Article
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España
ABSTRACT
Introduction and objectives: The results of Magmaris implantation in the acute coronary syndrome setting is uncertain and more studies will be needed to assess the long-term safety profile of these devices. The objective of this work was to conduct an observational study to analyze the clinical safety profile of Magmaris implanted in a single hospital center in the acute coronary syndrome setting beyond 12 months.
Methods: Registry of 36 patients with Magmaris devices implanted between November 2016 through November 2018 with a diagnosis of acute coronary syndrome included consecutively. The primary endpoint was considered the device-oriented composite endpoint of target vessel myocardial infarction, target lesion failure, and cardiac death. Secondary endpoints included Magmaris related thrombosis.
Results: Regarding the device-oriented combination, no target vessel myocardial infarction was observed, 0 cases (0%), while target lesion failure was seen in 2 cases (5.6%). There were no cases of Magmaris thrombosis at the follow-up and only 1 case of cardiac death (2.8%) was found 36 months after Magmaris implantation. The cause of death could not be determined since no autopsy was performed.
Conclusions: Our results with long-term follow-up confirm that Magmaris has a favorable clinical profile in the acute coronary syndrome complex setting.
Keywords: Magmaris. Acute coronary syndrome. Bioresorbable scaffold thrombosis.
RESUMEN
Introducción y objetivos: Los resultados del Magmaris en el síndrome coronario agudo son controvertidos y se necesitan más estudios para evaluar su seguridad a largo plazo. El objetivo del trabajo fue analizar mediante un estudio observacional la seguridad clínica más allá de 12 meses de los Magmaris implantados en un único centro hospitalario en pacientes con síndrome coronario agudo.
Métodos: Se registraron de manera consecutiva 36 pacientes con Magmaris implantados entre noviembre de 2016 y noviembre de 2018 con diagnóstico de síndrome coronario agudo. Para el objetivo primario se consideró el combinado orientado al dispositivo de infarto de miocardio del vaso diana, fracaso de la lesión diana y muerte de causa cardiovascular. Como objetivo secundario se incluyó la trombosis del dispositivo.
Resultados: En cuanto al combinado orientado al dispositivo no se observó infarto de miocardio del vaso diana (0%), en 2 casos (5,6%) se observó fracaso de la lesión diana y se constató 1 caso de muerte cardiaca (2,8%) a los 36 meses del implante del Magmaris, sin poder conocer la causa por no disponer de autopsia. Con respecto a los objetivos secundarios, no hubo casos de trombosis del Magmaris durante el seguimiento.
Conclusiones: Nuestros resultados, con un seguimiento a largo plazo, apoyan que los Magmaris presentan un perfil clínico favorable en el escenario complejo del síndrome coronario agudo.
Palabras clave: Magmaris. Sindrome coronario agudo. Trombosis armazon bioabsorbible.
INTRODUCTION
Magnesium-based bioresorbable scaffolds (Magmaris) are safe devices with good results in the long run like the BIOSOLVE II1 and BIOSOLVE III2 clinical trials show where no device thrombosis was seen at the long-term 12- to 24-month follow-up. Despite this fact, the device own limitations (ill-advised in cases of calcified complex coronary anatomy or in long lesions) have reduced its use significantly in the routine clinical practice to the point that only 224 procedures with bioresorbable devices were performed in Spain in 2019 (0.2% of the total number of devices implanted).3
As already mentioned, the good results reported in long-term follow-ups have turned the Magmaris (Biotronik, Germany) into the only bioresorbable metal scaffold to receive the CE marking (Conformité Européenne).4
The role Magmaris plays in the acute coronary syndrome setting is not widely known and further studies will be needed before its safety profile can be assessed. The objective of this work was to analyze—through an observational study in the routine clinical practice—the long-term (> 12 months) clinical safety of Magmaris scaffolds implanted in patients with acute coronary syndrome in the cath lab of a single center.
METHODS
Consecutive observational registry of patients diagnosed with acute coronary syndrome implanted with magnesium-based bioresorbable scaffolds between November 2016 and November 2018. The study was approved by the hospital ethics committee and all patients gave their signed written informed consent to participate in the study. The study primary endpoint was a composite of target vessel myocardial infarction, target lesion failure, and cardiovascular death. The study secondary endpoint included the device thrombosis. The PSP strategy (predilation, sizing, and postdilation) derived from the GHOSTEU registry was used in all the cases.5 In 100% of the patients the optical coherence tomography was used for the right characterization of the lesion and size of the vessel.
RESULTS
A total of 36 patients (29 males, 80%) were included with a median age of 59.61 ± 9.74 years. The follow-up period was 1001 days with an interquartile range of 342 days. Table 1 summarizes the baseline clinical characteristics of the sample as well as the main angiographic characteristics.
Table 1. Baseline clinical characteristics and angiographic parameters of the patients
| N (%) | |||
|---|---|---|---|
| Family history of ischemic heart disease | 11 (30.6) | ||
| Arterial hypertension | 19 (52.8) | ||
| Diabetes mellitus | 7 (19.4) | ||
| Dyslipidemia | 23 (63.9) | ||
| Smoker | 23 (63.9) | ||
| Type of acute coronary syndrome: | |||
| NSTEACS | 23 (63.9) | ||
| STEACS | 8 (22.2) | ||
| Unstable angina | 5 (13.9) | ||
| Number of diseased vessels: | |||
| 1 vessel | 16 (44.4) | ||
| 2 vessels | 15 (41.7) | ||
| 3 vessels | 5 (13.9) | ||
| Location of the lesion treated with Magmaris: | |||
| LAD | 27 (75%) | ||
| RCA | 10 (27.8%) | ||
| LCX | 4 (11.1%) | ||
| AHA classification of coronary lesions: | |||
| Type A | 16 (44.5%) | ||
| Type B | 12 (33.3%) | ||
| Type C | 8 (22.2%) | ||
| Immediate success after device implantation | 36 (100%) | ||
| Drug-eluting stent implantation | 12 (36%) | ||
| Normal LVEF | 26 (72.2%) | ||
| Antiplatelet therapy at discharge: | |||
| Acetylsalicylic acid | 36 (100%) | ||
| Ticagrelor | 29 (80.6%) | ||
| Clopidogrel | 6 (16.7%) | ||
| Prasugrel | 1 (2.8%) | ||
| Prolonged DAPT > 12 months | 14 (38.9%) | ||
| Statins | 36 (100%) | ||
| Beta-blockers | 31 (86.1%) | ||
| Angiographic parameters | Length (mm) | Diameter (mm) | Peak inflation pressure (atm) |
| Target lesion | 29.2 ± 13.4 | 3.4 ± 0.2 | |
| Predilation (noncompliant balloon) | 16.8 ± 2.9 | 3.2 ± 0.4 | 20.2 ± 1.2 |
| Magmaris | 22.5 ± 3.05 | 3.4 ± 0.2 | 15.9 ± 0.9 |
| Postdilation (noncompliant balloon) | 21.4 ± 1.5 | 3.7 ± 0.3 | 21.4 ± 1.5 |
|
AHA, American Heart Association; DAPT, dual antiplatelet therapy; LAD, left anterior descending coronary artery; LCX, left circumflex artery; LVEF, left ventricular ejection fraction; NSTEACS, non-ST-segment elevation acute coronary syndrome; RCA, right coronary artery; STEACS, ST-segment elevation acute coronary syndrome. |
|||
A total of 100% of the patients received a Magmaris device in the target lesion causing the study acute coronary event. Drug-eluting stents were implanted at the discretion of the operator in 12 of the 36 patients (33.3%), and in 1 patient only (2.8%) the implantation of the stent and the Magmaris scaffold overlapped. However, in the remaining patients they were not implanted in the target vessel.
Only 1 Magmaris scaffold was used in 15 patients (41.7%), 2 in 12 cases (33.3%), 3 in 2 cases (5.6%), 4 in 3 cases (8.3%), 5 in 3 cases (8.3%), and a maximum of 6 Magmaris devices in 1 single patient (2.8%). In 20 patients (55.6%) the stent and the Magmaris device implantation overlapped.
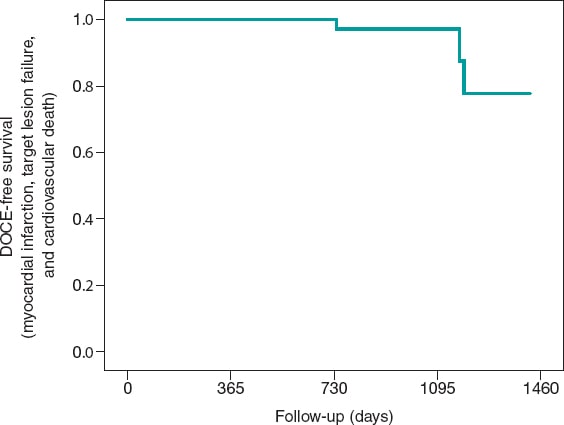
Regarding the device-oriented composite endpoint, 0 cases of target vessel myocardial infarction occurred (0%). However, 2 cases of target lesion failure were confirmed (5.6%), and 1 noncardiovascular death (2.8%) was reported at the 36 months following Magmaris implantation. However, the cause remained elusive for the lack of an autopsy (figure 1). Regarding the secondary endpoints, no Magmaris thrombosis was reported at the follow-up (table 2).
Figure 1. Kaplan-Meier survival curve with respect to the study primary endpoint: a device-oriented composite endpoint of target vessel myocardial infarction, target lesion failure, and cardiovascular death. DOCE, device-oriented composite endpoint.
Table 2. Primary (device-oriented composite endpoint) and secondary clinical events of the patients at the follow-up (N = 36, 100%)
| Event | Patients | Percentage |
|---|---|---|
| Target lesion failure | 2 | 5.6 % |
| Target vessel myocardial infarction | 0 | 0 % |
| Cardiovascular death* | 1 | 2.8 % |
| Magmaris device thrombosis | 0 | 0 % |
|
* 1084 days after Magmaris implantation. |
||
A total of 11 admissions were reported at the follow-up (30.6%). Among these, 8 were due to recurrent angina (22.2%), 2 to heart failure or de novo atrial fibrillation (5.6%), and 1 to atrioventricular block that required pacemaker implantation (2.8%).
The coronary angiography was repeated in 10 cases (27.8%), the absence of lesions was confirmed in 3 patients (8,3%), stent restenosis was reported in 1 patient 2.8%), Magmaris restenosis in 2 (5.6%), and de novo lesions in 4 patients (11.1%).
When the cases of Magmaris restenosis were studied across time it was found that all of these occurred after the 24-months follow-up (1 case after 737 days and the other after 1189 days). The cardiovascular death reported in 1 patient occurred 1084 days after implantation.
DISCUSSION
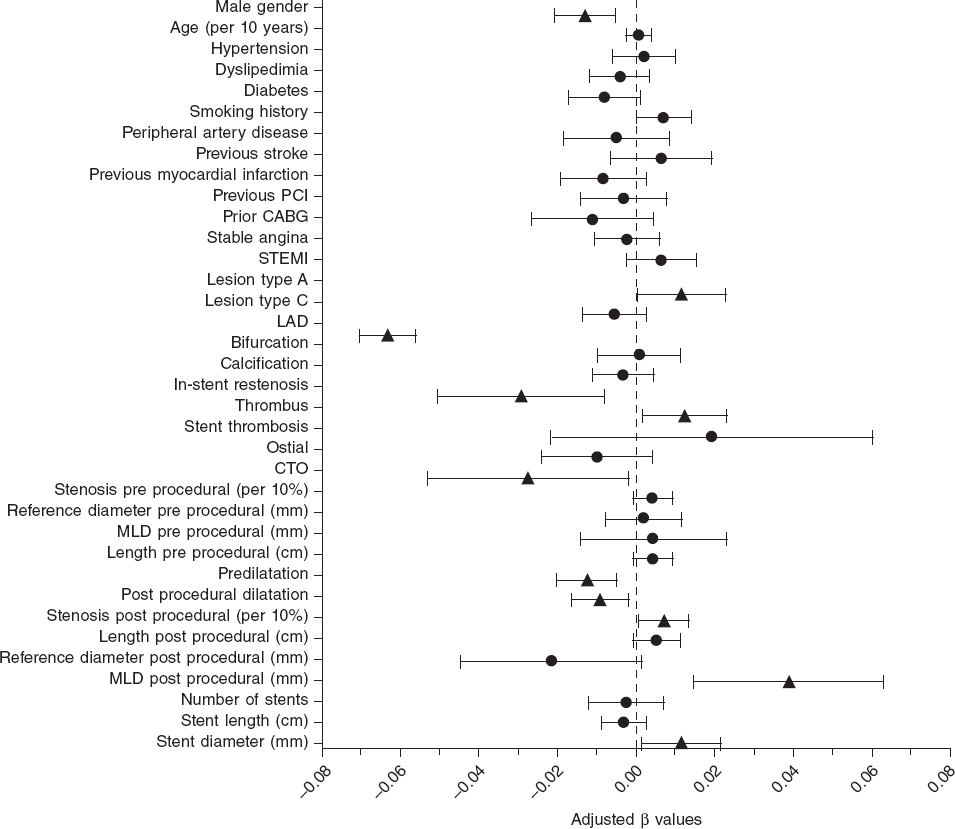
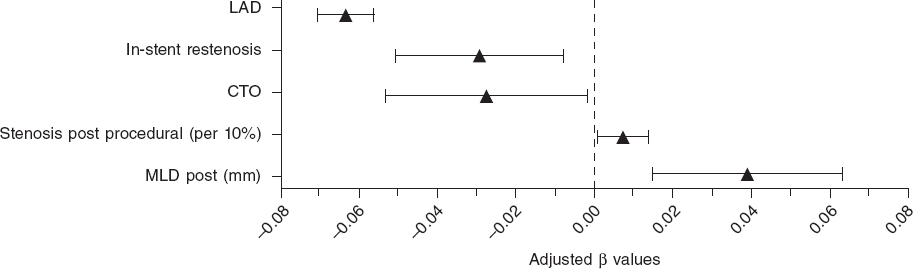
The magnesium-based bioresorbable scaffold (Magmaris) is a highly successful device when implanted following the recommendations made by the manufacturer6 including the right predilation and optimization of the lesions using intracoronary imaging modalities. A study proved that an optimal PSP technique was not associated with a lower rate of the device-oriented composite endpoint. However, patients with optimal PSP-3 had numerically fewer episodes compared to patients without optimal PSP-3 (0.5% vs 2.9%, P = .085, and 0.5% vs 1.8%, P = .248, respectively).7
The real-world 12-month follow-up results of a cohort registry with Magmaris have recently been published. These results have confirmed the Magmaris safety profile and its low rate of events (target lesion revascularization in 4.7%) and lack of thrombosis.8
Also, the use of magnesium-based bioresorbable scaffolds has been studied in a group of 50 patients with non-ST-segment elevation acute coronary syndrome. This device reached angiographic success in 100% of the cases. One case of failed target vessel revascularization was reported the day after the procedure that required the implantation of a bare-metal stent. No device-related events were reported at the 6-month follow-up.9
The Magmaris scaffold and the sirolimus-eluting stent were compared in a controlled, randomized, blinded, multicenter study of patients with ST-segment elevation acute coronary syndrome. This study proved that in 150 patients the primary endpoint of a greater vasomotor response to medication was better in the Magmaris group at the 1-year follow-up. However, the Magmaris scaffold was associated with a worse angiographic progression and a greater late luminal loss compared to the bare-metal stent. It was also associated with a higher rate of target lesion revascularization without a significantly higher number of thrombotic events being reported.10
In our registry, the immediate rate of successful device implantation was 100%. Device overlapping occurred in 55.6% of the cases with a high frequency of treatment of 2 and 3 vessels (55.4%). Despite the complexity of the lesions, device restenosis was only reported in 2 cases (5.6%) at the > 3-year follow-up. The rate of thrombosis at the follow-up was 0 cases and the rate of cardiovascular death—considering 1 case with the devices implanted 36 months beforehand (and without necropsy or previous clinical assessment)—was 2.8%. In our study of real-world clinical practice this confirms the device safety profile in the short and mid-term in the acute coronary syndrome setting. The results of the BIOSOLVE IV registry reported a target lesion failure rate of 4.3%,11 similar to the one seen in our registry—5.5%—despite our patients’ profile of higher ischemic risk (all of them with acute coronary syndrome and an average lesion length of 29.17 mm ± 13.39 mm with Magmaris overlapping in 55.6% of the cases).
Limitations
Our study main limitation is its small sample size, which shows the low penetration of resorbable scaffolds in our hospital setting. Another limitation is the registry observational nature with an inherent selection bias, without defined inclusion and exclusion criteria, and the use of second-generation drug-eluting stents in 33.3% of the patients outside de target vessel except for 1 case where the stent and the Magmaris device overlapped (2.8%).
Another limitation was the lack of an independent clinical event adjudication committee. However, 100% of the patients were followed-up (through electronic health records and phone calls) by the research working team.
CONCLUSIONS
In our registry of patients with acute coronary syndrome who received the Magmaris scaffold the primary endpoint of target lesion failure or target vessel myocardial infarction did not increase compared to registries previously published. No cases of scaffold definitive thrombosis were reported at the follow-up, and only 1 cardiovascular death was reported 36 months after implantation without knowing the definitive cause. Considering the aforementioned limitations, our results confirm that Magmaris scaffolds could have a favorable clinical profile in the complex setting of acute coronary syndrome.
FUNDING
No external funding has been received.
AUTHORS’ CONTRIBUTIONS
All authors contributed equally during the collection of clinical data and the performance of the interventional procedures including the follow-up of all of the patients.
CONFLICTS OF INTEREST
None reported.
WHAT IS KNOWN ABOUT THE TOPIC?
- Magnesium-based bioresorbable scaffolds (Magmaris) have proven safe and effective in former studies and registries.
- Despite the recent studies published on the acute coronary syndrome setting, the long-term safety of these devices has still not been confirmed yet.
WHAT DOES THIS STUDY ADD?
- A real-world registry with a very long-term follow-up showing a low rate of device-related events.
- Further multicenter registries with a high number of patients will be needed before solid conclusions can be drawn.
REFERENCES
1. Haude M, Ince H, Abizaid A, et al. Safety and performance of the second? generation drug?eluting absorbable metal scaffold in patients with de?novo coronary artery lesions (BIOSOLVE?II):6 month results of a prospective, multicentre, non?randomised, first?in?man trial. Lancet. 2016;387:31-39.
2. Haude M, Ince H, Kische S, et al. Sustained safety and clinical performance of a drug?eluting absorbable metal scaffold up to 24 months:pooled outcomes of BIOSOLVE?II and BIOSOLVE?III. EuroIntervention. 2017;13:432-439.
3. Datos de la Sección de Hemodinámica 2019. Available online: https://www.hemodinamica.com/wp-content/uploads/2020/12/Presentacion-Registro.pdf. Accessed 15 Nov 2021.
4. Sotomi Y, Onuma Y, Collet C, et al. Bioresorbable scaffold:the emerging reality and future directions. Circ Res. 201;120:1341-1352.
5. Ortega-Paz L, Capodanno D, Gori T, et al. Predilation, sizing, and post-dilation scoring in patients undergoing everolimus-eluting bioresorbable scaffold implantation for prediction of cardiac adverse events:development and internal validation of the PSP score. EuroIntervention. 2017;12:2110-2117.
6. Fajadet J, Haude M, Joner M, et al. Magmaris preliminary recommendation upon commercial launch:a consensus from the expert panel on 14 April 2016. Eurointervention. 2016;18:828-833.
7. Ortega-Paz L, Bruggaleta S, Capodanno D, et al. Efecto de la técnica de implantación en los resultados en pacientes tratados con armazón bioabsorbible en diferentes escenarios clínicos. REC Interv Cardiol. 2019;1:83-91.
8. Abellas-Sequeiros RA, Ocaranza-Sánchez R, Bayon-Lorenzo J, et al. 12-month clinical outcomes after Magmaris percutaneous coronary intervention in a real-world cohort of patients:Results from CardioHULA registry. Rev Port Cardiol. 2020;39:421-425.
9. Wlodarczak A, Lanocha M, Jastrzebski A, et al. Early outcome of magnesium bioresorbable scaffold implantation in acute coronary syndrome-the initial report from the Magmaris-ACS registry. Catheter Cardiovasc Interv. 2019;93:E287-E292.
10. SabatéM, Alfonso F, Cequier A, et al. Magnesium-Based Resorbable Scaffold Versus Permanent Metallic Sirolimus-Eluting Stent in Patients With ST-Segment Elevation Myocardial Infarction:The MAGSTEMI Randomized Clinical Trial. Circulation. 2019;140:1904-1916.
11. Verheye S, Wlodarczak A, Montorsi P, et al. Safety and performance of a reservable magnesium scaffold under real-world conditions:12 month outcomes of the first 400 patients enrolled in the BIOSOLVEIV registry. Eurointervention. 2020;15:e1383-e1386.
ABSTRACT
Introduction and objectives: There is scarce information available on the long-term outcomes of primary angioplasty (PA) in patients over 80 years of age. Our objective was to analyze the characteristics and results of PA in these patients and recognize the prognostic factors and long-term survival.
Methods: Observational and retrospective single-center study of consecutive patients over 80 years of age treated with PA between January 2013 and September 2019. A long-term clinical follow-up was performed (mean follow-up of 29 ± 25 months).
Results: The study included 133 patients (mean age 85.3 ± 3.8 years and 57 women [43%]). Seventeen percent of the patients were in Killip class III-IV at admission. The mean Charlson Comorbidity index was 2.3 ± 1.6. During the hospitalization, almost half of the patients developed heart failure and mortality rate was 18%. The overall mortality rate at the follow-up was 23%, yet 97.2% of the deaths were due to non-cardiac causes. The independent predictors of overall mortality at the follow-up were chronic kidney disease (HR, 5.7; 95%CI, 1.29-25.5; P = .022), and a Charlson Comorbidity index > 2 (HR, 2.57; 95%CI, 1.07-6.18; P = .035).
Conclusions: Patients over 80 years of age treated with PA have high in-hospital and long-term mortality rates. Comorbidities and chronic kidney disease were the only independent predictors of long-term mortality.
Keywords: Elderly. Myocardial infarction. Primary angioplasty.
RESUMEN
Introducción y objetivos: Existe poca información sobre los resultados a largo plazo de la angioplastia primaria (AP) en pacientes mayores de 80 años. Nuestro objetivo fue analizar las características y los resultados de la AP en estos pacientes, y valorar los predictores pronósticos y la supervivencia a largo plazo.
Métodos: Estudio observacional, retrospectivo y unicéntrico de pacientes mayores de 80 años consecutivos sometidos a AP entre enero de 2013 y septiembre de 2019. Se efectuó un seguimiento clínico a largo plazo (media de 29 ± 25 meses).
Resultados: Se incluyeron 133 pacientes (57 [43%] mujeres) con una edad media de 85,3 ± 3,8 años. El 17% se encontraban en clase Killip III o IV. El índice de Charlson medio fue de 2,3 ± 1,6. En cuanto a la evolución hospitalaria, casi la mitad de los pacientes desarrollaron insuficiencia cardiaca y un 18% fallecieron durante el ingreso. La mortalidad total en el seguimiento a largo plazo fue del 23%, siendo el 97,2% de las muertes de causa no cardiaca. Los predictores independientes de mortalidad total en el seguimiento a largo plazo fueron la enfermedad renal crónica (hazard ratio [HR] = 5,7; intervalo de confianza del 95% [IC95%], 1,29-25,5; p = 0,022) y el índice de Charlson mayor de 2 (HR = 2,57; IC95%, 1,07-6,18; p = 0,035).
Conclusiones: Los pacientes mayores de 80 años sometidos a AP tienen una elevada mortalidad hospitalaria y en el seguimiento a largo plazo. La comorbilidad y la enfermedad renal crónica resultaron ser los únicos predictores independientes de mortalidad a largo plazo.
Palabras clave: Anciano. Infarto de miocardio. Angioplastia primaria.
Abbreviations
PA: primary angioplasty.
INTRODUCTION
The current estimates reveal the population gradual aging, which will be more evident in the coming years.1 Based on these estimates, by the year 2050, our country will become one of the oldest worldwide with more than 4 million people over 80 years of age. This means that the percentage of patients treated with primary angioplasty (PA) is on the rise in our setting.
Although old age is associated with worse prognoses, PA is still the best reperfusion strategy for these patients.2-5 This segment of the population has a high prevalence of comorbidities, is often recommended fewer treatments, and has a higher risk of complications during revascularization procedures. Also, these patients are often misrepresented in the clinical trials, meaning that there is little scientific evidence available on the clinical characteristics, results, and long-term prognosis after PA.6
The objectives of this study were to analyze the characteristics, results, mortality, and prognostic predictors of patients > 80 treated with PA in our center.
METHODS
Single-center, retrospective, and observational study. All patients > 80 treated with PA in our center from January 2013 through September 2019 were included. Different clinical and epidemiological variables like age, sex, cardiovascular risk factors, presence of comorbidities, and the total ischemic time were prospectively registered in the unit database. The Charlson Comorbidity was retrospectively obtained at admission to stratify the patients’ overall comorbidities.7,8 This study was approved by Hospital Universitario Fundación Alcorcón ethics committee and waiver of informed consent was accepted.
Catheterization and treatment
Most cases were treated with percutaneous coronary intervention using the standard technique via radial access. The contrast agents used in all the cases were iohexol (Omnipaque 350, and Omnipaque 300), and iodixanol (Visipaque 320). The number of main vessels damaged seen on the coronary angiography, the access route, the dose of contrast used, the x-ray image time, and the number and type of stent use were recorded. Angiographic success and the presence of complications during the procedure were recorded as well. The operator chose the type of stent he would use during the procedure, although the local protocol recommended the use of conventional stents preferably. Drug-eluting stents were spared for situations of high risk of restenosis.
Follow-up and endpoints
Follow-up data were obtained after reviewing our hospital electronic health records. Also, phone calls to the patient or his family were made followed by a standard survey when appropriate. The endpoints studied were in-hospital mortality and complications, cardiovascular events, and cardiac death at the long-term follow-up.
Definitions
Left ventricular systolic function was estimated on the echocardiogram. The presence of a left ventricular ejection fraction < 45% was considered moderate-to-severe left ventricular dysfunction. Cardiogenic shock was defined as systolic arterial pressure < 90 mmHg for, at least, 1 hour followed by tissue hypoperfusion that required inotropic support and/or intra-aortic balloon pump implantation. Cardiac deaths were due to acute coronary syndrome, heart failure or ventricular arrhythmia. Angiographic success was defined as the presence of TIMI grade ≥ 2 flow in the absence of residual stenosis > 50%. The glomerular filtration rate was estimated using the simplified modification of diet in renal disease (MDRD) equation.9 Chronic kidney disease was defined as a glomerular filtration rate < 60 mL/min/ 1.73 m2 at admission. Bleeding complications associated with vascular access were classified based on the Bleeding Academic Research Consortium (BARC) definitions.10 BARC type > 2 hemorrhages were considered major bleeding. Target lesion revascularization was defined as the need for a new revascularization procedure (whether percutaneous or surgical) of the coronary segment with stenting in the presence of angiographic restenosis (stenosis > 50%) and symptoms or signs of myocardial ischemia.
Statistical analysis
The statistical software package SPSS version 20 was used for the analysis of data. Quantitative variables were expressed as mean ± standard deviation. The categorical ones were expressed as absolute value and percentage.
Univariate and multivariate modified Poisson regression analyses were conducted to determine the independent prognostic factors of in-hospital mortality. The variables included in the multivariate analyses were those considered of the greatest clinical relevance: Killip Class > I at admission, age > 85, chronic kidney disease, Charlson Comorbidity index > 2, and presence of moderate-to-severe left ventricular dysfunction. Results were expressed as relative risks and their 95% confidence interval (95%CI).
Univariate and multivariate Cox regression analyses were conducted to determine the independent predictors of overall mortality at the long-term follow-up. The variables included in the multivariate analyses were those associated with a higher mortality rate in the univariate analysis and also those of the greatest clinical relevance: Killip Class > I at admission, age > 85, chronic kidney disease, Charlson Comorbidity index > 2, and presence of moderate-to-severe left ventricular dysfunction. Results were expressed as hazard ratios (HR) and their 95%CI. P values < .05 were considered statistically significant. The inter-group overall mortality-free survival rates based on the presence of chronic kidney disease and a Charlson comorbidity index > 2 were compared using the Kaplan-Meier Curves (log-rank test).
RESULTS
Clinical characteristics and of the interventional procedure
A total of 1269 PAs were performed in our center from January 2013 through September 2019. A total of 10.5% were ≥ 80 years old at admission. The study group included 133 patients (57 women [43%]) with a median age of 85.3 ± 3.8 years treated with PA. The study population had a high prevalence of cardiovascular risk factors. A total of 66.2% of the patients had chronic kidney disease. The anterior was the most common location of the infarction. A total of 16.6% of the patients were Killip Class III-IV. In 28.5% of the cases delays of more than 6 hours between the beginning of pain and reperfusion were reported. The mean Charlson Comorbidity index used to assess the comorbidities of the patients included in our series was 2.3 ± 1.6 (table 1).
Table 1. Clinical angiographic, and interventional procedure data
| Patients | N = 133 |
|---|---|
| Age (years) | 85.3 ± 3.8 |
| Sex (woman) | 57 (43%) |
| Diabetes mellitus | 46 (34.6%) |
| Dyslipidemia | 77 (57.9%) |
| Arterial hypertension | 110 (82.7%) |
| Active smoking | 4 (3%) |
| Charlson Comorbidity index | 2.3 ± 1.6 |
| Body mass index | 26.4 ± 3.3 |
| Previous infarction | 23 (17.3%) |
| Previous angioplasty | 18 (13.5%) |
| Previous coronary artery bypass surgery | 3 (2.3%) |
| Atrial fibrillation | 31 (23.3%) |
| LVEF echocardiogram | 47.1 ± 11 |
| LVEF < 50% | 61 (45.8%) |
| Creatinine levels at admission (mg/dL) | 1.25 ± 0.44 |
| GFR-MDRD (mL/min/1.73 m2) | 52.2 ± 18.5 |
| Chronic kidney disease* | 88 (66.2%) |
| Location of the infarction | |
| Anterior | 62 (46.6%) |
| Inferior | 48 (36.1%) |
| Lateral | 11 (8.3%) |
| Undetermined | 9 (6.8%) |
| Cardiac arrest | 3 (2.3%) |
| Killip Class | |
| I | 100 (75.1%) |
| II | 11 (8.3) |
| III | 7 (5.3) |
| IV | 15 (11.3) |
| Total ischemic time > 6h | 37 (28.5%) |
| Median of total ischemic time (min) | 268 [177-406] |
| Median of time from symptom onset until arrival at the PA-capable left (min) | 203 [124-330] |
| Median of time from the arrival at the PA-capable leftuntil guidewire passage (min) | 50 [37-77] |
| X-ray image time (min) | 16.6 ± 13 |
| Volume of contrast (mL) | 173 ± 72 |
| Radial access | 107 (80.5%) |
| Number of diseased vessels | |
| 1 | 70 (52.6%) |
| 2 | 39 (29.3%) |
| 3 | 24 (18%) |
| Number of stents implanted | 1.04 ± 0.2 |
| Thrombus aspiration | 34 (25.6%) |
| Glycoprotein IIb/IIIa inhibitors | 17 (12.8%) |
| Drug-eluting stent | 41 (30.8%) |
| PCI of NC lesions in the acute phase | 4 (3%) |
| PCI of NC lesions in another procedure at admission | 11 (8.3%) |
| Complete revascularization | 69 (51.9%) |
| Angiographic success | 127 (95.5%) |
| In-hospital mortality | 24 (18%) |
|
GFR-MDRD, glomerular filtration rate (Modification of Diet in Renal Disease); LVEF, left ventricular ejection fraction; NC, non-culprit; PA, primary angioplasty; PCI, percutaneous coronary intervention. Data are expressed as no. (%), mean ± standard deviation or median [interquartile range]. * Defined as a glomerular filtration rate < 60 mL/min/1.73 m2. |
|
Regarding the angiographic and procedural data, the radial access was used in 80.5% of the patients of whom 47.4% had multivessel disease. Almost half of the patients were released from the hospital with incomplete angiographic revascularizations. Thrombus aspiration was performed in one fourth of the patients and drug-eluting stents were implanted in 30.8% of these patients (table 1).
Patient progression at the hospital setting
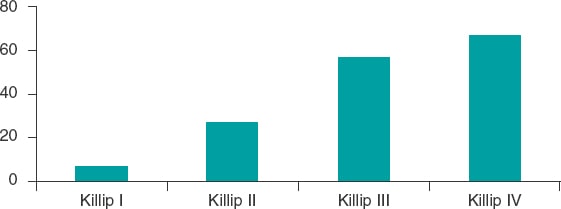
Regarding patient progression at the hospital setting, 63 patients (49%) developed heart failure and 24 patients (18%) died during admission. Two patients (1.5%) had stent thrombosis during their hospital stay. The cause of death of 21 of the dead patients (87.5%) was cardiovascular. There was a statistically significant higher in-hospital mortality rate in patients with Charlson comorbidity indices > 2 (28.9% vs 13.7%, P = .039), Killip Class > I (51.5% vs 7%; P < .001), and worse ventricular (26% vs 4.3%, P = .003) and renal functions (23.9% vs 6.7%; P = .031). The Killip Class-based mortality rate based was 7% for Killip Class I, 27.3% for Killip Class II, 57.1% for Killip Class III, and 66.7% for Killip Class IV (figure 1).
Figure 1. In-hospital mortality based on Killip Class.
In the multivariate modified Poisson regression analysis, the only independent prognostic factor of in-hospital mortality was the Killip Class at admission (relative risk, 6.5; 95%CI, 2.01-20.36; P = .001) (table 2).
Table 2. Factors associated with a higher in-hospital mortality rate. Univariate and multivariate modified Poisson regression analyses
| RR | 95%CI | P | |
|---|---|---|---|
| Univariate analysis | |||
| Age | 1.06 | 0.8-1.15 | .135 |
| Sex (woman) | 1.58 | 0.76-3.27 | .221 |
| Diabetes Mellitus | 1.35 | 0.65-2.81 | .42 |
| Killip Class > I | 7.36 | 3.34-16.22 | < .001 |
| Moderate-to-severe left ventricular dysfunction | 6.07 | 1.81-20.28 | .003 |
| Total ischemic time (hours) | 1.05 | 0.98-1.22 | .163 |
| Atrial fibrillation | 1.65 | 0.78-3.48 | .193 |
| Charlson Comorbidity index > 2 | 2.12 | 1.04-4.31 | .039 |
| Chronic kidney disease | 3.58 | 1.12-11.41 | .031 |
| Anterior location | 1.60 | 0.77-3.36 | .211 |
| Multivessel disease | 1.49 | 0.70-3.17 | .297 |
| Incomplete revascularization | 1.38 | 0.67-2.86 | .383 |
| Drug-eluting stent | 1.1 | 0.43-2.82 | .846 |
| Mulivariate analysis | |||
| Age | 1.1 | 0.99-1.21 | .074 |
| Killip Class > I | 6.5 | 2.01-20.36 | .001 |
| Chronic kidney disease | 1.23 | 0.26-5.96 | .793 |
| Charlson Comorbidity index > 2 | 2.2 | 0.9-5.38 | .083 |
| Moderate-to-severe left ventricular dysfunction | 3.05 | 0.95-9.81 | .062 |
|
95%CI, 95% confidence interval; RR, relative risk.Statistically significant results are highlighted in bold. |
|||
Long-term follow-up
A long-term follow-up was conducted of the 109 survivors. The median clinical follow-up was 24.3 months (interquartile range, 6.9-49.4 months) with 3 patients (2.8%) lost to follow-up. The clinical events occurred at the follow-up are shown on table 3. The overall mortality rate at the long-term follow-up was 23% with 97.2% of deaths due to noncardiac deaths.
Table 3. Events at the long-term follow-up
| Patients | N = 106 |
|---|---|
| New acute coronary syndrome | 10 (9.2%) |
| Target lesion revascularization | 4 (3.7%) |
| Stent thrombosis | 3 (2.8%) |
| BARC bleeding type > 2 | 19 (17.4%) |
| Stroke | 9 (8.3%) |
| Overall mortality | 25 (22.9%) |
| Cardiovascular mortality | 3 (2.8%) |
| Infection | 6 (5.5) |
| Neoplasm | 6 (5.5) |
| Respiratory failure | 5 (4.6) |
| Unknown | 5 (4.6) |
|
BARC, Bleeding Academic Research Consortium. |
|
In the univariate Cox regression analysis, the variables associated with a higher overall mortality rate were Killip Class > I (HR, 4.26; 95%CI, 2.38-7.62; P = .001), chronic kidney disease (HR, 7.24; 95%CI, 1.7-30.8; P = .007), and a Charlson Comorbidity index > 2 (HR, 2.74; 95%CI, 1.18-6.36; P = .019) (table 4). Patients with chronic kidney disease had a higher percentage of cases with Charlson Comorbidity indices > 2, but this difference was not statistically significant (19% vs 28,4%; P = .27).
Table 4. Factors associated with a higher mortality rate at the long-term follow-up. Univariate and multivariate Cox regression analyses
| HR | 95%CI | P | |
|---|---|---|---|
| Univariate analysis | |||
| Age | 1.1 | 0.99-1.23 | .076 |
| Sex (woman) | 1.66 | 0.71-3.91 | .244 |
| Diabetes Mellitus | 1.98 | 0.89-4.41 | .094 |
| Killip Class > I | 4.26 | 2.38-7.62 | .001 |
| Moderate-to-severe left ventricular dysfunction | 2.16 | 0.97-4.84 | .06 |
| Total ischemic time (hours) | 1.05 | 0.98-1.12 | .159 |
| Atrial fibrillation | 1.54 | 0.61-3.9 | .361 |
| Charlson Comorbidity index > 2 | 2.74 | 1.18-6.36 | .019 |
| Chronic kidney disease | 7.24 | 1.7-30.81 | .007 |
| Anterior location | 1.36 | 0.77-2.40 | .287 |
| Multivessel disease | 1.43 | 0.81-2.53 | .214 |
| Incomplete revascularization | 1.590 | 0.898-2.817 | .112 |
| Drug-eluting stent | 0.949 | 0.46-1.957 | .887 |
| Multivariate analysis | |||
| Age | 1.07 | 0.95-1.21 | .258 |
| Charlson Comorbidity index > 2 | 2.57 | 1.07-6.18 | .035 |
| Chronic kidney disease | 5.7 | 1.29-25.5 | .022 |
| Killip Class > I | 0.96 | 0.31-2.98 | .943 |
| Moderate-to-severe left ventricular dysfunction | 1.77 | 0.77-4.04 | .177 |
|
95%CI, 95% confidence interval; HR, hazard ratio. Statistically significant results are highlighted in bold. |
|||
In the multivariate Cox regression analysis, the only independent predictors of overall mortality were chronic kidney disease (HR, 5.7; 95%CI, 1.29-25.5; P = .022), and a Charlson Comorbidity index > 2 (HR, 2.57; 95%CI, 1.07-6.18; P = .035) (table 4).
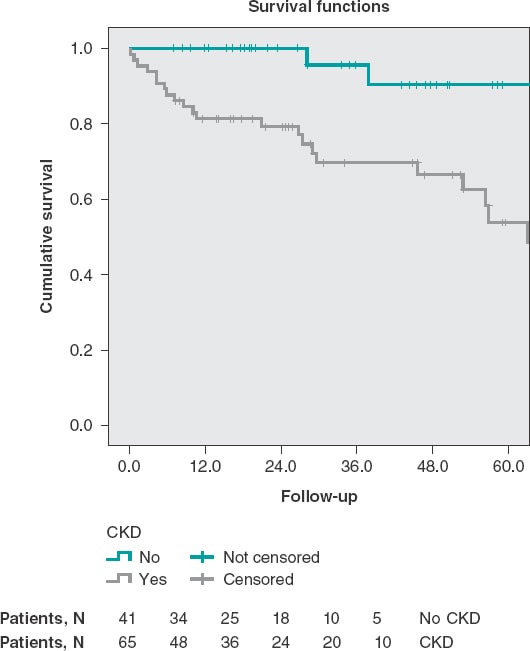
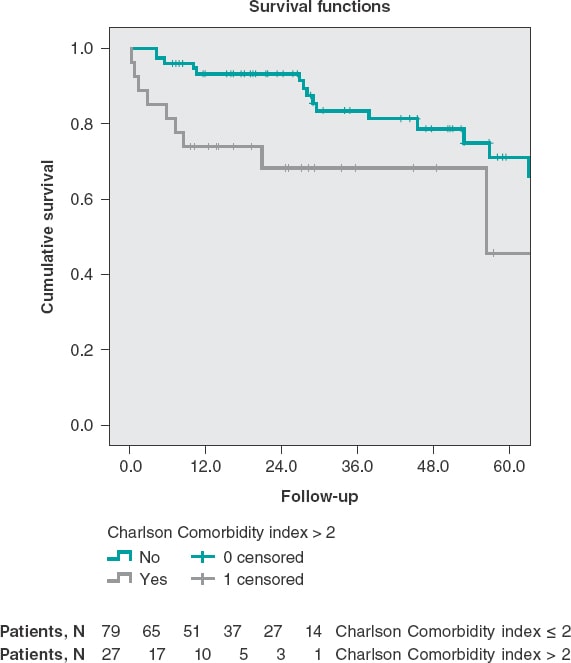
Patients with chronic kidney disease had lower survival rates at the long-term follow-up (56 ± 4.4 months vs 75 ± 3 months; P = .002) (figure 2). Patients with Charlson comorbidity indices > 2 also had lower survival rates at the long-term follow-up (45.5 ± 5.9 months vs 65.8 ± 3.3 months; P = .015) (figure 3).
Figure 2. Survival curves at the long-term follow-up stratified based on the presence of chronic kidney disease (log rank test, P = .002). ERC, chronic kidney disease.
Figure 3. Survival curves at the long-term follow-up stratified based on the presence of Charlson comorbidity indices > 2 (log rank test, P = .015).
DISCUSSION
Information on the results of PA in elderly patients and its long-term prognosis is scarce because this group of patients is often misrepresented in clinical trials.6 Our study emphasizes these patients’ high mortality rate (mainly due to cardiac causes)—both in-hospital and at the long-term follow-up—with a significant contribution from noncardiac mortality and comorbidities as prognostic predictors.
This segment of the population has special characteristics that pose an added risk. These are patients with a high prevalence of comorbidities and worse renal function.5 Diagnosis is not always easy because of the atypical symptoms reported and possible presence of previous changes on the EKG, factors that contribute to delaying reperfusion therapy.11 Finally, these are patients with a higher risk of bleeding and other complications during PA.12
Regarding the clinical profile of patients > 80 treated with PA in our center we should mention the higher percentage of women (43%) compared to other series from the general population, and the high prevalence of chronic kidney disease (66%), delays of more than 6 hours (29%), and advanced Killip Class (17%). All these characteristics are consistent with what has already been described by former studies in this population.5,13
Regarding the procedural aspects, the radial access was used in 80.5% of the cases. Elderly patients, especially women, have higher rates of failure with this access, but at the same time, these patients have the highest risk of bleeding with the femoral access. Rodríguez-Leor et al.14 reported on the possibility of achieving radial access in 95.1% in a population of patients > 75 treated with PA.
The in-hospital mortality seen in our study (18) is obviously higher to that of the general population, but not significantly different from that reported by other registries of elderly patients.12-17 In a group of 34 80-year-old patients treated with PA Sim et al.5 reported an in-hospital mortality rate of 18%. However, it went up to 37% in patients with ST-segment elevation acute coronary syndrome not treated with PA. In their prospective registry of 496 patients > 80 who received invasive treatment, Kvakkestad et al.12 reported an in-hospital mortality rate of 13%. In our series the main prognostic factor during admission was the patient’s hemodynamic situation measured using Killip Class. It is a well-known prognostic factor that has been widely described in PA studies.18
The mortality rate at the long-term follow-up was 23% with a striking contribution from noncardiac mortality, which is a differential factor with respect to series from the general population. This lower rate of adverse cardiovascular events in elderly patients who survive a myocardial infarction was found in other registries and may be due to the high early selection during the acute phase.13,17 In the aforementioned registry of Kvakkestad et al.12 the mortality rate at the 3-year follow-up was 29%. In the Swedish registry of 80-year-old patients treated with PA from 2001 to 2010, the annual mortality rate reported was 25%.17 In our series, the fact that mortality at the long-term follow-up was mostly noncardiac contributed to the fact that the main prognostic predictors at the long-term follow-up are extracardiac factors like renal function and the Charlson Comorbidity index. These factors may be understudied at the follow-up after PA.
The effect of comorbidities in the prognosis of patients is often quantified using the Charlson Comorbidity index.7,8 This index assigns a given score to a series of comorbidities based on the risk of mortality of every comorbidity. The overall score is associated with a given mortality risk. Over the last few years, interest has been growing on the analysis of comorbidities and other variables associated with age. However, data are still scarce on their prognostic influence on patients with infarction treated invasively. The existing growing heterogeneity among 80-year-old patients with infarction requires prognostic indices to stratify these patients into risk groups based on uniform criteria. Using a tool to guide us in the long-term prognosis of these patients may help us decide what the most suitable follow-up is. Several studies have proven the utility of the Charlson Comorbidity index in the acute coronary syndrome as a predictor of mortality. Núñez et al. determined the prognostic predictive value of this index in patients with myocardial infarction mostly treated conservatively.19 They found that the comorbidities present at admission were associated with higher rates of mortality or reinfarction at the 30-day and 1-year follow-up. In our series of invasive management, we found that a Charlson Comorbidity index > 2 was an independent predictor of mortality at the long-term follow-up. However, it is not a predictor of patient progression at the hospital setting where the most important thing is the patient’s hemodynamic situation. Therefore, in this population the Charlson Comorbidity index can help us plan their long-term follow-up.
Glomerular filtration rate impairment is a powerful predictor of mortality in different conditions including myocardial infarction.20 Same as it happens with the Charlson Comorbidity index, in our series of patients, renal function impairment was also an independent predictor of long-term mortality. This confirms that a more comprehensive assessment of 80-year-old survivors of a PA including an accurate assessment of comorbidities and renal function can optimize the management of this population after hospital discharge.
Risk stratification and decision-making are especially complex in 80-year-old patients with myocardial infarction because these a highly heterogeneous patients in whom chronological age may not reflect their actual biological situation. In view of our study findings we believe that in elderly patients it is important to include the measurement of the glomerular filtration rate and, above all, the assessment of comorbidities in the decision-making process at the long-term follow-up after PA. The close follow-up of these patients with several comorbidities can help diagnose potential decompensations (both cardiac and noncardiac) to prevent new hospitalizations. On the other hand, comorbidities determine a high use of drugs which favors the appearance of adverse events, interactions, and therapeutic compliance mistakes. The best thing to do would be to maximize compliance in this population, specify the benefits expected, and minimize the risks associated with the therapy used. Also, optimizing the management of noncardiac diseases can be the key to stabilize coronary artery disease. For all this, keeping a close collaboration with geriatric units after the hospital discharge of 80-year-old patients treated with PA improves their prognosis.
Limitations and strengths
Although the demographic, clinical, and angiographic data were collected prospectively, this was a retrospective analysis with the corresponding limitations of this type of studies. The size of the sample may have limited the statistical power of our study to detect the statistical significance of some associations. Also, the low number of events may have limited the reliability of the multivariate analysis regarding in-hospital mortality and mortality at the long-term follow-up since it included 5 variables in each of these 2 analyses. Since this was a single-center study, results may not be generalizable to other settings.
One of the strengths of the study is that results are based on a thorough and consecutive registry of patients from our setting who were hospitalized after a PA. Also, that a great deal of clinical, analytical, and angiographic information was obtained during their hospital stay and several evolutionary variables were registered at the very long follow-up.
CONCLUSIONS
Patients over 80 treated with PA have a high in-hospital mortality rate (18% in our series). The only independent predictor of in-hospital mortality was Killip Class. Over the next 2 years, mortality is still very high (23%), but is basically associated with noncardiac problems. The independent predictors of overall mortality at the long-term follow-up were chronic kidney disease and a Charlson Comorbidity index > 2
FUNDING
None.
AUTHORS’ CONTRIBUTIONS
L. Hernando Marrupe and J. Botas Rodríguez had the study idea. L. Hernando Marrupe, J. Botas Rodríguez, C. Marco Quirós, and R. Gayoso Gayo designed the study. L. Hernando Marrupe, C. Marco Quirós, R. Gayoso Gayo, V. Espejo Bares, V. Artiaga de la Barrera, C. Jiménez Martínez, R. Del Castillo Medina, and A. Núñez García collaborated in the study data mining. L. Hernando Marrupe, and E. Pérez Fernández conducted the statistical analysis. L. Hernando Marrupe, C. Marco Quirós, and R. Gayoso Gayo interpreted the results and wrote the manuscript first draft. L. Hernando wrote the manuscript final version, and J. Botas conducted the manuscript critical review.
CONFLICTS OF INTEREST
None reported.
WHAT IS KNOWN ABOUT THE TOPIC?
- Primary angioplasty has been considered the best reperfusion strategy in patients with ST-segment elevation myocardial infarction for years. However, elderly patients have special characteristics that pose an added risk. Also, they are misrepresented in the clinical trials. Risk stratification and the decision-making process are especially complex in 80-year-old patients with myocardial infarction because this is a highly heterogeneous population.
WHAT DOES THE STUDY ADD?
- In 80-year-old patients treated with PA it seems that the main prognostic factor of in-hospital mortality is the patient’s hemodynamic situation at admission. However, if the patient survives the index event his prognosis is more associated with the presence of comorbidities. Our study proved that measuring the glomerular filtration rate and Charlson Comorbidity index can help us treat these patients more effectively at the long-term follow-up.
REFERENCES
1. Christensen K, Doblhammer G, Rau R, et al. Ageing populations:the challenges ahead. Lancet. 2009;374:1196-1208.
2. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Rev Esp Cardiol. 2017;70:1082.
3. Bueno H, Betriu A, Heras M, et al. Primary angioplasty vs. fibrinolysis in very old patients with acute myocardial infarction:TRIANA (TRatamiento del Infarto Agudo de miocardio eN Ancianos) randomized trial and pooled analysis with previous studies. Eur Heart J. 2011;32:51-60.
4. Doizon T, Orion L, Dimet J, et al. ST elevation myocardial infarction (STEMI) in patients aged 85 and over. Invasive management versus exclusive medical treatment:Departmental study. Ann Cardiol Angeiol (Paris). 2015;64:345-351.
5. Sim WL, Mutha V, Ul-Haq MA, et al. Clinical characteristics and outcomes of octogenarians presenting with ST elevation myocardial infarction in the Australian population. World J Cardiol. 2017;9:437-441.
6. Lee PY, Alexander KP, Hammill BG, et al. Representation of elderly persons and women in published randomized trials of acute coronary syndromes. JAMA. 2001;286:708-713.
7. Charlson M, Szatrowski TP, Peterson J, et al. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245-1251.
8. Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies:development and validation. J Chronic Dis. 1987;40:373-383.
9. Levey AS, Bosch JP, Lewis JB, et al. A more accurate method to estimate glomerular filtration rate from serum creatinine:a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130:461-470.
10. Hicks KA, Stockbridge NL, Targum SL, et al. Bleeding Academic Research Consortium consensus report:the Food and Drug Administration perspective. Circulation. 2011;123:2664-2665.
11. Ladwig KH, Fang X, Wolf K, et al. Comparison of delay times between symptom onset of an acute ST-elevation myocardial infarction and hospital arrival in men and women <65 years versus >65 years of age:findings from Multicenter Munich Examination of Delay in Patients Experiencing Acute Myocardial Infarction (MEDEA) study. Am J Cardiol. 2017;120:2128-2134.
12. Kvakkestad KM, Abdelnoor M, Claussen PA, et al. Long-term survival in octogenarians and older patients with ST-elevation myocardial infarction in the era of primary angioplasty:A prospective cohort study. Eur Heart J Acute Cardiovasc Care. 2016;5:243-252.
13. De la Torre Hernández JM, Brugaletta S, Gómez Hospital JA, et al. Primary Angioplasty in Patients Older Than 75 Years. Profile of Patients and Procedures, Outcomes, and Predictors of Prognosis in the ESTROFA IM+75 Registry. Rev Esp Cardiol. 2017;70:81-87.
14. Rodríguez-Leor O, Fernández-Nofrerias E, Carrillo X, et al. Results of primary percutaneous coronary intervention in patients ≥75 years treated by the transradial approach. Am J Cardiol. 2014;113:452-456.
15. DeGeare VS, Grines CL. Primary percutaneous intervention in octogenarians with acute myocardial infarction:the treatment of choice. Am J Med. 2000;108:257-258.
16. Forman DE, Chen AY, Wiviott SD, et al. Comparison of outcomes in patients aged <75, 75 to 84, and ≥85 years with ST-elevation myocardial infarction (from the ACTION Registry-GWTG). Am J Cardiol. 2010;106:1382-1388.
17. Velders MA, James SK, Libungan B, et al. Prognosis of elderly patients with ST-elevation myocardial infarction treated with primary percutaneous coronary intervention in 2001 to 2011:A report from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR) registry. Am Heart J. 2014;167:666-673.
18. DeGeare VS, Boura JA, Grines LL, et al. Predictive value of the Killip classification in patients undergoing primary percutaneous coronary intervention for acute myocardial infarction. Am J Cardiol. 2001;87:1035-1038.
19. Nunez JE, Nunez E, Facila L, et al. Prognostic value of Charlson comorbidity index at 30 days and 1 year after acute myocardial infarction. Rev Esp Cardiol. 2004;57:842-849.
20. Szummer K, Lundman P, Jacobson SH, et al. Relation between renal function, presentation, use of therapies and in-hospital complications in acute coronary syndrome:data from the SWEDEHEART register. J Intern Med. 2010;268:40-49.

ABSTRACT
Introduction and objectives: The objective of this study was to describe our experience with coronary physiology assessment using the instantaneous wave-free ratio (iFR) and/or a Syncvision-guided iFR-pullback study [Syncvision version 4.1.0.5, Philips Volcano, Belgium] in all-comer patients.
Methods: Consecutive patients undergoing coronary physiology assessment with the iFR (and/or a Syncvision-guided iFR-pullback study) at our center between January 2017 and December 2019 were included. The iFR cut-off value was 0.89. The primary endpoint was a composite of cardiac death, myocardial infarction, probable or definitive stent thrombosis, and target lesion revascularization.
Results: A total of 277 patients with 433 lesions evaluated were included. The mean age was 65 ± 10 years and 74% were men. Personal history of diabetes mellitus was present in 41% of patients. Clinical presentation was stable angina in 160 patients (58%), and acute coronary syndrome in 117 patients (42%). iFRs > 0.89 were obtained in 266 lesions (61.4%) on which the PCI was postponed. The remaining lesions were revascularized. The Syncvision software was used to guide the iFR-pullback study in 155 lesions (36%) and the decision-making process, mainly in long, diffuse or sequential lesions (91 lesions, 58.7%), and intermediate lesions (52 lesions, 33.5%). After a median follow-up of 18 months, the primary endpoint occurred in 17 patients (6.1%) without differences regarding the baseline iFR (≤ 0.89 or > 0.89) (4.2% vs 3.8%; P = .9) or the clinical presentation (stable angina or acute coronary syndrome) (4.4% vs 8.5%; P = .1)
Conclusions: The use of coronary physiology assessment with the iFR and the Syncvision-guided iFR-pullback study in the routine daily practice and in all-comer patients seems safe with a low percentage of major adverse cardiovascular events at the mid-term follow-up.
Keywords: Physiological assessment. All-comer patients. Syncvision-guided iFR-pullback study.
RESUMEN
Introducción y objetivos: El propósito del estudio fue describir nuestra experiencia con el uso del índice diastólico instantáneo sin ondas (iFR) para la evaluación fisiológica coronaria o el uso del software Syncvision/iFR (Syncvision versión 4.1.0.5, Philips Volcano, Bélgica) en todo tipo de pacientes.
Métodos: Se incluyeron todos los pacientes consecutivos a quienes, entre enero de 2017 y diciembre de 2019, se realizó en nuestro centro una evaluación fisiológica coronaria con iFR o con Syncvision/iFR. El valor de corte establecido para el iFR fue 0,89. El objetivo primario fue un compuesto de muerte cardiaca, infarto de miocardio, trombosis de stent probable o definitiva y nueva revascularización de la lesión evaluada.
Resultados: Se incluyeron 277 pacientes con 433 lesiones evaluadas. La edad media fue de 65 ± 10 años y el 74% eran varones. El 41% tenía antecedente de diabetes mellitus. La presentación clínica fue angina estable en 160 pacientes (58%) y síndrome coronario agudo en 117 pacientes (42%). Se obtuvo un iFR > 0,89 en 266 lesiones (61,4%), en las cuales la intervención coronaria percutánea fue diferida. Las lesiones restantes se revascularizaron. El software Syncvision/iFR se usó en 155 lesiones (36%) para guiar la toma de decisiones, principalmente lesiones largas, difusas o secuenciales (91 lesiones, 58,7%) y lesiones intermedias (52 lesiones, 33,5%). Tras un periodo de seguimiento de 18 meses, el objetivo primario se observó en 17 pacientes (6,1%), sin diferencias en función del iFR basal (≤ 0,89 o > 0,89) (4,2 frente a 3,8%; p = 0,9) ni de la presentación clínica (angina estable o síndrome coronario agudo) (4,4 frente a 8,5%; p = 0,1).
Conclusiones: La evaluación fisiológica coronaria con iFR y el software Syncvision/iFR en la práctica diaria y en todo tipo de pacientes parece ser segura, con un bajo porcentaje de eventos cardiacos adversos mayores a medio plazo.
Palabras clave: Evaluacion fisiologica. Todo tipo de pacientes. Software Syncvision/iFR.
Abbreviations
iFR: instantaneous wave-free ratio. PCI: percutaneous coronary intervention. MACE: major adverse cardiovascular events.
INTRODUCTION
Physiological assessment using the fractional flow reserve (FFR) or the instantaneous wave-free ratio (iFR) is strongly recommended by the European guidelines to the guide percutaneous coronary intervention (PCI) decision-making process to treat intermediate coronary stenosis (indication I, level of evidence A) and multivessel disease (indication IIa, level of evidence B).1-7
The established cut-off values based on landmark trials to safely postpone treatment of a coronary lesion are FFRs > 0.80 and iFRs > 0.89.2-7 Unlike the FFR, the new iFR resting index allows us to analyze the physiological significance of each segment in the presence of coronary arteries with several lesions. Syncvision is a new software that analyzes the specific contribution of each coronary segment allowing us to predict physiological improvement after percutaneous treatment.8,9 It’s not necessary to use any vasodilators either, thus reducing any potential side effects.3,4
However, the evidence supporting the use of coronary physiology assessment with both indices and the use of the Syncvision software in other type of lesions and other clinical scenarios is scarce.8-10 For this reason, it is not quite clear whether the same cut-off value established in the landmark trials should be used; or if safety, utility, and efficacy will be the same.
The objective of this study is to describe our experience with coronary physiology assessment using the iFR (and/or the Syncvision- guided iFR-pullback study) in all-comer patients undergoing invasive coronary angiography.
METHODS
We performed a single-center retrospective study including all patients who underwent functional assessments (using the iFR) and/or the Syncvision software at our center between January 2017 and December 2019 on a PCI decision-making process. The cut-off value to consider the need for revascularization was the same one established by the landmark clinical trials (iFR ≤ 0.89).3,4 The pressure guidewires used for the functional assessment were the Volcano Verrata, and the Volcano Verrata Plus (Philips Volcano, Belgium). The use of the Syncvision software to guide the iFR study as well as the lesions assessed were left to the operator’s discretion.
All subjects included in the study gave their informed consent to undergo the procedure and for data analysis and publication. Additionally, the study received the proper ethical oversight and was approved by our center ethics committee.
Inclusion and exclusion criteria
Patients with the following criteria were included: a) consecutive patients in whom an invasive coronary angiography was performed due to stable or unstable symptoms or silent ischemia; b) presence of, at least, a lesion or vessel physiologically assessed with the iFR during the index procedure. The following exclusion criteria were stablished: a) impossibility to understand the informed consent during the index procedure; b) written informed consent to use data for research purposes not provided.
Lesion classification
The lesions physiologically assessed were classified based on their angiographic characteristics and/or clinical setting: a) intermediate lesions: lesions with a 40% to 80% angiographic stenosis as seen on the quantitative coronary angiography (QCA); b) sequential or diffuse coronary lesions: presence of, at least, 2 sequential lesions or a coronary segment with diffuse disease (coronary vessel with multiple plaques in most of the epicardial territory) with a total length of 25 mm; c) bifurcation lesions: presence of a coronary stenosis at bifurcation level with a side branch size large enough to be protected; d) in-stent restenosis: presence of focal or diffuse in-stent restenosis with a a 40% to 80% angiographic stenosis as seen on the QCA; e) coronary bypass lesion, defined as, at least, a lesion in the coronary artery bypass grafting or native vessel presenting with proximal total occlusion.
Endpoints
The primary endpoint of the study was the rate of major adverse cardiovascular events (MACE) at the follow-up. The MACE were defined as a composite of cardiac death, myocardial infarction (MI), definitive or probable stent thrombosis, and new target lesion revascularization (TLR). All deaths were considered cardiovascular unless unequivocal non-cardiac causes would be established. Myocardial infarction included spontaneous ST-segment elevation MI or non-ST-segment elevation acute myocardial infarction. The TLR was defined as a new revascularization of a baseline physiologically negative lesion at the follow-up or as a repeat revascularization of a baseline physiologically positive lesion percutaneously treated during the index procedure.
The secondary endpoints established were: a) analysis of the primary endpoint components separately; b) rate of MACE based on the clinical setting (stable angina or acute coronary syndrome), non-ST-segment elevation acute myocardial infarction (NSTEMI), and ST segment elevation myocardial infarction (STEMI); c) rate of MACE based on the baseline iFR; d) to determine the type of lesions where the Syncvision software was used for the iFR-pullback study.
Follow-up
The patients’ follow-up was performed through phone calls, hospital record reviews or outpatient visits.
Quantitative coronary measurements
Quantitative coronary measurements were performed using a validated system (CAAS system, Pied Medica Imaging, The Netherlands). These were the measurements analyzed: reference vessel diameter, minimum lumen diameter, percent diameter stenosis, and lesion length. All measurements were performed at baseline and after the PCI.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation, and the Student t test was used to establish comparisons. The categorical variables were expressed as frequency and percentage, and compared using the chi-square test. The univariate analysis was performed with the following covariates: age, male sex, current smoking status, dyslipidemia, left ventricular ejection fraction, acute coronary syndrome, multivessel disease, clopidogrel, ticagrelor, right coronary artery as the study vessel, other vessels analyzed, and baseline iFRs ≤ 0.89. Results were reported using odds ratios (OR), and two-sided 95% confidence intervals. In all the cases, P values < .05 were considered statistically significant. The statistical analysis was performed using the IBM-SPSS statistical software package (version 24.0 for Macintosh, SPSS Corp., United States).
RESULTS
The study flowchart is shown on figure 1. During the study period, a total of 2951 patients underwent coronary angiography at our center. The iFR-based physiological assessment was performed in 277 patients (9.4%) with 433 lesions. The baseline clinical data are shown on table 1. The mean age was 65 ± 10 years, and 74% of the patients (204) were men. The prevalence of comorbidities was high (diabetes mellitus, 41%; previous MI, 32%; peripheral arterial disease, 4%; cerebrovascular disease, 6%; chronic kidney disease, 13%). The clinical presentation included stable angina in 160 patients (58%), NSTEMI in 91 patients (33%), and STEMI in 26 patients (9%).
Table 1. Baseline clinical data
| Patients | Total (N = 277) | Stable angina (N = 160) | ACS (N = 117) | P |
|---|---|---|---|---|
| Age, years | 65 ± 10 | 65 ± 10 | 64 ± 11 | .071 |
| Sex, male, N (%) | 204 (74) | 116 (72) | 94 (80) | .112 |
| Hypertension, N (%) | 175 (63) | 101 (63) | 77 (66) | .645 |
| Diabetes mellitus, N (%) | 114 (41) | 58 (36) | 52 (44) | .169 |
| Dyslipidemia, N (%) | 157 (57) | 101 (63) | 58 (50) | .024 |
| Current smoker, N (%) | 72 (26) | 29 (18) | 42 (36) | .001 |
| Previous myocardial infarction, N (%) | 89 (32) | 53 (33) | 37 (32) | .792 |
| Previous revascularization, N (%) | 94 (34) | 50 (31) | 32 (27) | .518 |
| Percutaneous, N (%) | 80 (85) | 50 (31) | 30 (26) | .336 |
| Surgical, N (%) | 14 (15) | 8 (16) | 6 (19) | .095 |
| Atrial fibrillation, N (%) | 39 (14) | 19 (12) | 13 (11) | .844 |
| Heart failure, N (%) | 8 (3) | 7 (4) | 2 (2) | .216 |
| Prior ACE, N (%) | 17 (6) | 11 (7) | 9 (8) | .795 |
| Peripheral arterial disease, N (%) | 11 (4) | 7 (4) | 5 (4) | .967 |
| Previous bleeding, N (%) | 3 (1) | 2 (1) | 2 (2) | .752 |
| Chronic kidney disease, N (%) | 36 (13) | 19 (12) | 17 (15) | .486 |
| Hemoglobin, g/dL | 13.96 ± 1.7 | 13.87 ± 1.8 | 14.13 ± 1.8 | .365 |
| Creatinine, g/dL | 0.98 ± 0.47 | 1 ± 0.63 | 1 ± 0.37 | .584 |
| Left fentricular ejection fraction, % | 59 ± 15 | 57 ± 16 | 60 ± 13 | .098 |
|
ACE, acute cerebrovascular event; ACS, acute coronary syndrome. Data are expressed as number (N) and percentage (%). |
||||
Angiographic and procedural data
Angiographic and procedural data are shown on table 2. Radial access was the access of choice in most of the cases (392 lesions, 91%). A total of 186 patients (67%) showed angiographic multivessel disease. Regarding the angiographic Syntax I score, 232 patients (84%) had Syntax scores < 22, 41 patients (15%) between 22 and 32, and only 4 patients (1%) > 32 without any differences being reported between stable and unstable patients. The vessel most frequently analyzed was the left anterior descending coronary artery (180, 42%) followed by the right coronary artery (99, 23%). The left main coronary artery was evaluated in 23 patients (5%).
Table 2. Angiographic and procedural data
| Patients | Total (N = 277) | Stable angina (N = 160) | ACS (N = 117) | P |
|---|---|---|---|---|
| Radial access, N (%) | 251 (90) | 147 (92) | 104 (89) | .329 |
| Multivessel disease, N (%) | 165 (59) | 84 (52) | 81 (69) | .004 |
| Syntax score | 11 ± 8 | 10 ± 8 | 12 ± 8 | .885 |
| Low risk (< 22) | 45 (16) | 25 (16) | 20 (17) | .184 |
| Intermediate risk (22-32) | 6 (2) | 1 (1) | 5 (4) | .066 |
| High risk (> 32) | 1 (1) | 1 (1) | 0 | .331 |
| Acetylsalicylic acid, N (%) | 245 (88) | 142 (88) | 103 (88) | .740 |
| P2Y12 inhibitor, N (%) | 195 (71) | 98 (61) | 97 (83) | |
| Clopidogrel | 63 (23) | 40 (25) | 23 (20) | .011 |
| Ticagrelor | 127 (46) | 56 (35) | 71 (61) | .019 |
| Prasugrel | 65 (2) | 2 (1) | 3 (3) | .642 |
| Vessel analyzed, N (%) | ||||
| LAD | 121 (44) | 66 (41) | 55 (47) | .318 |
| LCx | 40 (14) | 26 (16) | 14 (12) | .327 |
| RCA | 75 (27) | 50 (31) | 25 (21) | .072 |
| LMCA | 15 (5) | 8 (5) | 7 (6) | .712 |
| Other | 27 (10) | 11 (7) | 16 (14) | .057 |
| Reference vessel diameter (mm) | 3.3 ± 3 | 3.3 ± 3 | 3.3 ± 3 | .971 |
| Vessel stenosis (%) | 49 ± 16 | 49 ± 17 | 49 ± 16 | .816 |
| Vessel minimal lumen diameter (mm) | 1.6 ± 0.6 | 1.5 ± 0.6 | 1.5 ± 0.5 | .203 |
| Vessel lesion length (mm) | 21 ± 12 | 21 ± 13 | 20 ± 11 | .174 |
| Vessel stent diameter (mm) | 2.8 ± 0.4 | 2.8 ± 0.4 | 2.8 ± 0.4 | .581 |
| Type of stent implanted (%) | ||||
| DES | 100 | |||
| BMS | 0 | |||
| Other | 0 | |||
| Immediate angiographic optimal result (%) | 100 | |||
| Contrast used (mL) | 142 ± 91 | 151 ± 110 | 164 ± 72 | .166 |
| Intracoronary imaging, N (%) | 6 (2) | 6 (4) | 0 | .034 |
| Procedural complications, N (%) | 3 (1) | 2 (1) | 1 (1) | .754 |
| Baseline iFR | 0.88 ± 0.12 | 0.89 ± 0.12 | 0.86 ± 0.14 | .097 |
| Final iFR | 0.93 ± 0.04 | 0.93 ± 0.04 | 0.93 ± 0.04 | .951 |
| Syncvision-guided iFR-pullback study, N (%) | 155 lesions (36) | 94 lesions (36) | 61 lesions (35) | .4 |
| Lesions evaluated | Total (N = 433) | Stable angina (N = 258) | ACS (N = 175) | P |
| Angiographically moderate lesions, N (%) | 244 (56.4) | 149 (58) | 95 (54) | .475 |
| Sequential/diffuse coronary lesions, N (%) | 118 (27.3) | 64 (25) | 53 (30) | .208 |
| Bifurcation lesions, N (%) | 51 (11.8) | 31 (12) | 20 (11) | .853 |
| In-stent restenosis, N (%) | 15 (3.5) | 11 (4.3) | 4 (2.3) | .269 |
| Coronary artery bypass grafting, N (%) | 2 (0.5) | 0 (0) | 2 (1.1) | .085 |
| Other lesions, N (%) | 3 (0.75) | 2 (0.8) | 1 (0.6) | .802 |
|
ACS, acute coronary syndrome; BMS, bare metal stent; DES, drug-eluting stent; iFR, instantaneous wave-free ratio; LAD, left anterior descending coronary artery; LCx, left circumflex artery; LMCA, left main coronary artery; RCA, right coronary artery. Data are expressed as number (N) and percentage (%). |
||||
The mean reference diameter was 3.3 mm ± 3 mm with a mean vessel stenosis of 49% ± 16%, and a mean lesion length of 21 mm ± 12 mm. The mean diameter of the stent implanted was 2.8 ± 0.4. All the stents implanted were drug-eluting stents (100%). Intracoronary imaging was used in 14 patients (3%).
The instantaneous wave-free ratio was obtained in 433 lesions, with a baseline value of 0.89 ± 0.12. The physiological assessment results after the PCI were obtained in 129 lesions (29.8%) with a final iFR of 0.93 ± 0.04.
The lesions physiologically assessed are shown on table 2. The most common type of lesions undergoing physiological assessment were angiographically moderate lesions (244, 56.4%) followed by sequential and diffuse lesions (118, 27.3%). Physiological assessment was used in 51 bifurcation lesions (11.8%) basically to guide the intervention over the side branch while using a provisional stenting strategy.
The Syncvision software for the iFR-pullback study was used in 155 lesions to guide the decision-making process (35.8%). Sequential and diffuse coronary lesions were the most common lesions analyzed by the iFR-pullback study (91 vessels, 58.7%, figure 2) followed by angiographically moderate lesions (52 vessels, 33.5%). This software was used in 5 bifurcation lesions (3.2%) to establish a baseline physiological classification or confirm an optimal physiological result after the PCI in both branches. The remaining lesions assessed by the iFR-pullback study were 6 focal or diffuse in-stent restenoses (3.9%) and 1 saphenous vein bypass graft with diffuse disease (0.6%).
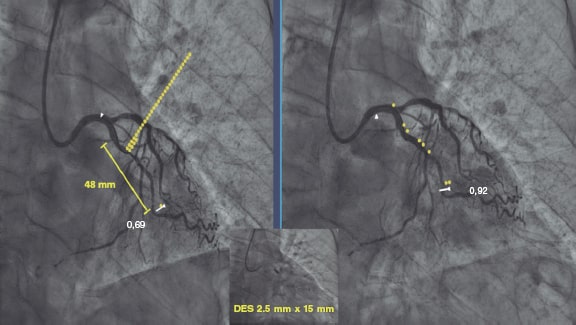
Figure 2. Images of iFR-coregistration with the Syncvision software from a left circumflex artery with diffuse disease in its middle segment (48 mm of lesion length). The baseline distal iFR was 0.69. The Syncvision-guided iFR-pullback study demonstrated physiological significance only in the proximal segment. Direct implantation of a 2.5 mm × 15 mm DES was performed with a final iFR of 0.92. The stent length reduction regarding the angiographic lesion was 33 mm.
Follow-up
Follow-up data were available for 274 out of 277 patients (99%). After a mean 18 ± 10-month follow-up, 17 patients (6.1 %) presented with a major adverse cardiovascular events (table 3), 7 patients (2.5 %) with TLR, 2 of them over a lesion treated during the index procedure (0.7%) and 5 (1.8%) due to disease progression of a baseline physiologically negative lesion; 6 patients (2.2 %) suffered from acute myocardial infarction (1 patient due to acute stent thrombosis, another to a new lesion not evaluated at the index procedure, another to a baseline physiologically non-significant lesion, and the remaining 3 patients due to failed previously revascularized lesions); also, 4 patients (1.4%) presented with unclear or cardiac death. There were no differences regarding MACE between baseline physiologically negative and positive lesions (table 3).
Table 3. Rate of major adverse cardiovascular events at the follow-up based on the clinical presentation
| MACE (277 patients, 433 lesions) | iFR ≤ 0.89 (N = 167 lesions) | iFR > 0.89 (N = 266 lesions) | P | Stable angina (N = 160) | ACS (N = 117) | P | |
|---|---|---|---|---|---|---|---|
| Overall, N (%) | 17 (6.1) | 7 (4.2) | 10 (3.8) | .9 | 7 (4.4) | 10 (8.5) | .1 |
| Unclear or cardiac death, N (%) | 4 (1.4) | 2 (1.2) | 2 (0.8) | .2 | 3 (1.9) | 1 (0.8) | .9 |
| Myocardial infarction, N (%) | 6 (2.2) | 1 (0.6) | 5 (1.9) | .46 | 1 (0.6) | 5 (4.3) | < .05 |
| Target lesion revascularization, N (%) | 7 (2.5) | 4 (2.4) | 3 (1.1) | .09 | 3 (1.9) | 4 (3.4) | .2 |
|
ACS, acute coronary syndrome; iFR, instantaneous wave-free ratio; MACE, major adverse cardiovascular events. Data are expressed as number (N) and percentage (%). |
|||||||
Based on their clinical signs, patients who presented with ACS had an increased rate of new myocardial infarction at the follow-up (5.3% vs 0.6%; P < .05), although no differences were found regarding unclear or cardiac death (0.9% vs 1.8%; P = .9) and the overall MACE (8.5% vs 4.4%; OR, 2.056, 0.759-5.572; P = .156 (table 3).
Finally, we performed a univariate analysis and found no risk or protective factors for MACE in this cohort of patients (table 4).
Table 4. Univariate analysis of the different variables with potential impact in the rate of major adverse cardiovascular events between groups
| Variable | Univariate analysis | |
|---|---|---|
| OR (95%CI) | P | |
| Age | 1.01 (0.97-1.06) | .608 |
| Male | 2.54 (0.57-11.40) | .224 |
| Current smoker | 1.23 (0.42-3.60) | .713 |
| Dyslipidemia | 1.39 (0.50-3.87) | .531 |
| Left ventricular ejection fraction (%) | 0.99 (0.95-1.04) | .684 |
| Acute coronary syndrome | 2.06 (0.76-5.57) | .156 |
| Multivessel disease | 0.90 (0.33-2.45) | .842 |
| Clopidogrel | 0.75 (0.23-2.44) | .623 |
| Ticagrelor | 1.52 (0.46-4.96) | .490 |
| Right coronary artery as examined vessel | 1.52 (0.54-4.26) | .428 |
| Other vessel analyzed | 1.26 (0.27-5.82) | .769 |
| Baseline iFR ≤ 0.89 | 1.43 (0.88-2.32) | .152 |
|
95%CI, confidence interval; iFR, instantaneous wave-free ratio; OR, odds ratio. |
||
DISCUSSION
This study tried to describe our experience using the physiological assessment and the Syncvision software in all-comer patients who underwent percutaneous coronary evaluations. The main findings of our study are: a) the use of the iFR in lesions of all-comer patients with the same cut-off values than established in the main trials showed a low percentage of MACE at the mid-term follow-up (6.1%); b) patients who presented with acute coronary syndrome showed an increased rate of myocardial infarction at the mid-term follow-up, and a trend towards a higher rate of MACE (OR, 2.056, 0.759-5.572; P = .156); c) The Syncvision-guided iFR-pullback study provided additional information to guide the PCI decision-making process, especially in complex lesions like sequential lesions and diffuse coronary artery disease.
The fractional flow reserve was the first physiological index that demonstrated its utility, safety, and efficacy guiding the revascularization decision-making process.2,5-7 To obtain it, the use of a hyperemic agent to reduce vascular resistance is mandatory. Adenosine is the most commonly used drug, but it presents a series of side effects and contraindications.3,4,11,12 The more recent resting index (the instantaneous wave-free ratio) has demonstrated similar utility, safety, and efficacy to the FFR.3,4 Furthermore, it has 2 main advantages: first, it is not necessary to use vasodilators, thus reducing side effects, contraindications for use, and procedural time; secondly, it allows us to assess the contribution of each lesion when the vessel presents several lesions, with the specific Syncvision-guided iFR-pullback study.8,9
For these reasons, the coronary physiology assessment is already the routine practice at the cath lab for the assessment of intermediate lesions,2-5 and multivessel disease.6,7 The main clinical setting included in these studies was stable angina. Patients with NSTEMI could be included if the lesion evaluated was identified as a non-culprit lesion. However, patients with STEMI, left main coronary artery lesions, and coronary artery bypass grafting lesions were not represented in the trials; also, the percentage of bifurcation lesions and sequential or diffuse coronary lesions is tiny. The cut-off value for the FFR and the iFR is well defined in those trials, being safe to postpone a lesion with a FFR > 0.80 or an iFR > 0.89. However, information is scarce on the utility and efficacy of physiological assessment and the same cut-off values in other types of lesions and clinical presentations.13 A multicenter registry that used the iFR to guide revascularization in patients with left main coronary artery stenosis has just been published. Using a cut-off value of 0.89, the authors conclude that postponing a left main coronary artery lesion with a iFR > 0.89 seems to be safe.10
Our study results suggest that the use of physiological assessment and the Syncvision software to guide the PCI decision-making process in all-comer patients with the same cut-off values as established by the landmark trials seems useful and safe regardless of the lesion and clinical presentation undergoing evaluation. Also, the MACE rates are similar to those reported by the landmark trials with selected lesions and patients.3,4 The iFR was the index used more often. The reasons are the faster and more comfortable use,3,4 and the possibility of lesion assessment with the Syncvision software.8,9
An important point of the study was to evaluate the rate of MACE based on the clinical presentation. Although no significant differences in the overall rate of MACE were found, patients who presented with acute coronary syndrome showed a significantly higher rate of MI at the follow-up, and a trend towards a higher rate of overall MACE. We think that this absence of statistical significance could be associated with a lack of statistical power.
A type of lesion included in the study was bifurcation lesions. Physiological assessment was used mainly to guide the side branch results during a provisional stenting strategy, thus keeping the pressure wire jailed as previously described.14,15 However, another interesting use of the iFR-pullback study with the Syncvision software was to stablish the baseline physiological contribution of every segment included in the most accepted classification.16
Finally, the Syncvision-guided iFR-pullback study was used in 155 lesions (36%). The main type of lesions where this software was used were diffuse and tandem lesions. This software can predict the physiological contribution of each lesion or coronary segment, which is why we believe that it is a very useful tool to avoid treating lesions without any physiological contribution and probably without clinical benefits. That is why this software seems to reduce the total stent length implanted regarding angiographically-guided revascularization with potential benefits at long-term follow-up.17,18 A clinical trial is currently in the recruitment phase to demonstrate the efficacy of this software reducing the length of the stent implanted in this type of lesions without detriment to the adverse events.19
In our experience, the key aspects to properly perform this technique are: a) a perfect aortic pressure curve allows the accurate detection of diastole through the software; b) passing the pressure sensor as distally as possible; c) finding a projection where the artery can be seen completely and with the least foreshortening possible; d) withdrawing the pressure guidewire very slowly so that the software can perfectly recognize the length of each arterial segment; e) checking that there is not drift when the pressure guidewire reaches the coronary ostium (iFR different to 1 ± 0.02) to avoid erroneous results; f) performing the coronary angiography in the same position as the guidewire withdrawal without any modifications to the height of the table or the C-arm, and with a higher flow and volume of contrast to facilitate the software recognition of all the lesions. The main problem when using this technique is the presence of lesions with complicated wiring. The pressure wire has a hydrophilic non-polymeric coating that is useful in most lesions. However, it may be very challenging to reach the distal part of the artery in very complex lesions (calcified, angled lesions…), and our experience with previous normalization, wire disconnection, the microcatheter exchange technique, and reconnection is very limited, but still there is a significant level of drift.
Limitations
The study presents several limitations. It is a retrospective, single-center analysis with a low number of patients and lesions. Therefore, the results should be interpreted with caution, although it could be a hypothesis-generating study for future larger scale randomized clinical trials.
CONCLUSIONS
The use of coronary physiology assessment using the iFR and the Syncvision-guided iFR-pullback study in the routine daily practice and in all-comer patients seems safe with a low percentage of MACE at the mid-term follow-up. The Syncvision-guided iFR-pullback study provides additional information to guide the PCI decision-making process.
FUNDING
The study has not had funding.
AUTHORS’ CONTRIBUTION
F.J. Hidalgo-Lesmes prepared the main draft of the manuscript. S. Ojeda-Pineda participated in the drafting of the manuscript. C. Pericet-Rodríguez, R. González-Manzanares, A. Fernández-Ruiz, and M.G. Flores-Vergara all contributed to the analysis and interpretation of data. A. Luque-Moreno, J. Suárez de Lezo, and F. Mazuelos-Bellido participated in the conception and design of the study. M.A. Romero-Moreno, and J.M. Segura Saint-Gerons revised the manuscript critically for important intellectual content. M. Pan Álvarez-Ossorio approved the final version of the manuscript.
CONFLICTS OF INTEREST
F.J. Hidalgo-Lesmes received minor fees from Philips Volcano Europe unrelated to the manuscript; S. Ojeda-Pineda received minor fees from Terumo and Philips Volcano Europe unrelated to the manuscript; M. Pan Álvarez-Ossorio received minor fees from Terumo, Abbott Vascular, and Philips Volcano Europe unrelated to the manuscript. The remaining authors declared no conflicts of interest.
WHAT IS KNOWN ABOUT THE TOPIC?
- Physiological assessments with the iFR are strongly recommended by the European guidelines on coronary revascularization to guide the PCI decision-making process in intermediate coronary stenosis.
- However, the evidence supporting the use of coronary physiology assessment, and the new Syncvision-iFR software in other type of lesions and clinical settings is scarce.
WHAT DOES THIS STUDY ADD?
- This study describes our experience with the iFR and the Syncvision-iFR software in all-comer patients and demonstrates an acceptable percentage of MACE at the mid-term follow-up.
- Furthermore, the study shows that the Syncvision-guided iFR-pullback study provides additional information to guide the PCI decision-making process, particularly in complex lesions like sequential lesions and diffuse coronary artery disease.
REFERENCES
1. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
2. Pijls NHJ, van Schaardenburgh P, Manoharan G, et al. Percutaneous Coronary Intervention of Functionally Nonsignificant Stenosis. 5-Year Follow-Up of the DEFER Study. J Am Coll Cardiol. 2007;49:2105-2111.
3. Davies JE, Sen S, Dehbi H-M, et al. Use of the Instantaneous Wave-free Ratio or Fractional Flow Reserve in PCI. N Engl J Med. 2017;376:1824-1834.
4. Götberg M, Christiansen EH, Gudmundsdottir IJ, et al. Instantaneous Wave-free Ratio versus Fractional Flow Reserve to Guide PCI. N Engl J Med. 2017;376:1813-1823.
5. Pijls NHJ, de Bruyne B, Peels K, et al. Measurement of Fractional Flow Reserve to Assess the Functional Severity of Coronary-Artery Stenoses. N Engl J Med. 1996;334:1703-1708.
6. Tonino AL, Bruyne B De, Pijls NHJ, et al. Fractional Flow Reserve versus Angiography for Guiding Percutaneous Coronary Intervention Pim. N Engl J Med. 2015:687-696.
7. Van Nunen LX, Zimmermann FM, Tonino PAL, et al. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME):5-year follow-up of a randomised controlled trial. Lancet. 2015;386:1853-1860.
8. Nijjer SS, Sen S, Petraco R, Mayet J, Francis DP, Davies JER. The Instantaneous wave-Free Ratio (iFR) pullback:A novel innovation using baseline physiology to optimise coronary angioplasty in tandem lesions. Cardiovasc Revasc Med. 2015;16:167-171.
9. Nijjer SS, Sen S, Petraco R, et al. Pre-angioplasty instantaneous wave-free ratio pullback provides virtual intervention and predicts hemodynamic outcome for serial lesions and diffuse coronary artery disease. JACC Cardiovasc Interv. 2014;7:1386-1396.
10. Warisawa T, Cook CM, Rajkumar C, et al. Safety of Revascularization Deferral of Left Main Stenosis Based on Instantaneous Wave-Free Ratio Evaluation. JACC Cardiovasc Interv. 2020;13:1655-1664.
11. Gili S, Barbero U, Errigo D, et al. Intracoronary versus intravenous adenosine to assess fractional flow reserve:A systematic review and meta-analysis. J Cardiovasc Med. 2018;19:274-283.
12. Patel HR, Shah P, Bajaj S, Virk H, Bikkina M, Shamoon F. Intracoronary adenosine-induced ventricular arrhythmias during fractional flow reserve (FFR) measurement:case series and literature review. Cardiovasc Interv Ther. 2017;32:374-380.
13. Ihdayhid AR, Koh JS, Ramzy J, et al. The Role of Fractional Flow Reserve and Instantaneous Wave-Free Ratio Measurements in Patients with Acute Coronary Syndrome. Curr Cardiol Rep. 2019;21.
14. Burzotta F, Lassen JF, Banning AP, et al. Percutaneous coronary intervention in left main coronary artery disease:The 13th consensus document from the European Bifurcation Club. EuroIntervention. 2018;14:112-120.
15. Hidalgo F, Pan M, Ojeda S, et al. Feasibility and Efficacy of the Jailed Pressure Wire Technique for Coronary Bifurcation Lesions. JACC Cardiovasc Interv. 2019;12:109-111.
16. Medina A, Suárez de Lezo J, Pan M. A New Classification of Coronary Bifurcation Lesions. Rev Esp Cardiol. 2006;59:183.
17. Mauri L, O'Malley AJ, Popma JJ, et al. Comparison of thrombosis and restenosis risk from stent length of sirolimus-eluting stents versus bare metal stents. Am J Cardiol. 2005;95:1140-1145.
18. Kikuta Y, Cook CM, Sharp ASP, et al. Pre-Angioplasty Instantaneous Wave-Free Ratio Pullback Predicts Hemodynamic Outcome In Humans With Coronary Artery Disease:Primary Results of the International Multicenter iFR GRADIENT Registry. JACC Cardiovasc Interv. 2018;11:757-767.
19. Hidalgo F, Ojeda S, de Lezo JS, et al. Usefulness of a co-registration strategy with iFR in long and/or diffuse coronary lesions (iLARDI):study protocol. REC Interv Cardiol. 2021;3:190-195.
Abstract
Introduction and objectives: Patients with a low post-percutaneous coronary intervention (PCI) fractional flow reserve (FFR) are at a higher risk for future adverse cardiac events. The objective of the current study was to assess specific patient and procedural predictors of post-PCI FFR.
Methods: The FFR-SEARCH study is a prospective single-center registry of 1000 consecutive all-comer patients who underwent FFR measurements after an angiographically successful PCI with a dedicated microcatheter. Mixed effects models were used to search for independent predictors of post-PCI FFR.
Results: The mean post-PCI distal coronary pressure divided by the aortic pressure (Pd/Pa) was 0.96 ± 0.04 and the mean post-PCI FFR, 0.91 ± 0.07. After adjusting for the independent predictors of post-PCI FFR, the left anterior descending coronary artery as the measured vessel was the strongest predictor of post-PCI FFR (adjusted β = -0.063; 95%CI, -0.070 to -0.056; P < .0001) followed by the postprocedural minimum lumen diameter (adjusted β = 0.039; 95%CI, 0.015-0.065; P = .002). Additionally, male sex, in-stent restenosis, chronic total coronary occlusions, and pre- and post-dilatation were negatively associated with postprocedural FFR. Conversely, type A lesions, thrombus-containing lesions, postprocedural percent stenosis, and stent diameter were positively associated with postprocedural FFR. The R2 for the complete model was 53%.
Conclusions: Multiple independent patient and vessel related predictors of postprocedural FFR were identified, including sex, the left anterior descending coronary artery as the measured vessel, and postprocedural minimum lumen diameter.
Keywords: Percutaneous coronary intervention. Post-PCI FFR. Predictors.
RESUMEN
Introducción y objetivos: Los pacientes con una reserva fraccional de flujo (FFR) posintervención coronaria percutánea (ICP) baja tienen mayor riesgo de futuros eventos cardiacos adversos. El objetivo del presente estudio fue evaluar predictores específicos de pacientes y procedimientos de FFR tras una ICP.
Métodos: El estudio FFR-SEARCH es un registro prospectivo de un solo centro que incluyó 1.000 pacientes consecutivos que se sometieron a una evaluación de la FFR tras una ICP con éxito angiográfico utilizando un microcatéter específico. Se utilizaron modelos de efectos mixtos para buscar predictores independientes de FFR tras la ICP.
Resultados: La media de presión distal dividida entre la presión aórtica tras la ICP fue de 0,96 ± 0,04, y la media de la FFR tras la ICP fue de 0,91 ± 0,07. Tras ajustar por predictores independientes de FFR tras la ICP, la arteria descendente anterior izquierda como vaso medido fue el predictor más fuerte (β ajustado = −0,063; IC95%, −0,070 a −0,056; p < 0,0001), seguida del diámetro luminal mínimo posprocedimiento (β ajustado = 0,039; IC95%, 0,015 a 0,065; p = 0,002). Además, el sexo masculino, la reestenosis del stent, las oclusiones totales crónicas y la pre- y posdilatación se correlacionaron negativamente con la FFR posprocedimiento. Por el contrario, las lesiones de tipo A, las lesiones con trombos, el porcentaje de estenosis posprocedimiento y el diámetro del stent se correlacionaron positivamente con la FFR posprocedimiento. El R2 para el modelo completo fue del 53%.
Conclusiones: Se identificaron diversos predictores independientes relacionados con los pacientes y con los vasos para la FFR posprocedimiento, incluyendo el sexo, la arteria descendente anterior izquierda como vaso medido y el diámetro luminal mínimo posprocedimiento.
Palabras clave: Intervención coronaria percutánea. FFR post-ICP. Predictores.
Abbreviations:
FFR: fractional flow reserve. LAD: left anterior descending coronary artery. MLD: minimum luminal diameter. PCI: percutaneous coronary intervention.
INTRODUCTION
The limitations of an accurate assessment of the hemodynamic significance of coronary artery lesions through angiographic guidance alone are well-known.1 Instead, the fractional flow reserve (FFR) has proven to be a useful technique to address the coronary physiology and the hemodynamic significance of coronary segments before and after performing an intervention.2-4 Also, measuring FFR post-stenting has proven to be a strong and independent predictor of major adverse cardiovascular events at the 2-year follow-up.3-5
While FFR primarily takes into account the relative luminal narrowing and the amount of viable myocardium perfused by a specific vessel, several factors have been shown to impact the FFR values prior to performing a percutaneous coronary intervention (PCI). Therefore, longer lesion length, high syntax scores, calcifications, and tortuosity are associated with significantly lower FFR values. Conversely, the presence of microvascular dysfunction, chronic kidney disease and female gender have been associated with higher FFR values.6-11
At the present time, there is lack of data on independent predictors of post-PCI FFR. Therefore, the objective of the present study was to assess the patient and procedural characteristics associated with low post-PCI FFR in an all-comer patient population.
METHODS
The FFR-SEARCH study is a prospective single-center registry that assessed the routine distal pressure divided by the aortic pressure (Pd/Pa) and FFR values of all consecutive patients after an angiographically successful PCI. The primary endpoint was to study the impact of post-PCI FFR on the rate of major adverse cardiovascular event at the 2-year follow-up. Accordingly, no further actions were taken to improve post-PCI FFR. The study was performed in full compliance with the Declaration of Helsinki. The study protocol was approved by the local ethics committee. All patients gave their written informed consent to undergo the procedure. Also, anonymous datasets for research purposes were used in compliance with the Dutch Medical Research Act. A total of 1512 patients treated between March 2016 and May 2017 at the Erasmus Medical Center were eligible to enter our study. A total of 504 of these patients were excluded due to hemodynamic instability (156), a rather small distal outflow (129), the operator’s decision not to proceed with post-PCI hemodynamic assessment (148) or other reasons (79). A total of 1000 patients were included in the study. The microcatheter could not cross the treated lesion in 28 patients, technical issues with the catheter prevented post-PCI assessments in 11 patients, and in 2 patients the post-PCI FFR measurements had to be aborted prematurely due to adenosine intolerance. This left 959 patients whose post-PCI FFR values were measured in at least 1 angiographically successfully treated lesion.
Quantitative coronary angiography
The preprocedural lesion type was defined according to the ACC/AHA guidelines12 and divided into 4 categories: A, B1, B2, and C. Comprehensive quantitative coronary angiography analyses were performed pre- and post-stent implantation in all the treated lesions. An angiographic view with minimal foreshortening of the lesion and minimal overlapping with other vessels was selected. Similar angiographic views were used pre- and post-stent implantation. Measurements included pre- and postprocedural percent diameter stenosis, reference vessel diameter, lesion length, and minimum luminal diameter (MLD). In case of a total occlusion in patients presenting with ST-segment elevation myocardial infarction (STEMI) or chronic total coronary occlusion (CTO), the MLD was considered zero and the percent diameter stenosis, 100%. The reference vessel diameter and the lesion length were measured from the first angiographic view with restored flow. All measurements were taken using CAAS for Windows, version 2.11.2 (Pie Medical Imaging, The Netherlands).
Fractional flow reserve measurements
All FFR measurements were acquired using the Navvus RXi system (ACIST Medical Systems, United States), a dedicated FFR microcatheter with optical pressure sensor technology.13,14 Measurements were performed after an intracoronary bolus of nitrates (200 µg). The catheter was advanced while mounted over the previously used guidewire approximately 20 mm distal to the most distal border of the stent. The FFR was defined as the mean distal coronary artery pressure divided by the mean aortic pressure during maximum hyperemia achieved by the continuous IV infusion of adenosine at a rate of 140 µg/kg/min via the antecubital vein. In this study no vessels were assessed using intracoronary adenosine.
Statistical analysis
At baseline, the categorical variables were expressed as counts (percentage) and the continuous ones as mean ± standard deviation. To assess the independent predictors of post-PCI FFR, all the patient and vessel characteristics were primarily assessed through an univariate test using a mixed effects model (LME-model) with a random effect for the patients and a fixed effect for the post-PCI FFR. All variables were subsequently inserted in a multivariate LME-model using the enter method that resulted in all the significant independent predictors of post-PCI FFR values. A forest plot was developed to depict all variables with the corresponding 95% confidence intervals (95%CI). Beta (β) values show the average increase or decrease of the FFR values in the case of dichotomous variables or the increment per unit increase in the case of continuous variables. Statistical analyses were performed using the statistical software package R (version 3.5.1, packages: Hmisc, lme4 and nlme, RStudio Team, United States).
RESULTS
Demographic characteristics
The mean age was 64.6 ± 11.8 years and 72.5% were males. In 959 patients, at least, 1 lesion was measured with an overall 1165 successfully treated and measured lesions. The patient demographics and baseline characteristics are shown on table 1. Up to 70% of the patients presented with an acute coronary syndrome, and 18% had confirmed thrombus as seen on the angiography. Intravascular imaging modalities were used in 9.6% of the patients to guide the procedure. Overall, 1.4 ± 0.6 lesions were treated per patient and in 1.2 ± 0.5 lesions per patient the post-PCI FFR was successfully assessed. The average overall stent length per vessel was 29 mm ± 17 mm with an average stent diameter of 3.2 mm ± 0.5 mm.
Table 1. Baseline patient and vessel characteristics
| Variable | Total FFR-SEARCH registry |
|---|---|
| Patient characteristics | (N = 1000) |
| Age | 64.6 ± 11.8 |
| Sex, male | 725 (73) |
| Hypertension | 515 (52) |
| Hypercholesterolemia | 451 (45) |
| Diabetes | 191 (19) |
| Smoking history | 499 (50) |
| Previous stroke | 77 (8) |
| Peripheral arterial disease | 76 (8) |
| Previous myocardial infarction | 203 (20) |
| Previous PCI | 264 (26) |
| Previous CABG | 57 (6) |
| Indication for PCI | |
| Stable angina | 304 (30) |
| NSTEMI | 367 (37) |
| STEMI | 329 (33) |
| Vessel characteristics | (N = 1165) |
| Lesion type | |
| A | 125 (11) |
| B1 | 233 (20) |
| B2 | 379 (33) |
| C | 428 (37) |
| LAD | 593 (51) |
| Bifurcation | 138 (12) |
| Calcified | 402 (35) |
| In-stent restenosis | 39 (3) |
| Thrombus | 214 (18) |
| Stent thrombosis | 14 (1) |
| Ostial | 97 (8) |
| CTO | 42 (4) |
| Stenosis pre procedural | 69 ± 22 |
| Reference diameter pre procedural (mm) | 2.6 ± 0.6 |
| Length pre procedural (cm) | 21 ± 11 |
| MLD pre (mm) | 0.9 ± 0.6 |
| Predilatation | 769 (66) |
| Postdilatation | 691 (59) |
| Stenosis post procedural | 44 ± 13 |
| Reference diameter post procedural (mm) | 2.7 ± 0.5 |
| Length post procedural (cm) | 24 ± 13 |
| MLD post procedural (mm) | 2.6 ± 0.5 |
| Number of stents | 1.4 ± 0.6 |
| Stent length (cm) | 29 ± 17 |
| Stent diameter (mm) | 3.2 ± 0.5 |
| Mean post-PCI Pd/Pa | 0.96 ± 0.04 |
| Mean post-PCI FFR | 0.91 ± 0.07 |
|
CABG, coronary artery bypass graft; CTO, chronic total coronary occlusion; FFR, fractional flow reserve; LAD, left anterior descending artery; MLD, minimum luminal diameter; NSTEMI, non-ST segment elevation acute myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction; Pd/Pa, ratio of mean distal coronary artery pressure to mean aortic pressure; Values are expressed as mean ± standard deviation or no. (%). |
|
The mean post-PCI FFR was 0.91 ± 0.07 and 7.7% of vessels had a post-PCI FFR ≤ 0.80. In the LME-model and after adjusting for independent predictors of post-PCI FFR, the left anterior descending coronary artery (LAD) as the measured vessel was the strongest predictor of post-PCI FFR (adjusted β = -0.063; 95%CI, -0.070 to -0.056; P < .0001) followed by the postprocedural MLD (adjusted β = 0.039; 95%CI, 0.015-0.065]; P = .002). Additionally, male sex, in-stent restenosis, CTO, and pre- and post-dilatation were negatively correlated with postprocedural FFR. Conversely, type A lesions, thrombus-containing lesions, postprocedural percent diameter stenosis, and stent diameter were positively correlated with postprocedural FFR. The R2 for the entire model was 53%. Figure 1 shows all significant and non-significant adjusted predictors included in the LME-model. Table 2 shows all adjusted and unadjusted predictors with corresponding β values and 95%CI. The most important predictors are shown on figure 2.
Figure 1. Forest plot of independent predictors of post-PCI FFR. Adjusted beta values with 95% confidence intervals. Triangles indicate significant predictors while circles are indicative of non-significant predictors in the multivariate generalized mixed model to predict post-PCI FFR. ACS, acute coronary syndrome; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction; LAD, left anterior descending coronary artery; CTO, chronic total coronary occlusion; MLD, minimum lumen diameter.
Table 2. Predictors for post-PCI FFR
| Variable | Unadjusted | Adjusted | ||
|---|---|---|---|---|
| P | β(95%CI) | P | β(95%CI) | |
| Patient characteristics | ||||
| Male sex | .214 | -0.006 (-0.015 – 0.003) | .001 | -0.013 (-0.021 – -0.005) |
| Age (per 10 years) | .976 | 0.000 (-0.03 – 0.03) | .724 | 0.001 (-0.002 – 0.003) |
| Hypertension | .013 | -0.010 (-0.018 – -0.002) | .610 | 0.002 (-0.006 – 0.010) |
| Hypercholesterolemia | < .001 | -0.019 (-0.027 – -0.011) | .287 | -0.004 (-0.012 – 0.004) |
| Diabetes | < .001 | 0.018 (0.008 – 0.042) | .081 | -0.008 (-0.017 – 0.001) |
| Smoking history | .007 | 0.020 (0.010 – 0.019) | .054 | 0.007 (-0.0001 – 0.014) |
| Previous stroke | .831 | -0.002 (-0.017 – 0.013) | .342 | 0.006 (-0.0007 – 0.019) |
| Peripheral arterial disease | .022 | -0.017 (-0.032 – -0.003) | .460 | -0.005 (-0.018 – 0.008) |
| Previous myocardial infarction | .002 | -0.016 (-0.026 – -0.006) | .137 | -0.008 (-0.019 – 0.003) |
| Previous PCI | < .001 | -0.016 (-0.025 – -0.007) | .569 | -0.032 (-0.014 – 0.008) |
| Previous CABG | .896 | -0.001 (-0.019 – 0.017) | .166 | -0.011 (-0.014 – 0.004) |
| Indication for PCI | ||||
| Stable angina | < .001 | -0.025 (-0.034 – -0.016) | .563 | -0.002 (-0.011 – 0.005) |
| STEMI | < .001 | 0.032 (0.025 – 0.041) | .171 | 0.006 (-0.003 – 0.015) |
| Vessel characteristics | ||||
| Lesion type | ||||
| A | <.001 | 0.022 (0.009 – 0.035) | .040 | 0.012 (0.0005 – 0.023) |
| C | .045 | -0.008 (-0.016 – -0.0002) | .172 | -0.006 (-0.014 – 0.002) |
| LAD | <.001 | -0.070 ( -0.077 – -0.064) | <.001 | -0.063 (-0.070 – -0.056) |
| Bifurcation | < .001 | -0.024 (-0.036 – - 0.012) | .883 | 0.001 (-0.010 – 0.011) |
| Calcified | < .001 | -0.025 (-0.033 – -0.017) | .409 | -0.003 (-0.011 – 0.005) |
| In-stent restenosis | .006 | -0.031 (-0.053 – -0.009) | .007 | -0.029 (-0.051 – -0.008) |
| Thrombus | < .001 | 0.031 (0.021 – 0.042) | .026 | 0.012 (-0.001 – 0.023) |
| Stent thrombosis | .920 | 0.002 (-0.034 – 0.038) | .362 | 0.019 (-0.022 – 0.060) |
| Ostial | .181 | -0.010 (-0.024 – 0.005) | .165 | -0.010 (-0.024 – 0.004) |
| CTO | .002 | -0.034 (-0.056 – -0.013) | .036 | -0.027 (-0.053 – -0.002) |
| Stenosis pre procedural (per 10%) | <.001 | 0.007 (0.005 – 0.009) | .105 | 0.004 (-0.0009 – 0.009) |
| Reference diameter pre procedural (mm) | <.001 | 0.030 (0.023 – 0.037) | .704 | 0.002 (-0.008 – 0.011) |
| Length pre procedural (cm) | .900 | -0.00002 (-0.004 – 0.003) | .101 | 0.004 (0.0008 – 0.009) |
| MLD pre procedural (mm) | <.001 | -0.015 (-0.022 – -0.008) | .638 | 0.004 (-0.014 – 0.023) |
| Predilatation | <.001 | -0.019 (-.027 – -0.011) | .002 | -0.012 (-0.020 – -0.005) |
| Postdilatation | <.001 | 0.027 (-0.035 – -0.019) | .015 | -0.009 (-0.016 – -0.002) |
| Stenosis post procedural (per 10%) | .077 | 0.003 (-0.0003 – 0.006) | .029 | 0.01 (0.0007 – 0.01) |
| Reference diameter post procedural (mm) | <.001 | 0.035 (0.027 – 0.042) | .067 | -0.022 (-0.045 – 0.002) |
| Length post procedural (cm) | .312 | -0.002 (-0.005 – 0.001) | .086 | 0.001 (-0.0007 – 0.001) |
| MLD post procedural (mm) | <.001 | 0.032 (0.024 – 0.040) | .002 | 0.039 (0.015 – 0.063) |
| Number of stents | <.001 | -0.012 (-0.018 – -0.006) | .620 | -0.002 (-0.012 – 0.007) |
| Stent length (cm) | <.001 | 0.019 (0.009 – 0.041) | .286 | -0.003 (-0.009 – 0.002) |
| Stent diameter (mm) | <.001 | 0.033 (0.025 – 0.042) | .026 | 0.012 (0.001 – 0.022) |
|
Beta (β) values are indicative of the average increase or decrease of the FFR values in cases of dichotomous variables or the increment per unit increase in cases of continuous variables. 95%CI, 95% confidence interval; CABG, coronary artery bypass graft; CTO, chronic total coronary occlusion; FFR, fractional flow reserve; LAD, left anterior descending coronary artery; MLD, minimum lumen diameter; STEMI, ST-segment elevation myocardial infarction. |
||||
Figure 2. Forest plot of most important predictors of post-PCI FFR. Adjusted beta values with 95% confidence intervals. The figure includes all significant predictors from the multivariate generalized mixed model predicting post-PCI FFR except for categorical variables with beta values < 0.02. LAD, left anterior descending coronary artery; CTO, chronic total coronary occlusion; MLD, minimum lumen diameter.
DISCUSSION
This study is the largest report to this day of predictors of post-PCI FFR. Based on data derived from the FFR-SEARCH registry, we could identify several patient and procedural predictors of post-PCI FFR. These predictors will bring more in-depth interpretations of post-PCI FFR values to be able to identify correctly which vessels are prone to future events. At first, male gender appeared to be negatively correlated with postprocedural FFR. This finding is consistent with the findings of former studies that focused on the impact of gender on pre-PCI FFR measurements.6,11,15,16 Compared to females, males are known to have a lower prevalence of microvascular dysfunction.8,17 The concept of FFR is based on drug-induced maximal hyperemia to minimize microvascular resistance. Microvascular dysfunction may hamper this vasodilator response and consequently result in a dampened flow response and high FFR.15 Subsequently, on average, males have larger myocardial masses and myocardial perfusion territories compared to females.18,19 The importance of the latter is illustrated by the second and strongest predictor of post-PCI FFR in this study, the FFR measurements in the LAD. FFR values are associated with the myocardial mass and the outflow territory of the measured vessel. As such, the LAD—the vessel with the largest perfusion area—has previously been associated with lower pre- and postprocedural FFR values.20-22
The diameters of the stents implanted in the RCA are larger, on average, but the outflow territory of the LAD is even larger.23 This discrepancy between luminal dimensions and myocardial mass may explain why the optimal improvement of the FFR measurements in the LAD is difficult to achieve.23
Thirdly, larger stent diameters and larger post-PCI MLDs were associated with higher post-PCI FFR values. However, higher postprocedural percent stenosis was also associated with higher post-PCI FFR values. While these findings may seem contradictory, post procedural percent stenosis was not associated with post-PCI physiology in the DEFINE PCI study either.24
In the intravascular ultrasound substudy of the FFR-SEARCH registry, van Zandvoort et al. showed that evident signs of residual luminal narrowing including focal lesions, underexpansion, and malapposition were present in a significant amount of vessels with post-PCI FFR values ≤ 0.85. These findings were not readily apparent on the comprehensive quantitative coronary angiography.25 Percent diameter stenosis was 20% in the cohort of patients with post-PCI FFR values ≤ 0.85 and > 0.85.26
Together with the latter predictors of post-PCI FFR we identified several others. A dedicated analysis of 26 CTOs recently showed that postprocedural FFR values are typically low initially; however they seem to increase at the 4-month follow-up. The initially low post-PCI FFR values is thought to be due to the microvascular dysfunction of the recently opened vessel, a phenomenon that improves after several months.27 In-stent restenosis and pre- and postdilatation were associated with lower post-PCI FFR values. A finding that is consistent with former studies that showed that, in general, complex lesions are associated with lower post-PCI FFR values.20,21,26,28
Also, it was interesting to see the impact of clinical presentation on post-PCI FFR values in the study population in which most patients presented with acute coronary syndrome. Contrary to former studies that questioned the validity of invasive hyperemic physiological indices in patients with acute coronary syndrome, we could not confirm the impact of clinical presentation on post-PCI FFR values. However, the identification of a thrombus, that often occurs after a ruptured plaque in patients with acute coronary syndrome, was associated with significantly higher FFR values. Despite the restoration of epicardial flow by the PCI, a relatively large number of patients with STEMI have abnormal myocardial perfusion at the end of the procedure.29 This phenomenon is thought to be related to microvascular obstruction due to distal embolization (reperfusion injury) and tissue inflammation due to myocyte necrosis.30,31 The latter may explain the significantly higher post-PCI FFR values reported in patients presenting with thrombus-containing lesions compared to those without such lesions. Conversely, our findings also show that in patients without thrombus-containing lesions the post-PCI FFR may be a valuable diagnostic tool for the identification of patients at a high risk of future adverse cardiac events.
Limitations
This study was conducted with the Navvus microcatheter, a dedicated rapid exchange microcatheter with a mean diameter of 0.022 in that proved its utility in a slight but significant underestimation of the FFR compared to conventional 0.014 in pressure guidewires.32 That is why we cannot directly extrapolate the current findings to wire-based FFR devices.14 Based on the study protocol, no further action was taken in the presence of low post-PCI FFR values. The Target FFR and FFR REACT studies (NCT03259815 and NTR6711) will provide further information on post-PCI FFR and the potential of further actions to improve post-PCI FFR and clinical outcomes.33,34 These studies should also focus on the trade-off of potential benefits and harm when performing additional interventions in order to improve the final FFR values.
CONCLUSIONS
In this substudy of the FFR-SEARCH registry, the largest real-world post-PCI FFR registry conducted to this day, we identified sex, LAD vessels, postprocedural MLD, and several other independent predictors of postprocedural FFR.
FUNDING
The FFR SEARCH study was conducted with institutional support from ACIST Medical Inc.
AUTHORS' CONTRIBUTION
Conception and design: L.J.C. van Zandvoort, N.M. van Mieghem, and J. Daemen. Data aquisition: L.J.C. van Zandvoort, K. Masdjedi, J. Wilschut, W. Den Dekker, R. Diletti, F. Zijlstra, N.M. van Mieghem, and J. Daemen. Statistical analysis and manuscript writing: L.J.C. van Zandvoort and J. Daemen. Providing criticial feedback to the manuscript and approving the final content: L.J.C. van Zandvoort, K. Masdjedi, T. Neleman, M.N Tovar Forero, J. Wilschut, W. Den Dekker, R. Diletti, F. Zijlstra, N.M. van Mieghem, and J. Daemen.
CONFLICTS OF INTEREST
L.J.C. van Zandvoort received institutional research support from Acist medical Inc. J. Daemen received institutional research support from Pie Medical, ACIST Medical Inc., PulseCath, Medtronic, Boston Scientific, Abbott Vascular, Pie Medical and speaker and consultancy fees from PulseCath, Medtronic, ReCor Medical, ACIST Medical Inc. and Pie Medical. The remaining authors declared no conflicts of interest.
WHAT IS KNOWN ABOUT THE TOPIC?
- FFR has proven to be a useful technique to address coronary physiology and the hemodynamic significance of coronary segments pre- and post-intervention.
- Also, the FFR post-stenting has proven to be a strong and independent predictor of major adverse cardiovascular events at the 2-year follow-up.
- Unfortunately, at present, there is lack of data on independent predictors of post PCI FFR.
WHAT DOES THIS STUDY ADD?
- This study is the largest report to this day on predictors of post-PCI FFR.
- Based on data from the FFR-SEARCH registry, we could identify several patient and procedural predictors of post-PCI FFR.
- The main predictors included sex, LAD vessels, and postprocedural lumen dimensions. These predictors will help us interpret post-PCI FFR values and identify correctly the vessels that are prone to future events.
REFERENCES
1. Topol EJ, Nissen SE. Our preoccupation with coronary luminology. The dissociation between clinical and angiographic findings in ischemic heart disease. Circulation. 1995;92:2333-2342.
2. De Bruyne B, Fearon WF, Pijls NHJ, et al. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med. 2014;1533-4406.
3. Wolfrum M, Fahrni G, de Maria GL, et al. Impact of impaired fractional flow reserve after coronary interventions on outcomes:a systematic review and meta-analysis. BMC Cardiovasc Disord. 2016;16:177.
4. Rimac G, Fearon WF, De Bruyne B, et al. Clinical value of post-percutaneous coronary intervention fractional flow reserve value:A systematic review and meta-analysis. Am Heart J. 2017;183:1-9.
5. Kasula S, Agarwal SK, Hacioglu Y, et al. Clinical and prognostic value of poststenting fractional flow reserve in acute coronary syndromes. Heart. 2016;102:1988-1994.
6. Sareen N, Baber U, Kezbor S, et al. Clinical and angiographic predictors of haemodynamically significant angiographic lesions:development and validation of a risk score to predict positive fractional flow reserve. EuroIntervention. 2017;12:e2228-e2235.
7. Baranauskas A, Peace A, Kibarskis A, et al. FFR result post PCI is suboptimal in long diffuse coronary artery disease. EuroIntervention. 2016;12:1473-1480.
8. Crystal GJ, Klein LW. Fractional flow reserve:physiological basis, advantages and limitations, and potential gender differences. Curr Cardiol Rev. 2015;11:209-219.
9. Ahmadi A, Leipsic J, Ovrehus KA, et al. Lesion-Specific and Vessel-Related Determinants of Fractional Flow Reserve Beyond Coronary Artery Stenosis. JACC Cardiovasc Imaging. 2018;11:521-530.
10. Tebaldi M, Biscaglia S, Fineschi M, et al. Fractional Flow Reserve Evaluation and Chronic Kidney Disease:Analysis From a Multicenter Italian Registry (the FREAK Study). Catheter Cardiovasc Interv. 2016;88:555-562.
11. Fineschi M, Guerrieri G, Orphal D, et al. The impact of gender on fractional flow reserve measurements. EuroIntervention. 2013;9:360-366.
12. Ryan TJ, Faxon DP, Gunnar RM, et al. Guidelines for percutaneous transluminal coronary angioplasty. A report of the American College of Cardiology/American Heart Association Task Force on Assessment of Diagnostic and Therapeutic Cardiovascular Procedures (Subcommittee on Percutaneous Transluminal Coronary Angioplasty). Circulation. 1988;78:486-502.
13. Diletti R, Van Mieghem NM, Valgimigli M, et al. Rapid exchange ultra-thin microcatheter using fibre-optic sensing technology for measurement of intracoronary fractional flow reserve. EuroIntervention. 2015;11:428-432.
14. Menon M, Jaffe W, Watson T, Webster M. Assessment of coronary fractional flow reserve using a monorail pressure catheter:the first-in-human ACCESS-NZ trial. EuroIntervention. 2015;11:257-263.
15. van de Hoef TP, Meuwissen M, Escaned J, et al. Fractional flow reserve as a surrogate for inducible myocardial ischaemia. Nat Rev Cardiol. 2013;10:439-452.
16. Kim HS, Tonino PA, De Bruyne B, et al. The impact of sex differences on fractional flow reserve-guided percutaneous coronary intervention:a FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation) substudy. JACC Cardiovasc Interv. 2012;5:1037-1042.
17. Reis SE, Holubkov R, Lee JS, et al. Coronary flow velocity response to adenosine characterizes coronary microvascular function in women with chest pain and no obstructive coronary disease. Results from the pilot phase of the Women's Ischemia Syndrome Evaluation (WISE) study. J Am Coll Cardiol. 1999;33:1469-1475.
18. Iqbal MB, Shah N, Khan M, Wallis W. Reduction in myocardial perfusion territory and its effect on the physiological severity of a coronary stenosis. Circ Cardiovasc Interv. 2010;3:89-90.
19. Lin FY, Devereux RB, Roman MJ, et al. Cardiac chamber volumes, function, and mass as determined by 64-multidetector row computed tomography:mean values among healthy adults free of hypertension and obesity. JACC Cardiovasc Imaging. 2008;1:782-786.
20. Nam CW, Hur SH, Cho YK, et al. Relation of fractional flow reserve after drug-eluting stent implantation to one-year outcomes. Am J Cardiol. 2011;107:1763-1767.
21. Doh JH, Nam CW, Koo BK, et al. Clinical Relevance of Poststent Fractional Flow Reserve After Drug-Eluting Stent Implantation. J Invasive Cardiol. 2015;27:346-351.
22. Agarwal SK, Kasula S, Hacioglu Y, Ahmed Z, Uretsky BF, Hakeem A. Utilizing Post-Intervention Fractional Flow Reserve to Optimize Acute Results and the Relationship to Long-Term Outcomes. JACC Cardiovasc Interv. 2016;9:1022-1031.
23. Kimura Y, Tanaka N, Okura H, et al. Characterization of real-world patients with low fractional flow reserve immediately after drug-eluting stents implantation. Cardiovasc Interv Ther. 2016;31:29-37.
24. Jeremias A, Davies JE, Maehara A, et al. Blinded Physiological Assessment of Residual Ischemia After Successful Angiographic Percutaneous Coronary Intervention:The DEFINE PCI Study. JACC:Cardiovasc Interv. 2019;12:1991-2001.
25. van Zandvoort LJC, Masdjedi K, Witberg K, et al. Explanation of Postprocedural Fractional Flow Reserve Below 0.85. Circ Cardiovasc Interv. 2019;12:e007030.
26. van Zandvoort LJC, Witberg K, Ligthart J, et al. Explanation of post procedural fractional flow reserve below 0.85:a comprehensive ultrasound analysis of the FFR Search registry. In Cardiovascular Research Technologies (CRT) Conference 2018 March 3-6;Washingtong DC, United States. 2018.
27. Karamasis GV, Kalogeropoulos AS, Mohdnazri SR, et al. Serial Fractional Flow Reserve Measurements Post Coronary Chronic Total Occlusion Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2018;11:e006941.
28. Pijls NH, Klauss V, Siebert U, et al. Coronary pressure measurement after stenting predicts adverse events at follow-up:a multicenter registry. Circulation. 2002;105:2950-2954.
29. Stone GW, Webb J, Cox DA, et al. Distal microcirculatory protection during percutaneous coronary intervention in acute ST-segment elevation myocardial infarction:a randomized controlled trial. JAMA. 2005;293:1063-1072.
30. Shah NR, Al-Lamee R, Davies J. Fractional flow reserve in acute coronary syndromes:A review. Int J Cardiol Heart Vasc. 2014;5:20-25.
31. Cuculi F, De Maria GL, Meier P, et al. Impact of microvascular obstruction on the assessment of coronary flow reserve, index of microcirculatory resistance, and fractional flow reserve after ST-segment elevation myocardial infarction. J Am Coll Cardiol. 2014;64:1894-904.
32. Pouillot C, Fournier S, Glasenapp J, et al. Pressure wire versus microcatheter for FFR measurement:a head-to-head comparison. EuroIntervention. 2018;13:e1850-e1856.
33. van Zandvoort LJC, Masdjedi K, Tovar Forero MN, et al. Fractional flow reserve guided percutaneous coronary intervention optimization directed by high-definition intravascular ultrasound versus standard of care:Rationale and study design of the prospective randomized FFR-REACT trial. Am Heart J. 2019;213:66-72.
34. Collison D, McClure JD, Berry C, Oldroyd KG. A randomized controlled trial of a physiology-guided percutaneous coronary intervention optimization strategy:Rationale and design of the TARGET FFR study. Clin Cardiol. 2020;43:414-422.
ABSTRACT
Introduction and objectives: The presence of comorbidities in elderly patients with non-ST-segment elevation acute coronary syndrome worsens its prognosis. The objective of the study was to analyze the impact of the burden of comorbidities in the decision of using invasive management in these patients.
Methods: A total of 7211 patients > 70 years old from 11 Spanish registries were included. Individual data were analyzed in a common database. We assessed the presence of 6 comorbidities and their association with coronary angiography during admission.
Results: The mean age was 79 ± 6 years and the mean CRACE score was 150 ± 21 points. A total of 1179 patients (16%) were treated conservatively. The presence of each comorbidity was associated with less invasive management (adjusted for predictive clinical variables): cerebrovascular disease (OR, 0.78; 95%CI, 0.64-0.95; P = .01), anemia (OR, 0.64; 95%CI, 0.54-0.76; P < .0001), chronic kidney disease (OR, 0.65; 95%CI, 0.56-0.75; P < .0001), peripheral arterial disease (OR, 0.79; 95%CI, 0.65-0.96; P = .02), chronic lung disease (OR, 0.85; IC95%, 0.71-0.99; P = .05), and diabetes mellitus (OR, 0.85; 95%CI, 0.74-0.98; P < .03). The increase in the number of comorbidities (comorbidity burden) was associated with a reduction in coronary angiographies after adjusting for the GRACE score: 1 comorbidity (OR, 0.66; 95%CI, 0.54-0.81), 2 comorbidities (OR, 0.55; 95%CI, 0.45-0.69), 3 comorbidities (OR, 0.37; 95%CI, 0.29-0.47), 4 comorbidities (OR, 0.33; 95%CI, 0.24-0.45), ≥ 5 comorbidities (OR, 0.21; 95%CI, 0.12-0.36); all P values < .0001 compared to 0.
Conclusions: The number of coronary angiographies performed drops as the number of comorbidities increases in elderly patients with non-ST-segment elevation acute coronary syndrome. More studies are still needed to know what the best management of these patients should be.
Keywords: Comorbidities. Elderly. Acute coronary syndrome. Coronary angiography.
Resumen
Introducción y objetivos: La comorbilidad en ancianos con síndrome coronario agudo sin elevación del segmento ST empeora el pronóstico. El objetivo fue analizar la influencia de la carga de comorbilidad en la decisión del tratamiento invasivo en ancianos con SCASEST.
Métodos: Se incluyeron 7.211 pacientes mayores de 70 años procedentes de 11 registros españoles. Los datos se analizaron en una base de datos conjunta. Se evaluó la presencia de 6 enfermedades simultáneas y su asociación con la realización de coronariografía durante el ingreso.
Resultados: La edad media fue de 79 ± 6 años y la puntuación GRACE media fue de 150 ± 21 puntos. Fueron tratados de manera conservadora 1.179 pacientes (16%). La presencia de cada enfermedad se asoció con un menor abordaje invasivo (ajustado por variables clínicas predictivas): enfermedad cerebrovascular (odds ratio [OR] = 0,78; intervalo de confianza del 95% [IC95%], 0,64-0,95; p = 0,01), anemia (OR = 0,64; IC95%, 0,54-0,76; p < 0,0001), insuficiencia renal (OR = 0,65; IC95%, 0,56-0,75; p < 0,0001), arteriopatía periférica (OR = 0,79; IC95%, 0,65-0,96; p = 0,02), enfermedad pulmonar crónica (OR = 0,85; IC95%, 0,71-0,99; p = 0,05) y diabetes mellitus (OR = 0,85; IC95%, 0,74-0,98; p = 0,03). Asimismo, el aumento del número de enfermedades (carga de comorbilidad) se asoció con menor realización de coronariografías, ajustado por la escala GRACE: 1 enfermedad (OR = 0,66; IC95%, 0,54-0,81); 2 (OR = 0,55; IC95%, 0,45-0,69); 3 (OR = 0,37; IC95%, 0,29-0,47); 4 (OR = 0,33; IC95%, 0,24-0,45); ≥ 5 (OR = 0,21; IC95%, 0,12-0,36); todos p < 0,0001, en comparación con ninguna enfermedad.
Conclusiones: Conforme aumenta la comorbilidad disminuye la realización de coronariografías en ancianos con síndrome coronario agudo sin elevación del segmento ST. Se necesitan estudios que investiguen la mejor estrategia diagnóstico-terapéutica en estos pacientes.
Palabras clave: Comorbilidad. Ancianos. Síndrome coronario agudo. Coronariografía.
Abbreviations:
ACS: acute coronary syndrome. DM: diabetes mellitus. NSTEACS: non-ST-segment elevation acute coronary syndrome.
INTRODUCTION
Population ageing leads to an increase in the number of elderly patients who suffer non-ST-segment elevation acute coronary syndrome (NSTEACS). This population group, that has been misrepresented in large studies, has a great comorbidity burden that increases with age1 and an important impact on prognosis.2-4 The ideal therapeutic strategy for the management of these patients is still unknown. The benefit of an invasive strategy in elderly patients with NSTEACS and comorbidities is still unclear.5-9 In general, elderly patients with comorbidities undergo fewer coronary angiographies despite their worse prognosis.10 This clinical practice —apparently in contrast with the recommendations published in the clinical practice guidelines11— seems to be based on the perception of a scarce benefit due to the worse intrinsic prognosis associated with comorbidities.
In this study the data of 11 Spanish NSTEACS registries were collected to set up a common database with over 7000 elderly patients with NSTEACS. In this preliminary analysis, the objective was to study the impact of comorbidities on the decision to go with invasive approach.
METHODS
Study design
The study was conducted from 11 cohorts of Spanish registries of patients with NSTEACS (annex).2,12-20 All cases were included in a single database of patients with chest pain and a diagnosis of NSTEACS, > 70 years of age and with, at least, a 1-year follow-up.
ANNEX. Registries included in the study.
| Hospital Clínico Universitario, Valencia2 |
| Hospital Universitario Joan XXIII, Tarragona12 |
| Hospital Universitario de Bellvitge, Barcelona13 |
| Hospital Ramón y Cajal, Madrid14 |
| Hospital Universitario de San Juan, Alicante15 |
| LONGEVO multicenter registry16 |
| ACHILLES multicenter registry17 |
| Hospital Álvaro Cunqueiro, Vigo18 |
| Hospital Clínico Universitario, Santiago de Compostela19 |
| Hospital Universitario Vall d’Hebron, Barcelona20 |
| Hospital Universitario de La Princesa, Madrid* |
|
* Unpublished data. |
The anthropometric and social-demographic data, main cardiovascular risk factors, and analytical and hemodynamic data at admission or during hospitalization were registered.
Patients were treated according to each center routine clinical practice and the decision to treat the NSTEACS invasively, with or without a coronary angiography, was left to the discretion of the treating physician. The 6-month mortality GRACE risk score was determined in all the patients.21
A total of 6 conditions that proved to have a higher prognostic impact on elderly patients hospitalized due to acute coronary syndrome (ACS) in a previous study were included:22 renal failure (glomerular filtration rate < 60mL/min/1.73m2), anemia (hemoglobin levels < 11 g/dL), diabetes mellitus (DM), cerebrovascular disease, peripheral arterial disease, and chronic pulmonary disease.
Endpoints
The study primary endpoint was to assess how the presence of comorbidities impacted the decision to perform a coronary angiography during admission.
Statistical analysis
Categorical variables were expressed as absolute values (percentages) and compared using the unpaired Student t test or the ANOVA. The continuous ones were expressed as mean ± standard deviation and compared using the chi-square test.
Initially, the correlation between each disease and the performance of a coronary angiography through univariable analysis were assessed. Then, a first binary logistics regression model was conducted including the 6 conditions and the clinical variables associated with the performance of the coronary angiography in the univariable analysis. The odds ratio (OR) and the 95% confidence intervals (95%CI) were estimated. Afterwards, patients were classified according to their comorbidity burden, defined by the number of concomitant conditions (from 0 to 6). A second logistics regression model was conducted where comorbidity burden was adjusted for the predictive clinical variables in the previous analysis. Finally, a third logistics regression model was conducted where the comorbidity burden was adjusted based on the GRACE risk score. Differences were considered statistically significant with P values < .05
RESULTS
A total of 7211 patients with a mean age of 79 ± 6 years were included; 62% were males. Table 1 shows the population baseline characteristics. The prevalence of comorbidities was DM in 2874 patients (40%), chronic kidney disease in 3070 patients (42.6%), anemia in 1025 (14.2%), peripheral arterial disease in 1006 (14%), chronic pulmonary disease in 1161 (16%), and previous stroke in 831 (11.5%).
Table 1. Differences in the baseline characteristics based on the therapeutic approach
| All N = 7211 | Conservative approach N = 1 179 (16) | Invasive approach N = 6 032 (84) | P | |
|---|---|---|---|---|
| Age (years) | 79 ± 6 | 82 ± 6 | 78 ± 5 | .001 |
| Males | 4 441 (61.6) | 597 (50.6) | 3 844 (63.7) | .0001 |
| Smoking | 621 (8.6) | 72 (6.1) | 549 (9.1) | .0001 |
| Hypertension | 5 723 (79.4) | 943 (80) | 4 780 (79.2) | .58 |
| Dyslipidemia | 4 262 (59) | 609 (51.7) | 3 653 (60.6) | .0001 |
| Previous myocardial infarction | 1 682 (23.3) | 371 (31.7) | 1 308 (21.7) | .0001 |
| Pervious percutaneous coronary intervention | 1 334 (19) | 175 (14.8) | 1 159 (19.2) | .0001 |
| Previous coronary surgery | 573 (7.9) | 104 (8.8) | 469 (7.8) | .24 |
| Previous heart failure | 641 (8.9) | 198 (16.8) | 443 (7.3) | .0001 |
| Killip ≥ 2 | 1 889 (26.2) | 463 (39.3) | 1 426 (23.6) | .0001 |
| ST-segment depression | 2 638 (36.6) | 396 (33.6) | 2 242 (37.2) | .02 |
| High troponin levels | 5 319 (73.7) | 920 (78) | 4 399 (73) | .001 |
| Left ventricular ejection fraction (%) | 54 ± 11 | 54 ± 12 | 55 ± 11 | .03 |
| GRACE | 150 ± 21 | 159 ± 21 | 147 ± 19 | .0001 |
| Comorbidities | ||||
| Anemia | 1 025 (14.2) | 273 (23.2) | 752 (12.5) | .0001 |
| Peripheral arterial disease | 1 006 (14) | 196 (16.6) | 810 (13.4) | .04 |
| Chronic pulmonary disease | 1 161 (16.1) | 210 (17.8) | 951 (15.8) | .08 |
| Diabetes mellitus | 2 874 (39.9) | 522 (44.3) | 2 352 (39) | .0001 |
| Cerebrovascular disease | 831 (11.5) | 186 (15.8) | 645 (10.7) | .0001 |
| Chronic kidney disease | 3 070 (42.6) | 716 (60.7) | 2 354 (39) | .0001 |
|
Data are expressed as no. (%) or mean ± standard deviation. |
||||
During admission 6032 patients (84%) underwent a coronary angiography. A total of 4339 patients (60%) were revascularized: 3848 (53%) of them through percutaneous coronary intervention and 491 (7%) through surgery. Patients on conservative management (1179, 16%) were predominantly women with higher scores in the GRACE score, and a past medical history of infarction or heart failure. Conversely, smoking and high levels of troponins or ST-segment depressions on the electrocardiogram performed at admission and a previous percutaneous coronary intervention were associated with a higher invasive approach (table 1). The GRACE risk score was lower in patients who underwent catheterization (147 ± 19 vs 159 ± 21; P = .0001).
The presence of each of the 6 conditions studied was associated with fewer coronary angiographies performed: chronic kidney disease, 60.7% vs 39% (P = .0001); anemia, 23.2% vs 12.5% (P = .0001); DM, 44.3% vs 39% (P = .0001); cerebrovascular disease, 15.8% vs 10.7% (P = .0001); peripheral arterial disease, 16.6% vs 13.4% (p = .04); and chronic pulmonary disease, 17.8% vs 15.8% (P = .08) (table 1).
In the multivariable analysis adjusted for the main cardiovascular risk factors and clinical variables that were statistically significant in the univariable analysis, the 6 conditions associated with a lower probability of an indication for coronary angiography were: cerebrovascular disease, OR, 0.78 (IC95%, 0.64-0.95; P = .01); anemia, OR, 0.64 (IC95%, 0.54-0.76; P < .0001); chronic kidney disease, OR, 0.65 (IC95%, 0.56-0.75; P < .0001); peripheral arterial disease, OR, 0.79 (IC95%, 0.65-0.96; P = .02); chronic pulmonary disease, OR, 0.85 (IC95%, 0.71-0.99; P = .05); and DM, OR, 0.85 (IC95%, 0.74-0.98; P = .03). Table 2 shows the clinical variables associated with the indication for coronary angiography.
Table 2. Results: multivariable analysis for the indication of a coronary angiography
| Variable | OR | 95%CI | P | |
|---|---|---|---|---|
| Age (years) | 0.89 | 0.88-0.91 | .0001 | |
| Males | 1.48 | 1.28-1.71 | .0001 | |
| Dyslipidemia | 1.44 | 1.26-1.66 | .0001 | |
| Previous myocardial infarction | 0.46 | 0.39-0.54 | .0001 | |
| Previous heart failure | 0.68 | 0.56-0.84 | .0001 | |
| Previous percutaneous coronary intervention | 1.91 | 1.55-2.34 | .0001 | |
| Killip ≥ 2 | 0.68 | 0.56-0.80 | .0001 | |
| ST-segment depression | 1.44 | 1.25-1.66 | .0001 | |
| Left ventricular ejection fraction (by 5%) | 0.98 | 0.98-0.99 | .001 | |
| Anemia | 0.64 | 0.54-0.76 | .0001 | |
| Peripheral artery disease | 0.79 | 0.65-0.96 | .02 | |
| Chronic pulmonary disease | 0.85 | 0.71-0.99 | .05 | |
| Diabetes mellitus | 0.85 | 0.74-0.98 | .03 | |
| Cerebrovascular disease | 0.78 | 0.64-0.95 | .01 | |
| Chronic kidney disease | 0.65 | 0.56-0.75 | .0001 | |
|
95%CI: 95% confidence interval; OR: odds ratio. |
||||
Comorbidity burden was defined as the number of present conditions (from 0 to 6). This was their distribution: 0 conditions, N = 1891 (26%); 1 condition, N = 2413 (33.5%); 2 conditions, N = 1638 (22.7%); 3 conditions, N = 879 (12.2%); 4 conditions, N = 314 (4.4%); and 5 or 6 conditions, N = 76 (1.1%). The analysis of the comorbidity burden adjusted for the clinical variables associated with the indication for coronary angiography showed a negative correlation between the number of conditions and the probability to perform a coronary angiography: 1 condition, OR, 0.66 (95%CI, 0.54-0.81); 2 conditions, OR, 0.55 (95%CI, 0.45-0.69); 3 conditions, OR, 0.37 (95%CI, 0.29-0.46); 4 conditions, OR, 0.32 (95%CI, 0.23-0.45); and 5 or 6 conditions, OR, 0.21 (95%CI, 0.12-0.37); All P values < .0001 compared to no condition.
With more conditions, higher GRACE risk scores (table 3). The negative correlation between the comorbidity burden and the performance of the coronary angiography was kept after adjusting for the GRACE risk score. Figure 1 shows that with more conditions, the probability to perform a coronary angiography increased too (figure 1A) despite the higher risk posed by higher GRACE risk scores (figure 1B, table 3).
Table 3. Distribution of comorbidity burden and the score obtained in the GRACE risk score (P < .0001 for the tendency)
| Conditions | N = 7 211 | GRACE risk score |
|---|---|---|
| 0 | 1891 (26) | 141 ± 18 |
| 1 | 2413 (33.5) | 148 ± 19 |
| 2 | 1638 (22.7) | 153 ± 20 |
| 3 | 879 (12.2) | 160 ± 19 |
| 4 | 314 (4.4) | 162 ± 19 |
| ≥ 5 | 76 (1.1) | 166 ± 17 |
|
Data are expressed as no. (%) or mean ± standard deviation. |
||
Figure 1. A: chances of undergoing a coronary angiography based on the number of concomitant conditions. The odds ratio (OR) with a 95% confidence interval (95%IC) can be seen. Analysis adjusted for the GRACE risk score. B: Representation of the correlation between comorbidity burden and the GRACE risk score. The mean ± standard deviation (SD) of the GRACE risk score can be seen.
DISCUSSION
The main findings of our study were: a) the 6 conditions studied (cerebrovascular disease, anemia, chronic kidney disease, peripheral arterial disease, chronic pulmonary disease, and DM) were independently associated with a lower probability to use the invasive approach; b) with higher comorbidity burdens, considered as the number of concomitant conditions, lower chances of performing coronary angiographies.
There is a high prevalence of comorbidities in elderly patients with NSTEACS that greatly impacts prognosis in the short and mid-term.2,4 The Charlson index is the most commonly used tool to assess comorbidities.23,24 However, the analysis of the 6 conditions studied (chronic kidney disease, anemia, DM, cerebrovascular disease, peripheral arterial disease, and chronic pulmonary disease) has proven to be a useful risk stratification tool and have great predictive discriminatory capabilities that are similar to the Charlson index.22
Comorbidity burden is very important for the in-hospital management of NSTEACS.2,3,6,9,10 Although the optimal therapeutic strategy for the management of elderly patients with NSTEACS is still unknown, several studies show certain benefits with revascularization.5,7,8,25-30
Our study shows that with higher comorbidity burdens, lower chances of undergoing coronary angiographies. This may be due to the fact that comorbidities are seen as contraindications for the invasive approach.10 However, the risk of suffering an acute myocardial infarction according to the GRACE risk score increases parallel to the number of concomitant conditions. Actually, these may be the patients who would benefit the most from an invasive approach.31,32
The presence of each one of these 6 conditions was independently associated with fewer invasive approaches. On the one hand, cerebrovascular disease and peripheral arterial disease are responsible for a greater spread of atherosclerotic disease.33 Anemia has proven to be a powerful predictor of mortality in the ACS setting;34-36 we used the 11 g/dL threshold as the cut-off value that had the greatest impact on mortality in former studies.34 Its specific weight in the decision to administer conservative treatment may be justified by its clear association with the occurrence of hemorrhagic events in the ACS setting.37 Chronic kidney disease is an expression of a greater spread of cardiovascular disease and is independently associated with more mortality after an ACS. There is a linear correlation between the risk of death due to cardiovascular causes and lower glomerular filtration rates.17,38 DM is a powerful predictor of mortality, and not only due to cardiovascular causes. There is a clear correlation between DM and major adverse cardiovascular events, and these are patients at very high risk.39 Chronic pulmonary disease is associated with a worse short-term prognosis after an acute myocardial infarction. Also, in the management of NSTEACS it is associated with diagnostic delays, fewer invasive approaches, and a lower use of drugs for secondary prevention purposes.40
In the multivariable analysis, age, previous acute myocardial infarctions, previous heart failure, Killip class ≥ 2 at admission, and a reduced ejection fraction were associated with fewer invasive approaches. Elderly patients receive fewer evidence-based therapies. The older the age, the lower the rate of performing coronary angiographies.41 On top of age, a past medical history of infarction, heart failure, a reduced ejection fraction, and scores ≥ 2 in the Killip classification are important aspects in the prognosis of ACS that, in general, translate into a worse ventricular function. Paradoxically, our findings suggest that the higher the risk, the lower the chances of performing a coronary angiography. Actually, these findings are consistent with former studies published.10,42 It is possible that the perception of fewer benefits from revascularization or higher risk in the revascularization procedures may explain these results.2 On the other hand, male sex, dyslipidemia, previous percutaneous coronary interventions, and ST-segment depressions at admission were associated with more invasive approaches. Several studies suggest that women undergo fewer invasive approaches compared to men despite the mortality benefits seen.43 Previous angioplasties, ST-segment depressions, and dyslipidemia are probably interpreted as ischemic risk factors, which may explain their association with a higher frequency of invasive approaches.20,28,44
Limitations
The main limitation of our study is that it is an observational registry with its corresponding selection bias and differences in the management of patients depending on the different centers involved. On the other hand, although the multivariable model was adjusted for percutaneous coronary intervention or previous coronary surgeries, it was not adjusted for previous coronary angiographies. It is possible that the previous knowledge of the coronary anatomy impacted the decision to perform fewer coronary angiographies in patients at higher risk.
CONCLUSIONS
The presence of comorbidities greatly impacts the therapeutic decision in elderly patients with ACS. With more conditions, higher GRACE risk scores, and lower chances of indicating a coronary angiography
This paradox of higher-risk and more conservative treatment justifies conducting new studies to determine the benefits of the invasive strategy in elderly patients with NSTEACS and comorbidities to establish the best therapeutic decision.
FUNDING
This article was funded by a grant from the Carlos III Health Institute: CIBERCV 16/11/00420, Madrid, Spain.
CONFLICTS OF INTEREST
J. Sanchis is an associate editor of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. J. Núñez has received funding from Novartis, Vitor Pharma, and Boehringer Ingelheim, and a grant from Astra Zeneca and Vitor Pharma. J.A. Barrabés has received funding for the educational activities conducted for AstraZeneca, and for his job as consultor for Bayer. The remaining authors did not declare any conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Elderly patients with NSTEACS have a higher comorbidity burden. Concomitant conditions are associated with worse prognosis. Elderly patients with comorbidities undergo fewer coronary angiographies despite their worse prognosis, which is in sharp contrast with the recommendations published in the clinical practice guidelines.
WHAT DOES THIS STUDY ADD?
- This analysis of a multicenter registry shows the correlation between comorbidity burden and invasive therapeutic approach in elderly patients with NSTEACS. With more concomitant conditions, higher GRACE risk scores, but lower chances of indicating a coronary angiography.
REFERENCES
1. Barnett K, Mercer SW, Norbury M, Watt G, Wyke S, Guthrie B. Epidemiology of multimorbidity and implications for health care, research, and medical education:a cross-sectional study. Lancet. 2012;380:37-43.
2. Sanchis J, Núñez J, BodíV, Núñez E, García-Alvarez A, Bonanad C, et al. Influence of comorbid conditions on one-year outcomes in non-ST-segment elevation acute coronary syndrome. Mayo Clin Proc. 2011;86:291-296.
3. Chirinos JA, Veerani A, Zambrano JP, Schob A, Perez G, Mendez AJ, et al. Evaluation of comorbidity scores to predict all-cause mortality in patients with established coronary artery disease. Int J Cardiol. 2007;117:97-102.
4. Sanchis J, Bonanad C, Ruiz V, Fernández J, García-Blas S, Mainar L, et al. Frailty and other geriatric conditions for risk stratification of older patients with acute coronary syndrome. Am Heart J. 2014;168:784-791.
5. Bardaji A, Barrabés JA, Ribera A, et al. Revascularization in older adult patients with non-ST-segment elevation acute coronary syndrome:effect and impact on 6-month mortality [published online ahead of print, 2019 May 14]. Eur Heart J Acute Cardiovasc Care. 2019;2048∖19849922.
6. Chuang AM, Hancock DG, Halabi A, et al. Invasive management of acute coronary syndrome:Interaction with competing risks. Int J Cardiol. 2018; 269:13-18.
7. Tegn N, Abdelnoor M, Aaberge L, et al. Invasive versus conservative strategy in patients aged 80 years or older with non-ST-elevation myocardial infarction or unstable angina pectoris (After Eighty study):an open-label randomized controlled trial. Lancet. 2016;387:1057-1065.
8. Sanchis J, Núñez E, Barrabés JA, et al. Randomized comparison between the invasive and conservative strategies in comorbid elderly patients with non-ST elevation myocardial infarction. Eur J Intern Med. 2016;35:89-94.
9. Palau P, Núñez J, Sanchis J, et al. Differential prognostic effect of revascularization according to a simple comorbidity index in high-risk non-ST- segment elevation acute coronary syndrome. Clin Cardiol. 2012;35:237-243.
10. Savonitto S, Morici N, De Servi S. Treatment of acute coronary syndromes in the elderly and in patients with comorbidities. Rev Esp Cardiol. 2014;67:564-573.
11. Roffi M, Patrono C, Collet JP, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation:Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:267-315.
12. Camprubi M, Cabrera S, Sans J, et al. Body Mass Index and Hospital Mortality in Patients with Acute Coronary Syndrome Receiving Care in a University Hospital. J Obes. 2012;2012:287939.
13. Ariza-SoléA, Sánchez-Salado JC, Lorente V, et al. Is it possible to separate ischemic and bleeding risk in patients with non-ST segment elevation acute coronary syndromes?Int J Cardiol. 2014;171:448-450.
14. Alonso Salinas GL, Sanmartín Fernández M, Pascual Izco M, et al. Frailty predicts major bleeding within 30 days in elderly patients with Acute Coronary Syndrome. Int J Cardiol. 2016;222:590-593.
15. Cordero A, López-Palop R, Carrillo P, et al. Prevalence and Postdischarge Incidence of Malignancies in Patients With Acute Coronary Syndrome. Rev Esp Cardiol. 2018;71:267-273.
16. Alegre O, Formiga F, López-Palop R, et al. An Easy Assessment of Frailty at Baseline Independently Predicts Prognosis in Very Elderly Patients With Acute Coronary Syndromes. J Am Med Dir Assoc. 2018;19:296-303.
17. Rivera-Caravaca JM, Ruiz-Nodar JM, Tello-Montoliu A, et al. Disparities in the Estimation of Glomerular Filtration Rate According to Cockcroft-Gault, Modification of Diet in Renal Disease-4, and Chronic Kidney Disease Epidemiology Collaboration Equations and Relation With Outcomes in Patients With Acute Coronary Syndrome. J Am Heart Assoc. 2018;7:e008725.
18. Abu-Assi E, Raposeiras-Roubin S, Cobas-Paz R, et al. Assessing the performance of the PRECISE-DAPT and PARIS risk scores for predicting one-year out-of-hospital bleeding in acute coronary syndrome patients. EuroIntervention. 2018;13:1914-1922.
19. Álvarez Álvarez B, Abou Jokh Casas C, Cordero A, et al. Early revascularization and long-term mortality in high-risk patients with non-ST-elevation myocardial infarction. The CARDIOCHUS-HUSJ registry. Rev Esp Cardiol. 2020;73:35-42.
20. MilàL, Barrabés JA, Lidón RM, et al. Prior adherence to recommended lipid control targets in patients admitted for acute coronary syndrome. Rev Esp Cardiol. 2019;73:376-382.
21. Eagle KA, Lim MJ, Dabbous OH, et al. A validated prediction model for all forms of acute coronary syndrome:estimating the risk of 6-month postdischarge death in an international registry. JAMA. 2004:291:2727-2733.
22. Sanchis J, Soler M, Núñez J, et al. Comorbidity assessment for mortality risk stratification in elderly patients with acute coronary syndrome. Eur J Intern Med. 2019;62:48-53.
23. Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies:development and validation. J Chronic Dis. 1987;40:373-383.
24. Núñez J, Núñez E, Fácila L, et al. Prognostic value of Charlson comorbidity index at 30 days and 1 year after acute myocardial infarction. Rev Esp Cardiol. 2004;57:842-849.
25. De Servi S, Cavallini C, Dellavalle A, et al. Non-ST-elevation acute coronary syndrome in the elderly:treatment strategies and 30-day outcome. Am Heart J. 2004;147:830-836.
26. Sillano D, Resmini C, Meliga E, et al. Retrospective multicenter observational study of the interventional management of coronary disease in the very elderly:the NINETY. Catheter Cardiovasc Interv. 2013;82:414-421.
27. Kolte D, Khera S, Palaniswamy C, et al. Early invasive versus initial conservative treatment strategies in octogenarians with UA/NSTEMI. Am J Med. 2013;126:1076-1083.
28. Núñez J, Ruiz V, Bonanad C, et al. Percutaneous coronary intervention and recurrent hospitalizations in elderly patients with non ST-segment acute coronary syndrome:The role of frailty. Int J Cardiol. 2017;228:456-458.
29. LlaóI, Ariza-SoléA, Sanchis J, et al. Invasive strategy and frailty in very elderly patients with acute coronary syndromes. EuroIntervention. 2018;14:e336-342.
30. Sanchis J, Ariza-SoléA, Abu-Assi E, et al. Invasive Versus Conservative Strategy in Frail Patients With NSTEMI:The MOSCA-FRAIL Clinical Trial Study Design. Rev Esp Cardiol. 2019;72:154-159.
31. de Groot V, Beckerman H, Lankhorst GJ, et al. How to measure comorbidity. A critical review of available methods. J Clin Epidemiol. 2003;56:221-229.
32. Park JY, Kim MH, Bae EJ, et al. Comorbidities can predict mortality of kidney transplant recipients:comparison with the Charlson comorbidity index. Transplant Proc. 2018;50:1068-1073.
33. Chirinos JA, Veerani A, Zambrano JP, et al. Evaluation of comorbidity scores to predict all-cause mortality in patients with established coronary artery disease. Int J Cardiol. 2007;117:97-102.
34. Lawler PR, Filion KB, Dourian T, et al. Anemia and mortality in acute coronary syndromes:a systematic review and meta-analysis. Am Heart J. 2013;165:143-153.
35. Ford I, Bezlyak V, Stott DJ, et al. Reduced glomerular filtration rate and its association with clinical outcome in older patients at risk of vascular events:secondary analysis. PLoS Med. 2009;6:e16.
36. Sabatine MS, Morrow DA, Giugliano RP, et al. Association of hemoglobin levels with clinical outcomes in acute coronary syndromes. Circulation. 2005;111:2042-2049.
37. Vicente-Ibarra N, Marín F, Pernias-Escrig V, et al. Impact of anemia as risk factor for major bleeding and mortality in patients with acute coronary syndrome. Eur J Intern Med. 2019;61:48-53.
38. Goldenberg I, Subirana I, Boyko V, et al. Relation between renal function and outcomes in patients with non-ST-segment elevation acute coronary syndrome:real-world data from the European Public Health Outcome Research and Indicators Collection Project. Arch Intern Med. 2010;170:888-895.
39. Rao Kondapally Seshasai S, Kaptoge S, Thompson A, et al. Diabetes mellitus, fasting glucose, and risk of cause-specific death [published correction appears in N Engl J Med. 2011;364:1281]. N Engl J Med. 2011;364:829-841.
40. Rothnie KJ, Smeeth L, Herrett E, et al. Closing the mortality gap after a myocardial infarction in people with and without chronic obstructive pulmonary disease. Heart. 2015;101:1103-1110.
41. Avezum A, Makdisse M, Spencer F, et al. Impact of age on management and outcome of acute coronary syndrome:observations from the Global Registry of Acute Coronary Events (GRACE). Am Heart J. 2005;149:67-73.
42. Itzahki Ben Zadok O, Ben-Gal T, Abelow A, et al. Temporal Trends in the Characteristics, Management and Outcomes of Patients With Acute Coronary Syndrome According to Their Killip Class. Am J Cardiol. 2019;124:1862-1868.
43. Mehta LS, Beckie TM, DeVon HA, et al. Acute Myocardial Infarction in Women:A Scientific Statement From the American Heart Association. Circulation. 2016;133:916-947.
44. Yudi MB, Clark DJ, Farouque O, et al. Trends and predictors of recurrent acute coronary syndrome hospitalizations and unplanned revascularization after index acute myocardial infarction treated with percutaneous coronary intervention. Am Heart J. 2019;212:134-143.
- Angiographic and clinical results of the STENTYS Xposition S self-apposing stent. A single-center experience
- Usefulness of a co-registration strategy with iFR in long and/or diffuse coronary lesions (iLARDI): study protocol
- Rotational atherectomy for the management of bifurcation lesions: a pilot randomized study
- Microalbuminuria predicts contrast-induced nephropathy in patients with acute coronary syndrome
Interviews
An interview with Bruno Scheller
aServicio de Cardiología, Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria de La Princesa (IIS-IP), Universidad Autónoma de Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain