant); myocardial infarction, 8% vs 0% (P = .014), target lesion revascularization; 9.1% vs 0% (P = .013); target vessel revascularization, 10.2 vs 1.2% (P = .02); and major adverse cardiovascular events, 12.5% vs 3.5% (P = .05).
Conclusions: Treatment of the SB with a DCB in AD-diagonal bifurcation lesions appears to be safe and demonstrates very high long-term effectiveness, with a significantly lower rate of adverse events compared with a conventional strategy. Large randomized clinical trials are needed to confirm these findings.
Keywords: Drug-coated balloon. Coronary bifurcation lesions. Follow-up study.
RESUMEN
Introducción y objetivos: El balón farmacoactivo (BFA) es una alternativa válida en el tratamiento de las lesiones en bifurcación, en especial en la rama lateral (RL). Dentro de las bifurcaciones, la localización descendente anterior (DA)-diagonal es la más frecuente. Nuestro objetivo fue determinar la efectividad a largo plazo del BFA en la RL en dicho escenario, en comparación con una estrategia convencional.
Métodos: Estudio de casos y controles para comparar lesiones en la bifurcación DA-diagonal con las RL tratadas con BFA o con estrategia convencional de stent condicional o 2 stents, con un seguimiento medio de 3 años. Se registró la incidencia de muerte, infarto agudo de miocardio, revascularización de la lesión diana, revascularización del vaso diana y la combinación de eventos cardiovasculares adversos mayores.
Resultados: Se incluyó a 86 pacientes con lesión en la bifurcación DA-diagonal en el grupo de tratamiento con BFA y se compararon con una cohorte de 88 pacientes con abordaje convencional de la RL. El seguimiento medio fue de 1.008 días (rango 5-2.411). La estrategia en el grupo control fue stent solo en la rama principal 76,2%, stent solo en la RL 6,8% y stent en ambas ramas 17%. La incidencia comparativa (control frente a BFA) de eventos adversos fue: muerte cardiaca 1,1 frente a 2, 3% (no significativo); infarto de miocardio 8 frente a 0% (p = 0,014); revascularización de la lesión diana 9,1 frente a 0% (p = 0,013); revascularización del vaso diana 10,2 frente a 1,2% (p = 0,02); y eventos cardiovasculares adversos mayores 12,5 frente a 3,5% (p = 0,05).
Conclusiones: El tratamiento con BFA de la RL en lesiones en la bifurcación DA-diagonal es seguro y presenta una muy alta efectividad a largo plazo, con una incidencia significativamente menor de eventos adversos en comparación con la estrategia convencional. Se requerirían amplios ensayos clínicos aleatorizados para confirmar estos hallazgos.
Palabras clave: Balón farmacoactivo. Lesiones en bifurcación. Estudio de seguimiento.
DCB: drug-coated balloon. MACE: major adverse cardiovascular events. MB: main branch. SB: side branch. TLR: target lesion revascularization. TVR: target vessel revascularization.
INTRODUCTION
Coronary bifurcation lesions represent 20% of all lesions treated in the cath lab; among them, left anterior descending coronary artery (LAD)-diagonal lesions are the most frequent (between 50% and 70% according to published series).1,2 Currently, the most widely accepted strategy is provisional stenting.3-5 The 2-stent strategy is usually reserved for long lesions in the side branch (SB) or as bailout strategy if the SB worsens after main branch (MB) stenting. Drug-coated balloon (DCB) has emerged as a valid strategy for the treatment of these lesions, both in the MB, mainly in Medina 0,1,1 lesions, and in the SB, which is a much more extensively studied scenario.6 Studies have demonstrated significant benefit in angiographic parameters; however, controversy remains regarding clinical benefit, and only recently have studies confirmed significant improvements in clinical events. The theoretical advantages7 of DCB in the management of the SB would mainly be drug delivery at the SB coronary ostium, absence of distortion of its original anatomy, and minimization of strut deformation at the carina if MB stenting is performed.
We present a nonrandomized comparative trial of LAD-diagonal bifurcation lesions with a long-term follow-up (mean 3 years) treated with DCB in the SB vs a control group with conventional SB management.
