ABSTRACT
Introduction and objectives: Drug-coated balloons are an emerging stentless therapeutic option for the treatment of de novo coronary artery disease. Although paclitaxel-coated balloons remain the current standard, concerns regarding delayed vascular healing and potential local toxicity have driven the development of sirolimus-eluting balloons (SEB). This study aimed to compare the safety and efficacy profile of the novel SELUTION SLR SEB with the established Pantera Lux paclitaxel-coated balloon in a real-world cohort of patients with de novo coronary artery disease.
Methods: We conducted a prospective, single-center registry included 257 patients with 316 de novo coronary lesions treated with drug-coated balloon–only percutaneous coronary intervention from 2023 through 2024. Patients were treated with either the SELUTION SLR sirolimus-eluting balloon or the Pantera Lux paclitaxel-coated balloon. A 1-year follow-up was conducted to assess major adverse cardiovascular events, target lesion failure (TLF), and target lesion revascularization (TLR), which were descriptively compared across groups.
Results: Baseline clinical and angiographic characteristics were well balanced. At the 1-year follow-up, the overall rate of major adverse cardiovascular events was 5.8%, with no significant differences across treatment groups. The rates of TLF and TLR were low and comparable. However, patients treated with the SELUTION SLR experienced fewer procedural complications, including a lower rate of mild coronary dissections (6.7% vs 14.7%; P = .043) and no-reflow events (0.0% vs 2.7%; P = .036).
Conclusions: The SELUTION SLR SEB demonstrated efficacy vs the Pantera Lux, with fewer procedural complications. These findings support the selective use of SELUTION SLR as a stentless treatment strategy and highlight the need for larger randomized trials to confirm long-term comparative outcomes.
Keywords: Coronary artery disease. Drug-coated balloon. Percutaneous coronary intervention.
RESUMEN
Introducción y objetivos: Los balones farmacoactivos representan una alternativa emergente sin stent para el tratamiento de la enfermedad coronaria de novo. Aunque los balones recubiertos de paclitaxel constituyen el estándar actual, las preocupaciones sobre el retraso en la cicatrización y la toxicidad vascular han favorecido el desarrollo de balones liberadores de sirolimus. El objetivo fue comparar la seguridad y la eficacia del balón liberador de sirolimus SELUTION SLR frente al balón recubierto de paclitaxel Pantera Lux en pacientes con enfermedad coronaria de novo en la práctica clínica real.
Métodos: Registro prospectivo y unicéntrico que incluye 257 pacientes con 316 lesiones de novo tratados mediante intervención coronaria percutánea exclusivamente con balón farmacoactivo entre 2023 y 2024. Los pacientes recibieron SELUTION SLR (sirolimus) o Pantera Lux (paclitaxel). El seguimiento a 1 año evaluó los eventos cardiacos adversos mayores, el fracaso de la lesión diana y la revascularización de la lesión diana, analizados de forma descriptiva.
Resultados: Las características clínicas y angiográficas basales fueron comparables entre los grupos. Al año, la tasa global de eventos cardiacos adversos mayores fue del 5,8%, sin diferencias significativas. El fracaso de la lesión diana y la revascularización de la lesión diana fueron bajos y similares. Sin embargo, el grupo con SELUTION SLR presentó menos complicaciones intraprocedimiento, incluidas disecciones coronarias leves (6,7% frente a 14,7 %, p = 0,043) y fenómenos de no reflujo (0,0 % frente a 2,7 %, p = 0,036).
Conclusiones: El balón liberador de sirolimus SELUTION SLR mostró una eficacia comparable al Pantera Lux, con menos complicaciones intraprocedimiento, lo que apoya su uso selectivo como estrategia sin stent y señala la necesidad de ensayos aleatorizados de mayor tamaño.
Palabras clave: Enfermedad coronaria. Balón farmacoactivo. Intervención coronaria percutánea.
Abbreviations
CAD: coronary artery disease. DCB: drug-coated balloon. DES: drug-eluting stent. PCB: paclitaxel-coated balloon. PCI: percutaneous coronary intervention. SEB: sirolimus-eluting balloon.
INTRODUCTION
Percutaneous coronary intervention (PCI) has revolutionized the treatment of coronary artery disease (CAD), particularly with the advent of drug-eluting stents (DES). However, restenosis, late thrombosis, and very late thrombosis, and the need for prolonged dual antiplatelet therapy have increased interest in no-footprint percutaneous coronary intervention (PCI) strategies like drug-coated balloons (DCBs).1 Initially developed and widely adopted for treating in-stent restenosis, DCBs have more recently gained acceptance for the management of de novo coronary lesions, particularly in small-vessel disease and in patients at high risk of hemorrhage.2,3
Paclitaxel-coated balloons (PCBs) have dominated the field due to drug lipophilicity and potent antiproliferative effect.4 However, concerns regarding delayed vascular healing, cytotoxicity, and downstream embolization have encouraged the development of newer DCB technologies. Sirolimus, by being cytostatic rather than cytotoxic, offers several theoretical advantages over paclitaxel, including a wider therapeutic window, improved endothelial healing, and reduced inflammatory response. However, its low lipophilicity (making tissue retention difficult) and slow onset of action has limited its use in DCB platforms.
The SELUTION SLR (Cordis, United States) aimed to overcome the unfavorable pharmacodynamics of sirolimus by incorporating microreservoirs and a phospholipid coating designed for controlled, sustained release of sirolimus over 90 days. Preclinical and early clinical evidence suggests favorable vessel healing, drug retention, and patency.5 However, comparative data vs paclitaxel-based DCBs in de novo lesions remain limited. Given the evolving landscape of interventional cardiology and increasing interest in no-footprint PCI, a direct head-to-head evaluation of the safety and efficacy profiles of these 2 DCB platforms is warranted.
