To the Editor,
Evaluation of chest pain remains a frequent clinical challenge, with ongoing debate surrounding the optimal diagnostic strategy. Among the available modalities, coronary computed tomography angiography (CCTA) has emerged as a reliable diagnostic imaging modality because of its high sensitivity and negative predictive value. However, its positive predictive value remains moderate (60%–70%) vs invasive coronary angiography (ICA) and stress imaging modalities. This limitation often results in increased downstream testing, as demonstrated by the PROMISE trial.1 Recent technological advancements have enabled non-invasive assessment of the hemodynamic significance of coronary lesions, allowing fractional flow reserve derived from computed tomography (FFR-CT) to be calculated directly from CCTA images without the need for pharmacological stress agents or additional radiation exposure. Previous prospective trials have demonstrated a significant enhancement in the diagnostic accuracy of FFR-CT vs CCTA alone. As a result, the United States, the United Kingdom and European clinical practice guidelines currently endorse the use of FFR-CT. However, uncertainties remain regarding its clinical effectiveness and cost-efficiency across different health care systems and clinical settings.
We evaluated the potential clinical and economic impact of integrating FFR-CT into the routine practice in Spain vs standard care in patients with moderate stenosis on coronary computed tomography angiography (CCTA). We conducted a retrospective, observational, single-center intervention simulation study including outpatients with stable chest pain who underwent CCTA from July 2021 through December 2022 at a Spanish university hospital. Eligible patients had lesions classified as CAD-RADS 2.0 grade 3 (50%–69% stenosis) in vessels ≥ 1.5 mm,2 body mass index (BMI < 35 kg/m²) and coronary artery calcium score < 1000 Agatston Units (AU). Patients were identified through administrative databases, and electronic health records were reviewed to collect baseline clinical and imaging data. CCTA reports were independently reviewed by 5 clinical cardiologists, who recommended additional tests or treatments according to their clinical judgment. The same process was repeated after incorporation of FFR-CT to determine how functional information influenced decision-making. For each scenario, cardiologists answered questions, both before and after FFR-CT analysis: a) What additional diagnostic tests would you request? b) Would you recommend hospital admission, outpatient management, or discharge? c) What treatment strategy would you propose? d) Was the FFR-CT information useful? (figure 1). This structure enabled direct comparison of anatomical-only decisions vs those incorporating functional data. Significant FFR was defined as at least 1 vessel with FFR ≤ 80%.
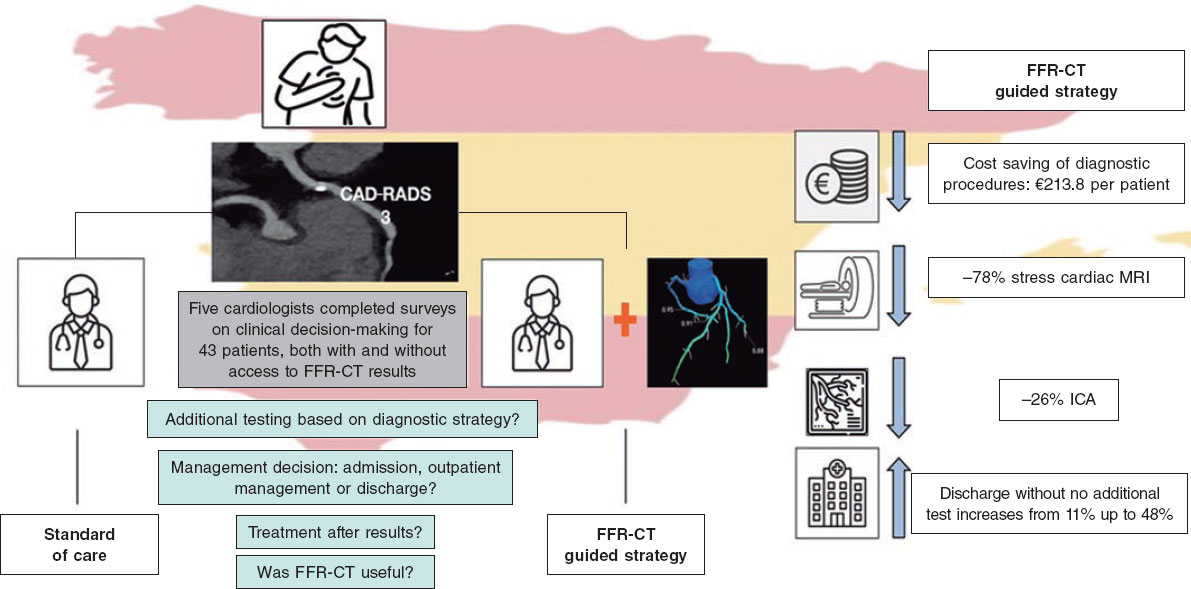
Figure 1. Study design and outcomes. CAD-RADS, coronary artery disease-reporting and data system; CT, computed tomography; FFR, fractional flow reserve; ICA, invasive coronary angiography; MRI, magnetic resonance imaging.