METHODS
We conducted a comprehensive single-center retrospective registry at Hospital General Dr. Balmis (Alicante, Spain), a high-volume center performing > 1200 angioplasties per year including all consecutive LAD-diagonal bifurcation lesions with SB ≥ 2 mm in which the SB was treated with a paclitaxel-coated SeQuent Please NEO DCB (B. Braun, Germany). Procedures had to conclude successfully (including absence of post-DCB dissection requiring stenting, residual percent diameter stenosis < 50%, and final TIMI grade-3 flow). In our cath lab, the routine clinical practice was to use DCB after MB stenting, recrossing and dilating the cell adjacent to the SB coronary ostium before SB treatment (at the operator’s discretion, the DCB could be used before stenting if recrossing was anticipated to be difficult).
The inclusion period spanned from October 2018 through July 2024. There were no exclusion criteria. The interventional cardiology team was instructed to adhere to device recommendations, including SB predilatation preferably with a noncompliant or scoring balloon at a vessel/balloon diameter ratio of 0.8–1, and DCB use only if acceptable angiographic results were achieved (including TIMI grade-3 flow, no significant dissection, and residual percent diameter stenosis < 30%). The control group included all consecutive procedures performed during the first half of 2021 in which successful LAD-diagonal bifurcation intervention (diagonal diameter ≥ 2 mm) was performed using the provisional or 2-stent technique at operator discretion.
We analyzed clinical patient variables, lesion anatomical characteristics, and procedural data. Mean clinical follow-up was 3 years, conducted via telephone contact or digital health record review. Clinical events were selected according to the Drug Coated Balloon Academic Research Consortium recommendations.8 Recorded events included all-cause mortality (cardiac and noncardiac), acute myocardial infarction (elevation of myocardial injury biomarkers > 99th percentile upper reference limit with clinical evidence of ischemia), lesion thrombosis, target lesion revascularization (TLR), and target vessel revascularization (TVR) (revascularization either of the target lesion included in the study or other segments within the same vessel) at the follow-up. The composite endpoint of mayor adverse cardiovascular events (MACE) included TLR, TVR, hospitalization for acute myocardial infarction, and cardiac death. All patients signed informed consent, and the study was approved by Hospital General Dr. Balmis ethics committee.
Statistical analysis
Continuous variables were expressed as mean and SD and compared using Student t test. Categorical variables were expressed as percentages and were compared using the chi-square test or Fisher’s exact test when expected cell counts were < 5. Variables showing significant differences in univariate analysis between the DCB group and the control group were entered into a logistic regression model to determine independent predictors of higher rates of TLR. Similarly, we constructed event-free survival curves using the Kaplan-Meier method, and the log-rank test to compare the DCB treatment group vs the control group.
RESULTS
A total of 86 patients were included in the DCB group and 88 in the control group. The patients’ clinical and anatomical characteristics are shown in table 1. The rate of true bifurcations (Medina 1,1,1; 1,0,1; or 0,1,1 with SB diameter ≥ 2.5 mm) was 34.9% in the DCB group and 28.4% in the control group (P = .36). Among such variables, significant differences were observed across groups only in multivessel disease and prior coronary intervention, whose incidence rates were both higher in the DCB group. In-stent restenosis lesions were more than twice as frequent in the DCB group, with borderline statistical significance. Procedural variables are shown in table 2.
Table 1. Clinical characteristics of the patients
| Clinical characteristics | DCB group (n = 86) | Control group (n = 88) | P |
|---|---|---|---|
| Age (years) | 67 ± 11 | 65 ± 13 | .28 |
| Male sex | 81.8 | 77.9 | .41 |
| Hypertension | 60.5 | 58 | .73 |
| Dyslipidemia | 75.6 | 67 | .21 |
| Smoking | 25.6 | 31.8 | .36 |
| Diabetes | 24.4 | 34.1 | .16 |
| Prior PTA | 41.9 | 22.7 | < .01 |
| LVEF | 54.3 | 54.4 | .96 |
| Indication for coronary angiography | |||
|
|
|
|
| Multivessel disease | 67.4 | 51.1 | .029 |
| ISR | 15.1 | 6.8 | .079 |
| Medina classification | |||
|
|
|
|
| Lesion in the SB | |||
|
|
|
|
|
DCB, drug-coated balloon; ISR, in-stent restenosis lesion; LV, left ventricle; LVEF, left ventricular ejection fraction; NSTEACS, non–ST-segment elevation acute coronary syndrome; PTA, percutaneous transluminal angioplasty; SB, side branch; STEACS, ST-segment elevation acute coronary syndrome. Data are expressed as No. (%) or mean ± SD. |
|||
Table 2. Procedural characteristics
| Characteristics | DCB group (n = 86) | Control group (n = 88) | P |
|---|---|---|---|
| Radial access | 98.8 | 96.6 | .62 |
| 6-Fr | 96.5 | 90.9 | .78 |
| Predilatation | |||
|
|
|
|
| MB stenting | 84.9 | 93.1 | .079 |
| SB diameter ≥ 2.5 mm | 65.1 | 63.1 | .84 |
| Postdilatation | |||
|
|
|
|
| OCT/IVUS use | 9.3 | 2.3 | .056 |
|
DCB, drug-coated balloon; Fr, French; IVUS, intravascular ultrasound; MB, main branch; OCT, optical coherence tomography; POT, proximal optimization technique; SB, side branch. Data are expressed as percentages. |
|||
In the control group, the predominant strategy was 1 MB stent (76.1%). Stents were implanted in both branches in 17% of the cases and only in the SB in 6.9%. The rates of SB predilatation, MB postdilatation, and use of kissing-balloon or proximal optimization techniques were significantly more frequent in the DCB group. DCB was applied after MB stenting in most cases; pre-stent use occurred in 7 cases (8.1%). The use of intracoronary imaging, such as optical coherence tomography (OCT) or intravascular ultrasound (IVUS) was low in both groups, with a trend toward greater use in the DCB group but remaining < 10%.
The rate of adverse events during follow-up is shown in table 3. After a mean of 3 years, 13 deaths were reported in the DCB group and 11 in the control group, mostly noncardiac. Among the 2 cardiac deaths reported in the DCB group, 1 was due to heart failure progression (after catheterization the patient underwent transfemoral aortic valve implantation and permanent pacemaker implantation), and the other was sudden death at home. No TLR, myocardial infarction, or definite lesion thrombosis occurred in the DCB group. A total of 8 myocardial infarctions were reported in the control group, although only 1 was not target-vessel related (right coronary revascularization in the context of a non-ST-segment elevation acute coronary syndrome). There was 1 case of TVR (figure 1) in the active group in a patient in whom the LAD-diagonal lesion was treated with orbital atherectomy due to severe calcification and who was readmitted 1 year later for unstable angina, without changes on electrocardiogram or echocardiogram and without enzyme elevation. However, repeat catheterization revealed progression of a lesion at the distal margin of the LAD stent, far from the bifurcation treated with DCB. This revascularization was not ischemia-guided, and the result at the bifurcation was optimal. The rate of TLR in the control group was 9.1%, with the MB responsible in 4 of the 8 cases, the SB in 2 cases, and both branches in the remaining 2. The rates of TLR and TVR were significantly lower in the DCB group (TLR, 0 vs 9.1%; P = .013; TVR, 1.2 vs 10.5%; P = .02). The logistic regression model (table 4) showed that none of the analyzed variables were independent predictors of events.