This study provides a real-world comparison between the SELUTION SLR SEB and the Pantera Lux (Biotronik, Germany) PCB in the management of de novo coronary lesions. By analyzing clinical and angiographic outcomes, we aim to determine whether the novel SEB platform offers a feasible and potentially superior alternative to the current standard of care.
METHODS
Study population
We conducted an analysis of a single-center prospective registry of 257 patients and 316 de novo coronary lesions treated with DCBs from January 2023 through December 2024. Patients presenting with chronic or acute coronary syndrome were eligible if at least 1 lesion was treated with either the SELUTION SLR or the Pantera Lux. The selection of the DCB platform was left to the operator’s discretion. We excluded patients with cardiac arrest or cardiogenic shock and those in whom > 1 brand of DCB was used. In patients with acute coronary syndrome and multivessel disease, complete revascularization was preferably performed in staged procedures to allow for opportunistic angiographic reevaluation of previously treated lesions. Patients were clinically monitored for 1 year and the rates of all-cause mortality, cardiac death, myocardial infarction (MI), unplanned revascularization, target lesion restenosis, and target lesion revascularization (TLR) were compared between the 2 groups. Target lesion failure (TLF) was determined as a composite endpoint of cardiac death, target lesion MI and TLR. Major adverse cardiovascular events (MACE) were defined as a composite endpoint of cardiac death, any MI and any unplanned revascularization. In patients undergoing opportunistic angiographical follow-up, positive vessel remodeling was defined by a ≥ 50% increment in lumen diameter and restenosis as a ≥ 50% lumen reduction. The protocol of the study was revised and approved by the local ethics committee.
Lesion preparation and DCB deployment
All types of coronary lesions were included and aggressive plaque preparation techniques (using scoring or cutting balloons) were incentivized, as was the use of intracoronary imaging to guide balloon size selection and deployment. Only lesions with residual percent diameter stenosis < 30% after optimal preparation and without significant recoil or flow impairment were eligible for DCB inflation. DCBs were selected according to target vessel reference diameter in a 1:1 ratio and had to be inflated for at least 60 seconds. Only 1 inflation per DCB was allowed. Use of multiple DCB was allowed only if the same brand was used. In lesions with persistent residual percent diameter ≥ 30% after aggressive preparation, PCI with DES was recommended. Although post-PCB bailout stenting was recommended only in the presence of flow limiting dissections or persistent contrast staining, the final decision was left to the operator’s discretion.
Statistical analysis
Continuous variables with normal distribution were expressed as mean ± standard deviation, and categorical ones as absolute count and respective percentages. The 95% confidence intervals (95%CI) of the means of continuous variables and percentages of categorical variables were calculated using t tests and Clopper-Pearson (exact) approaches, respectively. The Student t and Mann Whitney U tests were used to compare variables with normal and non-normal distribution, respectively, while the chi-square test was used to compare prevalences across groups. A subgroup analysis stratified by target vessel diameter was performed using a 3-mm lumen diameter cutoff. Kaplan-Meier survival curves were generated for descriptive time-to-event assessment. Due to the low number of events, propensity score weighting, adjusted Cox proportional hazards models, and other multivariable approaches were not performed, as these approaches would be underpowered and potentially yield unreliable or misleading estimates. All analyses used SPSS v30 (IBM, United States). Although statistical significance was defined as P < .05, findings should be interpreted as exploratory.
RESULTS
Study population
A total of 257 patients with 316 coronary lesions were included: 104 patients (133 lesions) treated with SELUTION SLR and 153 patients (183 lesions) treated with Pantera Lux. Baseline demographics and clinical characteristics were balanced across the groups (table 1). There were no significant differences in target vessel location or lesion complexity according to ACC/AHA lesion classification. True bifurcation lesions represented 44.0% of all treated lesions, reflecting the real-world nature and complexity of the cohort (table 2).
