A few years ago I had the opportunity to write a review article for Revista Española de Cardiología together with Elazer R. Edelman.1 In that paper, we reviewed the different stages and modalities of biomedical research, ranging from nonclinical studies in animal or computational models to randomized clinical trials and clinical registries. We also discussed the limitations and challenges they pose, while proposing potential solutions and alternative approaches to help overcome them.
Nearly a decade later, the aim of the present article is to offer a more personal perspective on clinical research in interventional cardiology, primarily intended for those who wish to being or are already taking their first steps in this field.
WHY CONDUCT RESEARCH?
All physicians should be involved in research in some capacity, although the degree of involvement may vary considerably. Clinical groups that actively conduct research also tend to improve the quality of their clinical care. Therefore, research should not be viewed as an optional activity within a medical team, but rather as an essential professional responsibility. Within each team, participation will naturally differ among members, encompassing both leadership and collaborative roles. Ultimately, research should serve first and foremost to improve patient care and, only secondarily, to enhance individual academic credentials.2
In my case, as in that of many physicians engaged in research, the motivations for undertaking research include the following:
- – Because I ask questions.
- – Because I wish to improve medical care.
- – Because I wish to improve clinical outcomes.
- – Because I feel fulfilled; it makes me feel better; it fascinates me.
- – And yes, also because I find satisfaction in seeing my studies published.
The primary motivation for research is, ultimately, the desire to answer questions related to patients’ cardiac diseases, including their diagnosis, treatment, and prognosis.3
If this is the case, however, why is it sometimes so difficult to initiate, sustain, or direct efforts toward research? Numerous barriers exist. Among the most notable are the scarcity of financial resources—often reflecting the limited sociopolitical priority given to research—the lack of a strong research culture and social motivation, the heavy clinical workload, the instability and job insecurity faced by researchers, and the limited professional and social recognition of research activities.2,4 Additional obstacles include insufficient training in research methodology, lack of financial compensation, convenience, and simple lack of interest.
WHAT CAN I RESEARCH?
When beginning a research career—often modestly—one of the first questions we ask ourselves is: what can I investigate that has not already been studied? Frequently, particularly among younger physicians, there is a temptation to replicate previously conducted studies and reproduce them at a local level. In certain circumstances this approach may be appropriate, especially when prior studies have clear limitations and the proposed design offers meaningful improvements. However, this is often not the case, and such studies, even if completed, are unlikely to have a meaningful impact or achieve publication.
The proportion of recommendations in cardiology clinical practice guidelines supported by high-level evidence—defined as level A, typically derived from multiple randomized clinical trials or meta-analyses—is relatively low. In the clinical practice guidelines of the American College of Cardiology and the American Heart Association (ACC/AHA), this proportion is approximately 8.5%.5 In the European Society of Cardiology (ESC) guidelines, it ranges between 14% and 16%.6
Although much of the available clinical evidence is based on randomized clinical trials, these studies may also present certain limitations:
- – Small sample size or inadequate representation of certain subgroups or specific clinical indications.
- – Use of nonclinical (surrogate) endpoints or “soft” clinical outcomes (eg, outcomes that do not include death or myocardial infarction).
- – Short duration of follow-up.
- – Use of a control group that does not represent the reference standard.
- – Absence of an appropriate placebo (sham procedure) in trials involving certain interventional techniques.
In many settings, evidence derived from randomized clinical trials is lacking, and the available knowledge is based primarily on observational registries (level of evidence B). In cardiology, recommendations supported by level of evidence C—based on expert consensus, small studies, or retrospective registries—still account for a substantial proportion of clinical practice guidelines.
Therefore, it is undeniable that substantial knowledge gaps remain, and identifying them is essential for designing studies that, even if modest in scope, may still provide meaningful contributions.
Accordingly, there are numerous opportunities for clinical research, including the following:
- – Registries evaluating the use of new technologies, including:
- Outcomes in real-world clinical practice.
- Outcomes in specific patient subgroups.
- Assessment of rare complications.
- – Comparisons between “old” and “new” technologies in clinical contexts that have not been addressed in previous trials.
- – Clinical questions typically not addressed by industry-sponsored research, such as:
- Conditions with very low case volumes.
- Commercially “orphan” techniques.
- – Evaluation of health care processes, including:
- Safety and effectiveness of care delivery.
METHODOLOGICAL DESIGNS: STARTING WITH OBSERVATIONAL STUDIES
At the beginning of a clinical research career, conducting randomized clinical trials is often very difficult, particularly multicenter trials. However, other study designs are more feasible and accessible, and they provide valuable opportunities to develop and refine research skills.
Observational registries represent an excellent starting point. In order of increasing complexity of implementation—and, therefore, potential scientific value—the following study designs can be considered1:
- – Single-center retrospective observational registries.
- – Single-center prospective observational registries.
- – Multicenter retrospective observational registries.
- – Multicenter prospective observational registries.
In addition, hybrid study designs may also be used, such as ambispective registries, which incorporate both retrospective and prospective components.
Within these registries, the control group may be historical, based on a previous time series in which one treatment replaced another, or concurrent, when the treatments being compared coexist during the same time period. However, selection bias often limits the validity of comparisons between study and control groups, particularly when both treatments were available simultaneously. For this reason, registries frequently employ statistical adjustment techniques, such as methods based on the propensity score, to improve comparability between groups.7
A key question when designing a new observational registry is identifying where the opportunity lies. A real-world registry evaluating the use of a new drug or a new diagnostic or therapeutic technique may provide meaningful value if it meets at least one of the following conditions:
- – It is the first registry addressing the topic.
- – It includes a larger sample size than previously published studies.
- – It provides longer follow-up than existing data.
- – It explores new indications beyond established uses (off-label).
- – It incorporates novel methods for evaluating or measuring outcomes.
- – It evaluates its use in a specific national, regional, or local context.
COOPERATIVE, MULTICENTER, AND MULTIDISCIPLINARY RESEARCH
Modern medical research has evolved beyond the model of the solitary investigator toward a cooperative, multicenter, and multidisciplinary approach.
Multidisciplinarity
Many contemporary research projects require collaboration among cardiologists with different areas of expertise, cardiac surgeons, physicians from other specialties, nursing professionals, statisticians, bioinformaticians, and other specialists. This multidisciplinary framework allows cardiovascular diseases and interventions to be studied from multiple perspectives, fostering innovative solutions that would be difficult to achieve within a single discipline.
Multicenter approach
Research studies are increasingly conducted across multiple institutions to include larger and more diverse patient populations. This approach enhances statistical power and improves the generalizability of results to different populations worldwide. Multicenter collaborations frequently extend across countries and health systems. To develop such collaborations, establishing connections with professionals interested in the same field of research is essential. Scientific conferences and in-person courses provide valuable opportunities for networking, but they are not the only pathways. Currently, remote interaction and communication among professionals are widespread, both within countries and internationally. Email has facilitated these exchanges for decades, and more recently, online events and social media platforms have enabled researchers to connect, share ideas, and develop collaborative projects with colleagues around the world.
PROGRESSION OF THE CLINICAL RESEARCH CAREER
Earlier we highlighted the different levels of complexity of observational studies. This has important implications for how a research career may evolve, including the types of study designs undertaken and the strategies used to disseminate their results.
The sequence of progression I propose is the following. It should be considered illustrative rather than prescriptive, as research careers may follow different paths:
- – Local observational studies conducted within one’s own center:
- Presentation at regional or national congresses.
- Publication in regional or national journals.
- – Multicenter retrospective and prospective observational studies:
- Presentation at national or international congresses.
- Publication in national or international journals.
- – Randomized clinical trials (single-center or multicenter):
- Submission as late-breaking trials to international congresses.
- Publication in international journals.
As an example of this progression, the ESTROFA study group (Estudio español sobre trombosis de stents farmacoactivos) is noteworthy, having generated up to 10 publications. Focused on the investigation of clinical outcomes associated with drug-eluting stents, this collaborative project began with large multicenter retrospective registries, followed by prospective registries, and ultimately culminated in a collaborative randomized trial conducted together with a British research group.8-10
Another illustrative example is the LITRO study group (Lesiones intermedias en tronco común), which explored different aspects of the use of intravascular ultrasound in the assessment and management of left main coronary artery lesions.11-13
STATISTICS AND DATABASES
For basic and moderately complex analyses, it is often preferable to work independently or with a trusted collaborator, consulting external experts only for highly complex analyses that cannot otherwise be addressed. For this reason, training in biostatistics—through dedicated courses or master’s programs—is highly valuable.
Statistical software used in clinical research can generally be divided into powerful commercial platforms and open-source or user-friendly tools designed for academic researchers.14,15
Among the most powerful commercial programs is SAS (Statistical Analysis System), which provides a robust biostatistical platform designed to meet strict regulatory standards. SPSS Statistics (IBM) is widely used in the health sciences because of its intuitive interface based on drop-down menus, making it suitable for analyses commonly applied in clinical research without requiring programming skills. MedCalc, designed specifically for biomedical research, is another commercial option that, although less comprehensive than other platforms, is among the most intuitive and therefore particularly suitable for beginners. Among free and open-source tools, R (and the RStudio environment) stands out as one of the most powerful and flexible statistical platforms currently available. It includes thousands of libraries specifically developed for biostatistical analyses and is widely used in academic research because it is freely available. However, its effective use requires greater training and programming experience.
Regarding databases, researchers often begin by creating them in Excel (Microsoft). Nevertheless, due to its limitations for research purposes, it is advisable to progress toward databases specifically designed for clinical research. Among these, REDCap (Research Electronic Data Capture) is particularly notable for its strong academic orientation. It is a secure, web-based platform designed for creating and managing online databases and surveys to facilitate data collection in research projects, especially in biomedical and academic settings. REDCap allows the design of complex studies—such as clinical trials and longitudinal studies—supports straightforward data import and export, and incorporates features such as branching logic, automated functions, and data validation. As a result, it has become an essential platform for researchers who need to manage sensitive information efficiently and securely.
ARTIFICIAL INTELLIGENCE AND CLINICAL RESEARCH
Artificial intelligence (AI) can assist research projects through general-purpose applications such as ChatGPT (OpenAI), which is widely used for writing, programming support, and general inquiries, as well as through other tools specifically oriented toward scientific research.16,17
- – Literature search and review: traditionally, PubMed, the free public search engine maintained by the US National Library of Medicine, has been the most widely used resource for biomedical literature searches. However, AI-based tools are now emerging that allow clinical evidence to be identified more rapidly and efficiently than traditional search engines. One example is OpenEvidence, a platform designed to provide medical answers based exclusively on up-to-date clinical evidence.
- – Data analysis: several AI tools are designed to process large volumes of medical information, including both structured and unstructured data. Amazon Comprehend Medical uses natural language processing to extract clinical information—such as diagnoses, medications, and symptoms—from unstructured medical notes. Owkin focuses on biomarker discovery and clinical trial design using machine learning models. PubMed.ai allows public health researchers to analyze and synthesize findings from multiple studies efficiently.
- – Writing and editing of manuscripts: various applications assist with drafting scientific texts and ensuring compliance with academic standards. Paperpal provides real-time suggestions to improve academic writing and enhance the quality of scientific language. Jenny AI assists with article structure, proposes titles, and suggests citations based on the bibliography provided by the user. QuillBot is useful for paraphrasing and summarizing complex texts, helping improve clarity and reduce the risk of plagiarism. DeepL is a highly effective AI-based translation tool widely used in scientific writing.
- – Workflow support: NotebookLM (Google) allows researchers to upload their own documents to ask specific questions, generate summaries, or create study guides. Scite.ai helps validate academic citations by indicating whether a study has been supported, cited, or contrasted by subsequent research.
Of note, although these tools can facilitate several steps of the research process, their outputs require careful verification and responsible, transparent use.
PUBLIC MASSIVE CLINICAL DATA SOURCES
Several public databases provide information on health care activity at the national level. Among these, the Minimum Basic Data Set (Conjunto Mínimo Básico de Datos, CMBD) is particularly relevant. This database is derived from hospital discharge reports, from which coding units at each hospital collect a standardized set of clinical and administrative variables. The main advantages of the CMBD include its large sample size, the high reliability of certain variables—such as age, sex, discharge diagnosis, and mortality—and the presence of a robust data auditing system that ensures overall data quality. However, several limitations must also be considered, including the potential undercoding of some variables and the absence of coding for others.18
THE SEARCH FOR FUNDING
The search for funding for cardiovascular research in Spain typically focuses on competitive public calls (national and regional), European programs, scientific societies, private foundations, and the pharmaceutical and medical device industry.
- – Public funding (national and regional): the Agencia Estatal de Investigación (AEI), attached to the Ministerio de Ciencia, Innovación y Universidades, supports basic and translational research through annual R&D&i calls. The Instituto de Salud Carlos III (ISCIII) is the main source of funding for biomedical and clinical research in Spain through its Strategic Action in Health (Acción Estratégica en Salud, AES) programs.
- – Scientific societies: the Spanish Society of Cardiology (SEC), including its associations and sections—such as the Interventional Cardiology Association of the SEC—offers grants, research awards, and funding for specific research projects.
- – Centers of excellence: the Centro Nacional de Investigaciones Cardiovasculares (CNIC) receives competitive funding, including the Severo Ochoa accreditation, to support research in cardiovascular prevention and treatment.
- – Private foundations and other entities: examples include CaixaResearch, Fundación BBVA, and Fundación Ramón Areces, which provide competitive grants to support biomedical research.
- – Independent research supported by industry: these are investigator-initiated clinical studies proposed and led by researchers outside the pharmaceutical or medical device industry that receive full or partial financial support from these companies.
- – European funding: the European Research Council (ERC) offers grants to principal investigators of any nationality working in Europe. In addition, Horizon Europe, the European Union’s framework program for research and innovation, includes substantial funding opportunities for health research. However, if obtaining funding at the national level is already challenging, applications for European funding typically involve considerably greater administrative and bureaucratic complexity.
ADMINISTRATIVE AND REGULATORY SUPPORT PLATFORMS
To conduct multicenter studies, whether prospective registries or randomized trials, particularly the latter, it is necessary to have appropriate expert support to manage, supervise, and develop the study while ensuring regulatory and ethical compliance. For this purpose, contract research organizations (CRO) are often used; however, their services are typically very costly. Consequently, alternative platforms developed by scientific institutions and foundations have emerged. Examples include the SEC’s Agencia de Inves- tigación19 and initiatives promoted by foundations and research associations, such as Fundación EPIC20 and Fundación FIC.21 These platforms provide comprehensive support for study design, coordination, and regulatory management, offering a closer and more accessible framework for independent investigators working with limited research funding.
FINAL REFLECTIONS
One of the greatest challenges in research is the substantial personal effort required, often combined with funding that is difficult to obtain or simply scarce. In addition, collaborations are frequently altruistic or poorly compensated, centralized data monitoring may be limited or absent, the quality of statistical analysis may vary, and the capacity for publication is often constrained. These difficulties commonly lead to a frustrating sequence of editorial rejections (figure 1).
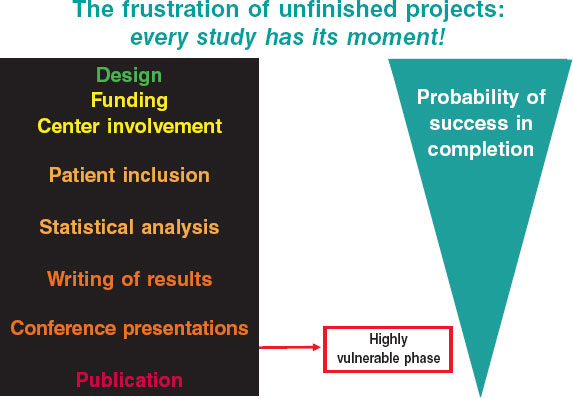
Figure 1. Stages of a study and levels of execution risk.
To successfully undertake a clinical research project, it is essential to define a clear and original objective with practical implications and to design a study that interferes as little as possible with routine clinical practice. Only essential and well-defined information should be requested. Flexible and individualized contributions from participating centers should be encouraged, prioritizing data quality over quantity. Maintaining close communication with collaborators —offering support during patient enrollment and follow-up—is also crucial. Furthermore, investigators must demonstrate a genuine commitment to the dissemination and publication of results and establish authorship structures that reflect the magnitude of each contribution while avoiding any form of favoritism (figure 2).
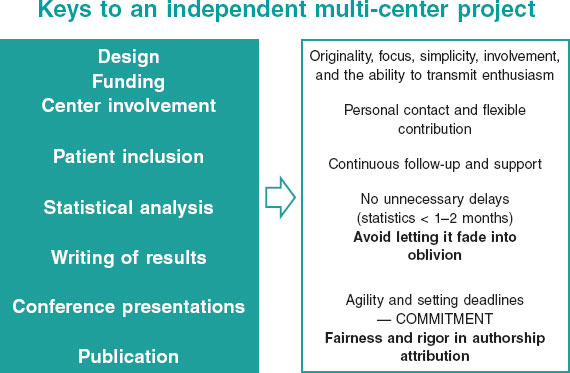
Figure 2. Essential aspects to guarantee success in an independent multicenter study.
Finally, as in any collaborative endeavor, the group of individuals involved in research largely determines the project’s ultimate success. Each investigator should aim to build a collaborative network that includes both close colleagues and peers from other centers, while remaining willing to delegate tasks and assume shared responsibilities. The level of involvement and motivation among collaborators will inevitably vary; recognizing and managing these differences can help ensure the success of each project and foster durable research collaborations.
FUNDING
None.
CONFLICTS OF INTEREST
J.M. de la Torre-Hernández declared having received speaker and consultant fees from Abbott, Medtronic, Philips, Biotronik, Amgen, Daiichi Sankyo, Boston Sci, and Novartis, unrelated to the present article.
REFERENCES
1. de la Torre Hernández JM, Edelman ER. From Nonclinical Research to Clinical Trials and Patient-registries:Challenges and Opportunities in Biomedical Research. Rev Esp Cardiol. 2017;70:1121-1133.
2. Cohen JJ, Siegel EK. Academic Medical Centers and Medical Research:The Challenges Ahead. JAMA. 2005;294:1367-1372.
3. Mokhtari B, Badalzadeh R, Ghaffarifar S. The next generation of physician-researchers:undergraduate medical students'and residents'attitudes, challenges, and approaches towards addressing them. BMC Med Educ. 2024;24:1313.
4. Lloyd T, Phillips BR, Aber RC. Factors that influence doctors'participation in clinical research. Med Educ. 2004;38:848-851.
5. Fanaroff AC, Califf RM, Windecker S, Smith SC, Lopes RD. Levels of Evidence Supporting American College of Cardiology/American Heart Association and European Society of Cardiology Guidelines, 2008-2018. JAMA. 2019;321:1069-1080.
6. Boriani G, Venturelli A, Imberti JF, Bonini N, Mei DA, Vitolo M. Comparative analysis of level of evidence and class of recommendation for 50 clinical practice guidelines released by the European Society of Cardiology from 2011 to 2022. Eur J Intern Med. 2023;114:1-14.
7. Benedetto U, Head SJ, Angelini GD, Blackstone EH. Statistical primer:propensity score matching and its alternatives. Eur J Cardiothorac Surg. 2018;53:1112-1117.
8. De la Torre-Hernández JM, Alfonso F, Hernández F, et al. ESTROFA Study Group. Drug-eluting stent thrombosis:results from the multicenter Spanish registry ESTROFA (Estudio ESpañol sobre TROmbosis de stents FArmacoactivos). J Am Coll Cardiol. 2008;51:986-990.
9. De la Torre Hernández JM, Alfonso F, Gimeno F, et al. ESTROFA-2 Study Group. Thrombosis of second-generation drug-eluting stents in real practice results from the multicenter Spanish registry ESTROFA-2 (Estudio Español Sobre Trombosis de Stents Farmacoactivos de Segunda Generacion-2). JACC Cardiovasc Interv. 2010;3:911-919.
10. De Belder A, de la Torre Hernandez JM, Lopez-Palop R, et al.;XIMA Investigators. A prospective randomized trial of everolimus-eluting stents versus bare-metal stents in octogenarians:the XIMA Trial (Xience or Vision Stents for the Management of Angina in the Elderly). J Am Coll Cardiol. 2014;63:1371-1375.
11. De la Torre Hernandez JM, Hernández Hernandez F, Alfonso F, et al. LITRO Study Group (Spanish Working Group on Interventional Cardiology). Prospective application of pre-defined intravascular ultrasound criteria for assessment of intermediate left main coronary artery lesions results from the multicenter LITRO study. J Am Coll Cardiol. 2011;58:351-358.
12. De la Torre Hernandez JM, Baz Alonso JA, Gómez Hospital JA, et al.;IVUS-TRONCO-ICP Spanish study. Clinical impact of intravascular ultrasound guidance in drug-eluting stent implantation for unprotected left main coronary disease:pooled analysis at the patient-level of 4 registries. JACC Cardiovasc Interv. 2014;7:244-254.
13. Rodriguez-Leor O, de la Torre Hernández JM, García-Camarero T, et al. Instantaneous Wave-Free Ratio for the Assessment of Intermediate Left Main Coronary Artery Stenosis:Correlations With Fractional Flow Reserve/Intravascular Ultrasound and Prognostic Implications:The iLITRO-EPIC07 Study. Circ Cardiovasc Interv. 2022;15:861-871.
14. Dembe AE, Partridge JS, Geist LC. Statistical software applications used in health services research:analysis of published studies in the U.S. BMC Health Serv Res. 2011;11:252.
15. Goldmann E, Barnard-Mayers R, Glymour MM, Healey MA. Statistical software skills for master's-level jobs in epidemiology:an analysis of 15 years of job posting data. Am J Epidemiol. 2025:kwaf243.
16. Sen CK. Artificial Intelligence Tools in Biomedical Research:Part 1 - Literature Search and Knowledge Mining. Antioxid Redox Signal. 2026;44:1-10.
17. Haug CJ, Drazen JM. Artificial Intelligence and Machine Learning in Clinical Medicine, 2023. N Engl J Med. 2023;388:1201-1208.
18. Fernández-Navarro P, López-Abente G, Salido-Campos C, Sanz-Anquela JM. The Minimum Basic Data Set (MBDS) as a tool for cancer epidemiological surveillance. Eur J Intern Med. 2016;34:94-97.
19. Sociedad Española de Cardiología. Agencia de Investigación (AISEC). Available at: https://secardiologia.es/cientifico/investigacion/agencia-de-investigacion. Accessed 9 Feb 2026.
20. Fundación Epic. Available at: https://fundacionepic.org/. Accessed 9 Feb 2026.
21. Fundación FIC. Available at: https://fundacionfic.es/proyectos-de-investigacion/. Accessed 9 Feb 2026.