Table 3. Rate of adverse events during follow-up
| Adverse events | DCB group (n = 86) | Control group (n = 88) | P |
|---|---|---|---|
| Follow-up days | 1009 ± 586 | 1028 ± 233 | .66 |
| Cause of death | |||
| Cardiac | 2.3 | 1.1 | .69 |
| Noncardiac | 12.8 | 11.4 | |
| TLR | 0 | 9.1 | .013 |
| TVR | 1.2 | 10.2 | .02 |
| Lesion thrombosis | |||
| Yes | 0 | 3.4 | .15 |
| Possible | 1.2 | 0 | |
| Myocardial infarction | 0 | 8 | .014 |
| Major adverse cardiovascular events | 3.5 | 12.5 | .05 |
|
DCB: drug-coated balloon; TLR: target lesion revascularization; TVR: target vessel revascularization. Data are expressed as No. (%) or mean ± SD. |
|||
Figure 1. Single case of target vessel revascularization in the drug-coated balloon (DCB) treatment group. A: bifurcation lesion with severe calcification in the main branch. B: optimal result after orbital atherectomy, stent implantation in the left anterior descending coronary artery, and drug-coated balloon in the diagonal branch. C: 1-year follow-up result showing a significant lesion at the distal stent margin (orange arrow), far from the bifurcation treated with DCB (black arrows), which demonstrated sustained procedural success.
Table 4. Logistic regression model for independent predictors of target lesion revascularization
| Factors | P | 95%CI | |
|---|---|---|---|
| Prior PCI | .81 | 0.13-13.2 | |
| Multivessel disease | .45 | 0.11-2.7 | |
| ISR | 1 | 0 – > 30 | |
| SB predilatation | .22 | 0.5-21 | |
| MB stent | 1 | 0 – > 30 | |
| MB postdilatation | .73 | 0.2-8.7 | |
| POT | .85 | 0.09-18.2 | |
| Kissing-balloon | .85 | 0.09-18.2 | |
| OCT/IVUS | 1 | 0 – ≥ 30 | |
|
95%CI, 95% confidence interval; ISR, in-stent restenosis; IVUS, intravascular ultrasound; MB, main branch; OCT, optical coherence tomography; PCI, prior coronary intervention; POT, proximal optimization technique; SB, side branch. |
|||
The rate of MACE was significantly lower in the DCB group (3.5 vs 12.5%; P = .05). Kaplan-Meier curves for primary and secondary endpoints are shown in figure 2.
Figure 2. Kaplan-Meier curves for adverse-event–free survival. MACE, major adverse cardiovascular events; TLR, target lesion revascularization; TVR, target vessel revascularization.
Figure 3. Central illustration. Kaplan-Meier curves for event-free survival. MACE, major adverse cardiovascular events; TLR, target lesion revascularization.
DISCUSSION
Despite the vast body of literature on coronary bifurcation treatment, the real importance of the SB and its implication in target lesion failure are not well defined; therefore, there are no clear clinical practice guidelines on the optimal therapeutic approach for this branch. Few studies have exclusively analyzed the effectiveness of DCB in SB treatment, although this trend appears to be changing. Early studies published from 2011 onward, such as the DEBIUT,9 the BABILON,10 the DEBSIDE,11 the study by Herrador et al.,12 the PEPCAD V,13 the PEPCAD-BIF,14 and the BEYOND15 showed discrepant data regarding DCB effectiveness, although generally favorable. These studies demonstrated improved quantitative angiographic parameters in terms of restenosis or late lumen loss; however, this was not always accompanied by lower revascularization rates, and there were concerns on a potential increase in late thrombosis, as suggested by some of them. A meta-analysis including 10 studies evaluating the effect of DCB in the SB concluded that this technique resulted in significantly better angiographic outcomes; however, this did not translate into a significant improvement of the outcomes (mainly target lesion failure), according to the authors, due to the low rate of this adverse event and insufficient statistical power because of small sample size.16
The optimal timing for DCB use remains controversial, and no study has demonstrated superiority of its use before or after MB stenting. Although expert consensus recommends its use before stenting, the timing ultimately depends on operator or team experience. Conversely, in the randomized DCB-BIF study17—the most relevant to date—the design required DCB application after stenting.