Table 1. General characteristics of the study population (per patient analysis)
| Baseline characteristics | Population (N = 257) | SELUTION SLR (n = 104) | Pantera Lux (n = 153) | P |
|---|---|---|---|---|
| Baseline demographics | ||||
| Age (years) | 67.3 ± 11.1 | 67.3 ± 11.4 | 67.3 ± 10.0 | .994 |
| Male | 224 (83.7) | 91 (87.5) | 133 (86.9) | .771 |
| BMI (kg/m2) | 27.0 [22.9-29.4] | 26.2 [24.5-29.2] | 27.0 [24.9-29.6] | .409 |
| Past medical history | ||||
| Prior MI | 138 (53.7) | 54 (51.9) | 84 (54.9) | .678 |
| Prior PCI | 128 (49.8) | 51 (49.0) | 77 (50.3) | .839 |
| Prior CABG | 9 (3.5) | 6 (5.8) | 3 (2.0) | .103 |
| Hypertension | 192 (74.7) | 80 (76.9) | 112 (73.2) | .501 |
| Dyslipidemia | 200 (77.8) | 79 (75.9) | 121 (79.1) | .201 |
| Smoker | 74 (28.8) | 32 (30.8) | 42 (27.5) | .564 |
| Congestive heart failure | 51 (19.8) | 22 (21.2) | 29 (18.9) | .512 |
| Diabetes mellitus | 102 (39.7) | 40 (38.5) | 62 (40.5) | .740 |
| Renal dysfunction | 52 (20.2) | 23 (22.1) | 29 (19.0) | .532 |
| Pulmonary disease | 12 (4.7) | 7 (6.7) | 5 (3.3) | .197 |
| Peripheral arterial disease | 30 (11.7) | 11 (10.6) | 19 (12.4) | .652 |
| Atrial fibrillation/flutter | 18 (7.0) | 7 (6.7) | 11 (7.2) | .888 |
| Family history of CAD | 21 (8.2) | 6 (5.8) | 15 (9.8) | .246 |
| Clinical presentation | ||||
| STEMI | 57 (22.2) | 23 (22.1) | 34 (22.2) | .984 |
| NSTEMI | 77 (30.0) | 26 (25.0) | 51 (33.3) | .152 |
| Unstable angina | 9 (3.5) | 3 (2.9) | 6 (3.9) | .657 |
| CCS | 114 (44.3) | 52 (50.0) | 62 (40.5) | .113 |
| Procedural characteristics | ||||
| Multivessel/segment disease | 205 (79.7) | 86 (82.7) | 119 (77.8) | .650 |
| > 1 lesion treated | 181 (70.4) | 79 (75.9) | 102 (66.7) | .109 |
| No. of treated lesions | 1.0 [1.0-2.0] | 2.0 [1.0-2.0] | 1.0 [1.0-2.0] | .201 |
|
BMI, body mass index; CABG, coronary artery bypass grafting; CAD, coronary artery disease; CCS, chronic coronary syndrome; MI, myocardial infarction; NSTEMI, non-ST segment myocardial infarction; PCI, percutaneous coronary interventions; STEMI, ST-segment elevation myocardial infarction. Data are express as No. (%), mean ± standard deviation or median [interquartile range]. |
||||
Table 2. General characteristics of the target lesion treated (per lesion analysis)
| Vessel/segment | Population (N = 316) | SELUTION SLR (n = 133) | Pantera Lux (n = 183) | P |
|---|---|---|---|---|
| LAD | 141 (44.6) | 56 (42.1) | 85 (46.4) | .331 |
| Proximal LAD | 25 (7.9) | 10 (7.5) | 15 (8.2) | - |
| Mid LAD | 15 (4.7) | 6 (4.5) | 9 (4.9) | - |
| Distal LAD | 31 (9.8) | 11 (8.3) | 20 (10.9) | - |
| First diagonal | 59 (18.7) | 24 (18.0) | 35 (19.1) | - |
| Second or third diagonal | 11 (3.5) | 5 (3.8) | 6 (3.3) | - |
| LCx | 93 (29.4) | 44 (33.1) | 49 (26.7) | .109 |
| Proximal LCx | 20 (6.3) | 8 (6.0) | 12 (6.6) | - |
| Distal LCx | 23 (7.3) | 12 (9.0) | 11 (6.0) | - |
| First obtuse marginal | 37 (11.7) | 16 (12.0) | 21 (11.5) | - |
| Second obtuse marginal | 11 (3.5) | 6 (4.5) | 5 (2.7) | - |
| Third obtuse marginal | 2 (0.6) | 2 (1.5) | 0 (0.0) | - |
| Intermediate branch | 9 (2.8) | 3 (2.3) | 6 (3.3) | .743 |
| RCA | 70 (22.2) | 29 (21.8) | 41 (22.4) | .828 |
| Proximal RCA | 8 (2.5) | 3 (2.3) | 5 (2.7) | - |
| Mid RCA | 9 (2.8) | 2 (1.5) | 7 (3.8) | - |
| Distal RCA | 15 (4.7) | 6 (4.5) | 9 (4.9) | - |
| PDA | 21 (6.6) | 10 (7.5) | 11 (6.0) | - |
| Postero-lateral branch | 17 (5.4) | 8 (6.0) | 9 (4.9) | - |
| CABG | 3 (0.9) | 1 (0.8) | 2 (1.0) | .901 |
| Arterial graft | 1 (0.3) | 0 (0.0) | 1 (0.5) | - |
| Venous graft | 2 (0.6) | 1 (0.8) | 1 (0.5) | - |
| Lesion characteristics | ||||
| Target vessel diameter ≥ 3 mm | 59 (18.7) | 23 (17.2) | 36 (19.7) | .384 |
| AHA class A | 93 (29.4) | 39 (29.3) | 54 (29.5) | .911 |
| AHA class B1/B2 | 195 (61.7) | 83 (62.4) | 112 (61.2) | .873 |
| AHA class C | 28 (8.9) | 11 (8.3) | 17 (9.3) | .795 |
| CTO | 29 (9.2) | 14 (10.5) | 15 (8.2) | .479 |
| True bifurcation | 139 (44.0) | 61 (45.9) | 78 (42.6) | .559 |
|
AHA, American Heart Association; CABG, coronary artery bypass graft; CTO, chronic total coronary occlusion; LAD, left anterior descending coronary artery; LCx, left circumflex artery; PDA, posterior descending artery; RCA, right coronary artery. Data are expressed as No. (%). |
||||
Procedural characteristics
Lesion preparation details are shown in table 3. Most lesions were prepared with non-compliant (NC) balloons (82.0%). Intracoronary imaging to guide PCI was used in 25.9% of cases. There were no differences in mean DCB diameter (2.25 [2.00-2.50] vs 2.25 [2.00-2.50] for SELUTION SLR and Pantera Lux, respectively, P = .205) and the mean DCB length (20.0 [15.0-25.0] vs 20.0 [15.0-29.0], for SELUTION SLR and Pantera Lux, respectively, P = .432). Nearly one-third (n = 100, 31.6%) of all DCB utilization occurred in the setting of hybrid bifurcation PCI (DES in the main branch plus DCB in the side branch), reflecting the increasing adoption of this strategy to avoid 2-stent PCI technique outside of the left main coronary artery. SELUTION SLR was used more frequently than Pantera Lux in pure DCB bifurcation PCI (DCB in both the main and side branches) (15.7% vs 9.8%, for SELUTION SLR and Pantera Lux, respectively; P = .012).
Table 3. Lesion preparation, devices used for PCI and procedural results (per lesion analysis)
| Devices used and procedural results | Population (N = 316) | SELUTION SLR (n = 133) | Pantera Lux (n = 183) | P |
|---|---|---|---|---|
| Predilatation | ||||
| SC balloon | 66 (20.9) | 29 (21.8) | 37 (20.2) | .732 |
| NC balloon | 259 (82.0) | 110 (82.7) | 149 (81.4) | .769 |
| Plaque preparation/calcium modification | ||||
| Cutting/scoring balloon | 62 (19.6) | 27 (20.3) | 35 (19.1) | .907 |
| Rotational atherectomy | 2 (0.6) | 1 (0.8) | 1 (0.5) | .820 |
| Orbital atherectomy | 9 (2.8) | 4 (3.0) | 5 (2.7) | .885 |
| IVL | 7 (2.2) | 3 (2.3) | 4 (2.2) | .898 |
| Intracoronary imaging | ||||
| IVUS | 70 (22.2) | 28 (21.1) | 42 (23.0) | .792 |
| OCT | 12 (3.8) | 6 (4.5) | 6 (3.3) | .789 |
| PCI | ||||
| DCB diameter | 2.25 [2.00-2.50] | 2.25 [2.00-2.50] | 2.25 [2.00-2.50] | .205 |
| DCB length | 20.0 [15.0-25.0] | 20.0 [15.0-25.0] | 20.0 [15.0-29.0] | .432 |
| PCI with DES in a different lesion | 181 (57.3) | 76 (57.1) | 105 (59.7) | .480 |
| Hybrid bifurcation PCI (DES + DCB) | 100 (31.6) | 43 (32.3) | 57 (31.1) | .703 |
| DCB-only bifurcation PCI | 39 (12.3) | 21 (15.7) | 18 (9.8) | .012 |
| Procedural results | ||||
| Post-DCB residual percent diameter stenosis > 30% | 19 (6.0) | 11 (8.4) | 8 (4.8) | .201 |
| Mild dissection (type A-B) | 36 (11.4) | 9(6.7) | 27 (14.7) | .043 |
| Severe dissection (type C-F) | 3 (0.9) | 0 (0.0) | 3 (1.6) | .370 |
| Coronary perforation | 2 (0.6) | 1 (0.8) | 1 (0.5) | .820 |
| Coronary rupture | 2 (0.6) | 1 (0.8) | 1 (0.5) | .820 |
| No-reflow | 5 (1.6) | 0 (0.0) | 5 (2.7) | .036 |
| Bailout DES | 31 (9.8) | 9 (6.8) | 22 (12.0) | .121 |
| Procedural mortality | 0 (0.0) | 0 (0.0) | 0 (0.0) | - |
|
DCB, drug-coated balloon; DES, drug eluting stent; IVL, intravascular lithotripsy; IVUS, intravascular ultrasound; NC, non-compliant; OCT, optical coherence tomography; PCI, percutaneous coronary intervention; SC, semi-compliant. Data are expressed as No. (%), mean ± standard deviation or median [interquartile range]. |
||||
Several important differences emerged in procedural findings. Mild coronary dissections, defined as non–flow-limiting dissections of types A–B and considered expected angiographic findings after preparation or DCB inflation, occurred significantly more often in the Pantera Lux than in the SELUTION SLR group (14.7% vs 6.7%; P = .043). Severe dissections were rare. There were no-reflow phenomena exclusively in the Pantera Lux group, with 5 cases being reported overall, corresponding to 1.6% of all lesions (0.0% vs 2.7% within each respective treatment group; P = .036). There were no periprocedural deaths in either arm.
1-year clinical outcomes
Clinical follow-up at 12 months was available for all patients, whereas angiographic follow-up was performed in 35.4% of lesions (n = 91) at the operator’s discretion and was not protocol mandated. At the patient level, the overall rates of adverse clinical events were low (table 4). All-cause mortality occurred in 2.3% of patients, cardiovascular mortality in 1.6%, and myocardial infarction in 2.3%. The composite MACE rate was 5.8% overall, with no statistically significant difference between SELUTION SLR and Pantera Lux (2.9% vs 7.8%; P = .168). At the lesion level, TLF occurred in 3.8% of lesions, again without significant differences being reported across groups (2.3% vs 5.8%; P = .252). TLR was rare, recorded in 0.9% of lesions, with no difference across devices. Figure 1 illustrates the respective Kaplan-Meier survival curves for MACE, cardiac death and MI.
Table 4. Events at the follow-up
| Outcomes | Population | SELUTION SLR | Pantera Lux | P |
|---|---|---|---|---|
| Patient oriented outcomes | N = 257 | n = 104 | n = 153 | |
| All-cause mortality | 6 (2.3) | 1 (0.9) | 5 (3.2) | .574 |
| Cardiac death | 4 (1.6) | 1 (0.9) | 3 (1.9) | .410 |
| Myocardial infarction | 6 (2.3) | 1 (0.9) | 5 (3.2) | .574 |
| UR for angina | 5 (1.9) | 1 (0.9) | 4 (2.6) | .630 |
| MACE (cardiac death + MI + UR) | 15 (5.8) | 3 (2.9) | 12 (7.8) | .168 |
| Lesion oriented outcomes | N = 316 | n = 133 | n = 183 | |
| TLF (CV death + target vessel MI + TLR) | 12 (3.8) | 3 (2.3) | 9 (5.8) | .252 |
| Target lesion revascularization | 3 (0.9) | 1 (0.8) | 2 (1.1) | .721 |
| Angiographic control | N = 91 | n = 34 | n = 57 | |
| Restenosis (> 50% diameter) | 7 (7.7) | 1 (2.9) | 6 (10.5) | .229 |
| Positive remodeling (> 50% lumen gain) | 22 (24.2) | 12 (35.3) | 10 (17.5) | .097 |
|
MACE: major adverse cardiovascular events; MI, myocardial infarction; TLR, target lesion revascularization; UR: unplanned revascularization. Data are expressed as No. (%). |
||||
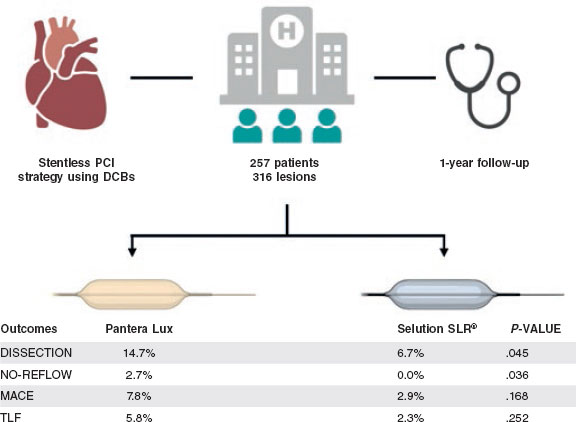
Figure 1. Kaplan Meier survival curves for MACE (A), CV death (B) and MI (C) after 1 year follow-up. CV, cardiovascular; DCB, drug-coated balloon; MACE, major adverse cardiac events; MI, myocardial infarction.
Among the lesions that underwent repeat angiography, restenosis was diagnosed in 7.7% of cases, with no meaningful differences across the different balloon types. Positive vessel remodeling was numerically more common in SELUTION SLR-treated lesions (35.3% vs 17.5%; P = .097), although the sample was limited due to the opportunistic nature of follow-up.
A subgroup analysis according to reference vessel diameter showed that 18.7% of lesions were treated in vessels ≥ 3 mm (table S1). From a procedural standpoint, large arteries did not exhibit higher rates of mild (types A and B) dissections (18.6% vs 10.1%; P = .066) but higher rates of bailout stenting (18.6% vs 7.8%; P = .011) (reasons for stent bailout are summarized in table S2). Despite this, there were no differences in safety outcomes such as severe dissection, coronary rupture, or no-reflow. Moreover, lesion-oriented outcomes, including TLF and TLR rates, were low and comparable across groups. Of note, only 1 TVF event was registered in the large artery group, in 1 patient treated with Pantera Lux. These subgroup observations should be interpreted as exploratory because the study was not powered for interaction testing and event rates were low.
DISCUSSION
In this prospective, real-world registry comparing the SELUTION SLR sirolimus-eluting balloon with the Pantera Lux paclitaxel-coated balloon for the treatment of de novo coronary lesions, the 2 devices demonstrated low and comparable clinical event rates at 12 months (figure 2). The study population included complex, all-comer patients, and despite the inherent limitations of a single-center observational design, the findings contribute to the growing body of evidence supporting DCB strategies for selected de novo lesions.
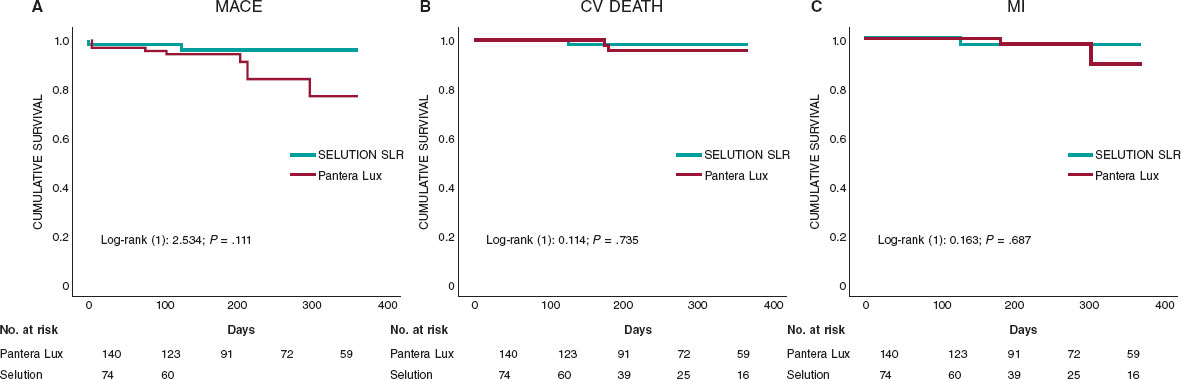
Figure 2. Central illustration. Study design and main outcomes. A total of 257 patients with 316 de novo coronary lesions were treated using a stentless percutaneous coronary intervention strategy with paclitaxel-coated balloon and 1-year follow-up. Outcomes were compared between the Pantera Lux paclitaxel-coated balloon and SELUTION SLR sirolimus-eluting balloon. SELUTION SLR was associated with fewer procedural complications, including lower rates of coronary dissection and no-reflow, with comparable clinical outcomes at follow-up. CV, cardiovascular; MI, myocardial infarction; MACE, major adverse cardiovascular events; PCI, percutaneous coronary intervention; TLF, target lesion failure.
