Article
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
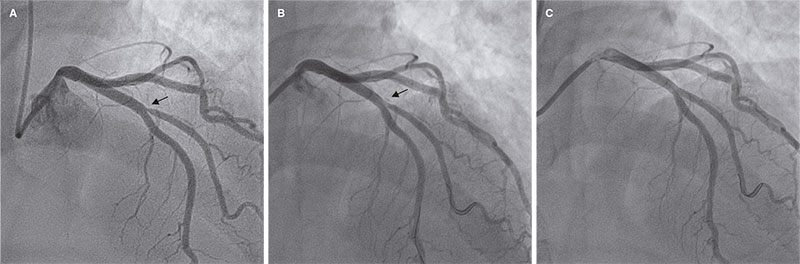
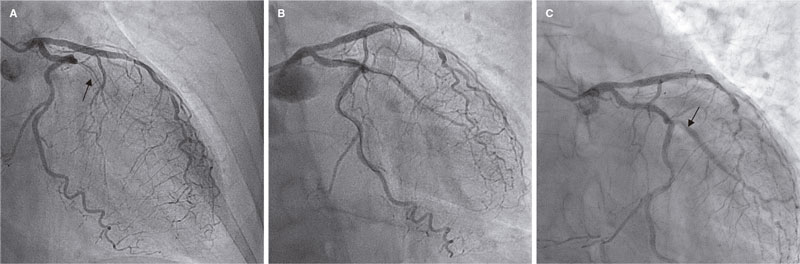
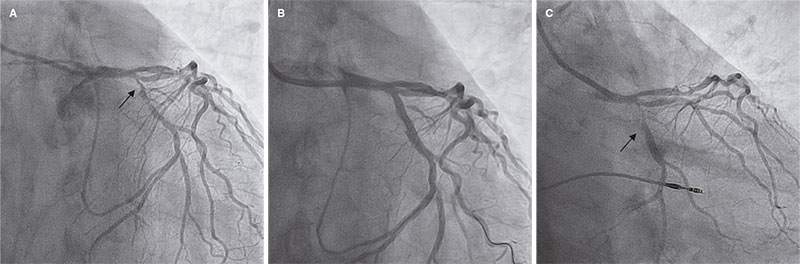
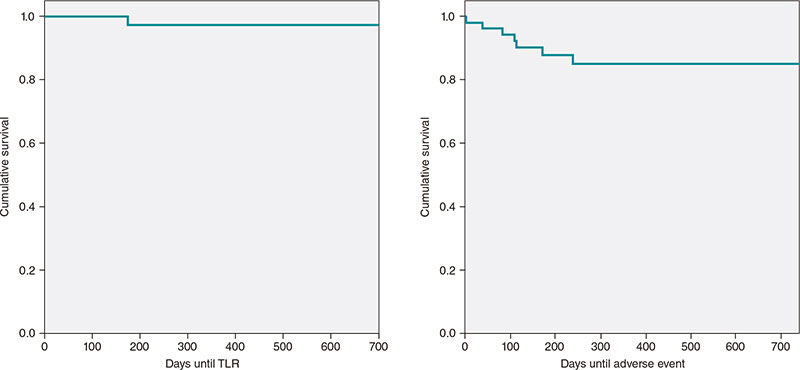
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España
ABSTRACT
Introduction and objectives: Geographical and organizational differences between different autonomous communities (AC) can generate differences in care for ST-segment elevation myocardial infarction (STEMI). A total of 17 heart attack code programs have been compared in terms of incidence rate, clinical characteristics, reperfusion therapy, delay to reperfusion, and 30-day mortality.
Methods: National prospective observational study (83 centers included in 17 infarction networks). The recruitment period was 3 months (April 1 to June 30, 2019) with clinical follow-up at 30 days.
Results: 4366 patients with STEMI were included. The incidence rate was variable between different AC (P < .0001), as was gender (P = .003) and the prevalence of cardiovascular risk factors (P < .0001). Reperfusion treatment was primary angioplasty (range 77.5%-97.8%), fibrinolysis (range 0%-12.9%) or no treatment (range 2.2%- 13.5%). The analysis of the delay to reperfusion showed significant differences (P < .001) for all the intervals analyzed. There were significant differences in 30-days mortality that disappeared after adjusting for clinical and healthcare network characteristics.
Conclusions: Large differences in STEMI care have been detected between the different AC, in terms of incidence rate, clinical characteristics, reperfusion treatment, delay until reperfusion, and 30-day mortality. The differences in mortality disappeared after adjusting for the characteristics of the patient and the care network.
Keywords: STEMI. Population characteristics. Angioplasty.
RESUMEN
Introducción y objetivos: Las diferencias geográficas y organizativas entre distintas comunidades autónomas (CCAA) pueden generar diferencias en la atención al infarto agudo de miocardio con elevación del segmento ST (IAMCEST). Se han comparado 17 programas de Código Infarto en términos de incidencia, características clínicas, tratamiento de reperfusión, retraso hasta la reperfusión y mortalidad a 30 días.
Métodos: Estudio observacional prospectivo nacional (83 centros en 17 redes de infarto). El periodo de selección fue de 3 meses (1 de abril a 30 de junio de 2019), con seguimiento clínico a 30 días.
Resultados: Se incluyeron 4.366 pacientes con IAMCEST. La tasa de incidencia fue variable entre las CCAA (p < 0,0001), igual que el sexo (p = 0,003) y la prevalencia de factores de riesgo cardiovascular (p < 0,0001). El tratamiento de reperfusión fue angioplastia primaria (rango 77,5-97,8%), fibrinolisis (rango 0- 12,9%) o ninguno (rango 2,2-13,5%). El análisis del retraso hasta la reperfusión mostró diferencias significativas (p < 0,001) para todos los intervalos analizados. Hubo diferencias significativas en la mortalidad cruda a 30 días que desaparecieron tras ajustar por las características clínicas y dependientes de la red asistencial (primer contacto, tiempo hasta la reperfusión y abordaje de críticos).
Conclusiones: Se han detectado diferencias en la atención al IAMCEST entre las distintas CCAA, en términos de incidencia, características clínicas, tratamiento de reperfusión, retraso hasta la reperfusión y mortalidad a 30 días. Las diferencias en mortalidad desaparecen tras ajustar por las características del paciente y de la red asistencial.
Palabras clave: IAMCEST. Características de la población. Angioplastia.
Abbreviations
ACI-SEC: Interventional Cardiology Association at the Spanish Society of Cardiology. AC: autonomous communities. pPCI: primary percutaneous coronary intervention. STEMI: ST-segment elevation myocardial infarction.
INTRODUCTION
Infarction Code networks are key to treat ST-segment elevation myocardial infarction (STEMI) in the shortest time possible while optimizing reperfusion therapy.1 In Spain we have 17 different public regional STEMI networks, 1 in each autonomous community (AC) for a total of 83 pPCI-capable hospitals in programs on a 24/7/365 basis.2 According to data from the Annual Activity Registry of the Interventional Cardiology Association of the Spanish Society of Cardiology (ACI-SEC), back in 2019, a total of 22 529 interventional procedures were performed in patients with infarction.3 Recently, an analysis of the ACI-SEC Infarction Code Registry revealed the characteristics of infarction care in Spain with 87.5%, 4.4%, and 8.1% of the patients with STEMI being treated with pPCI, fibrinolysis, and without reperfusion, respectively. The 30-day mortality rate of STEMI was 7.9% dropping down to 6.8% in patients treated with pPCI.4
The geographical differences and heterogeneity of the organizational infrastructure among the different Infarction Code programs available can lead to regional differences as a survey conducted among health professionals involved in these programs revealed recently.5 These organizational differences can have an impact on the management of patients with STEMI. Their analysis and AC-based comparison facilitates finding matters where there is room for improvement to optimize treatment.
This analysis compared the incidence rate, clinical characteristics, type and time to reperfusion, the characteristics of pPCI, and the 30-day mortality rate of 17 different regional programs of the Infarction Code in Spain.
METHODS
Study design
The Registry design has already been introduced4. In conclusion, this was a national, observational, and prospective study of 83 centers from 17 different regional STEMI networks. The patients’ recruitment period was 3 months—from April 1 through June 30, 2019—with a 30-day clinical follow-up.
Registry protocol was approved by the reference central ethics committee that did not deem the obtention of the informed consent necessary since data anonymity was guaranteed at any time.
Inclusion criteria
All consecutive patients who, during the study period, triggered the activation of different regional infarction care networks with a final diagnosis of STEMI and met the following criteria were included in the study: a) diagnosis of ST-segment elevation acute coronary syndrome with symptoms consistent with acute coronary syndrome, electrocardiogram showing ST-segment elevation or new-onset left bundle branch block or suspected posterior infarction of, at least, 24-hour evolution since symptom onset or b) recovered cardiac arrest with suspected coronary etiology or c) cardiogenic shock with suspected coronary etiology.
Definition and collection of variables
Clinical variables were registered in an online form and previously published.4 The definitions of the different time intervals since symptom onset until reperfusion were given based on the recommendations established by the European clinical practice guidelines on the management of STEMI.1 Subjective judgment from a local investigator was requested on the delay sustained by the patient since his first medical contact (existence of unjustified delay—yes/no—and reason why). To estimate the incidence rate (number of cases per million inhabitants) population data from the National Statistics Institute from 2019 were used.6 Regarding the mortality adjusted analysis, the following characteristics of the care network were defined: the individual responsible for the first medical contact (emergency medical services, health center, non-pPCI-capable hospital, pPCI-capable hospital), time to reperfusion, and location where critical care was administered (intensive care unit or cardiac surgery intensive care unit).
Statistical analysis
Continuous variables were expressed as mean ± standard deviation. The categorical ones were expressed as frequencies and percentages. Inter-group comparisons of baseline variables were conducted using the chi-square test or the Student t test, when appropriate. Times to reperfusion were expressed as median and interquartile range and compared using the Mann-Whitney U test.
Poisson regression coefficient was used to estimate the 30-day mortality rate of each AC including patient-dependent factors (the confounding factors included were age, sex, hypertension, diabetes, dyslipidemia, smoking, previous ischemic heart disease, Killip classification, and anterior location of STEMI), and the healthcare network involved (location of the first medical contact, time between the onset of pain and reperfusion, and location where critically ill patients were treated).
The variable AC was introduced in the model in a second step, and a test of ratio of verisimilitude was performed to verify its statistical significance. When the AC variable was added, adjusted associations were obtained between AC and mortality. The Poisson regression coefficients became incidence rates using the marginal effect function. The estimated 30-day mortality rate for each AC was obtained from a mean distribution of confounding factors, which facilitated comparing mortality rate across the different AC. This method had been previously used in the acute myocardial infarction setting.7-9 Since there could be a selection bias across the different AC in patients without reperfusion therapy, these were not included in the adjusted mortality analysis.
P values < .05 were considered statistically significant. The STATA statistical software package version 15 SE (Stata Corp, College Station, United States) was used.
RESULTS
Patients
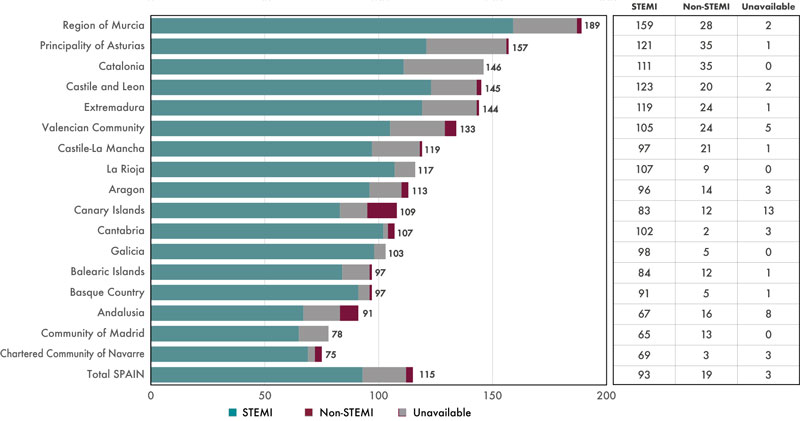
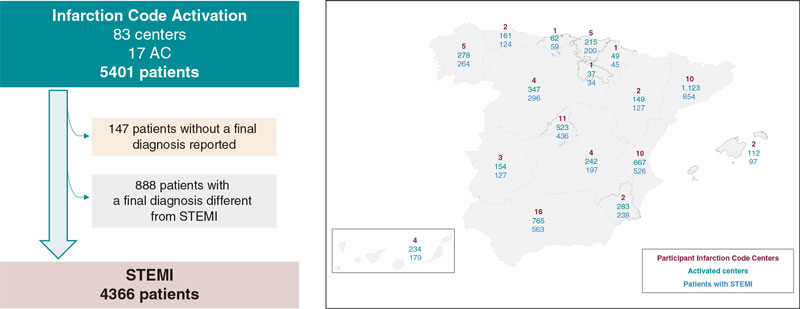
The registry included a total of 5401 patients, 4366 (81.2%) of whom had a final diagnosis of STEMI. The 888 patients (16.4%) with a diagnosis different from STEMI and the 147 (2.7%) without a final diagnosis were excluded from the analysis. Figure 1 shows the flow of patients and the AC-based distribution. Figure 2 shows the number of patients treated across the different AC plus the final diagnosis achieved adjusted by million inhabitants.6 Table 1 shows the clinical characteristics of patients with STEMI across the different AC.
Figure 1. Flow of patients and distribution across the different autonomous communities (AC) based on participant centers, number of codes activated, and number of patients with ST-segment elevation myocardial infarction (STEMI) as final diagnosis.
Figure 2. Patients treated across the different autonomous communities (AC) adjusted for million inhabitants. AC were arranged from largest to smallest number of patients treated per million inhabitants. Regarding the population estimate per million inhabitants, population data from the National Statistics Institute were used.6 STEMI, ST-segment elevation myocardial infarction.
Table 1. Clinical characteristics of patients with ST-segment elevation myocardial infarction treated in the Infarction Code networks per autonomous community
| Age, years | Sex, women | AHT | Diabetes | Dyslipidemia | Active smoking | Previous IHD | Previous PCI | Previous stroke | Early Killip I | Early Killip IV | Anterior location | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Andalusia | 63 ± 13 | 110/563 (19.5) | 297/560 (53.0) | 159/558 (28.5) | 252/559 (45.1) | 264/557 (47.4) | 60/561 (10.7) | 59/559 (10.6) | 31/556 (5.6) | 423/541 (78.2) | 31/541 (5.7) | 223/521 (42.8) |
| Aragon | 65 ± 14 | 30/127 (23.6) | 62/127 (48.8) | 28/125 (22.4) | 56/127 (44.1) | 59/124 (47.6) | 13/124 (10.5) | 17/126 (13.5) | 7/122 (5.7) | 99/124 (79.8) | 13/124 (10.5) | 56/120 (46.7) |
| Principality of Asturias | 66 ± 13 | 40/124 (32.3) | 61/124 (49.2) | 34/122 (27.9) | 54/124 (43.6) | 41/123 (33.3) | 20/123 (16.3) | 19/123 (15.4) | 7/123 (5.6) | 96/123 (78.1) | 11/123 (8.9) | 57/122 (46.7) |
| Balearic Islands | 63 ± 12 | 28/97 (28.9) | 44/94 (46.8) | 21/94 (22.3) | 49/93 (52.7) | 49/93 (52.7) | 14/93 (15.1) | 14/94 (14.9) | 4/92 (4.4) | 71/96 (74.0) | 5/96 (5.2) | 30/92 (32.6) |
| Canary Islands | 60 ± 12 | 40/178 (22.5) | 99/178 (55.6) | 52/178 (29.2) | 102/177 (57.6) | 93/178 (52.3) | 22/178 (12.4) | 18/178 (10.1) | 8/176 (4.6) | 146/168 (86.9) | 14/168 (8.3) | 65/163 (39.9) |
| Cantabria | 62 ± 13 | 15/59 (25.4) | 31/59 (52.5) | 21/58 (36.2) | 27/58 (46.6) | 31/57 (54.4) | 10/58 (17.2) | 10/59 (17.0) | 3/57 (5.3) | 46/56 (83.9) | 2/56 (3.6) | 25/58 (43.1) |
| Castile and Leon | 64 ± 13 | 56/296 (18.9) | 146/293 (49.8) | 73/291 (25.1) | 126/292 (43.2) | 117/292 (40.1) | 31/293 (10.6) | 31/294 (10.5) | 12/176 (4.1) | 236/287 (82.2) | 17/287 (5.9) | 138/280 (49.3) |
| Castile-La Mancha | 64 ± 13 | 26/197 (13.2) | 108/194 (55.7) | 58/192 (30.2) | 99/196 (50.5) | 92/193 (47.7) | 19/192 (9.9) | 18/194 (9.3) | 9/194 (4.6) | 157/196 (80.1) | 12/196 (6.1) | 89/194 (45.9) |
| Catalonia | 63 ± 13 | 195/854 (22.8) | 393/854 (46.0) | 198/854 (23.2) | 340/854 (39.8) | 354/854 (41.4) | 60/854 (7.0) | 62/854 (7.3) | 30/854 (3.5) | 683/826 (82.7) | 67/826 (8.1) | 351/767 (45.8) |
| Extremadura | 63 ± 13 | 18/127 (14.2) | 74/127 (58.3) | 26/126 (20.6) | 52/126 (41.3) | 48/127 (37.8) | 17/126 (13.5) | 14/126 (11.1) | 4/127 (3.2) | 91/122 (74.6) | 11/122 (9.0) | 56/121 (46.3) |
| Galicia | 63 ± 13 | 63/264 (23.9) | 130/262 (49.6) | 48/259 (18.5) | 138/261 (52.9) | 100/215 (46.5) | 18/261 (6.9) | 25/262 (9.5) | 12/263 (4.6) | 195/251 (77.7) | 31/251 (12.4) | 103/233 (44.2) |
| La Rioja | 59 ± 12 | 8/34 (23.5) | 14/34 (41.2) | 3/34 (8.8) | 16/34 (46.1) | 20/34 (58.8) | 1/34 (3.0) | 2/34 (5.9) | 0/34 (0) | 30/34 (88.2) | 3/34 (8.8) | 11/34 (32.4) |
| Community of Madrid | 63 ± 13 | 105/436 (24.1) | 212/432 (49.1) | 88/430 (20.5) | 208/431 (48.3) | 177/428 (41.4) | 41/429 (9.6) | 43/429 (10.0) | 11/429 (2.6) | 347/424 (81.8) | 35/424 (8.3) | 174/419 (41.5) |
| Region of Murcia | 64 ± 13 | 43/238 (18.1) | 127/237 (53.6) | 71/237 (30.0) | 100/237 (42.4) | 110/237 (46.4) | 41/237 (17.3) | 24/151 (15.9) | 3/151 (2.0) | 196/237 (82.7) | 18/237 (7.6) | 101/231 (43.7) |
| Chartered Community of Navarre | 65 ± 14 | 14/45 (31.1) | 18/44 (40.9) | 9/45 (20.0) | 29/45 (64.4) | 16/45 (35.6) | 3/45 (6.7) | 4/44 (9.1) | 3/45 (6.7) | 31/43 (72.1) | 4/43 (9.3) | 16/44 (36.4) |
| Basque Country | 64 ± 14 | 52/200 (26.0) | 101/197 (51.3) | 39/197 (19.8) | 101/198 (51.0) | 89/197 (45.2) | 26/195 (13.3) | 32/196 (16.3) | 11/193 (5.7) | 169/200 (84.5) | 12/200 (6.0) | 83/199 (41.7) |
| Valencian Community | 63 ± 13 | 119/526 (22.6) | 293/519 (56.5) | 163/514 (31.7) | 212/514 (41.3) | 235/514 (45.7) | 56/515 (10.9) | 53/511 (10.4) | 21/513 (4.1) | 445/520 (85.6) | 34/520 (6.5) | 217/503 (43.1) |
| P | .054 | .003 | .038 | < .0001 | < .0001 | .007 | < .0001 | .011 | .61 | .016 | .25 | .44 |
| Total | 63 ± 13 | 962/4365 (22.0) | 2210/4335 (51.0) | 1091/4314 (25.3) | 1961/4326 (45.3) | 1895/4268 (44.4) | 452/4318 (10.5) | 445/4234 (10.5) | 176/4222 (4.2) | 3462/4248 (81.5) | 320/4248 (7.5) | 1795/4101 (43.8) |
|
AHT, arterial hypertension; IHD, ischemic heart disease; PCI, percutaneous coronary intervention. |
||||||||||||
Reperfusion therapy used in patients with ST-segment elevation myocardial infarction
Out of the 4366 patients with STEMI, 3792 (86.9%) received pPCI, 189 (4.3%) fibrinolysis, and 353 (8.1%) no reperfusion therapy whatsoever. No reperfusion therapy was reported in 32 patients (0.7%). Figure 3 shows treatment distribution based on AC. Table 2 shows, across different AC and patients treated with cardiac catheterization, the angiographic findings and characteristics of interventional therapy had this procedure been performed.
Figure 3. Distribution of reperfusion therapy in patients with ST-segment elevation myocardial infarction by autonomous communities. pPCI, primary percutaneous coronary intervention.