In the present registry, although not randomized, we consider both groups comparable when interpreting differences in the rate of adverse event, which favored the DCB treatment group. Clinical and anatomical differences were unfavorable to the DCB group (higher rate of in-stent restenosis lesions and multivessel disease), and some procedural differences may be explained by the intrinsic characteristics of each approach in each of the groups (such as a higher SB predilatation rate in the DCB group, considered practically mandatory). Furthermore, multivariate analysis including all these factors did not identify any independent predictor of events, thereby minimizing the likelihood of confounding. Despite this, in the study by Oh et al.,18 treatment of the SB with either a conventional balloon or a stent in 1089 patients with true LAD-diagonal bifurcation lesions, compared with no SB treatment, was associated with a lower rate of target vessel failure. However, this difference did not reach statistical significance overall, although it became significant in the low-risk subgroup.
Very few studies have demonstrated clinical, not only angiographic, benefit after DCB use in the SB. The most relevant one is the 2025 study by Gao et al.,17 in which DCB in the SB after provisional stenting resulted in a significant reduction of the composite adverse event driven by fewer myocardial infarctions, although without significant differences in TLR. The cause of these findings remains controversial.19
Another study published in 2022 randomized 219 true de novo bifurcation lesions to SB treatment with conventional balloon vs DCB.20 At the 12-month clinical and angiographic follow-up, significant improvements were observed both in angiographic parameters (lower late lumen loss and greater late minimum lumen diameter) and clinical outcomes, with a lower rate of MACE; however, this improvement did not translate into significant reductions in new revascularizations or target vessel failure.
In the present study, the rate of adverse events at the 3-year follow-up in the DCB group was exceptionally low, with 0% TLR and 1.2% TVR. The 9% TLR rate in the control group is similar to that reported in former studies of high-risk coronary lesions such as bifurcations. In the abovementioned study by Oh et al.,17 the 3-year rates of TLR went from 6.6% to 9% among > 1000 LAD- diagonal lesions. In another study comparing conventional vs DCB approaches in various complex lesions (including bifurcations: 36% in the first group and 24% in the second), the 2-year rate of TVR was 7.6% in the DCB group and 8.1% in the stent group.21
In a very recent Swedish study, the rate of adverse events was compared in nearly 6800 LAD-diagonal bifurcation lesions treated with a simple (MB only) vs complex strategy (both branches, both with balloon and stent).22 At the 1-year follow-up, the rate of MACE was lower in the complex strategy group (6.2% vs 7.9%; HR, 0.74; 95%CI, 0.59–0.93; P = .010), driven by a lower all-cause mortality rate, without differences in TLR. This benefit persisted at 5 years (17% vs 19.8%; HR, 0.83; 95%CI, 0.72–0.96; P = .010).
The relative importance of the SB in repeat revascularizations remains controversial. Although some studies, such as the BABILON10 showed that new revascularizations originated in the MB, in our registry, in the control group, up to 50% of the cases originated in the SB. This supports the need for exhaustive SB treatment during provisional stenting, both with DCB (as suggested by our results) and any other technique (conventional balloon or stent), particularly if significant SB ostial stenosis persists even without flow limitation.
Our group recently published a study analyzing adverse predictors after DCB use in SB bifurcation lesions regardless of location.23 Only the presence of long SB lesions (> 10 mm) was identified as a negative prognostic factor. Our findings suggest that in the LAD-diagonal location this factor may be less relevant than in other locations, although adequately powered randomized trials are needed to confirm or refute this hypothesis.
Limitations
The main limitations of this study are the lack of randomization, absence of perioperative myocardial infarction recording due to its retrospective design, inability to extract relevant IVUS/OCT data given low usage, relatively small sample size, and lack of systematic angiographic reevaluation, which may have identified subclinical events.
CONCLUSIONS
These results correspond to a single center with a very low long-term rate of adverse events in patients with LAD-diagonal bifurcation lesions whose SB was treated with DCB, significantly lower than in the control group. Currently, this is the first study demonstrating a significant improvement in an important endpoint such as TLR. Therefore, proper lesion selection (possibly those without long SB lesions), meticulous lesion preparation technique, and greater use of IVUS and OCT are essential. Randomized clinical trials with sufficient statistical power are needed to confirm these promising results and definitively establish the superiority of DCB for SB treatment in bifurcation lesions, particularly in the most frequent location, LAD-diagonal.
WHAT IS KNOWN ABOUT THE TOPIC?