The results of our study offer important insights into the real-world performance of sirolimus-DCB, particularly the novel SELUTION SLR platform, in the management of de novo coronary lesions. The findings are 2-fold: first, the SELUTION SLR demonstrated a safety and efficacy profile comparable to that of an established PCB; and second, overall, outcomes were consistent across vessel diameters, with no significant reduction in performance observed between small- and large-caliber arteries.
The principal observation of the present analysis is that SELUTION SLR yielded clinical outcomes similar to those obtained with the Pantera Lux, while being associated with fewer procedural findings such as mild, non–flow-limiting dissections, and no-reflow phenomena. This finding may reflect differences in mechanical interaction or coating characteristics between the 2 platforms. However, because factors such as lesion preparation, severity of calcification, balloon-to-artery ratio, and operator technique strongly influence dissection rates—and were neither controlled nor adjusted for in this analysis—this interpretation remains speculative. Furthermore, mild angiographic type A–B dissections are widely regarded as expected and benign features of DCB angioplasty, provided TIMI grade-3 flow is maintained and no significant recoil is present. Recent consensus documents indicate that such dissections rarely require stenting and may even facilitate drug transfer. Of note, all 5 cases of no-reflow occurred in Pantera Lux–treated lesions, an observation consistent with prior preclinical and clinical work documenting the embolic behavior of crystalline paclitaxel particles.6 Although this mechanistic explanation is biologically plausible, further studies in humans are required to confirm this association. Given the small number of events observed in both groups, the clinical relevance of these findings remains uncertain.
Paclitaxel has remained the backbone of DCB therapy for CAD for over a decade, primarily due to its lipophilic properties, rapid tissue uptake, and strong antiproliferative effects.7 However, emerging concerns regarding delayed healing, potential toxicity, and long-term safety have prompted the development of balloons with alternative antiproliferative agents.8 Sirolimus, although more hydrophilic and requiring innovative delivery systems to ensure sustained arterial wall absorption, has demonstrated superior biocompatibility, endothelial healing properties, and reduced inflammatory responses in both preclinical and early clinical settings. Nonetheless, the results from the TRANSFORM I trial were disappointing.9 By not being as cytotoxic, it became clear from this trial that platforms using sirolimus had to ensure sustained drug delivery to the artery over a long period of time and become a “drug-eluting balloon” rather than a “drug-coated balloon”. Hence, the SELUTION SLR incorporates a proprietary micro-reservoir technology with biodegradable polymers to facilitate sustained sirolimus release over 60–90 days, aiming to mimic the drug release kinetics of second-generation DES, and ensure drug activity over the traditional restenosis cycle period of coronary arteries after balloon barotrauma9,10.
In our study cohort comprising both small and large vessels, the procedural success and short-term lesion-oriented outcomes, namely TLF and TLR, were low and statistically similar between the SELUTION SLR and the Pantera Lux platforms. These observations are consistent with data from the PRESTIGE trial, which have demonstrated non-inferior late lumen loss (LLL) and binary restenosis rates with SELUTION SLR vs a paclitaxel DCB in a smaller cohort of patients including de novo lesions and in-stent restenosis.11 We reported very low rates of TLR (0.9% of treated lesions). We believe this finding is likely multifactorial. First, this was a selected population in which most treated vessels were small (< 3.0 mm). In small vessels, the amount of myocardium at risk is limited, and both procedural failures and late vessel occlusions may remain clinically silent and therefore undetected. In our subgroup analysis, we did not find any differences in TLR between small and larger vessels, however, our sample size is underpow- ered to detect such differences. Notably, in the REC-CAGEFREE I trial, the higher event rates observed in larger vessels ultimately drove the trial results.12 Second, angiographic follow-up was not mandated by protocol, and we acknowledge that asymptomatic restenosis or late vessel occlusion may have gone undetected and may or may not be equally distributed across treatment groups.
The bulk of the existing evidence favoring PCB comes from large randomized trials such as the PEPCAD II, the FEMPAC, and the BELLO, which provided robust comparative data across various lesion subsets and anatomical territories.13-15 However, PCB are not without limitations, including concerns regarding late thrombotic events, dose-dependent cytotoxicity, and secondary vasomotor and endothelial dysfunction.16 In fact, the toxicity profile of PCB was made evident in patients who were treated for chronic total coronary occlusions (CTO) and demonstrated post-PCI late coronary aneurysms, either due to > 1 PCB being applied in the same territory, as paclitaxel has a very narrow therapeutic window, or by having the drug delivered in false lumen segments and being directly applied to the vascular smooth muscle cell layer.17 Moreover, animal studies have shown that, while PCB and SEB have been shown to equally reduce neointimal formation, medial muscle cell layer loss was greater with PCB, underscoring their higher toxicity profile.16 Furthermore, various reports have noted the occurrence of paclitaxel crystal embolization into distal vascular territories after balloon inflation both in peripheral and myocardial tissues.18,19 This phenomenon might help explain the increased occurrence of no-reflow in the PCB group in our study.
The differential performance of DCBs in small vs large coronary arteries is another critical consideration. Traditionally, small vessels have posed a greater challenge for DES due to increased risk of restenosis, higher neointimal proliferation, and limited expansion capacity.20 By avoiding permanent implants and associated foreign body responses, DCBs are ideally suited for small vessel interventions. Our analysis confirms this advantage, with low rates of TLF and TLR being reported in small arteries, and no significant increase in procedural complications vs larger vessels. In fact, the overall MACE rate at the 1-year follow-up was 5.8%, which is well within the margins of current DES platforms, with 1 exception being the FireBird stent in the REC-CAGEFREE I trial. In this trial, patients with de novo coronary lesions treated with PCB (Swide, Shenqi Medical, China) exhibited a 2-year MACE rate of 6.4%, while patients treated with the FireBird stent, a sirolimus eluting stent, exhibited a 2-year MACE rate of 3.4%, well under the expected rate for DES platforms.12 The good performance of DES in this study was likely due to the inclusion of simple lesions and high rates of plaque modification techniques before stenting (scoring and cutting balloons being used in > 60% of cases), suggesting that good plaque preparation not only is fundamental for DCB PCI outcomes, but also seems to improve them in the context of DES PCI.
Interestingly, the large vessel subgroup in our cohort demonstrated a higher rate of bailout DES implantation (18.6% vs 7.8%; P = .011), despite comparable pre-treatment angiographic severity and lesion characteristics. This finding may reflect either a procedural bias—operators may be more inclined to stent larger vessels when faced with apparently suboptimal DCB results—or true differences in biomechanical response and vessel wall compliance between vessel calibers. Nevertheless, these findings did not translate into differences in safety endpoints or short-term clinical outcomes, highlighting the procedural adaptability and safety of the DCB-only approach in larger arteries when managed judiciously.
In the SELUTION DeNovo trial (NCT04859985),21 a strategy using the SELUTION SLR sirolimus-eluting balloon with provisional stenting achieved a 1-year TVF rate of approximately 5.3% vs 4.4% in the systematic DES arm, meeting the prespecified non-inferiority margin. Notably, nearly 80% of patients randomized to the SELUTION arm were successfully treated without any stenting, reinforcing the feasibility of a “leave-nothing-behind” approach in appropriately prepared de novo lesions. These results are consistent with the observations of our registry, where SELUTION SLR demonstrated comparable clinical outcomes to its direct PCB comparator, Pantera Lux, along with fewer procedural disturbances such as mild dissections and no-reflow. While the SELUTION DeNovo findings support the safety and efficacy profile of SELUTION in a controlled randomized environment,21 it is important to note that the trial included carefully selected patients—excluding left main disease, STEMI, CTO, and other high-risk anatomies—whereas our registry reflects real-world lesion complexity and operator-driven device selection. Consequently, although the procedural and clinical performance of SELUTION in this study appears directionally consistent with DeNovo, extrapolation should remain cautious given differences in inclusion criteria, lesion profiles, and the non-randomized nature of our dataset. Together, both datasets reinforce the potential of SELUTION SLR as a viable stentless strategy, while highlighting the need for further randomized clinical trials with broader inclusion criteria and extended follow-up to determine long-term durability > 1 year. Larger, randomized trials with extended clinical endpoints are essential to confirm the long-term efficacy and durability of SEB, particularly in complex anatomical settings such as bifurcations, diffuse disease, and high-risk diabetic or chronic kidney disease populations.
Limitations
Interpretation of the present results must balance the strengths of an all-comers design with the inherent weaknesses of observational registries. The absence of randomization introduces the possibility of confounding by indication, particularly regarding device selection and bailout stenting decisions. Notably, clinical event rates in this analysis were low across the 2 devices, limiting statistical power to detect small or moderate differences and rendering subgroup analyses descriptive rather than inferential. Given the observational nature of the study and the low number of clinical events, formal propensity score adjustment was not performed, which limits causal inference despite comparable baseline prevalence of key complexity markers. Furthermore, angiographic follow-up was opportunistic rather than protocol-mandated, which may introduce verification bias, as patients returning for angiography may differ meaningfully from those who did not. Despite these constraints, the consistency of the clinical findings, together with the observed procedural advantages, lends support to the feasibility of a sirolimus-eluting balloon strategy in well-prepared de novo lesions. Finally, given the nature of our study, our results are mainly hypothesis generating rather than hypothesis testing and larger studies are required to validate our observations.
CONCLUSIONS
In conclusion, the SELUTION SLR demonstrated comparable 12-month clinical outcomes to the Pantera Lux while showing fewer procedural disturbances and numerically favorable angiographic trends. These results reinforce the role of DCB as a viable stentless strategy in appropriately selected de novo lesions and support continued evaluation of sirolimus-based DCB technologies in larger randomized clinical trials.
ETHICAL CONSIDERATIONS
Ethical approval was obtained from our institutional review board. The study was conducted in full compliance with the principles outlined in the Declaration of Helsinki and follows the SAGER guidelines
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
A large language model was used to improve text readability and fluency.
AUTHORS’ CONTRIBUTIONS
D. Faria was responsible for the study design, data collection, statistical analysis and drafting. D. Neves was responsible for review and drafting. L. Hamann was responsible for data collection. J. Guedes, J. Bispo, F. Soares and H. Vinhas were responsible for manuscript review.
CONFLICTS OF INTEREST
D. Faria has received speaker fees from Cordis. H. Vinhas declared to have received speaker fees from Cordis. F. Soares declared to have received consultant fees from Cordis and Biotronik. The remaining authors declared no conflicts of interest whatsoever.
FUNDING
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- DCBs are an established stentless treatment option for selected coronary lesions, with PCB being supported by extensive clinical evidence.
- More recently, SEB technologies have been developed to address potential limitations of paclitaxel-based platforms, but comparative real-world data in de novo coronary lesions remain limited.
WHAT DOES THIS STUDY ADD?
- In this prospective cohort of 257 patients with de novo coronary artery disease, the SELUTION SLR SEB demonstrated similar 1-year clinical outcomes vs the Pantera Lux paclitaxel-coated balloon, with fewer procedural complications.
- These findings support the safety and feasibility of SEB as a stentless treatment strategy, while underscoring the need for confirmation in randomized clinical trials.
REFERENCES
1. Alfonso F, Byrne RA, Scheller B, van Belle E, Mehilli J. Drug-coated balloon angioplasty for in-stent restenosis:pros and cons. EuroIntervention. 2025;21:e102-e104.
2. Cortese B, Di Palma G, Guimaraes MG, et al. Drug-Coated Balloon Versus Drug-Eluting Stent for Small Coronary Vessel Disease:PICCOLETO II Randomized Clinical Trial. JACC Cardiovasc Interv. 2020;13:2840-2849.
3. Jeger RV, Farah A, Ohlow MA, et al. BASKET-SMALL 2 Investigators. Drug-coated balloons for small coronary artery disease (BASKET-SMALL 2):an open-label randomised non-inferiority trial. Lancet. 2018;392:849-856.
4. Sedhom R, Hamed M, Elbadawi A, et al. Outcomes With Limus- vs Paclitaxel-Coated Balloons for Percutaneous Coronary Intervention:Meta-Analysis of Randomized Controlled Trials. JACC Cardiovasc Interv. 2024;17:1533-1543.
5. Madanchi M, Cioffi GM, Attinger-Toller A, et al. Metal free percutaneous coronary interventions in all-comers:First experience with a novel sirolimus-coated balloon. Cardiol J. 2022;29:906-916.
6. Kawai K, Kolodgie FD, Kawakami R, et al. Vascular Response, Downstream Effect, and Pharmacokinetics After Sirolimus- and Paclitaxel-Coated Balloons in Porcine Coronary Arteries. Catheter Cardiovasc Interv. 2025;105:1434-1444.
7. Loh JP, Waksman R. Paclitaxel drug-coated balloons:a review of current status and emerging applications in native coronary artery de novo lesions. JACC Cardiovasc Interv. 2012;5:1001-1012.
8. Shin D, Singh M, Shlofmitz E, et al. Paclitaxel-coated versus sirolimus-coated balloon angioplasty for coronary artery disease:A systematic review and meta-analysis. Catheter Cardiovasc Interv. 2024;104:425-436.
9. Ninomiya K, Serruys PW, Colombo A, et al. A Prospective Randomized Trial Comparing Sirolimus-Coated Balloon With Paclitaxel-Coated Balloon in De Novo Small Vessels. JACC Cardiovasc Interv. 2023;16:2884-2896.
10. Costa MA, Simon DI. Molecular basis of restenosis and drug-eluting stents. Circulation. 2005;111:2257-2273.
11. Costa RA, Mandal SC, Hazra PK, et al. Sirolimus-Coated Balloon With a Microsphere-Based Technology for the Treatment of De novo or Restenotic Coronary Lesions. Cardiovasc Revasc Med. 2022;45:18-25.
12. Gao C, He X, Ouyang F, et al. Drug-coated balloon angioplasty with rescue stenting versus intended stenting for the treatment of patients with de novo coronary artery lesions (REC-CAGEFREE I):an open-label, randomised, non-inferiority trial. Lancet. 2024;404:1040-1050.
13. Unverdorben M, Vallbracht C, Cremers B, et al. Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis:the three-year results of the PEPCAD II ISR study. EuroIntervention. 2015;11:926-34.
14. Latib A, Colombo A, Castriota F, et al. A randomized multicenter study comparing a paclitaxel drug-eluting balloon with a paclitaxel-eluting stent in small coronary vessels:the BELLO (Balloon Elution and Late Loss Optimization) study. J Am Coll Cardiol. 2012;60:2473-2480.
15. Werk M, Langner S, Reinkensmeier B, et al. Inhibition of restenosis in femoropopliteal arteries:paclitaxel-coated versus uncoated balloon:femoral paclitaxel randomized pilot trial. Circulation. 2008;118:1358-1365.
16. Nakamura T, Brott BC, Brants I, et al. Vasomotor function after paclitaxel-coated balloon post-dilation in porcine coronary stent model. JACC Cardiovasc Interv. 2011;4:247-255.
17. Jun EJ, Shin ES, Kim B, et al. Coronary artery aneurysm formation after paclitaxel-coated balloon-only intervention for de novo coronary chronic total occlusion. Front Cardiovasc Med. 2023;9:1039316.
18. Kawai K, Kolodgie FD, Kawakami R, et al. Vascular Response, Downstream Effect, and Pharmacokinetics After Sirolimus- and Paclitaxel-Coated Balloons in Porcine Coronary Arteries. Catheter Cardiovasc Interv. 2025;105:1434-1444.
19. Boitet A, Grassin-Delyle S, Louedec L, et al. An Experimental Study of Paclitaxel Embolisation During Drug Coated Balloon Angioplasty. Eur J Vasc Endovasc Surg. 2019;57:578-586.
20. Giustino G, Colombo A, Camaj A, et al. Coronary In-Stent Restenosis:JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;80:348-372.
21. Spaulding C, Krackhardt F, Bogaerts K, et al. Comparing a strategy of sirolimus-eluting balloon treatment to drug-eluting stent implantation in de novo coronary lesions in all-comers: Design and rationale of the SELUTION DeNovo Trial. Am Heart J. 2023;258:77-84.














