ABSTRACT
Introduction and objectives: Rapid ventricular pacing is essential to ensure accurate valve deployment during transcatheter aortic valve implantation (TAVI). Although it is traditionally performed via right ventricular (RV) pacing using a temporary pacemaker, left ventricular (LV) pacing with pre-shaped guidewires has emerged as an effective alternative, associated with shorter procedural and fluoroscopy times. There are no data available on the use of the Abbott Circulo guidewire. The aim of this study was to evaluate its safety and efficacy profile for LV pacing during TAVI.
Methods: This was a prospective, multicenter, observational study with patients undergoing transfemoral TAVI with LV pacing using the Circulo guidewire. The primary endpoint was pacing efficacy, defined as a reduction in systolic blood pressure < 60 mmHg. Secondary endpoints included the pacing threshold, crossover to RV pacing, and perioperative complications.
Results: A total of 50 patients were enrolled from March through June 2025 (mean age, 80.56 ± 6.26 years; 62% women). Rapid LV pacing was successful in 100% of cases, with a mean pacing threshold of 7.00 ± 3.48 mV. A total of 3 patients required crossover to RV pacing due to atrioventricular block after balloon predilatation. The overall rate of complications was 14%, with rhythm disturbances being the most common adverse events. There were no complications associated with the use of the Circulo guidewire.
Conclusions: LV pacing using the Circulo guidewire during TAVI is safe and effective, and avoids the routine use of temporary transvenous pacemakers.
Keywords: Transcatheter aortic valve implantation. Left ventricular pacing. Pre-shaped guidewire. Rapid pacing. Pacemaker. Structural heart procedure.
RESUMEN
Introducción y objetivos: La estimulación ventricular rápida es esencial para la correcta colocación de la prótesis durante el implante percutáneo de válvula aórtica (TAVI). Aunque clásicamente se realiza mediante estimulación del ventrículo derecho (VD) con marcapasos transitorio, la estimulación del ventrículo izquierdo (VI) con guías preformadas es una alternativa eficaz, asociada a menor tiempo de procedimiento y de fluoroscopia. No existen estudios publicados sobre el uso de la guía Circulo de Abbott. El objetivo fue evaluar su eficacia y seguridad para la estimulación del VI durante el TAVI.
Métodos: Estudio observacional, prospectivo y multicéntrico que incluyó pacientes sometidos a TAVI transfemoral con estimulación del VI mediante la guía Circulo. El objetivo primario fue la eficacia de la estimulación, definida como una reducción de la presión arterial sistólica por debajo de 60 mmHg. Los objetivos secundarios incluyeron el umbral de estimulación, la conversión a estimulación del VD y las complicaciones periprocedimiento.
Resultados: Entre marzo y junio de 2025 se incluyó a 50 pacientes (edad media 80,56 ± 6,26 años; 62% mujeres). La estimulación rápida del VI fue efectiva en el 100% de los casos, con un umbral medio de 7,00 ± 3,48 mV. Un total de 3 pacientes requirieron estimulación del VD por bloqueo auriculoventricular tras la predilatación con balón. La tasa global de complicaciones fue del 14%, principalmente trastornos de la conducción, sin complicaciones atribuibles a la guía Circulo.
Conclusiones: La estimulación del VI con la guía Circulo durante el TAVI es eficaz y segura, y evita el implante sistemático de un marcapasos transitorio.
Palabras clave: Implante percutáneo de válvula aórtica. Estimulación ventricular izquierda. Guía preformada. Estimulación rápida. Marcapasos. Intervención estructural cardiaca.
Abbreviations
AVB: atrioventricular block. LV: left ventricle. RV: right ventricle. TAVI: transcatheter aortic valve implantation.
INTRODUCTION
Transcatheter aortic valve implantation (TAVI) is one of the treatments that has been shown to improve prognosis and reduce mortality in patients with symptomatic severe aortic stenosis.1 Currently, according to the clinical practice guidelines outlined by the European Society of Cardiology (ESC),2 TAVI is the treatment of choice in patients aged ≥ 70 years, while the American Heart Association and the American College of Cardiology (AHA/ACC) clinical practice guidelines indicate that it may be considered from the age of 65 years.3,4.