- There is a limited number of studies analyzing the role of DCB in SB treatment of coronary bifurcation lesions, particularly in LAD-diagonal.
- Although DCB appears beneficial in angiographic parameters, this has not consistently translated into significant clinical improvement such as target vessel failure or need for repeat revascularization.
WHAT DOES THIS STUDY ADD?
- Experience from a high-volume center with very long-term follow-up representative of real-world practice.
- The favorable results may support DCB as a useful tool to improve long-term outcomes in LAD-diagonal bifurcation intervention.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The study followed the Helsinki Declaration guidelines and was approved by Hospital General Dr. Balmis ethics committee (Alicante, Spain). Informed consent was obtained. SAGER guidelines regarding sex/gender bias were followed.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the preparation of this work or manuscript.
AUTHORS’ CONTRIBUTIONS
J. Valencia: patient treatment, data collection, manuscript drafting and revision. F. Torres-Mezcua and M. Herrero-Brocal: patient treatment, data collection and revision. P. Bordes, F. Torres-Saura, J. Pineda and J.M. Ruiz-Nodar: patient treatment and revision. All authors approved the final version.
CONFLICTS OF INTEREST
None declared.
REFERENCES
1. Valencia J, Torres-Mezcua F, Herrero-Brocal M, et al. Long-term effectiveness of drug-coated balloon in the side branch treatment of bifurcation lesions. REC Interv Cardiol. 2023;5:7-13.
2. Kurt M, Tanboga IH, Karakas MF, et al. Clinical and morphological evaluation of coronary bifurcation lesions. Arch Turk Soc Cardiol. 2013;41:207-211.
3. Albiero R, Burzotta F, Lassen JF, et al. Treatment of coronary bifurcation lesions, part I:implanting the first stent in the provisional pathway. The 16th expert consensus document of the European Bifurcation Club. EuroIntervention. 2022;18:e362-e376.
4. Burzotta F, Lassen JF, Louvard Y, et al. European Bifurcation Club white paper on stenting techniques for patients with bifurcated coronary artery lesions. Catheter Cardiovasc Interv. 2020;96:1067-1079.
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
6. Jeger RV, Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease:Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13:1391-1402.
7. Sawaya FJ, Lefèvre T, Chevalier B, et al. Contemporary Approach to Coronary Bifurcation Lesion Treatment. JACC Cardiovasc Interv. 2016;9:1861-1878.
8. Fezzi S, Scheller B, Cortese B, et al. Definitions and standardized endpoints for the use of drug-coated balloon in coronary artery disease:consensus document of the Drug Coated Balloon Academic Research Consortium. Eur Heart J. 2025;46:2498-2519.
9. Stella PR, Belkacemi A, Dubois C, et al. A multicenter randomized comparison of drug-eluting balloon plus bare-metal stent versus bare-metal stent versus drug-eluting stent in bifurcation lesions treated with a single-stenting technique:six-month angiographic and 12-month clinical results of the drug-eluting balloon in bifurcations trial. Catheter Cardiovasc Interv. 2012;80:1138-1146.
10. López Mínguez JR, Nogales Asensio JM, Doncel Vecino LJ, et al. A prospective randomised study of the paclitaxel-coated balloon catheter in bifurcated coronary lesions (BABILON trial):24-month clinical and angiographic results. EuroIntervention. 2014;10:50-57.
11. Berland J, Lefèvre T, Brenot P, et al. DANUBIO —a new drug-eluting balloon for the treatment of side branches in bifurcation lesions:six-month angiographic follow-up results of the DEBSIDE trial. EuroIntervention. 2015;11:868-876.
12. Herrador JA, Fernandez JC, Guzman M, Aragon V. Drug-eluting vs. conventional balloon for side branch dilation in coronary bifurcations treated by provisional T stenting. J Interv Cardiol. 2013;26:454-462.
13. Mathey DG, Wendig I, Boxberger M, Bonaventura K, Kleber FX. Treatment of bifurcation lesions with a drug-eluting balloon:the PEPCAD V (Paclitaxel Eluting PTCA Balloon in Coronary Artery Disease) trial. EuroIntervention. 2011;7 (Suppl K):K61-65.