Table 2. Angiographic findings and characteristics of interventional procedures in patients with ST-segment elevation myocardial infarction treated with cardiac catheterization per autonomous community
| Radial access | No. of diseased vessels | Early TIMI grade-0/1 flow | Final TIMI grade-3 flow | Need for hemodynamic support | Thrombus aspiration in IRA | BMS implantation in IRA | DES implantation in IRA | pPCI | Bailout PCI | Elective PCI after fibrinolysis | Coronary angiography without PCI | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Andalusia | 456/534 (85.4) | 1.49 ± 0.69 | 416/535 (77.8) | 502/536 (93.7) | 15/563 (2.7) | 76/563 (13.5) | 48/563 (8.5) | 456/563 (81.0) | 471/557 (84.6) | 36/557 (6.5) | 27/557 (4.9) | 23/557 (4.1) |
| Aragon | 111/122 (91.0) | 1.62 ± 0.78 | 90/120 (75.0) | 114/122 (93.4) | 5/127 (3.9) | 41/127 (32.3) | 0/127 (0) | 103/127 (81.1) | 108/124 (87.1) | 6/124 (4.8) | 1/124 (0.8) | 9/124 (7.3) |
| Principality of Asturias | 99/121 (81.8) | 1.54 ± 0.77 | 106/121 (87.6) | 111/121 (91.7) | 5/124 (4.0) | 39/124 (31.5) | 10/124 (8.1) | 98/124 (79.0) | 118/123 (95.9) | 0/123 (0) | 0/123 (0) | 5/123 (4.1) |
| Balearic Islands | 79/92 (85.9) | 1.46 ± 0.67 | 67/92 (72.8) | 85/92 (92.4) | 0/124 (0) | 27/97 (27.8) | 4/97 (4.1) | 80/97 (82.5) | 89/96 (92.7) | 4/96 (4.2) | 0/96 (0) | 3/96 (3.1) |
| Canary Islands | 138/169 (81.7) | 1.54 ± 0.76 | 131/170 (77.1) | 155/169 (91.7) | 6/179 (3.6) | 29/179 (16.2) | 3/179 (1.7) | 150/179 (83.8) | 145/176 (82.4) | 6/176 (3.4) | 15/176 (8.5) | 10/176 (5.7) |
| Cantabria | 17/56 (30.4) | 1.50 ± 0.68 | 51/57 (89.5) | 55/56 (98.2) | 1/59 (1.7) | 31/59 (52.5) | 0/59 (0) | 51/59 (86.4) | 57/59 (96.6) | 0/59 (0) | 1/59 (1.7) | 1/59 (1.7) |
| Castile and Leon | 263/281 (93.6) | 1.55 ± 0.74 | 192/241 (79.7) | 225/247 (91.1) | 15/296 (5.1) | 27/296 (9.1) | 9/296 (3.0) | 249/296 (84.1) | 255/291 (96.6) | 12/291 (4.1) | 16/291 (5.5) | 8/291 (2.8) |
| Castile-La Mancha | 164/191 (85.9) | 1.68 ± 0.73 | 164/192 (85.4) | 186/190 (97.9) | 9/197 (4.6) | 75/197 (38.1) | 10/197 (5.1) | 172/197 (97.3) | 185/196 (94.4) | 2/196 (1.0) | 4/196 (2.0) | 5/196 (2.6) |
| Catalonia | 727/781 (93.1) | 1.48 ± 0.70 | 594/844 (70.4) | 787/827 (95.2) | ND | 259/854 (30.3) | 117/854 (13.7) | 653/854 (76.5) | 807/849 (95.1) | 8/849 (0.9) | 3/849 (0.4) | 31/849 (3.7) |
| Extremadura | 119/121 (98.4) | 1.65 ± 0.79 | 104/122 (85.3) | 104/122 (85.3) | 6/127 (4.7) | 18/127 (14.2) | 12/127 (11.0) | 98/127 (77.2) | 112/126 (88.9) | 8/126 (6.4) | 2/126 (1.6) | 4/126 (3.2) |
| Galicia | 228/242 (94.2) | 1.53 ± 0.84 | 182/229 (79.5) | 214/229 (93.5) | 20/264 (7.6) | 77/264 (29.2) | 4/264 (1.5) | 215/264 (81.4) | 246/264 (93.2) | 0/264 (0) | 0/264 (0) | 18/264 (6.8) |
| La Rioja | 29/34 (85.3) | 1.15 ± 0.36 | 30/34 (88.2) | 31/34 (91.2) | 0/24 (0) | 10/34 (29.4) | 3/34 (8.8) | 27/34 (79.4) | 33/34 (97.1) | 0/34 (0) | 0/34 (0) | 1/34 (2.9) |
| Community of Madrid | 395/421 (93.8) | 1.48 ± 0.69 | 329/402 (81.8) | 392/425 (92.2) | 23/436 (5.3) | 80/436 (18.4) | 15/436 (3.4) | 352/436 (80.5) | 421/434 (97.0) | 3/434 (0.7) | 0/434 (0) | 10/434 (2.3) |
| Region of Murcia | 213/237 (89.9) | 1.48 ± 0.64 | 175/234 (74.8) | 223/236 (94.5) | 4/238 (1.7) | 56/238 (23.5) | 5/238 (2.1) | 209/238 (87.2) | 226/238 (95.0) | 7/238 (2.9) | 0/238 (0) | 5/238 (2.1) |
| Chartered Community of Navarre | 31/36 (86.1) | 2.00 ± 0.86 | 34/43 (79.1) | 39/45 (86.7) | 6/45 (13.3) | 22/45 (48.9) | 2/45 (4.4) | 39/45 (86.7) | 44/45 (97.8) | 0/45 (0) | 0/45 (0) | 1/45 (2.2) |
| Basque Country | 179/198 (90.4) | 1.51 ± 0.67 | 153/198 (77.3) | 191/199 (96.0) | 7/200 (3.5) | 100/200 (50.0) | 3/200 (1.5) | 174/200 (87.0) | 194/199 (97.5) | 4/199 (2.0) | 1/199 (0.5) | 0/199 (0) |
| Valencian Community | 484/514 (94.2) | 1.59 ± 0.76 | 390/496 (78.6) | 461/497 (92.8) | 8/256 (1.5) | 145/526 (27.6) | 34/526 (6.5) | 423/526 (80.4) | 482/518 (93.1) | 10/518 (1.9) | 4/518 (0.8) | 22/518 (4.3) |
| P | < .0001 | .84 | < .0001 | .002 | < .0001 | < .0001 | < .0001 | .004 | < .0001 | |||
| Total | 3732/4150 (89.9) | 1.50 ± 0.71 | 3208/4130 (77.7) | 3875/4147 (93.4) | 110/4366 (2.5) | 1112/4366 (25.5) | 281/4366 (6.4) | 3548/4366 (81.3) | 3992/4329 (92.2) | 106/4329 (2.5) | 74/4329 (1.7) | 157/4329 (3.6) |
|
BMS, bare metal stent; CL, cath lab; DES, drug-eluting stent; ECG, electrocardiogram; EMS, emergency medical services; FMC, first medical contact; IRA, infarct-related artery; PCI, percutaneous coronary intervention; pPCI, primary percutaneous coronary intervention. |
||||||||||||
Time intervals between symptom onset and reperfusion in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention
Table 3 shows time intervals between symptom onset and reperfusion. Figure 4 shows the different time intervals analyzed for every AC with significant differences in all of them. Figure 5 summarizes the causes of unjustified delays between the first medical contact and reperfusion for every AC.
Table 3. Location of the first medical contact and time intervals between the first medical contact and reperfusion per autonomous community
| First EMS care | First care provided at the health center | First non-pPCI-capable center care | First pPCI-capable center care | Transfer without going to the CL right away* | Time of onset of pain to FMC | Time of FMC to ECG | Time of FMC to pPCI-capable center in transferred patients | Time from FMC to reperfusion | Time from onset of pain to reperfusion | |
|---|---|---|---|---|---|---|---|---|---|---|
| Andalusia | 206/537 (38.4) | 138/537 (25.7) | 93/537 (17.3) | 100/537 (18.6) | 188/427 (44.0) | 60 [30-123] | 5 [3-10] | 80 [50-120] | 113 [70-170] | 195 [135-330] |
| Aragon | 46/123 (37.4) | 23/123 (18.7) | 42/123 (34.1) | 12/123 (9.8) | 23/110 (20.9) | 62.5 [18.5-170] | 7 [4-12.5] | 84.5 [45-145] | 116.5 [70.5-177.5] | 229 [126-345] |
| Principality of Asturias | 32/123 (26.0) | 18/123 (14.6) | 36/123 (29.3) | 37/123 (30.1) | 4/86 (4.7) | 80 [32-210] | 10 [5-22] | 85 [60-119] | 108 [73-137] | 215 [134.5-351] |
| Balearic Islands | 33/95 (34.7) | 26/95 (27.4) | 27/95 (28.4) | 9/95 (9.5) | 3/85 (3.5) | 70 [30-164] | 6 [5-10] | 100 [55-139] | 124 [85-169] | 197.5 [143.5-391] |
| Canary Islands | 28/178 (15.7) | 103/178 (57.9) | 22/178 (12.4) | 25/178 (14.0) | 77/152 (50.7) | 75 [37.5-150] | 9 [5-15] | 85 [55-133] | 122 [95-172] | 220 [159-385] |
| Cantabria | 15/58 (25.9) | 19/58 (32.8) | 13/58 (22.4) | 11/58 (19.0) | 26/46 (56.5) | 53 [25-145] | 5 [4.5-10] | 60 [35-93] | 110 [81-188] | 210 [134-303.5] |
| Castile and Leon | 97/290 (33.5) | 70/290 (27.2) | 68/290 (23.5) | 46/290 (15.9) | 70/237 (29.5) | 90 [35-221] | 8 [4-15] | 115 [70-165] | 135 [85-197] | 242.5 [163-432.5] |
| Castile-La Mancha | 69/196 (35.2) | 61/196 (31.1) | 30/196 (17.3) | 36/196 (18.4) | 49/160 (30.6) | 68 [30-160] | 10 [5-15] | 86.5 [58-114] | 109 [80-155] | 205 [150-322] |
| Catalonia | 332/847 (39.2) | 161/847 (19.0) | 256/847 (30.2) | 98/847 (11.6) | 115/730 (15.8) | 63 [30-160] | 6 [3-14] | 75 [55-105] | 104 [80-138] | 180 [127-288] |
| Extremadura | 43/126 (34.1) | 36/126 (28.6) | 22/126 (17.5) | 25/126 (19.8) | 27/93 (29.0) | 81.5 [44-135] | 10 [5-12] | 91.5 [60-143] | 121 [90-178] | 240 [160-360] |
| Galicia | 84/264 (31.8) | 111/264 (42.1) | 28/264 (10.6) | 41/264 (15.5) | ND | 60 [26-179] | 9 [5-19] | 95 [70-140] | 115 [88.5-163] | 194 [134-353] |
| La Rioja | 10/34 (29.4) | 9/34 (26.5) | 6/34 (17.7) | 9/34 (26.5) | 3/25 (12.0) | 76.5 [35-110] | 4.5 [1-10] | 70 [46-86] | 90.5 [67-114] | 159.5 [118.5-212.5] |
| Community of Madrid | 196/429 (45.7) | 37/429 (8.6) | 80/429 (18.7) | 116/429 (27.0) | 142/309 (45.6) | 63 [35-140] | 6 [3-12] | 60 [42-85] | 95 [75-130] | 178.5 [135-257.5] |
| Region of Murcia | 102/238 (42.9) | 36/238 (15.1) | 74/238 (31.1) | 26/238 (10.9) | 25/212 (11.8) | 56.5 [24-131] | 5 [5-10] | 80 [60-120] | 103 [79-160] | 175 [130-305] |
| Chartered Community of Navarre | 22/45 (48.9) | 7/45 (15.6) | 3/45 (6.7) | 13/45 (28.9) | 12/32 (37.5) | 63.5 [29.5-124.5] | 1 [0-5] | 50 [35-91] | 90 [69-140] | 175 [128-262] |
| Basque Country | 76/199 (38.2) | 28/199 (14.1) | 37/199 (18.6) | 58/199 (29.2) | 61/138 (44.2) | 80 [32-184] | 6.5 [3-11] | 61 [49-77] | 97 [75-135] | 210 [134-345] |
| Valencian Community | 128/521 (24.6) | 146/521 (28.0) | 128/521 (24.6) | 119/521 (22.8) | 98/398 (24.6) | 82 [35-180] | 5 [0-10] | 94 [65-135] | 120 [93-165] | 220 [146-348] |
| P | < .0001 | < .0001 | < .0001 | < .0001 | < .001 | .001 | .0001 | .0001 | .0001 | .0001 |
| Total | 1519/4303 (35.3) | 1038/4303 (24.1) | 965/4303 (22.4) | 781/4303 (18.2) | 923/3240 (28.5) | 67 [30-165] | 7 [4-15] | 80 [55-120] | 110 [80-154] | 197 [135-330] |
|
CL, cath lab; ECG, electrocardiogram; EMS, emergency medical services; FMC, first medical contact; pPCI, primary percutaneous coronary intervention. * Patients treated early in a non-pPCI-capable center requiring immediate transfer to a pPCI-capable center. Data are expressed as no. (%) or mean [interquartile range]. Times are expressed in minutes. |
||||||||||
Figure 4. Time intervals between symptom onset and reperfusion in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention (pPCI) for every autonomous community. A: time in min from the onset of pain to the first medical contact. B: time in min from the first medical contact to the electrocardiogram (ECG). C: time in min from the first medical contact to reperfusion. D: time in min from the onset of pain to reperfusion. E: time in min from the first medical contact to the arrival at the pPCI-capable center in patients requiring transfer from a non-pPCI-capable center.
Figure 5. Causes of unjustified time delays between the first medical contact and reperfusion. Unjustified time delays did not imply, necessarily, that the time between the first medical contact and reperfusion was > 120 min. As a matter of fact, overall, in 53.2% of the cases the time between the first medical contact and reperfusion was < 120 min, and, among these, excessive time delays were reported in 21.5%. EMS, emergency medical services; pPCI, primary percutaneous coronary intervention.
Mortality analysis in patients with ST-segment elevation myocardial infarction
Table 4 includes unadjusted mortality data at hospital admission and 30 days, and mortality for the adjusted model.
Table 4. Mortality analysis in patients treated with primary percutaneous coronary intervention per autonomous community
| Unadjusted hospital mortality | Unadjusted 30-day mortality | Adjusted 30-day mortality | |
|---|---|---|---|
| Andalusia | 30/563 (5.3) | 37/523 (7.1) | 6.0 [5.3-6-7] |
| Aragon | 8/127 (6.3) | 8/124 (6.5) | 5.5 [4.0-6.9] |
| Principality of Asturias | 9/124 (7.3) | 10/118 (8.5) | 6.7 [5.4-8.0] |
| Balearic Islands | 6/97 (6.2) | 6/88 (6.8) | 5.0 [3.3-6.7] |
| Canary Islands | 15/179 (8.4) | 15/155 (9.7) | 7.0 [5.5-8.6] |
| Cantabria | 0/59 (0) | 0/59 (0) | 0 |
| Castile and Leon | 18/296 (6.1) | 23/270 (8.5) | 8.4 [7.1-9.8] |
| Castile-La Mancha | 9/197 (4.6) | 10/191 (5.2) | 3.1 [2.3-3.8] |
| Catalonia | 29/854 (3.4) | 58/801 (7.2) | 6.0 [5.4-6.6] |
| Extremadura | 12/127 (9.5) | 16/125 (12.8) | 8.1 [6.6-9.5] |
| Galicia | 22/264 (8.3) | 28/260 (10.8) | 6.8 [5.6-7.9] |
| La Rioja | 1/34 (2.9) | 1/33 (3.0) | 5.6 [2.3-8.9] |
| Community of Madrid | 14/436 (3.2) | 21/421 (5.0) | 3.9 [3.3-4.6] |
| Region of Murcia | 21/237 (8.9) | 24/226 (10.6) | 9.2 [8.0-10.5] |
| Chartered Community of Navarre | 5/45 (11.1) | 5/45 (11.1) | 9.5 [6.7-12.3] |
| Basque Country | 12/200 (6.0) | 16/197 (8.1) | 8.9 [7.4-10.4] |
| Valencian Community | 47/526 (8.9) | 55/499 (11.0) | 10.2 [9.2-11.2] |
| P | < .001 | < .001 | .19 |
| Total | 258/4365 (5.9) | 337/4166 (8.1) | – |
|
Data are expressed as no. (%) or mean [interquartile range]. |
|||
30-day mortality rate was different across different AC (P < .001). When the analysis was adjusted for patient-dependent factors and the healthcare network, mortality difference across the AC lost its statistical significance (P = .19).
DISCUSSION
This study is a comparative of how the different STEMI care programs work in Spain. Results show differences in the incidence rate, the patients’ clinical profile, revascularization therapy, the characteristics of the interventional procedure performed, infarction care times, and the 30-day unadjusted mortality rate. Although mortality differences reduce, they’re still significantly different after adjusting for the patients’ risk and clinical characteristics. Also, they disappear after adjusting for whoever is responsible for the first medical contact, time to reperfusion, and location where critical care is administered, all of them factors associated with the way each network is organized.
Both functioning and results of infarction care networks are highly influenced by different factors like geography, the number of capable centers, transfer times, the availability of the right resources, infrastructure, and the characteristics of each healthcare system.2 In Spain, the plan of each AC has been designed independently. Also, the services rendered by the different AC is not homogeneous since resource allocation by the different administrations of the 17 Spanish AC is decentralized2 in such a way that there are inequalities in the ways these networks are organized.2,5,10,11 A recent consensus document on the requirements and sustainability of pPCI programs in Spain proposed measures to homogenize and secure their sustainability.2,12 Our study data reinforce the need for taking measures like the proposals made in the said consensus document.
Differences in the patients’ clinical profile
Registry data demonstrated a difference in the number of codes activated per million inhabitants. Also, in the number of patients with STEMI per million inhabitants across the different AC. These differences are multifactorial and can be seen, historically, in the ACI-SEC annual activity registry reports.3 Some AC have older populations and more cardiovascular risk factors, which could account for the higher rate of infarction reported.6 However, the lack of a unified criterion on the indication for Infarction Code activation could also account for these differences seen.5
Differences in reperfusion therapy
pPCI is the treatment of choice for the management of STEMI.1 The geographical (populations far from pPCI-capable centers) and organizational characteristics (availability of medical service transport with ECG monitorization) across the different AC lead to a variable number of patients be treated with fibrinolysis. A previous analysis of data on the Codi Infart in Catalonia revealed that patients treated with fibrinolysis in non- pPCI-capable centers had worse disease progression compared to those transferred to pPCI-capable centers within the first 140 min after diagnosis.13
Different time delays to reperfusion
Patient-dependent time delays (from symptom onset to first medical contact) were highly variable. Although the geographic distribution of the population could partially account for these differences, public campaigns should be run to increase awareness on STEMI symptoms and the need for calling out-of-hospital emergency care.1
System-dependent time delays (from first medical contact to reperfusion) is much easier to change with organizational measures. Also, it determines prognosis.14 Time delays to reperfusion depend on whoever is involved in the first medical contact. Therefore, patients treated by emergency medical services—those with the shortest times—showed high variability across the different programs. Better access to these systems for the population would also improve time delays to reperfusion.15
European clinical practice guidelines on the management of STEMI describe quality indicators that should be observed by the infarction networks to reduce the time to reperfusion, among these, a single coordination centralized center, interpreting the ECG before arriving at the hospital to achieve diagnosis and activate the system early, the direct transfer of patients to the cath lab without ER or ICU admissions or the follow-up of infarction care times, among other.1 Our study demonstrated that not all programs meet these recommendations meaning that, in many cases, there is a huge room for improvement. For example, currently, it does not seem reasonable that a significant number of patients who need to be transferred to the pPCI (up to 50% in some cases) wouldn’t end up at the cath lab right away. This simple measure can reduce time to reperfusion in 20 min and have a direct impact on prognosis.16,17
The presence of unjustified delayed reperfusion times was highly variable across the different AC, as well as the causes for these delays, which is indicative of the characteristics of each AC.
Mortality differences
A study conducted by Cequier et al.18 analyzed standardized mortality based on the risk of patients with STEMI across different AC from 2003 through 2012 and detected significant differences. However, across this period, not all regions had implemented Infarction Code programs and the rate of pPCI was highly variable. Our study demonstrated that there are still differences in crude mortality that disappear after adjusting for the clinical variables and care network-related variables (location of first medical contact, delay to reperfusion, and management of critically ill patients). We have already mentioned the importance that the first medical contact should be performed by emergency medical services and the measures used to reduce time to reperfusion. Regarding the management of critically ill patients, a study conducted by Sánchez-Salado et al.19 of 20 000 patients with cardiogenic shock demonstrated that the availability of cardiac surgery intensive care units was associated with a lower mortality rate. Data from this study added to the finding of our registry support the need for expanding the availability of cardiac surgery intensive care units in large volume centers of patients with acute coronary syndrome. In conclusion, the results of mortality study suggest that the organization of the different networks would increase the crude mortality rate seen in some AC.
Limitations
This study has some limitations. In the first place, it is based on self-reported data without external auditing. However, data on interventional cardiology are rather standardized across the world, and the electronic form for data curation was designed to be applied both intuitively and universally. Also, data from Catalonia and Galicia were collected from their official registries, reviewed, and then audited.
Secondly, the profile of patients may have been different across the different AC. To address this limitation and its possible impact on the different crude mortality rates reported, a mortality study was conducted across different AC after adjusting for different clinical variables and care networks. Therefore, some models may be over-adjusted, which is why formal statistical comparisons across AC should be interpreted as cautious as the associations described in any observational trial. The model did not include patients lacking some of the variables included in the model. Table 1 of the supplementary data shows patients discarded from the study for every AC.
Thirdly, patients with STEMI treated outside the infarction networks were not included in this study, although this is probably indicative of a mild selection bias due to its reduced number. Therefore, the greater bias occurs in patients without reperfusion therapy, who, at times, are not covered by these networks. For this reason, these patients were not considered in the mortality analysis. Similarly, patients with myocardial infarction and subacute presentation without emergency reperfusion criteria were not included in the study.
Fourthly, the way of collecting times may have presented some differences between centers and AC. However, since this was a prospective study with previously established definitions, we believe that these differences may have been minimized.
In the fifth place, the data presented date back to 2019. Since then, no big organizational changes have occurred to justify changes in the dynamics of functioning or relevant changes have been made in the European guidelines on the management of STEMI (published back in 2017). Also, in a study conducted during the first wave of the COVID-19 pandemic no differences were seen regarding the type of reperfusion therapy used or time between the first medical contact and reperfusion. However, an increased mortality rate was seen attributed, among other causes, to longer ischemia times.20
Finally, this study only included patients for a period of 3 months. However, we think these data can be generalized to what happens in a much larger period.
CONCLUSIONS
This registry showed significant differences in STEMI care across the different Spanish AC regarding incidence rate, the patients’ clinical characteristics, reperfusion therapy, time delays to reperfusion, and 30-day crude mortality rate. After adjusting for the clinical characteristics and variables associated with the care network, no differences mortality differences were reported across the different AC.
Standardizing the organization and functioning of Infarction Code networks could correct some of the differences seen in the management of STEMI.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
Drafting of the manuscript: O. Rodríguez-Leor, A.B. Cid-Álvarez, A. Pérez de Prado, and X Rosselló. Process of manuscript revision: all the authors. Statistical analysis: O. Rodríguez-Leor, and X. Rosselló. Database review: O. Rodríguez-Leor, A.B. Cid-Álvarez, and A. Pérez de Prado. Data coordination across the different regional network: all the authors.
CONFLICTS OF INTEREST
A. Pérez de Prado received numerous personal fees from iVascular, Boston Scientific, Terumo, Bbraun, and Abbott Vascular. Á. Cequier received personal fees from Ferrer International, Terumo, Astra Zeneca, and Biotronik. R. Moreno, S. Ojeda, R. Romaguera, and A. Pérez de Prado are associate editors of REC: Interventional Cardiology. The journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. The remaining authors did not declare any conflicts of interest associated with the content of this manuscript.
ACKNOWLEDGEMENTS
The authors wish to thank all health professionals involved in STEMI care programs for their not always rewarded work, effort, and dedication. Also, they wish to thank Meia Faixedas, and Josepa Mauri from the Departament de Salut de la Generalitat de Catalunya for granting us access to data from the Catalonian Registre de Codi Infart, and the entire personnel from Servicio Gallego de Salud (SERGAS) involved in the coordination of the REGALIAM registry for facilitating access to its data.
REFERENCES
1. Ibañez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology. Eur Heart J. 2018;39:39:119-177.
2. Cequier A, Pérez de Prado A, Cid-Álvarez AB, et al. Requisitos y sostenibilidad de los programas de ICP primaria en España en el IAMCEST. Documento de consenso de SEC, AEEC y SEMES. REC Interv Cardiol. 2019;1:108-119.
3. Ojeda S, Romaguera R, Cruz-González I, Moreno R. Registro español de hemodinámica y cardiología intervencionista. XXIX Informe Oficial de la Asociación de Cardiología Intervencionista de la Sociedad Española de Cardiología (1990-2019). Rev Esp Cardiol. 2020;73:927-936.
4. Rodríguez-Leor O, Cid-Álvarez AB, Pérez de Prado A, et al. Análisis de la atención al infarto con elevación del segmento ST en España. Resultados del Registro de Código Infarto de la ACI-SEC. Rev Esp Cardiol. 2022;75:669-680.
5. Rodríguez-Leor O, Cid-Álvarez AB, Moreno R, et al. Encuesta sobre las necesidades de los programas de angioplastia primaria en España. REC Interv Cardiol. 2020;1:8-14.
6. Instituto Nacional de Estadística. Datos de población de comunidades autónomas. Available online: https://www.ine.es/jaxiT3/Datos.htm?t=2915#!tabs-tabla. Accessed 24 Jan 2022.
7. Bueno H, Rosselló X, Pocock SJ, et al. In-hospital coronary revascularization rates and post-discharge mortality risk in non-ST-segment elevation acute coronary syndrome. J Am Coll Cardiol. 2019;74:1454-1461.
8. Rosselló X, Huo Y, Pocock S, et al. Global geographical variations in ST-segment elevation myocardial infarction management and post-discharge mortality. Int J Cardiol. 2017;245:27-34.
9. Bueno H, Rosselló X, Pocock S, et al. Regional variations in hospital management and post-discharge mortality in patients with non-ST-segment elevation acute coronary syndrome. Clin Res Cardiol. 2018;107:836-844.
10. Alter DA, Austin PC, Tu JV, et al. Canadian cardiovascular outcomes research. Community factors, hospital characteristics and inter-regional outcome variations following acute myocardial infarction in Canada. Can J Cardiol. 2005;21:247-255.
11. Bertomeu V, Cequier A, Bernal JL, et al. In-hospital mortality due to acute myocardial infarction. Relevance of type of hospital and care provided. RECALCAR study. Rev Esp Cardiol. 2013:66:935-942.
12. Moreno R, Ojeda S, Romaguera R, et al. Actualización de las recomendaciones sobre requisitos y equipamiento en cardiología intervencionista. Documento de consenso de la Asociación de Cardiología Intervencionista y la Asociación de Cardiopatía Isquémica y Cuidados Agudos Cardiovasculares de la Sociedad Española de Cardiología y la Asociación Española de Enfermería en Cardiología. REC Interv Cardiol. 2021;3:33-44.
13. Carrillo X, Fernandez-Nofrerias E, Rodriguez-Leor O, et al. Early ST elevation myocardial infarction in on-capable percutaneous coronary intervention centres: in situ fibrinolysis vs. percutaneous coronary intervention transfer. Eur Heart J. 2016;37:1034-1040.
14. Terkelsen CJ, Sorensen JT, Maeng M, et al. System delay and mortality among patients with STEMI treated with primary percutaneous coronary intervention. JAMA. 2010;304:763-771.
15. Rodríguez-Leor O, Fernández-Nofrerías E, Mauri F, et al. Analysis of reperfusion delay in patients with acute myocardial infarction treated with primary angioplasty based on first medical contact and time of presentation. Rev Esp Cardiol. 2011;64:476-483.
16. Rodríguez-Leor O, Fernández-Nofrerías E, Mauri J, et al. Integration of a local into regional primary angioplasty action plan (the Catalan Codi Infart network) reduces time to reperfusion. Int J Cardiol. 2013;168:4354-4357.
17. Bagai A, Jollis JG, Dauerman HL, et al. Emergency department bypass for ST-segment-elevation myocardial infarction patients identified with a prehospital electrocardiogram: a report from the American Heart Association Mission: Lifeline program. Circulation. 2013;128:352-359.
18. Cequier A, Ariza-Sole A, Elola FJ, et al. Impacto en la mortalidad de diferentes sistemas de asistencia en red para el tratamiento del infarto agudo de miocardio con elevación del segmento ST. La experiencia de España. Rev Esp Cardiol. 2017;70:155-161.
19. Sánchez-Salado JC, Burgos V, Ariza-Solé A, et al. Trends in cardiogenic shock management and prognostic impact of type of treating center. Rev Esp Cardiol. 2020;73:546-553.
20. Rodríguez-Leor O, Cid-Álvarez AB, Pérez de Prado A, et al. Impact of COVID-19 on ST-segment elevation myocardial infarction care. The Spanish experience. Rev Esp Cardiol. 2020;73:994-1002.
ABSTRACT
Introduction and objectives: The role of inflammation in the pathogenesis of coronary artery disease, and that resulting from percutaneous coronary intervention (PCI) is increasingly recognized, yet the effect of colchicine in attenuating peri-PCI inflammation remains unknown. This meta-analysis investigated the efficacy of colchicine in patients undergoing PCI for secondary prevention of coronary artery disease.
Methods: The Web of Science, PubMed, Ovid MEDLINE, Embase, Cochrane Central Register of Controlled Trials and ClinicalTrials.gov databases were searched. Data on studies assessing the efficacy profile of colchicine in patients undergoing PCI were pooled using a random-effects model.
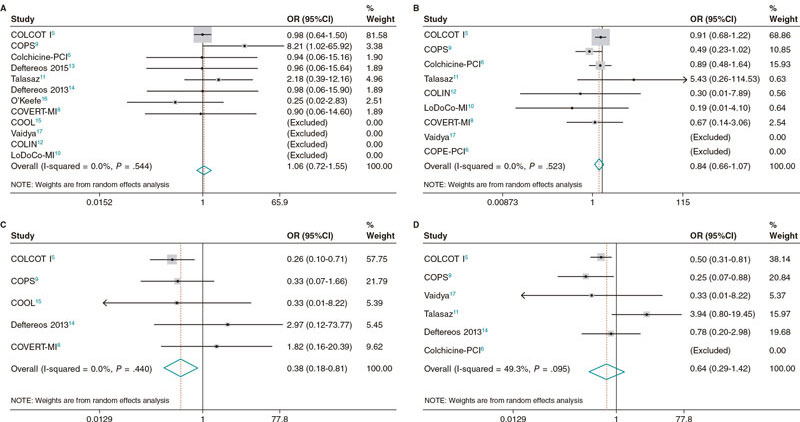
Results: In 13 studies of 7414 patients, no differences were observed between patients treated with colchicine compared to those without for all-cause mortality (OR, 1.1; 95%CI, 0.72-1.56; I2 = 0%), cardiovascular mortality (OR, 0.98; 95%CI, 0.42-2.28; I2 = 14.2%), myocardial infarction (OR, 0.84; 95%CI, 0.65-1.08; I2 = 1.4%) or coronary revascularization (OR, 0.64; 95%CI, 0.28-1.42; I2 = 49.3%). However, patients treated with colchicine had a lower risk of stroke (OR, 0.33; 95%CI, 0.15-0.72; I2 = 0%).
Conclusions: Adding colchicine to standard medical therapy in patients undergoing PCI did not decrease all-cause mortality, cardiovascular mortality or urgent revascularization. However, it showed a trend towards a lower risk of myocardial infarction and a significantly lower risk of stroke.
Keywords: Coronary artery disease. Percutaneous coronary intervention. Inflammation. Colchicine.
RESUMEN
Introducción y objetivos: La importancia de la inflamación en la patogénesis de la enfermedad coronaria, así como tras la angioplastia percutánea, es un fenómeno reconocido. Sin embargo, el efecto de la colchicina para atenuar la inflamación tras la intervención coronaria percutánea se desconoce. Este metanálisis investigó la eficacia de la colchicina en pacientes que se sometieron a intervención coronaria percutánea con el objetivo de prevención secundaria
Métodos: Se revisaron las bases de datos Web of Science, PubMed, OVID MEDLINE, Embase, Cochrane Central Register of Controlled Trials y ClinicalTrials.gov, y se analizaron los datos de los estudios que investigaban la eficacia de la colchicina en pacientes que se sometieron a angioplastia coronaria percutánea, usando un modelo de efectos aleatorios.
Resultados: En 13 estudios, que incluyeron un total de 7.414 pacientes, no se observó ninguna diferencia entre los tratados con colchicina y los no tratados con colchicina en cuanto a mortalidad por cualquier causa (OR = 1,1; IC95%, 0,72-1,56; I2 = 0%), mortalidad por causa cardiovascular (OR = 0,98; IC95%, 0,42-2,28; I2 = 14,2%), infarto de miocardio (OR = 0,84; IC95%, 0,65-1,08; I2 = 1,4%) y revascularización coronaria (OR = 0,64; IC95%, 0,28-1,42; I2 = 49,3%). Sin embargo, los pacientes tratados con colchicina mostraron un menor riesgo de accidente vascular cerebral (OR = 0,33; IC95%, 0,15-0,72; I2 = 0%).
Conclusiones: Agregar colchicina a la terapia medica estándar en pacientes sometidos a angioplastia coronaria percutánea no modificó la mortalidad por cualquier causa, la mortalidad por causa cardiovascular ni la revascularización coronaria, pero si mostró una tendencia a un menor riesgo de infarto de miocardio y un menor riesgo significativo de accidente vascular cerebral.
Palabras clave: Enfermedad coronaria. Angioplastia percutánea. Inflamación. Colchicina.
Abbreviations
ACS: acute coronary syndrome. MI: myocardial infarction. NSTEMI: non-ST-elevation acute myocardial infarction. PCI: percutaneous coronary intervention. RCT: randomized controlled trial.
INTRODUCTION
Despite increasingly effective primary and secondary preventive treatments, coronary artery-related events continue to be the leading cause of morbidity and mortality worldwide.1,2 Lifestyle changes (eg, weight loss, low-salt diet, smoking cessation), medical therapy (eg, anti-hypertensive, lipid-lowering, glucose-lowering, and antithrombotic regimens) in addition to coronary revascularization via percutaneous coronary intervention (PCI) and coronary artery bypass graft (CABG) constitute the multifaceted approach of this disease. Yet despite the advances made in this multimodality approach, cardiovascular morbidity and mortality remain high.
More recently, the central role played by inflammation in the pathogenesis of coronary artery disease from atherosclerotic plaque formation to acute coronary syndrome (ACS), and PCI itself have gained important recognition. Colchicine, an anti-inflammatory agent indicated for multiple inflammatory conditions including pericarditis, gout, and familial Mediterranean fever, has gained attention as a potential attenuator of atherosclerotic inflammation. Acting via the inhibition of tubulin polymerization and eventually blunting immune cell activation and inflammatory response,3,4 recent evidence suggests a benefit of colchicine in the management of the cardiovascular events of patients with clinical signs of coronary artery disease.5 However, its impact among patients in the peri-PCI period remain controversial.
Recent trials have begun exploring the effects of colchicine in the PCI setting, albeit with mixed results. In the Colchicine-PCI trial of patients with non-ST-segment elevation acute coronary syndrome, the administration of colchicine immediately before and after PCI resulted in lower interleukin-6 and high-sensitivity C-reactive protein (hsCRP) levels at 24 hours, but did not show fewer PCI-related myocardial injuries.6 This trial was followed by COPE-PCI that found that when administered 6-to-24 hours before the PCI, colchicine did in fact reduce PCI-related myocardial injuries in a population of patients with stable angina and non-ST-elevation acute myocardial infarction (NSTEMI).7 Nevertheless, the more recent COVERT-MI trial8 found no difference in infarct size or left ventricular remodeling on the cardiac magnetic resonance imaging in patients treated with colchicine compared to those untreated with this agent.
These individual studies may not provide properly powered analyses, particularly in low-rate events such as strokes, on the impact of colchicine regarding secondary prevention in patients in the peri-PCI period, thus prompting the need for a systematic appraisal and meta-analysis of the quality of evidence and treatment effects on major adverse cardiovascular events.
METHODS
Protocol
The search process of this meta-analysis was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and is registered with PROSPERO (CRD42021247704). The meta-analysis did not require specific institutional review board approval since it utilized results published in former studies. All relevant information can be found in the trials included. The corresponding author had full access to all the data and final responsibility on the decision to submit the manuscript for publication. The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Search strategy
We performed a comprehensive literature search of all published studies—retrospective, observational, and randomized controlled trials—available on Web of Science, Embase, PubMed, Ovid MEDLINE, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov (inception through August 23, 2021, without language restrictions. Case reports, letters to the editor, reviews, and book chapters were not included in this meta-analysis. The keywords used in the search were ‘colchicine,’ ‘coronary artery disease,’ ‘coronary heart disease,’ ‘angina,’ ‘myocardial infarction,’ ‘acute myocardial infarction,’ ‘myocardial ischemia,’ ‘acute coronary syndrome,’ ‘ischemic heart disease,’ ‘percutaneous coronary intervention,’ ‘percutaneous transluminal angioplasty,’ ‘percutaneous coronary revascularization,’ and ‘myocardial revascularization’ including their subheadings, MeSH terms, and all synonyms. References for each of the studies se lected were also screened (the detailed search strategy can be found on the supplementary data). The search process was reported according to the Preferred Reported Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Selection criteria
Studies were eligible if they included any the following criteria: a) compared the efficacy of colchicine treatment, at any dose and for any duration, to standard medical treatment with or without placebo; b) included populations of patients treated with PCI regardless of the indication; and c) reported, at least, 1 of the following cardiovascular outcomes: all-cause mortality, cardiovascular mortality, myocardial infarction (MI), stroke or urgent coronary revascularization. Study selection was conducted by 2 independent reviewers (C.E. Soria Jiménez, and J. Chang) first by screening titles and abstracts and then by reviewing full texts and their corresponding references. In case of disagreement over eligibility, a third reviewer (H.M. García-García) assessed discrepancy, and decisions were reached by consensus.
Data collection and study endpoints
Data on study characteristics, patient characteristics, and endpoint event rates were independently drawn and organized into a structured dataset by 2 reviewers (C.E. Soria Jiménez, and F. Hayat), and then compared. All discrepancies resulted in the re-evaluation of primary data and involvement of a third reviewer (H.M. García García). Disagreements were resolved by consensus.
Endpoints
The prespecified primary endpoint was all-cause mortality. Secondary clinical endpoints were cardiovascular mortality, MI, stroke, and any revascularization. Each endpoint was assessed according to the definitions reported in the original study protocols (summarized on table 1 of the supplementary data).
Risk of bias
The risk of bias in each study was assessed using the revised Cochrane Risk of Bias tool (RoB 2.0) for randomized controlled trials (RCTs), and the Risk of Bias in Non-randomized Studies of Interventions assessment Tool from the Cochrane handbook (ROBINS-I) for observational studies. Two investigators (C.E. Soria Jiménez, and J. Sanz Sánchez) independently assessed 5 domains of bias in RCTs: (1) randomization process, (2) deviations from intended procedures, (3) missing outcome data, (4) outcome measurement, and (5) selection of results reported. The same investigators independently assessed 7 domains of bias in observational studies: (1) confounding, (2) selection of participants, (3) classification of procedures, (4) deviations from intended interventions, (5) missing outcome data, (6) outcome measurement, and (7) selection of results reported (table 2 and 3 of the supplementary data).
Statistical analysis
Odds ratios (OR) and 95% confidence intervals (95%CI) were estimated using the DerSimonian and Laird random-effects model with the estimate of heterogeneity taken from the Mantel-Haenszel method. The presence of heterogeneity among the studies was evaluated using the Cochran Q test referred to chi-square distribution (P ≤ .10 was considered statistically significant) plus the I2 test to assess inconsistencies. Values of 0% indicated no observed heterogeneity, and values ≤ 25%, ≤ 50%, and > 50% indicated low, moderate, and high heterogeneity, respectively. The presence of publication bias was investigated using Harbord test and visual estimation with funnel plots. We conducted a leave-one-out sensitivity analysis for all outcomes by iteratively removing 1 study at a time to confirm that our findings were not driven by any single study. To account for the different follow-up durations across the studies, another sensitivity analysis was conducted using a Poisson regression model with random intervention effects to calculate the means of inverse-variance weighting of trial-specific log stratified incidence rate ratios. Results were shown as incidence rate ratios, which are exponential coefficients of the regression model.
A meta-regression analysis was conducted using the empirical Bayesian method to estimate the between-study variance tau-squared to assess the effect of colchicine dosage, follow-up duration, percentage of patients with ACS, and percentage of those with diabetes mellitus on treatment effects on the primary endpoint.
Two-tailed P values < .05 were considered statistically significant. Statistical analyses were conducted using the Stata software version 13.1 (StataCorp LP, College Station, United States).
RESULTS
Search results
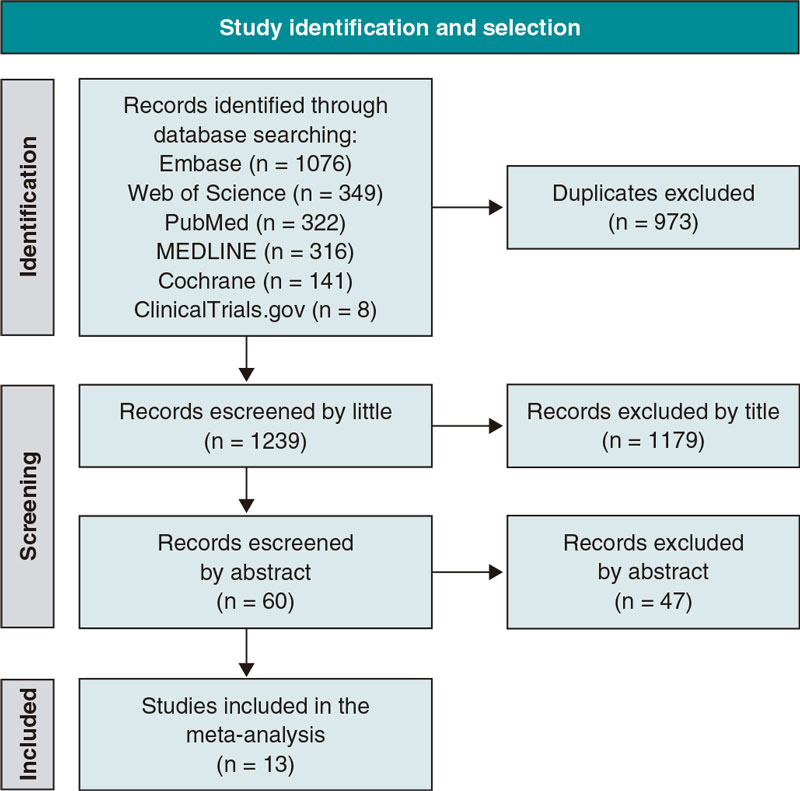
Figure 1 shows the PRISMA study search and selection process. Out of a total of 1239 unique reports, 12 RCTs5-16 and 1 observational study17 were identified and included in this analysis. The corresponding author of the COOL trial15 was contacted regarding data from a number of patients treated with PCI; 58 out of a total of 80 patients evaluated (72.5%) underwent PCI. The study ultimately met the inclusion criteria and was included in our analysis. The main features of the studies included are shown on table 1. Data on the outcomes, mortality, MI, stroke, and urgent revascularization were reported in 12, 9, 5, and 6 trials, respectively. A total of 3741 and 3673 patients treated with and without colchicine were included (for a total of 7414 patients). Time elapsed from the PCI to the start of colchicine went from immediately before PCI to 13.5 days later as shown on table 1.
Figure 1. Preferred Reported Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart of database search results and study selection.
