To the Editor,
Drug-coated balloon (DCB) angioplasty has been associated with late lumen enlargement (LLE), a phenomenon in which vessel dimensions increase over time in the absence of permanent scaffolding.1 Although this angiographic observation is well-documented, the temporal evolution of coronary physiology after DCB treatment remains incompletely characterized. Previous studies have shown that fractional flow reserve improves from baseline to the postoperative period and remains stable at the follow-up;2,3 however, paired functional improvement from post-procedure to follow-up has not been specifically documented. We therefore performed a paired temporal assessment of late functional gain (LFG), defined as progressive physiological improvement occurring after completion of the index procedure, in patients undergoing DCB angioplasty for de novo coronary lesions.
This study analyzed consecutive patients who underwent DCB treatment for de novo coronary lesions between January 2018 and December 2024 from the CARDIO-FR registry (NCT04185285), a single-center prospective registry that systematically enrolls all consecutive patients undergoing percutaneous coronary intervention at Hospital & University Fribourg. The study protocol was approved by the local ethics committee and conducted in full compliance with the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all patients. Eligible patients aged ≥ 18 years and had available baseline, postoperative, and follow-up coronary angiography performed ≥ 7 days after the index procedure, with adequate image quality for Murray’s law-based quantitative flow ratio (µQFR) analysis at all time points. Follow-up angiography was not protocol-mandated in the CARDIO-FR registry; however, for the present analysis, only patients with a follow-up angiography after a DCB angioplasty were included. µQFR assessment was performed using AngioPlus Gallery (Pulse Medical Imaging Technology, China) by 2 independent blinded analysts. For each lesion, the angiographic projection that best visualized the vessel from ostium to distal segment was selected, minimizing overlap and foreshortening. The same projection was used for µQFR assessment at all 3 time points to ensure reproducibility. Lesions with inadequate image quality or inability to match projections across time points were excluded. DCB procedures followed standardized institutional protocols, with lesion preparation at the operator’s discretion. Statistical analyses included paired tests for temporal comparisons and regression analyses to identify predictors of functional improvement.
Between January 1, 2018, and December 31, 2024, 4725 patients were included in the registry, of whom 120 underwent DCB-only percutaneous coronary intervention for de novo coronary lesions. Among these, 59 patients (66 lesions) met all inclusion criteria and were included in the analysis. We analyzed a total of 132 angiograms from these 59 patients (age 70 ± 8 years; 22%, women; 37%, diabetic) with 66 de novo lesions (reference vessel diameter. 2.5 [interquartile range (IQR) 1.8-2.8] mm). Lesion preparation used semi-compliant (88%), non-compliant (62%), cutting balloons (28.8%), and intravascular lithotripsy (5.1%). DCB platforms comprised SELUTION SLR (M.A. Med Alliance, Ireland; n = 46, 78.0%, sirolimus-coated), Prevail (Medtronic, United States; n = 10, 16.9%, paclitaxel-coated), MagicTouch (Concept Medical, United States; n = 2, 3.4%, sirolimus-coated), and AGENT DCB (Boston Scientific, United States; n = 1, 1.7%, paclitaxel-coated). At a median angiographic follow-up of 8.5 months [IQR, 6.2-12.3 months], target lesion revascularization (TLR) occurred in 14 (21.2%) of 66 lesions. µQFR improved significantly from 0.88 ± 0.08 at baseline to 0.90 ± 0.07 at follow-up (+0.02; 95%CI, 0.00-0.04; P = .007). Minimal luminal diameter increased from 1.68 ± 0.40 mm to 1.78 ± 0.40 mm (+0.09 mm; 95%CI, 0.01-0.18; P = .036) (figure 1). Physiological trajectories were heterogeneous, with functional improvement predominantly observed in lesions without TLR. After excluding TLR, LFG was even more pronounced at +0.04 (95%CI, 0.01-0.06; P < .001), with minimal luminal diameter increasing by +0.05 mm (95%CI, 0.01-0.18; P = .034).