14. Kleber FX, Rittger H, Ludwig J, et al. Drug eluting balloons as stand alone procedure for coronary bifurcational lesions:results of the randomized multicenter PEPCAD-BIF trial. Clin Res Cardiol. 2016;105:613-621.
15. Jing QM, Zhao X, Han YL, et al. A drug-eluting Balloon for the trEatment of coronarY bifurcatiON lesions in the side branch:a prospective multicenter ranDomized (BEYOND) clinical trial in China. Chin Med J. 2020;133:899-908.
16. Zheng Y, Li J, Wang L, et al. Effect of Drug-Coated Balloon in Side Branch Protection for de novo Coronary Bifurcation Lesions:A Systematic Review and Meta-Analysis. Front Cardiovasc Med. 2021;8:758560.
17. Gao X, Tian N, Kan J, et al. Drug-Coated Balloon Angioplasty of the Side Branch During Provisional Stenting:The Multicenter Randomized DCB-BIF Trial. J Am Coll Cardiol. 2025;85:1-15.
18. Oh GC, Park KW, Kang J, et al. Association of Side-Branch Treatment and Patient Factors in Left Anterior Descending Artery True Bifurcation Lesions:Analysis from the GRAND-DES Pooled Registry. J Interv Cardiol. 2020;2020:8858642.
19. Ali Z, Yong CM. Drug-coated balloons to keep interventions of the side branch simple (KISS):KISS or be KISS'd?J Am Coll Cardiol. 2025;85:16-18.
20. Li Y, Mao Q, Liu H, Zhou D, Zhao J. Effect of Paclitaxel-Coated Balloon Angioplasty on Side Branch Lesion and Cardiovascular Outcomes in Patients with De Novo True Coronary Bifurcation Lesions Undergoing Percutaneous Coronary Intervention. Cardiovasc Drugs Ther. 2022;36:859-866.
21. Joh HS, Kwon W, Shin D, et al. Efficacy of drug-coated-balloon and drug-eluting stent in percutaneous coronary intervention for complex lesions. JACC Asia. 2024;4:519-531.
22. Katona A, von Koch S, Andell P, et al. Long-term prognosis after coronary bifurcation PCI-A nationwide observational study. PLoS One.2025;20:e0317628.
23. Valencia J, Torres-Mezcua F, Herrero M, et al. Prognostic factors in drug-coated balloon interventions for treating the side branch of coronary bifurcation lesions. REC Interv Cardiol. 2025;7:57-59.
- SELUTION SLR sirolimus-eluting balloon compared with the Pantera Lux paclitaxel drug-coated balloon in the management of de novo coronary lesions
- Feasibility of implementing reperfusion strategies in STEMI codes in a mountainous population remote from a PCI-capable center
- Territorial longitudinal strain to predict the location and severity of significant stenosis in stable coronary artery disease
- Outcomes of left ventricular pacing with a pre-shaped guidewire during transcatheter aortic valve implantation
Editorials
Expanding the role of drug-coated balloons in native large coronary artery disease
aDepartment of Biomedical Sciences, Humanitas University, Pieve Emanuele, Milan, Italy
bCardio Center, IRCCS Humanitas Research Hospital, Rozzano, Milan, Italy
cDivision of Cardiology, Montefiore Medical Center, Bronx, New York, United States
Original articles
Editorials
The role of angiography-derived physiological assessment techniques in the post-FAVOR III Europe era?
aServicio de Cardiología, Hospital General Universitario Gregorio Marañón, Instituto de Investigación Sanitaria Gregorio Marañón, Universidad Complutense de Madrid, Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain
Original articles
Review Articles
Interviews
An interview with Camino Bañuelos
aServicio de Cardiología, Hospital Clínico San Carlos, Instituto de Investigación Sanitaria del Hospital Clínico San Carlos (IdISSC), Madrid, Spain
bServicio de Cardiología, Hospital Clínico Universitario de Santiago de Compostela, Santiago de Compostela, A Coruña, Spain