Although TAVI is considered a safe procedure, complications may occur;5 however, their incidence has declined over the years owing, among other factors, to advances in preoperative cardiac imaging, the development of improved models of transcatheter heart valves and delivery sheaths, and the increasing experience of the teams performing the procedure.6 However, not all complications are directly associated with valve implantation, and some are due to auxiliary devices used perioperatively. One example is the temporary transvenous pacemaker traditionally used for rapid right ventricular (RV) pacing, which requires an additional puncture (generally in the femoral, jugular, subclavian, or brachial vein) and the insertion of a catheter into the RV.
A meta-analysis published in 2019 reported that, even today, despite the availability of imaging modalities that facilitate implantation, transvenous temporary pacing remains associated with complications in 22.9% of patients.7 Many of these complications could be avoided by performing left ventricular (LV) pacing using the pre-shaped guidewire during TAVI, which eliminates the need for any additional vascular accesses or specific devices.8 In 2019, the randomized clinical trial EASY TAVI compared both techniques showing a similar safety and efficacy profile, resulting in lower costs and shorter procedural and fluoroscopy times with LV pacing.9 Although the utility of the SAFARI 2 (Boston Scientific, United States), Confida (Medtronic, United States), Lunderquist (Cook Medical Inc., United States), Amplatz Extra-Stiff (Cook Medical Inc.), Amplatz Super Stiff (Boston Scientific), and SavvyWire (OpSens Inc., Canada) guidewires has already been demonstrated10,11, there are no studies on the Circulo guidewire (Abbott, United States).12
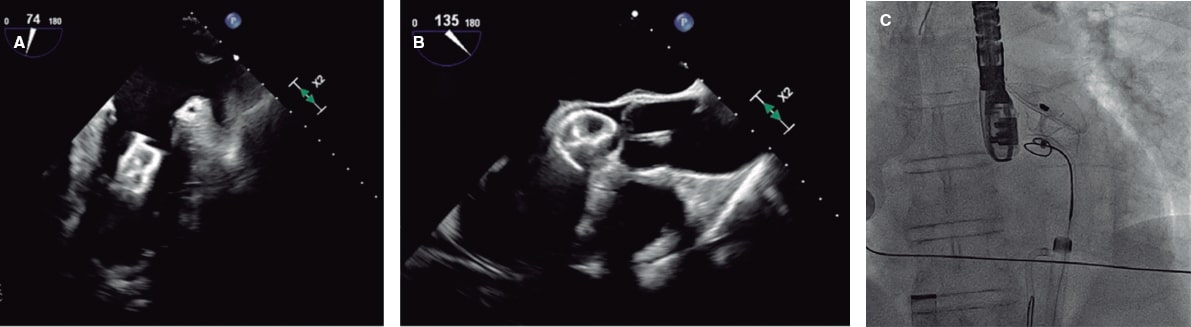
The aim of this study was to evaluate the performance of the pre-shaped Circulo guidewire for LV pacing during TAVI (figure 1A).
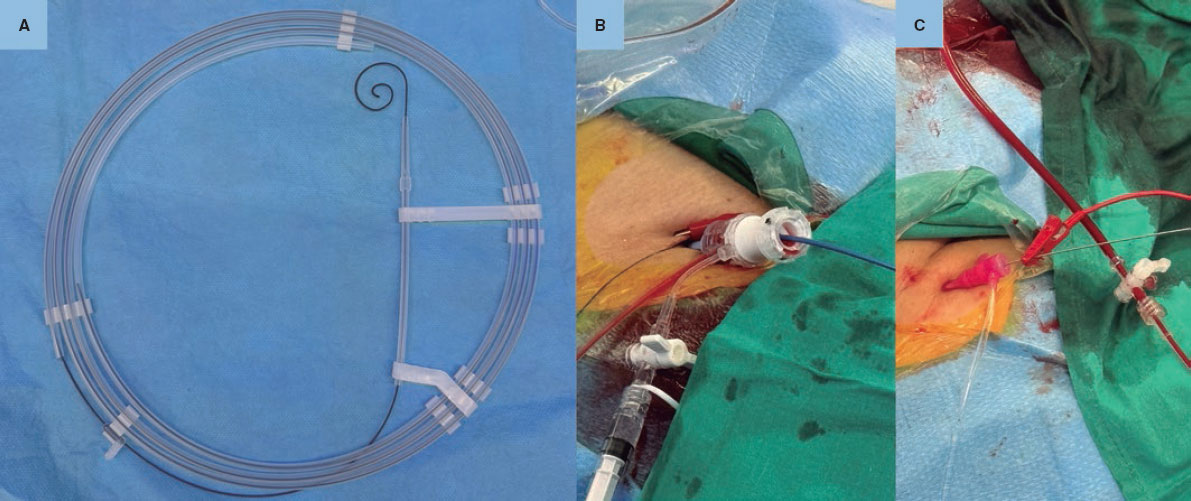
Figure 1. A: pre-shaped Circulo guidewire. B: grounding for left ventricular pacing through the skin. C: grounding for left ventricular pacing through a guidewire.