Table 1. Characteristics of trials selected
| Trial/Author | Year | Study design | Multicenter | Patients (n) | Population | Colchicine dose and duration | Time elapsed from PCI to start of colchicine | Follow-up |
|---|---|---|---|---|---|---|---|---|
| COVERT-MI8 | 2021 | RCT | Yes | 192 | Adults with a first-time STEMI referred for primary or bailout PCI | 2 mg oral loading dose followed by daily oral 0.5 mg twice daily for 5 days | Loading dose immediately before PCI; if not possible, immediately after PCI | 3 months |
| COPE-PCI7 | 2021 | RCT | No | 75 | Adults with stable angina or NSTEMI undergoing angiography and PCI | 1 mg followed by 0.5 mg 1 h later, 6 hrs to 24 hrs pre-PCI | 6 hrs to 24 hrs before coronary angiogram | 1 day |
| Colchicine-PCI6 | 2020 | RCT | No | 400 | Adults with suspected ischemic heart disease or ACS referred for angiography with possible PCI | 1.2 mg 1 h to 2 h pre-angiography, 0.6 mg 1 h later or immediately after the procedure if rushed for emergency angiography | 1 h to 2 h before coronary angiography | 1 month |
| COPS9 | 2020 | RCT | Yes | 795 | Adults presenting with ACS and evidence of CAD treated with angiography and managed with PCI or medical therapy | 0.5 mg twice daily for 1 month, then 0.5 mg daily for 11 months | Immediately after PCI and randomization | 13.2 months |
| LoDoCo-MI10 | 2019 | RCT | No | 237 | Adults who sustained a type 1 MI within the past 7 days | 0.5 mg daily for 30 days | 1.5 days following the index MI | 1 month |
| Talasaz11 | 2019 | RCT | No | 196 | Adults presenting with STEMI undergoing PCI | NA | NA | 1 month |
| COLCOT I5 | 2019 | RCT | Yes | 4745 | Adults with MI within the past 30 days who had completed some percutaneous revascularization | 0.5 mg once daily for, at least, 2 years | 13.5 days | 42 months |
| Vaidya17 | 2018 | Observational | No | 80 | Adults who presented with ACS < 1 month prior and underwent invasive coronary angiography and revascularization if indicated | 0.5 mg once daily for 1 year | NA (< 1 month from ACS per inclusion criteria) | 12.6 months |
| COLIN12 | 2017 | RCT (Open-label) | No | 44 | Adults admitted for STEMI with occlusion of 1 of the main coronary arteries treated with PCI | 1 mg once daily for 1 month | On the first day of the AMI | 1 month |
| Deftereos 201513 | 2015 | RCT (Pilot) | Yes | 151 | Adults presenting with STEMI of ≤ 12-hour evolution from pain onset treated with PCI | 2 mg loading dose, 0.5 mg twice daily for 5 days | Immediately after completion of diagnostic coronary angiography | 5 days |
| Deftereos 201314 | 2013 | RCT | No | 222 | Adults with diabetes, aged 40-80 treated with PCI with bare metal stent | 0.5 mg twice daily for 6 months | Within 24 hrs of index PCI | 6 months |
| COOL15 | 2012 | RCT | No | 80 | Adults with ACS or acute ischemic stroke | 1 mg once daily for 30 days | Immediately after randomization | 1 month |
| O’Keefe16 | 1992 | RCT | No | 197 | Adults who underwent elective angioplasty (single or multivessel, new or restenosed lesions) for silent, stable or unstable angina; CABG | 0.6 mg twice daily for 6 months | Somewhere between 12 hrs before and 24 hrs after balloon angioplasty | 6 months |
|
ACS, acute coronary intervention; CABG, coronary artery bypass graft; CAD, coronary artery disease; MI, myocardial infarction; NA, not available; NSTEMI, non-ST-elevation acute myocardial infarction; PCI, percutaneous coronary intervention; RCT, randomized controlled trial; STEMI, ST-segment elevation myocardial infarction. |
||||||||
Baseline characteristics
Main baseline characteristics of the patients included are shown on table 2. Most patients were men with a mean age of 60 years, had ACS, and underwent revascularization with drug-eluting stents.
Table 2. Baseline characteristics of patients from each trial
| Trial/Author | Mean Age | Men (%) | ACS (%) | DES (%) | HTN (%) | DM2 (%) | HLD (%) | Previous MI (%) | Previous PCI (%) | Previous CABG (%) | Underwent PCI (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| COVERT-MI8 | 60 | 80.3 | 100 | 95.7 | 30.8 | 13.1 | 33.1 | 0 | 0 | 0 | 93 |
| COPE-PCI7 | 64.7 | 71.5 | 58.7 | 97.0 | 54.5 | 22.9 | 63.5 | 17.5 | 16.0 | NA | 100 |
| Colchicine-PCI6 | 66.3 | 93.5 | 49.5 | NA | 91.7 | 57.8 | 88.8 | 25.8 | 37.6 | NA | 100 |
| COPS9 | 59.9 | 79.5 | 100.0 | NA | 50.5 | 19.0 | 46.0 | 15.0 | 13.0 | 4.5 | 88 |
| LoDoCo-MI10 | 61.0 | 77.0 | 100.0 | NA | 47.5 | 22.0 | NA | 15.0 | 11.5 | NA | 90 |
| Talasaz11 | NA | NA | 100.0 | NA | NA | NA | NA | NA | NA | NA | 100 |
| COLCOT I5 | 60.6 | 80.9 | 100.0 | NA | 51.1 | 20.2 | NA | 16.2 | 16.9 | 3.2 | 93 |
| Vaidya17 | 57.4 | 77.5 | 100.0 | NA | 53.8 | 31.3 | 85.0 | 51.3 | 63.8 | NA | 77.5 |
| COLIN12 | 59.9 | 79.4 | 100.0 | NA | 43.4 | 13.7 | 36.5 | NA | 4.6 | 2.4 | 100 |
| Deftereos 201513 | 58.0 | 69.0 | 100.0 | NA | 39.5 | 21.5 | 52.0 | 0.0 | NA | NA | 100 |
| Deftereos 201314 | 63.6 | 65.5 | 31.0 | 0 | 48.5 | 100.0 | NA | NA | NA | NA | 100 |
| COOL15 | 57.2 | 88.8 | 91.3 | NA | 42.5 | 16.3 | 47.5 | 17.5 | 0 | NA | 73 |
| O’Keefe16 | 60.5 | 86.0 | 39.5 | 0 | NA | 12.0 | NA | NA | NA | 25.5 | 100 |
|
ACS, acute coronary syndrome; CABG, coronary artery bypass graft; DES, drug-eluting stent; DM2, diabetes mellitus type 2; HLD, hyperlipidemia; HTN, hypertension; MI, myocardial infarction; NA, not available; PCI, percutaneous intervention. |
|||||||||||
Publication bias and asymmetry
Funnel-plot distributions of pre-specified outcomes indicate absence of publication bias for all the outcomes (figures 1 to 5 of the supplementary data).
Risk of bias assessment
Table 2 and table 3 of the supplementary data summarize the results of the risk of bias assessment. A total of 11 trials were ranked as trials with a low overall risk of bias, 1 presented some concerns while another one was ranked as a trial with a high overall risk of bias.
Outcomes
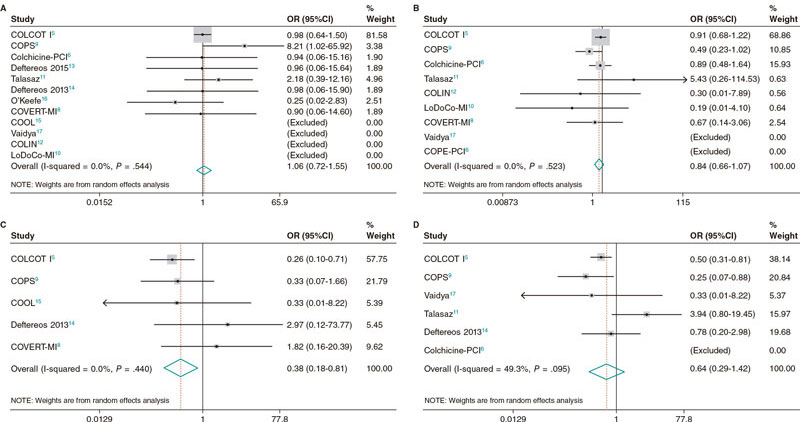
No differences were seen between patients treated with colchicine and those treated without it or placebo regarding all-cause mortality (OR, 1.06; 95%CI, 0.72-1.55; I2 = 0%), cardiovascular mortality (OR, 0.98; 95%CI, 0.42-2.28; I2 = 14.2%) or coronary revascularization (OR, 0.64; 95%CI, 0.29-1.42; I2 = 49.3%). However, patients treated with colchicine had a lower risk of stroke (OR, 0.38; 95%CI, 0.18-0.81; I2 = 0%), and a trend towards a lower risk of MI (OR, 0.84; 95%CI, 0.66-1.07; I2 = 0%) (figure 2).
Figure 2. Forrest plot analyses for the main outcomes of death (A), myocardial infarction (B), stroke (C), and revascularization (D). 95%CI, confidence interval; OR, odds ratio.
Sensitivity analyses
In the leave-one-out sensitivity analysis, results were consistent with the primary analysis (tables 4 to 8 of the supplementary data). Similarly, in a sensitivity analysis on the use of estimated incidence rate ratios to account for different lengths of follow-up, findings remained unchanged (table 9 of the supplementary data).
When the risk ratios with random-effects models were estimated, findings remained consistent with the main analysis for all endpoints (table 10 of the supplementary data). Random effect meta-regression analyses found no significant impact of colchicine dosage (P = .33), follow-up duration (P = .88), percentage of patients with ACS (P = .37) or percentage of patients with diabetes mellitus (P = .96) on treatment effect regarding the primary endpoint (table 11 of the supplementary data).
DISCUSSION
This meta-analysis included 7414 patients across 12 RCTs and 1 observational study. It showed some clinical benefits on cardiovascular events with the addition of colchicine to standard medical therapy in patients undergoing PCI. Specifically, we found that the addition of colchicine compared to no colchicine or placebo reduced the risk of stroke showing a trend towards a lower risk of MI both with no observed heterogeneity. Additionally, we observed no differences in all-cause mortality, cardiovascular mortality or coronary revascularization. Significantly, colchicine dosage, follow-up duration, percentage of patients with ACS or diabetes mellitus showed no impact on treatment effect (see PRISMA checklist on table 12 of the supplementary data).
Our outcomes regarding all-cause and cardiovascular mortality are consistent with a prior meta-analysis of 5 RCTs conducted by Fu et al.,18 that also found no significant reduction of mortality, MI, serious adverse events, and restenosis. One explanation for the lack of mortality benefit of both trials may be that although mortality rate was generally low and differences were largely not statistically significant in many of these trials, follow-up duration was generally short (< 30 days) in most studies, and it is possible that higher event rates may be seen with longer follow-up data. We should mention that the meta-analysis conducted by Fu et al.18 included 1 RCT of patients treated with CABG, not PCI. It is possible that the inflammatory profiles of this cohort of patients differ from those treated with PCI (eg, multivessel coronary artery disease, longer postoperative recovery, and higher risk of postoperative complications). As a matter of fact, this mixed population may have led to the lack of reduction seen in the overall rate of MI, serious adverse events, and restenosis. Similarly, a prior meta-analysis conducted by Fiolet et al.19 demonstrated that the addition of colchicine to standard medical therapy in patients with acute and chronic coronary syndromes reduced the risk of the primary endpoint significantly (a composite of MI, stroke, and cardiovascular mortality), and the individual endpoint of MI, stroke, and coronary revascularization with no differences whatsoever on all-cause or cardiovascular mortality. Our results demonstrating a lower risk of stroke and a trend towards a lower risk of MI are more consistent with this meta-analysis. A key difference among the different meta-analyses is the population of patients. Fiolet et al.19 included the LoDoCo20 and LoDoCo221 trials whose inclusion criteria were patients with chronic coronary disease and clinical stability for over 6 months. This amounted to > 50% of patients analyzed who were not in the peri-PCI period and likely had a different inflammatory profile at the time of colchicine administration. These 2 trials also had longer follow-ups (36 and 29 months, respectively) potentially allowing for more time to capture outcome differences like MI and urgent revascularization between the different treatment groups. In contrast, our meta-analysis only focused on patients in the peri-PCI as conducted by Fu et al.18 and expanded the total number of studies analyzed to 12 RCTs and 1 observational study. As far as we know, our study is the largest meta-analysis ever conducted to this date to assess the effects of colchicine on the clinical outcomes of patients in the peri-PCI period.
Alkouli et al.22 reported that the adjusted rate of ischemic stroke increased for patients treated with PCI due to ST-segment elevation myocardial infarction (STEMI) (0.6% to 0.96%), NSTEMI (0.5% to 0.6%), and unstable angina or stable ischemic heart disease (UA/SIHD, 0.3% to 0.72%). In turn, in-hospital mortality was higher (23.5% vs 11.0%, 9.5% vs 2.8%, and 11.5% vs 2.4% for STEMI, NSTEMI, and UA/SIHD cohorts, respectively), and post-PCI stroke was associated with a > 2-fold increase in LoS, a > 3-fold increase in non-home discharges, and a > 60% increase in cost. Given the increasing complexity of patients treated as well as the PCI techniques utilized over the past decade, effective preventive strategies and treatments are needed, and herein lies the opportunity for other anti-inflammatory drugs such as colchicine to further mitigate the morbidity and mortality of patients with post-PCI stroke. In the acute phase of MI, activated inflammasomes mount an intense inflammatory response.23 There is also endothelial damage after PCI, which may result in atherosclerotic plaque destabilization with subsequent thromboembolism causing cerebrovascular events.24 Colchicine may play a role preventing stroke by helping stabilize atherosclerotic plaques in patients undergoing PCI, though this effect may not be robust enough to overcome the direct endothelial injury present at the time of PCI.
Colchicine is a widely available drug with known anti-inflammatory properties. Its mechanism of action is yet to be fully elucidated but has been shown to work partly via the inhibition of NLRP3 (nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing protein 3) inflammasome, which ultimately downregulates interleukin-1B and interleukin-6, 2 known inflammatory mediators.23-27 It also causes microtubule disruption and decreased neutrophil activation and extravasation. Since elevated levels of inflammatory biomarkers are an independent predictor of major adverse cardiovascular events28-31 our results show that colchicine joining the current medical therapy is a potential addition to further attenuate inflammation regarding the secondary prevention of cardiovascular disease in patients undergoing PCI.
Some limitations of our study include the use of aggregate study-level data as opposed to patient-level data. While this limits subgroup analyses, the overall conclusions would remain the same. There was also a small percentage of patients in each of the studies analyzed who did not undergo PCI, which poses some limitations on the overall effects on a PCI population. However, in all studies, the vast majority of patients eventually underwent this procedure. Similarly, the LoDoCo221 trial enrolled patients who underwent PCI but was ultimately excluded from this analysis as patients required a period of clinical stability 6 months after PCI before starting colchicine therapy. A 6-month gap from PCI to colchicine initiation did not fit in with our period of interest (the peri-PCI period). The study conducted by O’keefe16 was completed in an era of balloon angioplasty, and colchicine treatment in this setting may not be comparable to patients who underwent PCI in the era of statins, modern stents, and antiplatelet agents. Additionally, most patients from our study underwent PCI due to the presentation of ACS, yet there were other clinical presentations including stable ischemic heart disease and unstable angina, and yet others that specifically excluded patients with acute MI. Given the different clinical status at presentation for PCI, it’s likely that the inflammatory profile of these different populations of patients also varied resulting in different clinical outcomes. Nevertheless, despite variation in the inclusion and exclusion criteria, outcome definitions, and colchicine dose and duration, this did not introduce heterogeneity into our results.
CONCLUSIONS
In patients undergoing PCI, the addition of colchicine to optimal medical therapy resulted in a significant reduction of strokes, and a trend towards a lower risk of MI. However, this did not result in lower all-cause and cardiovascular mortality rates, and urgent revascularization.
FUNDING
This research did not receive any specific grants from public, private or non-profit sectors.
AUTHORS’ CONTRIBUTIONS
M.B. Levine was involved in data curation and research. F. Hayat, and J. Chang were involved in data curation and research, as well as in drafting, editing, and reviewing the early draft of the manuscript. C.E. Soria Jiménez, J. Sanz Sánchez, and H. García-García were involved in project conceptualization, data curation, formal data analysis and investigation, methodology, project administration, resources, validation, visualization, as well as drafting, editing, and reviewing all manuscript drafts and its final version.
CONFLICTS OF INTEREST
H.M. García-García declared institutional grant support from Biotronik, Boston Scientific, Medtronic, Abbott, Neovasc, Shockwave, Phillips, and Corflow. The remaining authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Inflammation plays a central role in the pathogenesis of coronary artery disease, and it’s involved in percutaneous coronary interventions. Colchicine is a powerful anti-inflammatory drug. Its effect, however, attenuating peri-PCI inflammation remains unknown.
WHAT DOES THIS STUDY ADD?
- In this meta-analysis of 12 RCTs and 1 observational study, the addition of colchicine to patients undergoing PCI resulted in a lower risk of stroke. Other major adverse cardiovascular events did not show any significant differences.
REFERENCES
1. Fox KA, Poole-Wilson P, Clayton TC, et al. 5-year outcome of an interventional strategy in non-ST-elevation acute coronary syndrome: the British Heart Foundation RITA 3 randomised trial. Lancet. 2005;366:914-920.
2. Boden WE, O’Rourke RA, Teo KK, et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007;356:1503-1516.
3. Deftereos S, Giannopoulos G, Papoutsidakis N, et al. Colchicine and the heart: pushing the envelope. J Am Coll Cardiol. 2013;62:1817-1825.
4. Nidorf SM, Eikelboom JW, Thompson PL. Colchicine for secondary prevention of cardiovascular disease. Curr Atheroscler Rep. 2014;16:391.
5. Tardif JC, Kouz S, Waters DD, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019;381:2497-2505.
6. Shah B, Pillinger M, Zhong H, et al. Effects of Acute Colchicine Administration Prior to Percutaneous Coronary Intervention: COLCHICINE-PCI Randomized Trial. Circ Cardiovasc Interv. 2020;13:e008717.
7. Cole J, Htun N, Lew R, Freilich M, Quinn S, Layland J. Colchicine to Prevent Periprocedural Myocardial Injury in Percutaneous Coronary Intervention: The COPE-PCI Pilot Trial. Circ Cardiovasc Interv. 2021;14:e009992.
8. Mewton N, Roubille F, Bresson D, et al. Effect of Colchicine on Myocardial Injury in Acute Myocardial Infarction. Circulation. 2021;144:859-869.
9. Tong DC, Quinn S, Nasis A, et al. Colchicine in Patients With Acute Coronary Syndrome: The Australian COPS Randomized Clinical Trial. Circulation. 2020;142:1890-1900.
10. Hennessy T, Soh L, Bowman M, et al. The Low Dose Colchicine after Myocardial Infarction (LoDoCo-MI) study: A pilot randomized placebo controlled trial of colchicine following acute myocardial infarction. Am Heart J. 2019;215:62-69.
11. Talasaz AH, Jenab Y, Hosseini SH. P461. Colchicine before percutaneous coronary intervention in acute myocardial infarction. Eur Heart J. 2019(40):Suppl 1;ehz745.0994.
12. Akodad M, Lattuca B, Nagot N, et al. COLIN trial: Value of colchicine in the treatment of patients with acute myocardial infarction and inflammatory response. Arch Cardiovasc Dis. 2017;110:395-402.
13. Deftereos S, Giannopoulos G, Angelidis C, et al. Anti-Inflammatory Treatment With Colchicine in Acute Myocardial Infarction: A Pilot Study. Circulation. 2015;132:1395-1403.
14. Deftereos S, Giannopoulos G, Raisakis K, et al. Colchicine treatment for the prevention of bare-metal stent restenosis in diabetic patients. J Am Coll Cardiol. 2013;61:1679-1685.
15. Raju NC, Yi Q, Nidorf M, Fagel ND, Hiralal R, Eikelboom JW. Effect of colchicine compared with placebo on high sensitivity C-reactive protein in patients with acute coronary syndrome or acute stroke: a pilot randomized controlled trial. J Thromb Thrombolysis. 2012;33:88-94.
16. O’Keefe JH Jr, McCallister BD, Bateman TM, Kuhnlein DL, Ligon RW, Hartzler GO. Ineffectiveness of colchicine for the prevention of restenosis after coronary angioplasty. J Am Coll Cardiol. 1992;19:1597-1600.
17. Vaidya K, Arnott C, Martínez GJ, et al. Colchicine Therapy and Plaque Stabilization in Patients With Acute Coronary Syndrome: A CT Coronary Angiography Study. JACC Cardiovasc Imaging. 2018;11:305-316.
18. Fu C, Wang B. Colchicine administration for percutaneous coronary intervention: A meta-analysis of randomized controlled trials. Am J Emerg Med. 2021;46:121-125.
19. Fiolet A, Opstal T, Mosterd A, et al. Efficacy and Safety of Low-Dose Colchicine in Patients with Coronary Disease: A Systematic Review and Meta-Analysis of Randomized Trials. Eur Heart J. 2021;00:1-11.
20. Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL. Low-dose colchicine for secondary prevention of cardiovascular disease. J Am Coll Cardiol. 2013;61:404-410.
21. Nidorf SM, Fiolet ATL, Mosterd A, et al. Colchicine in Patients with Chronic Coronary Disease. N Engl J Med. 2020;383(19):1838-1847.
22. Alkhouli M, Alqahtani F, Tarabishy A, Sandhu G, Rihal CS. Incidence, Predictors, and Outcomes of Acute Ischemic Stroke Following Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2019;12:1497-1506.
23. Chen B, Frangogiannis NG. Immune cells in repair of the infarcted myocardium. Microcirculation. 2017;e12305.
24. de Winter RJ, Heyde GS, Koch KT, et al. The prognostic value of pre-procedural plasma C-reactive protein in patients undergoing elective coronary angioplasty. Eur Heart J. 2002;23:960-966.
25. Rajamäki K, Lappalainen J, Oörni K, et al. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS One. 2010;5:e11765.
26. Paschke S, Weidner AF, Paust T, Marti O, Beil M, Ben-Chetrit E. Technical advance: Inhibition of neutrophil chemotaxis by colchicine is modulated through viscoelastic properties of subcellular compartments. J Leukoc Biol. 2013;94:1091-1096.
27. Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237-241.
28. Buffon A, Liuzzo G, Biasucci LM, et al. Preprocedural serum levels of C-reactive protein predict early complications and late restenosis after coronary angioplasty. J Am Coll Cardiol. 1999;34:1512-1521.
29. Kwaijtaal M, van Diest R, Bär FW, et al. Inflammatory markers predict late cardiac events in patients who are exhausted after percutaneous coronary intervention. Atherosclerosis. 2005;182:341-348.
30. Patti G, Di Sciascio G, D’Ambrosio A, Dicuonzo G, Abbate A, Dobrina A. Prognostic value of interleukin-1 receptor antagonist in patients undergoing percutaneous coronary intervention. Am J Cardiol. 2002;89:372-376.
31. Walter DH, Fichtlscherer S, Sellwig M, Auch-Schwelk W, Schächinger V, Zeiher AM. Preprocedural C-reactive protein levels and cardiovascular events after coronary stent implantation. J Am Coll Cardiol. 2001;37:839-846.
ABSTRACT
Introduction and objectives: Delayed vascular healing may induce late stent thrombosis. Optical coherence tomography (OCT) is useful to evaluate endothelial coverage. The objective of this study was to compare stent coverage and apposition in non-complex coronary artery lesions treated with durable polymer-coated everolimus-eluting stents (durable-polymer EES) vs biodegradable polymer-coated everolimus-eluting stents (biodegradable-polymer EES) vs polymer-free biolimus-eluting stents (BES) 1 and 6 months after stent implantation.
Methods: Prospective, multicenter, non-randomized study that compared the 3 types of DES. Follow-up angiography and OCT were performed 1 and 6 months later. The primary endpoint was the rate of uncovered struts as assessed by the OCT at 1 month.
Results: A total of 104 patients with de novo non-complex coronary artery lesions were enrolled. A total of 44 patients were treated with polymer-free BES, 35 with biodegradable-polymer EES, and 25 with durable-polymer EES. A high rate of uncovered struts was found at 1 month with no significant differences reported among the stents (80.2%, polymer-free BES; 88.1%, biodegradable-polymer EES; 82.5%, durable-polymer EES; P = .209). Coverage improved after 6 months in the 3 groups without significant differences being reported (97%, 95%, and 93.7%, respectively; P = .172).
Conclusions: In patients with de novo non-complex coronary artery lesions treated with durable vs biodegradable vs polymer-free DES, strut coverage and apposition were suboptimal at 1 month with significant improvement at 6 months.
Keywords: Optical coherence tomography. Drug-eluting stents. Endothelization. Apposition. Restenosis.
RESUMEN
Introducción y objetivos: A pesar del desarrollo de los stents farmacoactivos, el retraso en la endotelización puede causar trombosis tardía. La tomografía de coherencia óptica puede evaluar la cobertura intimal. El objetivo de este estudio fue comparar la cobertura y la aposición en lesiones coronarias no complejas de 3 tipos de stent: stent de everolimus con polímero persistente, stent de everolimus con polímero bioabsorbible y stent de biolimus sin polímero, a 1 y 6 meses del implante.
Métodos: Se diseñó un estudio prospectivo, multicéntrico, no aleatorizado, que comparó 3 stents farmacoactivos. Se realizaron angiografía y tomografía de coherencia óptica a 1 o 6 meses. El objetivo primario fue comparar la cobertura.
Resultados: Se incluyeron 104 pacientes con lesiones coronarias de novo no complejas. Se implantó stent sin polímero a 44 pacientes, stent con polímero bioabsorbible a 35 pacientes y stent con polímero persistente a 25 pacientes. Al mes, se observó una alta tasa de struts no cubiertos, sin diferencias significativas entre los grupos (80,2% sin polímero, 88,1% con polímero bioabsorbible y 82,5% con polímero persistente; p = 0,209). La cobertura mejoró a los 6 meses en los 3 stents, sin diferencias significativas entre ellos (97, 95 y 93,7%, respectivamente; p = 0,172).
Conclusiones: En los pacientes con lesiones coronarias no complejas tratados con stent con polímero persistente, con polímero bioabsorbible o sin polímero, la cobertura y la aposición fueron subóptimas a 1 mes del implante, con mejoría significativa a los 6 meses.
Palabras clave: Tomografía de coherencia óptica. Stent farmacoactivo. Endotelización. Aposición. Reestenosis.
Abbreviations
DAPT: dual antiplatelet therapy. DES: drug-eluting stents. OCT: optical coherence tomography. PCI: percutaneous coronary intervention. QCA: quantitative coronary angiography.
INTRODUCTION
Uncovered stent struts is one of the key predictors of stent thrombosis,1,2 and dual antiplatelet therapy (DAPT) has shown to reduce its risk.3 However, DAPT increases the risk of hemorrhage, and nearly one third of the patients treated with percutaneous coronary intervention (PCI) are considered at high bleeding risk. Given the desire for earlier discontinuation of DAPT to reduce the risk of bleeding complications, healing of the stents at earlier time points is desirable.
Drug-eluting stents (DES) significantly reduce neointimal hyperplasia and restenosis compared to bare-metal stents (BMS). However, the main concern regarding first-generation DES was late stent thrombosis due to lack of endothelization of stent struts.1 Therefore, a new generation of DES was developed based on improved thinner metal platforms, new drugs (alternative antiproliferative -limus analogues),4-6 and more biocompatible polymers.7 The evolution of DES moved towards DES with biodegradable polymers.8-10 The comparative studies between DES with biodegradable polymers and BMS showed a lower rate of cardiac death, target vessel-related myocardial reinfarction, and revascularization at 1 year.10 Compared to biodegradable polymer DES, those with durable polymer were noninferior regarding acute coronary syndromes with respect to all-cause mortality, nonfatal myocardial infarction, and revascularization.11 Furthermore, the most recent advance to overcome stent thrombosis has been the polymer-free DES. This type of DES was initially designed to reduce the risk of stent thrombosis in patients with high bleeding risk who could only take short courses of DAPT. It was compared to BMS showing a better efficacy and safety profile.13 It has recently been compared to DES in large clinical trials, especially in patients with high bleeding risk and need for shorter DAPT courses. In these studies, the use of polymer-based zotarolimus-eluting stent was noninferior to polymer-free DES14, and non-measurable differences in device-oriented composite primary endpoints were found.15
However, despite these large clinical trials, there is scarce information on the difference between the characteristics of arterial healing among different types of latest generation DES. Optical coherence tomography (OCT) is a widely used high-resolution intracoronary imaging modality to assess vascular response after stent implantation, thus detecting stent strut coverage and its apposition to the vessel wall.16,17 Stent strut coverage as studied by the OCT is considered a valuable surrogate marker for vessel healing after DES implantation.
The objective of this study was to compare durable polymer-coated everolimus-eluting stents (durable-polymer EES) vs biodegradable polymer-coated everolimus-eluting stents (biodegradable-polymer EES) vs polymer-free BES using stent strut coverage as assessed by the optical coherence tomography (OCT) as a surrogate marker to evaluate short-term arterial healing.
METHODS
Patient population and data collection
This was a prospective, multicenter, non-randomized study that compared 3 different types of DES: a) the durable polymer-coated everolimus-eluting stent Xience DES (Abbott, United States); b) the biodegradable polymer-coated everolimus-eluting stent Synergy DES (Boston Scientific, United States), and c) the polymer-free BES Biofreedom DES (Biosensors International Ltd, Singapore). The study was conducted at 4 Spanish teaching hospitals.
A total of 144 patients were consecutively recruited from January 2018 through December 2019. Patients were eligible if they had been admitted due to stable coronary artery disease or acute coronary syndrome without cardiogenic shock. The medical team selected the type of stent that should be implanted. The detailed study flowchart is shown on figure 1. Inclusion criteria were a) de novo lesions; b) ≥ 1 target lesions in the same or different coronary artery; c) no need for stent overlapping, and a 10 mm minimal distance between the stents; d) stent length between 8 mm and 30 mm; e) use of stents with diameters ≥ 2.5 mm.
Figure 1. Flowchart of patient inclusion.
Exclusion criteria were a) complex lesions including ostial lesions, chronic total coronary occlusions, calcified lesions requiring calcium modification techniques, and bifurcations requiring the kissing balloon technique; b) target lesions in small vessels (< 2.5 mm) and long lesions (> 30 mm) requiring small diameter stents (2.25 mm) or overlapping stents; c) diabetic patients; d) very tortuous arteries that anticipated the impossibility of access with the OCT catheter for follow-up purposes; and e) complications during index procedure. Patients with diabetes mellitus were excluded from the study because of their pro-inflammatory status that facilitates both stent thrombosis and restenosis.18
Once recruited, patients were consecutively assigned to a 1- or 6-month OCT follow-up group. The baseline characteristics, angiographic and procedural data, follow-up data, and outcome data were prospectively collected by the study coordinators. Clinical data at follow-up were obtained from the clinical records. This study was performed following the principles established by the Declaration of Helsinki, ISO14155, and the clinical practice guidelines. The study protocol was approved by the Institutional Ethics Committee (IEC) and the hospital research committee. Informed consent was obtained from all the patients.
Percutaneous coronary intervention, angiographic analysis, and optical coherence tomography
In the index procedure stents were implanted according to the standard approach. Patients were medically treated following the European guidelines on the management of chronic ischemic heart disease or acute coronary syndrome.19
Regarding the initial angiographic analysis, 2 orthogonal projections without coronary guidewire were obtained after finishing the index procedure. These same projections were acquired at follow-up. The off-line analysis of the angiographic images (quantitative coronary angiography [QCA]) was performed in an independent core lab (Barcelona Cardiac Imaging Core-Lab [BARCICORElab]), following their standard protocol. They used a dedicated software (CAAS, version 5.9; Pie Medical BV, The Netherlands). Methods used in this core lab have been previously reported20.
The follow-up angiography was performed at 1-or-6-month follow-up. Angiographic and OCT images were obtained from each patient. The Dragonfly frequency domain OCT C7-XR system (St. Jude Medical, United States) was used. This analysis was performed at the same independent core lab with a dedicated software (St. Jude Medical). Further information can be found on the supplementary data. The struts were classified as non-covered if their surface was totally or partially exposed to the lumen, and without any tissue coverage above its high-density scaffold. Stent strut apposition was defined as the perpendicular distance between the luminal edge of the strut and the vascular wall. Incomplete apposition was considered when distance was higher compared to the total strut thickness considering the addition of strut plus polymer. Intimal hyperplasia was measured as the perpendicular distance between the luminal surface of the stent strut and the luminal surface of the neointima.
Endpoints
The study primary endpoint was the percentage of uncovered struts among durable-polymer EES vs biodegradable-polymer EES vs polymer-free BES as seen on the OCT at 1 month.
The study secondary endpoint was to compare the coverage and apposition of these 3 different types of DES on the OCT 1 vs 6 months after implantation. In addition, we evaluated the intimal hyperplasia in the 3 stent groups over time.
Statistical analysis
Continuous variables were expressed as mean and standard deviation except when they did not follow normal distribution, in which case they were expressed as median and 25th-75th percentile. Categorical variables were expressed as frequency and percentage. The analysis of the clinical differences was performed using the chi-square test or Fisher’s exact test for qualitative variables. Comparison among quantitative variables was performed using the 1-way ANOVA test. Generalized estimating equations, considering the clustering nature of the OCT data, were used to conduct analyses at strut level. All probability values were two-sided, P values < .05 were considered statistically significant. Statistical analysis was performed using the SPSS software package, version 22.0 (SPSS, United States). The sample size estimate is shown on the supplementary data.
RESULTS
Baseline clinical characteristics
A total of 104 patients from 4 different hospitals were included in the study; 44 patients were treated with a polymer-free BES, 35 with a biodegradable-polymer EES, and 25 patients with a durable-polymer EES. Of these, 37 patients underwent follow-up angiography and OCT 1 month after DES implantation, and 67 patients after 6 months. Mean age was 57 years; most patients were man (11% women). The inter-group baseline clinical characteristics are shown on table 1 according to the type of stent implanted. We observed a statistically significant difference in the left ventricular ejection fraction that was slightly lower in patients who received a polymer-free BES (54% vs 60%). The number of patients who needed postdilatation was higher in the durable-polymer EES group (68%) especially compared to the polymer-free BES group (38%).
Table 1. Baseline clinical, lesion, and procedural characteristics
| Polymer-free BES (N = 44) |
Biodegradable-polymer EES (N = 35) |
Durable-polymer EES (N = 25) |
P | |
|---|---|---|---|---|
| Age | 57 ± 8 | 61 ± 9 | 59 ± 10 | .094 |
| Women | 3 (7) | 4 (11) | 4 (16) | .482 |
| Dyslipidemia | 24 (55) | 19 (54) | 14 (56) | .990 |
| Hypertension | 17 (39) | 14 (40) | 13 (52) | .527 |
| Family history of ischemic heart disease | 10 (23) | 4 (11) | 6 (24) | .353 |
| Smoker | 26 (59) | 13 (37) | 9 (36) | .076 |
| LVEF % | 54 ± 9 | 60 ± 9 | 60 ± 8 | .006 |
| Chronic kidney disease (creatinine > 1.5 mg/dL) | 0 (0) | 1 (3) | 0 (0) | .370 |
| Previous MI | 2 (5) | 7 (20) | 4 (12) | .102 |
| Previous PCI | 2 (5) | 6 (17) | 4 (16) | .159 |
| Previous CABG | 1 (2) | 0 (0) | 1 (4) | .526 |
| Target lesion location | .101 | |||
| Left anterior descending coronary artery | 18 (41) | 10 (29) | 11 (44) | |
| Left circumflex artery | 10 (23) | 8 (23) | 19 (40) | |
| Right coronary artery | 15 (34) | 13 (37) | 4 (16) | |
| Secondary artery (diagonal, posterolateral, posterior descending) | 1 (2) | 4 (11) | 0 (0) | |
| Stent length, mm | 18.6 ± 5 | 18.8 ± 6 | 19.5 ± 6 | .769 |
| Stent diameter, mm | 3.4 ± 0.8 | 3.1 ± 0.5 | 3.1 ± 0.4 | .053 |
| Predilatation, % | 19 (43) | 16 (73) | 13 (53) | .076 |
| Postdilatation, % | 16 (38) | 12 (57) | 17 (68) | .05 |
|
Data are expressed as no. (%) or mean ± standard deviation. |
||||
Procedural and lesion characteristics
Procedural characteristics based on the type of stent implanted are shown on table 1. We found no significant differences in stent diameter or length in the 3 stent groups. The left anterior descending coronary artery was the most treated of all whereas secondary arteries were scarcely included in this study.
Angiographic analysis
The lesion angiographic characteristics are shown on table 2 and table 1 of the supplementary data. There were 2 patients with angiographic images with insufficient quality for analysis, both from the biodegradable-polymer EES group. There were no significant differences in the lesions before or after the PCI. After 1 month, no significant differences were reported regarding lumen loss or percent diameter stenosis. No differences were reported among the 3 stent groups at 6 months.
Table 2. Angiographic analysis
| Polymer-free BES (N = 44) | Biodegradable-polymer EES (N = 35) | Durable-polymer EES (N = 25) | P | |
|---|---|---|---|---|
| 1-month follow-up | (N = 7) | (N = 16) | (N = 12) | |
| Stent length, mm | 17.85 ± 4.32 | 19.24 ± 5.63 | 19.39 ± 4.41 | .788 |
| Reference lumen diameter, mm | 2.93 ± 0.60 | 2.80 ± 0.53 | 2.77 ± 0.55 | .827 |
| Minimal lumen diameter, mm | 2.75 ± 0.46 | 2.65 ± 0.50 | 2.51 ± 0.49 | .586 |
| Late lumen loss, mm | 0.03 ± 0.09 | 0.04 ± 0.10 | 0.03 ± 0.08 | .965 |
| Percentage diameter stenosis, % | 5.57 ± 6.27 | 6.50 ± 7.14 | 8.67 ± 9.27 | .658 |
| 6-month follow-up | (n = 37) | (n = 17) | (n = 13) | |
| Stent length, mm | 18.99 ± 4.92 | 20.03 ± 6.55 | 18.13 ± 4.95 | .627 |
| Reference lumen diameter, mm | 2.75 ± 0.57 | 2.79 ± 0.50 | 2.65 ± 0.34 | .757 |
| Minimal lumen diameter, mm | 2.54 ± 0.45 | 2.34 ± 0.41 | 2.39 ± 0.37 | .213 |
| Late lumen loss, mm | 0.19 ± 0.25 | 0.28 ± 0.24 | 0.20 ± 0.18 | .368 |
| Percentage diameter stenosis, % | 5.77 ± 15.30 | 15.18 ± 12.92 | 10.08 ± 7.40 | .065 |
|
Data are expressed as no. (%) or mean ± standard deviation. |
||||
OCT outcomes
OCT results are shown on table 3, and table 2 in the supplementary data. There were 6 patients with OCT images with insufficient quality for analysis, 2 from the polymer-free BES, 3 from the biodegradable-polymer EES group, and 1 from the durable-polymer EES group. Stent strut coverage and apposition were analyzed, as well as neointimal hyperplasia. Overall, 15 906 struts were examined. Of these, 4380 struts were from durable-polymer EES; 5122 from biodegradable-polymer EES; and 6404 from polymer-free BES; 6184 and 9722 struts were analyzed at 1 and 6 months, respectively.
Table 3. Optical coherence tomography analysis
| Polymer-free BES | Biodegradable-polymer EES | Durable-polymer EES | Pa | Pb | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Follow-up | 1-month (N = 7) |
6-months (N = 35) |
P | 1-month (N = 17) |
6-months (N = 15) |
P | 1-month (N = 12) |
6-months (N = 12) |
P | ||
| Strut level analysis | (N = 1242) | (N = 5162) | (N = 2673) | (N = 2449) | (N = 2269) | (N = 2111) | |||||
| Uncovered struts, n | 238 (19.2) | 154 (3.0) | < .001 | 318 (11.9) | 123 (5.0) | .007 | 396 (17.5) | 133 (6.3) | .001 | .209 | .172 |
| Malapposed struts, n | 66 (5.3) | 13 (0.3) | < .001 | 101 (3.8) | 21(0.9) | .001 | 138 (6.1) | 35 (1.7) | .029 | .497 | .071 |
| Neointimal thickness, µm | 50.7 ± 41.9 | 138.1 ± 102.9 | < .001 | 59.9 ± 45.1 | 88.3 ± 83.9 | .005 | 48.9 ± 38.1 | 85.5 ± 68.6 | < .001 | .083 | < .001 |
|
Data are expressed as no. or mean ± standard deviation. Categorical variables were estimated using the chi-square test; quantitative variables were estimated with 1-way ANOVA test, and strut level analyses were conducted using generalized estimating equations considering the clustering nature of OCT data. |
|||||||||||
A high rate of uncovered struts was found among stents 1 month after implantation, with no significant differences (P = .209). We observed ≥ 5% of uncovered struts in > 80% of the patients. There was better coverage in the 3 stent groups at 6 months compared to 1 month (P < .001 polymer-free BES; P = .007 biodegradable-polymer EES; P = .001 durable-polymer EES). No statistically significant differences were reported in strut coverage at 6 months among the different stents (P = .172) (figure 2, figure 3A).
Figure 2. Endpoint comparison at 1 and 6 months. A: percentage of struts covered at 1 and 6 months; B: percentage of struts apposed at 1 and 6 months; C: neointimal hyperplasia at 1 and 6 months. BES, biolimus-eluting stent; EES, everolimus-eluting stent.
Figure 3. Optical coherence tomography. A: optical coherence tomography showing covered and uncovered stent struts at 1 and 6 months; B: optical coherence tomography showing malapposed stent struts at 1 and 6 months. BES, biolimus-eluting stent; EES, everolimus-eluting stent; MO, months.
Regarding strut apposition to the artery walls, no significant differences were reported among the 3 stents after 1 month (P = .497). We observed ≥ 5% of malapposed struts in 29% to 30% of the patients with no differences among stents. No significant differences were reported among the stents after 6 months either. The rate of apposition was higher at 6 months compared to 1 month in all stent groups (P < .001 polymer-free BES; P = .001 biodegradable-polymer EES; P = .029 durable-polymer EES) (figure 2, figure 3B).
When we analyzed neointimal hyperplasia, no significant differences were found at 1 month among the 3 stent groups (P = .083). At 6 months, we found higher hyperplasia in the polymer-free BES compared to the durable-polymer EES (P < .001). We found more hyperplasia at 6 months compared to 1 month in all groups (P < .001 polymer-free BES; P < .001 biodegradable-polymer EES; P = .005 durable-polymer EES; figure 2).
DISCUSSION
The main findings of this prospective multicenter registry are: a) in non-diabetic patients with de novo non-complex coronary lesions treated with durable vs biodegradable vs polymer-free DES, strut coverage was similar and low (≥ 5% of uncovered struts in > 80% of patients) at 1 month; b) there was a similar high rate of malapposed stent struts (4% to 6%) at 1 month; c) intimal coverage and apposition improved significantly at 6 months; d) polymer-free BES had higher intimal hyperplasia at 6 months.
OCT findings suggest that, in non-diabetic patients with non-complex coronary lesions treated with 3 latest generation DES, there is a similar high rate of uncovered and malapposed struts at 1 month. It is after 6 months when we could see better coverage and apposition.