CCTA images were acquired using a dual-source scanner (Somatom Drive, Siemens Healthineers, Germany) and interpreted by cardiologists and cardiovascular radiologists. FFR-CT analysis was performed by experts blinded to the original CCTA findings using DEEPVESSEL-FFR (Keya Medical Technology Inc; China), a new machine learning-based software platform that uses artificial intelligence and deep learning algorithms to generate FFR-CT values directly from CCTA datasets.3
The primary endpoint was total cost generated or saved after incorporating FFR-CT. Secondary endpoints included changes in tests utilization, hospitalizations decisions, adjustments in optimal medical therapy and physician-perceived satisfaction after FFR-CT analysis.
Baseline characteristics are expressed as frequencies and percentages for categorical data, and as mean ± standard deviation for continuous data. A cost analysis assessed the financial impact of the clinical decisions made in the intervention simulation, calculating total costs generated or avoided accordingly. Reference prices were obtained from our regional health care system and updated to 2024 using the Consumer Price Index.4 The company-reported cost of each FFR-CT analysis was €400. All statistical analyses were performed using Stata 16.1; P values <.05 were considered statistically significant.
Among 631 outpatients who underwent CCTA, 43 (6.8%) met the inclusion criteria. Most were men, with a mean age of 64.7 ± 8.6 years. The mean Agatston score was 355.7 AU, reflecting a high coronary calcium burden. Dyslipidemia and hypertension were present in fewer than one-half of patients, and diabetes was documented in 11 (25.6%). The mean body mass index was 27.6 ± 4.3 kg/m², and mean radiation exposure was low (1.9 ± 1.1 millisievert). A total of 83.7% of patients underwent single-beat acquisition (Flash); 7%, prospective (‘step-and-shoot’) acquisition; and 9%, retrospective acquisition. FFR-CT analysis was not feasible in 3 cases (7%) due to motion artifacts. Among successful analyses, 5 patients (11.6%) had a positive FFR-CT result.
In the clinical simulation analysis, 215 clinical scenarios were finally evaluated, corresponding to the individual assessment of all 43 cases by each of the 5 participating cardiologists. Prior to FFR-CT, additional testing was recommended in 89.9%, based solely on CCTA findings. The most frequently requested imaging modalities were stress cardiac magnetic resonance (CMR) (n = 118; 54.8%), and ICA (n = 46; 21%). After FFR-CT, the need for further testing dropped down to 28% (P < .01), with marked reductions in stress CMR (to 12%, -78%; P < .01) and ICA (to 15%, -26%; P < .01), respectively (table 1). This shift resulted in notable cost savings: total diagnostic costs decreased from €301 690 to €255 720, yielding savings of €45 976.64 (€213.84 per case). Discharge recommendations increased from 11% to 48% after FFR-CT (P <.01). Clinicians found FFR-CT helpful in 81% of scenarios. There were no significant changes in decisions regarding medical therapy.
Table 1. Cost analysis based on clinical simulation analysis
| Complementary tests | Before CT-FFR | Total cost (CT + additional testsa) (€) | After CT-FFR | Total cost (CT + CT-FFR + additional testsa,b) (€) | P-value | Balance (€) |
|---|---|---|---|---|---|---|
| ICA-no. (%) | 46 (21%) | 80 865.99 | 32 (15%) | 69 024.64 | < .01 | |
| SPECT-no. (%) | 9 (4%) | 9180.98 | 1 (0.5%) | 1420.11 | .01 | |
| Stress MRI-no. (%) | 118 (55%) | 183 383.24 | 25 (12%) | 48 850.50 | < .01 | |
| Stress echo-no. (%) | 19 (8%) | 17 601.98 | 2 (0.9%) | 2652.04 | < .01 | |
| No test-no. (%) | 23 (10%) | 10 649.92 | 155 (72%) | 133 768.1 | < .01 | |
| Total | 301 692.04 | 255 715.4 | 45 976.64 | |||
|
CT, computed tomography; FFR, fractional flow reserve; ICA, invasive coronary angiography; MRI, magnetic resonance imaging; SPECT, single photon emission computed tomography. a Cost Regional Healthcare system December 2024 (updated to 2024 values based on the Consumer Price Index) (€): ICA = 1294.95; SPECT = 557.09; Stress MRI = 1091.16; CTA = 463.02; Stress echo = 463.40 b The cost of CT-FFR was estimated at € 400 per study. |
||||||
This intervention simulation study provides several clinically relevant insights. First, FFR-CT was associated with lower health care spending and fewer downstream testing. Second, it enabled completion of the diagnostic process in a high proportion of patients and was judged clinically useful in most cases. As the first study in Spain evaluating an FFR-CT–guided strategy, it suggests that routine implementation could decrease downstream investigations and associated costs in patients with stable chest pain.
Most cost-efficiency data comes from the United States and the United Kingdom, where FFR-CT adoption is higher. While in the United States, the PLATFORM study demonstrated cost reductions, primarily driven by decreased use of ICA,5 evidence from the United Kingdom is more heterogeneous. While the latest NICE update projected £391 savings per patient, Mittal et al. reported higher costs vs stress imaging, although these findings have been questioned because most patients had stenoses outside the 50%–69% range—where additional testing and interobserver variability are more frequent—and the observed cost increase was largely attributable to higher ICA rates, findings that contradict earlier reports.6 In our cohort, stress CMR (54.8%) and ICA (21%) decreased substantially after incorporation of FFR-CT (to 12% and 15%), resulting in savings of €213.84 per case.
In addition, FFR-CT effectively concluded the diagnostic process, increasing the discharge rate from 11% to 48% after FFR-CT (P < .01). This notable finding introduces an important dimension of organizational efficiency, as it may help optimize imaging resources and reduce waiting times for advanced diagnostic tests. Notably, previous cost evaluations have primarily focused on avoidance of unnecessary testing, and have not fully captured this potential management benefit. Furthermore, clinicians participating in the study considered FFR-CT highly useful in 87% of cases.
The main limitation of our study is its simulation-based design. Economic outcomes were influenced by local test utilization patterns and regional pricing structures, which may limit external validity and generalizability. Therefore, the results should be considered hypothesis-generating.
In conclusion, in this simulation study, incorporation of FFR-CT in patients with stable chest pain and moderate coronary stenosis on coronary CCTA was associated with reduced downstream testing, lower health care costs, and higher discharge rates within the Spanish National Health System.
DATA AVAILABILITY
The data underlying this study will be made available upon reasonable request. Requests should be directed to the corresponding author.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
This study was conducted in full compliance with the principles outlined in the Declaration of Helsinki and the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines on clinical research. As this was an interventional simulation study based on previously collected and fully anonymized data, formal ethical approval and informed consent were deemed unnecessary. Although this study included male and female patients alike, sex disaggregated analyses were not performed due to lack of expected sex related differences.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence has been used in the development of this paper.
CONFLICTS OF INTEREST
None declared.
AUTHORS’ CONTRIBUTIONS
Conceptualization, formal analysis, drafting of the original manuscript, and supervision were performed by J.A. Parada-Barcia and M. Barreiro-Pérez. Critical review and editing of the manuscript were undertaken by the remaining authors. All authors approved the final version of the manuscript for publication and accept all aspects of the work.
REFERENCES
1. Douglas PS, Pontone G, Hlatky MA, et al. Clinical outcomes of FFR-CT guided diagnostic strategies vs. usual care in patients with suspected coronary artery disease:the prospective longitudinal trial of FFR-CT:outcome and resource impacts study. Eur Heart J. 2015;36:3359-3367.
2. Cury RC, Leipsic J, Abbara S, et al. 2022 Coronary Artery Disease-Reporting and Data System:An Expert Consensus Document of the SCCT, ACC, ACR, and NASCI. J Cardiovasc Comput Tomogr. 2022;16:536-557.
3. Wang ZQ, Zhou YJ, Zhao YX, Shi DM, Liu YY, Liu W. Diagnostic accuracy of a deep learning approach to calculate FFR from coronary CT angiography. J Geriatr Cardiol. 2019;16:42-48.
4. Diario Oficial de Galicia. Decreto 56/2014. DOG. 2014;96:22788.
5. Hlatky MA, De Bruyne B, Pontone G, et al. Quality-of-life and economic outcomes of assessing FFR-CT:PLATFORM. J Am Coll Cardiol. 2015;66:2315-2323.
6. Mittal TK, Hothi SS, Venugopal V, et al. The Use and Efficacy of FFR-CT:Real-World Multicenter Audit of Clinical Data With Cost Analysis. JACC Cardiovasc Imaging. 2023;16:1056-1065.















