ABSTRACT
Introduction and objectives: To compare clinical outcomes of immediate vs staged multivessel percutaneous coronary intervention (PCI) for non-culprit lesions in hemodynamically stable patients with ST-segment elevation myocardial infarction and multivessel coronary artery disease.
Methods: We conducted this systematic review and meta-analysis according to PRISMA guidelines. PubMed, Embase, and Cochrane databases were searched throughout September 2025. Outcomes included cardiac death, all-cause mortality, reinfarction, acute kidney injury, and stroke. Risk ratios were calculated, and trial sequential analysis, incidence rate ratio (IRR) analyses, and meta-regression were conducted.
Results: Six randomized controlled trials including 4267 patients were analyzed. At the 1-year follow-up, there were no significant differences between immediate and staged PCI regarding cardiac death (RR, 1.26; 95%CI, 0.89-1.79), all-cause mortality (RR, 1.18; 95%CI, 0.93-1.50), acute kidney injury (RR, 0.91; 95%CI, 0.64-1.31), or stroke (RR, 1.17; 95%CI, 0.66-2.05). Immediate PCI was associated with a lower risk of reinfarction (RR, 0.63; 95%CI, 0.40-0.98), a finding confirmed by IRR analysis (IRR, 0.60; 95%CI, 0.39-0.94; I2 = 0%). Exploratory meta-regression analyses did not identify procedural timing or baseline comorbidities as significant effect modifiers.
Conclusions: In patients with ST-segment elevation myocardial infarction and multivessel disease, immediate PCI was associated with a lower risk of reinfarction, without any significant differences being reported in cardiac death or all-cause mortality compared with staged PCI. Although trial sequential analysis indicates that current evidence remains underpowered for certain outcomes, the consistency of findings across multiple analytical approaches supports the safety profile of immediate complete revascularization, pending confirmation in adequately powered randomized trials. (PROSPERO: CRD42023485163.I).
Keywords: Coronary artery disease. ST-segment elevation myocardial infarction. Multivessel disease. Immediate percutaneous coronary intervention. Staged percutaneous coronary intervention.
RESUMEN
Introducción y objetivos: Esta revisión sistemática con metanálisis evalúa si una estrategia de intervención coronaria percutánea (ICP) multivaso por etapas o inmediata proporciona resultados clínicos superiores en pacientes hemodinámicamente estables con infarto agudo de miocardio con elevación del segmento ST y enfermedad coronaria multivaso sometidos a revascularización de lesiones no culpables.
Métodos: Se realizaron búsquedas en PubMed, Embase y Cochrane hasta septiembre de 2025 según las directrices PRISMA. Los resultados incluyeron muerte por causa cardiaca, muerte por cualquier causa, reinfarto, lesión renal aguda y accidente vascular cerebral. Se estimaron los riesgos relativos y se realizó un análisis secuencial de ensayos en desenlaces seleccionados. Además, se calcularon las tasas de incidencia relativa (IRR) y se realizó una metarregresión.
Resultados: Se incluyeron 6 ensayos clínicos aleatorizados con 4.267 pacientes. En el seguimiento al año no se encontraron diferencias significativas entre la ICP por etapas y la inmediata en el riesgo de muerte por causa cardiaca (RR = 1,26; IC95%, 0,89-1,79), muerte por cualquier causa (RR = 1,18; IC95%, 0,93-1,50), lesión renal aguda (RR = 0,91; IC95%, 0,64-1,31) o accidente vascular cerebral (RR = 1,17; IC95%, 0,66-2,05). La ICP inmediata se asoció con un menor riesgo de reinfarto (RR = 0,63; IC95%, 0,40-0,98), confirmado por el análisis de tasa de incidencia relativa (IRR = 0,60; IC95%, 0,39-0,94; I² = 0%). La metarregresión no mostró asociación significativa entre el riesgo de reinfarto y el momento de la ICP por etapas o la comorbilidad basal.
Conclusiones: La ICP inmediata en pacientes con infarto agudo de miocardio con elevación del segmento ST y enfermedad multivaso se asoció con un menor riesgo de reinfarto, sin diferencias significativas en la mortalidad por causa cardiaca ni en la mortalidad por cualquier causa frente a la ICP por etapas. Aunque el análisis secuencial de ensayos indicó que la evidencia actual aún carece de suficiente poder estadístico, la consistencia en la dirección del efecto a través de múltiples enfoques analíticos respalda la seguridad y la posible ventaja clínica de la revascularización completa inmediata, lo que justifica su confirmación en futuros ensayos aleatorizados adecuadamente dimensionados. (PROSPERO: CRD42023485163.I).
Palabras clave: Enfermedad coronaria. Infarto agudo de miocardio con elevación del segmento ST. Enfermedad multivaso. Intervención coronaria percutánea inmediata. Intervención coronaria percutánea por etapas.
Abbreviations
AKI: acute kidney injury; PCI: percutaneous coronary intervention. STEMI: ST-segment elevation myocardial infarction.
INTRODUCTION
Multivessel coronary artery disease is defined as the presence of ≥ 70% luminal stenosis in at least 2 major epicardial coronary arteries due to atherosclerotic plaque. Approximately 40% of patients with ST-segment elevation myocardial infarction (STEMI) exhibit multivessel coronary disease, which is associated with an increased risk of recurrent myocardial infarction and mortality.1-3 Evidence from randomized controlled trials has underscored the advantages of comprehensive revascularization with multivessel percutaneous coronary intervention (PCI) over culprit-lesion-only PCI, notably reducing the risks of cardiac death, myocardial infarction, and ischemia-driven revascularization at 1 year.4-6 There are 2 strategies to approach non-culprit lesions: immediate PCI which involves the revascularization of both culprit and non-culprit lesions during the index procedure, and staged PCI, where the treatment of non-culprit lesions is deferred to a later stage.
The 2021 clinical practice guidelines outlined by the American College of Cardiology/American Heart Association (ACC/AHA) acknowledge non-culprit vessel revascularization as a class IIb recommendation for patients with STEMI.7 However, determining the optimal timing for revascularization of non-culprit lesions remains a clinical challenge7,8 While PCI for non-culprit lesions has demonstrated superiority over a strategy of treating the culprit lesion alone,4-6 the timing of intervention, whether immediate or staged, lacks conclusive evidence.8,9
The objective of this meta-analysis was to compare staged vs immediate PCI for non-culprit lesions in hemodynamically stable patients with STEMI and multivessel disease. We hypothesize that both the staged and immediate PCI targeting non-culprit vessels will demonstrate comparable levels of safety, efficacy, and rate of complications.
METHODS
We conducted the present study following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines.10 All steps were conducted in full compliance with the Cochrane Handbook of Systematic Reviews and Meta-analysis of Interventions (version 6.3).11 This meta-analysis protocol was registered on PROSPERO on 4 December, 2023, under protocol ID: CRD42023485163.l. During the peer-review process, the literature search was updated using the same search strategy, databases, and eligibility criteria described in supplementary data S1 to identify newly published randomized evidence.
Criteria of the included studies
We evaluated randomized controlled trials comparing immediate vs staged PCI for non-culprit lesions in adults with STEMI and multivessel coronary artery disease. Eligible studies were required to report extractable numerical data for at least 1 prespecified outcome of interest, as defined below. Trials reporting outcomes exclusively as Kaplan-Meier curves without corresponding event counts or effect estimates or reporting outcomes only as composite endpoints without separate data, were excluded from quantitative synthesis. No publication time limit was applied; however, the search was restricted to studies published in Spanish, English, or German. Case-control and cross-sectional studies, systematic reviews, meta-analyses, case reports, basic science research, conference abstracts, and letters to the editor were excluded.
Literature search strategy
We conducted a comprehensive and systematic search across 3 databases (PubMed, EMBASE, and COCHRANE) from their inception until 28 September, 2025. Keywords used for the search included “coronary artery disease”, “immediate multivessel percutaneous coronary intervention”, “staged multivessel percutaneous coronary intervention”, “acute coronary syndrome”, “multivessel disease”, “myocardial infarction”, “major adverse cardiovascular events”, “ST-segment elevation myocardial infarction”. These keywords were combined using Boolean operators AND and OR. The search details for each database are available in the supplementary data S1.
Screening of literature search results
All duplicates were removed using Zotero reference manager software. Records were initially screened by title and abstract by one author (E. Andrade-Arbaiza), followed by full-text assessment when potentially eligible. Two independent authors (D. Paulino-González, and L.H. García-Mena) reviewed full-text articles for inclusion and methodological quality, with disagreements being resolved by consensus or third-party adjudication (D. A. Navarro-Martínez). Studies were excluded at the full-text stage if they lacked extractable numerical data for prespecified outcomes or did not report outcomes separately, precluding quantitative synthesis. A detailed list of excluded studies with reasons for exclusion is provided in table S1. References cited in included studies were also manually screened for eligibility.
Data extraction
Data extraction was performed using standardized Excel spreadsheets to collect baseline population characteristics, key study features, outcome measures expressed as risk ratios, and domains for quality assessment. Outcome data were included only when numerical information was directly available or could be reliably derived without reconstruction from survival curves.
Assessing the risk of bias
The risk of bias of included randomized controlled trials was assessed usingvVersion 2 of the Cochrane risk-of-bias tool for randomized trials (ROB 2), in accordance with the Cochrane Handbook of Systematic Reviews of Interventions.11
Endpoints
The prespecified endpoints of this meta-analysis were cardiac death, all-cause mortality, stroke, reinfarction, and acute kidney injury (AKI). Given minor variations in endpoint definitions across trials, detailed outcome definitions as reported by each study are summarized in table S2. Outcomes were analyzed as dichotomous variables using event counts and percentages, and risk ratios with 95% confidence intervals (95%CI) were calculated.
Data analysis
Dichotomous outcomes were analyzed using pooled event data from the included studies and are reported as risk ratios with 95%CI. P values < .05 were considered statistically significant. Associations between outcomes and immediate vs staged PCI were evaluated using a random-effects model (DerSimonian–Laird method) to account for between-study heterogeneity.12 Statistical analyses and forest plots were generated using RevMan version 5.4.1 (Cochrane Collaboration) for MacOs.
Statistical analysis
Pooled risk ratios (RR) and incidence rate ratios (IRR) were estimated using random-effects models with appropriate adjustments, and heterogeneity was assessed using the I2 statistic. Sensitivity analyses, trial sequential analysis, and meta-regression were performed to evaluate the robustness of findings and explore potential effect modifiers. Detailed methodological procedures are shown in the supplementary data S2.
Assessment of heterogeneity
Heterogeneity was assessed using Cochran’s Q test (P < .10) and quantified with the I2 statistic, with values > 50% indicating substantial heterogeneity.
RESULTS
A total of 6 studies13-18 met the predefined inclusion criteria and were included in the analysis. The study selection process is summarized in the PRISMA flow diagram shown in figure 1. All included studies evaluated staged or immediate PCI in non-culprit arteries of patients with STEMI and multivessel disease.
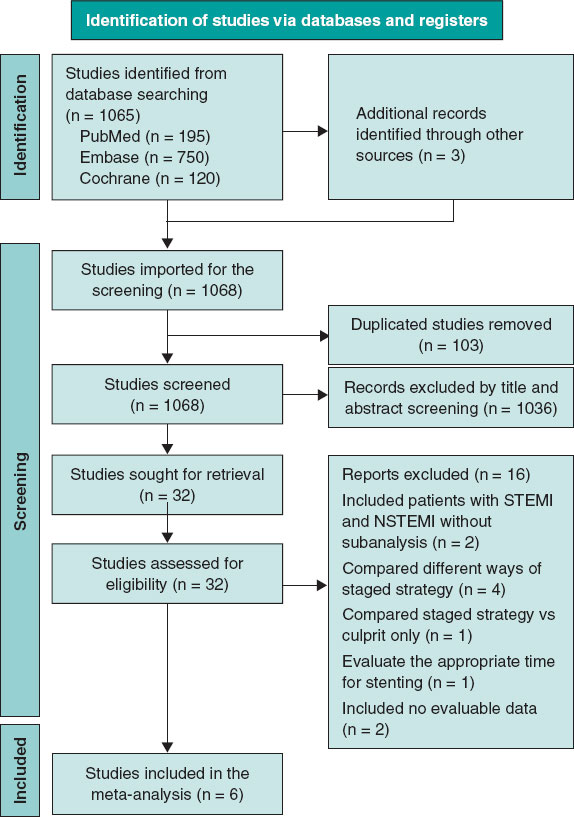
Figure 1. PRISMA flow diagram. The diagram reflects the initial search and the updated search performed during peer review using the same strategy and eligibility criteria (supplementary data S1). NSTEMI, non-ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction.
In the staged strategy, PCI of nonculprit lesions was performed a mean of 18.6 days (IQR, 7-34) after the index procedure. The total patient population included in our meta-analysis was 4267 patients, of whom 2479 underwent immediate PCI and 1788, staged PCI. Summary and baseline characteristics are shown in table 1. The characteristics of the included studies are summarized in table 2.
Table 1. Baseline characteristics and treatment of the included studies’ populations
| Study information | Baseline characteristics | Treatments and following intervention | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study ID | Country | Total population size | Intervention | Population size | Median [IQR] Age | Gender | Comorbidities | Culprit artery | Non-culprit artery | Access site index | Access site index + staged | Stent used index | Stent used index + staged | Volume of contrast index | Volume of contrast index + staged | Length of stay index | Length of stay index + staged | ||
| Kim et al.182025 | Multicenter | 994 | Immediate | 498 | 66 IQR [57-76] | F: 102 M: 396 |
Yes | Left main coronary artery | NR | Left main coronary artery | NR | Radial:371 Femoral: NR | Radial: 379 Femoral: NR | 2 | NR | 180 (140-225) | NR | 4 (3-6) | NR |
| NR | NR | ||||||||||||||||||
| Left anterior descending coronary artery | 221 | Left anterior descending coronary artery | 222 | ||||||||||||||||
| 234 | 212 | ||||||||||||||||||
| Staged | 496 | 65 IQR [58-76] | F: 103 M: 393 |
Left circumflex artery | 62 | Left circumflex artery | 218 | 1 | 2 | 130 (100-180) | 220 (170-298) | NR | 5 (4-8) | ||||||
| 67 | 215 | ||||||||||||||||||
| Right coronary artery | 215 | Right coronary artery | 162 | ||||||||||||||||
| 195 | 167 | ||||||||||||||||||
| Stähli et al.132023 | Multicentric | 840 | Immediate | 418 | 66 IQR [58-74] | F: 97 M:321 | Yes | Left main coronary artery | 0 | Left main coronary artery | 1 | Radial: 301 Femoral 117 | Radial: -- Femoral 296 | 3 (2-4) | NR | 250 (199-320) | NR | 4 (3-6) | NR |
| 1 | 4 | ||||||||||||||||||
| Left anterior descending coronary artery | 163 | Left anterior descending coronary artery | 205 | ||||||||||||||||
| 176 | 180 | ||||||||||||||||||
| Staged | 422 | 64 IQR [55-73] | F:81 M:341 | Left circumflex artery | 67 | Left circumflex artery | 196 | Radial: 311 Femoral 111 | Radial: -- Femoral 90 | 1 (1-2) | 3 (2-4) | 170 (130-220) | 333 (258-411) | 4 (3-6) | 5 (4-7) | ||||
| 77 | 152 | ||||||||||||||||||
| Right coronary artery | 188 | Right coronary artery | 134 | ||||||||||||||||
| 169 | 124 | ||||||||||||||||||
| Politi et al.142010 | Italy | 130 |
Immediate | 65 | 64.5 ± 11.7 | F: 15 M: 50 |
Yes | Left main coronary artery | NR | Left main coronary artery | NR | NR | NR | NR | NR | NR | NR | 4.8 ±2.6 | NR |
| NR | NR | ||||||||||||||||||
| Left anterior descending coronary artery | NR | Left anterior descending coronary artery | NR | ||||||||||||||||
| NR | NR | ||||||||||||||||||
| Staged | 65 | 64.5 ± 11.1 | F:13 M: 52 |
Left circumflex artery | NR | Left circumflex artery | NR | NR | NR | NR | NR | NR | NR | 5.4±3.1 | NR | ||||
| NR | NR | ||||||||||||||||||
| Right coronary artery | NR | Right coronary artery | NR | ||||||||||||||||
| NR | NR | ||||||||||||||||||
| Park et al.152023 | Multicentric | 209*Analysis of a subgroup | Immediate | 103 | 63.3 ± 10.4 | F:21 M:82 | Yes | Left main coronary artery | 1 | Left main coronary artery | 4 | Radial: 27 | -- | 2.58 ± 0.86 | -- | NR | -- | NR | NR |
| 1 | 3 | ||||||||||||||||||
| Left anterior descending coronary artery | 45 | Left anterior descending coronary artery | 79 | ||||||||||||||||
| 48 | 82 | ||||||||||||||||||
| Staged | 106 | 62.2 ± 10.9 | F 18 M: 88 |
Left circumflex artery | 12 | Left circumflex artery | 60 | Radial:25 | NR | 2.62 ± 1.00 | NR | NR | NR | NR | NR | ||||
| 17 | 55 | ||||||||||||||||||
| Right coronary artery | 46 | Right coronary artery | 78 | ||||||||||||||||
| 42 | 80 | ||||||||||||||||||
| 228 | 65 | ||||||||||||||||||
| Maamoun et al.162011 | Yemen | 78 | Immediate | 36 | 52.33 ± 7.1 | M: 32 F: 4 |
Yes | NR | NR | NR | NR | Femoral | Femoral | 2.38 ± 0.49 | 2.31 ± 0.47 | NR | NR | NR | NR |
| Staged | 42 | 54.52 ± 10.3 | M: 40 F: 2 |
||||||||||||||||
| Wood et al.172019 | Canada | 4041* | Immediate | 1353 | 61.8 ± 10.5 | M: 1079 F: 274 |
Yes | NR | NR | Left main coronary artery | 4 | Radial | Radial | NR | NR | NR | NR | NR | NR |
| 6 | |||||||||||||||||||
| Left anterior descending coronary artery | 683 | ||||||||||||||||||
| 384 | |||||||||||||||||||
| Staged | 663 | 61.2 ± 11.0 | M: 544 F: 119 |
Left circumflex artery | 654 | ||||||||||||||
| 294 | |||||||||||||||||||
| Right coronary artery | 463 | ||||||||||||||||||
| 207 | |||||||||||||||||||
|
F, female; IQR, interquartile range; M, male; NR, not reported. |
|||||||||||||||||||
Table 2. Summary of included studies.
| Author & year | Journal | Country | Study design | Population size (included) | Compared interventions | Timeframe by staged PCI | Mean follow-up | Key findings |
|---|---|---|---|---|---|---|---|---|
| Kim et al.182025 | South Korea | Ramdomized controlled trial | 994 | Immediate vs staged revascularization | 3 days | 1 year |
| |
| Stähli et al.132023 | Europe | Randomized controlled trial | 840 | Immediate vs staged revascularization | 19-45 days | 30 days (± 7 days), 6 months (± 14 days), 1 year (± 14 days) |
| |
| Park et al.152023 | South Korea | Randomized controlled trial | 209 | Immediate vs staged revascularization | 14 days | 7 days 1 month 1 year |
|
|
| Wood et al.172019 | Canada | Randomized controlled trial | 4041 (2016) | Immediate vs staged revascularization | 23 (12.5-33.5) days | 3 years |
|
|
| Maamoun et al.162011 | Yemen | Randomized controlled trial | 78 | Immediate vs staged revascularization | 7 days | 1 year |
|
|
| Politi et al.142010 | Italy | Randomized controlled trial | 263 (130) | Immediate vs staged revascularization | 56.8 ± 12.9 days | 2.5 ± 1.4 years |
|
|
|
95%CI, 95% confidence interval; CR, complete revascularization; HR, hazard ratio; IRA, infarct-related artery; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction; MACE, major adverse cardiovascular events; MI, myocardial infarction; RR, risk ratio; SR, staged revascularization. |
||||||||
Cardiac death
There were no significant difference between staged and immediate PCI regarding the risk of cardiac death (RR, 1.26; 95%CI, 0.89-1.79; P = .19). No heterogeneity was observed among the included studies (I2 = 0%). These findings are shown in figure 2A.
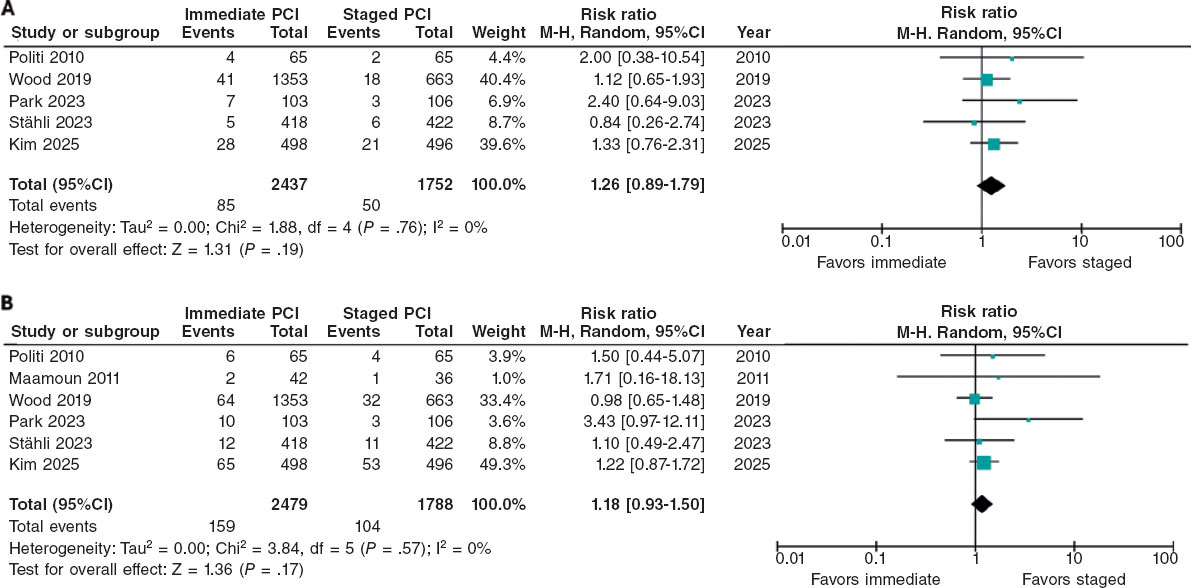
Figure 2. Risk Ratios of cardiac death and all-cause mortality. A: cardiac death: risk ratio (RR) across the included trials are represented by solid squares, with horizontal lines indicating 95% confidence intervals (95%CI). B: all-cause mortality: RR across trials are shown by solid squares, with 95%CI indicated by horizontal lines. The bibliographical references cited in this figure correspond to the following studies: Politi et al.14 (2010), Maamoun et al.16 (2011), Wood et al.17 (2019), Park et al.15 (2023), Stähli et al.13 (2023), and Kim et al.18 (2025).
All-cause mortality
There were no significant differences in the risk of all-cause mortality between patients undergoing staged PCI and those treated with immediate PCI (RR, 1.18; 95%CI, 0.93-1.50; P = .17), with no heterogeneity detected among studies (I2 = 0%) (figure 2B).
Reinfarction
The analysis demonstrated a statistically significant lower risk of reinfarction with immediate vs staged PCI (RR, 0.63; 95%CI, 0.40-0.98; P = .04), which amounts to an approximately 37% relative risk reduction with the former. There was no heterogeneity across trials (I2 = 0%). These findings are shown in figure 3A.
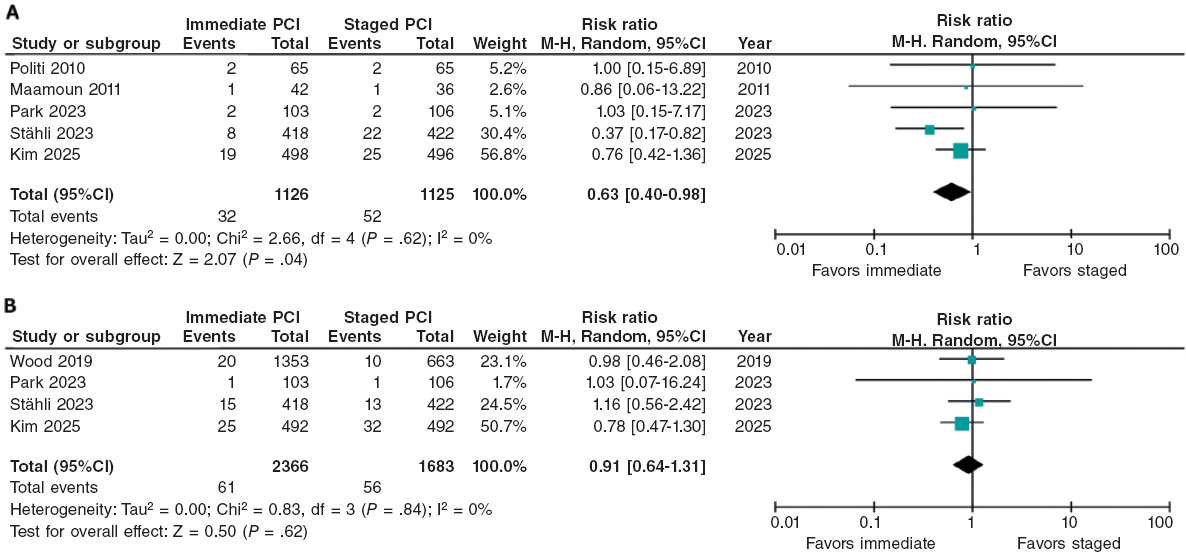
Figure 3. Risk ratios for reinfarction and acute kidney injury. A: reinfarction: risk ratio (RR) across the included trials are represented by solid squares, with horizontal lines indicating 95% confidence intervals (95%CI). B: acute kidney injury: RR across trials are shown by solid squares, with 95%CI indicated by horizontal lines. The bibliographical references cited in this figure correspond to the following studies: Politi et al.14 (2010), Maamoun et al.16 (2011), Wood et al.17 (2019), Park et al.15 (2023), Stähli et al.13 (2023), and Kim et al.18 (2025).
Acute kidney injury
There were no significant differences between the 2 strategies (RR, 0.91; 95%CI, 0.64-1.31; P = .62), indicating no clear association between the revascularization strategy and the risk of AKI. There was no heterogeneity among the included studies (I2 = 0%). These results are shown in figure 3B.
Stroke
Stroke was reported in only 3 of the studies included, and our analysis showed no significant differences between staged and immediate PCI for this outcome (RR, 1.17; 95%CI, 0.66-2.05; P = .59). There was no heterogeneity among the included studies (I2 = 0%). These results are shown in figure S1.
Publication bias was assessed using funnel plots, Egger’s regression, and trim-and-fill analyses for reinfarction, cardiac death, and all-cause mortality. There was no visual evidence of funnel-plot asymmetry (figure S2). For reinfarction, trim-and-fill imputed 1 potentially missing study, with the pooled effect remained statistically significant (IRR, 0.59; 95%CI, 0.38-0.90). For cardiac death and all-cause mortality, 2 studies were imputed in each analysis; however, pooled effects remained non-significant, with no heterogeneity detected (I2 = 0%). Overall, these results suggest no substantial publication bias, although the limited number of included trials reduces the power of formal tests for funnel plot asymmetry.
Sensitivity analyses
Leave-one-out sensitivity analyses confirmed the robustness of the pooled estimates across all primary endpoints (figure S3). For all-cause mortality and cardiac death, exclusion of individual studies did not materially alter the results, with pooled RRs remaining within the confidence intervals of the main models and I2 = 0% in all scenarios. For reinfarction, estimates consistently favored immediate PCI (RR range, 0.46-0.75), with I2 = 0% supporting the internal consistency of the findings.
Incidence rate ratios
At the 1-year follow-up, IRR analysis showed that immediate PCI was associated with a lower rate of reinfarction vs staged PCI. Under the random-effects model, the pooled IRR was 0.60 (95%CI, 0.39-0.94; I2 = 0%), which is consistent with the common-effect model (IRR, 0.59; 95%CI, 0.38-0.92), supporting the robustness of this finding (figure S4). In contrast, there were no significant differences between the strategies regarding cardiac death (IRR, 1.26; 95%CI, 0.90-1.77; I2 = 0%) or all-cause mortality (IRR, 1.17; 95%CI, 0.84-1.64; I2 = 0%), indicating stable estimates with negligible heterogeneity across studies (figures S5, S6).
Trial sequential analyses
Trial sequential analysis were conducted for cardiac death, all-cause mortality, and reinfarction (figure 4A-C). For cardiac death and all-cause mortality, the accrued sample represented 33% and 58% of the required information size, respectively; in both analysis, the cumulative Z-curves remained within nonsignificant boundaries, indicating insufficient evidence to confirm or exclude a 30% relative risk reduction. For reinfarction, only 3.4% of the required information size was reached, and the Z-curve remained well within the futility zone, showing a non-significant trend favoring immediate PCI. Collectively, these results demonstrate that current data remain underpowered to establish definitive conclusions across outcomes, despite a consistent numerical pattern supporting immediate revascularization.
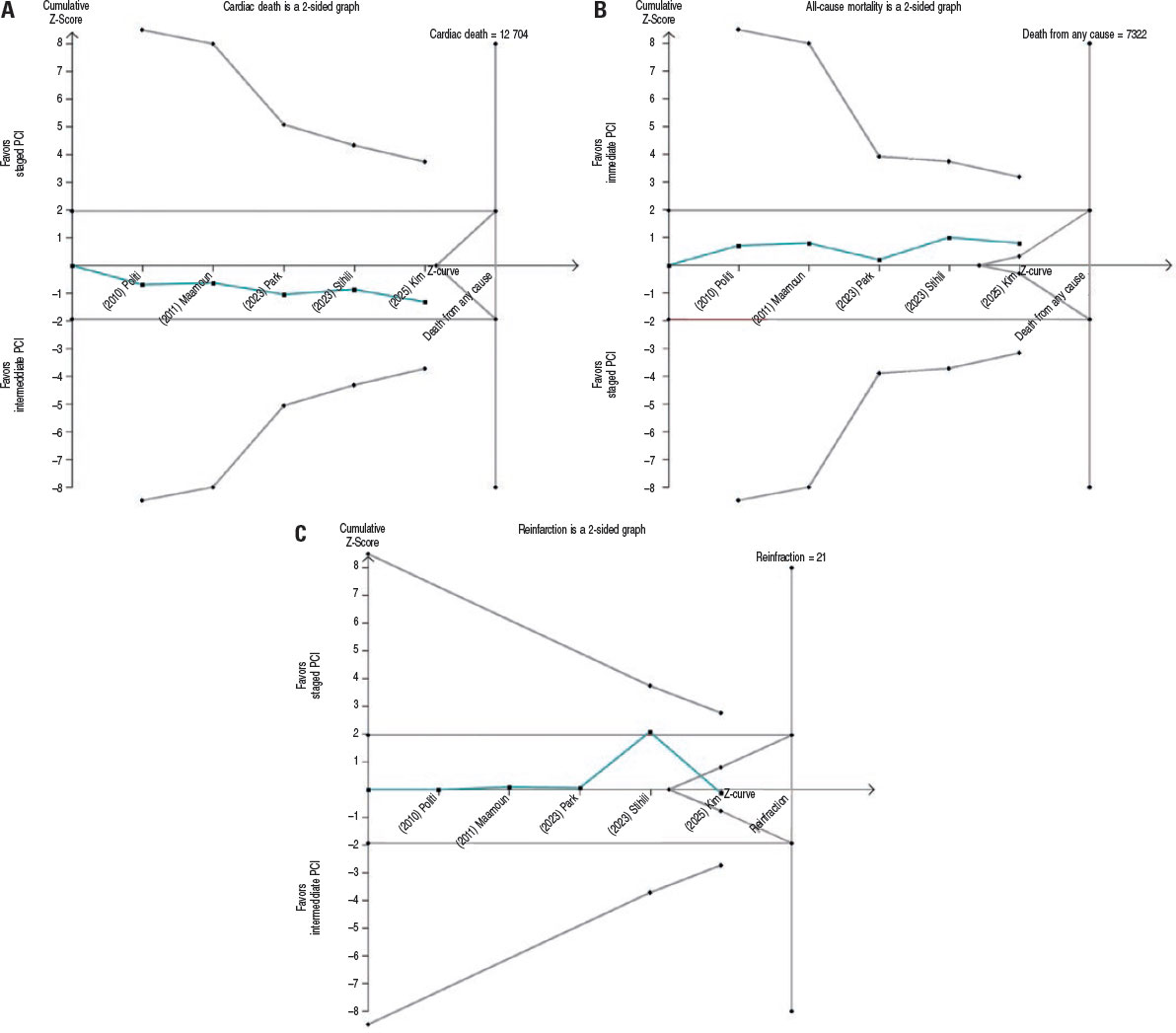
Figure 4. Trial sequential analysis. Trial sequential analysis for (A) cardiac death, (B) all-cause mortality, and (C) reinfarction. In all panels, the cumulative Z-curve is shown in blue, trial sequential monitoring boundaries in red, and conventional significance thresholds as horizontal magenta lines. PCI, percutaneous coronary intervention. The bibliographical references cited in this figure correspond to the following studies: Politi et al.14 (2010), Maamoun et al.16 (2011), Wood et al.17 (2019), Park et al.15 (2023), Stähli et al.13 (2023), and Kim et al.18 (2025).
Meta-regression
Exploratory meta-regression analyses were conducted to assess whether procedural timing or baseline comorbidities influenced reinfarction outcomes. The association between time to staged PCI and reinfarction risk showed a negative but non-significant slope (β = –0.016 per day; 95%CI, –0.042-0.010; P = .22), with no residual heterogeneity (I2 = 0%), indicating consistent effects across studies (figure S7). Similarly, diabetes prevalence (β = 0.021 per 1% increase; 95%CI, –0.017 to 0.058; P = .22) and hypertension prevalence (β = –0.064 per 1% increase; 95%CI, –0.214 to 0.085; P = .40) were not significantly associated with reinfarction risk, with no evidence of residual heterogeneity (I2 = 0% for both) (figures S8-S9). Collectively, these findings suggest that neither procedural timing nor comorbidity burden materially modified the comparative risk of reinfarction between immediate and staged PCI; however, given the study-level and exploratory design of these analyses, results should be interpreted with caution.
Risk of bias assessment
Risk of bias assessment was assessed using version 2 of the Cochrane risk-of-bias tool for randomized trials (ROB 2), as outlined in the Cochrane Handbook of Systematic Reviews of Interventions.11 Four clinical trials were judged to have a low overall risk of bias, whereas the study by Maamoun et al.16 was rated as the only clinical trial having some concerns (table 3).
Table 3. Risk of bias summary for randomized studies (RoB 2)
| Study | Bias arising from randomization process | Bias due to deviations from intended interventions | Bias due to missing outcome data | Bias in measurement of the outcomes | Bias in selection of the reported results | Overall risk of bias |
|---|---|---|---|---|---|---|
| Politi et al.142010 | Low | Low | Low | Low | Low | Low |
| Maamoun et al.162011 | Low | Some concerns | Low | Some concerns | Low | Some concerns |
| Wood et al.172019 | Low | Low | Low | Low | Low | Low |
| Park et al.152023 | Low | Low | Low | Low | Low | Low |
| Stähli et al.132023 | Low | Low | Low | Low | Low | Low |
| Kim et al.182025 | Low | Low | Low | Low | Low | Low |
DISCUSSION
The present meta-analysis provides a focused comparison of immediate vs staged PCI in patients with STEMI and multivessel disease. By integrating randomized evidence using complementary analytical approaches, our findings offer an updated and clinically relevant synthesis of the safety and efficacy profile of these 2 revascularization strategies in a well-defined population.
Although cardiac death and all-cause mortality are clinically relevant, they are relatively rare outcomes in contemporary STEMI populations undergoing multivessel revascularization. In the present analysis, there were no statistically significant differences between immediate and staged PCI for either endpoint. These neutral findings should be interpreted in the context of limited statistical power, as reflected by the low cumulative incidence of events and trial sequential analysis indicating that the required information size has not yet been reached.19,20 Additional IRR analyses yielded consistent results, further supporting the absence of a clinically meaningful survival difference between strategies. Accordingly, mortality outcomes do not appear to discriminate between immediate and staged PCI, and treatment selection should instead be guided by patient-specific clinical stability and procedural considerations rather than isolated mortality endpoints alone.
Regarding reinfarction risk, immediate PCI was consistently associated with a significantly lower rate of reinfarction vs staged PCI. Exploratory meta-regression analyses did not identify procedural timing, diabetes, or hypertension prevalence as significant modifiers of this association. Trial sequential and sensitivity analyses showed consistent results across the models, with no evidence of heterogeneity. Although the required information size was not fully reached, the consistency of findings across multiple analytical approaches supports a robust association between immediate PCI and reduced recurrent ischemic events.
Regarding AKI, there were no significant differences between immediate and staged PCI, with low event rates and no heterogeneity across trials. The limited number of events precluded meaningful trial sequential analysis, highlighting insufficient power to detect small differences. Although post-PCI AKI is influenced by multiple factors, including contrast exposure and procedural complexity,21 current evidence does not support the assumption that staged PCI reduces renal risk by distributing contrast load.22 None of the included trials were specifically powered for renal outcomes, and AKI definitions varied across studies. Therefore, until adequately powered trials with standardized definitions become available, PCI timing decisions should remain individualized, taking into account baseline renal function and overall procedural risk.23
There were no significant differences in the rates of stroke between staged and immediate PCI, with few events being reported across trials, limiting statistical power. Available evidence suggests that stroke risk after PCI in STEMI is more closely related to procedural factors than to the timing of non-culprit revascularization.15 Accordingly, stroke does not appear to represent a clinically discriminative endpoint when comparing immediate and staged revascularization strategies in this population.
Historically, international clinical practice guidelines favored a staged approach to complete revascularization in STEMI with multivessel disease, largely due to concerns regarding procedural complexity, contrast exposure, and hemodynamic instability during the index intervention. However, cumulative randomized evidence has challenged this paradigm, showing that immediate complete PCI can be performed safely in hemodynamically stable patients, with outcomes comparable or superior to staged strategies.13,17,24,25 Notably, most prior trials primarily enrolled clinically stable patients following successful reperfusion, which may limit the generalizability of their findings to higher-risk STEMI populations. In this context, the OPTION-STEMI trial extends the available evidence by including patients with transient or mild hemodynamic instability, thereby providing contemporary data that better reflect real-world clinical complexity.18
Consistent with our primary findings, immediate and staged PCI did not differ in cardiac death or all-cause mortality at 1 year, while immediate PCI was associated with a lower risk of reinfarction. Beyond clinical outcomes, immediate complete revascularization may offer economic advantages by reducing the length of stay, avoiding repeat cardiac catheterizations, and optimizing resource utilization. In contrast, staged PCI often requires a second procedure and hospitalization, increasing cumulative costs by up to 50% in some analyses,26,27 and mirroring the additional expenses associated with unplanned readmissions for recurrent ischemia. In this context, a single-session revascularization strategy may provide both clinical and economic value, particularly in health care systems with constrained resources, where contemporary PCI costs remain substantial.28
From a safety and efficacy perspective, both staged and immediate PCI represent acceptable revascularization strategies in appropriately selected patients with STEMI and multivessel disease. In the present analysis, immediate PCI was primarily associated with a reduction in recurrent ischemic events, while major adverse cardiovascular events, including cardiac death and all-cause mortality did not differ significantly across strategies. Accordingly, the choice between immediate and staged PCI may increasingly be guided by patient-specific clinical characteristics and logistical considerations rather than concerns regarding procedural safety.
Although not directly assessed in this meta-analysis, emerging evidence suggests that systemic biological factors—such as inflammation, hypercoagulability, and sympathetic activation—may influence recurrent ischemic risk beyond angiographic severity alone.29,30 In addition, features of plaque vulnerability have been associated with future ischemic events in non-culprit lesions, even when angiographically mild.31 Integrating biological risk markers and clinical stability into revascularization decision-making, therefore, represents a relevant direction for future research aimed at refining individualized strategies in patients with STEMI and multivessel disease.
Limitations
Several limitations should be acknowledged. First, only 6 randomized controlled trials were included, resulting in limited statistical power, particularly for rare outcomes. Second, this analysis was restricted to patients with STEMI and multivessel disease; therefore, the findings should not be extrapolated to other acute coronary syndrome populations, and the limited number of trials reporting all prespecified outcomes may reduce the robustness of certain estimates. Third, the use of aggregate study-level data precluded adjustment for individual patient characteristics and limited the assessment of patient-level effect modification; accordingly, meta-regression analyses were exploratory and should be interpreted with caution given the small number of included studies and the potential for ecological bias. Finally, variability in the timing of staged PCI across trials may have introduced residual clinical heterogeneity, and formal assessments of publication bias were underpowered due to the limited number of studies.
CONCLUSIONS
This meta-analysis, integrating additional analytical approaches, provides a focused comparison of immediate vs staged PCI in patients with STEMI and multivessel disease. Immediate PCI was consistently associated with a lower risk of reinfarction, without significant differences being reported in cardiac death or all-cause mortality, stroke, or renal injury. Although the cumulative evidence remains below the required information size for certain hard endpoints, the consistency of findings across multiple analyses supports the robustness of the observed reduction in reinfarction events. From a clinical perspective, these results suggest that immediate complete revascularization can be performed safely in appropriately selected patients and may reduce the burden of reinfarction. Accordingly, the choice between immediate and staged strategies should increasingly be guided by patient-specific clinical stability and practical considerations rather than by concerns regarding procedural safety alone. Future research incorporating biological risk markers may further refine individualized revascularization strategies in this population.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
This meta-analysis was based exclusively on data from previously published studies and involved secondary analyses of publicly available information. Therefore, approval by an Ethics Committee or institutional review board was deemed unnecessary. As no individual patient data or case reports were included, informed consent was not applicable. All the studies included were conducted in full compliance with the ethical standards of the respective institutions at the time of their original publication.
In line with the SAGER guidelines, we assessed sex and gender reporting in the included randomized trials. While sex was generally reported at baseline, sex-disaggregated outcome data and gender-related variables were rarely available, precluding analyses by sex or gender. This limitation should be considered when interpreting the results.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
During the preparation of this work the authors used ChatGPT 5.2 to review the syntaxis and grammar of this document. After using this tool/service, the authors reviewed and edited the content as needed and took full responsibility for the content of the published article.
AUTHORS’ CONTRIBUTIONS
D. Paulino-González: conceptualization, formal analysis, drafting – review and editing. L.H. García-Mena: formal analysis, investigation, drafting – review and editing. E. Andrade-Arbaiza: methodology, investigation, drafting – review and editing. D.A. Navarro-Martínez: methodology, formal analysis; J.L. Maldonado-García: formal analysis, drafting – review and editing; S.A. Xiloj-López: investigation. C.J. Palomino-Ojeda: investigation, drafting – review and editing, R.M. Arellano-Sánchez: data curating, formal analysis. N.E. Antonio-Villa: data curating, formal analysis, drafting – review and editing. A. Ricalde-Alcocer: drafting – review and editing, supervision. D. Araiza-Garaygordobil: drafting – review and editing, supervision.
CONFLICTS OF INTEREST
None declared.
ACKNOWLEDGEMENTS
The authors respectfully honor, in memory, a beloved family member of one of the coauthors, and gratefully acknowledge the support of Dr. Pavel Pichardo-Rojas.
WHAT IS KNOWN ABOUT THE TOPIC?
- Multivessel disease is present in approximately 40% of patients with ST-segment elevation myocardial infarction and is associated with an increased risk of recurrent myocardial infarction and mortality. Complete revascularization, including both culprit and non-culprit lesions, reduces adverse cardiovascular events compared with culprit-lesion-only PCI. Non-culprit lesions may be treated either during the index procedure or in a staged approach; however, the optimal timing of revascularization remains uncertain.
WHAT DOES THIS STUDY ADD?
- This meta-analysis of 6 randomized controlled trials provides a comprehensive comparison of immediate vs staged PCI for non-culprit lesions in STEMI with multivessel disease. Immediate PCI was associated with a lower risk of reinfarction, without significant differences being reported in cardiac death, all-cause mortality, stroke, or AKI. Exploratory meta-regression analyses did not identify procedural timing or baseline comorbidities as effect modifiers. Overall, these findings support the safety of immediate complete revascularization and suggest a potential clinical and economic advantage by reducing recurrent ischemic events and repeat procedures.
REFERENCES
1. Pimor A, Auffret V, Didier R, et al. Immediate complete revascularization in patients with ST-segment elevation myocardial infarction and multivessel disease treated by primary percutaneous coronary intervention:Insights from the ORBI registry. Arch Cardiovasc Dis. 2018;111:656-665.
2. Muller DWM, Topol EJ, Ellis SG, Sigmon KN, Lee K, Califf RM. Multivessel coronary artery disease:a key predictor of short-term prognosis after reperfusion therapy for acute myocardial infarction. Thrombolysis and Angioplasty in Myocardial Infarction (TAMI) Study Group. Am Heart J. 1991;121(4 Pt 1):1042-1049.
3. Jaski BE, Cohen JD, Trausch J, et al. Outcome of urgent percutaneous transluminal coronary angioplasty in acute myocardial infarction:comparison of single-vessel vs multivessel coronary artery disease. Am Heart J. 1992;124:1427-1433.
4. Wald DS, Morris JK, Wald NJ, et al. Randomized trial of preventive angioplasty in myocardial infarction. N Engl J Med. 2013;369:1115-1123.
5. Gershlick AH, Khan JN, Kelly DJ, et al. Randomized trial of complete vs lesion-only revascularization in patients undergoing primary percutaneous coronary intervention for STEMI and multivessel disease:the CvLPRIT trial. J Am Coll Cardiol. 2015;65:963-972.
6. Mehta SR, Wood DA, Storey RF, et al. Complete Revascularization with Multivessel PCI for Myocardial Infarction. N Engl J Med. 2019;381:1411-1421.
7. O'Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction:a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;127:362-425.
8. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation:The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39:119-177.
9. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
10. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement:an updated guideline for reporting systematic reviews. BMJ. 2021;372:71.
11. Higgins JPT, Thomas J, Chandler J, et al. Cochrane Handbook for Systematic Reviews of Interventions version 6.5 (updated August 2024). Cochrane. Available at www.cochrane.org/handbook. Accessed 11 Dec 2025.
12. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177-188.
13. Stähli BE, Varbella F, Linke A, et al. Timing of Complete Revascularization with Multivessel PCI for Myocardial Infarction. N Engl J Med. 2023;389:1368-1379.
14. Politi L, Sgura F, Rossi R, et al. A randomised trial of target-vessel vs multi-vessel revascularisation in ST-elevation myocardial infarction:major adverse cardiac events during long-term follow-up. Heart. 2010;96:662-667.
15. Park S, Rha SW, Choi BG, et al. Immediate versus staged complete revascularization in patients with ST-segment elevation myocardial infarction and multivessel coronary artery disease:results from a prematurely discontinued randomized multicenter trial. Am Heart J. 2023;259:58-67.
16. Maamoun W, Elkhaeat N, Elarasy R. Safety and feasibility of complete simultaneous revascularization during primary PCI in patients with STEMI and multi-vessel disease. Egypt Heart J. 2011;63:39-43.
17. Wood DA, Cairns JA, Wang J, et al. Timing of Staged Nonculprit Artery Revascularization in Patients With ST-Segment Elevation Myocardial Infarction:COMPLETE Trial. J Am Coll Cardiol. 2019;74:2713-2723.
18. Kim MC, Ahn JH, Hyun DY, et al. Immediate versus staged complete revascularisation during index admission in patients with ST-segment elevation myocardial infarction and multivessel disease (OPTION–STEMI):a multicentre, non-inferiority, open-label, randomised trial. Lancet. 2025;406:1032-1043.
19. Schoenfeld DA. Sample-Size Formula for the Proportional-Hazards Regression Model. Biometrics. 1983;39:499.
20. Olivier CB, StrußL, Sünnen N, et al. Accuracy of Event Rate and Effect Size Estimation in Major Cardiovascular Trials:A Systematic Review. JAMA Netw Open. 2024;7:248818-248818.
21. Mehran R, Dangas GD, Weisbord SD. Contrast-Associated Acute Kidney Injury. N Engl J Med. 2019;380:2146-2155.
22. Almendarez M, Gurm HS, Mariani J, et al. Procedural Strategies to Reduce the Incidence of Contrast-Induced Acute Kidney Injury During Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2019;12:1877-1888.
23. Nakamura T, Watanabe M, Sugiura J, et al. Prognostic impact and predictors of persistent renal dysfunction in acute kidney injury after percutaneous coronary intervention for acute myocardial infarction. Scientific Reports. 2024;14:1-9.
24. Rao S V, O'Donoghue ML, Ruel M, et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes:A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2025;151:771-862.
25. Diletti R, den Dekker WK, Bennett J, et al. Immediate versus staged complete revascularisation in patients presenting with acute coronary syndrome and multivessel coronary disease (BIOVASC):a prospective, open-label, non-inferiority, randomised trial. Lancet. 2023;401:1172-1182.
26. Virani SS, Newby LK, Arnold SV, et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA Guideline for the Management of Patients With Chronic Coronary Disease:A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2023;148:E9-E119.
27. Kim LK, Yeo I, Cheung JW, et al. Thirty?Day Readmission Rates, Timing, Causes, and Costs after ST?Segment–Elevation Myocardial Infarction in the United States:A National Readmission Database Analysis 2010-2014. J Am Heart Assoc. 2018;7:009863.
28. Hsia RY, Antwi YA, Weber E, Nath JB. A Cross-Sectional Analysis of Variation in Charges and Prices across California for Percutaneous Coronary Intervention. PLoS One. 2014;9:103829.
29. Seropian IM, Toldo S, Van Tassell BW, Abbate A. Anti-inflammatory strategies for ventricular remodeling following ST-segment elevation acute myocardial infarction. J Am Coll Cardiol. 2014;63:1593-1603.
30. Klingenberg R, Brokopp CE, Grivès A, et al. Clonal restriction and predominance of regulatory T cells in coronary thrombi of patients with acute coronary syndromes. Eur Heart J. 2015;36:1041-1048.
31. Spagnolo M, Giacoppo D, Laudani C, et al. Advances in the Detection and Management of Vulnerable Coronary Plaques. Circ Cardiovasc Interv. 2025;18:015529.