Several small-scale OCT studies have been performed to compare the coverage and apposition of stents with permanent and absorbable polymers.21-24 Conclusions of these studies differ with most of them stating that the absorbable polymer stent has better coverage than the permanent polymer, and 1 of them concluding the opposite.23 One of the studies found coverage to be sufficient after 3 months,22 whereas another stated that coverage improved at 12 months.24 In this study, we found no significant differences in stent strut coverage or apposition between permanent and absorbable polymer at 1 or 6 months on the OCT analysis (figure 2). Differences in stent strut coverage at follow-up may in part be explained by the stent platform, the polymers used to control drug release, and the antiproliferative drug itself. The stents of the study had a similar drug (-limus analogue) but different polymeric features (durable vs biodegradable vs polymer-free), and different platform thickness (the polymer-free BES had a thicker platform). Probably within the first month the drug effect is most important, and it was similar among the study stents (antiproliferative -limus analogues). However, over time (between 1 and 6 months), other stent features such as platform thickness or polymeric features might play a role that could explain the differences seen regarding coverage at 3 months in other studies.22,23
Accordingly, other studies have analyzed other types of polymer-free stents different to the one from our study.25,26 They found that coverage was achieved in a high percentage at 3 to 9 months, reaching conclusions that were similar to our study. One of the studies26 performed an OCT at 1, 3, and 9 months, demonstrating higher strut coverage over time, which is consistent with our results. Only 1 study analyzed the Biolimus A9 polymer-free stent with OCT without comparing it to other stents.27 It was a prospective single-center single-armed study that examined strut coverage of the Biolimus A9 polymer-free stent at 1, 2, 3, 4, 5, and 9 months. Researchers found that coverage was fast and improved over time with the stent remaining safe and effective. These results are similar to ours in the sense that coverage was significantly better at 6 months. However, we also compared polymer-free stents to other polymer-based stents, something that, to the best of our knowledge, has not been tested before.
One of the limitations of extrapolating the clinical safety of the stents and the degree of intimal coverage as seen on the OCT is that there is no consensus on the cut-off value of coverage that would allow safe discontinuation of DAPT. Few studies1,28 have tried to decide on a percentage of coverage, with the only in-vivo study estimating that > 5.9% of uncovered struts was an independent risk factor for stent thrombosis.28 However, these studies were limited in the number of patients included, and only some stents were tested. Larger studies are needed to decide on a security threshold for strut coverage that would make it safe to stop DAPT without increasing the risk of stent thrombosis. Therefore, the rate of coverage at 1 month (80% to 88%) in our study seems insufficient, reaching a very high percentage after 6 months (94% to 97%) in the 3 types of stents.
Finally, our study shows that intimal hyperplasia was significantly higher in polymer-free stents at 6 months. Polymer-free Biolimus A9 has a stainless steel and thicker platform, which has been associated with more intimal hyperplasia and in-stent restenosis in previous studies.27 The other 2 types of DES have a cobalt-chromium platform which has largely substituted stainless steel to provide sufficient strength and visibility with thinner struts of around 70-90 μm, resulting in lower rates of angiographic and clinical restenosis.29 Thus, inflammatory response to this thicker stent platform (130-140 μm) could be in part responsible for this finding.
Study limitations
This was an OCT-based study; unfortunately, it was not powered to assess clinical outcomes. Our study was non-randomized. However, we minimized the confounding factors through selected inclusion/exclusion criteria for patients and lesions (non-diabetic patients with non-complex coronary lesions). We analyzed the differences among the groups and no significant differences were found, except in the left ventricular ejection fraction, which was significantly lower in the group of polymer-free stents. It has not been described in the medical literature whether a lower left ventricular ejection fraction has any correlation with stent thrombosis. This study included selected non-diabetic patients with simple coronary artery lesions. Therefore, the conclusions cannot be extrapolated to other groups with different characteristics.
The distribution of patients who underwent the follow-up angiography in the polymer-free DES group at 1 or 6 months was uneven. More patients rejected the follow-up angiography at 1 month in the polymer-free DES group, which may account for this difference. Finally, the complexity of the analysis of 3 different groups in 2 different moments of time caused a disgregation of cases. This led to a small N in each group, with the potential biases associated.
CONCLUSIONS
In non-diabetic patients, a significantly high percentage of uncovered struts was detected at 1 month with OCT in latest generation DES regardless of the polymeric features of the stent (durable vs biodegradable vs polymer-free stent) in the non-complex coronary artery lesion setting. Our OCT findings do not support improved short-term healing characteristics of stents with biodegradable polymer or polymer-free based -limus elution compared to current generation of durable polymer DES.
FUNDING
This study received funding from Abbott Vascular and Biosensors International. Funders were not involved in the study design, collection, analysis, interpretation of data, drafting of this article or decision to submit it for publication.
AUTHORS’ CONTRIBUTIONS
A. Calvo-Fernández, R. Elosua, and B. Vaquerizo designed the study. J. Gómez-Lara, Héctor Cubero-Gallego, Helena Tizón-Marcos, N. Salvatella, A. Negrete, R. Millán, J.L. Díez, J.M. de la Torre Hernández, and B. Vaquerizo participated in the recruitment of patients and database contribution. A. Calvo-Fernández, C. Ivern, A. Sánchez-Carpintero, and B. Vaquerizo managed the database. A. Calvo-Fernández, X. Durán, N. Farré, and B. Vaquerizo conducted the data analysis and statistics. A. Calvo-Fernández, R. Elosua, N. Farré, H. Cubero-Gallego, and B. Vaquerizo drafted the paper with feedback from all the authors.
CONFLICTS OF INTEREST
J.M. de la Torre Hernandez is an associate editor of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed; he has received grants and research support from Abbott Medical, Biosensors, Bristol Myers Squibb, Amgen; honoraria or consultation fees from Boston Scientific, Medtronic, Biotronik, Astra Zeneca, Daiichi-Sankyo. N. Salvatella has received teaching honoraria from Abbott Vascular, and consultation fees from Boston Scientific. The remaining authors declared no other competing interests.
WHAT IS KNOWN ABOUT THE TOPIC?
- Current DES with biodegradable polymer coating or the new polymer-free biolimus stent have been proposed as the optimal solution to the problem of delayed coronary artery healing characteristics seen with first-generation durable polymer-coated DES.
WHAT DOES THIS STUDY ADD?
- This multicenter registry compared 3 types of DES with similar drug elution (-limus analogue), but different stent polymeric features (durable vs biodegradable vs polymer free).
- Using the percentage of uncovered struts at short term assessed by optical coherence tomography as a surrogate endpoint, healing characteristics at 1 month were similar among stents and insufficient after 1 month.
- Our findings do not support a preferential use of stents with biodegradable polymer-based or polymer-free stents to reduce the time of dual antiplatelet therapy at 1 month.
REFERENCES
1. Finn AV, Joner M, Nakazawa G, et al. Pathological Correlates of Late Drug-Eluting Stent Thrombosis: Strut Coverage as a Marker of Endothelization. Circulation. 2007;115:2435-2441.
2. Guagliumi G, Sirbu V, Musumeci G, et al. Examination of the In Vivo Mechanisms of Late Drug-Eluting Stent Thrombosis. J Am Coll Cardiol Intv. 2012;5:12-20.
3. Magnani G, Valgimigli M. Dual Antiplatelet Therapy After Drug-eluting Stent Implantation. Interv Cardiol. 2016;11:51-53.
4. Kelly CR, Teirstein PS, Meredith IT, et al. Long-Term Safety and Efficacy of Platinum Chromium Everolimus-Eluting Stents in Coronary Artery Disease: 5-Year Results From the PLATINUM Trial. J Am Coll Cardiol Intv. 2017;10:2392-2400.
5. Maeng M, Tilsted HH, Jensen LO, et al. Differential clinical outcomes after 1 year versus 5 years in a randomised comparison of zotarolimus-eluting and sirolimus-eluting coronary stents (the SORT OUT III study): a multicentre, open-label, randomised superiority trial. Lancet. 2014;383:2047-2056.
6. Jakobsen L, Christiansen EH, Maeng M, et al. Final five-year outcomes after implantation of biodegradable polymer-coated biolimus-eluting stents versus durable polymer-coated sirolimus-eluting stents. EuroIntervention. 2017;13:1336-1344.
7. Sarno G, Lagerqyist B, Fröbert O, et al. Lower risk of stent thrombosis and restenosis with unrestricted use of ‘new-generation’ drug-eluting stents: a report from the nationwide Swedish Coronary Angiography and Angioplasty Registry (SCAAR). Eur Heart J. 2021;33:606-613.
8. von Birgelen C, Kok MM, van der Heijden LC, et al. Very thin strut biodegradable polymer everolimus-eluting and sirolimus-eluting stents versus durable polymer zotarolimus-eluting stents in allcomers with coronary artery disease (BIO-RESORT): a three-arm, randomised, non-inferiority trial. Lancet. 2016;388:2607-2617.
9. Raungaard B, Jensen LO, Tilsted HH, et al. Zotarolimus-eluting durable-polymer-coated stent versus a biolimus-eluting biodegradable-polymer-coated stent in unselected patients undergoing percutaneous coronary intervention (SORT OUT VI): a randomised non-inferiority trial. Lancet. 2015;385:1527-1535.
10. Jensen LO, Thayssen P, Maeng M, et al. Randomized Comparison of a Biodegradable Polymer Ultrathin Strut Sirolimus-Eluting Stent With a Biodegradable Polymer Biolimus-Eluting Stent in Patients Treated With Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2019;9:e003610.
11. Räber L, Kelbæk H, Ostojic M, et al. Effect of biolimus-eluting stents with biodegradable polymer vs bare-metal stents on cardiovascular events among patients with acute myocardial infarction: the COMFORTABLE AMI randomized trial. JAMA. 2012;308:777-787.
12. Kim HS, Kang J, Hwang D, et al, HOST-REDUCE-POLYTECH-ACS Trial Investigators. Durable Polymer Versus Biodegradable Polymer Drug-Eluting Stents After Percutaneous Coronary Intervention in Patients with Acute Coronary Syndrome: The HOST-REDUCE-POLYTECH-ACS Trial. Circulation. 2021;143:1081-1091.
13. Urban P, Meredith IT, Abizaid A, et al. Polymer-free Drug-Coated Coronary Stents in Patients at High Bleeding Risk. N Engl J Med. 2015;373:2038-2047.
14. Windecker S, Latib A, Kedhi E, et al; ONYX-ONE Investigators. Polymer-based or Polymer-free Stents in Patients at High Bleeding Risk. N Engl J Med. 2020;382:1208-1218.
15. Kufner S, Ernst M, Cassese S, et al; ISAR-TEST-5 Investigators. 10-Year Outcomes From a Randomized Trial of Polymer-Free Versus Durable Polymer Drug-Eluting Coronary Stents. J Am Coll Cardiol. 2020;76:146-158.
16. Lee KS, Lee JZ, Hsu CH, et al. Temporal Trends in Strut-Level Optical Coherence Tomography Evaluation of Coronary Stent Coverage: A Systematic Review and Meta-Analysis. Catheter Cardiovasc Interv. 2016;88:1083-1093.
17. Tearney GJ, Regar E, Akasaka T, et al. Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J Am Coll Cardiol. 2012;59:1058-1072.
18. Yuan, J. Xu GM. Early and Late Stent Thrombosis in Patients with Versus Without Diabetes Mellitus Following Percutaneous Coronary Intervention with Drug-Eluting Stents: A Systematic Review and Meta-Analysis. Am J Cardiovasc Drugs. 2018;18:483-492.
19. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
20. Gomez-Lara J, Teruel L, Homs S, et al. Lumen enlargement of the coronary segments located distal to chronic total occlusions successfully treated with drug-eluting stents at follow-up. EuroIntervention. 2014;9:1181-1188.
21. Teeuwen K, Spoormans E, Bennett J, et al. Optical coherence tomography findings: insights from the “randomised multicentre trial investigating angiographic outcomes of hybrid sirolimus-eluting stents with biodegradable polymer compared with everolimus-eluting stents with durable polymer in chronic total occlusions” (PRISON IV) trial. EuroIntervention. 2017;13:e522-e530.
22. Kretov E, Naryshkin I, Baystrukov V, et al. Three-months optical coherence tomography analysis of a biodegradable polymer, sirolimus-eluting stent. J Interv Cardiol. 2018;31:442-449.
23. Adriaenssens T, Ughi GJ, Dubois C, et al. STACCATO (Assessment of Stent sTrut Apposition and Coverage in Coronary ArTeries with Optical coherence tomography in patients with STEMI, NSTEMI and stable/unstable angina undergoing everolimus vs biolimus A9-eluting stent implantation): a randomised controlled trial. EuroIntervention. 2016;11:e1619-e1626.
24. Kim BK, Hong MK, Shin DH, et al. Optical coherence tomography analysis of strut coverage in biolimus- and sirolimus-eluting stents: 3-Month and 12-month serial follow-up. Int J Cardiol. 2013;168:4617-4623.
25. Suwannasom P, Onuma Y, Benit E, et al. Evaluation of vascular healing of polymer-free sirolimus-eluting stents in native coronary artery stenosis: a serial follow-up at three and six months with optical coherence tomography imaging. EuroIntervention. 2016;12:e574-e583.
26. Worthley SG, Abizaid A, Kirtane AJ, et al. First-in-Human Evaluation of a Novel Polymer-Free Drug-Filled Stent. JACC Cardiovasc Interv. 2017;10:147-156.
27. Lee SWL, Tam FCC, Chan KKW, et al. Establishment of healing profile and neointimal transformation in the new polymer-free biolimus A9-coated coronary stent by longitudinal sequential optical coherence tomography assessments: the EGO-BIOFREEDOM study. EuroIntervention. 2018;14:780-788.
28. Won H, Shin DH, Kim BK, et al. Optical coherence tomography derived cut-off value of uncovered stent struts to predict adverse clinical outcomes after drug-eluting stent implantation. Int J Cardiovasc Imaging. 2013;29:1255-1263.
29. Pache J, Kastrati A, Mehilli J, et al. Intracoronary stenting and angiographic results: Strut thickness effect on restenosis outcome (ISAR-STEREO-2) trial. J Am Coll Cardiol. 2003;41:1283-1288.
ABSTRACT
Introduction and objectives: The optimal time to perform a diagnostic coronary angiography in patients admitted due to non-ST-segment elevation acute coronary syndrome (NSTEACS) and start pretreatment with dual antiplatelet therapy is controversial. Our study aims to identify the current diagnostic and therapeutic approach, and clinical progression of patients with NSTEACS in our country.
Methods: The IMPACT-TIMING-GO trial (Impact of time of intervention in patients with myocardial infarction with non-ST segment elevation. Management and outcomes) is a national, observational, prospective, and multicenter registry that will include consecutive patients from 24 Spanish centers with a clinical diagnosis of NSTEACS treated with diagnostic coronary angiography and with present unstable or causal atherosclerotic coronary artery disease. The study primary endpoint is to assess the level of compliance to clinical practice guidelines in patients admitted due to NSTEACS undergoing coronary angiography in Spain, describe the use of antithrombotic treatment prior to cardiac catheterization, and register the time elapsed until it is performed. Major adverse cardiovascular events will also be described like all-cause mortality, non-fatal myocardial infarction and non-fatal stroke, and the rate of major bleeding according to the BARC (Bleeding Academic Research Consortium) scale at 1- and 3-year follow-up.
Results: This study will provide more information on the impact of different early management strategies in patients admitted with NSTEACS in Spain, and the degree of implementation of current recommendations into the routine clinical practice. It will also provide information on these patients’ baseline and clinical characteristics.
Conclusions: This is the first prospective study conducted in Spain that will be reporting on the early therapeutic strategies—both pharmacological and interventional—implemented in our country in patients with NSTEACS after the publication of the 2020 European guidelines, and on the clinical short- and long-term outcomes of these patients.
Keywords: Acute coronary syndrome. Acute myocardial infarction. Non-ST-segment elevation acute coronary syndrome. Dual antiplatelet therapy. Pretreatment. Early invasive strategy. ESC guidelines. Diabetes mellitus. Hemorrhage. Revascularization.
RESUMEN
Introducción y objetivos: El momento óptimo para la realización de un cateterismo diagnóstico en pacientes con síndrome coronario agudo sin elevación del segmento ST (SCASEST) y la necesidad de pretratamiento con doble antiagregación son motivo de controversia. Este estudio pretende conocer el abordaje diagnóstico y terapéutico actual, así como la evolución clínica de los pacientes con SCASEST en España.
Métodos: El estudio IMPACT of Time of Intervention in patients with Myocardial Infarction with Non-ST seGment elevation. ManaGement and Outcomes (IMPACT-TIMING-GO) es un registro nacional observacional, prospectivo y multicéntrico, que incluirá pacientes consecutivos con diagnóstico de SCASEST tratados con coronariografía diagnóstica y que presenten enfermedad coronaria aterosclerótica inestable o causal en 24 centros españoles. El objetivo primario del estudio es conocer el grado de cumplimiento de las recomendaciones de las guías de práctica clínica en pacientes que ingresan por SCASEST tratados con coronariografía en España, describir el uso del tratamiento antitrombótico antes del cateterismo y determinar el tiempo hasta este en la práctica clínica real. Se describirán también los eventos adversos cardiovasculares mayores: mortalidad por cualquier causa, infarto no fatal e ictus no fatal, y también la incidencia de hemorragia mayor según la escala BARC (Bleeding Academic Research Consortium) durante el seguimiento a 1 y 3 años.
Resultados: Este registro permitirá mejorar el conocimiento en relación con el abordaje terapéutico inicial en pacientes que ingresan por SCASEST en España. Contribuirá a conocer sus características basales y su evolución clínica, así como el grado de adherencia y cumplimiento de las recomendaciones de las que se dispone actualmente.
Conclusiones: Se trata del primer estudio prospectivo realizado en España que permitirá conocer las estrategias terapéuticas iniciales, tanto farmacológicas como intervencionistas, que se realizan en nuestro país en pacientes con SCASEST tras la publicación de las guías europeas de 2020, y la evolución clínica de estos pacientes a corto y largo plazo.
Palabras clave: Síndrome coronario agudo. Infarto agudo de miocardio. Síndrome coronario agudo sin elevación del segmento ST. Doble antiagregación plaquetaria. Pretratamiento. Coronariografía precoz. Guía ESC. Diabetes mellitus. Hemorragia. Revascularización.
Abbreviations
IMPACT-TIMING-GO: Impact of Time of Intervention in patients with Myocardial Infarction with non-ST segment elevation ManaGement and Outcomes. SCA: síndrome coronario agudo. SCASEST: síndrome coronario agudo sin elevación del segmento ST.
INTRODUCTION
Ischemic heart disease is the leading cause of mortality in developed countries.1 The rate of acute coronary syndrome (ACS), specially non-ST-segment elevation ACS (NSTEACS), has increased over the last few years, in part, due to the ageing of the population.2-3 Given the underlying pathophysiology4 patients receive specific antithrombotic treatment, and invasive approach is used in most of the cases.1-3 The new guidelines published by the European Society of Cardiology (ESC) on the management of NSTEACS1 include changes compared to the guidelines published back in 2016. The most significant ones include antithrombotic treatment, the revascularization strategy, and several controversial innovations.
In the guidelines published in 2020, early cardiac catheterization within the first 24 hours after admission was advised (level of evidence IA) in patients diagnosed with acute myocardial infarction with GRACE scores (Global Registry of Acute Coronary Events) > 140 or dynamic electrocardiographic changes suggestive of ischemia.1 Also, the previous window of recommendation of 0 to 72 hours for moderate risk patients is now gone.4 On the other hand, the systematic use of pretreatment at admission with an P2Y12 inhibitor antiplatelet drug (ticagrelor, prasugrel or clopidogrel) in patients to be treated with an early invasive strategy is now ill-advised.1
The objective of the IMPACT registry (Time of intervention in patients with myocardial infarction with non-ST segment elevation, management and outcomes [IMPACT-TIMING-GO]) is to get the big picture on the current treatment of NSTEACS, in Spain, in association with catheterization times, use of pretreatment in these patients, and describe the possible prognostic implications of the different strategies used in real life.
METHODS
Study design and population
This is an observational, prospective, multicenter, and nationwide registry that will include all consecutive patients admitted with a diagnosis of NSTEACS to the different participant centers, treated with diagnostic coronary angiography, and with unstable or causal atherosclerotic disease regardless of further treatment administered by the heart team. The baseline characteristics of the patients included, and their clinical progression regarding in-hospital events will be studied. Patients will undergo a 1-and-3-year clinical follow-up period.
This registry has been promoted by the Spanish Society of Cardiology Young Cardiologists Working Group with scientific support from the Spanish Society of Cardiology Research Agency. Also, it has been approved by different Research Ethics Committees with drugs from all the participant hospitals. Finally, it has been designed according to the STROBE checklist for observational studies.
The list of centers that will eventually participate in the registry is shown on figure 1. Inclusion and exclusion criteria are shown on table 1. The presence of elevated markers of myocardial damage or electrocardiographic changes is not mandatory. Patients with a clinical diagnosis of unstable angina can be included as long as coronary angiography confirms the clinical diagnosis.
Figure 1. Map with the Spanish participant centers in the IMPACT-TIMING-GO registry.
Table 1. Inclusion and exclusion criteria of the IMPACT-TIMING-GO registry
| Inclusion criteria |
|---|
| NSTEACS with in-hospital invasive treatment regardless of when it is performed. |
| Evidence of causal or unstable atherosclerotic disease. |
| Age ≥ 18 years. |
| Capacity to give informed consent. |
| Exclusion criteria |
| Minors and those who withdraw their consent to be included or followed at any time during the study. |
| Assessment of myocardial damage markers associated with type 2 myocardial infarction. |
| Patients without any signs of coronary artery disease including those with myocarditis, Prinzmetal angina, takotsubo syndrome or MINOCA. |
| Patients diagnosed with spontaneous coronary artery dissection. |
| Patients with complete left bundle branch block or pacemaker rhythm on the electrocardiogram performed at admission. |
| Patients with a valve heart disease eligible for surgery. |
| Patients with a known past medical history of diffuse coronary artery disease noneligible for revascularization. |
| Patients with known or confirmed allergy to some antiplatelet drug. |
|
IMPACT-TIMING-GO, IMPACT of time of intervention in patients with myocardial infarction with non-ST segment elevation. Management and outcomes; MINOCA, Myocardial infarction with non-obstructive coronary artery disease; NSTEACS, non-ST-segment elevation acute coronary syndrome. |
Endpoints
The study primary endpoint is to know the degree of compliance of the recommendations included in the clinical practice guide-lines in patients admitted due to NSTEACS treated with coronary angiography, in Spain, describe the use of antithrombotic treatment before cardiac catheterization, and the time elapsed until it is performed in the real-world clinical practice.
The secondary endpoints are:
- – To describe the baseline, clinical, and epidemiological characteristics of the study population.
- – To study the rates of cardiovascular mortality, new revascularization, stent thrombosis, and hospitalizations due to heart failure during admission and at the 1-and-3-year follow-up.
- – To describe major cardiovascular adverse events of all-cause mortality, non-fatal stroke, non-fatal infarction, and the rate of major bleeding grades 3, 4, and 5 according to the BARC scale (Bleeding Academic Research consortium.5) Data will be analyzed during admission and at the 1-and-3-year follow-up.
- – To know the medical treatment at discharge and at follow-up of patients discharged in Spain after NSTEACS.
- – To know the degree of control of the different cardiovascular risk factors associated with the endpoints defined in the ESC guidelines 2021 on prevention of cardiovascular disease in the routine clinical practice.6
Data curation and definitions
Data will be collected prospectively by trained medical investigators from each participant center in a specific standard form. Demographic data, the baseline clinical characteristics, and all analytical, electrocardiographic, and echocardiographic data will be included as well.
Similarly, data on disease progression and the in-hospital stay, indication for coronary angiography and when it is be performed, type of treatment received (conservative, stent implantation or revascularization surgery), and the in-hospital complications occurred (hemorrhages and severity, heart failure or shock, reinfarction, stroke, confusional state, mechanical and arrhythmic complications, infectious complications requiring antibiotic therapy, and mortality causes) will be collected. Finally, the medical treatment at hospital discharge and level of compliance of the current recommendations based on the clinical practice guidelines will be studied too.
The definitions of the variables are shown on table 2.7-8
Table 2. Definitions of target variables
| Variable | Definition |
|---|---|
| All-cause mortality | All deaths regardless of their cause. |
| Cardiovascular death | All deaths of vascular causes both cardiac (heart failure/shock; malignant arrhythmias; myocardial infarction) and non-coronary vascular including cerebrovascular disease, pulmonary embolism, aneurysms/aortic dissections, acute ischemia of lower limbs, etc.
All sudden deaths of unknown causes will be adjudicated as cardiovascular death. |
| Non-cardiac death | All deaths that do not meet the previous definition like deaths due to infections, cancer, pulmonary diseases, accidents, suicide or trauma. |
| Myocardial infarction | It is defined based on the criteria established in the 4th and current Universal definition.4 Therefore, patients with type 2 infarction, extracardiac causes or without elevated markers of myocardial damage were excluded. |
| Stroke/Transient ischemic attack | New-onset neurological, focal or global deficit due to ischemia or hemorrhage, and as long as it is part of diagnostic judgement at hospital discharge. |
| Stent thrombosis | Defined based on the Academic Research Consortium of randomized clinical trials with stents.7 |
| New revascularization | All unscheduled revascularizations performed after hospital discharge, whether surgical or percutaneous, including target vessel failure and target lesion failure. |
| Admission due to heart failure | Unscheduled hospital admission > 24 hours with a primary diagnosis of heart failure based on the current defintion.8 |
Follow-up
Clinical follow-up to detect events will be conducted by medical investigators through on-site visits, health record reviews or phone calls with the patient, family members or treating physician at 1 and 3 years. Clinical variables, functional class, and additional variables (analytical, electrocardiographic, and echocardiographic, and treatment received) will be included. The overall mortality rate and its causes, need of emergency hospitalization (duration > 24 hours) and its causes, and the rates of non-fatal infarction and stroke will be collected as well. All deaths due to myocardial infarction, sudden death or heart failure will be considered cardiovascular deaths.
Sample size estimate
Taking the events seen in previous studies with a population of similar characteristics as the reference,9-14 a sample size of 800 patients will be enough to know the baseline characteristics of the study population, and the therapeutic approach currently used in Spain in our routine clinical practice. Patients lost to follow-up will be handled by multiple imputation.
Statistical analysis
Categorical variables will be expressed as number and percentage. Quantitative variables will be expressed as mean ± standard deviation. Quantitative variables with normal distribution will be expressed as median and interquartile range [25%-75%]. The normal distribution of quantitative variables will be assessed using the Kolmogorov-Smirnoff test. Regarding the reference variables, Student t test will be used to compare quantitative variables, and the chi-square test or Fisher’s exact test, if applicable, to compare categorical variables. Statistical analysis will be performed using the SPSS statistical software version 22.0 (IBM Corp., Armonk, United States).
Specific studies on subgroups of special interest will be conducted: feminine sex, patients ≥ 75 years, those with GRACE scores > 140, diabetic patients, those with a past medical history of renal failure, with an indication for chronic oral anticoagulation, with multivessel disease, acute myocardial infarction, ventricular dysfunction according to the current clinical practice guidelines and based on the day of admission (holiday vs working day), and patients who require transfer to tertiary centers to receive a coronary angiography.
Ethical principles
Inclusion in the study will not imply changes to the patients’ treatment. Instead, it will follow the routine clinical practice and the recommendations set forth by the current clinical practice guidelines. Therefore, antithrombotic treatment and additional examinations including the need for a coronary angiography and the time it is performed will all be decided by the heart team based on the routine clinical practice. Coronary angiography, vascular access, antithrombotic treatment during the procedure, and the material and devices used will all be decided by the treating operator in charge of the case. All patients will sign a written informed consent form before being included in the study that will be conducted in full compliance with the Declaration of Helsinki. This study will also observe all legal regulations applicable to this type of studies and follow the good clinical practice rules while being conducted.
DISCUSSION
The IMPACT-TIMING-GO registry will give us information on the current real-world management of patients with NSTEACS with invasive treatment and causal coronary artery disease, which will allow us to assess the degree of implementation of the current recommendations of ESC guidelines 2020 on cardiac catheterizations performed within the first 24 hours and no pretreatment with P2Y12 inhibitors. Similarly, different prognostic differences that early invasive treatment and no pretreatment could have in the real life of patients diagnosed with NSTEACS could be suggested.
Despite the clinical practice guidelines recommendations on the invasive treatment of patients with NSTEACS, the main clinical trials published to this date have been unable to demonstrate any clear benefits from systematic early invasive treatment.9-14 The VERDICT trial,9 published in 2018, randomized 2147 patients with NSTEACS on a 1:1 ratio to receive early (< 12 hours) or delayed (48 to 72 hours) cardiac catheterization. No significant differences were found in the composite endpoint of major cardiovascular events at 4-year follow-up. However, in the subgroup of patients with GRACE scores > 140 statistically significant differences were seen favorable to the early strategy regarding major adverse cardiovascular events (hazard ratio, 0.81; 95% confidence interval, 0.67-1.01; P = .023). Consistent with this, the TIMACS clinical trial10 published in 2008 of 3031 patients with NSTEACS found no differences in the primary endpoint when early invasive strategy (< 24 hours) and delayed approach (> 36 hours) were compared, except for, once again, in patients with GRACE scores > 140. Other randomized clinical trials with fewer patients show contradictory results11-14 some without significant differences.15 Also, in many cases, the results favorable to the early strategy are associated with refractory ischemia, not with hard endpoints like cardiovascular mortality or non-fatal myocardial infarction. In Spain, evidence on the management of NSTEACS is prior to the current clinical practice guidelines,16-17 and the most recent registry is retrospective, which is suggestive of a possible mortality benefit in patients with GRACE scores > 140.18 Over the last 2 decades, in our country, the use of an invasive strategy in patients with NSTEACS has increased significantly from 20% in the MASCARA registry in 200516 up to 80% in the DIOCLES study from 2012.17 However, evidence is scarce on catheterization times, our capacity to adapt to current recommendations (the median time of the DIOCLES trial was 3 days), the possible impact this time reduction can have, and on the consequencies from not starting antiplatelet pretreatment in patients who don’t meet the times recommended.
On the other hand, the current formal recommendation from the clinical practice guidelines of not pretreating systematically with a P2Y12 inhibitor (level of recommendation IIIA1) patients on early invasive treatment is mainly based on 3 clinical trials and their meta-analysis.19 In the ACCOAST trial, pretreatment with prasugrel did not reduce thrombotic events in patients with NSTEMI. However, cardiac surgery-related and potentially fatal hemorrhages increased.20 We should mention that the median time elapsed since the prasugrel loading dose until the coronary angiography was performed was 4 hours. In the ISAR-REACT 5 trial published in 2019, a non-pretreatment strategy with prasugrel in patients with ACS vs pretreatment with ticagrelor proved superior regarding the primary endpoint of thrombotic events with a tendency towards fewer hemorrhagic events.21 We should mention that the intrinsic effect of the drug used should not be obviated or else the fact that the median time elapsed since randomization until the prasugrel loading dose was received in the non-pretreatment group was 60 minutes. Finally, the first study that compared 2 different pretreatment strategies vs the intraoperative administration of ticagrelor did not show any clear benefits regarding thrombosis or a deleterious effect of pretreatment regarding bleeding.22 Once again, the median time elapsed until the cardiac catheterization was performed was < 24 hours since hospital admission (23 hours). Surprisingly, clinical practice guidelines leave the door opened to a weak level of recommendation (IIbC) regarding pretreatment of patients in whom early catheterization < 24 hours is not possible.1
In conclusion, current recommendations on early invasive treatment and no antiplatelet pretreatment in patients with NSTEACS are controversial and can also be difficult to implement in the routine clinical practice in our setting. The ultimate objective of the IMPACT-TIMING-GO registry is to shed light on the current management of NSTEACS in Spain. After the impact that the COVID-19 pandemic has had on the general structure of the healthcare system and the drop in the number of interventional procedures performed in 2020,23 we should expect to see pre-pandemic numbers in 2022 and cath labs and cardiac surgery intensive care units going back to normal. Therefore, moment seems ripe to conduct a real-world registry.
CONCLUSIONS
The IMPACT-TIMING-GO registry is the first prospective study ever conducted in Spain that will be giving us information on the early therapeutic strategies—both pharmacological and interventional—performed in our country in patients with NSTEACS after the publication of the ESC guidelines 2020, and the impact of these and other measures indicated in these patients at follow-up.
FUNDING
This unfunded study has been promoted by the Spanish Society of Cardiology Young Cardiologists Working Group with scientific endorsement from the Spanish Society of Cardiology.
AUTHORS’ CONTRIBUTIONS
Study design, data curation and review, statistical analysis, and manuscript drafting: P. Díez-Villanueva, F. Díez-Delhoyo, and M.T. López-LLuva. All the authors participated in the manuscript review and approval process.
CONFLICTS OF INTEREST
None reported.
ACKNOWLEDGEMENTS
We wish to thank the Spanish Society of Cardiology Young Cardiologists Working Group for their drive to engage the youth in medical research.
WHAT IS KNOWN ABOUT THE TOPIC?
- The management of patients with NSTEACS includes dual antiplatelet therapy with a P2Y12 inhibitor and, in most cases, invasive approach through cardiac catheterization for further revascularization. The current ESC clinical practice guidelines recommend early invasive approach (<24 hours) and no pretreatment systemically though both aspects are still controversial.
- The degree of implementation of such recommendations in the routine clinical practice, in Spain, is still unknown.
WHAT DOES THIS STUDY ADD?
- This study will improve our knowledge on early therapeutic approach, and its prognostic impact in patients admitted due to NSTEACS in Spain.
- Also, it will bring us information on the characteristics and clinical evolution of these patients in association with the recommendations and therapeutic targets we have today.
REFERENCES
1. Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42:1289-1367.
2. Díez-Villanueva P, Méndez CJ, Alfonso F. Non-ST elevation acute coronary syndrome in the elderly. J Geriatr Cardiol JGC. 2020;17:9-15.
3. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the management of patients with non-ST-elevation acute coronary syndromes: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;130:2354-2394.
4. Roffi M, Patrono C, Collet JP, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:267-315.
5. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123:2736-2747.
6. Visseren FLJ, Mach F, Smulders YM, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies With the special contribution of the European Association of Preventive Cardiology (EAPC). Rev Esp Cardiol. 2022;75:429.
7. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials: The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
8. McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599-3726.
9. Kofoed KF, Kelbæk H, Hansen PR, et al. Early Versus Standard Care Invasive Examination and Treatment of Patients With Non-ST-Segment Elevation Acute Coronary Syndrome. Circulation. 2018;138:2741-2750.
10. Mehta SR, Granger CB, Boden WE, et al. Early versus delayed invasive intervention in acute coronary syndromes. N Engl J Med. 2009;360:2165-2175.
11. Thiele H, Rach J, Klein N, et al. Optimal timing of invasive angiography in stable non-ST-elevation myocardial infarction: the Leipzig Immediate versus early and late PercutaneouS coronary Intervention triAl in NSTEMI (LIPSIA-NSTEMI Trial). Eur Heart J. 2012;33:2035-2043.
12. Milosevic A, Vasiljevic-Pokrajcic Z, Milasinovic D, et al. Immediate Versus Delayed Invasive Intervention for Non-STEMI Patients: The RIDDLE-NSTEMI Study. JACC Cardiovasc Interv. 2016;9:541-549.
13. Montalescot G, Cayla G, Collet JP, et al. Immediate vs delayed intervention for acute coronary syndromes: a randomized clinical trial. JAMA. 2009;302:947-954.
14. Lemesle G, Laine M, Pankert M, et al. Optimal Timing of Intervention in NSTE-ACS Without Pre-Treatment: The EARLY Randomized Trial. JACC Cardiovasc Interv. 2020;13:907-917.
15. Janssens GN, van der Hoeven NW, Lemkes JS, et al. 1-Year Outcomes of Delayed Versus Immediate Intervention in Patients With Transient ST-Segment Elevation Myocardial Infarction. JACC Cardiovasc Interv. 2019;12:2272-2282.
16. Ferreira-González I, Permanyer-Miralda G, Marrugat J, et al. MASCARA (Manejo del Síndrome Coronario Agudo. Registro Actualizado) study. General findings. Rev Esp Cardiol. 2008;61:803-816.
17. Barrabés JA, Bardají A, Jiménez-Candil J, et al. Prognosis and management of acute coronary syndrome in Spain in 2012: the DIOCLES study. Rev Esp Cardiol. 2015;68:98-106.
18. Álvarez Álvarez B, Abou Jokh Casas C, Cordero A, et al. Early revascularization and long-term mortality in high-risk patients with non-ST-elevation myocardial infarction. The CARDIOCHUS-HUSJ registry. Rev Esp Cardiol. 2020;73:35-42.
19. Dawson LP, Chen D, Dagan M, et al. Assessment of Pretreatment With Oral P2Y12 Inhibitors and Cardiovascular and Bleeding Outcomes in Patients With Non-ST Elevation Acute Coronary Syndromes: A Systematic Review and Meta-analysis. JAMA Netw Open. 2021;4:e2134322.
20. Montalescot G, Bolognese L, Dudek D, et al. Pretreatment with prasugrel in non-ST-segment elevation acute coronary syndromes. N Engl J Med. 2013;369:999-1010.
21. Schüpke S, Neumann FJ, Menichelli M, et al. Ticagrelor or Prasugrel in Patients with Acute Coronary Syndromes. N Engl J Med. 2019;381:1524-1534.
22. Tarantini G, Mojoli M, Varbella F, et al. Timing of Oral P2Y12 Inhibitor Administration in Non-ST Elevation Acute Coronary Syndrome. J Am Coll Cardiol. 2020;76:2450-2459.
23. Romaguera R, Ojeda S, Cruz-González I, collaborators of the ACI-SEC, REGISTRY COLLABORATORS. Spanish Cardiac Catheterization and Coronary Intervention Registry. 30th Official Report of the Interventional Cardiology Association of the Spanish Society of Cardiology (1990-2020) in the year of the COVID-19 pandemic. Rev Esp Cardiol. 2021;74:1095-1105.

ABSTRACT
Introduction and objectives: There are few data on the utility of drug-coated balloons (DCB) for the side branch treatment of bifurcated lesions. Our objective was to determine the long-term effectiveness of such device in this scenario.
Methods: Retrospective-prospective registry of all such lesions treated with DCB (paclitaxel coating) at our unit from 2018 until present day with clinical follow-up including a record of adverse events.
Results: A total of 56 lesions from 55 patients were included. The main demographic characteristics were mean age, 66.2 ± 11.3; and/or women, 27.3%; hypertension, 67.3%; dyslipidemia, 83.6%, and diabetes, 32.7%. The most common causes according to the coronary angiography were non-ST segment elevation acute coronary syndrome and stable angina. The main characteristics of the lesions were the location (circumflex-obtuse marginal, 19.6%; left anterior descending-diagonal, 64.3%; left main-circumflex, 8.9%; posterior descending-posterolateral trunk, 7.1%. The Medina classification was 1-1-1 37.5% of the times, and 1-1-0, 19.6% of the times. The rate of in-stent restenotic lesions was 32.1%. Procedural characteristics: radial access, 100%; side branch (SB) and main branch (MB) predilatation, 83.9% and 58.9%, respectively; MB stenting, 71.4%; POT technique, 35.7%; final kissing, 48.2%; optical coherence tomography/intravascular ultrasound, 7.1%. Procedural success was achieved in 98.2% of the cases. The median follow-up he all-cause mortality, myocardial infarction and lesion thrombosis, and target lesion revascularization rates were .7%, 0%, and 3.6%, respectively.
Conclusions: SB treatment with DCB in selected bifurcation lesions is safe and highly effective with a long-term success rate of 96.4%. Very large studies are still required to compare this strategy to SB conservative approach, and determine its optimal treatment.
Keywords: Drug-coated balloon. Bifurcation lesions. Follow-up study. Side branch.
RESUMEN
Introducción y objetivos: Hay pocos datos acerca de la utilidad del balón farmacoactivo (BFA) para el tratamiento de la rama lateral de las lesiones en bifurcación. El objetivo fue determinar la efectividad a largo plazo de dicho dispositivo en este escenario.
Métodos: Registro retrospectivo-prospectivo de todas las lesiones de este tipo tratadas con BFA recubierto de paclitaxel en nuestra unidad desde 2018 hasta la actualidad. Se realizó un seguimiento clínico con registro de eventos adversos.
Resultados: Se incluyeron 56 lesiones de 55 pacientes. Principales características demográficas: edad media 66,2 ± 11,3 años, 27,3% mujeres, 67,3% hipertensión arterial, 83,6% dislipemia y 32,7% diabetes. Las indicaciones más frecuentes para el cateterismo fueron síndrome coronario agudo sin elevación del ST y angina estable. Características de las lesiones tratadas: localización circunfleja-obtusa marginal 19,6%, descendente anterior-diagonal 64,3%, tronco común-circunfleja 8,9% y descendente posterior-tronco posterolateral 7,1%. Según la clasificación de Medina, el tipo más frecuente fue el 1,1,1 con el 37,3%, seguido del 1,1,0 con el 19,6%. Las lesiones tipo reestenosis en el interior del stent fueron del 32,1%. Características principales del procedimiento: acceso radial 100%, predilatación de rama lateral 83,9% y de rama principal 58,9%, stent en rama principal 71,4%, técnica POT 35,7%, kissing final 48,2% y tomografía de coherencia óptica/ecocardiografía intravascular 7,1%. Se logró el éxito del procedimiento en el 98,2%. Con un seguimiento medio de 12 meses, se registraron una incidencia de muerte por cualquier causa del 3,7%, trombosis lesional o infarto 0%, y revascularización de la lesión diana del 3,6%.
Conclusiones: El tratamiento con BFA de la rama lateral en lesiones bifurcadas seleccionadas es seguro y presenta una alta efectividad, con una tasa de éxito a largo plazo del 96,4%. Serían necesarios estudios muy amplios que permitieran comparar dicha estrategia con el abordaje conservador de la rama lateral y determinar cuál es su tratamiento óptimo.
Palabras clave: Balón farmacoactivo. Lesiones en bifurcación. Estudio de seguimiento. Rama lateral.
Abbreviations
DCB: drug-coated balloon. ISR: in-stent restenosis. MB: main branch. SB: side branch.
INTRODUCTION
Coronary bifurcation lesions are still challenging for interventional cardiologists. The complexity surrounding such lesions regarding their anatomical, functional, and even clinical aspects truly complicates the management of this entity despite its high incidence rate that can be up to 20% of all the lesions that are treated at a cath lab on a routine basis. The relentless publication of articles on such lesions over the last few decades, the creation of specific study groups like the European Bifurcation Club, and the periodic publication of consensus documents for the management of this entity shows, without a doubt, that this scenario is in constant change and has not been solved today yet. One of the most controversial aspects is the importance of the side branch (SB) regarding the long-term prognosis of such lesions. Drug-coated balloon (DCB) is part of the therapeutic armamentarium of interventional cardiologists to treat coronary bifurcation lesions. Its utility for the management of certain anatomical settings like in-stent restenosis (ISR) type of lesions has already been demonstrated. However, its effectiveness to treat the SB is much less evident with scarce studies available in the medical literature. The theoretical advances posed by this device to treat the SB would be the administration of antiproliferative drugs into the ostium mainly, the lack of distortion of its original anatomy, and the minimization of strut deformation at carina level.1
This article presents a registry with the results obtained in our unit with the management of SB with DCB with a longer than usual clinical follow-up in this type of studies.
METHODS
This was a single-center, prospective-retrospective registry started back in 2019 of all coronary bifurcation lesions where the SB was treated with paclitaxel-coated DCB from October 2018 through March 2022. The device used was the SeQuent Please NEO (Braun, Germany), a paclitaxel-iopromide coated polymer-free balloon using Paccocath technology. Inclusion criteria were the presence of coronary bifurcation lesions with 1 compromised SB of, at least, 2 mm in diameter through visual angiographic estimate regardless of the aprioristic presence of a diseased SB or the appearance of carina displacement or slow flow after treating the main branch (MB). Also, the operator should consider the DCB approach of clinical and prognostic interest. Patient recruitment in the registry was on the rise: 4 patients in 2018, another 4 in 2019, 9 patients in 2020, 31 in 2021, and finally 7 within the first 3 months of 2022. No exclusion criteria were established. Approach strategy consisted of an early provisional stenting or DCB technique to treat the MB when damaged. Further management of SB with DCB was left to the operator’s criterion if, after treating the MB, significant damage done to the SB would require stenting in such branch. In that case, the patient would not be included, and the SB would not be eligible for treatment with a DCB. If, after preparing the lesion, the operator would actually consider using the DCB option, that would be the time to include the patient in the study. The rate of procedural failure—defined as the impossibility to cross the lesion with the DCB once it was used or unsatisfactory angiographic outcomes after balloon inflation involving SB stenting. The protocol for using the DCB—based on the recommendations established on the use of such devices—consisted of SB predilatation with non-compliant or scoring balloons in a 0.8-1 vessel/balloon diameter ratio, use of the device if an acceptable angiographic result with TIMI grade-3 flow was achieved, lack of significant dissection, and residual stenosis < 30%. If other lesions different from the one that triggered the inclusion in the registry needed revascularization, this was scheduled for a second surgical act. The study design followed a per protocol analysis to estimate the benefits of the technique described compared to the routine clinical practice including cases with successful DCB treatment at the follow-up and excluding those with acute device failure or impossibility to use the device once opened for being unable to cross the lesion. The lack of dissection after DCB that required stenting with residual stenosis < 50%, and final TIMI grade-3 flow was considered as procedural success. Device failure, on the other hand, was considered as an impossible DCB inflation once used or the need for stenting the SB with unsatisfactory DCB results. Different clinical variables from the patient were analyzed, as well as the lesion anatomy, and the procedural intervention per se. Retrospective clinical follow-up of patients successfully treated with the DCB was conducted. Follow-up went on for a maximum of 2 years after the procedure, and prospectively since the registry started back in 2019 until present time. This follow-up was conducted through phone calls or by checking the patients’ electronic health records. The ARC-2 definitions2 were used to collect the adverse clinical events including a composite endpoint of all-cause mortality, cardiac death, myocardial infarction, device thrombosis, clinically driven target lesion failure and revascularization, target vessel failure outside the target lesion, and revascularization of other lesions occurred at follow-up. All patients signed their written informed consent forms, and the study was approved by our center research ethics committee.
Statistical analysis
Continuous variables are expressed as mean ± standard deviation. Categorical variables are expressed as frequency and percentage. Also, actuarial curves of adverse event-free survival using the Kaplan-Meier method were built, specifically target lesion failure-free and adverse event-free curves (all-cause mortality, target lesion revascularization, target vessel failure, and revascularization of other lesions).
RESULTS
A total of 55 patients and 56 lesions were included since 2 different bifurcations found in 1 of the patients were treated in the same procedure. The patient/lesion flowchart included in the study is shown on figure 1. The patients’ clinical characteristics are shown on table 1. Vascular access was radial in 100% of the cases using a 6-Fr introducer sheath also in all of them. Table 2 shows the anatomical characteristics of target lesions. Figure 2 shows a schematic representation of the type of lesion according to the Medina classification. Table 3 shows the variables associated with the procedure. We should mention that all the clinical and anatomical data shown here, the patients’ high-risk profile with high prevalence of cardiovascular risk factors, and the large number of ISR-type of lesions reached 32.1% of the sample. The rate of lesions included with damage to 2 or 3 different bifurcation segments was 71.4% (40 out of 56). Regarding procedural factors the high rate of procedural success was significant (low rate of acute device failure with only 1 case of a type A dissection image after DCB inflation without damage to the distal flow and > 30% residual stenosis). Therefore, because of lesion location at ostium level, and possible damage to the MB (the left anterior descending coronary artery in this case), the operator decided to perform drug-eluting stent implantation for sealing purposes (figure 3). In all the remaining procedures, the acute result of the DCB was successful. In our series, the scarce use of intracoronary imaging modalities (only 7.1%) was also remarkable.
Figure 1. Flowchart of patients/lesions included in the study.
Figure 2. Number of lesions based on the type of bifurcation damage according to the Medina classification.
Figure 3. Only case of acute device failure. A: diagonal branch ostial lesion prior to the intervention (arrow); B: suboptimal outcome after drug-coated balloon (arrow); C: final outcome after stenting the side branch.
Table 1. Patients’ clinical characteristics
| N | 55 |
| Age | 66.2 ± 11.3 years [range, 45-91] |
| Sex | |
| Men | 40 (72.7%) |
| Women | 15 (27.3%) |
| Hypertension | 37 (67.3%) |
| Dyslipidemia | 46 (83.6%) |
| Smoking | 17 (30.9%) |
| Diabetes | 18 (32.7%) |
| Previous PTA | 28 (50.9%) |
| Previous coronary artery bypass graft | 1 (1.8%) |
| Indication for coronary angiography | |
| NSTEACS | 20 (36.4%) |
| STEACS | 9 (16.4%) |
| Stable angina | 20 (36.4%) |
| Other | 6 (10.9%) |
|
NSTEACS, non-ST-segment elevation acute coronary syndrome; PTA, percutaneous transluminal angioplasty; STEACS, ST-segment elevation acute coronary syndrome. |
|
Table 2. Anatomical characteristics of the lesions
| N | 56 |
| Diseased vessel | |
| LMCA-LCx | 5 (8.9%) |
| LAD-diagonal | 36 (64.3%) |
| LCx-OMA | 11 (19.6%) |
| PDA-PLT | 4 (7.1%) |
| ISR-type of lesion | 18 (32.1%) |
|
ISR, in-stent restenosis; LAD, left anterior descending coronary artery; LCx, left circumflex artery; LMCA, left main coronary artery; OMA, obtuse marginal artery; PDA, posterior descending artery; PLT, posterolateral trunk. |
|
Table 3. Procedural characteristics
| N | 56 |
| Predilatation | |
| SB | 47 (83.9%) |
| MB | 33 (58.9%) |
| MB treatment | |
| Stent | 40 (71.4%) |
| DCB | 4 (7.1%) |
| DCB diameter for the SB (mm) | |
| 2 | 20 (35.7%) |
| 2.25 | 4 (7.1%) |
| 2.5 | 23 (41.1%) |
| 3 | 8 (14.3%) |
| 3.5 | 1 (1.8%) |
| Postdilatation | |
| MB | 36 (64.3%) |
| POT | 20 (35.7%) |
| SB | 17 (30.4%) |
| Final kissing balloon | 27 (48.2%) |
| OCT/IVUS | 4 (7.1%) |
| Procedural success | 55 (98.2%) |
|
DCB, drug-coated balloon; IVUS, intravascular ultrasound; MB, main branch; OCT, optical coherence tomography; POT, proximal optimization technique; SB, side branch. |
|
The rate of adverse events at follow-up is shown on table 4. After a median follow-up of 12 months (377 ± 244 days; range, 64-734 days) only 2 clinically driven target lesion revascularizations (3.6%) were reported. Both were performed due to in-stent lesions that did not reach the target lesion proximal or distal borders. The first one was performed in a case of ISR of the SB in a very small vessel without acute ischemia whose new revascularization was performed late, more specifically, 23 months after the index procedure (figure 4). The second one was performed 6 months after the procedure—also without acute ischemic signs—but with ISR in the main vessel while the SB remained patent without significant restenosis (figure 5). Both cases were treated with drug-eluting stent implantation. Two deaths were reported: 1 cardiac death due to advanced left ventricular dysfunction in an 80-year-old woman who, after percutaneous coronary intervention, was implanted with a transfemoral aortic valve and a definitive pacemaker, with poor disease progression that eventually led to her death. The other death was septic shock related. No admissions due to acute myocardial infarction or episodes of target lesion thrombosis (both probable and definitive) were reported. No cases of target vessel failure outside the target lesion were reported either. A total of 5 revascularizations of other lesions (9.3%) were performed—all of them scheduled—but none due to acute coronary syndrome. The Kaplan-Meier curves showing target lesion revascularization-free and adverse event-free survival are shown on figure 6.
Table 4. Rate of adverse cardiovascular events at follow-up
| N | 54/55 |
| Follow-up days | 377 ± 244 [range, 79-734] |
| All-cause mortality | 2/54 (3,7%) |
| Cardiac death | 1/54 (1,8%) |
| Myocardial infarction/target lesion device thrombosis | 0/55 (0%) |
| Target lesion revascularization | 2/55 (3,6%) |
| Target vessel failure outside the target lesion | 0/55 (0%) |
| Revascularization of other lesions outside the target vessel | 5/54 (9.3%) |
Figure 1. First case of target lesion failure due to late restenosis. A: early in-stent restenosis type of lesion in obtuse marginal artery (arrow); B: final outcomes after drug-coated balloon; C: new in-stent restenosis in the side branch at 23 months (arrow).
Figure 5. Second case of target lesion failure. A: early obtuse marginal artery bifurcation lesion with distal left circumflex artery (arrow); B: outcomes after stenting the main branch, and drug-coated balloon implantation into the side branch; C: 6-month follow-up with restenosis at main branch level (arrow).
Figure 6. Kaplan-Meier curve of actuarial target lesion revascularization (TLR)-free survival and composite adverse events-free survival (all-cause mortality, TLR, revascularization of other lesions).
DISCUSSION
The latest document of the European Bifurcation Club on the utility of DCBs to treat SBs in coronary bifurcation lesions pays little attention to it due to the lack of large enough clinical trials to be conclusive.3 Despite the huge amount of medical literature available on the management of coronary bifurcation lesions, the actual significance of the SB and its involvement in target lesion failure has not been properly explained to this date. A study conducted by Oh et al.4 conclude that treating the SB in 1089 patients with bifurcation lesions at left anterior descending coronary artery-diagonal branch level was associated with a lower—yet not statistically significant—rate of target vessel failure. However, this difference was statistically significant when the subgroup studied included low-risk patients. On the other hand, a different clinical trial that studied factors associated with failed revascularizations of the left main coronary artery bifurcation found that the presence of MB stent struts inside the SB ostium was one of them5 suggestive that the use of intracoronary imaging modalities like intracoronary ultrasound or optical coherence tomography could improve results, at least, on such location, by telling us what patients would benefit from specifically treating the SB.
The strongest evidence available to this date leans towards the utility of DCB to treat ISR-type of lesions without a word dedicated to the SB. Very few studies have focused on the effectiveness of DCB to treat the SB. Such document advocates for treating coronary bifurcation lesions with the provisional stenting strategy according to the latest clinical practice guidelines drafted by the European Cardiology Society followed by treating the SB with a DCB. The first clinical trials on this regard were published back in 2011 like the DEBIUT,6 BABILON,7 DEBSIDE,8 the study conducted by Herrador et al.,9 the PEPCAD V,10 and the PEPCAD-BIF11 clinical trials. These studies showed contradictory—yet overall satisfactory—data regarding the effectiveness of DCB. These studies presented better quantitative angiographic parameters regarding restenosis or late lumen loss. However, not in every one of them this was associated with a lower rate of revascularization. As a matter of fact, there were doubts around the possibility of a higher rate of late thrombosis suggested by some of these trials. The recently published BEYOND clinical trial conducted by Jing et al.12 compared the use of a conventional balloon vs DCB to treat the SB with a 9-month angiographic follow-up. This trial found that the DCB was associated with better results regarding less late lumen loss. However, such an improvement did not translate into a lower rate of clinical adverse events since surprisingly enough no new revascularizations were reported in any of the 2 groups. A recent meta-analysis13 that included 10 studies on the effect of DCB on the SB concluded that such technique improved the angiographic outcomes significantly. However, this did not translate either into statistically significant clinical outcomes (target lesion failure mainly) basically due, according to the authors, to the low rate of this adverse event reported, and the fact that the study was statistically underpowered due to its small sample size. In a different study published in 2022,14 the management of coronary bifurcation lesions of left main coronary artery using 2 strategies was compared: double stenting for the MB and the SB vs 1 stent into the MB, and 1 DCB into the SB. They found controversial results at follow-up between both groups in different angiographic parameters with similar rates of restenosis and adverse events. However, the group treated with DCB significantly improved all the parameters associated with the SB (left circumflex artery, in this study)—as opposed to those associated with the MB (left main coronary artery-left anterior descending coronary artery)—with less late lumen loss (0.43 vs -0.17; P < .001), less luminal narrowing (16.7 vs 32.1; P = .002), and greater minimal lumen diameter (2.4 vs 1.8; P = .0031). Still, the rate of restenosis in the left circumflex artery (SB in this study) was 4 times higher in the double stenting group compared to the DCB group (30.4% vs 7.7%) although this difference was not statistically significant (P = .09). This could be indicative of greater superiority of the DCB if studies with larger samples would be conducted. Another recent study published in 202115 randomized 219 true de novo coronary bifurcation lesions where the SB was treated with conventional balloon vs DCB. At 12-month clinical and angiographic follow-up, significant improvements were reported in both the angiographic (less late lumen loss and greater minimal lumen diameter) and clinical parameters with a lower rate of major adverse cardiovascular events being reported. This improvement, however, did not translate into significant reductions regarding new revascularizations or target vessel failure.
The rate of target lesion failure requiring new revascularization was 3.6%, a figure that is consistent with most former studies. However, the range found goes from a surprising 0% up to a whopping 22%. However, we should mention the truly unfavorable clinical and anatomical profile of our sample since in most clinical trials, ISR-type of lesions, left main coronary artery disease or ST-segment elevation acute coronary syndrome—all allowed in our registry—were considered exclusion criteria regarding.
Out of the only 2 cases reported of target lesion failure requiring new revascularization, 1 occurred in a patient with an ISR-type of lesion. This occurred precisely in the SB while in former studies7—as already explained—the main incidence rate of failure occurred in the MB, not in the SB. The exclusion of patients with ISR would account for this difference. In our sample this type of lesions were 32.7% of all the lesions included. This added to the high rate (30.6%) of Medina 1,1,1 coronary bifurcation lesions (the one with the greatest complexity of all bifurcations) demonstrates the truly unfavorable profile of our sample. As a matter of fact, the rate of lesions included with damage to, at least, 2 segments of 1 bifurcation according to the Medina classification reached 71.4%. Very few studies have been conducted on this subgroup of patients. One of the most significant ones is the one conducted by Harada et al.16 that included 177 patients with ISR-type of lesions both in the MB and the SB treated with DCB. The latter was used in 80.6% of the SBs. The rate of binary restenosis was 24% at 6-to-8-month angiographic follow-up while the 1-year rate of new target lesion revascularization was 22%.
Limitations
Our study main limitation is the lack of a control group with lesions of similar characteristics, which would have allowed us to compare both groups and determine exactly the impact DCB has on the prognosis of patients. Similarly, the lack of angiographic follow-up does not discard the possibility of device failure. However, this would probably occur in the SB, not the MB, since it is in the latter where target lesion failure occurs according to the BABILON clinical trial.7 Another study limitation we should take into consideration is the elevated presence of small SB with a rate of use, in our sample, of DCB sizes < 2.25 mm of 43.7%. This would make target lesion failure go clinically inadvertently in some of these cases. Finally, we should mention that this study is limited by the relatively small number of patients included. Also, because due to its observational nature, no selection biases can be excluded.
CONCLUSIONS
The findings presented here show the experience of a single center with a very low rate of acute procedural complications, and a low rate of long-term adverse events despite dealing very high-risk profile lesions and patients with a 3.6% rate of target lesion failure reported. It is crucial to select the right type of lesions that can benefit from such therapy (basically the lack of a large plaque burden in the SB), a very refined technique of lesion preparation, and a greater use of tools to guide the angioplasty like intracoronary ultrasound or optical coherence tomography, preferably in ISR-type of lesions whose clinical progression is more unfavorable compared to that of de novo lesions. Despite the low rate of adverse events reported since no comparison with a control group was made, no definitive conclusions can be drawn on the advantages of DCBs in this clinical setting. We can only say that both in the «real-world» and the routine clinical practice described here, such strategy yields good long-term results without prejudice to other strategies may have given better or worse results regarding effectiveness. Randomized clinical trials are needed with enough statistical power and large enough samples to corroborate the promising data obtained from former studies to confirm or discard the superiority of DCB in the management of the SB in coronary bifurcation lesions. Until that time, the DCB can be considered a therapeutic tool that can be tremendously useful to improve the long-term results obtained in this type of complex lesions.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
J. Valencia drafted the manuscript. J. Valencia, F. Torrez-Mezcua, and M. Herrero-Brocal participated in data curation, and in the clinical follow-up of the patients. J. Valencia, J. Pineda, P. Bordes, F. Torres-Saura, and J.M. Ruiz-Nodar participated in patient recruitment and in the manuscript critical review process. All the authors gave their final approval to the manuscript.
CONFLICTS OF INTEREST
None reported.
WHAT IS KNOWN ABOUT THE TOPIC?
- To this date, there is a limited number of studies that have analyzed the role of DCB to treat the SB of coronary bifurcation lesions. Although such role seems beneficial regarding the improvement of the parameters analyzed, this still has not translated into a clear significant improvement of clinical parameters like target lesion/vessel failure or need for new revascularizations. On the other hand, the exact relevance of the SB and the role it plays in the short- and long-term prognosis of coronary bifurcation lesions remains unknown.
WHAT DOES THIS STUDY ADD?
- Our registry provides the experience of a single large volume center treating this type of lesions with a long follow-up. Also, it represents the «real-world» setting, that has been considered cut off from large randomized clinical trials too many times. The favorable results obtained in our study in a very unfavorable clinical and anatomical setting can situate the DCB as an extremely useful tool to improve the long-term results of percutaneous coronary interventions performed on coronary bifurcation lesions at our cath labs.
REFERENCES
1. Sawaya FJ, Lefèvre T, Chevalier B, et al. Contemporary Approach to Coronary Bifurcation Lesion Treatment. JACC Cardiovasc Interv. 2016;9:1861-1878.
2. Spitzer A, McFadden E, Vranckx P, et al. Critical Appraisal of Contemporary Clinical Endpoint Definitions in Coronary Intervention Trials: A Guidance Document. JACC: Cardiovasc Interv. 2019;12:805-819.
3. Jeger RV, Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease: Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13:1391-1402.
4. Oh GC, Park KW, Kang J, et al. Association of Side-Branch Treatment and Patient Factors in Left Anterior Descending Artery True Bifurcation Lesions: Analysis from the GRAND-DES Pooled Registry. J Interv Cardiol. 2020. https://doi.org/10.1155/2020/8858642.
5. Mori H, Torii S, Harari E, et al. Pathological mechanisms of left main stent failure. Int J Cardiol. 2018;263:9-16.
6. Stella PR, Belkacemi A, Dubois C, et al. A multicenter randomized comparison of drug-eluting balloon plus bare-metal stent versus bare-metal stent versus drug-eluting stent in bifurcation lesions treated with a single-stenting technique: six-month angiographic and 12-month clinical results of the drug-eluting balloon in bifurcations trial. Catheter Cardiovasc Interv. 2012;80:1138-1146.
7. López Mínguez JR, Nogales Asensio JM, Doncel Vecino LJ, et al. A prospective randomised study of the paclitaxel-coated balloon catheter in bifurcated coronary lesions (BABILON trial): 24-month clinical and angiographic results. EuroIntervention. 2014;10:50-7.
8. Berland J, Lefèvre T, Brenot P, et al. DANUBIO — a new drug-eluting balloon for the treatment of side branches in bifurcation lesions: six-month angiographic follow-up results of the DEBSIDE trial. EuroIntervention. 2015;11:868-876.
9. Herrador JA, Fernandez JC, Guzman M, Aragon V. Drug-eluting vs. conventional balloon for side branch dilation in coronary bifurcations treated by provisional T stenting. J Interv Cardiol. 2013;26:454-462.
10. Mathey DG, Wendig I, Boxberger M, Bonaventura K, Kleber FX. Treatment of bifurcation lesions with a drug-eluting balloon: the PEPCAD V (Paclitaxel Eluting PTCA Balloon in Coronary Artery Disease) trial. EuroIntervention. 2011;7 Suppl K:K61-65.
11. Kleber FX, Rittger H, Ludwig J, et al. Drug eluting balloons as stand alone procedure for coronary bifurcational lesions: results of the randomized multicenter PEPCAD-BIF trial. Clin Res Cardiol. 2016;105:613-621.
12. Jing QM, Zhao X, Han YL, et al. A drug-eluting Balloon for the trEatment of coronarY bifurcatiON lesions in the side branch: a prospective multicenter ranDomized (BEYOND) clinical trial in China. Chin Med J. 2020;133:899-908.
13. Zheng Y, Li J, Wang L, et al. Effect of Drug-Coated Balloon in Side Branch Protection for de novo Coronary Bifurcation Lesions: A Systematic Review and Meta-Analysis. Front Cardiovasc Med. 2021;8:758560.
14. Liu H, Tao H, Han X, et al. Improved Outcomes of Combined Main Branch Stenting and Side Branch Drug-Coated Balloon versus Two-Stent Strategy in Patients with Left Main Bifurcation Lesions. J Interv Cardiol. 2022. https://doi.org/10.1155/2022/8250057.
15. Li Y, Mao Q, Liu H, Zhou D, Zhao J. Effect of Paclitaxel-Coated Balloon Angioplasty on Side Branch Lesion and Cardiovascular Outcomes in Patients with De Novo True Coronary Bifurcation Lesions Undergoing Percutaneous Coronary Intervention. Cardiovasc Drugs Ther. 2021. https://doi.org/10.1007/s10557-021-07225-8.
16. Harada Y, Colleran R, Pinieck S, et al. Angiographic and clinical outcomes of patients treated with drug-coated balloon angioplasty for in-stent restenosis after coronary bifurcation stenting with a two-stent technique. EuroIntervention. 2017;12:2132-2139.
- EPIC03-BIOSS observational prospective study. Performance analysis of the BIOSS LIM C dedicated stent in coronary bifurcation lesion angioplasty
- Clinical impact of complete revascularization on real-life diabetic patients
- Rationale and study design on the effectiveness of vasodilators and topical local anesthetics to prevent radial spasm. The E-RADIAL trial
- Effectiveness of spherical tip noncompliant balloon for stent postdilatation: the REPIC02-RECONQUISTHA study
Interviews
An interview with Bruno Scheller
aServicio de Cardiología, Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria de La Princesa (IIS-IP), Universidad Autónoma de Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain