Article
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España

ABSTRACT
Introduction and objectives: Ultrathin-strut stents (UTS) represent a significant advancement in percutaneous coronary intervention. This study aimed to evaluate the safety and short- to mid-term outcomes of stenting with the thinnest struts on the market (50 μm) using a biodegradable everolimus-eluting polymer (Evermine 50) in real-world patients with coronary artery disease.
Methods: A single-arm, multicenter, prospective study was conducted in real-world patients. A total of 161 patients with de novo lesions who received at least 1 UTS stent were enrolled. The primary safety endpoint was the occurrence of major adverse cardiovascular events, defined as cardiac death, target-vessel myocardial infarction, or the need for revascularization of the target lesion at 12 months. The incidence of stent thrombosis at 12 months was also analyzed.
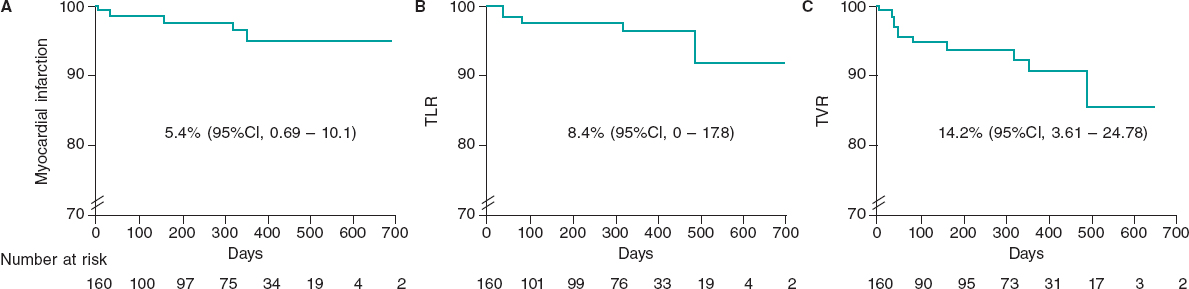
Results: The study included 161 patients with a mean age of 64 ± 14 years; 79% were male, 34% had diabetes, and 66% had hypertension. The most common indication for intervention was non-ST-segment elevation myocardial infarction (42%), followed by ST-segment elevation myocardial infarction (22%). The procedural success rate was 100%. At 12 months of follow-up, the incidence of MACE was 2.5%, and the definite stent thrombosis rate was 1.3%.
Conclusions: The use of the 50 μm UTS stent with a biodegradable everolimus-eluting polymer demonstrated a favorable safety profile and good clinical outcomes in unselected patients at 1 year of follow-up.
Keywords: Coronary artery disease. Percutaneous coronary intervention. Ultrathin struts.
RESUMEN
Introducción y objetivos: Los stents de struts ultrafinos (SUF) constituyen una mejora en el campo del intervencionismo coronario percutáneo. El objetivo de este estudio fue evaluar la seguridad y los resultados a corto y medio plazo del stent con los struts más finos del mercado (50 μm), con polímero biodegradable y liberador de everolimus (Evermine 50), en pacientes del mundo real con enfermedad coronaria.
Métodos: Se diseñó un estudio prospectivo, multicéntrico, de un solo grupo, en pacientes del mundo real. Se incluyeron 161 pacientes con lesiones de novo en los que se implantó al menos 1 stent de SUF. La variable principal de seguridad fueron los eventos adversos cardiovasculares mayores, compuesto de muerte cardiaca, infarto de miocardio atribuido al vaso diana y necesidad de revascularización de la lesión diana a los 12 meses de seguimiento. También se analizó la incidencia de trombosis del stent a los 12 meses del procedimiento.
Resultados: De los 161 pacientes incluidos (edad media 64 ± 14 años; 79% varones), el 34% eran diabéticos y el 66% eran hipertensos. La indicación más frecuente fue infarto sin elevación del segmento ST (42%), con un 22% de casos en contexto de infarto con elevación del segmento ST. El porcentaje de éxito del procedimiento fue del 100%. A los 12 meses de seguimiento, la incidencia de eventos adversos cardiovasculares mayores fue del 2,5%, con una tasa de trombosis del stent definitiva del 1,3%.
Conclusiones: El uso de stent con SUF de 50 μm, con polímero biodegradable y liberador de everolimus en pacientes no seleccionados mostró unos buenos resultados clínicos, así como un buen perfil de seguridad a 1 año de seguimiento.
Palabras clave: Enfermedad coronaria. Intervencionismo coronario percutaneo. Strut ultrafino.
Abbreviations
MACE: major adverse cardiovascular events. MI: myocardial infarction. PCI: percutaneous coronary intervention. ST: stent thrombosis. STEMI: ST-segment elevation myocardial infarction. UTS: ultra-thin strut.
INTRODUCTION
Percutaneous coronary intervention (PCI) has grown exponentially along with the technological evolution associated with this procedure. The continuous advancement of technology has enabled the development of stents with thinner struts, which offer a series of advantages over stents with thicker struts. One of the advantages of these new stents is the improved device profile—with increased flexibility—providing better navigability and greater lesion crossing capability. On the other hand, ultra-thin struts (UTS) cause fewer disturbances to normal laminar blood flow at target lesion level, due to the reduced protrusion of material into the vascular lumen. This seems to be associated with a lower degree of platelet activation and muscle cell proliferation,—the processes involved in stent failure—in terms of stent thrombosis (ST) and in-stent restenosis.1,2 In lesions located in small caliber vessels (≤ 2.5 mm), the use of UTS could provide additional advantages due to a higher ratio between the size of the struts and the lesion luminal area.3 Furthermore, UTS stents seem to be associated with less acute damage to the vascular endothelium during stent deployment. This reduced initial aggression could diminish the barotrauma-related inflammatory response and, therefore, prevent in-stent restenosis and promote faster device endothelialization.4,5 Studies have indicated that the use of UTS stents could be associated with lower rates of in-stent restenosis and a reduced need for new revascularizations.6,7
The Evermine 50 EES stent (Meril Life Sciences, India) is a UTS (50 μm) stent with CE marking consisting of a cobalt-chromium alloy platform with an everolimus-eluting biodegradable polymer. The aim of this study was to evaluate the 1-year safety and efficacy outcomes after UTS stent deployment in real-world patients with coronary artery disease.
METHODS
We conducted a prospective, non-randomized, multicenter study with patients who underwent UTS stent deployment at 4 different Spanish hospitals (data from the Everythin Registry). To be included in the study, patients had to be older than 18 years, with available coronary angiographies in the context of chronic or acute coronary syndrome, and have, at least, 1 target lesion with a 2 mm up to 4.5 mm reference vessel diameter on visual estimation. Overlapping stents was ill-advised and, if necessary, the overlap length should be ≤ 2 mm. PCI in multiple vessels and lesions during the same surgical act was allowed, and deferred procedures within the first 90 days since the initial procedure were also accepted. In these cases, any further procedures were not coded as an event—i.e. need for new revascularization—but as scheduled procedures. Only 1 case—1 target lesion treated with UTS stent deployment—was counted per patient. Deploying the study UTS stent was not mandatory in any of the other treated lesions, only in the target lesion/vessel.
The study followed the privacy policy of each research center, including regulations for the appropriate use of data from patient research. The study was approved by the Ethics Committee for Drug Research of the coordinating center. Moreover, the study was conducted in full compliance with the terms set forth in the Declaration of Helsinki. All patients signed specific informed consent forms prior to being included in the study.
Study device and procedure
The Evermine 50 EES (Meril Life Sciences, India) is a UTS (50 μm) stent with a cobalt-chromium platform coated with a biodegradable polymer composed of poly-L-lactic acid and poly(lactic-co-glycolic) acid. The Evermine stent—which has a hybrid design with an open cell in its central part and a closed cell at the edges—releases everolimus (1.25 μg/mm²) as the antiproliferative drug. The stent has received the corresponding CE marking and is available in several lengths from 8 mm up to 48 mm with diameters ranging from 2 mm up to 4.5 mm. The main features of the Evermine 50 EES device are illustrated in figure 1.

Figure 1. A: illustrative image of the Evermine 50 stent (Meril Life Sciences, India). B: description of the main characteristics of the stent. C: comparison of the study stent strut thickness vs major competing next-generation stents. PLGA, poly(lactic-co-glycolic acid); PLLA, poly-L-lactic acid (Images courtesy of Meril Life Sciences. Images reproduced with permission from Meril Life Sciences or its affiliates.)
PCI was performed following each center routine practice within the recommendations outlined in the clinical practice guidelines.8 The PSP algorithm (predilation, sizing [stent size selection], and postdilation) was recommended for optimal device implantation. The study protocol recommended postdilation, especially in cases where any degree of underexpansion was identified immediately after device implantation. Although the study protocol recommended the use of intravascular imaging modalities to guide the procedure, this was left to the operator’s discretion. All patients received a 300 mg loading dose of acetylsalicylic acid prior to the intervention followed by a loading dose of a second antiplatelet agent—clopidogrel, prasugrel, or ticagrelor—after the PCI, which was maintained for 3 up to 12 months and left to the discretion of the responsible investigator of the center.
Endpoints and definitions
The primary endpoint of the study was the occurrence of major adverse cardiovascular events (MACE) at 12 months. MACE were defined as the composite of cardiac death, non-fatal target vessel myocardial infarction (MI), or the need for target lesion revascularization. Secondary endpoints included each individual component of the composite endpoint, the overall mortality and ST (both definite and probable) according to the definitions of the Academic Research Consortium9 12 months after implantation. Additionally, the rates of device and procedural success were taken into consideration. Device success was defined as the deployment of the study stent in the target lesion with a final percent diameter residual stenosis < 30% by visual estimation. Procedural success was defined as the success of the device without any in-hospital complications, including death, MI, and target lesion revascularization.
Statistical analysis
Quantitative variables are expressed as mean and standard deviation or as median and interquartile range [IQR], depending on their distribution. Categorical variables are expressed as number and percentage. All analyses were performed using the statistical tool STATA 12 (StataCorp LLC, United States).
RESULTS
Demographic and baseline clinical characteristics
A total of 161 patients were included in the study from November 2020 through April 2022 whose demographic data and clinical characteristics are shown in table 1. The mean age was 64 ± 14 years, and 79% were male. A total of 66% of the patients were hypertensive; 53% had dyslipidemia; 34%, diabetes mellitus, and 59% a history of smoking. A total of 20% of the patients had experienced a prior MI, and 22% a previous PCI. The most common indication for the intervention was the diagnosis of non-ST-segment elevation acute myocardial infarction (42%), followed by ST-segment elevation myocardial infarction (STEMI) (22%) and chronic coronary syndrome (21%).
Table 1. Baseline characteristics of the study population
| Basal characteristics | Patients (n = 161) |
|---|---|
| Age (years) ± SD | 64 ± 14 |
| Male, n (%) | 126 (79) |
| BMI (kg/m²) | 28 ± 3.5 |
| Hypertension, n (%) | 106 (66) |
| Dyslipidemia, n (%) | 86 (53) |
| Diabetes mellitus, n (%) | 55 (34) |
| Smoking status, n (%) | |
| Non-smoker | 65 (40) |
| Former smoker | 49 (30) |
| Current smoker | 47 (29) |
| Previous AMI, n (%) | 33 (20) |
| Previous stroke, n (%) | 2 (1.2) |
| Atrial fibrillation, n (%) | 7 (4.3) |
| Peripheral vascular disease, n (%) | 10 (6.2) |
| Previous coronary angioplasty, n (%) | 36 (22) |
| Previous coronary artery bypass grafting, n (%) | 4 (2.5) |
| COPD, n (%) | 13 (8) |
| Chronic kidney disease, n (%) | 14 (9) |
| Glomerular filtration rate (mL/min/1.73 m²) | 61 ± 10 |
| Left ventricular function, (%) | 55 ± 11 |
| Indication for coronary angiography, n (%) | |
| Chronic coronary syndrome | 34 (21) |
| Unstable angina | 24 (15) |
| NSTEMI | 67 (42) |
| STEMI | 36 (22) |
|
AMI, acute myocardial infarction; BMI, body mass index; COPD, chronic obstructive pulmonary disease; NSTEMI, non-ST-segment elevation acute myocardial infarction; SD, standard deviation; STEMI, ST-segment elevation myocardial infarction. |
|
Angiographic and procedural characteristics
The lesion angiographic characteristics, and the results of the intervention are shown in table 2. Most patients had significant single-vessel disease (71%), being the presence of 2 or 3-vessel disease far less common (20% and 9%, respectively). The most widely treated vessel was the left anterior descending coronary artery (54%), followed by the right coronary artery (27%) and the left circumflex artery (17%). The target lesion median percent diameter stenosis by visual estimation was 90% [IQR, 75-99]. A total of 29% of the target lesions showed some degree of calcification on angiography. Intracoronary imaging modalities (7% optical coherence tomography) were used to guide the PCI in 11% of the cases. The mean number of stents deployed per lesion was 1.04 ± 0.22, with a median stent diameter of 3.0 mm [IQR 2.75-3.5] and a median stent length of 19 mm [IQR 19-24]. Pre- and postdilation were performed in 71% and 39% of the cases, respectively. The device and procedural success rates were 100%, without any procedure-related complications being reported in patients treated during the inpatient period.
Table 2. Angiographic, procedural and clinical follow-up characteristics of the cohort
| Angiographic and procedural characteristics | Patients (n = 161) |
|---|---|
| Radial access, n (%) | 158 (98) |
| Diseased vessels, n (%) | |
| 1-vessel disease | 114 (71) |
| 2-vessel disease | 32 (20) |
| 3-vessel disease | 15 (9) |
| Target lesion location, n (%) | |
| Left main coronary artery | 3 (1.8) |
| Proximal left anterior descending coronary artery | 37 (23) |
| Mid left anterior descending coronary artery | 40 (24.8) |
| Distal left anterior descending coronary artery | 10 (6.2) |
| Proximal left circumflex artery | 10 (6.2) |
| Mid left circumflex artery | 11 (6.8) |
| Distal left circumflex artery | 6 (3.7) |
| Proximal right coronary artery | 13 (8) |
| Mid right coronary artery | 18 (11.2) |
| Distal right coronary artery | 13 (8) |
| Bifurcation lesions, n (%) | 12 (7.5) |
| Calcified lesions, n (%) | 46 (29) |
| Visual percent diameter stenosis, median [IQR] | 90 [75-99] |
| Predilation, n (%) | 114 (71) |
| Postdilation, n (%) | 63 (39) |
| Intracoronary imaging modalities, n (%) | 18 (11) |
| Optical coherence tomography | 11 (7) |
| Intravascular ultrasound | 7 (4) |
| No. of stents deployed, mean ± SD | 1.04 ± 0.22 |
| Stent diameter (mm), median [IQR] | 3.0 [2.75-3.5] |
| Stent length (mm), median [IQR] | 19 [19-24] |
| Device success, n (%) | 161 (100) |
| Procedural success, n (%) | 161 (100) |
| Antiplatelet therapy after PCI, n (%) | |
| Acetylsalicylic acid | 161 (100) |
| Clopidogrel | 78 (48) |
| Ticagrelor | 68 (42) |
| Prasugrel | 15 (9) |
| Clinical follow-up | |
| 12-month follow-up, n (%) | 158 (98) |
| MACE, n (%) | 4 (2.5) |
| Cardiac death | 1 (0.6) |
| Target vessel MI | 2 (1.3) |
| Target lesion revascularization | 2 (1.3) |
| Overall mortality, n (%) | 3 (1.9) |
| Stent thrombosis, n (%) | |
| Definite | 2 (1.3) |
| Probable | 1 (0.6) |
|
IQR, interquartile range; MACE, major adverse cardiovascular events; MI, myocardial infarction; PCI, percutaneous coronary intervention; SD, standard deviation. |
|
Clinical outcomes at the follow-up
The 12-month follow-up was completed in 158 patients (98%). One year after implantation, 4 patients exhibited MACE (2.5%), and 3 patients died (1.9%). The cause of death was cardiac in 1 patient (due to a probable ST 7 days after the procedure) and non-cardiac in the remaining 2 (one due to lung neoplasm and the other to multiple organ failure). There were 2 non-fatal MIs (1.3%), both due to late definite ST (1 occurred 8 months after stent deployment and was associated with the study UTS stent, while the other one occurred 9 months after deployment due to a different thrombosed non-UTS stent implanted in a lesion of the target lesion same vessel. Only 2 patients required target lesion revascularization at the follow-up (1 due to ST and the other one due to in-stent restenosis).
DISCUSSION
The present study prospectively and multicentrically evaluates the safety and efficacy profile of implanting an UTS stent in a real-world population. Its main findings are that the UTS stent demonstrated a high procedural success rate, without in-hospital complications, acceptable midterm clinical outcomes, and a 2.5% rate of MACE 12 months after implantation.
The baseline characteristics of the study population are similar to the ones reported in previous studies that analyzed various stent technologies in patients with atherosclerotic coronary artery disease.10-12 However, it is noteworthy that in this study, 79% of cases were performed in the context of an acute coronary syndrome, including 22% of patients diagnosed with STEMI. In acute coronary syndrome—especially STEMI—there are factors associated with poorer outcomes of the implanted device, both in the short and long term. Firstly, the state of generalized vasoconstriction of the coronary tree and high thrombotic burden can complicate the appropriate selection of the size of the stent, thus leading to the implantation of smaller devices in relation to the actual size of the vessel, a mechanism involved in ST and in-stent restenosis. Furthermore, in the context of acute lesions, there is a higher risk of embolization and no-reflow or slow-flow phenomena, which can sometimes condition suboptimal final outcomes in terms of distal coronary flow, involving a greater risk of further ST. In our study, no ST occurred in patients with an early diagnosis of STEMI. Although it is worth mentioning that the results of the study stent were good—even in demanding contexts such as STEMI—the absolute number of STEMI patients included was low, meaning that data should be contrasted in larger series.
UTS stents provide better navigability, flexibility, and conformity to the vessel geometry. However, there may be doubts on whether the presence of UTS can lead to a reduction of the stent radial strength, which could have further implications for treating more unfavorable lesions, such as calcified lesions. Although, in the present study, 29% of the treated lesions showed some degree of calcification on angiography, the success rate of the stent reached 100%. This demonstrates the good performance of this UTS stent across different scenarios, achieving excellent radial strength even in the most challenging situations, such as calcified coronary lesions. These results are especially relevant in the specific context of the study, where, despite the recommendation for systematic postdilation, the final rate of stent postdilation was relatively low (39%).
Previous studies have consistently shown good clinical follow-up results for UTS stents with low rates of ST.13-15 The reason for this low rate of ST would be strut thickness per se, which would favor early neointimal coverage, thereby reducing the risk of ST (especially late and very ST).4 In the specific case of the study device (Evermine 50 EES), Patted et al.13 described the 6-month follow-up results of 251 patients. In this single-center, prospective experience, the authors describe a 6-month rate of MACE of 0.8%, with no ST at the follow-up. Regarding differences with respect to our series, nearly one-third of the cases were procedures in asymptomatic patients or with silent ischemia. Additionally, the rate of postdilation (57%) was higher than that of our cohort, which may have influenced the ST outcomes. The same group retrospectively described the results of 171 patients treated with the Evermine 50 EES stent,16 with 2-year rates of procedural success and MACE of 100% (same as in our study) and 2.4%, respectively. Again, the authors noted the absence of definite or probable ST at the follow-up. In this single-center cohort, the rate of stent postdilation was not reported, which may have implications for the prevention of MACE, especially ST. A meta-analysis that analyzed various types of UTS stents found no significant differences in the likelihood of stent failure, including ST across different stents with struts < 70 μm.17 In the present study, although the 1-year rate of definite ST after stent deployment was 1.3%, only 1 of these STs was attributed to the study device. The rate of ST is similar to that of other real-world experiences with second and third-generation stents,18-20 which confirms the good performance of the Evermine 50 EES in unselected real-world patients.
Limitations
The main limitations of the study are the relatively low number of patients included, and the absence of a comparator group. Furthermore, although the events reported at the follow-up were reviewed by the principal investigator of the coordinating center based on the case reports submitted by each principal investigator from the collaborating centers, these events were not allocated by an independent event adjudication committee. The fact that, in our cohort, few intracoronary imaging modalities were used to guide the PCI—reflecting real clinical practice—could be interpreted as a limitation of the study.
CONCLUSIONS
With data from a prospective, multicentric study of real-world patients, the PCIs performed with a 50 μm UTS stent, with a biodegradable polymer and everolimus elution had good clinical outcomes and a favorable safety profile at the 12-month follow-up.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The study was approved by the Drug Research Ethics Committee of the coordinating center. The study was conducted in full compliance with the terms outlined in the Declaration of Helsinki. All patients signed specific informed consent forms prior to the intervention and before being included in the study.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used for this work.
AUTHORS’ CONTRIBUTIONS
J. Casanova-Sandoval and M. García-Guimarães participated in the conception and design of the study, analysis and interpretation of results, and drafting the manuscript. G. Miñana Escrivà, E. Bosch-Peligero, J.F. Muñoz-Camacho, D. Fernández-Rodríguez, K. Rivera, A. Fernández-Cisnal, and D. Valcárcel-Paz participated in data acquisition and critically reviewed the content of the manuscript. All authors gave their final approval for the publication of the latest draft of the manuscript.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- The use of UTS stents may be associated, through various mechanisms, with better clinical outcomes compared with thicker-strut stents. Previous studies suggest that UTS stents are associated with less stent failure, preventing in-stent restenosis and ST.