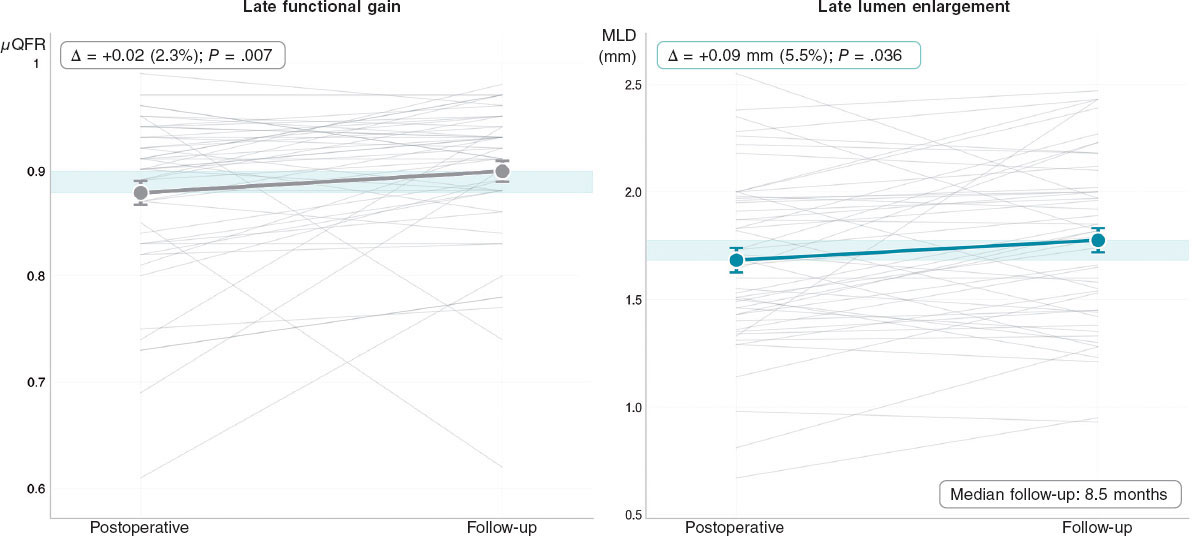
Figure 1. Paired analysis of minimum lumen diameter and Murray’s law-based quantitative flow ratio from the postoperative period to follow-up (n = 66 paired lesions). Individual patient trajectories are shown in grey lines and mean values with standard error bars in grey (LFG, left) and green (LLE, right). Absolute and relative changes are displayed. LFG, late functional gain; LLE, late lumen enlargement; µQFR, Murray’s law-based quantitative flow ratio.
Higher DCB diameter-to-vessel ratio and longer inflation time predicted functional improvement (+0.01 per 0.1 ratio increase, P = .015; +0.0041 per second, P = .011; respectively). The predilatation maximal diameter-to-vessel ratio emerged as the strongest binary predictor (odds ratio [OR], 17.23; P = .006), followed by DCB maximal diameter-to-vessel ratio (OR, 14.38; P = .011). Exploratory thresholds were identified at DCB balloon-to-vessel ratio > 1.31 (95%CI, 1.03-1.45) and inflation time > 72 seconds, both of which were associated with greater functional gain (+0.05 vs +0.02; P = .012; +0.04 vs +0.02; P = .008, respectively). Lesion preparation characteristics beyond diameter-to-vessel ratio, including specialty balloon type, showed no predictive value for functional improvement (all P > .20). For LLE, only DCB inflation time predicted enlargement (+0.011 mm per second; P = .015). The use of sirolimus-coated balloons showed a trend toward lower likelihood of lumen gain (OR, 0.22; 95%CI, 0.04-1.10; P = .07).
This exploratory study reports a paired temporal µQFR assessment of LFG following DCB angioplasty, suggesting progressive coronary physiological improvement from the postoperative period to mid-term follow-up in lesions without revascularization. LFG refers to continued functional recovery beyond the acute procedural result, conceptually distinct from, yet complementary to the established angiographic phenomenon of LLE.1 The temporal association between anatomical LLE and functional improvement suggests a potential mechanistic relationship; however, the observational design precludes definitive causal inference. Previous physiological assessments using µQFR in the PICCOLETO II trial showed comparable functional performance between DCB and drug-eluting stents at follow-up, with DCB showing relative functional recovery over time.4 Recent data have also described heterogeneous late physiological trajectories after DCB treatment, including late QFR deterioration in a subset of patients.5 Our findings extend these observations by identifying procedural determinants associated with functional improvement within a paired within-patient design. This hypothesis-generating observation provides further insight into the biological effects of DCB therapy, suggesting that vascuular healing may continue beyond the index procedure.6 In contrast to drug-eluting stents, where fractional flow reserve may deteriorate over time, and plain balloon angioplasty, which is often associated with functional decline, DCB treatment may promote progressive physiological improvement, potentially through antiproliferative effects, positive vessel remodeling,1 and restoration of endothelial function.
Our findings identify 2 procedural determinants associated with functional improvement. First, adequate lesion preparation induces plaque disruption, facilitating optimal drug penetration into the vessel wall. Second, optimized DCB delivery, through appropriate sizing and prolonged inflation, provides mechanical support that prevents elastic recoil and stabilizes dissection planes, thereby reducing the need for bail-out stenting. While drug transfer from sirolimus-coated balloons occurs within 30–45 seconds, prolonged inflation likely provides an additional mechanical benefit beyond drug delivery. Current consensus recommends DCB sizing with a balloon-to-vessel ratio of 0.9–1.3.6 Our results suggest that ratios toward the upper range may be associated with greater functional gain; however, these findings remain exploratory and should be individualized according to vessel anatomy, lesion characteristics, and operator experience.
Several limitations warrant consideration. The observational single-center design and modest sample size limit the generalizability of these findings, which should be considered hypothesis-generating. Second, the cohort included heterogeneous DCB platforms with different drug coatings, limiting the ability to draw conclusions regarding drug-class effects. The observed trend toward a lower likelihood of LLE with sirolimus-coated balloons may reflect device-specific differences in drug formulation, coating technology, or release kinetics rather than a true drug-class effect. As no class effect has been established among DCBs, caution is warranted when extrapolating these findings, and device-specific analyses were limited by the small sample size. Third, the absence of an independent imaging core laboratory may have introduced variability in image acquisition and µQFR analysis. Fourth, the observed TLR rate exceeds rates typically reported in randomized DCB clinical trials, which likely reflects the study’s inclusion criteria and limits the generalizability of the observed physiological findings. Finally, because follow-up angiography was not protocol-mandated, the study introduced inherent selection bias toward individuals with clinical indications for repeat imaging, resulting in heterogeneous clinical indications and variable follow-up timing.
In conclusion, this study provides evidence of LFG assessed by µQFR, suggesting progressive physiological improvement from the postoperative period to mid-term follow-up after DCB angioplasty.
FUNDING
This study was funded by the Fonds Scientifique Cardiovasculaire and Fondation Cecil de Recherche Cardiovasculaire.
ETHICAL CONSIDERATIONS
This study was conducted in full compliance with the principles outlined in the Declaration of Helsinki and was approved by the local ethics committee (analysis from the CARDIO-FR registry, ClinicalTrials.gov identifier: NCT04185285). Written informed consent was obtained from all participati patients prior to enrollment. The SAGER guidelines were considered throughout the design, analysis, and reporting of this study. However, sex- and gender-based subgroup analyses were not performed due to the limited sample size, which precluded meaningful comparisons. Sex and gender distribution of participants is reported in the baseline characteristics.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Artificial intelligence-based tools were used solely for English language proofreading during the preparation of this manuscript. All aspects of the study, including conception, design, data collection, analysis, interpretation of results, and manuscript drafting, were performed entirely by the authors without the assistance of artificial intelligence. The authors take full responsibility for the content and integrity of the work.
AUTHORS’ CONTRIBUTIONS
D. Garin conceived and designed the study. D. Garin and R. Mohand Saidi performed data collection, data analysis, and drafted the manuscript. J.F. Iglesias and C. Seydoux contributed to data interpretation and critically revised the manuscript for relevant intellectual content. M. Togni and S. Cook provided senior supervision, contributed to study design, and critically revised the manuscript. D. Garin supervised the overall project, contributed to data interpretation, and provided critical revision of the manuscript. All authors read and approved the final version of the manuscript.
CONFLICTS OF INTEREST
J.F. Iglesias reports having received speaker fees or honoraria from Abbott Vascular, Biotronik, Biosensors, Bristol Myers Squibb/Pfizer, Cordis, Concept Medical, Medtronic, Penumbra Inc., and ReCor Medical, and consulting fees from Biotronik, ReCor Medical, Cordis, and Medtronic. J.F. Iglesias’s institution received unrestricted research grants from Biotronik, Concept Medical, Terumo Corp., and SMT. None of these relationships influenced the design, conduct, or reporting of this research. The remaining authors declared no conflicts of interest whatsoever.
REFERENCES
1. Kleber FX, Schulz A, Waliszewski M, et al. Local paclitaxel induces late lumen enlargement in coronary arteries after balloon angioplasty. Clin Res Cardiol. 2015;104:217–225.
2. Ann SH, Balbir Singh G, Lim KH, et al. Anatomical and physiological changes after paclitaxel-coated balloon for atherosclerotic de novo coronary lesions:Serial IVUS-VH and FFR study. PLoS One.2016;11:e0147057.
3. Shin E-S, Bang LJ, Jun EJ, et al. Fractional flow reserve-guided coronary angioplasty using paclitaxel-coated balloons without stent implantation:Feasibility, safety and 6-month results by angiography and optical coherence tomography. EuroIntervention. 2016;12:e1004–1014.
4. Fezzi S, Trevisanello A, Buccheri D, et al. Physiological performance of drug-coated balloons in small coronary arteries PICCOLETO II µFR. Catheter Cardiovasc Interv. 2025;105:643–649.
5. Zhu Y, Hu T, Bian Q, et al. Predictors for quantitative flow ratio loss in patients with de novo coronary artery disease treated with drug-coated balloons. Scientific Reports. 2025;15:21479.
6. Fezzi S, Scheller B, Cortese B, et al. Definitions and standardized endpoints for the use of drug-coated balloon in coronary artery disease:Consensus document of the Drug Coated Balloon Academic Research Consortium. Eur Heart J. 2025;46:2498-2519.