METHODS
We conducted an observational, prospective, multicenter study. The primary endpoint was to demonstrate the efficacy of rapid LV pacing, defined as a decrease in systemic systolic pressure < 60 mmHg. Secondary endpoints included pacing threshold, the need for conversion to RV pacing, and safety.
The Circulo guidewire
Circulo is a 275-cm–long polytetrafluoroethylene–coated guidewire. It features a double curve at its distal end, with a circular configuration slightly different from that of other guidewires, which reduces ventricular compression. This characteristic is particularly notable in the smaller curve.
Study population
The study protocol was approved by Hospital Clínic de Barcelona Ethics Committee (Barcelona, Spain) in full compliance with the ethical principles outlined in the Declaration of Helsinki. A total of 50 consecutive patients with severe aortic stenosis or regurgitation undergoing TAVI in 8 Spanish centers with the pre-shaped Circulo guidewire for LV pacing were prospectively included. Patients were excluded if, in the investigators’ judgment, they required RV pacing, lacked femoral access, or underwent TAVI without the need for rapid pacing.
Baseline characteristics were collected, including sex, age, weight, height, surgical risk, history of pacemaker implantation, atrial fibrillation, ischemic heart disease, or bundle branch block. Echocardiographic characteristics included LV size, LV ejection fraction, and the presence of severe valvular heart disease.
TAVI with left ventricular pacing
TAVI was performed according to the clinical practice of each center. The grounding configuration for LV pacing and the type of TAVI were decided by each center based on patient characteristics (figure 1B-C). During LV pacing, systemic blood pressure and pacing threshold were measured. To determine the LV pacing threshold, pacemaker output was initially increased to the maximum and then gradually reduced until loss of capture was observed. The lowest voltage capable of capturing the ventricle on all beats was selected. Systemic blood pressure was measured using an arterial catheter.
Procedural characteristics were recorded, including venous access, grounding configuration for LV pacing, LV pacing threshold, systemic blood pressure during rapid LV pacing, conversion to pacing using a different LV guidewire or RV pacing, type and size of the transcatheter heart valve, need for balloon pre- or postdilatation, and complications according to the Valve Academic Research Consortium-3 (VARC-3) criteria, including vascular and access-related complications, hemorrhages, neurological events, new conduction disturbances and arrhythmias, acute kidney injury, structural heart complications, length of stay, and mortality.5
Follow-up
Follow-up was conducted 30 days after TAVI, either by telephone or in person. Complications were adjudicated according to VARC-3 criteria.5
Statistical analysis
Quantitative variables are expressed as mean and standard deviation or median and interquartile range (IQR), depending on whether distribution was normal, as previously assessed using the Kolmogorov–Smirnov test. The qualitative ones are expressed as total number and percentage. Statistical analyses were performed using IBM SPSS Statistics (version 27) and RStudio (version 4.5.1). Depending on the variable, Fisher’s exact test, the Kruskal–Wallis test, or ANOVA was applied.
RESULTS
Baseline characteristics
Baseline characteristics are shown in table 1. A total of 50 patients were included in the study, 31 (62%) women and 19 (38%) men. The mean age of the cohort was 80.56 ± 6.26 years. In terms of past medical history, the mean body mass index was 27.38 ± 4.6 kg/m2; 17 patients (34%) had ischemic heart disease; 12 (24%), atrial fibrillation; 3 (6%), a previously implanted pacemaker; 4 (11.8%), first-degree atrioventricular block (AVB); and 6 (12%), bundle branch block (4 patients had left bundle branch block and 2, right bundle branch block). The PR interval ranged from 140 ms to 240 ms, with a median of 170 ms (IQR, 158-190), and QRS duration ranged from 72 ms to 170 ms, with a median of 90 ms (IQR, 80-110). Median surgical risk, assessed using the EuroSCORE II, was 2.06% (IQR, 1.62-3.36).
Table 1. Baseline characteristics of the patients
| Variable | Total (n = 50) |
|---|---|
| Demographic data | |
| Age (years) | 80.56 ± 6.26 |
| Women | 31 (62) |
| Past medical history | |
| Body mass index (kg/m2) | 27.38 ± 4.6 |
| Ischemic heart disease | 17 (34) |
| Atrial fibrillation | 12 (24) |
| Previous pacemaker | 3 (6) |
| PR duration (ms) | 170 [158-190] |
| QRS duration (ms) | 90 [80-110] |
| First-degree AVB | 4 (11.8) |
| LBBB | 4 (8) |
| RBBB | 2 (4) |
| Surgical risk | |
| EuroSCORE II (%) | 2.06 [1.62-3.36] |
| Echocardiographic characteristics | |
| LV ejection fraction (%) | 58.48 ± 8.63 |
| LV end-diastolic diameter (mm) | 45.28 ± 7.83 |
| Mean gradient (mmHg) | 47.24 ± 19.25 |
| Severe aortic regurgitation | 6 (12) |
| Severe tricuspid regurgitation | 0 (0) |
|
AVB, atrioventricular block; RBBB, right bundle branch block; LBBB, left bundle branch block; LV, left ventricle. Data express n (%), mean ± standard deviation, or median [interquartile range]. |
|
Baseline echocardiographic evaluation demonstrated a mean LV end-diastolic diameter of 45.28 ± 7.83 mm, a mean LV ejection fraction of 58.48 ± 8.63%, and a mean transaortic gradient of 47.24 ± 19.25 mmHg. Severe aortic regurgitation was present in 6 patients (12%) and was the primary valvular heart disease in 2 cases (4%). There were no patients with severe tricuspid regurgitation.
Procedural characteristics and performance of the pre-shaped guidewire
LV pacing avoided venous access in nearly half of the patients (n = 24; 48%). Among those requiring venous access, the femoral vein was the most widely used site (n = 20; 77%), followed by the brachial (n = 4; 15%) and jugular veins (n = 2; 8%). Grounding methods for LV pacing varied, with skin grounding being the one most frequently used (n = 22; 44%), followed by grounding to a guidewire via venous access (n = 19; 38%), and needle-based grounding methods (n = 9; 18%) (table 2).
Table 2. Procedural characteristics and perioperative complications and 30-day follow-up
| Procedural characteristics | Total (n = 50) |
|---|---|
| Venous access | |
| None | 24 (48%) |
| Femoral | 20 (40%) |
| Jugular | 2 (4%) |
| Brachial | 4 (8%) |
| Grounding for LV pacing | |
| Needle | 9 (18%) |
| Guidewire | 19 (38%) |
| Skin | 22 (44%) |
| Predilatation | 71 (82%) |
| Systolic blood pressure < 60 mmHg during rapid LV pacing | 50 (100%) |
| LV pacing threshold (mV) | 7.00 ± 3.48 |
| Conversion to RV pacing | 3 (6%) |
| Conversion to a different LV guidewire | 0 (0%) |
| Type of transcatheter heart valve | |
| Navitor | 28 (56%) |
| Evolut | 5 (10%) |
| ACURATE | 5 (10%) |
| SAPIEN | 6 (12%) |
| Myval | 6 (12%) |
| Transcatheter heart valve size | 25 [23-27] |
| Pacing during TAVI | |
| None | 21 (42%) |
| LV | 27 (54%) |
| RV | 2 (4%) |
| Postdilatation | 10 (20%) |
| Length of stay | 3 (2-5) |
| Total complications | 7 (14%) |
| Stroke | 0 (0%) |
| Vascular | 1 (2%) |
| Bleeding | 0 (0%) |
| Coronary obstruction | 0 (0%) |
| Cardiac tamponade | 0 (0%) |
| Device embolization | 0 (0%) |
| Perioperative AVB | 6 (12%) |
| Permanent pacemaker | 4 (8%) |
| In-hospital mortality | 0 (0%) |
| 30-day readmission | 0 (0%) |
| 30-day mortality | 0 (0%) |
|
AVB, atrioventricular block; LV, left ventricle; RV, right ventricle. |
|
The Navitor valve (Abbott, United States) was the most frequently used transcatheter heart valve (n = 28; 56%), followed by the SAPIEN (Edwards Lifesciences, United States) and the Myval (Meril Life Sciences, India) valves (n = 6; 12%). The least frequently used ones were the Evolut (Medtronic, United States) and the ACURATE (Boston Scientific, United States) (n = 5; 10%). Self-expanding valves accounted for 76% of all implanted transcatheter heart valves. Valve sizes ranged from 23 mm to 34 mm, with a median of 25 mm (IQR, 23-27). Predilatation was performed in most patients (n = 41; 82%), whereas postdilatation was required in only 20% (n = 10) (table 2). The mean LV pacing threshold was 7 ± 3.48 mV, and in 100% of patients (n = 50), a decrease in systolic blood pressure < 60 mmHg was achieved without loss of capture during rapid LV pacing. Comparative data across the different guidewires are presented in table 3. No patient required conversion to an alternative LV pacing guidewire, and only 3 (6%) required RV pacing, all due to AVB following balloon predilatation or during advancement of the valve into the LV. No guidewire exchange was required to advance the TAVI delivery system (table 2).
Table 3. Performance of the preformed left ventricular pacing guidewire
| Study and guidewire | LV pacing threshold (mV) | Procedural success | |||
|---|---|---|---|---|---|
| Mean | SD | Median | IQR | n (%) | |
| Circulo (n = 50) | 7.00 | 3.48 | 7 | 3.8-9.25 | 50 (100) |
| Tamura et al. (2022)10 (n = 204) | 202 (99.02) | ||||
| CONFIDA (n = 34) | 3.36 | 1.66 | 3 | 2-5 | 34 (100) |
| SAFARI (n = 163) | 5.85 | 2.68 | 5 | 4-7 | 161 (98.8) |
| SAFARI Small (n = 110) | 5.97 | 3.01 | 5 | 4-7 | 108 (98.2) |
| SAFARI Extra Small (n = 53) | 5.62 | 1.96 | 6 | 4-7 | 53 (100) |
| Lunderquist (n = 7) | 7.5 | 5.94 | 5 | 3.25-10.5 | 7 (100) |
|
IQR, interquartile range; LV, left ventricle; SD, standard deviation. |
|||||
Complications
The overall rate of complications was 14% (n = 7), with conduction disturbances being the most frequent. Perioperative AVB requiring temporary pacemaker implantation occurred in 6 patients (12%), 4 of whom (8%) exhibited postoperative persistent complete AVB and eventually required permanent pacemaker implantation. One patient experienced a minor vascular complication in the form of vascular occlusion, which was successfully treated by transcatheter procedure and resolved without sequelae. There were no strokes or other major adverse events. The median length of stay from TAVI to discharge was 3 days (IQR, 2-5). The in-hospital survival rate was 100%, and no readmissions or deaths were recorded at the 1-month follow-up. Perioperative complications and those occurring within 1 month after the procedure are shown in table 2.
DISCUSSION
The main findings of the study were these: a) LV pacing using the pre-shaped Circulo guidewire was effective, achieving a reduction in systolic blood pressure < 60 mmHg in 100% of patients; b) no patient required switching to an alternative LV pacing guidewire; c) only 3 patients required conversion to RV pacing, all due to AVB following predilatation; d) the procedural success rate of TAVI was 100%; and e) there were no complications related to the Circulo guidewire. These results demonstrate the safety and efficacy profile of LV pacing using the Circulo guidewire and support its usefulness during transfemoral TAVI.
In this study, the success rate of LV pacing with the Circulo guidewire was comparable to that reported in former studies using other guidewires.8-11,13,14 Only 4% of patients presented right bundle branch block on baseline electrocardiography, indicating a cohort with a low risk of developing conduction disturbances. No patient required switching to an alternative LV pacing guidewire, and only 3 (6%) required conversion to RV pacing due to the onset of AVB after balloon predilatation or during valve advancement into the LV. In 2 of these 3 cases, LV pacing had been effective, with adequate pacing thresholds of 2.5 mV and 3.0 mV; however, although the procedure could have been continued using LV pacing, the operators decided to implant a transvenous temporary pacemaker before valve implantation, anticipating the need for pacing support after the procedure. In the third case, LV pacing was attempted, but due to the absence of adequate capture, conversion to RV pacing with implantation of a transvenous temporary pacemaker was performed. These patients did not present notable clinical differences, and aside from predilatation, no additional predictive factors were identified. The pacing thresholds recorded were slightly higher than those observed in former studies (7 mV vs 3.36 mV-7.5 mV);10 however, these discrepancies were not clinically relevant and are plausibly explained by inter-operator variability inherent to the technique.
Because transvenous temporary pacemaker implantation was only required in patients who developed AVB during or after the procedure, LV pacing with the Circulo guidewire avoided the implantation of 44 temporary pacemakers among 50 cases (88%). In former studies, this approach has been shown to reduce both procedural and fluoroscopy times.9,15,16 Therefore, although LV pacing can be performed in all patients, it is most beneficial in those at low risk of AVB and subsequent pacing requirements. In such cases, transvenous temporary pacemaker implantation can be avoided, a procedure associated with potential complications, such as inadvertent arterial puncture (0.3%), major hemorrhage (2.7%), deep vein thrombosis (0.3%), pulmonary thromboembolism (0.1%), pneumothorax (0.1%), cardiac perforation (0.7%), lead dislodgement (3.4%), pacing failure (7.7%), and the need for multiple placement attempts (0.9%).7
Although it is evident that LV pacing reduces most complications associated with transvenous RV temporary pacemaker implantation, the main concerns regarding LV pacing have focused on the risk of aortic bioprosthesis embolization due to loss of capture and the risk of perforation related to advancing the guidewire inside the LV during pacing. However, multiple studies have shown no significant differences in valve embolization rates between the 2 techniques, with some reporting a trend toward a lower risk of cardiac tamponade with LV pacing, likely attributable to the greater myocardial thickness of the LV vs the RV.9,14,15 In our study, there were no cases of device embolization or cardiac tamponade or LV pacing-related complications.
Study limitations
One limitation of the present study is that guidewire selection was left to the operator’s discretion, which may have introduced selection bias. In the cohort presented, only 4% of patients exhibited right bundle branch block on baseline electrocardiography, which is representative of a population at a low risk of developing arrhythmic complications. However, the operator-dependent choice of guidewire enhances the external validity of the results, as it more accurately reflects real-world clinical decision-making. Another limitation is the absence of a comparison group undergoing RV or LV pacing with other guidewires, which limits the ability to directly compare outcomes between techniques. Instead, results were compared with those of previously published studies.
CONCLUSIONS
The use of the Circulo guidewire for LV pacing during TAVI proved to be safe and effective, with a low overall rate of TAVI-related complications and no guidewire-related complications. This approach avoids the need for transvenous temporary pacemaker implantation in most patients at low risk of arrhythmic complications, thus potentially reducing procedural risks and simplifying perioperative management.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The study protocol was approved by Hospital Clínic de Barcelona Clinical Research Ethics Committee in full compliance with the ethical principles outlined in the Declaration of Helsinki. All participants signed informed consent prior to inclusion in the study. No sex-based subgroup analysis was performed, as this could compromise the statistical power of the study and both sexes were adequately represented in the cohort; furthermore, there is no prior evidence suggesting sex-related differences in response to LV pacing using a pre-shaped guidewire.
DECLARATION ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the development of this article.
AUTHORS’ CONTRIBUTIONS
A. Regueiro designed the protocol, database, and study framework, and coordinated statistical analysis, data interpretation, and manuscript drafting. I. Anduaga contributed to protocol design, database development, and study framework, performed statistical analysis and data interpretation, and drafted the manuscript. A. Ruberti, V. Vilalta, I.J. Amat-Santos, F. Díez-Delhoyo, L.L. Gheorghe, J.A. Baz, J.F. Díaz Fernández, L. Gutiérrez-Alonso, and X. Carrillo participated in data collection and critically reviewed the manuscript. All authors approved the final published version.
CONFLICTS OF INTEREST
A. Regueiro is a consultant for Abbott, Edwards, Haemonetics, Medtronic, and Meril. J.F. Díaz Fernández is a consultant for Abbott, Boston Scientific, and Medtronic. I.J. Amat-Santos is a proctor for Boston Scientific, Medtronic, Meril, and MicroPort. The remaining authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- LV pacing using pre-shaped guidewires has emerged as an effective alternative to RV pacing with transvenous temporary pacemaker implantation.
- LV pacing has been associated with shorter procedural and fluoroscopy times.
- Although LV pacing has proven safe and effective with several guidewires, not all guidewires currently used in TAVI have proven useful for this purpose.
WHAT DOES THIS STUDY ADD?
- The pre-shaped Circulo guidewire, with its circular design, proved to be a highly effective option for LV pacing during transfemoral TAVI.
- The overall TAVI complication rate was low, and no complications attributable to the Circulo guidewire were observed.
- The Circulo guidewire avoids the need for transvenous temporary pacemaker implantation in a considerable proportion of patients and is particularly useful in those at low risk of perioperative conduction disturbances.
REFERENCES
1. Toff WD, Hildick-Smith D, Kovac J, et al. Effect of Transcatheter Aortic Valve Implantation vs Surgical Aortic Valve Replacement on All-Cause Mortality in Patients With Aortic Stenosis:A Randomized Clinical Trial. JAMA. 2022;327:1875-1887.
2. Praz F, Borger MA, Lanz J, et al. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2025;46:4635-4736.
3. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease:A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143:e72-e227.
4. Ando T, Onishi T, Kuno T, et al. Transcatheter Versus Surgical Aortic Valve Replacement in the United States (From the Nationwide Readmission Database). Am J Cardiol. 2021;148:110-115.
5. Généreux P, Piazza N, Alu MC, et al. Valve Academic Research Consortium 3:Updated Endpoint Definitions for Aortic Valve Clinical Research. J Am Coll Cardiol. 2021;77:2717-2746.
6. Ludman PF. UK TAVI registry. Heart. 2019;105(Suppl 2):s2-s5.
7. Tjong FVY, de Ruijter UW, Beurskens NEG, Knops RE. A comprehensive scoping review on transvenous temporary pacing therapy. Neth Heart J. 2019;27:462-473.
8. Faurie B, Abdellaoui M, Wautot F, et al. Rapid pacing using the left ventricular guidewire:Reviving an old technique to simplify BAV and TAVI procedures. Catheter Cardiovasc Interv. 2016;88:988-993.
9. Faurie B, Souteyrand G, Staat P, et al. Left Ventricular Rapid Pacing Via the Valve Delivery Guidewire in Transcatheter Aortic Valve Replacement. JACC Cardiovasc Interv. 2019;12:2449-2459.
10. Tamura Y, Tamura Y, Konami Y, et al. Comparison of left ventricular pacing performance among pre-shaped guidewires designed for transfemoral-approach transcatheter aortic valve implantation. Heart Vessels. 2022;37:460-466.
11. Regueiro A, Alperi A, Vilalta V, et al. Safety and Efficacy of TAVR With a Pressure Sensor and Pacing Guidewire:SAFE-TAVI Trial. JACC Cardiovasc Interv. 2023;16:3016-3023.
12. Blusztein D, Raney A, Walsh J, Nazif T, Woods C, Daniels D. Best Practices in Left Ventricular Pacing for Transcatheter Aortic Valve Replacement. Struct Heart. 2023;7:100213.
13. Hilling-Smith R, Cockburn J, Dooley M, et al. Rapid pacing using the 0.035-in. Retrograde left ventricular support wire in 208 cases of transcatheter aortic valve implantation and balloon aortic valvuloplasty. Catheter Cardiovasc Interv. 2017;89:783-786.
14. Sta˛pór M, Tre˛bacz J, Wiewiórka Ł, et al. Direct left ventricular wire pacing during transcatheter aortic valve implantation. Kardiol Pol. 2020;78:882-888.
15. Hokken TW, de Ronde M, Wolff Q, et al. Insights in a restricted temporary pacemaker strategy in a lean transcatheter aortic valve implantation program. Catheter Cardiovasc Interv. 2022;99:1197-1205.
16. Savvoulidis P, Mechery A, Lawton E, et al. Comparison of left ventricular with right ventricular rapid pacing on tamponade during TAVI. Int J Cardiol. 2022;360:46-52.