WHAT DOES THIS STUDY ADD?
- In this prospective, multicentric study of real-world patients, the use of a 50 μm UTS stent with a biodegradable polymer and everolimus elution was associated with good clinical outcomes, and a favorable safety profile at the 12-month clinical follow-up.
REFERENCES
1. Duraiswamy N, Schoephoerster RT, Moreno MR, Moore JE. Stented artery flow patterns and their effects on the artery wall. Annu Rev Fluid Mech. 2007;39:357-382.
2. Chiu JJ, Chien S. Effects of disturbed flow on vascular endothelium:Pathophysiological basis and clinical perspectives. Physiol Rev. 2011;91:327-387.
3. Buiten RA, Ploumen EH, Zocca P, et al. Outcomes in Patients Treated with Thin-Strut, Very Thin-Strut, or Ultrathin-Strut Drug-Eluting Stents in Small Coronary Vessels:A Prespecified Analysis of the Randomized BIO-RESORT Trial. JAMA Cardiology. 2019;4:659-669.
4. Miura T, Ueki Y, Senda K, et al. Early vascular response of ultra-thin bioresorbable polymer sirolimus-eluting stents assessed by optical frequency domain imaging:the EVALUATION study. Cardiovasc Interv Ther. 2021;36:281-288.
5. Otaegui Irurueta I, González Sucarrats S, Barrón Molina JL, et al. Can an ultrathin strut stent design and a polymer free, proendothelializing probucol matrix coating improve early strut healing?The FRIENDLY-OCT trial. An intra-patient randomized study with OCT, evaluating early strut coverage of a novel probucol coated polymer-free and ultra-thin strut sirolimus-eluting stent compared to a biodegradable polymer sirolimus-eluting stent. Int J Cardiol. 2022;360:13-20.
6. Kastrati A, Mehilli J, Dirschinger J, et al. Intracoronary stenting and angiographic results:Strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation. 2001;103:2816-2821.
7. Farb A, Weber DK, Kolodgie FD, Burke AP, Virmani R. Morphological predictors of restenosis after coronary stenting in humans. Circulation. 2002;105:2974-2980.
8. Neumann F-J, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
9. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials. Eur Heart J. 2018;39:2192-2207.
10. Nakamura M, Kadota K, Nakagawa Y, et al. Ultrathin, Biodegradable-Polymer Sirolimus-Eluting Stent vs Thin, Durable-Polymer Everolimus-Eluting Stent. JACC Cardiovasc Interv. 2022;15:1324-1334.
11. de la Torre Hernández JM, Ocaranza Sanchez R, Santas Alvarez M, et al. Comparison of One-Year Outcomes Between the ihtDEStiny BD Stent and the Durable-Polymer Everolimus- and Zotarolimus-Eluting Stents:A Propensity-Score-Matched Analysis. Cardiovasc Revasc Med. 2021;31:1-6.
12. De Silva K, Li Kam Wa ME, Wells T, et al. The everolimus eluting Synergy MegatronTM drug-eluting stent platform:Early outcomes from the European Synergy MegatronTM Implanters'Registry. Catheter Cardiovasc Interv. 2023;102:1222-1228.
13. Patted SV, Patted AS, Turiya PK, Thakkar AS. Clinical Outcomes of World's Thinnest (50 µm) Strut Biodegradable Polymer Coated Everolimus-Eluting Coronary Stent System in Real-World Patients. Cardiol Res. 2018;9:370-377.
14. Araujo GN, Machado GP, Moura M, et al. Real-World Assessment of an Ultrathin Strut, Sirolimus-Eluting Stent in Patients with ST-Elevation Myocardial Infarction Submitted to Primary Percutaneous Coronary Intervention (INSTEMI Registry). Arq Bras Cardiol. 2023;120:e20220594.
15. Kasturi S, Polasa S, Sowdagar MA, et al. Safety and Clinical Performance of Biodegradable Polymer-Coated Ultra-Thin Everolimus-Eluting Stents in “Real-World“Patients:A Multicenter Registry (PERFORM-EVER). Anatol J Cardiol. 2022;26:619-628.
16. Patted SV, Thakkar AS. Clinical outcomes of ultrathin strut biodegradable polymer-coated everolimus-eluting stent in patients with coronary artery disease. ARYA Atheroscler. 2020;16:130-135.
17. Marengo G, Bruno F, Scudeler L, et al. Comparison Among Ultra-Thin Coronary Stents:A Network Meta-Analysis. Am J Cardiol. 2024;216:9-18.
18. Park S, Rha S-W, Choi BG, et al. Efficacy and Safety of Sirolimus-Eluting Stent with Biodegradable Polymer UltimasterTM in Unselected Korean Population:A Multicenter, Prospective, Observational Study From Korean Multicenter Ultimaster Registry. Korean Circ J. 2024;54:339-350.
19. Yu HY, Ahn J, Choi BG, et al. Three-Year Clinical Outcomes With the Cilotax Dual Drug-Eluting Stent vs Everolimus-Eluting Stents in Patients With Acute Myocardial Infarction. Texas Hear Inst J. 2024;51:e238271.
20. Nakao S, Ishihara T, Tsujimura T, et al. Two-year real world clinical outcomes after intravascular imaging device guided percutaneous coronary intervention with ultrathin-strut biodegradable-polymer sirolimus-eluting stent. Int J Cardiol. 2024;399:131686.
ABSTRACT
Introduction and objectives: The optimal treatment of nonculprit angiographic intermediate lesions (diameter stenosis 40%-69%) in patients with ST-segment elevation myocardial infarction (STEMI) is still unknown. Lesions with fractional flow reserve (FFR) ≤ 0.80 are indicative of ischemia and benefit from revascularization. However, lesions with FFR > 0.80 and optical coherence tomography (OCT) findings of vulnerability have been hypothesized to cause adverse events during follow-up. The study aims to compare the efficacy of a preventive treatment with stent implantation plus optimal medical therapy vs optimal medical therapy alone for nonculprit intermediate lesions with FFR > 0.80 and OCT findings of plaque vulnerability in STEMI patients at 4 years of follow-up.
Methods: This parallel-group, multicenter, controlled, single-blind, and 1:1 randomized trial will enroll a total of 600 STEMI patients with ≥ 1 intermediate nonculprit lesions with FFR > 0.80 and OCT findings of plaque vulnerability. The primary endpoint is target vessel failure, defined as the composite of cardiac death, target vessel myocardial infarction, or target vessel revascularization. The study will include a parallel registry of patients with FFR > 0.80 but without OCT findings of vulnerability. Vulnerable plaques are defined as lipid-rich fibroathermas with plaque burden ≥ 70% and a thin fibrous cap (≤ 80 μm).
Results: The VULNERABLE trial will reveal the role of preventive treatment with stent implantation for nonculprit and functionally nonsignificant vulnerable plaques in STEMI patients.
Conclusions: This is the first randomized trial of OCT-guided treatment of vulnerables plaques. Registered at ClinicalTrials.gov (NCT05599061).
Keywords: Fractional flow reserve. Optical coherence tomography. ST-segment elevation myocardial infarction. Vulnerable plaque.
RESUMEN
Introducción y objetivos: El tratamiento óptimo de las lesiones angiográficas intermedias (diámetro de estenosis 40-69%) no culpables en pacientes con infarto agudo de miocardio con elevación del segmento ST (IAMCEST) está por determinar. La reserva fraccional de flujo (RFF) permite diagnosticar lesiones causantes de isquemia (RFF ≤ 0,80) que se benefician de una revascularización. No obstante, las lesiones con RFF > 0,80 y criterios de vulnerabilidad por tomografía de coherencia óptica (OCT) también se ha hipotetizado que pueden causar eventos adversos en el seguimiento. El objetivo es comparar la eficacia del tratamiento preventivo con implantación de stent más tratamiento médico óptimo de lesiones intermedias no culpables con RFF > 0,80 y características de placa vulnerable frente a solo tratamiento médico óptimo en pacientes con IAMCEST a 4 años de seguimiento.
Métodos: Estudio de grupos paralelos, multicéntrico, controlado, aleatorizado 1:1 y simple ciego. Se incluirán 600 pacientes con IAMCEST y al menos una lesión intermedia no culpable que presenten RFF > 0,80 y características de placa vulnerable por OCT. El objetivo primario se define como fallo del vaso diana, compuesto de muerte cardiaca, infarto del vaso diana y necesidad de revascularización del vaso diana. El estudio incluye un registro paralelo para pacientes con RFF > 0,80 sin características de placa vulnerable. Se define placa vulnerable como fibroateromas lipídicos con carga de placa ≥ 70% y capa fibrosa fina (≤ 80 µm).
Resultados: El estudio VULNERABLE permitirá conocer el papel del tratamiento preventivo con stent de placas vulnerables no culpables funcionalmente no significativas en pacientes con IAMCEST.
Conclusiones: Se trata del primer estudio aleatorizado para el tratamiento de placas vulnerables guiado por OCT. Registrado en ClinicalTrials.gov (NCT05599061).
Palabras clave: Reserva fraccional de flujo. Tomografía de coherencia óptica. Infarto agudo de miocardio con elevación del segmento ST. Placa vulnerable.
Abbreviations
FFR: fractional flow reserve. MLA: minimum lumen area. OCT: optical coherence tomography. OMT: optimal medical therapy. PDE: percent diameter stenosis. STEMI: ST-segment elevation myocardial infarction.
INTRODUCTION
The presence of multivessel disease, defined as angiographic lesions with a percent diameter stenosis (PDS) ≥ 50% by visual estimation in patients with ST-segment elevation myocardial infarction (STEMI), is estimated to be approximately 50%.1 The COMPLETE trial compared angiography-guided preventive revascularization with stent implantation added to optimal medical therapy (OMT) for nonculprit lesions with a PDS ≥ 70% vs OMT alone.2 The trial found that angiography-guided preventive revascularization significantly reduced adverse cardiovascular events at 3 years of follow-up.2 Although the COMPLETE trial required physiological assessment using fractional flow reserve (FFR) for lesions with a PDS between 50% and 69% to guide the decision on revascularization, in practice, it was performed in only a very small percentage of patients.
The FLOWER-MI and FRAME-AMI trials3,4 investigated preventive stenting of FFR-guided nonculprit lesions—obtained through intracoronary pressure wire—compared with angiography-guided complete revascularization (visual estimation). Both trials mainly included intermediate lesions and demonstrated that pressure wire-guided preventive revascularization significantly reduces the need for revascularization, with similar or superior efficacy to angiography-guided complete revascularization.3,4 Despite these findings, clinical practice guidelines based on the COMPLETE trial recommend preventive stenting of nonculprit lesions guided by angiography alone.5,6
It is important to note that FFR is considered the gold standard for detecting myocardial ischemia (FFR ≤ 0.80). However, deferring treatment of nonculprit lesions that do not cause ischemia (FFR > 0.80) through OMT raises concerns in selected cases in which the anatomical features of the lesion suggest signs of vulnerability. In the FLOWER-MI trial, the group of patients randomized to undergo pressure-wire-guided revascularization with an FFR > 0.80 (referred for OMT) had more adverse events than those in the same group with FFR values ≤ 0.80 (referred for percutaneous revascularization).7 Several studies using intravascular imaging modalities have also demonstrated an association between the presence of fibro-lipid plaques with high lipid content and thin fibrous caps—known as vulnerable plaques—and the development of future adverse events due to plaque rupture.8,11
The VULNERABLE trial aims to evaluate the efficacy of a combined strategy using intracoronary physiological techniques and intravascular imaging to guide the treatment of intermediate nonculprit lesions in STEMI patients. The study hypothesis is that preventive stenting—in addition to OMT—in intermediate nonculprit lesions with FFR values > 0.80 and characteristics of vulnerable plaque will be superior to OMT alone. The present article includes the rationale and design of the study.
METHODS
Design
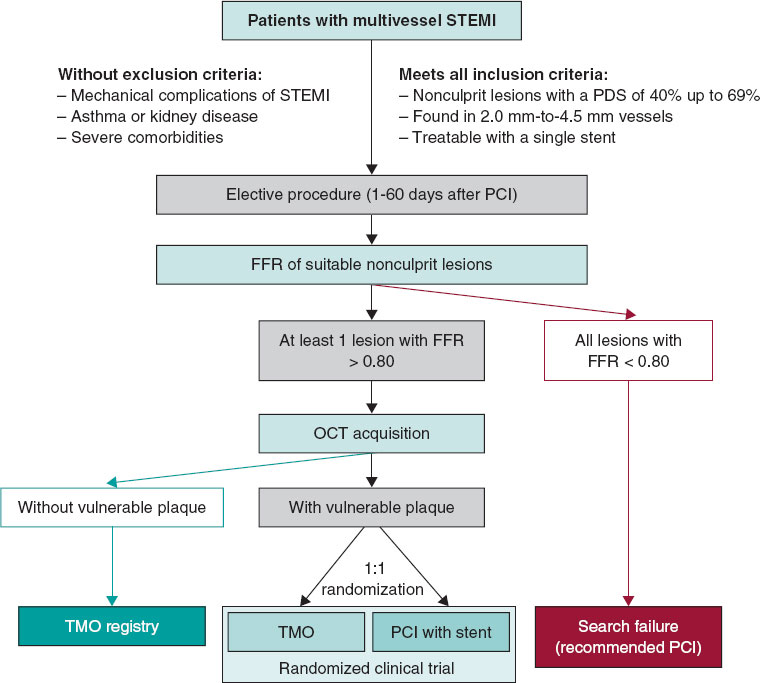
The VULNERABLE trial (NCT05599061) includes 3 groups based on the results obtained during the combined functional and anatomical assessment using pressure wires and optical coherence tomography (OCT). Figure 1 shows the study flowchart, which illustrates the 3 groups: patients with FFR ≤ 0.80 treated with stent (search failures), patients with FFR > 0.80 without vulnerable plaque characteristics (included in the registry group), and patients with FFR > 0.80 and vulnerable plaque characteristics (included in the randomized clinical trial).
This is a multicenter, controlled, prospective, randomized, parallel-group, single-blind study with patients included in the clinical trial group. The study will be conducted in accordance with the recommendations outlined in the Declaration of Helsinki on clinical research and has been approved by the lead ethics committee (Hospital Universitari de Bellvitge) and endorsed by the remaining ethics committees of participating centers. The participating centers and principal investigators are shown in table 1 of the supplementary data.
Table 1. Objectives of the VULNERABLE trial
| Primary endpoint |
|---|
| Compare the percentage of TVF between the 2 groups of patients assigned to the randomized clinical trial (FFR > 0.80 with characteristics of vulnerable plaque by OCT): preventive revascularization with stent + OMT vs OMT alone |
| Key secondary endpoints |
| Compare the percentage of TVF between patients allocated to the registry group (FFR > 0.80 without characteristics of vulnerable plaque by OCT and treated with the OMT) and patients allocated to the randomized OMT group (FFR > 0.80 with characteristics of vulnerable plaque) |
| Other secondary endpoints |
| Compare the rate of all-cause mortality reported between the 2 subgroups of randomized patients |
| Compare the percentage of cardiac deaths reported between the 2 subgroups of randomized patients |
| Compare the percentage of all myocardial infarctions reported between the 2 subgroups of randomized patients |
| Compare the percentage of target vessel myocardial infarctions reported between the 2 subgroups of randomized patients |
| Compare the percentage of target vessel revascularization needs between the 2 subgroups of randomized patients |
| Evaluate the percentage of restenosis and stent thrombosis in the preventive revascularization group with stent + OMT of the randomized clinical trial |
| * Although all objectives are marked with a complete 4-year follow-up, an interim study will be conducted at 2 years. ** All objectives will be calculated on an intention-to-treat basis according to the statistical plan. An exploratory per-protocol analysis will also be conducted based on the assessment by the study’s core imaging laboratory. |
|
FFR: fractional flow reserve; OCT: optical coherence tomograph; OMT: optimal medical treatment; TVF: target vessel failure. |
The study has been entirely designed and initiated by researchers and is sponsored by the Spanish Society of Cardiology Working Group on Intracoronary Diagnostic Techniques, which includes a steering committee, a data and safety monitoring board, and an independent event adjudication committee. The members of these committees are listed in table 2 of the supplementary data. The steering committee and all study investigators are committed to accurate data collection and adherence to the study protocol. The funding entity (Abbott Vascular, United States) plays no role in the study design, data collection, analysis, or the writing of the study results. The study sponsor (Foundation for Education in Interventional Cardiology Procedures [EPIC]), along with the principal investigators, is responsible for data management and confidentiality.
Table 2. Inclusion and exclusion criteria of the VULNERABLE trial
| Inclusion criteria |
|---|
| Patients older than 18 years |
| With STEMI (ST-segment elevation > 1 mm in, at least, 2 contiguous leads or true posterior ST-segment elevation with > 2 mm depression in anterior leads or new onset left bundle branch block) treated with successful revascularization of the culprit lesion within 72 hours from symptom onset |
| Presenting with multivessel disease with, at least, 1 angiographically intermediate lesion (PDS of 40% up to 69% by visual estimation) in a native vessel different from the culprit vessel |
| Planned FFR-guided percutaneous revascularization with a single 2.0 mm-to- 4.5 mm stent |
| Between 1 and 60 days after the index procedure (revascularization of the STEMI culprit vessel) |
| Exclusion criteria |
| Life expectancy < 4 years |
| Women of childbearing age who wish to become pregnant |
| Known intolerance to acetylsalicylic acid, heparin, everolimus, or iodinated contrast |
| Unresolved mechanical complications or infarct-related cardiogenic shock |
| Lesions suitable for the study located in the left main coronary artery, vessels with previous revascularization, in coronary bifurcations with > 2.5 mm side branches, severe angulations, or segments with severe calcification |
| History of severe asthma |
| Chronic kidney disease with glomerular filtration rate < 45 mL/min |
|
FFR: fractional flow reserve; PDS: percent diameter stenosis; STEMI: ST-segment elevation myocardial infarction. |
Endpoints
The primary objective of the VULNERABLE study (NCT05599061) is to compare the efficacy of preventive stenting combined with OMT vs OMT alone for intermediate lesions in noninfarct-related arteries with an FFR > 0.80 and vulnerable plaque characteristics as identified by OCT over a 4-year follow-up period. The primary endpoint of the study is the rate of target vessel failure (TVF), which is defined as a composite of cardiac death, target vessel myocardial infarction, or the need for target vessel revascularization.
The study also aims to evaluate several secondary endpoints, which are summarized in table 1. Among these secondary objectives, a key focus is the comparison of the TVF rate (the primary endpoint) between the registry group (patients with FFR > 0.80 without vulnerable plaque characteristics treated with OMT) and the randomized OMT arm of the clinical trial (patients with FFR > 0.80 and vulnerable plaque characteristics). The study endpoints are defined in table 3 of the supplementary data.12,13
Patient inclusion and exclusion criteria
The inclusion and exclusion criteria for the study are detailed in table 2. In brief, all patients with STEMI who have undergone successful revascularization of the culprit lesion and have at least 1 intermediate lesion (visually defined as having a DS of 40%-69%) in a noninfarct-related artery will be eligible for the study if percutaneous revascularization with a single stent guided by FFR is being considered. The study procedure must be conducted between 1 and 60 days after the revascularization of the culprit lesion. Patients must provide informed consent prior to the elective procedure for evaluating the nonculprit lesion.
Study protocol for nonculprit lesions and randomization
Eligible lesions will first be assessed with a pressure wire following the standard procedures in each center. Lesions with an FFR ≤ 0.80 will be considered search failures, and revascularization will be recommended based on clinical indications.5,6
Lesions with an FFR > 0.80 will be further evaluated with OCT according to the standard acquisition methods to detect vulnerable plaques in each center. The decision on whether a lesion meets the criteria for vulnerable plaque will be made by an accredited local investigator during the study procedure.
Patients with at least 1 lesion with an FFR > 0.80 without vulnerable plaque characteristics on OCT will be included in the registry group of the study. The protocol recommends OMT for all lesions with an FFR > 0.80 without vulnerable plaque characteristics. These patients will receive the same clinical follow-up as those in the randomized clinical trial group.
Patients with at least 1 lesion with an FFR > 0.80 that meets the criteria for a vulnerable plaque on OCT will be included in the clinical trial group. These patients will be randomized 1:1 to either preventive stenting combined with OMT or OMT alone (figure 1). Randomization will be conducted without stratification by center or clinical condition, using telematic algorithms. This process will be carried out online via the data collection platform provided by pInvestiga (Pontevedra, Spain).
Figure 1. Study diagram. FFR, fractional flow reserve; OCT, optical coherence tomography; OMT, optimal medical treatment; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction.
The supplementary data provide additional details on the FFR assessment method, including special situations where the lesion under study could not be fully evaluated, instances of unstable nonculprit plaques, complications related to diagnostic techniques, or patients with more than 1 nonculprit lesion.
Study device and implantation procedure
Patients with an FFR > 0.80 and vulnerable plaque characteristics identified by OCT assigned to the percutaneous coronary intervention group will be treated with an everolimus-eluting stent (Xience, Abbott, United States). According to the protocol, stent implantation must be guided by OCT. The criteria for OCT-guided stent implantation are detailed in table 4 of the supplementary data.
Optimal medical therapy
All patients included in both the randomized clinical trial and the registry must receive treatment in accordance with the European Society of Cardiology guidelines for managing acute coronary syndromes.5 The study protocol emphasizes managing modifiable risk factors—such as diet, smoking, obesity, exercise, and psychological status—as well as nonmodifiable risk factors, with set targets for blood pressure (systolic < 130 mmHg and diastolic < 80 mmHg), low-density lipoprotein cholesterol (< 55 mg/dL), and glycated hemoglobin A1c (< 7%). Pharmacological therapy should include beta-blockers and renin-angiotensin system inhibitors. Dual antiplatelet therapy is also recommended, but only during the first year after the index procedure, at the discretion of each center. As per the protocol, patient treatment details will be reported annually, and 2 lipid profile tests will be conducted throughout the study.
Vulnerable plaque criteria on optical coherence tomography and investigator training
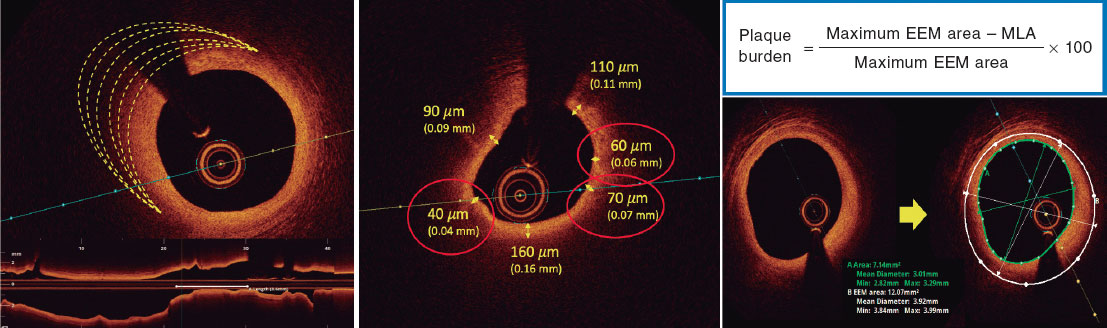
Based on histopathological data, a plaque is defined as vulnerable when it is caused by a fibroatheroma with a large necrotic core composed of cellular debris and a high number of inflammatory cells, covered by a thin fibrous cap (≤ 65 µm).14 The criteria for identifying a vulnerable plaque in the study are adapted from the classic histopathological definition but modified for OCT assessment. These criteria are shown in figure 2.
Figure 2. Vulnerable plaque criteria by optical coherence tomography. EEM, external elastic membrane; minimal lumen area.
According to the protocol, 3 simultaneous criteria are required to define a vulnerable plaque by OCT:
The presence of a fibro-lipid plaque with a necrotic core covering more than 90º of the perimeter of the vessel over a length of more than 5 mm. A necrotic core is defined as a hypointense image with poorly defined borders that attenuates the OCT light beam, preventing visualization of the artery behind the core.
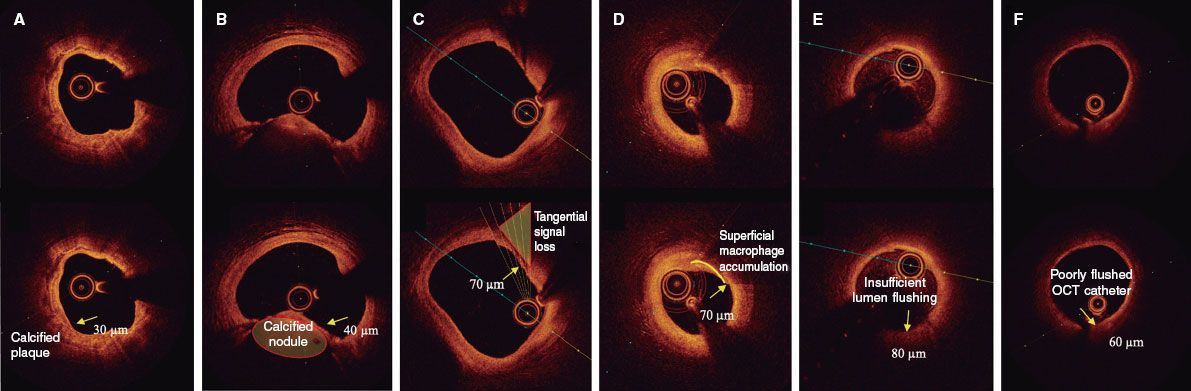
The presence of a thin fibrous cap, defined as ≤ 80 µm (65 + 15 µm axial resolution) in ≥ 3 consecutive images. The fibrous cap is defined as the tissue separating the necrotic core from the vessel lumen. Investigators will be trained to differentiate other findings that could be mistaken for a thin cap on OCT. Figure 3 shows examples of analogous OCT images that may mimic a thin fibrous cap but do not correspond to vulnerable plaques.
Figure 3. Distinction between vulnerable plaques and other findings by optical coherence tomography (OCT). A: plaque with superficial calcium (hypointense core with well-defined margins that do not attenuate the passage of light; arrow) and a thin fibrous cap. B: calcified nodule (arrow) protruding into the lumen and attenuating the signal, despite being composed of calcium. C: tangential signal loss (arrow) due to insufficient light beams caused by the peripheral, noncentral position of the OCT probe. D: superficial accumulation of macrophages (arrow) with a hyperintense appearance relative to the adjacent intima, with signal attenuation behind. E: presence of blood in the lumen due to inadequate flushing (arrow) during image acquisition, which distorts the arterial wall image, creating the appearance of hypointense regions. F: presence of blood between the probe and the OCT catheter (arrow) due to inadequate flushing, which distorts the arterial wall image and mimics hypointense regions.
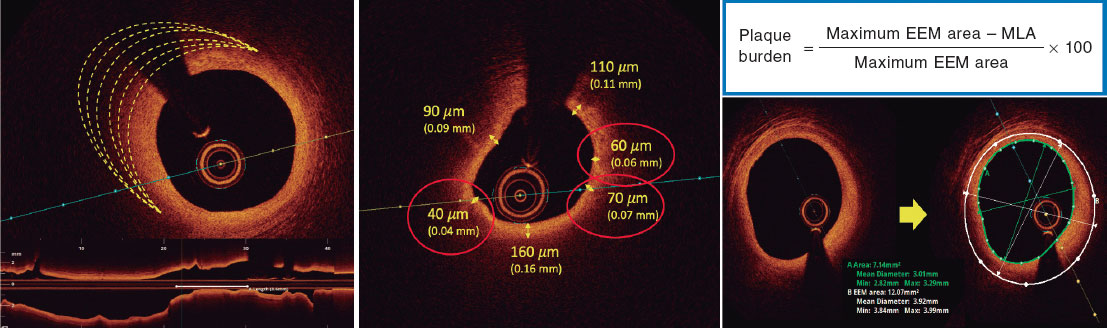
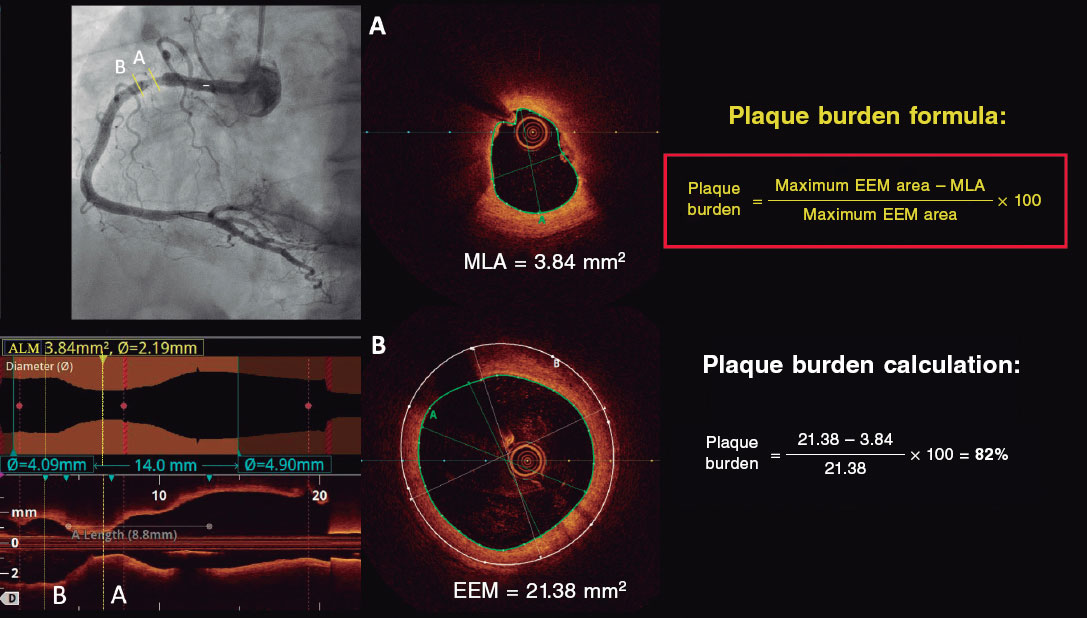
Investigators will be required to measure a plaque burden of ≥ 70% in the cross-sectional area corresponding to the minimal luminal area (MLA) within the lesion. To perform this assessment, it is necessary to measure the vessel perimeter by delineating the external elastic membrane (EEM). Due to the difficulty of assessing the vessel perimeter in fibro-lipid plaques, especially at the MLA site, investigators will be trained to choose a section as close as possible to the MLA, where at least 60% of the vessel perimeter can be visualized if it is not possible at the same point. This allows for calculation using the following formula (figure 4):
Figure 4. Plaque burden assessment by optical coherence tomography. A: cross-section of the minimal lumen area. B: cross-section where the external elastic membrane (EEM) was measured. Since the EEM cannot usually be assessed in the cross-section corresponding to the MLA, an approximate estimation is made by measuring the EEM within 10 mm proximal or distal to the MLA (preferably distal) in the absence of side branches. The EEM will be assessed in the first cross-section where 60% of the EEM perimeter can be evaluated.
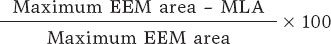
As per protocol, at least 1 local investigator from each participating center must have completed an online training course for the detection and assessment of vulnerable plaques using OCT, following the study criteria. Upon completing this course and passing a specific questionnaire, the investigator will be certified and approved to participate in the study.
Angiographic and optimal coherence tomography quantification analyses
The study includes an independent imaging laboratory for angiographic quantification and OCT analysis (Barcelona Cardiac Imaging Core Laboratory [BARCICORE-Lab]) to monitor adherence to the study criteria for diagnosing vulnerable plaques. A blinded analysis of the study results will be conducted, and patients will be assigned according to the protocol for exploratory analysis. A detailed explanation of the angiographic and OCT analysis conducted by the study laboratory is shown in the supplementary data.
Clinical follow-up and blinding
Patients in both the registry group and the randomized clinical trial group will undergo clinical follow-up for 4 years. Follow-up will include telephone consultations at 1 and 3 years, and in-person visits at 2 and 4 years. Each follow-up will involve an electrocardiogram and blood tests with cholesterol determination.
Patients in the randomized clinical trial group will be blinded to their assigned treatment group (single-blind). The details of blinding and monitoring are specified in the supplementary data.
Sample size calculation
The sample size has been calculated for the randomized clinical trial group. The number of patients included in the registry and search failures will depend on the total number needed to achieve the estimated sample size for the randomized trial.
According to previous studies on patients with acute coronary syndrome, theTVF rate for nonculprit lesions meeting vulnerable plaque criteria treated with OMT is estimated to be around 8% to 10% at 4 years. In similar lesions treated with stenting, the rate is approximately 4%.2,7,9 The studies used for the sample size calculation are summarized in table 5 of the supplementary data. Based on the study hypothesis, preventive stenting in nonculprit lesions with an FFR > 0.80 and vulnerable plaque characteristics is expected to reduce the primary endpoint by 60%. The estimated rate of TVF in the OMT group at 4 years is 10%. Assuming an annual loss to follow-up rate of 1.5% (total 6%), randomizing 600 participants 1:1 to preventive stenting plus OMT vs OMT alone will provide 80% power to demonstrate the superiority of preventive stenting with a 2-sided alpha error of .05.
Statistical analysis plan
The primary and secondary endpoints will be analyzed using the intention-to-treat principle at the 4-year follow-up. Comparisons will estimate event proportions between groups using logistic regression and will be reported as odds ratios with 95% confidence intervals. Only 1 event per patient will be counted for the primary endpoint. P values < .05 will be considered statistically significant for the primary endpoint. Kaplan-Meier curves will be used to visualize the time to the first event between groups.
For primary endpoint composites with missing data, a specific monitoring plan will determine if the missing data are random. In cases where data are adjudicated as missing at random, imputation methods will be used. For nonrandom missing data, sensitivity analyses using worst-case and last observation carried forward methods will be conducted.
Subgroup analyses will be performed for the primary and secondary endpoints, which involves comparing TVF rates between registry patients and those randomized to OMT in the clinical trial. Prespecified subgroups include: age > 75 years, sex, diabetes mellitus, left ventricular ejection fraction ≤ 35% at the time of the procedure, lesions in the proximal or mid-left anterior descending artery, and lesions in vessels with a reference diameter ≤ 2.75 mm.
Additionally, a hypothesis-generating parallel analysis will be conducted according to the study protocol. Patients will be included in the analysis only if the imaging laboratory confirms that their assigned treatment group, as determined by the local investigator, is consistent with the presence of vulnerable plaque identified by OCT. Patients will be excluded if there is a discrepancy between the investigator’s assignment and the imaging laboratory’s findings.
Interim analysis
After 2 years of follow-up, an interim analysis of the data is planned to monitor the primary endpoint in the randomized clinical trial group. Clinical follow-up will be extended if the events observed in the OMT arm of the randomized clinical trial are less than 4%.
DISCUSSION
The VULNERABLE trial aims to investigate the combined use of intracoronary physiology and images to guide the treatment of intermediate nonculprit lesions in STEMI patients.
Several lipid-lowering and anti-inflammatory drugs have been shown to reduce thrombotic events in patients with STEMI, likely by stabilizing functionally nonsignificant vulnerable plaques.15,17 In the PACMAN-AMI trial, treatment with alirocumab in addition to statins significantly reduced atheroma, decreased lipid content, and led to thickening of the fibrous cap compared with placebo in coronary regions with angiographically nonobstructive atherosclerosis (DS, 20%-50%).18 However, it is noteworthy that only 31% of patients in that study exhibited all 3 markers of reduced atherosclerosis, and data on more significant plaques (eg, 40%-69% stenosis with vulnerability criteria) were not specified.19
The use of stents in patients with vulnerable plaques is intended to enhance neointimal healing of the struts, which thickens the fibrous cap and stabilizes the plaque. The randomized PREVENT trial assessed the effectiveness of preventive stenting for functionally nonsignificant vulnerable lesions in patients with chronic coronary syndrome compared with OMT. Vulnerable plaques were identified using various intravascular imaging techniques, with most being guided solely by intravascular ultrasound. The study found that preventive stenting resulted in a statistically significant reduction in the rate of TVF at 2 years of follow-up (0.4% vs 3.4%; P = .0003).11
Finally, several observational trials have demonstrated that OCT is an effective method for detecting vulnerable plaques and monitoring the response to intensive treatments aimed at stabilizing these plaques through fibrous cap thickening.18,20 The PECTUS-obs trial included 438 acute coronary syndrome patients with nonculprit lesions with FFR > 0.80 treated with the OMT alone.10 All lesions were examined using OCT, with criteria similar to those used in the VULNERABLE trial to define vulnerable plaques. In that study, 34% of patients had at least 1 vulnerable lesion, which was associated with a higher risk of adverse events (15.4% vs 8.2% for the composite endpoint of death, myocardial infarction, or revascularization in the groups with and without vulnerable plaques, respectively). The VULNERABLE trial is the first to use OCT to guide the treatment of vulnerable plaques in functionally nonsignificant lesions.
CONCLUSIONS
The VULNERABLE trial aims to evaluate the effectiveness of preventive stenting plus OMT vs OMT alone for vulnerable plaques, as defined by OCT, in functionally nonsignificant intermediate lesions in nonculprit vessels of patients with STEMI. In addition, the study will provide information on the clinical relevance of the presence of vulnerable plaques in nonculprit lesions.
FUNDING
This study has been funded by Abbott Vascular.
ETHICAL CONSIDERATIONS
The study is being conducted following the recommendations outlined in the Declaration of Helsinki on clinical research, has been approved by Hospital Universitari de Bellvitge research ethics committee, and endorsed by the remaining ethics committees of participating centers. Informed consent acceptance and signature are required prior to performing any elective procedures to study the nonculprit lesion. Potential sex and gender biases are considered.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the drafting of this manuscript.
AUTHORS’ CONTRIBUTIONS
J. Gómez-Lara and E. Gutiérrez-Ibañes drafted this document. The remaining signatories reviewed the document, made changes at their discretion, and approved the final text.
CONFLICTS OF INTEREST
J. Gómez-Lara and E. Gutiérrez-Ibañes received a grant from Abbott Vascular for this study. A. Jurado-Román has received fees from Abbott, Boston, and Shockwave. E. Fernández received fees from Abbott and Hexacath. C. Cortés received a Río Hortega Contract from Instituto de Salud Carlos III. S. Brugaletta received fees from Abbott, Microport, and General Electric. T. García-Camarero received fees from Medtronic and Boston. J.A. Linares Vicente received fees from Abbott Vascular, Braun, AstraZeneca, Bayer, and IZASA. O. Rodríguez-Leor received fees from Shockwave, WorlsMedica, and Medtronic. S. Ojeda received fees from Abbott, Boston, WorldMedica, and Biosensors. A. Pérez de Prado received grants and fees from Abbot, Boston, iVascular, and Terumo. H.M. García-García received fees from ACIST, Boston Scientific, Medis, Biotronik, InfraRedx/Nipro, Chiesi, and Cordis. S. Ojeda and A. Pérez de Prado are associate editors of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial processing of the manuscript has been followed. The remaining authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Thin-cap fibroatheromas, also known as vulnerable plaques, are responsible for most acute coronary syndromes. Approximately 50% of patients with STEMI have additional angiographic lesions beyond the culprit lesion, which are associated with a significant number of adverse ischemic events. Preventive stenting for severe nonculprit lesions (DS ≥ 70%) has been shown to reduce the number of adverse events. However, the effectiveness of preventive stenting for angiographically intermediate nonculprit lesions (SD, 40%-69%) that have characteristics of vulnerable plaques remains to be determined.
WHAT DOES THIS STUDY ADD?
- VULNERABLE is the first randomized trial to evaluate the preventive treatment of angiographically intermediate, nonculprit lesions that exhibit features of vulnerability identified by OCT in patients with STEMI.
REFERENCES
1. Park DW, Clare RM, Schulte PJ, et al. Extent, location, and clinical significance of non-infarct-related coronary artery disease among patients with ST-elevation myocardial infarction. JAMA 2014;312:2019-2027.
2. Mehta SR, Wood DA, Storey RF, et al. Complete Revascularization with Multivessel PCI for Myocardial Infarction. N Engl J Med. 2019;381:1411-1421.
3. Lee JM, Kim HK, Park KH, et al. Fractional flow reserve versus angiography-guided strategy in acute myocardial infarction with multivessel disease:a randomized trial. Eur Heart J. 2023;44:473-484.
4. Puymirat E, Cayla G, Simon T, et al. Multivessel PCI Guided by FFR or Angiography for Myocardial Infarction. N Engl J Med. 2021;385:297-308.
5. Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44:3720-3826.
6. Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization:Executive Summary:A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:4-17.
7. Denormandie P, Simon T, Cayla G, et al. Compared Outcomes of ST-Elevation Myocardial Infarction Patients with Multivessel Disease Treated with Primary Percutaneous Coronary Intervention and Preserved Fractional Flow Reserve of Non-Culprit Lesions Treated Conservatively and of Those with Low Fractional Flow Reserve Managed Invasively:Insights from the FLOWER MI trial. Circ Cardiovasc Interv. 2021;14:011314.
8. Stone GW, Maehara A, Lansky AJ, et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364:226-235.
9. Erlinge D, Maehara A, Ben-Yehuda O, et al. Identification of vulnerable plaques and patients by intracoronary near-infrared spectroscopy and ultrasound (PROSPECT II):a prospective natural history study. Lancet. 2021;397:985-995.
10. Mol JQ, Volleberg R, Belkacemi A, et al. Fractional Flow Reserve-Negative High-Risk Plaques and Clinical Outcomes After Myocardial Infarction. JAMA Cardiol. 2023;8:1013-1021.
11. Park SJ, Ahn JM, Kang DY, et al. Preventive percutaneous coronary intervention versus optimal medical therapy alone for the treatment of vulnerable atherosclerotic coronary plaques (PREVENT):a multicentre, open-label, randomised controlled trial. Lancet. 2024;403:1753-1765.
12. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials:a case for standardized definitions. Circulation. 2007;115:2344-2351.
13. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40:237-269.
14. Virmani R. Pathology of the vulnerable plaque. J Am Coll Cardiol. 2006;47:C13-8.
15. Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med. 2004;350:1495-1504.
16. Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N Engl J Med. 2018;379:2097-2107.
17. Tardif JC, Kouz S, Waters DD, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019;381:2497-2505.
18. Raber L, Ueki Y, Otsuka T, et al. Effect of Alirocumab Added to High-Intensity Statin Therapy on Coronary Atherosclerosis in Patients With Acute Myocardial Infarction:The PACMAN-AMI Randomized Clinical Trial. JAMA. 2022;327:1771-1781.
19. Biccire FG, Haner J, Losdat S, et al. Concomitant Coronary Atheroma Regression and Stabilization in Response to Lipid-Lowering Therapy. J Am Coll Cardiol. 2023;82:1737-1747.
20. Kedhi E, Berta B, Roleder T, et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve:the COMBINE OCT-FFR trial. Eur Heart J. 2021;42:4671-4679.

ABSTRACT
Introduction and objectives: The use of transradial access for percutaneous coronary procedures has increased due to its advantages over the femoral approach. However, this benefit comes at the expense of a higher rate of radial artery occlusion (RAO). Our objective was to assess the incidence and predictors of RAO following transradial catheterization. Additionally, we studied anatomic variations of the radial artery (RA).
Methods: This prospective study enrolled 427 patients who underwent coronary angiography or angioplasty via transradial access. The forearm arteries were evaluated by ultrasound. If RAO was present, follow-up ultrasound examinations were performed at 1 and 3 months postprocedure.
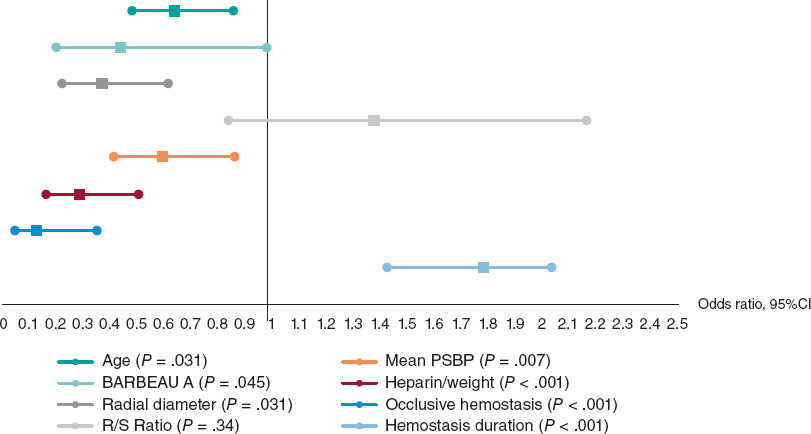
Results: Our study population included 288 men (67.4%) and 139 women (32.6%). The mean age was 61.9 ± 11.1 years. RAO occurred in 48 patients (11.24%), and spontaneous recanalization was observed within 3 months in 15 patients (32.6%). On multivariate analysis, independent predictors of RAO were younger age (OR, 0.642; 95%CI, 0.480-0.858; P = .031), low periprocedural systolic blood pressure (OR, 0.598; 95%CI, 0.415-0.862; P = .007), a small radial diameter (OR, 0.371; 95%CI, 0.323-0.618; P = .031), insufficient anticoagulation (OR, 0.287; 95%CI, 0.163-0.505; P < .001), occlusive hemostasis (OR, 0.128; 95%CI, 0.047-0.353; P < .001), and long duration of hemostasis. The overall incidence of RA anatomic variations was 14.8% (n = 63). Among these, 40 patients (63.5%) had a high radial origin, 18 (28.6%) had extreme RA tortuosity, and 5 (7.9%) had a complete radioulnar loop.
Conclusions: The main modifiable predictors of RAO are insufficient heparinization and occlusive hemostasis. Preventive strategies should focus primarily on these 2 predictive factors to reduce the risk of RAO.
Keywords: Anatomic variations. Cardiac catheterization. Doppler ultrasound. Percutaneous coronary intervention. Predictors. Radial artery occlusion. Transradial access.
RESUMEN
Introducción y objetivos: El acceso transradial para procedimientos coronarios percutáneos ha crecido en popularidad debido a sus ventajas sobre el abordaje femoral. Sin embargo, este beneficio se ve ensombrecido por una mayor tasa de oclusión de la arteria radial (OAR). Nuestro objetivo fue evaluar la incidencia y los factores predictivos de OAR tras el cateterismo transradial. También se estudiaron las variaciones anatómicas de la arteria radial (AR).
Métodos: En este estudio prospectivo participaron 427 pacientes a los que se había realizado angiografía coronaria o angioplastia mediante acceso transradial. Se realizó una evaluación ecográfica de las arterias del antebrazo. En caso de OAR, se llevó a cabo otro control ecográfico al mes y a los 3 meses de la intervención.
Resultados: La población de estudio incluyó a 288 varones (67,4%) y 139 mujeres (32,6%). La edad media fue de 61,9 ± 11,1 años. La OAR se produjo en 48 pacientes (11,24%), de los cuales en 15 (32,6%) se produjo recanalización espontánea en el plazo de 3 meses. En el análisis multivariante, la edad más joven (OR = 0,642; IC95%, 0,480-0,858; p = 0,031), la presión arterial sistólica periprocedimiento baja (OR = 0,598; IC95%, 0,415-0,862; p = 0,007), el diámetro radial pequeño (OR = 0,371; IC95%, 0,323-0,618; p = 0,031), la anticoagulación insuficiente (OR = 0,287; IC95%, 0,163-0,505; p < 0,001), la hemostasia oclusiva (OR = 0,128; IC95%, 0,047-0,353; p < 0,001) y la larga duración de la hemostasia aparecieron como predictores independientes de OAR. La incidencia global de variaciones anatómicas de la AR fue del 14,8% (n = 63). Entre estos pacientes, 40 (63,5%) tenían un origen radial alto, 18 (28,6%) presentaban una tortuosidad extrema de la AR y 5 (7,9%) tenían un asa radiocubital completa.
Conclusiones: La heparinización insuficiente y la hemostasia oclusiva son los principales predictores de OAR modificables. La estrategia preventiva debe centrarse principalmente en estos 2 factores predictivos.
Palabras clave: Variaciones anatómicas. Cateterismo cardiaco. Ecografía Doppler. Intervención coronaria percutánea. Predictores. Oclusión de la arteria radial. Acceso transradial.
Abbreviations
RA: radial artery. RAO: radial artery occlusion.
INTRODUCTION
The use of the transradial approach for coronary interventions has become increasingly widespread in interventional cardiology due to its numerous advantages.1 As a result, current guidelines recommend it as the first-line approach.2
However, the benefits of this technique are tempered by the risk of radial artery occlusion (RAO), with reported rates ranging from 5% to 30%.3,4 The aim of this study was to assess the incidence and predictors of RAO following transradial catheterization using Doppler ultrasound for evaluation.
METHODS
Patient population
This longitudinal, single-center prospective study was conducted in the cardiology department of the Military Central Hospital in Algiers. After applying exclusion criteria (hemodynamic instability and ST-segment elevation myocardial infarction), we included 427 consecutive patients undergoing transradial coronary procedures between January 2019 and March 2020. The study adhered to the principles of the Declaration of Helsinki and the International Conference on Harmonization Good Clinical Practices and was approved by the local ethics committee. All patients provided written informed consent.
Radial artery cannulation and retrograde radial arteriography
After radial artery (RA) puncture, a radial hydrophilic sheath (Radiofocus II, TERUMO Medical, Japan, or Prelude, MERIT Medical, United States) was introduced. An antispastic cocktail was then administered into the RA through the sheath, consisting of a saline solution, a vasodilator (1 mL of nicardipine), and a bolus of unfractionated heparin, which was administered either intravenously or directly into the RA as part of the spasmolytic cocktail, depending on the operator’s preference. In patients on vitamin K antagonists, these medications were not discontinued prior to the procedure.
Retrograde radial arteriography was performed by injecting a mixture of 4 mL of contrast and 4 mL of isotonic saline through the sheath. Radiographic images were then obtained in an anteroposterior projection.
Transradial coronary procedure
The standard approach was conventional right radial access. For coronary angiography, 5-French (Fr) hydrophilic sheaths and catheters were usually used. If the patient required revascularization, an ad hoc percutaneous coronary intervention was performed, using 6-Fr guiding catheters after exchanging the sheath from 5-Fr to 6-Fr. The usual dose of heparin is 5000 IU (2500 IU for oral anticoagulation with a vitamin K antagonist).
Hemostasis procedure
At the end of the procedure, the sheath was removed, and hemostasis was achieved using a hemostatic compression device (TR BAND, TERUMO Medical, Japan). A reverse Barbeau test5 was systematically performed. The hemostasis device was removed by nurses in the hospitalization unit. No standardized protocol for the duration of hemostasis was followed.
Assessment of postprocedural radial artery patency
Radial Doppler assessments were conducted before and after each transradial procedure. To evaluate RAO, pulsed Doppler was performed bilaterally on the radial and ulnar arteries. Normal arterial flow was indicated by a biphasic or triphasic signal, reflecting good perfusion. In cases of RAO, 2 additional ultrasonographic examinations were performed at 1 and 3 months, following the same protocol. Artery patency was assessed by an independent operator.
Classifications and definitions
RAO was defined as the absence of anterograde flow in the RA on ultrasound (figure 1). The location of the radial occlusion was identified using color and pulsed Doppler. We delineated 3 anatomical territories: the distal third, extending from the radial styloid to approximately 7 to 10 cm proximally; the proximal third, from the elbow folds to approximately 7 to 10 cm distally; and the middle third, located between the previous 2 regions (middle part of the forearm).
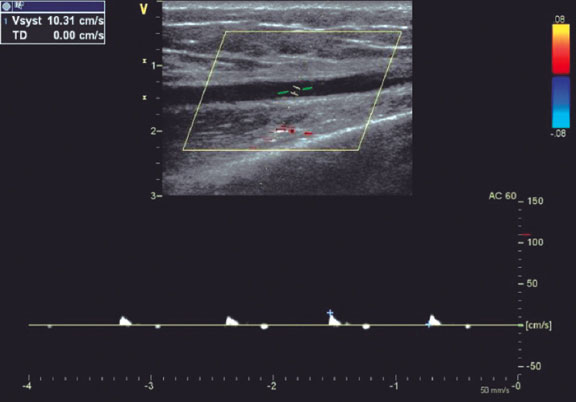
Figure 1. Radial artery with occlusion in the distal third. Pulsed Doppler flow targets a stop flow indicating radial occlusion.
The type of hemostasis, whether occlusive or patent, was assessed: patent hemostasis was indicated by the presence of a plethysmographic signal in the RA during the reverse Barbeau test,5 which involves compression of the ulnar artery. The operator did not intervene during this process but simply recorded whether the artery remained patent or not.
The internal luminal diameter of the RA was defined as the distance between the leading edges of the intima-lumen interface on the superficial wall and the lumen-intima interface on the deep wall.6
The R/S ratio (radial/sheath) was calculated by dividing the luminal diameter of the RA by the external diameter of the sheath (Radiofocus II: 5-Fr = 2.29 mm, 6-Fr = 2.62 mm, 7-Fr = 2.97 mm; Prelude: 5-Fr = 2.52 mm, 6-Fr = 2.83 mm). This ratio was categorized qualitatively as < 1 or ≥ 1.
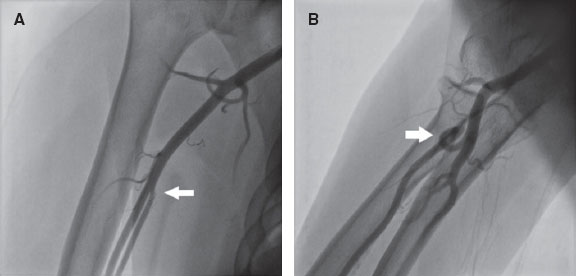
RA anatomical variations of clinical relevance were classified according to definitions provided in the literature.7,8 A high origin (high bifurcation) of the RA (figure 2) was defined with reference to the intercondylar line of the humerus. A radioulnar loop was characterized by the presence of a complete 360° loop of the RA, while radial tortuosity was identified by a curvature greater than 45°.
Figure 2. Anatomic variations of the radial artery. A: high origin of the radial artery. The radial and ulnar arteries separate at the level of the middle third of the humerus (arrow). B: radioulnar loop was defined as a complete 360° loop of the radial artery distal to the bifurcation of the brachial artery (arrow).
A blood pressure profile was obtained on the same side as the radial access. Forearm hematomas were classified according to the “EASY” study9: type I: < 5 cm in diameter; type II: < 10 cm; type III: > 10 cm but not extending to the elbow; type IV: extending beyond the elbow; type V: resulting in an ischemic lesion.
Statistical analysis
The statistical analysis was performed using IBM SPSS Software version 25. Parameters of interest are reported with their 95% confidence intervals (95%CI). For all tests, a significance threshold of 5% was retained. All tests were performed bilaterally. The following tests were used to compare groups: the chi-square test was used to compare 2 qualitative variables; the Student t-test or analysis of variance was used to compare a quantitative variable with a qualitative variable, with the Fisher test being applied when variances were unequal; and logistic regression was used to identify predictors of RAO.
RESULTS
Clinical and procedural characteristics of the study population
During the study period, 441 patients were screened. Of these, transradial access failed in 14 patients, who were excluded from the study, resulting in an eligible sample of 427 patients (mean age 61.9 ± 11.1 years, 67.4% male). Among the patients, 260 had hypertension (60.9%), and nearly half had diabetes (48.9%).
Table 1 summarizes the procedural data. The sheaths used were mainly 6-Fr (83.6%), and heparin was injected intra-arterially in 63.5% of patients. The mean heparin dose was 5669 ± 1394 IU, with a higher dose given when percutaneous coronary intervention was performed (4940 ± 339 IU vs 7491 ± 1368 IU; P < .001).
| Procedural characteristics | Patients N (%) |
|---|---|
| Indication | |
| CCS | 227 (53.2%) |
| ACS (NSTEMI) | 200 (46.8%) |
| Type of procedure | |
| Diagnostic angiography | 305 (71.4%) |
| PCI | 122 (28.6%) |
| Previous radial procedures | 68 (15.9%) |
| Right radial access | 410 (96.0%) |
| Puncture attempts | |
| 1 attempt | 258 (60.4%) |
| 2 attempts | 99 (23.2%) |
| ≥ 3 attempts | 70 (16.4%) |
| Sheath size | |
| 5-Fr | 68 (15.9%) |
| 6-Fr | 357 (83.6%) |
| 7-Fr | 2 (0.5%) |
| Heparin administration | |
| Intra-arterial | 271(63.5%) |
| Intravenous | 156 (36.5%) |
| Heparin dose (IU) | 5669 ± 1394 |
| Angiography | 4940 ± 339 |
| PCI | 7491 ± 1368 |
| Catheter diameter | |
| 5-Fr | 300 (70.3%) |
| 6-Fr | 125 (29.3%) |
| 7-Fr | 2 (0.5%) |
| Number of catheters used | |
| 1 | 43 (10.1%) |
| 2 | 271 (63.5%) |
| ≥ 3 | 113 (26.4%) |
| Fluoroscopy time (min) | 11.22 ± 12.09 |
| Radiation dose (mGy) | 564 ± 538 |
| Contrast amount (mL) | 98.97 ± 54.09 |
| Procedure time (min) | 39.16 ± 34.6 |
| Angiography | 21.63 ± 9.98 |
| PCI | 82.99 ± 35.39 |
| Coronary lesions | |
| Normal coronaries | 134 (31.4%) |
| 1 vessel disease | 131 (30.7%) |
| 2 vessel disease | 87 (20.4%) |
| 3 vessel disease | 75 (17.6%) |
|
ACS, acute coronary syndrome; CCS, chronic coronary syndrome; Fr, French; IU, international unit; NSTEMI, non–ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention. |
|
Incidence and characteristics of radial artery occlusion
RAO occurred in 48 patients (11.24%). Of these, 89.6% were asymptomatic, and the radial pulse remained palpable in 14 patients (29.2%). At 1 month, 2 patients were lost to follow-up. Among the remaining 46 patients, spontaneous recanalization occurred in 13 patients (28.3%). At the 3-month follow-up, the recanalization rate increased to 32.6% (15 cases).
The site of RAO was the distal third in 7 patients (14.6%), the middle third in 21 patients (43.8%), and the proximal third in 20 patients (41.7%).
Predictors of radial artery occlusion
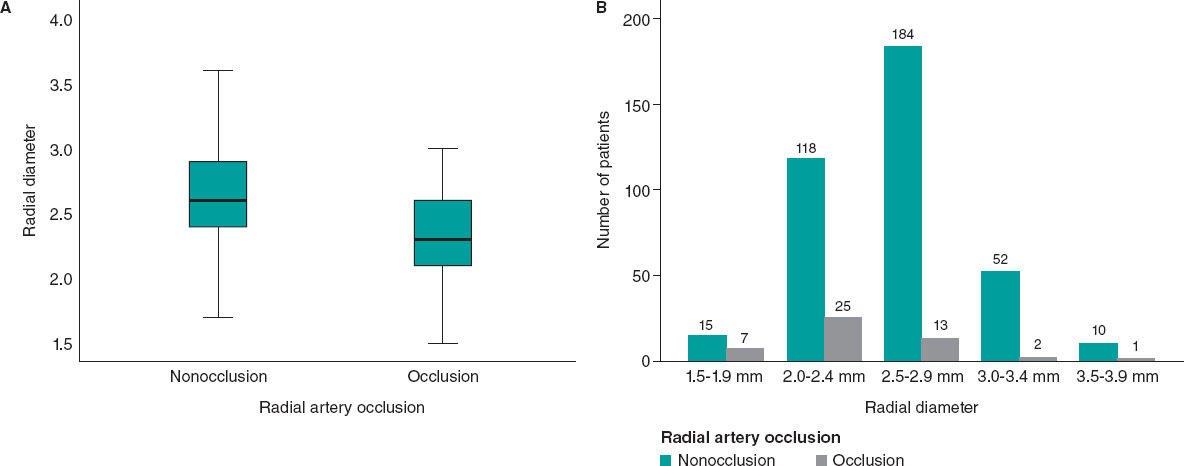
Patients with RAO were significantly younger (table 2). The mean periprocedural systolic blood pressure in the RAO group was significantly lower (138.04 mmHg ± 21.92, vs 145.84 mmHg ± 21.10; P = .017). Type A Barbeau test was associated with a higher risk of RAO compared with types B and C, and patients with occlusion had a smaller RA diameter (2.34 mm ± 0.40 vs 2.61 mm ± 0.37; P < .001) (figure 3).
Table 2. Comparison of patients with and without RAO
| Clinical data | Procedural data | ||||||
|---|---|---|---|---|---|---|---|
| Non-RAO (n= 379) | RAO (n= 48) | P | Non-RAO (n= 379) | RAO (n= 48) | P | ||
| Age | 62.6 ± 10.6 | 56.4 ± 14.0 | < .001* | Previous TRA | 61 (16.0%) | 7 (14.5) | .63 |
| Female sex | 122 (32.1%) | 17 (35.4%) | .65 | Diagnostic angiography | 266 (70.1%) | 39 (81.2%) | .11 |
| Hypertension | 237 (62.5%) | 23 (47%) | .051 | ≥ 2 puncture attempts | 147 (38.7%) | 22 (45.8%) | .41 |
| Diabetes | 182 (48%) | 27 (56%) | .28 | IV heparin | 136 (35.8%) | 20 (41.6%) | .43 |
| Dyslipidemia | 44 (11.6%) | 6 (12.5%) | .85 | Heparin dose (IU) | 5754 ± 1378 | 5007 ± 1352 | < .001* |
| Smoking | 75 (19.7%) | 9 (18.7%) | .86 | Spasm | 60 (15.8%) | 12 (25.0%) | .11 |
| BMI ≥ 30 kg/m2 | 123 (32.4%) | 16 (33.3%) | .90 | Procedure time (min) | 39.85 ± 34.56 | 33.73 ± 34.78 | .249 |
| Mean PSBP (mm Hg) | 145.84 ± 21.10 | 138.04 ± 21.92 | .017* | Number of catheters | 2.30 ± 0.88 | 2.21 ± 0.92 | .75 |
| Barbeau test type A | 99 (26.1%) | 20 (41.6%) | .044* | Occlusive hemostasis | 241 (63.5%) | 42 (87.5%) | .001* |
| RAD (mm) | 2.61 ± 0.37 | 2.34 ± 0.40 | < .001* | Hemostasis duration (h) | 4.29 ± 1.22 | 5.15 ± 1.41 | .006* |
| APT | 336 (88%) | 41 (85%) | .51 | ||||
| VKA (INR ≥ 2) | 16 (4.2%) | 5 (10.4%) | .061 | ||||
| MVCD | 149 (39.3%) | 13 (27.1%) | .20 | ||||
|
APT, antiplatelet therapy; BMI, body mass index; INR, international normalized ratio; IV, intravenous; IU, international unit; MVCD, multivessel coronary disease = ≥ 2 lesions ; PSBP, periprocedural systolic blood pressure; RAD , radial artery diameter; RAO, radial artery occlusion; TRA , transradial access; VKA, vitamin K antagonist. * Statistically significant. |
|||||||
Figure 3. Radial artery diameter as a predictor of occlusion. A: the radial diameter is significantly smaller if there is RAO. B: less than 2.5 mm, the risk of occlusion becomes greater.
RAO procedural factors are listed in table 2. An R/S ratio < 1 was found in 35 patients in the RAO group vs 153 patients in the non-RAO group (72.9% vs 40.3%, P < .001). The mean heparin dose was significantly lower in patients with RAO (5007 ± 1352IU vs 5754 ± 1378 IU; P < .001), and the dose adjusted to weight was also significantly lower in the RAO group (62.31 ± 17.82 IU/kg vs 75.73 ± 22.57 IU/kg; P < .001). In addition, the RAO rate decreased significantly when the heparin dose exceeded 70 IU/kg.
Forty-two patients in the RAO group had occlusive hemostasis vs 241 in the non-RAO group (87.5% vs 63.5%; P = .001). Surprisingly, two-thirds of our patients (283 [66.3%]) had occlusive hemostasis. The mean duration of hemostasis was longer if there was RAO (5.15 h ± 1.41 vs 4.29 h ± 1.22; P < .001).
On multivariate logistic regression analysis (figure 4), the following factors were independent predictors of RAO: young age (odds ratio [OR], 0.642; 95%CI, 0.480-0.858; P = .031), low periprocedural systolic blood pressure (OR, 0.598; 95%CI, 0.415-0.862; P = .007), type A Barbeau test (OR, 0.441; 95%CI, 0.198-0.981; P = .045), small RA diameter (OR, 0.371; 95%CI, 0.323-0.618; P = .031), insufficient anticoagulation (OR, 0.287; 95%CI, 0.163-0.505; P < .001), occlusive hemostasis (OR, 0.128; 95%CI, 0.047-0.353; P < .001), and a long hemostasis duration (OR, 1.786; 95%CI, 1.428-2.039; P < .001).
Figure 4. Independent factors predictive of radial artery occlusion. Multiple logistic regression analysis revealed that the independent factors predictive of radial occlusion were young age, low periprocedural systolic blood pressure, type A Barbeau test, small radial artery diameter, insufficient anticoagulation, occlusive hemostasis, and long hemostasis duration. 95%CI, 95% confidence interval; PSBP, periprocedural systolic blood pressure; RAO, radial artery occlusion.
Anatomic variations of the radial artery
The mean radial diameter was 2.58 mm ± 0.39, and the diameter was larger in men (2.69 mm ± 0.37 vs 2.36 mm ± 0.31; P < .001) and smaller in patients with diabetes (2.53 mm ± 0.38 vs 2.64 mm ± 0.38; P = .003). The mean radial diameter was significantly larger than the mean ulnar diameter (2.58 mm ± 0.39 vs 2.22 mm ± 0.43; P < .001).
Radial anatomical variations affected 63 patients (14.8%). The most common variation was a high origin of the RA, observed in 63.5% of cases (40 patients), followed by radial tortuosity in 28.6% (18 patients), radioulnar loop in 7.9% (5 patients). Anatomical variations were more frequent in women (23% vs 10.8%; P = .001) and in older patients, with a mean age of 66.3 years ± 10.2 vs 61.2 years ± 11.2 in those without variations (P = .001).
Periprocedural complications
Radial spasm occurred in 72 patients (16.9%). This complication was more frequent in women (29% vs 10.1%; P < .001), patients with diabetes (22.5% vs 11.5%; P = .002), and when 6-Fr catheters were used (14% vs 24%; P = .035). Forearm hematoma occurred in 25 patients (5.85%). According to the EASY classification,9 most hematomas were type I (17 patients, 68%), followed by type II (6 patients, 24%), with type III occurring in only 2 patients (8%).
DISCUSSION
The rate of RAO remains relatively high in some institutions.10,11 In the PROPHET study, the acute incidence of RAO (12%) was almost halved in 28 days (7%).3 Recanalization occurs as a result of activation of primary fibrinolysis.12 In the present study, the rate of radial recanalization at 3 months was 32.6%. The only predictor of recanalization was radial diameter: the larger the diameter, the higher the rate of spontaneous recanalization.
Zankl et al.13 found that RAO was located in the distal third of the forearm in 49% of patients, in the distal and middle third in 13.7%, and in the entire forearm (proximal third) in 37.3%. Dissections of the media also occur in the proximal RA, likely due to catheter progression or manipulation without protection of the sheath.14 In our opinion, this would explain the location of RAO in the proximal part of the artery.
Among our patients with RAO, 29.2% had a radial pulse. According to Uhlemann et al.,4 in 19.5% of patients with RAO on Doppler, the RA pulse was still palpable. This was likely due to retrograde filling of the RA by collaterals. Therefore, the diagnosis of RAO should be confirmed using a more objective method, such as Doppler ultrasound.
Young age is a predictor of RAO, possibly due to higher sympathetic reactivity in younger individuals, which increases their risk of spasm. However, this characteristic does not influence the rate of recanalization, likely because prolonged radial spasm leads to the formation of a permanent intra-arterial thrombus.
Low mean systolic blood pressure was also a predictor of radial occlusion. We speculate that hypertension and arterial stiffness may prevent complete interruption of flow during compression, thereby helping to maintain radial patency.15
There was a higher incidence of occlusion with type A Barbeau test. We believe that in cases with well-developed ulnar circulation, the ulnar artery generates a competitive retrograde flow that opposes the radial flow, promoting occlusion and hindering recanalization.
The likelihood of developing RAO is related to the size of the sheath,16 or more precisely, the R/S ratio.17 A prospective registry showed that 5-Fr sheaths reduced the rate of RAO by up to 55% compared with 6-Fr.4
A study by Pancholy et al.,18 demonstrated that intravenous heparin is as effective as intra-arterial heparin in reducing the incidence of RAO, suggesting that the systemic effect of heparin is more important than its local effect. A recently published meta-analysis identified higher heparin doses as the most significant measure for decreasing RAO.12 This results is in line with our finding that a dose of less than 70 IU/kg seems to promote the occurrence of RAO. The high prevalence of RAO and the benefit of higher doses of unfractionated heparin (≥ 50 IU/kg) in this setting were also highlighted by a meta-analysis of 112 studies.19 In a randomized superiority trial comparing high-dose (100 IU/kg) and standard-dose (50 IU/kg) heparin, the RAO rate was significantly lower in the high-dose group.20 Recent evidence suggests that a small dose of rivaroxaban, given orally after a transradial procedure, may decrease the occurrence of RAO at 1 month.21,22
Using the reverse Barbeau test, Sanmartin et al.23 found that 60% of patients had an absence of radial flow during compression. These observations led to the concept of nonocclusive hemostasis (patent hemostasis). In the PROPHET study,3 RAO was significantly less frequent in the group that underwent nonocclusive hemostasis than in the control group.
The duration of hemostatic compression has been studied in large, randomized trials.24-26 The authors concluded that compression duration was a strong predictor of RAO.
In a meta-analysis by Rashid et al.,27 the incidence of RAO after diagnostic coronary angiography was notably higher compared with percutaneous coronary intervention, possibly due to the use of higher anticoagulation doses during interventions.12 However, opposite findings have been reported by other studies.
In our sample, the mean radial diameter was 2.58 mm ± 0.39 and was significantly larger in men. Velasco et al.28 reported a mean arterial diameter of 2.22 ± 0.35 mm, while a Polish study found a mean diameter of 2.17 ± 0.53 mm for the right RA and 2.25 ± 0.43 mm for the left RA.29 The ulnar artery is also used in interventional cardiology,30 although there is no consensus on its size compared with the RA.
Autopsy studies of arterial anatomic variations of the upper extremity have reported frequencies between 4% and 18.5%.8 In the literature, the most frequent anatomic variation of the RA is high bifurcation. Yoo et al.31 reported a 2.4% incidence of high radial origin in 1191 Korean patients. Tortuosity of the RA frequently affects patients with high radial origin, possibly due to the elongated course of the RA predisposing it to tortuosity, which is considered one of the most common causes of procedural failure, along with radial spasm.32
Radioulnar loop is the most common cause of procedural failure with experienced operators.33 Angiographic evaluation of the radioulnar anastomosis is mandatory in such cases, as there is often a negotiable anastomosis between the radial and ulnar arteries.
In our study, radial spasm was the leading cause of procedural failure, occurring in 50% of the 14 patients who experienced such failures. Ruiz-Salmerón et al.34 found that RA anatomic variations were strongly associated with radial spasm in a multivariate analysis. The relationship between radial spasm and anatomic variations is mainly explained by the strong correlation with high radial origin and the radioulnar loop.
Study limitations
Since this study is a prospective registry and not a randomized trial, selection bias cannot be excluded. Our study represents a single-center experience with a limited number of patients, despite being one of the largest prospective registries of vascular ultrasound in radial catheterization to date. Among the other limitations of the study, we note the lack of standardized protocols for both heparin use and compression.
CONCLUSIONS
With the increasing number of transradial procedures and the greater age of patients undergoing these interventions, leading to more complex procedures, it is essential to maintain the patency of the RA for future access. Although predictors of RAO after cardiac catheterization have been identified, implementing preventive measures in practice remains a challenge. The main modifiable predictors associated with the risk of RAO are insufficient heparinization and occlusive hemostasis. Therefore, preventive strategies should primarily focus on addressing these 2 factors.
FUNDING
None.
ETHICAL CONSIDERATIONS
The study was conducted in accordance with the provisions of the Declaration of Helsinki and with the International Conference on Harmonization Good Clinical Practices and was approved by the local ethics committee. All patients included in the study provided written informed consent, which is archived and available. Our study population included both sexes. Gender had no influence on the occurrence of radial occlusion.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence software was used in the preparation of this study.
AUTHORS’ CONTRIBUTIONS
All authors meet the criteria for authorship as defined by the International Committee of Medical Journal Editors. M.S. Lounes, A. Meftah, C. Belhadi, K. Allal, H. Boulaam, A. Sayah, I. Hafidi, and E. Tebache contributed to the acquisition and analysis of data for this article. M.S. Lounes, A. Bedjaoui, A. Allali, and S. Benkhedda were responsible for the study design and the writing of the article. M.S. Lounes, A. Allali, and S. Benkhedda contributed to writing and critical revision of the content. All authors have read and approved the final version of the article and agree to be accountable for all aspects of the work, including the accuracy and integrity of all its parts.
CONFLICTS OF INTEREST
None.
WHAT IS KNOWN ABOUT THE TOPIC?
- Despite recommendations on the prevention of RAO in interventional cardiology, its incidence remains relatively high in some centers.
- Spontaneous recanalization of the artery may occur during follow-up.
- Permanent occlusion of the radial artery prevents any possibility of its further use (interventional procedures, dialysis, etc.)
WHAT DOES THIS STUDY ADD?
- RAO is not limited to the distal part of the artery and can affect the entire length of the vessel.
- Diagnosis of RAO should be confirmed using Doppler ultrasound, which remains the gold standard.
- The 2 independent modifiable predictors of RAO are the anticoagulation protocol and hemostasis technique.
- Anatomic variations of the RA may impact the procedure. A high origin of the RA is the most frequent, followed by radial tortuosities. After radial spasm, the radioulnar loop is the most common cause of procedural failure with experienced operators.
REFERENCES
1. Cruden NL, Teh CH, Starkey IR, Newby DE. Reduced vascular complications and length of stay with transradial rescue angioplasty for acute myocardial infarction. Catheter Cardiovasc Interv. 2007;70:670-675.
2. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
3. Pancholy SB, Coppola J, Patel T, Roke-Thomas M. Prevention of radial artery occlusion-patent hemostasis evaluation trial (PROPHET study):a randomized comparison of traditional versus patency documented hemostasis after transradial catheterization. Catheter Cardiovasc Interv. 2008;72:335-340.
4. Uhlemann M, Möbius-Winkler S, Mende M, et al. The Leipzig prospective vascular ultrasound registry in radial artery catheterization:impact of sheath size on vascular complications. JACC Cardiovasc Interv.2012;5:36-43.
5. Da Silva RL, Britto PF, Joaquim RM, et al. Clinical accuracy of reverse Barbeau test in the diagnosis of radial artery occlusion after transradial catheterization. J Transcat Intervent. 2021;29:eA20200037.
6. Costa F, van Leeuwen MA, Daemen J, et al. The Rotterdam Radial Access Research:Ultrasound-Based Radial Artery Evaluation for Diagnostic and Therapeutic Coronary Procedures. Circ Cardiovasc Interv. 2016;9:003129.
7. Uglietta JP, Kadir S. Arteriographic study of variant arterial anatomy of the upper extremities. Cardiovasc Intervent Radiol. 1989;12:145-148.
8. Rodríguez-Niedenführ M, Vázquez T, Nearn L, et al. Variations of the arterial pattern in the upper limb revisited:a morphological and statistical study, with a review of the literature. J Anat. 2001;199:547-566.
9. Bertrand OF. Acute forearm muscle swelling post transradial catheterization and compartment syndrome:prevention is better than treatment!Catheter Cardiovasc Interv. 2010;75:366-368.
10. Sadaka MA, Etman W, Ahmed W, et al. Incidence and predictors of radial artery occlusion after transradial coronary catheterization. Egypt Heart J. 2019;71:12.
11. Dousi M, Sotirakou K, Fatsi A. The Use of Acetylsalicylic Acid As A Measure of Prevention of Radial Artery Occlusion in Patients Who Perform Coronary Angiography with Tra Technique. J Radiol Clin Imaging. 2020;3:13-21.
12. Rashid M, Kwok CS, Pancholy S, et al. Radial Artery Occlusion After Transradial Interventions:A Systematic Review and Meta-Analysis. J Am Heart Assoc. 2016;5:002686.
13. Zankl AR, Andrassy M, Volz C, et al. Radial artery thrombosis following transradial coronary angiography:incidence and rationale for treatment of symptomatic patients with low-molecular-weight heparins. Clin Res Cardiol. 2010;99:841-847.
14. Bi XL, Fu XH, Gu XS, et al. Influence of Puncture Site on Radial Artery Occlusion After Transradial Coronary Intervention. Chin Med J (Engl). 2016;129:898-902.
15. Buturak A, Gorgulu S, Norgaz T, et al. The long-term incidence and predictors of radial artery occlusion following a transradial coronary procedure. Cardiol J. 2014;21:350-356.
16. Dahm JB, Vogelgesang D, Hummel A, et al. A randomized trial of 5 vs. 6 French transradial percutaneous coronary interventions. Catheter Cardiovasc Interv. 2002;57:172-176.
17. Saito S, Ikei H, Hosokawa G, Tanaka S. Influence of the ratio between radial artery inner diameter and sheath outer diameter on radial artery flow after transradial coronary intervention. Catheter Cardiovasc Interv.1999;46:173-178.
18. Pancholy SB. Comparison of the effect of intra-arterial versus intravenous heparin on radial artery occlusion after transradial catheterization. Am J Cardiol. 2009;104:1083-1085.
19. Hahalis GN, Aznaouridis K, Tsigkas G, et al. Radial Artery and Ulnar Artery Occlusions Following Coronary Procedures and the Impact of Anticoagulation:ARTEMIS Systematic Review and Meta-Analysis. J Am Heart Assoc. 2017;6:005430.
20. Hahalis GN, Leopoulou M, Tsigkas G, et al. Multicenter Randomized Evaluation of High Versus Standard Heparin Dose on Incident Radial Arterial Occlusion After Transradial Coronary Angiography:The SPIRIT OF ARTEMIS Study. JACC Cardiovasc Interv. 2018;11:2241-2250.
21. Liang D, Lin Q, Zhu Q, et al. Short-Term Postoperative Use of Rivaroxaban to Prevent Radial Artery Occlusion After Transradial Coronary Procedure:The RESTORE Randomized Trial. Circ Cardiovasc Interv. 2022;15:011555.
22. Hammami R, Abid S, Jihen J, et al. Prevention of radial artery occlusion with rivaroxaban after trans-radial access coronary procedures:The RIVARAD multicentric randomized trial. Front Cardiovasc Med. 2023;10:1160459.
23. Sanmartin M, Gomez M, Rumoroso JR, et al. Interruption of blood flow during compression and radial artery occlusion after transradial catheterization. Catheter Cardiovasc Interv. 2007;70:185-189.
24. Politi L, Aprile A, Paganelli C, et al. Randomized clinical trial on short-time compression with Kaolin-filled pad:a new strategy to avoid early bleeding and subacute radial artery occlusion after percutaneous coronary intervention. J Interv Cardiol. 2011;24:65-72.
25. Dharma S, Kedev S, Patel T, et al. A novel approach to reduce radial artery occlusion after transradial catheterization:postprocedural/prehemostasis intra-arterial nitroglycerin. Catheter Cardiovasc Interv.2015;85:818-825.
26. Aminian A, Saito S, Takahashi A, et al. Impact of sheath size and hemostasis time on radial artery patency after transradial coronary angiography and intervention in Japanese and non-Japanese patients:A substudy from RAP and BEAT randomized multicenter trial. Catheter Cardiovasc Interv. 2018;92:844-851.
27. Sinha SK, Jha MJ, Mishra V, et al. Radial Artery Occlusion. Incidence, Predictors and Long-term outcome after TRAnsradial Catheterization:clinico-Doppler ultrasound-based study (RAIL-TRAC study). Acta Cardiol. 2017;72:318-327.
28. Velasco A, Ono C, Nugent K, et al. Ultrasonic evaluation of the radial artery diameter in a local population from Texas. J Invasive Cardiol. 2012;24:339-341.
29. Peruga JP, Peruga JZ, Kasprzak JD, et al. Ultrasound evaluation of forearm arteries in patients undergoing percutaneous coronary intervention via radial artery access:results of one-year follow-up. Kardiologia Polska. 2015;73:502-510.
30. Knebel AV, Cardoso CO, Correa Rodrigues LH, et al. Safety and feasibility of transulnar cardiac catheterization. Tex Heart Inst J. 2008;35:268-272.
31. Yoo BS, Yoon J, Ko JY, et al. Anatomical consideration of the radial artery for transradial coronary procedures:arterial diameter, branching anomaly and vessel tortuosity. Int J Cardiol. 2005;101:421-427.
32. Pristipino C, Roncella A, Trani C, et al. Identifying factors that predict the choice and success rate of radial artery catheterisation in contemporary real world cardiology practice:a sub-analysis of the PREVAIL study data. EuroIntervention. 2010;6:240-246.
33. Louvard Y, Lefèvre T. Loops and transradial approach in coronary diagnosis and intervention. Catheter Cardiovasc Interv. 2000;51:250-252.
34. Ruiz-Salmerón RJ, Mora R, Vélez-Gimón M, et al. Radial artery spasm in transradial cardiac catheterization. Assessment of factors related to its occurrence, and of its consequences during follow-up. Rev Esp Cardiol. 2005;58:504-511.
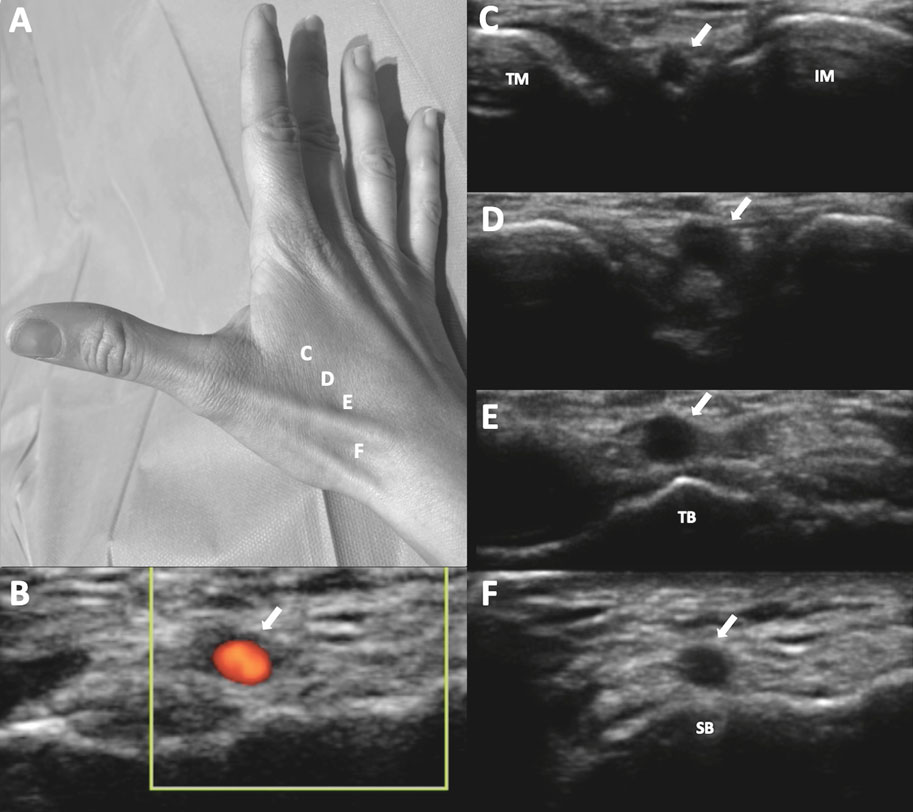
ABSTRACT
Introduction and objectives: Distal radial access (DRA) for coronary procedures is currently recognized as an alternative to conventional transradial access, with documented advantages primarily related to access-related complications. However, widespread adoption of DRA as the default approach remains limited. Therefore, this prospective cohort study aimed to present our initial experience with DRA for coronary procedures in any clinical settings.
Methods: From August 2020 to November 2023, we included 1000 DRA procedures (943 patients) conducted at a single center. The study enrolled a diverse patient population. We recommended pre- and postprocedural ultrasound evaluations of the radial artery course, with ultrasound-guided DRA puncture. The primary endpoint was DRA success, while secondary endpoints included coronary procedure success, DRA performance metrics, and the incidence of access-related complications.
Results: The DRA success rate was 97.4% (n = 974), with coronary procedure success at 96.9% (n = 969). The median DRA time was 40 [interquartile range, 30-60] seconds. Diagnostic procedures accounted for 64% (n = 644) of cases, while 36% (n = 356) involved percutaneous coronary intervention (PCI), including primary PCI in 13% (n = 128). Pre-procedure ultrasound evaluation and ultrasound-guided DRA were performed in 83% (n = 830) and 85% (n = 848) of cases, respectively. Access-related complications occurred in 2.9% (n = 29).
Conclusions: This study shows the safety and feasibility of DRA for coronary procedures, particularly when performed under ultrasound guidance in a diverse patient population. High rates of successful access and coronary procedure outcomes were observed, together with a low incidence of access-related complications. The study was registered on ClinicalTrials.gov (NTC06165406).
Keywords: Vascular access. Distal radial artery. Coronary angiography. Percutaneous transluminal coronary angioplasty. Doppler ultrasound. Access-related complications.
RESUMEN
Introducción y objetivos: Actualmente, el acceso radial distal (ARD) para procedimientos coronarios es una alternativa al acceso radial convencional, con algunas ventajas descritas principalmente en términos de complicaciones relacionadas con el acceso. A pesar de la evidencia, pocos centros han establecido el ARD como acceso sistemático para procedimientos coronarios. El objetivo de esta cohorte prospectiva es presentar la experiencia inicial en nuestro centro con el ARD en pacientes con indicación de procedimientos coronarios en cualquier escenario clínico.
Métodos: Se incluyeron 1.000 procedimientos de ARD (943 pacientes) realizados en un único centro de agosto de 2020 a noviembre de 2023. El estudio fue realizado con pacientes en cualquier escenario clínico. Se recomendó la valoración por ultrasonido del trayecto de la arteria radial antes y después del procedimiento, así como la punción ecoguiada. El objetivo principal fue el éxito del ARD. Como objetivos secundarios se consideraron el éxito del procedimiento coronario, el desempeño del ARD y las complicaciones relacionadas con el acceso.
Resultados: El éxito del ARD fue del 97,4% (n = 974) y el éxito del procedimiento coronario fue del 96,9% (n = 969). El tiempo de acceso del ARD fue de 40 segundos [rango intercuartílico, 30-60]. Se realizaron procedimientos diagnósticos en el 64% (n = 644) e intervencionismo coronario percutáneo (ICP) en el 36% (n = 356), incluyendo ICP primario en el 13% (n = 128) de los pacientes. La valoración por ultrasonido antes del procedimiento se llevó a cabo en el 83% (n = 830) y la punción ecoguiada en el 85% (n = 848). La incidencia de complicaciones relacionadas con el acceso fue del 2,9% (n = 29).
Conclusiones: Este estudio muestra la viabilidad y la seguridad del ARD principalmente guiado por ultrasonido para los procedimientos coronarios en cualquier escenario clínico, con un alto porcentaje de éxito del acceso y de éxito del procedimiento, además de una baja incidencia de complicaciones relacionadas con el acceso. El estudio fue registrado en ClinicalTrials.gov (NTC06165406).
Palabras clave: Acceso vascular. Arteria radial distal. Coronariografía. Angioplastia coronaria transluminal percutánea. Ultrasonido Doppler. Complicaciones relacionadas con el acceso.
Abbreviations
CAG: coronary angiography. DRA: distal radial access. DRart: distal radial artery. PRart: proximal radial artery. TRA: transradial access.
INTRODUCTION
Currently, distal radial access (DRA) in the anatomical snuffbox for both noncoronary and coronary procedures is gaining popularity. Since its introduction by Babunashvili et al.,1 in 2011, several observational studies have validated the feasibility and safety of DRA,2-4 comparing it with conventional transradial access (TRA). DRA has shown advantages such as a lower incidence of radial artery occlusion (RAO) and shorter hemostasis time, with minimal access-related complications.5,6 The usefulness of ultrasound to guide DRA and evaluate access-related complications has also been described.7,8 Recent randomized trials comparing DRA with TRA have reported conflicting results regarding RAO incidence, crossover rates, and access times.9-11 Nevertheless, meta-analyses consistently support the benefits of DRA, albeit with a higher crossover rate.12-13 One of the limitations of most studies on DRA is the restricted inclusion of patients in emergent situations or complex percutaneous coronary interventions (PCI), such as ST-segment elevation myocardial infarction (STEMI); therefore, the feasibility of the approach in this context is somewhat scarce.2,9-11,14 Despite current evidence, the use of DRA as the default access for coronary procedures is still not widely implemented in most centers. Hence, this prospective single-center cohort aimed to present the experience of our first 1000 DRA in patients undergoing coronary procedures in any clinical settings.
METHODS
Population and study design
The Distal Radial Access for Diagnostic and Interventional Coronary Procedures in an all-comer population (DISTAL) registry is a prospective observational investigation aiming to assess the performance of DRA and compare clinical and procedural characteristics in a diverse population undergoing coronary procedures. This interim analysis presents our initial experience with DRA conducted at a single center. All DRA procedures performed by 4 experienced operators, previously proficient in TRA, were included in the study from August 2020 to November 2023.
This study was approved by the Ethics Committee of our institution (CEIC-2804) and was conducted following the principles of the Declaration of Helsinki. All patients gave their informed written consent before the procedure.
Inclusion and exclusion criteria
The study included patients aged 18 years and older undergoing diagnostic or therapeutic coronary procedures using DRA in any clinical setting. Patients with an unsuitable distal radial artery (DRart) assessed by ultrasound (non-permeable or diameter <1 .8 mm) were excluded, as were patients with no palpable pulse of DRart with such unsuitability characteristics. Additional exclusion criteria encompassed participation in other clinical trials, known allergy to iodinated contrast, inability to provide informed consent, and women of childbearing age without a negative pregnancy test. While the Barbeau test was recommended, it was not mandatory for inclusion.15
Endpoints
The primary endpoint was the success of DRA and the main secondary endpoint was the success of the coronary procedure. Other secondary endpoints included DRA procedure time, total procedure duration, the incidence of radial artery spasm, exposure to ionizing radiation, patient comfort levels, hemostasis time, access-related complications, and the impact of ultrasound guidance on DRA performance. Detailed definitions of these endpoints are provided in the supplementary data.
Distal radial access technique
The DRA technique has been previously described,2,4,16-18 and is explained in detail in the supplementary data. Key aspects of interest included patient selection, the decision to use ultrasound-guided puncture19 (figure 1) vs blind with palpation puncture at the discretion of the operator, patient positioning for right (r) or left (l) DRA, the puncture technique itself, and the hemostasis procedure (figure 2).
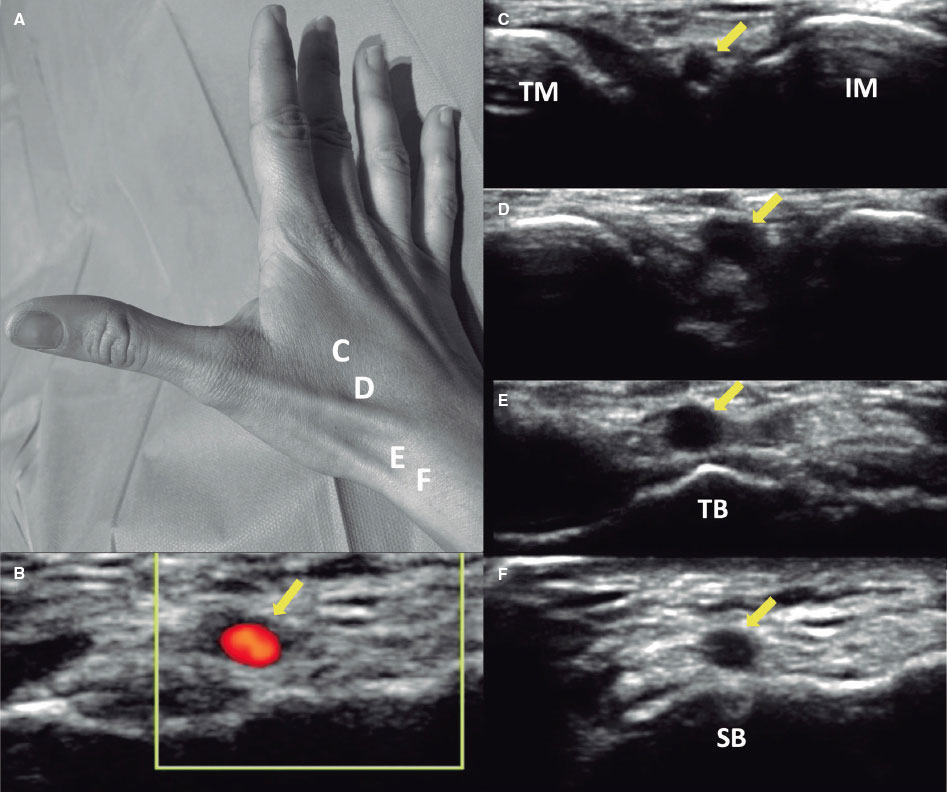
Figure 1. A: markers for ultrasound positioning in the anatomical snuffbox. B: patency of the distal radial artery (DRart) confirmed by color Doppler ultrasound. C-D: course of DRart between the metacarpal bones. E-F: recommended puncture sites of the DRart on a surface bone. IM, index metacarpal; SB, scaphoid bone; TB, trapezium bone; TM, thumb metacarpal.
Figure 2. Distal radial access (DRA) technique. Position of the hand for A) right DRA and B) left DRA. C: ultrasound-guided DRA technique. D: blind with palpation DRA puncture. E: final position of the introducer sheaths on the right and left DRA. F: hemostasis devices in DRA.
Statistical analysis
Sample size and statistical power calculations were performed using the GRANMO calculator.20 A sample size of 1000 procedures was determined to provide a statistical power greater than 99% to detect a difference of 3% or more in the proportion of DRA success (primary endpoint) at our center, assuming an alpha risk of 1%. This calculation was based on a reference proportion from previous medical literature estimated around 95%.11,18,21
Categorical variables are presented as counts (percentages), while continuous variables were assessed for normal distribution using the Kolmogorov-Smirnov test. Normally distributed variables are expressed as mean (standard deviation), and nonnormally distributed variables as median [interquartile range].
To evaluate the impact of the learning curve, comparisons were made among quartiles of the study period for variables including access failure, DRA time, total procedure time, and access-related complications. Analysis of variance or the Kruskal-Wallis test was used depending on the normality of the variable. Logistic regression analysis (logit command) was used with the first quartile as the reference to compare percentages among quartiles.
Statistical analyses were conducted using SPSS Statistics 20.0 software (IBM, United States) and STATA 12 (StataCorp, College Station, United States). A p-value < 0.05 was considered statistically significant for all tests.
RESULTS
From August 2020 to November 2023, a total of 1000 DRA procedures (943 patients) were performed. Table 1 shows the patients’ baseline clinical characteristics. The mean age was 68 years, and 29% of the patients were women. A total of 47% of the procedures were performed on an outpatient basis. In 35% of cases, the indication was acute coronary syndrome (13% STEMI).
Table 1. Baseline clinical characteristics
| Clinical characteristics | n = 1000 |
|---|---|
| Age, (years), mean (SD) | 68.1 (11.7) |
| Female, n (%) | 289 (28.9) |
| Weight, (kg), mean (SD) | 78.0 (14.8) |
| Height, (cm), mean (SD) | 167.9 (8.1) |
| Body mass index, (kg/m2), mean (SD) | 28.0 (4.5) |
| Hypertension, n (%) | 735 (73.5) |
| Dyslipidemia, n (%) | 578 (57.8) |
| Diabetes mellitus, n (%) | 353 (35.3) |
| Current smoker, n (%) | 246 (24.6) |
| Family history of premature coronary heart disease, n (%) | 54 (5.4) |
| Previous peripheral artery disease, n (%) | 50 (0.5) |
| Previous stroke, n (%) | 41 (4.1) |
| Previous heart failure, n (%) | 252 (25.2) |
| GFR (mL/minute/1.73m2), mean (SD) | 72.4 (20.0) |
| Dialysis, n (%) | 27 (2.7) |
| Left ventricular ejection fraction, mean (SD) | 52.6 (16.2) |
| Atrial fibrillation, n (%) | 170 (17.0) |
| OAC | |
| Acenocoumarin, n (%) | 170 (17.0) |
| Direct OAC, n (%) | 81 (8.1) |
| Previous CAG, n (%) | 251 (25.1) |
| Previous CABG, n (%) | 43 (4.3) |
| Previous PCI, n (%) | 218 (21.8) |
| Previous ischemic heart disease | |
| Previous STEMI, n (%) | 133 (13.3) |
| Previous NSTEMI, n (%) | 69 (6.9) |
| Previous CCS, n (%) | 53 (5.3) |
| CAG indication | |
| Chronic coronary syndrome, n (%) | 207 (20.7) |
| STEMI, n (%) | 128 (12.8) |
| NSTEMI, n (%) | 224 (22.4) |
| Staged PCI, n (%) | 60 (6.0) |
| Diagnostic, n (%) | 381 (38.1) |
| Preoperative CAG in patients with VHD, n (%) | 183 (18.3) |
| Dilated cardiomyopathy, n (%) | 158 (15.8) |
| Ventricular tachycardia, n (%) | 24 (2.4) |
| Others, n (%) | 16 (1.6) |
| Outpatient coronary arteriography, n (%) | 470 (47) |
|
CABG, coronary artery bypass grafting; CAG, coronary angiography; CCS, chronic coronary syndrome; GFR, glomerular filtration rate; NSTEMI, non−ST-segment elevation myocardial infarction; OAC, oral anticoagulation; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction; VHD, valvular heart disease. Data are expressed as No. (%) or mean ± standard deviation. |
|
Table 2 presents the characteristics of the radial artery and the DRA procedure. High rates of preprocedure ultrasound evaluation and ultrasound-guided technique for DRA were noted (83% and 85%, respectively). Notably, the percentage of coronary procedures showing insufficient catheter length due to DRA was low (3.7%).
Table 2. Characteristics of the DRA procedure
| Procedure characteristics | n = 1000 |
|---|---|
| Preprocedure characteristics | |
| Arterial pulse strength scale | |
| Absent, n (%) | 12 (1.2) |
| Weak, n (%) | 167 (16.7) |
| Normal, n (%) | 652 (65.2) |
| Strong, n (%) | 169 (16.9) |
| Radial artery preprocedure ultrasound evaluation, n (%) | 830 (83.0) |
| Arterial tortuosity | |
| Radial, n (%) | 23 (2.3) |
| Subclavian, n (%) | 62 (6.2) |
| Calcified radial artery, n (%) | 26 (2.6) |
| Distal radial artery size, mm (SD) | 2.3 (0.3) |
| Proximal radial artery size, mm (SD) | 2.5 (0.4) |
| Depth of the distal radial artery, mm (SD) | 3.8 (1.0) |
| DRA technique | |
| CAG by the same DRA, n (%) | 57 (5.7) |
| Ultrasound-guided access, n (%) | 848 (84.8) |
| DRA side | |
| Right DRA, n (%) | 627 (62.7) |
| Left DRA, n (%) | 373 (37.3) |
| Introducer size | |
| 5 French, n (%) | 256 (25.6) |
| 6 French, n (%) | 744 (74.4) |
| Introducer sheath type | |
| Prelude Ideal (Merit Medical) Introducer Kit, n (%) | 950 (95.0) |
| Radifocus Introducer II Kit A (Terumo Corporation), n (%) | 50 (5.0) |
| Short length of the radial catheter | 37 (3.7) |
| Postprocedure arterial patency evaluation, n (%) | 907 (90.7) |
| Postprocedure puncture site bleeding, n (%) | 55 (5.5) |
|
CAG, coronary angiography; DRA, distal radial access. Data are expressed as No. (%) or mean ± standard deviation. |
|
Table 3 summarizes the characteristics of coronary procedures, including the extent of coronary artery disease, types of procedures, and features of patients who underwent PCI. In general, 64% of the procedures were only diagnostic, while 36% included PCI.
Table 3. Characteristics of the coronary procedure
| Procedure characteristics | n = 1000 |
|---|---|
| Coronary disease extent | |
| One vessel, n (%) | 285 (28.5) |
| Two vessels, n (%) | 174 (17.4) |
| Three vessels, n (%) | 176 (17.6) |
| LMCAD, n (%) | 55 (5.5) |
| Coronary bypass graft, n (%) | 27 (2.7) |
| Characteristics of the coronary procedure | |
| Type of coronary procedures | |
| Diagnostic, n (%) | 644 (64.4) |
| PCI, n (%) | 356 (35.6) |
| Ambulatory PCI, n (%) | 90 (9.0) |
| PCI culprit lesion | |
| LMCAD, n (%) | 9 (0.9) |
| Left anterior descending artery, n (%) | 164 (16.4) |
| Circumflex coronary artery, n (%) | 95 (9.5) |
| Right coronary artery, n (%) | 100 (10.0) |
| Coronary bypass graft | 2 (0.2) |
| Specific techniques | |
| Wire-based intracoronary physiological assessment, n (%) | 57 (5.7) |
| Optical coherence tomography, n (%) | 21 (2.1) |
| Intravascular ultrasound, n (%) | 30 (3.0) |
| Guide catheter extension system, n (%) | 15 (1.5) |
| Rotational atherectomy, n (%) | 16 (1.6) |
| Cutting balloon, n (%) | 34 (3.4) |
| Intracoronary lithotripsy, n (%) | 8 (8.0) |
| Thrombus aspiration, n (%) | 81 (8.1) |
| Intracoronary perfusion catheter, n (%) | 7 (0.7) |
| Special PCI procedures | |
| Complex bifurcation, n (%) | 60 (6.0) |
| Chronic total occlusion, n (%) | 16 (1.6) |
| Volume of contrast, (mL), mean (SD) | 85.0 (53.1) |
| Heparin dose, (IU), median [IQR] | 5000 (3000-8500) |
|
LMCAD, left main coronary artery disease; PCI, percutaneous coronary intervention. |
|
Table 4 depicts the clinical endpoints. The DRA success rate was 97.4% and the coronary procedure success rate was 96.9%. The median access time was 40 (interquartile range [IQR], 30-60) seconds, and 4% of patients experienced radial artery spasm. The overall rate of access-related complications was low (2.9%).
Table 4. Clinical endpoints
| Clinical endpoints | n = 1000 |
|---|---|
| Primary endpoint | |
| DRA success, n (%) | 974 (97.4) |
| Coronary procedure success by DRA, n (%) | 969 (96.9) |
| Secondary endpoints | |
| Access time, (sec), median [IQR] | 40 (30-60) |
| Procedure time, (min), median [IQR] | 29.0 [17.3-45.0] |
| Radial artery spasm, n (%) | 44 (4.4) |
| DAP, (Gy.m2), median [IQR] | 32.7 [19.2-63.0] |
| Fluoroscopy time (min), median [IQR] | 4.6 [2.5-10.0] |
| VAS patient comfort for access, mean (SD) | 2.2 (0.6) |
| VAS patient comfort for hemostasis, mean (SD) | 2.1 (0.4) |
| Hemostasis time, (hour), mean, (SD) | 2.9 (1.1) |
| Access-related complications (all), n (%) | 29 (2.9) |
| Radial artery occlusion, n (%) | 10 (1.0) |
| Hematoma, n (%) | |
| Type I-a, n (%) | 11 (1.1) |
| Type I-b, n (%) | 1 (0.1) |
| Type II, n (%) | 1 (0.1) |
| Type III, n (%) | 1 (0.1) |
| Type IV, n (%) | 0 (0) |
| Radial pseudoaneurysm, n (%) | 0 (0) |
| Radial dissection, n (%) | 5 (0.5) |
| Arteriovenous fistula, n (%) | 0 (0) |
|
DAP, dose-area product; DRA, distal radial access; VAS, visual analog scale. Data are expressed as No. (%), mean ± standard deviation, or median [interquartile range]. |
|
Combined preprocedure ultrasound evaluation and ultrasound-guided puncture were performed in 82.8% of cases, with successful DRA achieved in 97.7% compared with 95.9% in those who did not undergo ultrasound guidance (P = .183). Based on the strength of the arterial pulse—absent, weak, normal, and strong—ultrasound-guided puncture was performed in 100%, 91%, 89.7%, and 45.5% of cases, respectively. Access time was longer with ultrasound-guided puncture than with nonultrasound-guided puncture (40 s [30-70] vs 35 s [30-45]; P < .001). The success of DRA in relation to the use of ultrasound-guided technique among all strengths of arterial pulse is detailed in table 1 of the supplementary data.
Arterial patency after removal of the hemostatic device was assessed in 907 patients (90.7%), revealing RAO in only 1% (n = 10).
In the quartile analysis, a shift in the selection of DRA side was observed, with lDRA initially more commonly used, shifting to rDRA as the preferred access in later quartiles (figure 3A). DRA failure rates were low in all quartiles but decreased significantly from the third quartile onwards (figure 3B). Access time decreased significantly from the second quartile onwards and remained stable thereafter (figure 3C). However, no significant differences were found in total procedure duration between quartiles (figure 3D).
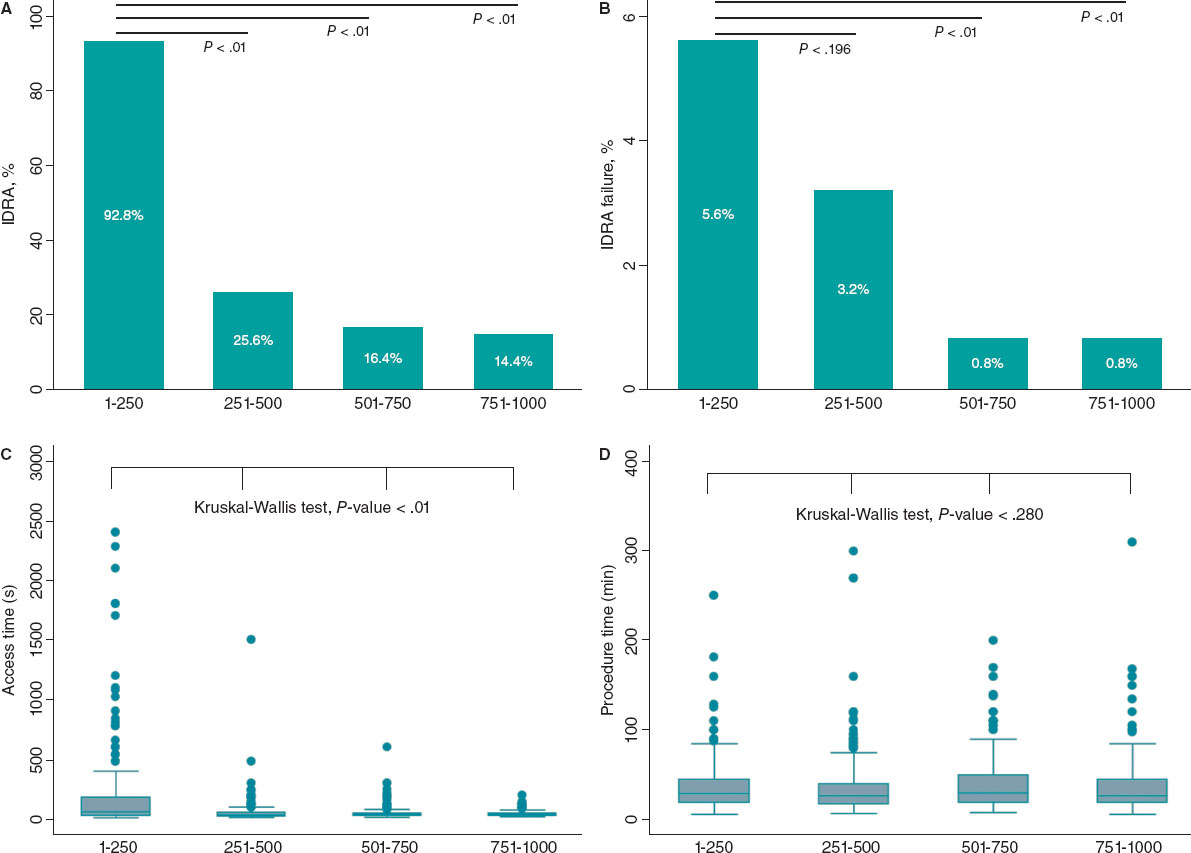
Figure 3. Stratified analysis by quartiles of patients over the study period. A: use of left vs right distal radial access (DRA). B: DRA access failure rate by quartile. C: DRA access time in seconds. D: total procedural time in minutes.
DISCUSSION
Using data from a large prospective registry of patients who underwent DRA for coronary procedures, with high use of ultrasound-guided techniques, our study showed that DRA achieves high rates of access and procedural success, coupled with a low incidence of access-related complications in an all-comer population.
The usefulness of ultrasound in the distal radial access technique
Understanding the anatomy of the anatomical snuffbox is crucial for successful DRA, and ultrasound serves as a valuable tool in achieving this, offering demonstrated advantages.5,16,17,22 In our study, preprocedure ultrasound evaluation and ultrasound-guided DRA techniques were used in most patients. In addition to assessing arterial diameters and evaluating calcification and tortuosity, ultrasound enabled us to exclude patients with unsuitable distal radial arteries. Overall, we found no significant differences between ultrasound-guided and nonultrasound-guided DRA, although the former was associated with longer access times. However, the role of ultrasound is particularly noteworthy in cases of weak or absent arterial pulses, which are often underrepresented in prior studies. The presence of a suboptimal arterial pulse can stem from various factors, including small DRart, hypotension, collateral blood supply, or depth of DRart.11 In our study, most patients with weak pulses underwent ultrasound-guided puncture, with a favorable trend toward successful access in those who did. However, in patients with normal to strong pulses, no differences in DRA success were found, and even prolongation of access time was observed with its use. Therefore, in this type of pulse, an ultrasound-guided puncture is probably not necessary.
Feasibility, safety, and technical issues in distal radial access
This study corroborates the previously reported advantages of DRA,3,9,10,12,13,18 such as a low rate of RAO, acceptable access time, short hemostasis time, and adequate patient comfort.
Furthermore, the absence of an increased risk of hand dysfunction after DRA has been demonstrated,23 even compared with TRA at 12 months of follow-up, documented by Al-Azizi et al.24 Here, we focus on controversial issues that may have hampered wider adoption of this technique, and our results may provide additional support for DRA.
High success rates of DRA in coronary procedures have been reported in numerous studies.2-4,17,18,25 In addition, recent clinical trials and meta-analyzes describe a higher crossover rate compared with TRA.9-13
In contrast to our results, trials comparing DRA with TRA have reported lower access success and longer puncture times.9-11 Conversely, our study demonstrates remarkably high success rates for DRA and coronary procedures, as well as shorter access time, consistent with registries in which DRA is the default approach among experienced operators, as shown by the largest registries published to date, the DISTRACTION and KODRA studies.2-4,18,21
The KODRA trial included 4977 DRA procedures from a Korean registry.21 The authors reported a DRA success rate of 94.4%, with a crossover rate of 6.7%. In contrast to our work, the use of ultrasound-guided puncture in KODRA was low (6.4%). Additionally, the authors found predictors of DRA failure, such as the presence of a weak pulse and limited operator experience (less than 100 cases).
The equivalence of rDRA and lDRA has previously been demonstrated, and contemporary studies use mainly rDRA.9-11,17 As in the first registries, which suggested a potential advantage of lDRA, we started our experience with lDRA but, based on operator comfort and preference, the use of the rDRA increased over time.
Although the feasibility and benefits of DRA over TRA in STEMI have been observed, the literature on the topic remains scarce.2,9-11 In our registry, all attempted DRA procedures in patients with STEMI were successful. However, the first DRA in STEMI was performed after the operators had surpassed the learning curve for the technique (up to case 320). Similarly, the use of DRA for complex PCI has been previously described.22,26,27 In our cohort, all complex PCI procedures were performed without crossover.
The puncture site in DRA, situated 5 cm distal to TRA, may lead to an inadequate catheter length in specific contexts (such as tall patients, dilated aorta, subclavian artery tortuosity, and the need for retrograde access to PCI for chronic total occlusions).28 We found a low incidence of short catheter length during DRA procedures, with only 1 crossover due to severe tortuosity of the subclavian artery.
DRA-related complications have been consistently reported to be low.2,9-11,18 Similarly, we found a very low rate of complications, the most common being type I-a hematoma. In our study, the incidence of in-hospital RAO was 1%.
The number of DRA procedures to overcome the learning curve and maintain a success rate above 94% is around 150 to 200.2,8 However, in our early experience, we achieved this percentage after the first 20 cases per operator.17 In this study, operators navigated the learning curve in the first quartile; however, success significantly improved to more than 99% in the last 2 quartiles, probably because DRA became the default access for coronary procedures among operators.
Limitations
First, this study was an interim analysis of the leading participating site and coordinator of the DISTAL registry (NTC06165406), conducted because substantial enrollment from other sites was lacking. Although the data cannot be fully extrapolated to other centers, recalculation of the sample size was considered sufficient to evaluate the results.
Second, patient enrollment was not consecutive because the decision to use DRA was at the operators’ discretion. Only one-third of coronary procedures during the study period used this approach. However, we included all patients in whom operators intended to use DRA in any clinical setting were included, with only 21 patients excluded due to DRart ≤1.8mm. Third, this was a descriptive cohort of DRA, without a comparison control group. Fourth, the scale used to assess the arterial pulse is subjective. However, this scale is widely used in routine clinical practice and has been used in multiple DRA studies. Finally, radial artery patency was not evaluated in 9.7% of the patients before discharge, and no evaluation was conducted at 1 month; therefore, the in-hospital rate of radial artery occlusion may be underestimated and no mid-term data are available on the patency of the DRart.
CONCLUSIONS
This study shows the safety and feasibility of DRA primarily guided by ultrasound for coronary procedures in an all-comer population, with high rates of both access and procedural success, in addition to a very low rate of access-related complications.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
This study was approved by the Ethics Committee of our institution (CEIC-2804) and was conducted following the principles of the Declaration of Helsinki. All patients gave their informed written consent before the procedure.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Not used.
AUTHORS’ CONTRIBUTIONS
K. Rivera and D. Fernández-Rodríguez conceived and designed the study. K. Rivera, D. Fernández-Rodríguez, M. García-Guimarães, J. Casanova-Sandoval, and J. L. Ferreiro analyzed data, and drafted the manuscript. All authors contributed to the treatment of patients, data acquisition and mining, and review and approval of the final version of the manuscript.
CONFLICTS OF INTEREST
J. L. Ferreiro reports a) honoraria for lectures from Eli Lilly Co, Daiichi Sankyio, Inc, AstraZeneca, Pfizer, Abbott, Boehringer Ingelheim, Bistol-Myers Squibb, Rovi, Terumo and Ferrer; b) consulting fees from AstraZeneca, Eli Lilly Co, Ferrer, Boston Scientific, Pfizer, Boehringer Ingelheim, Daiichi Sankyo, Inc, Bristol-Myers Squibb and Biotronik; c) research grants from AstraZeneca. The remaining authors have no conflicts of interest to declare.
WHAT IS KNOWN ABOUT THE TOPIC?
- Previous studies have demonstrated the safety and feasibility and safety DRA. Compared with TRA, DRA has several advantages, despite the high prevalence of crossover and controversial incidence of radial artery occlusion.
WHAT DOES THIS STUDY ADD?
- The results of this cohort show the safety and feasibility of DRA in an all-comer population throughout the spectrum of DRart pulses. Our study demonstrates that preprocedure ultrasound evaluation and the ultrasound-guided DRA technique help to achieve a low crossover rate, which is especially useful in patients with an unfavorable arterial pulse. According to our observations, DRA in urgent/emergent procedures and complex PCI is feasible and safe once the learning curve has been overcome and the operator is familiar with the technique.
REFERENCES
1. Babunashvili A, Dundua D. Recanalization and reuse of early occluded radial artery within 6 days after previous transradial diagnostic procedure. Catheter Cardiovasc Interv. 2011;77:530-536.
2. Lee JW, Park SW, Son JW, Ahn SG, Lee SH. Real-world experience of the left distal transradial approach for coronary angiography and percutaneous coronary intervention:A prospective observational study (LeDRA). EuroIntervention. 2018;14:e995-e1003.
3. Oliveira MDP, Navarro EC, Kiemeneij F. Distal transradial access as default approach for coronary angiography and interventions. Cardiovasc Diagn Ther. 2019;9:513-519.
4. Kiemeneij F. Left distal transradial access in the anatomical snuffbox for coronary angiography (ldTRA) and interventions (ldTRI). EuroIntervention. 2017;13:851-857.
5. Sgueglia GA, Di Giorgio A, Gaspardone A, Babunashvili A. Anatomic Basis and Physiological Rationale of Distal Radial Artery Access for Percutaneous Coronary and Endovascular Procedures. JACC Cardiovasc Interv. 2018;11:2113-2119.
6. Lu H, Wu D, Chen X. Comparison of Distal Transradial Access in Anatomic Snuffbox Versus Transradial Access for Coronary Angiography. Heart Surg Forum. 2020;23:E407-E410.
7. Ghose T, Kachru R, Dey J, Khan WU, et al. Safety and Feasibility of Ultrasound-Guided Access for Coronary Interventions through Distal Left Radial Route. J Interv Cardiol. 2022;2022:2141524.
8. Roh JW, Kim Y, Lee OH, et al. The learning curve of the distal radial access for coronary intervention. Sci Rep. 2021;11:13217.
9. Tsigkas G, Papageorgiou A, Moulias A, et al. Distal or Traditional Transradial Access Site for Coronary Procedures:A Single-Center, Randomized Study. JACC Cardiovasc Interv. 2022;15:22-32.
10. Aminian A, Sgueglia GA, Wiemer M, et al. Distal Versus Conventional Radial Access for Coronary Angiography and Intervention:The DISCO RADIAL Trial. JACC Cardiovasc Interv. 2022;15:1191-1201.
11. Kozin´ski Ł, Orzałkiewicz Z, Da˛browska-Kugacka A. Feasibility and Safety of the Routine Distal Transradial Approach in the Anatomical Snuffbox for Coronary Procedures:The ANTARES Randomized Trial. J Clin Med. 2023;12:7608.
12. Ferrante G, Condello F, Rao SV, et al. Distal vs Conventional Radial Access for Coronary Angiography and/or Intervention:A Meta-Analysis of Randomized Trials. JACC Cardiovasc Interv. 2022;15:2297-2311.
13. Barbarawi M, Barbarawi O, Jailani M, Al-Abdouh A, Mhanna M, Robinson P. Traditional versus distal radial access for coronary angiography:A meta-Analysis of randomized controlled trials. Coron Artery Dis. 2023;34:274-280.
14. Erdem K, Kurtogˇlu E, Küçük MA, Ilgenli TF, Kizmaz M. Distal transradial versus conventional transradial access in acute coronary syndrome. Turk Kardiyoloji Dernegi Arsivi. 2021;49:257-265.
15. Valgimigli M, Campo G, Penzo C, Tebaldi M, Biscaglia S, Ferrari R. Transradial coronary catheterization and intervention across the whole spectrum of allen test results. J Am Coll Cardiol. 2014;63:1833-1841.
16. Sgueglia GA, Lee BK, Cho BR, et al. Distal Radial Access:Consensus Report of the First Korea-Europe Transradial Intervention Meeting. JACC Cardiovasc Interv. 2021;14:892-906.
17. Rivera K, Fernández-Rodríguez D, Casanova-Sandoval J, et al. Comparison between the Right and Left Distal Radial Access for Patients Undergoing Coronary Procedures:A Propensity Score Matching Analysis. J Interv Cardiol. 2022;2022:7932114.
18. Oliveira MD, Navarro EC, Caixeta A. Distal transradial access for coronary procedures:A prospective cohort of 3,683 all-comers patients from the DISTRACTION registry. Cardiovasc Diagn Ther. 2022;12:208-219.
19. Hadjivassiliou A, Kiemeneij F, Nathan S, Klass D. Ultrasound-guided access to the distal radial artery at the anatomical snuffbox for catheter-based vascular interventions:A technical guide. EuroIntervention. 2021;16:1342-1348.
20. Calculadora de tamaño muestral GRANMO. Available at:https://www.imim.cat/media/upload/arxius/granmo/granmo_v704.html. Accessed 25 Mar 2024.
21. Lee JW, Kim Y, Lee BK, et al. Distal Radial Access for Coronary Procedures in a Large Prospective Multicenter Registry:The KODRA Trial. JACC Cardiovasc Interv. 2024;17:329-340.
22. Zong B, Liu Y, Han B, Feng CG. Safety and feasibility of a 7F thin-walled sheath via distal transradial artery access for complex coronary intervention. Front Cardiovasc Med. 2022;9:959197.
23. Sgueglia GA, Hassan A, Harb S, et al. International Hand Function Study Following Distal Radial Access:The RATATOUILLE Study. JACC Cardiovasc Interv. 2022;15:1205-1215.
24. Al-Azizi K, Moubarak G, Dib C, et al. Distal Versus Proximal Radial Artery Access for Cardiac Catheterization:1-Year Outcomes. Am J Cardiol. 2024;220:102-110.
25. Rivera K, Fernández-Rodríguez D, Bullones J, et al. Impact of sex differences on the feasibility and safety of distal radial access for coronary procedures:a multicenter prospective observational study. Coron Artery Dis. 2024;35(5):360-367.
26. Rivera K, Fernández-Rodríguez D, García-Guimarães M, Ramírez Martínez T, Casanova-Sandoval J. Intravascular ultrasound-guided percutaneous exclusion of a complicated coronary artery aneurysm presenting as ST-segment elevation myocardial infarction. Coron Artery Dis. 2023;34:527-528.
27. Nikolakopoulos I, Patel T, Jefferson BK, et al. Distal Radial Access in Chronic Total Occlusion Percutaneous Coronary Intervention:Insights From the PROGRESS-CTO Registry. J Invasive Cardiol. 2021;33:E717-E722.
28. Davies RE, Gilchrist IC. Back hand approach to radial access:The snuff box approach. Cardiovasc Revasc Med. 2018;19:324-326.
ABSTRACT
Introduction and objectives: Drug-eluting balloons (DEB) are an established treatment option for in-stent restenosis (ISR). This study aimed to assess the safety and efficacy of a novel DEB in patients with ISR.
Methods: This prospective, single-center study enrolled a consecutive cohort of patients diagnosed with ISR who underwent coronary angioplasty with a new second-generation paclitaxel-eluting balloon. The 3 main endpoints were myocardial infarction, target lesion revascularization, and target vessel revascularization. Baseline variables were collected, including patient and procedure characteristics. Follow-up data were collected through medical records or telephone contact.
Results: The study included 160 consecutive patients with 206 treated lesions (mean age, 71.4 ± 14.9 years, 15.5% women) undergoing percutaneous coronary intervention with DEB for ISR. A total of 53.3% of patients had acute coronary syndrome. The average diameter of the treated vessel was 3.10 ± 0.7 mm. The DEB used had a mean diameter of 3.1 ± 0.6 mm and a mean length of 23.1 ± 6.8 mm. Predilatation was performed in 98% of the lesions, and a noncompliant balloon was used in 80%. Intracoronary imaging was used in 24% of cases. At the end of the procedure, 98.5% of patients had Thrombolysis in Myocardial Infarction flow grade 3, residual stenosis was > 30% in 3.4%, and dissection occurred in 1.4%. Bail-out stenting was required in 4.8% of patients. Mortality was nil during follow-up (maximum 768 days). The incidence of myocardial infarction, target lesion revascularization, and target vessel revascularization were 5.4% (95%CI, 0.69-10.1), 8.4% (95%CI, 0-17.8), and 14.2% (95%CI, 3.61-24.78), respectively.
Conclusions: In this cohort of patients with ISR treated with DEB, we observed a low rate of adverse events in both the short- and mid-term. These results support the safety and efficacy of this new generation of DEB for treating ISR.
Keywords: In-stent restenosis. Drug-eluting balloon. Paclitaxel.
RESUMEN
Introducción y objetivos: El balón farmacoactivo (BFA) es un tratamiento establecido para tratar la reestenosis intrastent (RIS). El objetivo de este estudio fue valorar la eficacia y la seguridad de un nuevo BFA en pacientes con RIS.
Métodos: Cohorte prospectiva, unicéntrica y consecutiva de pacientes con RIS tratados con angioplastia coronaria con un nuevo balón liberador de paclitaxel de segunda generación. Los 3 eventos principales del estudio fueron infarto de miocardio, revascularización de la lesión diana y revascularización del vaso diana. Se recogieron variables basales, incluidas las características del paciente y del procedimiento. Los datos referentes al seguimiento se obtuvieron de registros médicos o por contacto telefónico.
Resultados: Se incluyeron 160 pacientes consecutivos con 206 lesiones tratadas (71,4 ± 14,9 años, el 15,5% mujeres) que fueron tratados con una intervención coronaria percutánea con BFA debido a RIS. El 53,3% de los pacientes presentaban síndrome coronario agudo. El diámetro medio del vaso tratado fue de 3,1 ± 0,7 mm. El diámetro y la longitud del BFA empleado fueron de 3,1 ± 0,6 mm y 23,1 ± 6,8, respectivamente. El 98% de las lesiones se predilataron y en el 80% se empleó un balón no distensible. El 24% de las angioplastias fueron guiadas por imagen intracoronaria. El 98,5% de los pacientes presentaban un flujo Thrombolysis in Myocardial Infarction de grado 3 al final de la angioplastia. Hubo estenosis residual > 30% en el 3,4%, y el 1,4% presentaron disección. El 4,8% de los pacientes requirieron stent de rescate. Al finalizar el seguimiento (máximo 768 días), ningún paciente había fallecido. Las incidencias de infarto de miocardio, de revascularización de la lesión diana y de revascularización del vaso diana fueron del 5,4% (IC95%, 0,69-10,1), el 8,4% (IC95%, 0-17,8) y el 14,2% (IC95%, 3,61-24,78), respectivamente.
Conclusiones: En esta cohorte de pacientes con RIS tratados con BFA se observa una baja tasa de eventos clínicos adversos, tanto a corto como a mediano plazo. Estos resultados respaldan la eficacia y la seguridad de esta nueva generación de BFA para pacientes con RIS.
Palabras clave: Reestenosis intrastent. Balón farmacoactivo. Paclitaxel.
Abbreviations
DEB: drug-eluting balloon. ISR: in-stent restenosis. TLR: target lesion revascularization. TVR: target vessel revascularization.
INTRODUCTION
Patients with coronary in-stent restenosis (ISR) represent a clinical challenge.1 Evidence indicates that these patients are at increased risk of recurrent symptoms, myocardial infarction, and repeated coronary revascularizations.2 The use of drug-eluting balloons (DEB) is a novel alternative therapeutic strategy in patients with ISR.1,3,4 The effect of DEBs in coronary angioplasty is based on the rapid and uniform transfer of antiproliferative drugs into the vessel wall using a single balloon through a lipophilic matrix without the need for permanent implants.5
Over time, new DEB technologies are developed and launched onto the market. The Essential Pro (iVascular, Spain) is a paclitaxel-eluting balloon catheter with advancements to enhance catheter pushability and drug delivery. We believe it is essential to report outcomes from real-world settings. In this study, we report our findings on the safety and efficacy of this new DEB in patients with ISR.
METHODS
Design and population
This prospective, single-center study included a cohort of consecutive patients undergoing DEB angioplasty with the Essential Pro. The center treating these patients performs more than 1500 percutaneous coronary interventions per year. The 2 inclusion criteria for this analysis were: a) use of an Essential Pro DEB and b) its application for ISR treatment. ISR was defined as stenosis more than 50% within the stented segment, and treatment was indicated according to the treating physician’s judgment.6 The use of the Essential Pro DEB was prioritized during the study period to treat all eligible patients for DEB angioplasty, while other DEB devices were rarely used due to inventory constraints. There were no exclusion criteria. Patients may have undergone stent coronary angioplasty of other lesions in the same or a different setting.
Drug-eluting balloon characteristics
The Essential Pro is a paclitaxel-eluting balloon with a uniform 3 μg/mm2 eluting formulation, consisting of paclitaxel (80%) and a biocompatible amphiphilic excipient (20%).7 The balloon incorporates the proprietary TransferTech technology (iVascular, Spain), which is based on the ultrasonic deposition of nanodrops, followed by a dry-off process, resulting in a homogeneous microcrystalline drug coating. This allows more uniform and complete treatment of the vessel with the antiproliferative drug. The microcrystalline structure, coupled with the lipophilic nature of both paclitaxel and the excipient, facilitates drug transfer within 45 to 60 seconds. The Essential Pro balloon has been designed with a smooth transition and a very low tip profile of 0.016 inches, enhancing flexibility, trackability, and device crossability. The balloon is compatible with 5-Fr sheaths in all available diameters.
Procedures
All procedures and decisions in this study reflect real-world clinical practice. Therefore, clinical indications, the use and selection of DEBs, procedural steps, and medical treatments were decided by treating physicians without following any specific guidelines. All coronary angiograms performed during follow-up were part of routine clinical practice and were assessed by our research team when available. Baseline and follow-up data were collected in a single anonymized dedicated database. Procedural aspects, as well as both baseline and follow-up angiograms, were independently evaluated by 3 different interventional cardiologists. Physicians were trained to consult senior staff if they had doubts when assessing angiograms or clinical records. Follow-up was conducted using clinical records, and patients with no on-site clinical visits during follow-up were contacted by telephone following standard clinical practice in our institution. This study was approved by our local institutional review board and patients provided consent for the use of their anonymized information for research purposes before inclusion. This was an investigator-initiated study with no sponsoring or funding.
Outcome definitions
Device delivery was defined as successful DEB insufflation in the affected coronary segment. Procedural, angiographic, and other standard outcomes were defined according to the Second Academic Research Consortium criteria.8 Cardiovascular mortality was defined as any death without a clear noncardiovascular cause. Acute myocardial infarction was defined as any myocardial infarction meeting the fourth version of the Universal Myocardial Infarction Criteria.9 Target lesion revascularization (TLR) was defined as any revascularization within or 5 mm beyond the treated segment.8 Target vessel revascularization (TVR) was defined as revascularization of the index treated vessel.8 Coronary-related hospitalization was defined as a new hospitalization in which a coronary origin was suspected as the main reason for admission. The 3 main efficacy outcomes were myocardial infarction, TLR, and TVR.
Statistical analysis
Categorical variables are presented as percentages, and continuous variables as mean standard deviation (SD) when appropriate. Since the same patient may receive more than 1 DEB (for the same or different territory), the denominator for balloon-specific variables was based on the total DEBs used (such as treated vessel, vessel diameter, DEB diameter, and length), while the denominator of patient-level variables (such as age, sex, or clinical outcomes) was each single individual. Clinical outcomes during follow-up are presented at 30 days, 1 year, and total follow-up. The Kaplan-Meier method was used for estimating both the total follow-up risk and generating survival curves. Data were analyzed using IBM SPSS Statistics 25.
RESULTS
From December 2020 to June 2023, 290 patients with 352 coronary lesions were treated with DEB. Among them, 160 patients (206 lesions) underwent DEB angioplasty due to ISR. Out of the 160 patients receiving DEB for ISR, 46 patients (29%) received more than 1 DEB angioplasty for ISR, either during the same procedure or staged to a different lesion.
The patients’ baseline characteristics are summarized in table 1. The mean age was 71.4 ± 14.9 years, 15.5% were women, and 35.5% had diabetes. Clinical presentation was stable angina in 29.7%, unstable angina in 30.5%, non–ST-segment elevation myocardial infarction in 9.9%, ST-segment elevation myocardial infarction in 12.9%, and 16.7% were asymptomatic.
Table 1. Baseline characteristics
| Patient characteristics | |
| Age, y | 71.4 (14.9) |
| Sex women | 20 (15.5) |
| BMI, kg/m2 | 29.2 (10.5) |
| Hypertension | 115 (87.7) |
| Active smoking | 8 (6.1) |
| Diabetes mellitus | 46 (35.3) |
| Previous MI | 67 (51.5) |
| Previous CABG | 26 (20) |
| Reduced LVEF (< 30%) | 10 (7.6) |
| Laboratory parameters | |
| Hemoglobin, g/dL | 13.9 (1.5) |
| GFR, mL/min/1.73 m2 | 82.9 (25.4) |
| Active medication | |
| Aspirin | 110 (84.6) |
| Clopidogrel | 75 (57.6) |
| Ticagrelor | 3 (2.3) |
| Prasugrel | 2 (1.5) |
| Anticoagulation | 20 (15.2) |
| Clinical presentation | |
| Silent ischemia | 22 (16.7) |
| Stable angina | 39 (29.7) |
| Unstable angina | 40 (30.5) |
| NSTEMI | 13 (9.9) |
| STEMI | 17 (12.9) |
|
BMI, body mass index; CABG, coronary artery bypass grafting; GFR, glomerular filtration rate; LVEF, left ventricular ejection fraction; MI, myocardial infarction; NSTEMI, non–ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction. Data are expressed as No. (%). |
|
Procedural characteristics are detailed in table 2. The most commonly treated vessel was the left anterior descending artery (48.7%), followed by the left circumflex (30.7%), and the right coronary artery (17%). Bifurcation was present in 10.7%. Lesion preparation was performed in 98.2% of cases (80% with a noncompliant balloon). Intracoronary imaging was used in 24% of patients. None of the patients underwent rotational atherectomy, and 2.4% underwent balloon lithotripsy before DEB delivery. The mean vessel diameter was 3.1 ± 0.65 mm. The mean DEB diameter was 3.1 ± 0.6 mm, and the mean length was 23.1 ± 6.8 mm. Device delivery was successful in 100% of cases (figure 1). The final angiographic assessment revealed a final dissection in 1.4%, Thrombolysis in Myocardial Infarction flow less than 3 in 1.5%, and residual stenosis more than 30% in 3.4%. Bail-out stenting was needed in 4.8%.
Table 2. Characteristics of the treated lesion
| Treated vessel | |
| LAD | 100 (48.7) |
| LCx | 63 (30.7) |
| Right coronary artery | 35 (17) |
| Left main coronary artery | 5 (2.4) |
| Graft | 2 (0.9) |
| Anatomical characteristics | |
| Bifurcation lesion | 22 (10.7) |
| Vessel diameter, mm | 3.1 (0.65) |
| Procedural characteristics | |
| IVUS-guided PCI | 51 (24) |
| Lesion predilatation | 202 (98) |
| Predilatation with NC balloon | 165 (80) |
| Intravascular lithotripsy | 5 (2.4) |
| DEB diameter, mm | 3.1 (0.6) |
| DEB length, mm | 23.1 (6.8) |
| Result after DEB PCI | |
| Vessel dissection | 3 (1.4) |
| TIMI flow 3 | 203 (98.5) |
| Residual stenosis > 30% | 194 (3.4) |
| Bail-out stenting | 10 (4.8) |
|
DEB, drug-eluting balloon; IVUS, intravascular ultrasound; LAD, left anterior descending artery; LCx, left circumflex artery; NC, noncompliant; PCI, percutaneous coronary intervention; RCA, right coronary artery; TIMI, Thrombolysis in Myocardial Infarction. Data are expressed as No. (%). |
|
Figure 1. Central illustration. Main findings on the safety and efficacy of the Essential Pro drug-eluting balloon in patients with in-stent restenosis. Kaplan-Meier shows freedom from TLR. MI, myocardial infarction; TLR, target lesion revascularization; TVR, target vessel revascularization.
After discharge, 93.3% of the patients were successfully contacted. The median follow-up was 361 days, including censored patients, with a maximum of 768 days. At 30 days of follow-up, there were no deaths or TLR, there was 1 myocardial infarction (0.6%), TVR occurred in 0.6%, and 6 patients were readmitted to hospital due to a coronary syndrome (4.1%). At the 1-year follow-up, mortality was 0%, myocardial infarction occurred in 3.4%, TLR in 2.5%, TVR in 6.3%, and coronary-related rehospitalizations in 11.8%. At 18 months, the TLR rate was 4.3%. When all available follow-up was included (figure 2), mortality was 0%, myocardial infarction occurred in 5.4% (95% confidence interval [95%CI], 0.69-10.1), TLR in 8.4% (95%CI, 0-17.8), and TVR in 14.2% (95%CI, 3.61-24.78). During follow-up, none of the patients underwent surgical revascularization.
Figure 2. Survival curves of key clinical outcomes. Kaplan-Meier estimates for survival free from myocardial infarction (A), target lesion revascularization (B), and target vessel revascularization (C) in days. 95%CI, 95% confidence interval; TLR, target lesion revascularization; TVR, target vessel revascularization.
DISCUSSION
This is the first study to describe a real-world experience with the Essential Pro DEB for the treatment of ISR. In this cohort, all attempts at DEB delivery were successful, and less than 1 in 20 patients required bail-out stenting. The use of this new-generation DEB catheter was associated with high efficacy and a low incidence of adverse clinical outcomes during follow-up.
Patients with ISR are at higher risk of recurrent events than those undergoing non-ISR angioplasty.10 The annual rate of ISR requiring TLR is around 2%,3 representing up to 11% of all percutaneous coronary interventions performed in the United States.11,12 Notably, 52% of patients presenting with symptomatic ISR have unstable angina, and up to 27% have an acute myocardial infarction.12 Therefore, ISR poses a significant clinical challenge due to both its frequency and severity. The use of DEB in the ISR scenario avoids the addition of extra stent layers, which may have detrimental effects in the long term.
The use of DEB in ISR poses certain challenges. DEB platforms commonly have lower lesion crossability than regular coronary balloon catheters. DEBs also have larger profiles than conventional balloons making it difficult to cross the lesion and requiring aggressive maneuvers that could lead to a loss of coating drug during delivery.13 However, in our study, all attempted DEB deployments were successful. This high success rate may be due to improvements in second-generation DEBs, as well as better lesion evaluation and lesion preparation.
In the present study, TLR occurred in 2.5% of the patients and TVR in 6.3% at 1 year, while TLR occurred in 4.3% at 18 months. This event rate may seem low when compared with a prior systematic review of randomized and observational studies, which reported a TVR rate after DEB treatment of 11.3% with a calculated weighted mean follow-up of 18 months.14 In a recent investigational device exemption randomized trial for a paclitaxel-coated balloon in ISR, the rate of TLR at 1 year was 13%.15 However, prior evidence stems from diverse settings, designs, and populations, making it difficult to draw strong conclusions.
The rate of TLR with the previous generation of the Essential Pro DEB in a smaller cohort (n = 31) was 10% at 6 months.16 While this rate may seem higher than that reported in our study, the small number of events (n = 3) makes comparisons challenging.
Limitations
This study has some limitations. First, it was based on a real-world cohort involving different operators from the same center, which does not follow specific protocols. Only a quarter of the patients underwent angioplasty assessment guided by intracoronary imaging. The lack of sponsorship to cover intracoronary imaging costs and its limited use reflects the usual clinical practice of this center. During the performance of this study, few patients with ISR were treated with other DEB catheters due to the lack of specific DEB sizes in stock. Since this situation was rare and was unrelated to clinical or medical coverage characteristics, it is unlikely to introduce significant bias. Since this was a substudy of a larger DEB cohort, some variables specific to ISR, such as the time from prior stent implantation or the type of stent used, were not available.
Second, there were no dedicated follow-up visits for this study. Although most of these patients were followed up by local cardiologists who maintained regular medical records, some required telephone contact for follow-up. Third, angiographic assessment was not duplicated, and no core lab was available. Finally, the number of events was low despite consecutive enrollment from late 2020, impacting the precision of Kaplan-Meier estimates for key clinical outcomes. Some limitations are related to real-world practice settings, which, on the other hand, enhance external validity with less selection bias compared with other more controlled designs.
CONCLUSIONS
Among patients with ISR, the Essential Pro DEB catheter had a high delivery rate and a low incidence of adverse clinical outcomes during follow-up. These results further underscore the safety and efficacy of this new-generation DEB for patients with ISR.
FUNDING
This work received no industry sponsoring or funding.
ETHICAL CONSIDERATIONS
This study was approved by our local institutional review board at the Instituto Cardiovascular de Buenos Aires, and patients provided written informed consent to use their anonymized information for research purposes before their inclusion. Possible sex/gender biases have been considered in the preparation of this paper.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence tool was used in the preparation of this study.
AUTHORS’ CONTRIBUTIONS
L. Padilla conceived and oversaw all the process. F. Liberman, J. Tello, P. Rosas, P. Spaletra, G. Pedernera, P. Mascolo, S. Ordoñez, P. Santilli, and A. Candiello collected data and analyzed coronary angiograms. F. Cura and J. Belardi provided senior advice. P. Lamelas performed the statistical analysis and generated the first draft of the manuscript.
CONFLICTS OF INTEREST
L. Padilla has received proctoring and consulting honoraria from Terumo and Boston Scientific. P. Spaletra has received honoraria from Boston Scientific. F. Cura has received honoraria from Medtronic, Boston Scientific, Terumo, and Meril. P. Lamelas has received proctoring and consulting honoraria from Medtronic, Boston Scientific, Meril, Microport. The remaining authors have no conflicts of interest to declare.
WHAT IS KNOWN ABOUT THE TOPIC?
- Patients with ISR are at high risk of recurrent events and are commonly treated with DEB. New or newer generation DEBs are frequently launched onto the market. It is important to report the real-world safety and efficacy of interventional devices. The Essential Pro is a secondgeneration paclitaxel-eluting balloon. Enhancements of this DEB include improvements in forward pushability, crossover capacity, and drug delivery capabilities.
WHAT DOES THIS STUDY ADD?
- Using this new-generation DEB, all attempts at treating ISR (n = 206) were successful. Intravascular ultrasound was used in 24%. The incidence of adverse events, from the procedure to mid-term follow-up, was infrequent and probably lower than that previously reported. These realworld results emphasize the safety and efficacy of this novel generation DEB for patients with ISR.
REFERENCES
1. Giacoppo D, Saucedo J, Scheller B. Coronary Drug-Coated Balloons for De Novo and In-Stent Restenosis Indications. J Soc Cardiovasc Angiogr Interv. 2023. https://doi.org/10.1016/j.jscai.2023.100625.
2. Pleva L, Kukla P, Hlinomaz O. Treatment of coronary in-stent restenosis:A systematic review. J Geriatr Cardiol. 2018;15:173-184.
3. Giustino G, Colombo A, Camaj A, et al. Coronary In-Stent Restenosis:JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;80:348-372.
4. Indermuehle A, Bahl R, Lansky AJ, et al. Drug-eluting balloon angioplasty for in-stent restenosis:A systematic review and meta-analysis of randomised controlled trials. Heart. 2012;99:327-333.
5. Jeger R V., Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease:Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13:1391-1402.
6. Klein LW, Nathan S, Maehara A, et al. SCAI Expert Consensus Statement on Management of In-Stent Restenosis and Stent Thrombosis. J Soc Cardiovasc Angiogr Interv. 2023;2:100971.
7. Pérez de Prado A, Pérez-Martínez C, Cuellas Ramón C, et al. Safety and Efficacy of Different Paclitaxel-eluting Balloons in a Porcine Model. Rev Esp Cardiol. 2014;67:456-462.
8. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized end point definitions for coronary intervention trials:The academic research consortium 2 consensus document. Circulation. 2018;137:2635-2650.
9. Domienik-Karlowicz J, Kupczyn´ska K, Michalski B, et al. Fourth universal definition of myocardial infarction. Selected messages from the european society of cardiology document and lessons learned from the new guidelines on st-segment elevation myocardial infarction and non-st-segment elevation-acute coronary syndrome. Cardiol J. 2021;28:195-201.
10. Steinberg DH, Pinto Slottow TL, Buch AN, et al. Impact of In-Stent Restenosis on Death and Myocardial Infarction. Am J Cardiol. 2007;100:1109-13.
11. Madhavan M V, Kirtane AJ, Redfors B, et al. Stent-Related Adverse Events >1 Year After Percutaneous Coronary Intervention. J Am Coll Cardiol. 2020;75:590-604.
12. Moussa ID, Mohananey D, Saucedo J, et al. Trends and Outcomes of Restenosis After Coronary Stent Implantation in the United States. J Am Coll Cardiol. 2020;76:1521-1531.
13. Yoshida R, Ishii H, Morishima I, et al. Impact of adjunctive use of guide extension catheter on midterm outcome of drug-coated balloon angioplasty. EuroIntervention. 2019;15:688-691.
14. Cui KY, Lyu SZ, Zhang M, Song XT, Yuan F, Xu F. Drug-Eluting Balloon versus New-Generation Drug-Eluting Stent for the Treatment of In-Stent Restenosis:An Updated Systematic Review and Meta-Analysis. Chin Med J (Engl). 2018;131:600-607.
15. Yeh RW, Shlofmitz R, Moses J, et al. Paclitaxel-Coated Balloon vs Uncoated Balloon for Coronary In-Stent Restenosis:The AGENT IDE Randomized Clinical Trial. JAMA. 2024;331:1015-1024.
16. de la Torre Hernández JM, Garcia Camarero T, Lozano Ruiz-Poveda F, et al. Angiography and Optical Coherence Tomography Assessment of the Drug-Coated Balloon ESSENTIAL for the Treatment of In-Stent Restenosis. Cardiovasc Revasc Med. 2020;21:508-513.
- Percutaneous treatment of the left main coronary artery in older adults. Impact of frailty on mid-term results
- Discordance between fractional flow reserve and nonhyperemic index with a fiber-optic pressure wire. READI EPIC-14
- Plaque modification and impact on the microcirculation territory after drug-coated balloon angioplasty. The PLAMI study design
- On- vs off-hours primary percutaneous coronary intervention: a single-center 5-year experience
Interviews
An interview with Bruno Scheller
aServicio de Cardiología, Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria de La Princesa (IIS-IP), Universidad Autónoma de Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain