Article
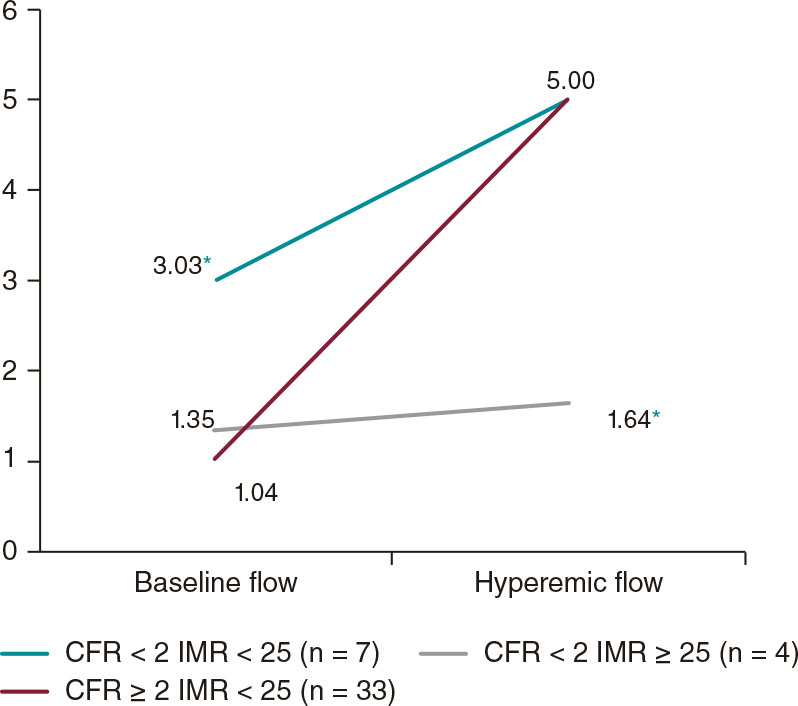
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España
ABSTRACT
Introduction and objectives: No previous studies have established the contemporary use and outcomes of Excimer laser coronary atherectomy (ELCA) in percutaneous coronary intervention (PCI) of severely calcified coronary lesions. The aim of this study was to assess the safety, efficacy, and 1-year outcomes of ELCA in this setting.
Methods: We retrospectively examined the clinical and angiographic characteristics and procedural outcomes of severely calcified lesions treated with ELCA-assisted PCI in our institution between 2016 and 2022.
Results: Seventy-eight consecutive patients (80 procedures) were included (mean age 71.2 ± 8.6 years, 80.5% men). Clinical presentation was stable coronary artery disease in 45 patients (56.2%) and acute coronary syndromes in 33 (43.8%). All the lesions were severely calcified. In addition, 40% were uncrossable lesions, 28.75% were undilatable lesions, 2.5% showed in-stent restenosis, 6.25% showed stent underexpansion, and 7.5% were chronic total occlusions. The combination of ≥ 2 of the above anatomic settings was found in 12.5% of the procedures. The maximum fluence was 73 ± 9.6 mJ/mm2, and the maximum frequency was 72.7 ± 10.4 Hz. The saline flushing technique was initially used in all the procedures, while contrast was used in 2 procedures. The ELCA success and technical success rates were both 91.25%. Adjuvant plaque modification therapies were required in 4 patients. The clinical success rate was 87.5%. ELCA-related complications occurred in 2 procedures (2.5%). After a median follow-up of 15.5 months [IQR, 5.0-29.3], major adverse cardiac events (MACE) (target lesion revascularization, myocardial infarction or cardiac death) occurred in 9 patients (11.25%).
Conclusions: Despite the complexity of PCI in severely calcified lesions, ELCA was effective with a relatively low incidence of ELCA-related complications and MACE during follow-up.
Keywords: Complex PCI. Excimer laser coronary atherectomy. Calcified coronary lesions.
RESUMEN
Introducción y objetivos: El uso contemporáneo y los resultados de la aterectomía coronaria con láser Excímer (ELCA) en el intervencionismo coronario percutáneo (ICP) de lesiones coronarias gravemente calcificadas no están establecidos. El objetivo de este estudio fue evaluar la eficacia, seguridad y resultados a 1 año de ELCA en este escenario.
Métodos: Se revisaron de forma retrospectiva las características clínicas y angiográficas, y los resultados de los procedimientos de revascularización de lesiones gravemente calcificadas tratadas con ICP asistido por ELCA en nuestro centro entre 2016 y 2022.
Resultados: Se incluyeron 78 pacientes consecutivos (80 procedimientos) (edad media 71,2 ± 8,6 años, 80,5% varones). La presentación clínica fue enfermedad arterial coronaria estable en 45 (56,2%) pacientes y síndromes coronarios agudos en 33 (43,8%). Todas las lesiones presentaban calcificación grave. Además, el 40% eran lesiones incruzables, el 28,75% lesiones indilatables, el 2,5% reestenosis intrastent, el 6,25% infraexpansión del stent y el 7,5% oclusiones crónicas. La combinación de ≥ 2 de los escenarios anatómicos anteriores existió en el 12,5% de los procedimientos. La fluencia máxima fue de 73 ± 9,6 mJ/mm2 y la frecuencia máxima de 72,7 ± 10,4 Hz. ELCA con lavado con solución salina se utilizó inicialmente en todos los procedimientos y se utilizó contraste en 2 procedimientos. La tasa de éxito de ELCA y de éxito técnico fueron del 91,25 %. Fueron necesarias terapias adyuvantes de modificación de placa en 4 casos. La tasa de éxito clínico fue del 87,5%. Ocurrieron complicaciones relacionadas con ELCA en 2 (2,5%) procedimientos. Tras una mediana de seguimiento de 15,5 meses (IQR, 5,0-29,3), se produjeron eventos cardiovasculares adversos mayores (MACE) (nueva revascularización de la lesión diana, infarto de miocardio o muerte cardiaca) en 9 pacientes (11,25%).
Conclusiones: A pesar de la complejidad de la ICP en lesiones gravemente calcificadas, ELCA demostró ser efectivo con una incidencia relativamente baja de complicaciones relacionadas con ELCA y MACE en el seguimiento.
Palabras clave: ICP compleja. Láser coronario. Lesiones coronarias calcificadas.
Abbreviations
CTO: chronic total occlusion. ELCA: excimer laser coronary atherectomy. ISR: in-stent restenosis. MACE: major adverse cardiovascular events. PCI: percutaneous coronary intervention. RA: rotational atherectomy.
INTRODUCTION
Moderate or severe coronary artery calcification is relatively common in patients undergoing percutaneous coronary interventions (PCI).1 This is closely related to advancing age and the high prevalence of comorbidities such as diabetes or chronic kidney disease. Coronary artery calcification is associated with a lower rate of successful PCI and complete revascularization, increased procedural-related complications, and a higher rate of major adverse cardiovascular events (MACE).2
Despite the availability of several plaque modification techniques, severely calcified lesions continue to pose a challenge to the successful performance of PCI.
Excimer laser coronary atherectomy (ELCA) is a plaque modification technique that has proved to be useful in various scenarios such as balloon failure (uncrossable or undilatable lesions), chronic total occlusions (CTO), stent underexpansion, in-stent restenosis (ISR), and thrombotic lesions. In recent years, incremental operator experience along with the standardization of the laser technique, has expanded its indications and decreased its complication rates.3,4
However, its effectiveness in calcified lesions is controversial. On one hand, some ELCA series have described a relationship between severe calcification and laser failure.5-8 On the other hand, moderate-to-severe calcification is found in more than 60% of cases in some ELCA series with a high success rate,9 suggesting that it could be useful in this setting.10
Due to the lack of evidence in this specific scenario, the aim of our study was to assess the safety and efficacy of ELCA in severely calcified coronary lesions, as well as the mid-term follow-up outcomes in a single center registry.
METHODS
Patient population
This single center retrospective observational study included all consecutive patients undergoing ELCA-assisted PCI for severely calcified lesions. From March 2016 to August 2022.
We excluded procedures using ELCA for any indication other than severe calcification. In all patients, PCI was indicated based on the presence of symptoms consistent with angina, demonstrated ischemia, or both. The study followed the international recommendations of clinical investigation (Declaration of Helsinki). All participants gave written informed consent and approval was obtained from the ethics committee of the center. The study took into consideration sex and gender variables according to SAGER guidelines. Patients were followed up in cardiology clinics at their referral center 3 to 6 months after the procedure, and thereafter at time intervals established at the discretion of their treating physician.
We analyzed data on clinical and angiographic characteristics, technical aspects of the procedure, and cardiovascular events during hospitalization and after discharge.
Procedure
All procedures were carried out by 5 different operators experienced in the use of ELCA. The decision to use ELCA was based on the presence of angiographically severe calcification.
Radial access was use by default. All cases were performed with the CVX-300 Excimer Laser System (Philips, Netherlands) using the 0.9 mm or 1.4 mm catheters. Saline infusion technique was used by default from the beginning, with fluence (mJ/mm2), frequency or repetition rate (Hertz), and the possibility to use ELCA without saline infusion or even with contrast left to the operator’s discretion. Additional dilatation with noncompliant balloons was performed in all procedures. Patients in which another plaque modification technique was used in combination with ELCA were included. All PCIs were performed following current recommendations.11
Definitions
Severely calcified lesions were angiographically defined as radiopacities observed on fluoroscopy without cardiac motion before contrast injection compromising 1 or both sides of the lumen.12 Balloon-uncrossable lesions were defined as lesions that could not be crossed with the lowest-profile balloon available or a microcatheter despite successful advancement of the guidewire distal to the lesion, having good guide catheter support with a guide extension catheter when required. Balloon-undilatable lesions were defined as those lesions in which a noncompliant balloon (diameter 1:1 according to the vessel diameter) failed to achieve adequate expansion. Anterograde flow was assessed by the Thrombolysis In Myocardial Infarction (TIMI) scale.
ELCA success was defined as the ability to cross the lesion completely with the laser catheter or, if crossing was not feasible, to allow the subsequent passage and expansion of a balloon sized 1:1 with the vessel diameter, after laser application. Technical success was defined as residual stenosis < 30% and anterograde TIMI 3 flow in the target vessel. Clinical success was defined as technical success and the absence of MACE during the current hospitalization (target lesion revascularization, procedure-related myocardial infarction [MI], or cardiovascular death). Procedural-related complications included coronary artery perforation leading to cardiac tamponade and requiring pericardial drainage, flow-limiting dissection, no-reflow, hemodynamic instability, MI type 4a according to the fourth universal definition of MI,13 ventricular arrhythmias, and major bleeding (bleeding requiring transfusion and/or surgical or percutaneous intervention). MACE occurring during follow-up were defined as a composite of target lesion revascularization, MI, or cardiac death.
Statistical analysis and data collection
All data were collected through the patients’ electronic medical records and were introduced in a local database. Angiograms were evaluated using local quantitative coronary analysis software and visual operators’ assessment. Categorical variables are reported as absolute values and percentages. Continuous variables are presented as the mean ± standard deviation (SD) or median (interquartile range [IQR] 25-75), depending on their normal or nonnormal distribution. All analyses were performed with StatIC 16.1 statistical software package.
RESULTS
Clinical characteristics
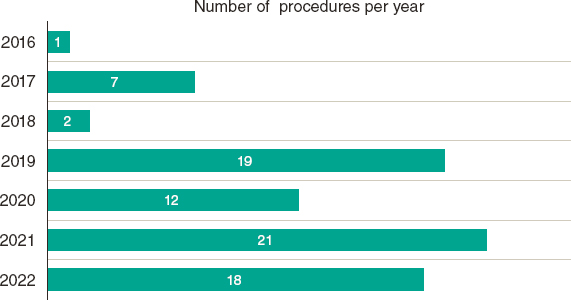
During the study period, a total of 78 patients with severely calcified coronary lesions underwent 80 ELCA-assisted PCIs and were included in the analysis. Patients undergoing ELCA for an indication other than severe calcification were excluded from the analysis. The distribution of the number of procedures per year, between March 2016 and May 2022, is shown in figure 1. A flowchart of patients in the present study is summarized in figure 2. Mean age was 71.2 ± 8.6 years, 62 (80.5%) were men, and there was a high prevalence of cardiovascular risk factors. Mean left ventricle ejection fraction was 52.9% ± 12.5%. Thirty-nine patients (50%) had a previous PCI. Clinical presentation was stable coronary artery disease in 45 procedures (56.2%), non–ST-segment elevation MI (NSTEMI) in 28 (35%), and ST-segment elevation MI (STEMI) in 7 (8.8%). Baseline clinical characteristics are summarized in table 1.
Figure 1. Distribution of the number of procedures per year (March 2016-May 2022).
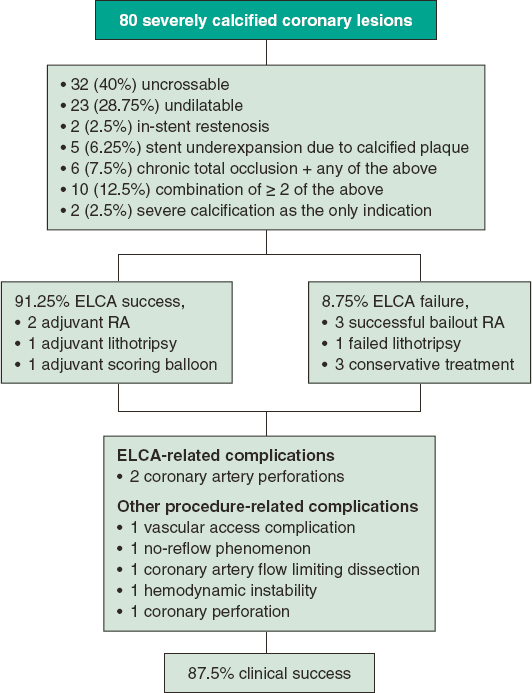
Figure 2. Flowchart of patients in the present study. ELCA, excimer laser coronary atherectomy; PCI, percutaneous coronary intervention; RA, rotational atherectomy.
Table 1. Baseline clinical characteristics
| Age | 71.2 ± 8.6 |
| Male sex | 62 (80.5%) |
| Body mass index (kg/m2) | 28.7 ± 4.2 |
| Hypertension | 70 (89.7%) |
| Dyslipidemia | 61 (78.2%) |
| Diabetes mellitus | 46 (59.0%) |
| Current smoker | 19 (24.4%) |
| Prior PCI | 39 (50.0%) |
| Prior CABG | 8 (10.3%) |
| Hb (g/dL) | 13.5 ± 5.3 |
| Serum creatinine (mg/dL) | 1.42 ± 1.8 |
| Ejection fraction (%) | 52.9% ± 12.5 |
| Clinical presentation (n = 98) | |
| Stable coronary artery disease | 45 (56.2%) |
| NSTEMI | 28 (35.0%) |
| STEMI | 7 (8.8%) |
|
CABG, coronary artery bypass graft surgery; NSTEMI, non–ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction. Data are expressed as no. (%) or mean ± standard deviation. |
|
Angiographic characteristics
Severe multivessel disease was present in 56 patients (71.8%). The most common target vessel was the left anterior descending artery (38.75%). In 7 procedures (8.75%), more than 1 target vessel were identified. The anatomical settings in the target vessel included uncrossable lesions in 32 (40%), undilatable lesions in 23 (28.75%), ISR in 2 (2.5%), and stent underexpansion related to calcified plaque in 5 (6.25%). In 6 (7.5%) procedures, the main indication for ELCA was CTO combined with any of the previous settings. In 10 procedures (12.5%), the indication for ELCA resulted from the combination of 2 or more of the above. ELCA was used with the sole indication of severely calcified lesion, not included in any of the previous anatomical settings, in 2 procedures (2.5%).
Procedural characteristics
The radial approach was performed in 44 (55%) cases. There was no need for access conversion when the radial approach was attempted.
Dual antiplatelet treatment consisted of pretreatment with aspirin and oral P2Y12 receptor blockers in 58 patients (72.5%). Selection of P2Y12 inhibitor was left to the physician’s discretion. Cangrelor was used in the patients without prior dual antiplatelet treatment. After the procedure and during follow-up, dual antiplatelet treatment was prescribed as follows: in stable coronary artery disease (n = 45) clopidogrel was used in 21 patients, ticagrelor in 10 and prasugrel in 3 patients. In acute settings (n = 35), ticagrelor was administered in 16 patients, prasugrel in 10, and clopidogrel in 7. GPIIB/IIIA inhibitors were used in 6 procedures (7.5%) (tirofiban in all cases).
Intracoronary imaging was used in 58 procedures (72.5%). Optical coherence tomography (OCT) was used in 48 procedures (60%) and intravascular ultrasound in 10 (12.5%).
Circulatory support with intra-aortic balloon counterpulsation was required in only 1 patient in the context of left-main revascularization.
Regarding the ELCA technique, most lesions were treated with 0.9 mm laser catheters (97.5%). In 2 patients, larger catheters (1.4 mm) were used (1 case of ISR in the left anterior descending artery and 1 calcified lesion in a saphenous vein graft). Flushing saline was used in all the procedures, and contrast was required in 2 procedures (figure 3). Maximum fluence was 73 ± 9.6 mJ/mm2 and the maximum frequency 72.7 ± 10.4 Hz. The highest fluence of 80 mJ/mm2 was required in 48 (60%) procedures and the highest frequency of 80 Hz in 48 (60%). A mean of 5103 ± 3120 pulses was delivered, and the median lasing time was 62 seconds [IQR 40-91].
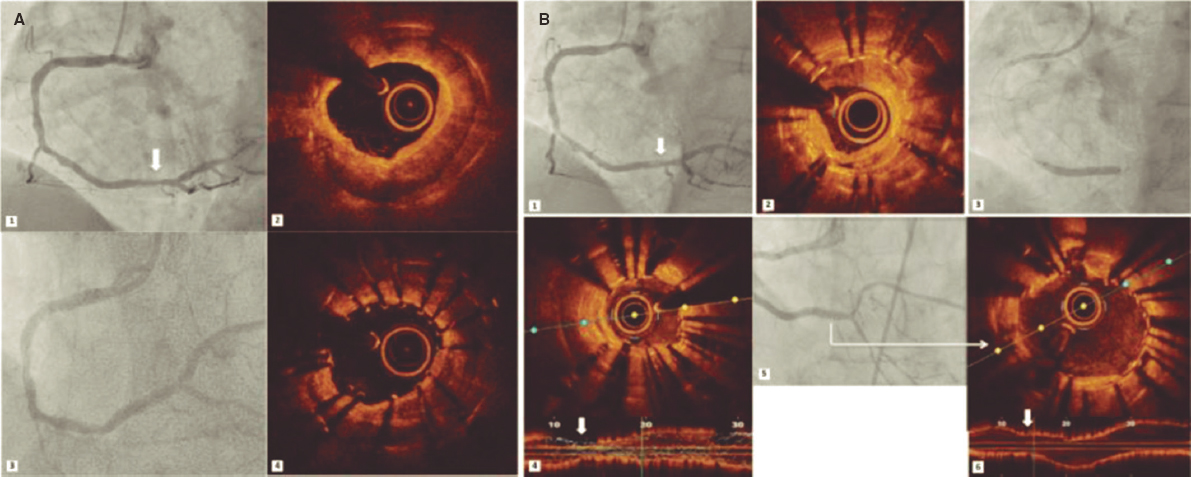
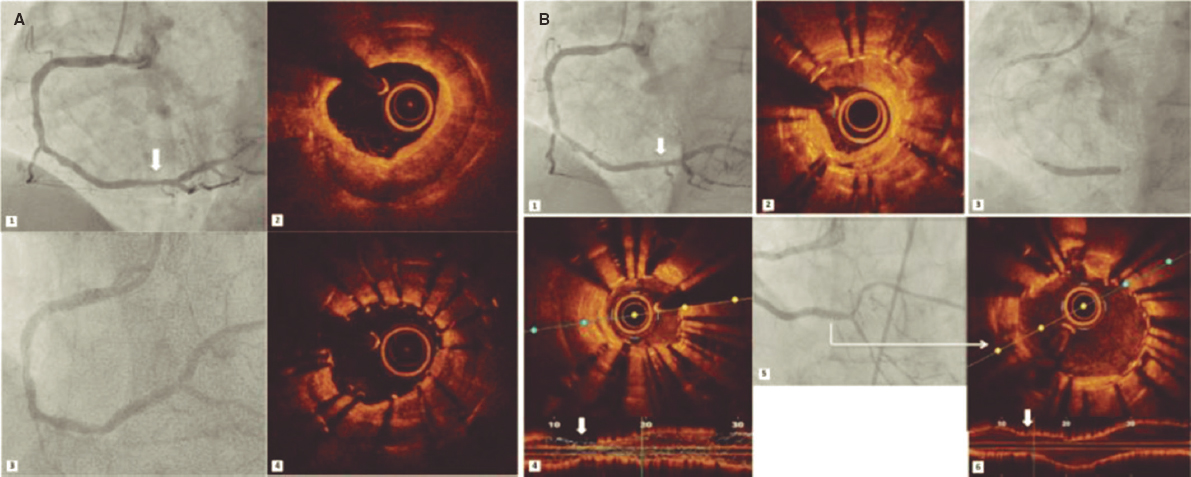
Figure 3. In-stent restenosis and stent underexpansion treated by excimer laser coronary atherectomy (ELCA). Severe in-stent restenosis (ISR) (A1) (arrow) of drug-eluting stent previously implanted in the right coronary artery. Optical coherence tomography (OCT) revealed calcified neoatherosclerosis with a minimum luminal area (MLA) of 1.25 mm2 (A2). An everolimus-eluting stent (2.75 × 20 mm) was implanted, and despite postdilatation with a 3-mm noncompliant (NC) balloon (A3), subsequent OCT confirmed stent underexpansion (MLA: 2.1 mm2) (A4). Sixteen months later, critical ISR of the previous stent (B1) (arrow) was noted with heterogeneous neointimal proliferation (B2). Laser atherectomy was performed, followed by dilation with 3- and 3.5-mm NC balloons up to 24 atm, and a 3-mm sirolimus-eluting stent was implanted with acceptable angiographic expansion (B3) but underexpansion on OCT (MLA: 1.5 mm2) (B4) (arrow). Laser application with contrast injection was repeated and was dilated with a 4 mm NC balloon, achieving adequate stent expansion (MLA: 4.5 mm2) (B5, B6) (arrow).
At least 1 new-generation drug-eluting stent was implanted in 70 procedures (87.5%). In the remaining procedures, stents were not delivered because of the presence of previous stents (6 ISR and 2 cases of stent underexpansion), which were treated with noncompliant and/or drug-eluting balloons, or due to ELCA failure (2 cases).
Angiographic and procedural characteristics and procedural strategy data are summarized in table 2.
Table 2. Angiographic and procedural characteristics
| Angiographic characteristics | |
|---|---|
| Target vessel | |
| Left anterior descending coronary artery | 31 (38.75%) |
| Right coronary artery | 28 (35.0%) |
| Circumflex artery | 10 (12.5%) |
| Left main coronary artery | 4 (5.0%) |
| Multivessel disease | 56 (71.8%) |
| Indication for ELCA | |
| Balloon-uncrossable lesion | 32 (40%) |
| Balloon-undilatable lesion | 23 (28.75%) |
| In-stent restenosis | 2 (2.5%) |
| Stent Underexpansion | 5 (6.25) |
| Chronic total occlusion | 6 (7.75%) |
| Combination of > 2 of the above | 10 (12.5%) |
| Severe calcification as sole indication | 2 (2.5%) |
| Bifurcation | 14 (17.7%) |
| Aorto-ostial | 2 (2.5%) |
| Procedural characteristics | |
| Access site | |
| Radial | 44 (55.0%) |
| Femoral | 33 (41.2%) |
| Femoral-radial | 3 (3.8%) |
| Guiding catheter French | |
| 6-Fr | 40 (50.0%) |
| 7-Fr | 34 (42.5%) |
| Intracoronary imaging | 58 (72.5%) |
| OCT | 48 (60.0%) |
| IVUS | 10 (12.5%) |
| Laser catheter | |
| 1.4 mm rapid-exchange catheter | 2 (2.5%) |
| 0.9 mm rapid-exchange catheter | 78 (97.5%) |
| Maximum fluence (mJ/mm2) | 72.97 ± 9.6 |
| Maximum frequency (Hz) | 72.7 ±10.4 |
| Number of pulses | 5 103 ± 3 120 |
| Total lasing time (sec) | 62 [40-91] |
| Contrast volume (mL) | 211 ± 68.0 |
| Fluoroscopy time (min) | 30 [22-39] |
| Radiation dose (Gy/cm2) | 103 [79-185] |
| Procedural time (min) | 72 [55-100] |
| Stent implantation | 70 [87.5%] |
| Stent diameter (mm | 3.04 ± 0.50 |
| Stents per procedure | 1.8 ± 1.14 |
| Total stent length (mm) | 43.7 ± 25.7 |
| Left ventricle assist device used | 1 (1.25%) |
| Timing of PCI (n = 98) | |
| Ad hoc | 22 (27.5%) |
| Deferred | 58 (72.5%) |
|
ELCA, excimer laser coronary atherectomy; OCT, optical coherence tomography; IVUS, intravascular ultrasound; PCI, percutaneous coronary intervention. Data are expressed as no. (%), mean ± standard deviation or median [interquartile range]. |
|
Procedural outcomes
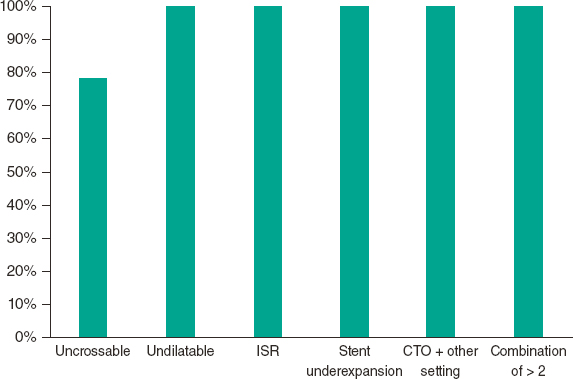
The ELCA success rate was 91.25%. The success rate was 78.1% in uncrossable lesions and 100% in the other anatomical settings (P < .001). The ELCA success rate in the different anatomical settings is shown in figure 4.
Figure 4. ELCA success rate in the different anatomical settings. ISR, in-stent restenosis, CTO, chronic total occlusion.
Among intracoronary imaging-guided procedures, the ELCA success rate was 98.3%, and dropped to 72.7% in non-coronary imaging-guided PCI (P < .001). Final stent expansion was analyzed with intracoronary imaging in 32 procedures. The median stent expansion was 80.3% [IQR, 68.2%-95.2%].
Despite ELCA success, adjuvant plaque modification therapies (other than noncompliant [NC] balloon inflation after ELCA) were used in 4 procedures, including rotational atherectomy (RA) in 2 procedures, lithotripsy in 1 procedure and scoring balloon in 1 procedure. The procedures in which ELCA allowed subsequent successful RA (RASER technique14) or successful lithotripsy (ELCA-tripsy technique15) were considered ELCA success.
In 7 procedures (8.75%), ELCA failed. In 2 of them, RA was successfully performed. In 1 procedure, intravascular lithotripsy was attempted, but failed. In 1 case, the procedure was prematurely interrupted at the request of the patient. In the remaining 2 patients, no bailout therapy was attempted, and they were managed conservatively. Cases in which ELCA did not facilitate the passage of RA or intravascular lithotripsy were not classified as RASER or ELCA-tripsy techniques. The overall technical success rate was 91.25%.
In-hospital and follow-up outcomes
ELCA-related complications occurred in 2 procedures (2.5%) due to coronary artery perforation after ELCA application, with immediate sealing after stent implantation (although pericardiocentesis was necessary in 2 of them). A third perforation was observed, not immediately after ELCA application, but after dilatation with NC balloons. In 2 of the perforations, the target lesion was a severely calcified and undilatable lesion located in the left anterior descending artery. The third perforation was observed in an uncrossable lesion at the right coronary artery. In all of them, the 0.9 mm catheter was used, and ELCA was applied with maximum fluency and repetition rate during saline infusion. Intracoronary imaging prior to ELCA application was not performed in any of these patients: the OCT catheter could not cross the lesion in 2 of them and crossing was not attempted in the third. After the application of coronary laser and stent implantation, OCT was performed in 2 of the procedures, which confirmed the good final result.
Other procedural complications not related to ELCA occurred in 4 patients. One patient developed a vascular access complication with retroperitoneal hemorrhage and severe bleeding requiring transfusion and transarterial embolization of a deep femoral artery branch, although his clinical course was favorable. One patient with severe aortic stenosis and impaired left ventricular function showed hemodynamic instability requiring support with inotropes and orotracheal intubation. In 1 patient, no-reflow phenomenon occurred after stent implantation but resolved after intracoronary adenosine infusion.
In the remaining patient, coronary dissection occurred during the guidewire advancement before ELCA application and was complicated with occlusive intracoronary hematoma, which resolved after emergent PCI with successful revascularization. No patient died during the procedure. Three patients died during admission despite successful revascularization due to cardiac causes not related to the procedure (mostly advanced heart failure) and 1 patient died from respiratory sepsis. There were no other in-hospital complications. Overall, the clinical success rate was 87.5%.
After a median follow-up of 15.5 months [IQR, 5.02-29.3], MACE occurred in 9 patients (11.25%). Target lesion revascularization occurred in 7 patients (8.9%), in all patients due to ISR. The median time to target lesion revascularization among patients with successful revascularization was 11.4 [IQR, 8.1-22.6] months. Cardiorespiratory arrest secondary to acute stent thrombosis occurred in 1 patient with successful revascularization, whose family reported poor antiplatelet therapy adherence. One patient died from advanced heart failure after 3 years of follow-up, despite successful revascularization. Three patients died from noncardiac causes.
The procedural outcomes, clinical outcomes, and major complications are summarized in table 3. No significant differences were observed in the results between male and female patients.
Table 3. Procedural and clinical outcomes
| Procedural and clinical success | n (%) |
|---|---|
| ELCA success | 73 (91.25%) |
| Balloon-uncrossable lesion | 25 (78.13%) |
| Balloon-undilatable lesion | 23 (100%) |
| In-stent restenosis | 2 (100%) |
| Stent underexpansion | 5 (100%) |
| Chronic total occlusion | 6 (100%) |
| Combination of > 2 of the above | 10 (100%) |
| Severe calcification as sole indication | 2 (100%) |
| Technical success | 73 (91.25%) |
| Clinical success | 70 (87.5%) |
| Procedural complications | |
| ELCA-related complications | |
| Coronary artery perforation | 2 (2.5%) |
| Complications not related to ELCA | |
| Vascular access complication with major bleeding | 1 (1.25%) |
| Coronary perforation | 1 (1.25%) |
| Flow-limiting dissection | 1 (1.25%) |
| Hemodynamic instability | 1 (1.25%) |
| No-reflow | 1 (1.25%) |
| Ventricular arrhythmia | 0 (0%) |
| In-hospital MACE | |
| Recurrent angina requiring TLR | 0 (0%) |
| Procedure-related myocardial infarction | 1 (1.25%) |
| New-onset heart failure | 0 (0%) |
| Stroke | 0 (0%) |
| Cardiovascular death | 3 (3.75%) |
| All-cause death | 4 (5.0%) |
| MACE after discharge | |
| TLR | 7 (8.75%) |
| MI due to stent thrombosis | 1 (1.25%) |
| Death from cardiovascular causes | 2 (2.5%) |
| Non-cardiovascular related death | 3 (3.75%) |
|
ELCA, excimer laser coronary atherectomy; MACE, major adverse cardiovascular events; MI, myocardial infarction; TLR, target lesion revascularization. |
|
DISCUSSION
The main findings of our study are as follows: a) ELCA was associated with a high rate of technical success in severely calcified coronary lesions, whether isolated or combined with other plaque modification techniques, with an acceptable ELCA-related complications rate. b) The success rate was higher in undilatable than in uncrossable lesions and was 100% in peri-stent lesions (stent underexpansion or ISR).
As described in previous series, calcified lesions are associated with higher rates of PCI failure, complications, morbidity, and mortality.2,16 Although ELCA is known to have no direct effect on calcium, calcified atheromatous plaques have a mixed composition, including lipids, collagen, and other protein fibers.1,17 The interaction of ELCA with these components, due to its photochemical, photothermal and photokinetic properties, modifies the plaque structure, thus facilitating angioplasty in lesions with severe calcification.17 Moreover, in some cases, as occurs in our series, ELCA is complementary to other plaque modification techniques, allowing the passage of the microcatheter to introduce specific atherectomy guidewires, or even to allow the passage of the lithotripsy balloon.14,15 The RASER technique was used in 2 patients and the ELCA-tripsy technique in another patient with technical success in all 3 of them.
There is a lack of contemporary specific series on the use of ELCA in lesions with severe calcification, and data available in the medical literature are contradictory. Bilodeau et al.18 reported high procedural (93%) and clinical (86%) success in a series of 95 patients with complex coronary lesions, of which 57 had significant calcification. The Laser Veterans Affairs (LAVA) Multicenter Registry7 evaluated the use of ELCA in 131 target complex coronary lesions, of which 62% were moderately or severely calcified lesions, globally reporting 90% technical and 88.8% procedural success rate, which is consistent with our results. In the LEONARDO study,19 in which 75% of lesions were calcified, high laser energy levels were shown to be safe and effective (success rate 93.7%). In our series, the highest fluence and frequency were required in 60% of the procedures, with a similar success rate.
Nowadays, the main indication of ELCA is treatment of uncrossable and undilatable lesions. In uncrossable lesions, the laser catheter can be advanced over any 0.014¨angioplasty guidewire that crosses the lesion, unlike other plaque modification techniques. In a multicenter US registry, the success rate for laser-assisted PCI in uncrossable balloon CTO was 95%, which was higher than that for RA (89%) in this setting.20 In a retrospective study by Karacsonyi et al.,21 laser use in balloon-uncrossable and balloon-undilatable CTO was associated with higher technical (91.5% vs 83.1%) and procedural (88.9% vs 81.6%) success rates compared with cases without the use of laser. Ojeda et al.9 conducted a multicenter registry of 126 uncrossable lesions and reported ELCA success of 81.8%. In that registry, severe calcification was independently associated with ELCA failure, a finding already described in a previous study.22 In our series (with severe calcification in 100% of patients), the overall ELCA success rate was 91.25%, but the ELCA success in uncrossable lesions was lower than in undilatable lesions (78.1% vs 100%) and similar to that in the series by Ojeda et al.9 The lower success rate in uncrossable and severely calcified lesions can probably be explained by the different plaque composition and calcium distribution. Furthermore, the higher rate of use of intracoronary imaging could also be associated with better results (72.5% in our series compared with 22.5% reported by Ojeda et al.9). Of note, an ELCA success of 78.1% in uncrossable lesions with severe calcification could be a reasonable result, considering that, if even a microcatheter cannot cross the lesion, ELCA may be the only alternative for revascularization.
In other scenarios, the ELCA success rate of our series was high and similar to that of other series. An ELCA success rate of 86% to 93% has been reported in CTOs.8,23 RA in CTO has been associated with similar success rates (89%-95.6%)24,25 but with a high rate of slow/no flow phenomena.24 In patients with stent underexpansion and ISR, ELCA is feasible and effective,26,27 with 100% ELCA success in our series.
Intravascular imaging is useful to guide calcified coronary stenosis PCI.28,29 Contemporary rates of intravascular imaging for complex PCI remain low.30 In our study, intracoronary imaging was used in 72 procedures (73.4%), and intracoronary imaging-guided procedures resulted in a higher success rate. Its lower use in uncrossable lesions can probably be explained by the fact that the intravascular ultrasound/OCT catheter cannot cross the lesion, rather than necessarily being the reason for the lower success rate in this setting.
Limitations
Our study has some limitations. First, it is an observational study with a small sample size. However, to the best of our knowledge, our study represents the largest series of ELCA specifically performed in severely calcified lesions in contemporary PCI. Second, the severity of lesion calcification was initially assessed by conventional coronary angiography, which has only low to moderate sensitivity compared with intravascular ultrasound or OCT. In addition, sometimes the calcium observed by conventional angiogra- phy is adventitious, thus not affecting balloon dilation or stent expansion with conventional techniques. However, the use of intracoronary imaging techniques was higher than in previous series and confirmed the severity of calcification in all patients. In addition, a significant number of cases consisted of uncrossable lesions, limiting the use of intracoronary imaging to define the calcification from the beginning of the procedure. Finally, the operators involved in this study were experienced ELCA operators. This may limit the generalizability of our results since ELCA is not available in most centers and requires a learning curve.
CONCLUSIONS
ELCA is a useful tool in severe calcification lesions, with a high success rate, especially in the setting of undilatable or peri-stent lesions. The technique is also reasonably safe, given that it is used in highly complex procedures. Future randomized studies will shed light on its role in the management of severe calcified coronary lesions.
FUNDING
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
ETHICAL CONSIDERATIONS
All patients signed an informed consent form and approval was obtained from the ethics committee of the center. The study has taken into consideration sex and gender variables according to SAGER guidelines.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence tool has been used during the preparation of this work.
AUTHORS’ CONTRIBUTIONS
A. Jurado-Román conceived and designed the study. L. Cobarro and A. Jurado-Román performed the analysis and wrote the initial draft. L. Cobarro, A. Jurado-Román, D. Tébar-Márquez, S. Vera-Vera, A. García-Escobar, C. Ugueto, C. Contreras, B. Rivero, S. Jiménez-Valero, G. Galeote, and R. Moreno collected the data and reviewed the final version of the manuscript.
CONFLICTS OF INTEREST
R. Moreno is associate editor of REC: Interventional Cardiology; the editorial procedure established in the journal has been followed to ensure impartial handling of the manuscript.
A. Jurado-Román is proctor of Philips-Biomenco, Boston Scientific, CSI-World Medica and Medtronic Inc and has received speaker fees from Boston Scientific, Abbott Vascular, World Medica, Biotronik, Philips-Biomenco, and Inari. R. Moreno has received speaker fees from Medtronic Inc, Boston Scientific, Abbott vascular, Biosensors, Biotronik, Edwards Lifesciences, AMGEN, AstraZeneca, and Daiichi Sankyo New Vascular Therapies and Biosensors.
WHAT IS KNOWN ABOUT THE TOPIC?
- Excimer laser coronary atherectomy (ELCA) is a plaque modification technique that has proved to be useful in several scenarios, such as balloon failure (uncrossable or undilatable lesions), chronic total occlusions (CTO), stent underexpansion, in-stent restenosis (ISR) and thrombotic lesions.
- In recent years, incremental operator experience along with the standardization of laser technique has expanded its indications and decreased its complication rates.
- The effectiveness of ELCA in calcified lesions is controversial. On one hand, some ELCA series have described a relationship between severe calcification and laser failure. In contrast, moderate-to-severe calcification is found in more than 60% of cases in some ELCA series with a high success rate, indicating that this technique could be useful in this setting.
- Due to the lack of evidence in this specific scenario, our study aimed to assess the contemporary safety and efficacy of ELCA in severely calcified coronary lesions.
WHAT DOES THIS STUDY ADD?
- ELCA is associated with a high rate of technical success in severely calcified coronary lesions, whether isolated or combined with other plaque modification techniques, with an acceptable ELCA-related complications rate.
- The success rate is higher in undilatable than in uncrossable lesions and was 100% in peri-stent lesions (stent underexpansion or restenosis). However, in uncrossable lesions, ELCA may be the only alternative for percutaneous revascularization.
- Clinical results after a median follow-up of 15.5 months were favorable, taking into account the complexity of this scenario.
REFERENCES
1. Mori H, Torii S, Kutyna M, Sakamoto A, Finn AV, Virmani R. Coronary Artery Calcification and its Progression:What Does it Really Mean?JACC Cardiovasc Imaging. 2018;11:127-142.
2. Huisman J, van der Heijden LC, Kok MM, et al. Impact of severe lesion calcification on clinical outcome of patients with stable angina, treated with newer generation permanent polymer-coated drug-eluting stents:A patient-level pooled analysis from TWENTE and DUTCH PEERS (TWENTE II). Am Heart J. 2016;175:121-129.
3. Jawad-Ul-Qamar M, Sharma H, Vetrugno V, et al. Contemporary use of excimer laser in percutaneous coronary intervention with indications, procedural characteristics, complications and outcomes in a university teaching hospital. Open Heart. 2021;8:e001522.
4. Sintek M, Coverstone E, Bach R, et al. Excimer Laser Coronary Angioplasty in Coronary Lesions:Use and Safety From the NCDR/CATH PCI Registry. Circ Cardiovasc Interv. 2021;14:e010061.
5. Stone GW, de Marchena E, Dageforde D, et al. Prospective, Randomized, Multicenter Comparison of Laser-Facilitated Balloon Angioplasty Versus Stand-Alone Balloon Angioplasty in Patients With Obstructive Coronary Artery Disease fn1fn1Funding for this study was provided in part by Eclipse Surgical Technologies, Inc., Sunnyvale, California. J Am Coll Cardiol. 1997;30:1714-1721.
6. Ocaranza-Sánchez R, Abellás-Sequeiros RA, Galvão-Braga C, Trillo-Nouche R, González-Juanatey JR. Uso de aterectomía coronaria con LASER Excimer como terapia coadyuvante en intervencionismo coronario percutáneo. Rev Esp Cardiol.2016;69:867-868.
7. Karacsonyi J, Armstrong EJ, Truong HTD, et al. Contemporary Use of Laser During Percutaneous Coronary Interventions:Insights from the Laser Veterans Affairs (LAVA) Multicenter Registry. J Invasive Cardiol. 2018;30:195-201.
8. Mohandes M, Rojas S, Moreno C, Fernández F, Fuertes M, Guarinos J. Excimer Laser in Percutaneous Coronary Intervention of Device Uncrossable Chronic Total and Functional Occlusions. Cardiovasc Revasc Med. 2020;21:657-660.
9. Ojeda S, Azzalini L, Suárez de Lezo J, et al. Excimer laser coronary atherectomy for uncrossable coronary lesions. A multicenter registry. Catheter Cardiovasc Interv. 2021;98:1241-1249.
10. Jurado-Román A, Gómez-Menchero A, Gonzalo N, et al. Plaque modification techniques to treat calcified coronary lesions. Position paper from the ACI-SEC. REC Interv Cardiol. 2023;5:46-61
11. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
12. Mintz GS, Popma JJ, Pichard AD, et al. Patterns of Calcification in Coronary Artery Disease. Circulation. 1995;91:1959-1965.
13. Thygesen K. 'Ten Commandments'for the Fourth Universal Definition of Myocardial Infarction 2018. Eur Heart J. 2019;40:226-226.
14. Egred M. RASER angioplasty. Catheter Cardiovasc Interv. 2012;79:1009-1012.
15. Jurado-Román A, García A, Moreno R. ELCA-Tripsy:Combination of Laser and Lithotripsy for Severely Calcified Lesions. J Invasive Cardiol.2021;33:E754-E755.
16. Copeland-Halperin RS, Baber U, Aquino M, et al. Prevalence, correlates, and impact of coronary calcification on adverse events following PCI with newer-generation DES:Findings from a large multiethnic registry. Catheter Cardiovasc Interv. 2018;91:859-866.
17. Tsutsui RS, Sammour Y, Kalra A, et al. Excimer Laser Atherectomy in Percutaneous Coronary Intervention:A Contemporary Review. Cardiovasc Revasc Med. 2021;25:75-85.
18. Bilodeau L, Fretz EB, Taeymans Y, Koolen J, Taylor K, Hilton DJ. Novel use of a high-energy excimer laser catheter for calcified and complex coronary artery lesions. Catheter Cardiovasc Interv. 2004;62:155-161.
19. Ambrosini V, Sorropago G, Laurenzano E, et al. Early outcome of high energy Laser (Excimer) facilitated coronary angioplasty ON hARD and complex calcified and balloOn-resistant coronary lesions:LEONARDO Study. Cardiovasc Revasc Med. 2015;16:141-146.
20. Karacsonyi J, Karmpaliotis D, Alaswad K, et al. Prevalence, indications and management of balloon uncrossable chronic total occlusions:Insights from a contemporary multicenter US registry. Catheter Cardiovasc Interv. 2017;90:12-20.
21. Karacsonyi J, Alaswad K, Choi JW, et al. Laser for balloon uncrossable and undilatable chronic total occlusion interventions. Int J Cardiol. 2021;336:33-37.
22. Bittl JA. Clinical results with excimer laser coronary angioplasty. Semin Interv Cardiol. 1996;1:129-134.
23. Rawlins J, Din JN, Suneel T, O´Kane P. Coronary Intervention with the Excimer Laser:Review of the Technology and Outcome Data. Interv Cardiol. 2016;1:27-32.
24. Azzalini L, Dautov R, Ojeda S, et al. Long-term outcomes of rotational atherectomy for the percutaneous treatment of chronic total occlusions. Catheter Cardiovasc Interv. 2017;89:820-828.
25. Pagnotta P, Briguori C, Mango R, et al. Rotational atherectomy in resistant chronic total occlusions. Catheter Cardiovasc Interv. 2010;76:366-371.
26. Latib A, Takagi K, Chizzola G, et al. Excimer Laser LEsion Modification to Expand Non-dilatable sTents:The ELLEMENT Registry. Cardiovasc Revasc Med. 2014;15:8-12.
27. Lee C, Shlofmitz R, Song L, et al. The effectiveness of excimer laser angioplasty to treat coronary in-stent restenosis with peri-stent calcium as assessed by optical coherence tomography. J Am Coll Cardiol.2017;69:1086-1086.
28. Fujino A, Mintz GS, Matsumura M, et al. A new optical coherence tomography-based calcium scoring system to predict stent underexpansion. EuroIntervention. 2018;13:e2182-e2189.
29. Zhang M, Matsumura M, Usui E, et al. Intravascular Ultrasound–Derived Calcium Score to Predict Stent Expansion in Severely Calcified Lesions. Circ Cardiovasc Interv. 2021;14:e010296.
30. Hannan EL, Zhong Y, Reddy P, et al. Percutaneous Coronary Intervention With and Without Intravascular Ultrasound for Patients With Complex Lesions:Utilization, Mortality, and Target Vessel Revascularization. Circ Cardiovasc Interv. 2022;15:e011687.
ABSTRACT
Introduction and objectives: Contrast-induced-acute kidney injury (CI-AKI) is a potential complication of angiographic procedures. The DyeVert Contrast Reduction system (Osprey Medical, United States) is a device to reduce the concentration of contrast medium (CM) in the kidneys by decreasing the amount of CM delivered to patients. Unlike manual systems, few data are available on the DyeVert Power XT system, which is used in conjunction with automated contrast injection. The main aim of our study was to evaluate its effectiveness during percutaneous coronary interventions (PCI).
Methods: Between 2020 and 2022, 101 patients who underwent PCI with the DyeVert Power XT system (case group) were enrolled to evaluate the amount of CM saved through the use of this device, as well as the rate, severity, and predictors of CI-AKI. Patients who underwent PCI without the use of the device (control group) were enrolled to create a matched group allowing assessment of differences in CM and the CI-AKI rate.
Results: : In the case group, the amount of CM saved was 114 ± 42 mL, representing an average of 32% of the total CM. Fourteen patients (13.9%) developed CI-AKI. The only independent predictors of CI-AKI were hematocrit (OR, 0.86; 95%CI, 0.74-0.99; P = .04) and ejection fraction (OR, 0.88; 95%CI, 0.82-0.95; P = .001). As a result of diversion by the device, the amount of CM delivered was lower in the case group than in controls (252 vs 267 mL; P = .42), but this difference was nonsignificant. Equally, the reduction in CI-AKI (14.3% vs 16.3%) was nonsignificant.
Conclusions: Hematocrit and ejection fraction may be more important predictors of CI-AKI than the CM volume normally used during PCI in the general population. The net practical benefit of DyeVert Power XT was low.
Keywords: Acute kidney injury. Contrast media. Percutaneous coronary intervention. DyeVert.
RESUMEN
Introducción y objetivos: La nefropatía inducida por contraste (NIC) es una potencial complicación de los procedimientos angiográficos. El sistema DyeVert Power (Osprey Medical, Estados Unidos) permite reducir la concentración renal del medio de contraste al disminuir la cantidad administrada a los pacientes. Al contrario que sobre los sistemas manuales, existen pocos datos disponibles sobre el sistema DyeVert, que se utiliza junto a la inyección automática de contraste. El objetivo principal de este estudio fue evaluar su eficacia en procedimientos de intervencionismo coronario percutáneo (ICP).
Métodos: Entre 2020 y 2022 se incluyó a 101 pacientes a quienes se realizó ICP utilizando el sistema DyeVert Power XT (grupo de casos) para evaluar la cantidad ahorrada de medio de contraste, así como la tasa, la gravedad y los predictores de NIC. Además, se seleccionó un grupo control de pacientes a los que se había realizado ICP sin utilizar el sistema DyeVert para comparar la cantidad de medio de contraste administrado y la tasa de NIC.
Resultados: En el grupo de casos se redujo la administración de medio de contraste en 114 ± 42 ml (una media del 32% del total). Desarrollaron NIC 14 pacientes (13,9%). Los predictores de NIC fueron el hematocrito (OR = 0,86; IC95%: 0,74-0,99; p = 0,04) y la fracción de eyección (OR = 0,88; IC95%: 0,82-0,95; p = 0,001). Como resultado de la utilización del sistema DyeVert, la cantidad administrada de medio de contraste fue menor, pero sin diferencias estadísticamente significativas (252 frente a 267 ml; p = 0,42). La tasa de NIC fue menor con el sistema DyeVert, pero sin alcanzar la significación estadística (14,3 frente a 16,3%; p = 1,0).
Conclusiones: El hematocrito y la fracción de eyección, más que la cantidad de contraste administrada, pueden ser predictores de NIC en los pacientes que reciben ICP. El beneficio del sistema DyeVert fue bajo.
Palabras clave: Insuficiencia renal aguda. Medios de contraste. Intervención coronaria percutánea. DyeVert.
Abbreviations
CI-AKI: contrast induced-acute kidney injury. CM/CMV: contrast medium/contrast medium volume. PCI: percutaneous coronary intervention.
INTRODUCTION
Contrast induced-acute kidney injury (CI-AKI) is a dreaded complication after diagnostic and interventional angiographic procedures and is linked to increased morbidity and mortality. In a large recent meta-analysis, the pooled incidence of CI-AKI after coronary angiography was 12.8%, with 95% confidence interval (95%CI) 11.7%-13.9%, and the associated mortality was 20.2% (95%CI, 10.7%-29.7%).1 Multiple risk factors have been identified: contrast medium volume (CMV), advanced age (> 75 years), diabetes, anemia, conditions associated with hypotension, and ejection fraction (EF) < 40%.2,3 Many of these risk factors are included in the Mehran score,2 which identifies 4 risk classes of contrast-induced nephropathy (CIN) after PCI: low (≤ 5 points), moderate (6-10 points), high (11-15 points), and very high (≥ 16 points). The Mehran score and the recent Mehran 2 score4 assign 1 point for each 100 mL of CMV up to a dose of 299 mL. Because volume depletion increases the CM concentration in renal tubules, the main preprocedural measure to reduce the occurrence of CI-AKI is intravenous administration of normal saline before and after the procedure, because other solutions provide no benefits5; hydration should be started 12 hours before and continued for 24 hours after the procedure at 1 mL/kg/h or 0.5 mL/kg/h if EF < 35% or New York Heart Association (NYHA) class > 2.6 Another means of decreasing CM concentration in the kidneys is the DyeVert Contrast Reduction system (Osprey Medical Inc, United States), which reduces the amount of CMV delivered to patients during angiographic procedures, with noninferior image quality as attested by independent reviewers.7,8 The DyeVert, DyeVert Plus and DyeVert Plus EZ are used in conjunction with manual contrast injection, and the DyeVert Power XT is used with automated contrast injection; the latter system has been little studied. The main aim of our study was to evaluate the effectiveness of the DyeVert Power XT system in reducing CM delivery during PCI.
METHODS
Study population
This single center, observational study was performed in patients who underwent PCI between September 2020 and December 2022 with the DyeVert Power XT system (case group) and in patients who underwent PCI during a similar period without the use of the device (control group).
Inclusion criteria for both groups were as follows: chronic kidney disease (CKD) [estimated glomerular filtration rate (eGFR) < 60 mL/min/m2] and/or need for a complex PCI with the likelihood of receiving a large amount of CM; previous coronary artery bypass graft (CABG); chronic total occlusion (CTO) (complete blockage of a coronary artery lasting at least 3 months); bifurcation; and left main and/or multivessel disease (at least 2 vessels involved).
The exclusion criterion for both groups was the presence of end-stage kidney failure on dialysis treatment. We collected laboratory, instrumental, clinical, and procedural variables in the case and control groups. Definitions of all these variables are reported in table 1, table 2, table 3, and table 4. For the variables included in the Mehran score, we used the same descriptions as those used in the score. eGFR was calculated by the Modification of Diet in Renal Disease (MDRD) 4-variable equation, left ventricular EF by 2-dimensional echocardiography during hospitalization and before arrival in the catheterization laboratory, and the risk of any post-PCI CIN by the Mehran score. Bifurcation/left main treatment (with single/double stent) consisted of the proximal optimization technique (POT) with kissing balloon inflation and eventually re-POT in all cases. Total CMV represents the volume that would have been delivered if DyeVert had not been used, ie, the sum of CMV delivered to patients and the CMV saved by DyeVert. CM injection flow was 4 and 3 mL/sec for the left and right coronary artery, respectively.
Table 1. Laboratory, instrumental, clinical characteristics, and Mehran score in the overall population and according to incidence of CI-AKI in the case group
| Characteristics | Overall population (n = 101) | No CI-AKI (n = 87) | CI-AKI (n = 14) | P |
|---|---|---|---|---|
| Laboratory and istrumental characteristics | ||||
| eGFR, mL/min | 51 ± 18 | 52 ± 19 | 45 ± 16 | .18 |
| HCT | 38.6 ± 4.9 | 39.1 ± 4.8 | 35.5 ± 4.8 | .01* |
| EF | 50 [35-55] | 50 [40-55] | 30 [28-36] | < .001* |
| CKD [eGFR < 60 mL/min/ 1.73 m2] | 73 (72.3) | 63 (72.4) | 10 (71.4) | 1 |
| Anemia [male HCT < 39%, female HCT < 36%] | 48 (47.5) | 38 (43.7) | 10 (71.4) | .10 |
| Clinical characteristics | ||||
| Age, years | 74 (68-80) | 73 (67-80) | 75 (74-81) | .09 |
| Age > 75 years | 39 (38.6) | 32 (36.8) | 7 (50) | .52 |
| Male sex | 80 (79.2) | 68 (78.2) | 12 (85.7) | .73 |
| Overweight [body mass index ≥ 25] | 52 (51.5) | 46 (52.9) | 6 (42.9) | .68 |
| Hypertension | 78 (77.2) | 70 (80.5) | 8 (57.1) | .08 |
| Diabetes | 48 (47.5) | 40 (46) | 8 (57.1) | .62 |
| Dyslipidemia | 68 (67.3) | 57 (66) | 11 (79) | .51 |
| Current smoker | 24 (23.8) | 20 (23) | 4 (28.6) | .74 |
| Former smoker | 35 (34.7) | 32 (36.8) | 3 (21.4) | .37 |
| CHF [NYHA class ≥ 3 and/or history of pulmonary edema] | 37 (36.6) | 25 (28.7) | 12 (85.7) | < .001* |
| Acute coronary syndrome presentation | 38 (37.6) | 31 (35.6) | 7 (50) | .46 |
| Hypotension [Systolic arterial pressure < 80 mmHg for ≥ 1 h requiring inotrope] | 4 (4) | 2 (2.3) | 2 (14.3) | .09 |
| Mehran score | ||||
| Mehran CI-AKI risk class: | ||||
| Low | 24 (23.8) | 24 (27.6) | 0 (0) | .04* |
| Moderate | 26 (25.7) | 24 (27.6) | 2 (14.3) | .51 |
| High | 34 (33.7) | 29 (33.3) | 5 (35.7) | 1 |
| Very high | 17 (16.8) | 10 (11.5) | 7 (50) | .002* |
| Mehran score, points | 11 ± 5 | 10 ± 5 | 15 ± 4 | < .001* |
|
Values are expressed as No. (%), mean ± standard deviation, or median [first quartile-third quartile]. *Statistically significant P-value (P < .05). CHF, congestive heart failure; CI-AKI, contrast induced-acute kidney injury; CKD, chronic kidney disease; EF, ejection fraction; eGFR, estimated glomerular filtration rate; HCT, hematocrit; NYHA, New York Heart Association. |
||||
Table 2. Procedural characteristics in the overall population and according to incidence of CI-AKI in the case group
| Characteristics | Overall population (n = 101) | No CI-AKI (n = 87) | CI-AKI (n = 14) | P |
|---|---|---|---|---|
| Procedural characteristics (angiography/PCI complexity/complications) | ||||
| Previous CABG | 20 (19.8) | 18 (20.7) | 2 (14.3) | .73 |
| CTO [complete blockage of a coronary artery lasting at least 3 months] | 12 (11.9) | 11 (12.6) | 1 (7.1) | 1 |
| No. vessels treated in the same procedure: | ||||
| 1 | 57 (56.4) | 52 (59.8) | 5 (35.7) | .09 |
| 2 | 40 (39.6) | 32 (36.8) | 8 (57.1) | .15 |
| 3 | 4 (4) | 3 (3.4) | 1 (7.1) | .45 |
| No. bifurcations treated in the same procedure: | ||||
| 0 | 67 (66.3) | 58 (66.7) | 9 (64.3) | 1 |
| 1 | 31 (30.7) | 27 (31) | 4 (28.6) | 1 |
| 2 | 3 (3) | 2 (2.3) | 1 (7.1) | .36 |
| Left main treatment | 25 (24.8) | 20 (23) | 5 (35.7) | .33 |
| Stent, number | 2 [1-3] | 2 [1-3] | 2 [1-3] | .75 |
| Stent lenght, mm | 52 [31-88] | 51 [30-91] | 57 [36-73] | .95 |
| Perforation | 3 (3) | 3 (3.4) | 0 (0) | 1 |
| IABP use | 1 (1) | 0 (0) | 1 (7.1) | .14 |
| Rotablator use | 3 (3) | 1 (1.1) | 2 (14.3) | .05 |
| Procedural characteristics (others) | ||||
| Radial access | 88 (87.1) | 75 (86.2) | 13 (92.9) | .69 |
| Femoral access | 27 (26.7) | 21 (24.1) | 6 (42.9) | .19 |
| Operator | ||||
| L | 52 (51.5) | 47 (54) | 5 (35.7) | .20 |
| A | 30 (29.7) | 26 (29.9) | 4 (28.6) | 1 |
| B | 4 (4) | 3 (3.4) | 1 (7.1) | .46 |
| V | 13 (12.9) | 10 (11.5) | 3 (21.4) | .38 |
| S | 2 (1.9) | 1 (1.1) | 1 (7.1) | .26 |
| Contrast medium type: | ||||
| Iomeprol 350 | 9 (8.9) | 7 (8) | 2 (14.3) | .61 |
| Iohexol 350 | 13 (12.9) | 11 (12.6) | 2 (14.3) | 1 |
| Iodixanol 320 | 79 (78.2) | 69 (79.3) | 10 (71.4) | .50 |
| Contrast medium dose delivered, mL | 242 [189-300] | 240 [188-306] | 258 [195-277] | .95 |
| Total contrast medium dose [delivered plus saved], mL | 355 ± 110 | 354 ± 79 | 356 ± 106 | .95 |
| IVUS use | 24 (23.8) | 23 (26.4) | 1 (7.1) | .18 |
|
Data are expressed as No. (%), mean ± standard deviation, or median [first quartile-third quartile]. CABG, coronary artery bypass graft; CI-AKI, contrast induced-acute kidney injury; CTO, chronic total occlusion; IABP, intra-aortic balloon pump; IVUS, intravascular ultrasound. |
||||
Table 3. Laboratory, instrumental, clinical characteristics, and Mehran score of cases and controls in the matched group
| Characteristics | No DyeVert (n = 49) | DyeVert (n = 49) | P | Standardized mean difference |
|---|---|---|---|---|
| Laboratory and istrumental characteristics | ||||
| eGFR, mL/min | 53 ± 18 | 51 ± 18 | .70 | 0.11 |
| HCT | 37.8 ± 4.1 | 38.2 ± 4.9 | .68 | 0.08 |
| EF | 50 [40-55] | 50 [35-55] | .68 | 0.13 |
| CKD [eGFR < 60 mL/min/1.73 m2] | 36 (73.5) | 34 (69.4) | .82 | 0.09 |
| Anemia [male HCT < 39, Female HCT < 36] | 27 (55.1) | 24 (49) | .69 | 0.12 |
| Clinical characteristics | ||||
| Age, years | 75 ± 9 | 75 ± 9 | .96 | 0.01 |
| Age > 75 years | 26 (53.1) | 24 (49) | .84 | 0.08 |
| Male sex | 38 (77.6) | 41 (83.7) | .61 | 0.15 |
| Overweight [body mass index ≥ 25] | 22 (44.9) | 24 (49) | .84 | 0.08 |
| Hypertension | 33 (67.3) | 37 (75.5) | .50 | 0.19 |
| Diabetes | 19 (38.8) | 20 (40.8) | 1 | 0.04 |
| Dyslipidemia | 28 (57.1) | 32 (65.3) | .53 | 0.17 |
| Current smoker | 11 (22.4) | 10 (20.4) | 1 | 0.05 |
| Former smoker | 16 (32.7) | 18 (36.7) | .83 | 0.09 |
| CHF [NYHA class ≥ 3 and/or history of pulmonary edema] | 15 (30.6) | 15 (30.6) | 1 | < 0.01 |
| Acute coronary syndrome presentation | 27 (55.1) | 25 (51) | .84 | 0.08 |
| Hypotension [systolic pressure < 80 mmHg for ≥ 1 h requiring inotrope] | 2 (4.1) | 2 (4.1) | 1 | < 0.01 |
| Mehran score | ||||
| Mehran CI-AKI risk class: | ||||
| Low | 12 (24.5) | 10 (20.4) | .63 | 0.09 |
| Moderate | 12 (24.5) | 15 (30.6) | .50 | 0.14 |
| High | 13 (26.5) | 15 (30.6) | .65 | 0.09 |
| Very high | 12 (24.5) | 9 (18.4) | .46 | 0.16 |
| Mehran score, points | 11 ± 6 | 11 ± 6 | .86 | 0.04 |
|
Data are expressed as No. (%), mean ± standard deviation, or median [first quartile-third quartile]. CHF, congestive heart failure; CI-AKI, contrast induced-acute kidney injury; CKD, chronic kidney disease; EF, ejection fraction; eGFR, estimated glomerular filtration rate; HCT, hematocrit; NYHA, New York Heart Association. |
||||
Table 4. Procedural characteristics of cases and controls in the matched group
| Characteristics | No DyeVert (n = 49) | DyeVert (n = 49) | P | Standardized mean difference |
|---|---|---|---|---|
| Procedural characteristics (angiography/PCI complexity/complications) | ||||
| Previous CABG | 8 (16.3) | 6 (12.2) | .56 | 0.10 |
| CTO [complete blockage of a coronary artery lasting at least 3 months] | 6 (12.2) | 8 (16.3) | .77 | 0.13 |
| No. vessels treated in the same procedure: | ||||
| 1 | 32 (65.3) | 29 (59.2) | .53 | 0.12 |
| 2 | 15 (30.6) | 17 (34.7) | .67 | 0.08 |
| 3 | 2 (4.1) | 3 (6.1) | 1 | 0.10 |
| No. bifurcations treated in the same procedure: | ||||
| 0 | 33 (67.3) | 31 (63.3) | .67 | 0.09 |
| 1 | 15 (30.6) | 16 (32.7) | .83 | 0.04 |
| 2 | 1 (2.1) | 2 (4) | 1 | 0.12 |
| Left main treatment | 12 (24.5) | 13 (26.5) | 1 | 0.05 |
| Stent, number | 2 [1-3] | 2 [1-3] | .30 | 0.15 |
| Stent lenght, mm | 46 [30-85] | 52 [33-97] | .41 | 0.13 |
| Perforation | 2 (4.1) | 1 (2) | 1 | 0.12 |
| IABP use | 0 (0) | 1 (2) | 1 | 0.20 |
| Rotablator use | 0 (0) | 2 (4.1) | .49 | 0.24 |
| Procedural characteristics (others) | ||||
| Radial access | 41 (83.7) | 45 (91.8) | .35 | 0.24 |
| Femoral access | 11 (22.4) | 15 (30.6) | .49 | 0.18 |
| Operator: | ||||
| L | 24 (49) | 24 (49) | 1 | < 0.01 |
| A | 17 (34.7) | 17 (34.7) | 1 | < 0.01 |
| B | 5 (10.2) | 3 (6.1) | .71 | 0.20 |
| V | 3 (6.1) | 5 (10.2) | .71 | 0.12 |
| Contrast medium type: | ||||
| Iomeprol 350 | 7 (14.3) | 4 (8.2) | .34 | 0.21 |
| Iohexol 350 | 9 (18.4) | 10 (20.4) | .80 | 0.06 |
| Iodixanol 320 | 33 (67.3) | 35 (71.4) | .66 | 0.10 |
| IVUS use | 10 (20.4) | 11 (22.4) | 1 | 0.05 |
|
Data are expressed as No. (%), mean ± standard deviation, or median [first quartile-third quartile]. CABG, coronary artery bypass graft; CI-AKI, contrast induced-acute kidney injury; CTO, chronic total occlusion; IABP, intra-aortic balloon pump; IVUS, intravascular ultrasound. |
||||
Image quality was evaluated by operators during the procedures. When quality was inadequate, exclusion of the device from the CM line was allowed for the shortest possible time.
AKI was defined as a rise in the concentration of serum creatinine ≥ 0.3 mg/dL within 48 hours after CM administration from the baseline value obtained before CM injection; further measurements after 48 hours were collected in patients with worsening kidney function; for its prevention, all patients received hydration with sodium chloride 0.9% intravenous solution at a rate of 1 or 0.5 mL/kg/h, as appropriate. The severity of AKI was defined according to Kidney Disease Improving Global Outcome (KDIGO) stages.
The research reported was performed in accordance with recommendations for clinical investigation (Declaration of Helsinki of the World Medical Association, October 2013) and was approved by an ethics committee. We declare that relevant informed consent was obtained from all participants and is available.
Objectives
In the case group, we evaluated the following: a) the amount of CMV saved using DyeVert and image quality; b) the rate and severity of CI-AKI and the rate of in-hospital all-cause death; c) laboratory, instrumental, clinical, and procedural differences in the 2 subgroups defined on the basis of the incidence of AKI; and d) independent predictors of CI-AKI.
In the overall population of the case and control groups, we performed propensity score matching (PSM) to obtain a group of patients with a sufficiently good balance (matched group), in which we evaluated the following: a) differences in CMV, and b) rate and severity of CI-AKI.
Statistical analysis
Categorical variables are expressed as the number and percentage of patients. Continuous parametric data are reported as the mean ± standard deviation and continuous nonparametric data as the median [lower and upper quartile]; for assessment of normality, the Kolmogorov test was used. Patients’ categorical variables were compared using the chi-squared test (with Yates’ correction for continuity in the case of variables with only 2 categories) or the Fisher exact test, as appropriate. The unpaired t-test was used for continuous parametric variables and the Mann-Whitney U-test for continuous nonparametric variables; the same tests were used in the matched group. On univariate analysis, significance was defined as P < .05. To establish the independent predictors of AKI, we performed multivariable logistic regression analysis. Variables were selected according to significance in the univariate analysis. The chosen method was stepwise backward regression with a maximum of 20 iterations. Multicollinearity was assessed with tolerance and variance inflation factor (VIF) values. Receiver operating characteristic (ROC) curves were used to establish the optimal cutoffs of independent predictors for the diagnosis of AKI. To perform PSM, the algorithm used was nearest neighbor matching 1:1 with a caliper size of ± 0.2. Statistical analyses were performed using SPSS for Windows, release 29, with R 4.2 implementation to perform PSM.
RESULTS
Analysis in the case group
A total of 101 patients (median age 74 [68-80] years, male sex 79.2%, CKD 72.3%) underwent PCI with the use of the DyeVert Power XT system.
In the overall population of the case group, mean hematocrit (HCT) was 38.6 ± 4.9 %, median EF was 50% [35%-55%], and mean Mehran score was 11 ± 5 points.
Congestive heart failure (CHF) was present in 37 patients (36.6%), Mehran CI-AKI very high-risk class was present in 17 patients (16.8%) and Mehran CI-AKI low-risk class was present in 24 patients (23.8%) (table 1).
We enrolled 20 patients (19.8%) with previous CABG, 12 (11.9%) with CTO, 34 (33.7%) with bifurcations, 25 (24.8%) with left main coronary artery disease, and 44 (43.6%) with multivessel disease. Delivered CM was 242 (189-300) mL, total CM was 355 ± 110 mL, and saved CM was 114 ± 42 mL, with an average of 32% of the total CMV (table 2). In almost all patients (n = 96, 95% of patients), image quality was adequate, while the device was excluded to make it adequate for the shortest possible time in 5 patients. Without these exclusions, saved CMV would have been slightly higher and with trivial changes with regard to the comparison with controls: 33% of the total, a value derived from patients without exclusions (n = 96).
A total of 14 (13.9%) patients developed CI-AKI (AKI-KDIGO 1, 2, 3: 6.9%, 3%, and 4%, respectively). The results of the univariate analysis for the overall population and according to the incidence of CI-AKI in the case group are reported in table 1, table 2, and figure 1.
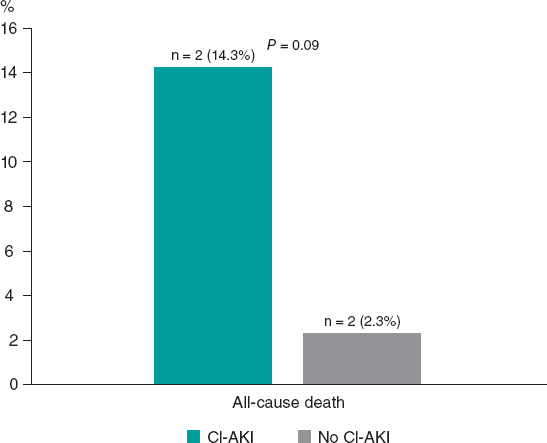
Figure 1. In-hospital all-cause mortality rate according to onset of CI-AKI in the case group. CI-AKI, contrast induced-acute kidney injury.
Compared with patients not developing CI-AKI, those in the CI-AKI subgroup had lower HCT values (35.5 ± 4.8 vs 39.1 ± 4.8; P = .01), lower EF values (30 [28-36] vs 50 [40-55]; P < .001) and higher Mehran score values (15 ± 4 vs 10 ± 5; P < .001).
In addition, the first patients more frequently had CHF [12 (85.7%) vs 25 (28.7%); P <.001] and Mehran CI-AKI very high-risk class (7 [50%] vs 10 [11.5%]; P =.002) and less frequently had Mehran CI-AKI low-risk class (0 [0%] vs 24 [27.6%]; P = .04).
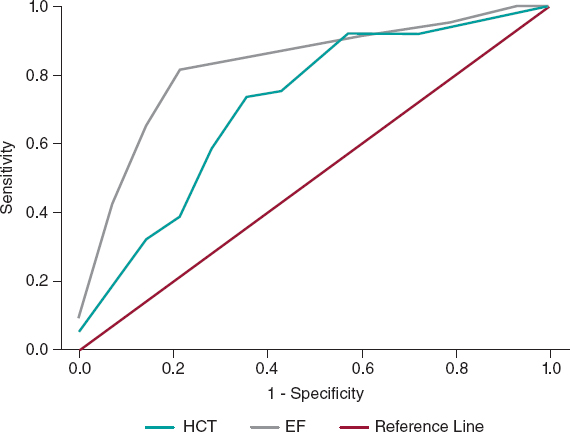
No significant differences were found in the remaining laboratory, instrumental, or clinical features or the procedural variables between the 2 subgroups; in particular, CM was slightly higher in CI-AKI patients: 258 [195-277] vs 240 [188-306] mL, total 356 ± 106 vs 354 ± 79 mL; P = .95 for both variables delivered. In the multivariate analyses, independent predictors of CI-AKI were HCT (OR, 0.86, 95%CI, 0.74-0.99; P = .04) and EF (OR, 0.88, 95%CI, 0.82-0.95; P = .001); the percentage accuracy in classification of the model was 88%, while tolerance and VIF values (0.99 and 1.01, respectively) showed no multicollinearity. The HCT ROC curve showed the following values: area under curve (AUC) 0.71 with 95%CI 0.56-0.87; P = .01; a cutoff of 36.3% had the best sensitivity (72%) and specificity (71%) for the outcome (figure 2). The EF ROC curve showed the following values: AUC 0.83 with 95%CI 0.72-0.94; P = .001; a cutoff of 37% had the best sensitivity (82%) and specificity (79%) (figure 2); therefore, our best predictor was EF < 40%.
Figure 2. Receiver operating characteristic curves showing the diagnostic ability of HCT and EF for the diagnosis of CI-AKI in the case group. CI-AKI, contrast induced-acute kidney injury; EF, ejection fraction; HCT, hematocrit.
There were 4 in-hospital all-cause deaths overall, 2 deaths in each subgroup (CI-AKI and no–CI-AKI subgroups), as shown in figure 1.
Analysis in the matched group
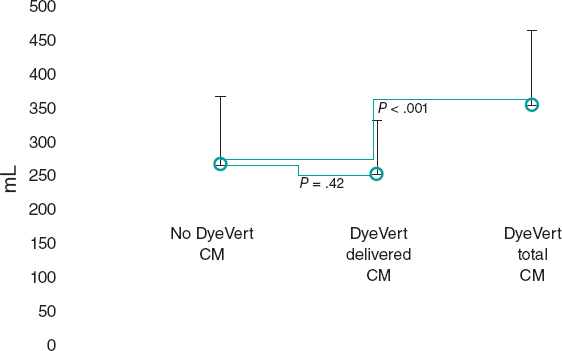
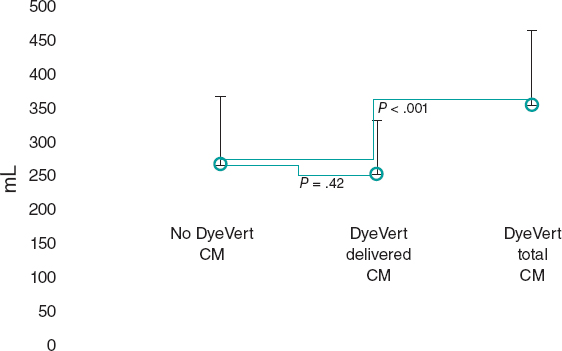
After the matching process, 49 patients remained in the control (no DyeVert) and case (DyeVert) groups with no significant imbalance (ie, standardized mean differences < ± 0.25), as reported in table 3 and table 4. As shown in figure 3, delivered CM was slightly lower in the DyeVert group than in the no-DyeVert group, with no significant difference (252 ± 80 vs 267 ± 101 mL; P = .42), while total CM was significantly higher in the DyeVert group (354 ± 110 vs 267 ± 101 mL; P < .001). The CI-AKI rate was slightly lower in the case group than in the control group (14.3% vs 16.3%; P = .99) with slightly more advanced stages of AKI in controls (table 1 of the supplementary data), without significance.
Figure 3. Contrast medium in cases and controls in the matched group. CM, contrast medium; blue dots represent the median values; vertical black lines represent the standard deviations.
DISCUSSION
In the case group, the DyeVert Power XT system saved 32% of CM and image quality was adequate in almost all cases; the only independent predictors of CI-AKI were HCT and EF.
In the matched group, total CM was higher in cases than in controls. After diversion by the device, delivered CM was slightly lower in cases than in controls, but without significance. The reduction in CI-AKI was also nonsignificant.
The DyeVert system is a second-generation device to reduce the amount of CM delivered to patients during angiographic procedures. The first generation was the AVERT system (Osprey Medical Inc), which showed a relative reduction of approximately 23% in CMV among PCI patients compared with controls; the use of the device did not reduce the AKI rate.9 DyeVert Power XT is used in combination with automatic injection; few data are available in this context, being limited to 2 studies that investigated 2610 and 9 patients,11 without a control group. There are more data on manual injection (1696 patients, 15 studies); all these 17 studies were collectively analyzed in the meta-analysis by Tarantini et al.12
In that meta-analysis, the mean saved CMV in the DyeVert group was reported by 7 observational studies and ranged from 34% to 47% of total CMV; the pooled estimate value was approximately 39.5% using manual CM injection systems; of note, the lowest value (34%) was achieved using DyeVert Power XT. We found a similar value in the DyeVert (case) group. These reduced values compared with manual systems may be related to different pressures generated during automatic contrast injection.
In our case group analysis, CMV was not significantly correlated with the occurrence of CI-AKI, which instead was independently predicted by lower HCT and EF values, which are known risk factors, as shown by Mehran scores.2,4 EF was also an independent predictor in the study by Briguori et al.13 Our findings confirm the importance of first identifying the variables (eg, those in the Mehran or Mehran 2 scores)2,4 that classify patients at higher risk of CI-AKI to apply appropriate preventive strategies. In the present study, these patients were identified by HCT and EF and consequently the latter variables (especially EF) may be more important predictors than CMV, which is normally used during PCI in the general population. In the above-mentioned scores, CMV was also an independent predictor of CI-AKI and, consequently, using the smallest possible value of CMV is still important, especially in higher risk patients. DyeVert thus has the potential benefit of reducing CIN, depending on its efficacy compared with controls, which was evaluated in the above-mentioned meta-analysis and in the present study.
In the meta-analysis, approximately half of the studies included controls for comparison. Delivered CM was usually lower in DyeVert patients than in controls. In these cases, the difference ranged from 22 to 50 mL,12 with the highest differences being reported in the studies by Tajti et al. (200 [153-256] vs 250 [170-303] mL; P = .04) and Briguori et al. (99 ± 50 vs 130 ± 50 mL; P <.001).13,14 Delivered CMV was slightly higher (difference of 2 mL) in the DyeVert group only in the study by Bunney et al.15 The pooled analysis showed a significant decrease in delivered CMV with DyeVert use relative to the control group. Of note, details about prior CABG, CTO and left main treatment were reported only in 1 work14 and the number of vessels treated was reported only in another work.13 The treatment of bifurcations and differences in operators were not reported. All these procedural characteristics, which may influence the amount of CM delivered during PCI, were included in our study and we used a matched group with a sufficiently good balance in the studied characteristics.
In our matched group, delivered CM was lower in the case group than in the control group, but the difference was slight and nonsignificant, while total CM (also called attempted in the meta-analysis) was significantly higher in the case group than in the control group. Consequently, the net practical benefit of the device in terms of spared CM was low. In our work, procedural characteristics (eg, procedural complexity), which could cause discrepancies in CM injections, were balanced in the matched group. Based on these findings, we believe that the control group required more prolonged and/or a greater number of contrast injections (and consequently more total CM) to achieve adequate image quality. In previous studies, adequate image quality was achieved with DyeVert in 98% of cases,12 a value similar to ours, but those studies did not discuss the need for prolonged injections and more total CM compared with controls to maintain image quality when DyeVert is used. Few data are available on total CM, but previous studies reporting this information indicate that total CM was higher in DyeVert patients than in controls (Briguori et al., P-value almost significant; Kutschman et al., P-value not reported).12,13,16
The reduction in CI-AKI in the present study was not statistically significant. In the meta-analysis, the pooled relative risk for CI-AKI associated with DyeVert system use was 0.60 (95%CI, 0.40-0.90; P = .01), which was a result derived from 5 studies. Moreover, in a recent abstract not included in the meta-analysis, postprocedure eGFR values among patients undergoing coronary and/or peripheral angiography were significantly more stable in the DyeVert group than in controls.17
Analysis of the 5 above-mentioned studies separately revealed that our results are mainly in agreement; indeed, the relative risk was significantly lower in only 1 study in the nonpooled analysis.13
The type of CM was not associated with the occurrence of CI-AKI; as recommended,18 we used iso-osmolar (Iodixanol 320) or low-osmolar (Iomeprol 350 or Iohexol 350) contrast agents to prevent CIN. Given the presence of more favorable evidence,19 we preferred to use the iso-osmolar agent and reserved the other agents to low-risk patients.
Study limitations
Our study has some limitations. First, the sample size was relatively small. Second, the study design was single center, observational and retrospective, although we performed PSM to reduce potential confounding bias. Third, we excluded patients not meeting the inclusion criteria, as they were usually at low risk of CI-AKI. Therefore, our results should be generalized with caution, since the analyzed patients may be not representative of the general population. In this work the variable of sex has not been taken into account in accordance with the SAGER guidelines.
CONCLUSIONS
The DyeVert Power XT system saved 32% of CM, but only HCT and EF were independent predictors of CI-AKI and the main predictor was EF < 40%. Therefore, these variables (especially EF) may be more important than CMV, which is normally used during PCI in the general population.
PCI with this system required more total CM compared with that in controls to achieve adequate image quality. Consequently, after CM saving by the device, delivered CM was only slightly lower than CM in controls (mean difference of 15 mL) and this difference was nonsignificant. Therefore, the net practical benefit of the system was low. Equally, the reduction in CI-AKI (14.3% vs 16.3%) was not statistically significant.
Future studies are needed to confirm these results.
FUNDING
The authors did not receive any grants for this research.
ETHICAL CONSIDERATIONS
The work has been approved by an Ethics Committee/institution. Informed consent of patients was obtained and archived for the publication of their cases. In this work the variable of sex has not been taken into account in accordance with the SAGER guidelines.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
We didn’t use artificial intelligence for the development of our work.
AUTHORS’ CONTRIBUTIONS
F. Vergni, M.Arioti, and M.Leoncini contributed to the design of the work. F. Vergni, M. Arioti, V. Boasi, F.A. Sánchez, M.Leoncini, and F. Ferrari contributed to the acquisition of data. F. Vergni analyzed the data. F. Vergni, M.Arioti, V. Boasi, F.A. Sánchez, M. Leoncini, and F. Ferrari contributed to the interpretation of the data. F. Vergni and M.Arioti contributed to the drafting of the work. F. Vergni, M. Arioti, V. Boasi, F.A. Sánchez, M.Leoncini, and F. Ferrari revised the work and approved the final version to be published.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.
What is known about the topic?
- The DyeVert Power XT system (which is used in conjunction with automated contrast injection) has been assessed in only 2 studies, which included a total of 35 patients investigated without a control group and mainly not during PCI.
What does this study add?
- Our study investigated the device in a larger population (n = 101) and during PCI. Moreover, we included a control group and performed propensity score matching to obtain a group of patients with a sufficiently good balance regarding laboratory, instrumental, clinical and procedural characteristics; in addition, among the latter features, we included the treatment of coronary bifurcations and differences between operators, which were not reported in previous studies. The above-mentioned characteristics may influence the outcome (ie, CI-AKI occurrence) and/or the volume of CM used and therefore their inclusion is important when assessing a device to spare CM.
REFERENCES
1. Lun Z, Liu L, Chen G, et al. The global incidence and mortality of contrast-associated acute kidney injury following coronary angiography:a meta-analysis of 1.2 million patients. J Nephrol. 2021;34:1479-1489.
2. Mehran R, Aymong ED, Nikolsky E, et al. A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention:development and initial validation. J Am Coll Cardiol. 2004;44:1393-1399.
3. Azzalini L, Spagnoli V, Ly HQ. Contrast-induced nephropathy:from pathophysiology to preventive strategies. Can J Cardiol. 2016;32:247-255.
4. Mehran R, Owen R, Chiarito M, et al. A contemporary simple risk score for prediction of contrast-associated acute kidney injury after percutaneous coronary intervention:derivation and validation from an observational registry. Lancet. 2021;398:1974-1983.
5. Almendarez M, Gurm HS, Mariani J Jr, et al. Procedural strategies to reduce the incidence of contrast-induced acute kidney injury during percutaneous coronary intervention. JACC Cardiovasc Interv. 2019;12:1877-1888.
6. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
7. Desch S, Fuernau G, Pöss J, et al. Impact of a novel contrast reduction system on contrast savings in coronary angiography –the DyeVert randomised controlled trial. Int J Cardiol. 2018;257:50-53.
8. Zimin VN, Jones MR, Richmond IT, et al. A feasibility study of the DyeVerttm plus contrast reduction system to reduce contrast media volumes in percutaneous coronary procedures using optical coherence tomography. Cardiovasc Revasc Med. 2021;30:40-46.
9. Mehran R, Faggioni M, Chandrasekhar J, et al. Effect of a contrast modulation system on contrast media use and the rate of acute kidney injury after coronary angiography. JACC Cardiovasc Interv. 2018;11:1601-1610.
10. Amoroso G, Christian J, Christopher A. First European experience using a novel contrast reduction system during coronary angiography with automated contrast injection. [Abstract]. Eurointervention. 2020;16(Suppl. AC):Euro20A-POS426.
11. Bruno RR, Nia AM, Wolff G, et al. Early clinical experiences with a novel contrast volume reduction system during invasive coronary angiography. Int J Cardiol Heart Vasc. 2019;23:100377.
12. Tarantini G, Prasad A, Rathore S, et al. DyeVert Contrast Reduction System Use in Patients Undergoing Coronary and/or Peripheral Angiography:A Systematic Literature Review and Meta-Analysis. Front Med (Lausanne). 2022;9:841876.
13. Briguori C, Golino M, Porchetta N, et al. Impact of a contrast media volume control device on acute kidney injury rate in patients with acute coronary syndrome. Catheter Cardiovasc Interv. 2021;98:76-84.
14. Tajti P, Xenogiannis I, Hall A, et al. Use of the DyeVert system in chronic total occlusion percutaneous coronary intervention. J Invasive Cardiol. 2019;31:253-299.
15. Bunney R, Saenger E, Shah C, et al. Contemporary use of contrast dye reduction technology in a tertiary academic hospital:patient characteristics and acute kidney injury outcomes following percutaneous coronary interventions. In:Acc 2019. 1st Quality Summit;2019 March 13-15;New Orleans, United States.
16. Kutschman R, Davison L, Beyer J. Comprehensive clinical quality initiative for reducing acute kidney injury in at-risk patients undergoing diagnostic coronary angiogram and/or percutaneous coronary interventions. In:Scai 2019. 42nd Scientific Sessions;2019 May 19-22;Las Vegas, United States.
17. Olubowale O, Ur Rahman E, U Okoro K, et al. The DyeVert contrast reduction system and contrast induced nephropathy:is it any better?J Am Coll Cardiol. 2022;79(Suppl 9):S903.
18. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
19. Zhao F, Lei R, Yang SK, et al. Comparative effect of iso-osmolar versus low-osmolar contrast media on the incidence of contrast-induced acute kidney injury in diabetic patients:a systematic review and meta-analysis. Cancer Imaging. 2019;19:38.
ABSTRACT
Introduction and objectives: Invasive diagnosis of vasoreactivity and microvascular function may be useful to optimize the management of patients with signs and/or symptoms of myocardial ischemia in the absence of significant coronary stenosis (INOCA). We analyzed the prevalence of the different endotypes, as well as the concordance between 2 diagnostic methods based on thermodilution assessment.
Methods: We prospectively included 60 patients with INOCA who underwent a vasoreactivity test with intracoronary acetylcholine, and measurement of absolute coronary blood flow (Q) and minimum microvascular resistance (R) using continuous thermodilution assessment. Finally, calculations of the coronary flow reserve (CFR) and index of microcirculatory resistance index (IMR) were made using the bolus thermodilution method considering CFR < 2 and MRI ≥ 25 as established pathological cut-off values.
Results: The invasive functional diagnostic procedure allowed patients to be categorized into 4 subgroups: microvascular dysfunction (40%), epicardial vasospasm (17%), mixed disorder (20%), and normal study (23%). No correlation was seen between the Q and the CFR. Using ROC curves, an R > 435 UW was estimated as the optimal cut-off value to identify patients with IMR ≥ 25 with an area under the curve of 0.67 (95%CI, 0.51-0.82; P = .04).
Conclusions: The invasive study of vasoreactivity and microcirculation was feasible and safe. Prevalence of vasospasm and microvascular dysfunction in patients with INOCA was high. The CFR/MRI/Q combined study allowed us to unmask a subtype of microvascular dysfunction characterized by an abnormally high coronary flow at baseline. The concordance seen between the microvascular resistance obtained by continuous thermodilution measurements and the reference method was low so future studies are justified to determine the usefulness of this technique.
Keywords: Microvascular dysfunction. Vasospasm. Acetylcholine. Continuous thermodilution measurements. Microvascular resistance. INOCA.
RESUMEN
Introducción y objetivos: El diagnóstico invasivo de la vasorreactividad y la función microvascular puede resultar de utilidad para optimizar el manejo de los pacientes con signos o síntomas de isquemia miocárdica en ausencia de estenosis coronarias significativas (INOCA). Se analizó la prevalencia de los distintos endotipos y la concordancia entre 2 métodos diagnósticos basados en la termodilución.
Métodos: Se incluyeron de forma prospectiva 60 pacientes con INOCA a quienes se realizó un test de vasorreactividad con acetilcolina intracoronaria, medida del flujo absoluto (Q) y la resistencia microvascular mínima (R) por termodilución continua y, por último, se calcularon la reserva de flujo coronario (RFC) y el índice de resistencia microvascular (IRM) por termodilución con bolos. Se consideraron como patológicos los puntos de corte establecidos de RFC < 2 e IRM ≥ 25.
Resultados: El procedimiento diagnóstico funcional invasivo permitió clasificar a los pacientes en 4 subgrupos: disfunción microvascular (40%), vasoespasmo epicárdico (17%), trastorno mixto (20%) y estudio normal (23%). No se observó correlación entre Q y RFC. Mediante curvas ROC se estimó una R > 435 UW como el punto de corte óptimo para identificar pacientes con IRM ≥ 25, con un área bajo la curva de 0,67 (IC95%, 0,51-0,82; p = 0,04).
Conclusiones: El estudio invasivo de la vasorreactividad y la microcirculación fue factible y seguro. La prevalencia de vasoespasmo y de disfunción microvascular en pacientes con INOCA fue elevada. El análisis conjunto de RFC, IRM y Q permitió desenmascarar un subtipo de disfunción microvascular caracterizado por un flujo coronario basal anormalmente elevado. La concordancia entre la resistencia microvascular obtenida por termodilución continua respecto al método de referencia fue baja, por lo que se requieren futuros estudios para determinar la utilidad de esta técnica.
Palabras clave: Disfunción microvascular. Vasoespasmo. Acetilcolina. Termodilución continua. Resistencia microvascular. INOCA.
Abbreviations
CFR: coronary flow reserve; INOCA: ischemia with nonobstructive coronary artery disease; IMR: index of microcirculatory resistance; Q: absolute coronary blood flow; R: coronary microvascular resistance.
INTRODUCTION
Over the past few years, the term INOCA (ischemia with nonobstructive coronary arteries) has established to define patients with signs or symptoms of ischemic heart disease without angiographically significant obstructive coronary artery disease.1 In these patients, coronary microvascular or epicardial vessel dysfunction could be the pathophysiological mechanism triggering the symptoms and ischemic impairment.2
Currently, the invasive study of microvascular function in patients with INOCA is a recommendation IIa according to the clinical practice guidelines of the European Society of Cardiology.3 What it does is measure the parameters that show its functional or structural status like coronary flow reserve (CFR) or index of microcirculatory resistance (IMR).4
Recently, the possibility of measuring absolute coronary blood flow (Q) and microvascular resistance (R) by continuous thermodilution with the infusion of a physiological saline solution through a specific coronary microcatheter has been described. This technique has potential advantages like its independence from the operator or not needing pharmacologically induced hyperemia.5
The objective of this study is to estimate the prevalence of the different endotypes of patients with INOCA and analyze the correlation between the measurements obtained by continuous thermodilution and the traditional method of intracoronary boluses of physiological saline solutions.
METHODS
This was a prospective and consecutive study of 60 referred patients due to symptoms or signs of myocardial ischemia without angiographically significant coronary artery stenosis on the visual estimate (< 50%) or after functional assessment (resting full-cycle ratio [RFR] > 0.89 or fractional flow reserve [FFR] > 0.80). Severe valvular heart disease, acute coronary syndrome, decompensated heart failure, and any clinical or anatomical condition where the study of microcirculation and vasoreactivity would be considered unnecessary were excluded.
All microcirculation and vasoreactivity studies were scheduled and second-staged. Nitrates and calcium antagonists were withdrawn prior to conducting the tests.
The coronary angiography was performed based on the routine clinical practice via radial access. A spasmolytic cocktail of 200 µg of nitroglycerin was administered. The target artery was the left main coronary artery.
The study was approved by the center ethics research committee and the patients’ written informed consent was obtained.
Vasoreactivity test
First, the vasoreactivity test was performed. Patient monitoring included precordial leads, and baseline angiograms were performed using 2 different projections. The sequential administration of acetylcholine was followed by increasing doses of 2 µg, 20 µg, and 100 µg in intracoronary bolus for 2 min. In the presence of significant bradycardia, the injection was interrupted, and if considered appropriate, it was re-administered at a slower rate. A follow-up angiogram was performed after every dose. In the presence of severe symptoms, changes to the echocardiogram or epicardial spasm 200 µg of intracoronary nitroglycerin were administered.
The test was considered positive based on the criteria established by the COVADIS (Coronary vasomotor disorders international study) group: epicardial spasm in the presence of chest pain, changes to the echocardiogram, and constriction ≥ 90%, and microvascular spasm in the presence of chest pain, and changes to the echocardiogram without epicardial spasm ≥ 90%.6
Indices obtained with continuous thermodilution
After the administration of unfractionated heparin (70 IU/kg), a pressure-temperature sensor guidewire Pressure Wire X (Abbott, United States) was inserted and pressures at the catheter distal border were equalized. The guidewire was advanced until it reached the left anterior descending coronary artery distal segment.
Resting full-cycle ratio was registered to confirm the lack of hemodynamically significant epicardial stenoses (RFR > 0.89).
Afterwards, a specific Rayflow (Hexacath, France) microcatheter for intracoronary infusion was placed in the left anterior descending coronary artery proximal segment. After confirmation that the guidewire sensor was, at least, 3 cm distal to the tip of the microcatheter, the intracoronary infusion of a physiological saline solution at room temperature and at a dose of 20 mL/min was started using an injector pump to induce hyperemia.
Pressure-temperatures curves were registered using Coroventis software (Abbott, United States). When the distal temperature drop was stabilized, the sensor was withdrawn up to the tip of the microcatheter to determine the infusion temperature.
Afterwards, the injection of the physiological saline solution stopped, and Q (L/min) and R (Wood units) values were obtained automatically (figure 1).
Figure 1. Measurements obtained by continuous thermodilution: DP, distal pressure; Q, absolute coronary blood flow; Qi, infusion flow (mL/min); R, microvascular resistance; T, distal temperature; Ti, infusion temperature.
Indices obtained with bolus thermodilution of a physiological saline solution
After completion of the continuous thermodilution study, and once the Rayflow microcatheter was removed, the pressure-temperature guidewire was repositioned in its previous location, and thermodilution curves were registered using the Coroventis software after the vigorous manual injection of 3 intracoronary boluses of 3 mL of a physiological saline solution. Measurements were taken at rest and after inducing hyperemia with a peripheral intravenous bolus of regadenoson (400 µg) resulting in the calculation of CFR and IMR.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation or median [interquartile range]. The categorical ones were expressed as absolute value or percentage. ROC (Receiver operating characteristic) curves were used to estimate the optimal cut-off values for the continuous variables Q and R. The cut-off values established as pathological for CFR < 2 and IMR ≥ 25 were used as the reference framework. Once dichotomized, the variables Q and R were compared to the CFR and IMR values using chi-square tests. One-way ANOVA was used to compare the different quantitative variables. The statistical analysis was performed using the SPSS v 20 statistical software package (IBM, United States).P values < .05 were considered statistically significant.
RESULTS
Study patients
Table 1 shows the baseline characteristics of the 60 patients included in the study. Women (55%) were predominant. Also, there was a high prevalence of cardiovascular risk factors. Most showed typical angina-like clinical signs (76%) and had tested positive to an ischemia test performed before the coronary angiography (60%).
Table 1. Clinical and angiographic characteristics (N = 60)
| Age (years) | 63 ± 10 |
| Women | 33 (55%) |
| Hypertension | 39 (65%) |
| Diabetes | 21 (35%) |
| Dyslipidemia | 35 (58%) |
| Smoking (current or past) | 28 (47%) |
| Previous percutaneous revascularization | 4 (7%) |
| Previous myocardial infarction | 3 (5%) |
| Left ventricular systolic dysfunction | 4 (7%) |
| Ejection fraction (%) | 63 ± 8 |
| Clinical presentation | |
| Exertional angina | 19 (32%) |
| Resting angina | 13 (22%) |
| Mixed angina | 14 (23%) |
| Other | 14 (24%) |
| Ischemia test | |
| Ergometry | 19 (32%) |
| Isotopic scintigraphy | 18 (30%) |
| Dobutamine stress echocardiography | 3 (5%) |
| None | 20 (33%) |
| Coronary angiography | |
| Atheromatous disease | 22 (37%) |
| Slow flow | 13 (22%) |
|
Data are expressed as no. (%) or mean ± standard deviation. |
|
The baseline coronary angiography confirmed that 37% of the patients showed parietal irregularities consistent with atheromatous disease, and 22% had slow coronary flow. The FFR and RFR values were normal in all the cases studied.
Coronary vasoreactivity
As shown on table 2, 60% of the cases (36/60) had a positive response to acetylcholine in the vasoreactivity test. A total of 32% of the cases (19/60) showed severe epicardial vasoconstriction, and 23% (14/60) met the criteria for microvascular spasm. In 3 patients (5%), microvascular spasm was observed concomitantly with the medium dose (20 µg), and epicardial spasm with the high dose (100 µg), which added to the impaired indices of microvascular function was consistent with a mixed endotype.
| Pathological vasoreactivity testing | 36 (60%) |
| Epicardial vasospasm | 19 (32%) |
| Microvascular vasospasm | 14 (23%) |
| Combined vasospasm | 3 (5%) |
| Structural microvascular dysfunction (IMR ≥ 25) | 20 (33%) |
| Isolated | 5 (8%) |
| Associated with epicardial spasm | 8 (13%) |
| Associated with microvascular spasm | 4 (7%) |
| Associated with combined spasm | 3 (5%) |
| CFR < 2 | 11 (18%) |
| CFR < 2.5 | 17 (28%) |
| RFR | 0.93 [0.91-0.94] |
| FFR | 0.90 [0.87-0.93] |
| Q (mL/min) | 170 ([138-219] |
| R (WU) | 496 [381-654] |
| CFR | 3.0 [2.3-4.2] |
| IMR | 20 [12-28] |
|
Data are expressed as no. (%) or median [interquartile range]. |
|
Indices of microvascular function
Both studies—bolus thermodilution and continuous infusion thermodilution—were performed uneventfully in all of the patients. Table 2 shows the values of the measurements of microvascular function obtained with both techniques.
In the continuous infusion study, a median of absolute flow in the left anterior descending coronary artery of 170 mL/min [138-219 mL/min] was described while the median of microvascular resistance was 496 WU [381-654 WU].
A total of 18% of the patients (11/60) had a reduced CFR (CFR < 2) while 33% (20/60) showed elevated resistances (IMR ≥ 25).
The group of patients with microvascular dysfunction due to low CFR with normal IMR (7/60, 12%) with respect to cases with microvascular dysfunction due to high IMR with normal CFR (16/60, 27%) had a clinical profile with a lower mean age (61 ± 11 vs 66 ± 8), and a higher predominance of women (86% vs 58%) although this tendency was not statistically significant.
Table 3 shows the mean transit times (MTT) of bolus thermodilution tests. The cases with low CFR showed significantly shorter baseline MTT (0.48 ± 0.45 vs 1.13 ± 0.70), especially the subgroup of patients with low CFR and high Q (0.31 ± 0.15 vs 0.77 ± 0.68).
Table 3. Mean transit times obtained by bolus thermodilution
| Overall (N = 60) |
CFR < 2 (N = 11) |
CFR < 2 Q > 170 (N = 7) |
CFR < 2 Q < 170 (N = 4) |
|
|---|---|---|---|---|
| Baseline MTT | 1.13 ± 0.70 | 0.48 ± 0.45* | 0.31 ± 0.15* | 0.77 ± 0.68 |
| Hyperemic MTT | 0.36 ± 0.25 | 0.35 ± 0.28 | 0.25 ± 0.14 | 0.51 ± 0.41 |
|
Values (in seconds) are expressed as mean ± standard deviation. |
||||
Figure 2 shows data of coronary flow estimated by MTT measurement divided into 3 groups based on CFR and IMR results. We should mention that patients with low CFR without elevated resistances had significantly high resting flows and hyperemic flows without significant differences compared to the rest while in patients with low CFR and elevated resistances, the opposite phenomenon was described.
Figure 2. Baseline and hyperemic mean flow estimated based on the MTT (1/MTT) and grouped based on the CFR and IMR results. Values are expressed as s–1. CFR, coronary flow reserve; IMR, index of microvascular resistance; MTT, mean transit time.
* P < .05.
Endotypes
Figure 3A shows the percentages of endotypes based on the result of the acetylcholine test and the measurements of CFR and IMR. The most common pattern was microvascular dysfunction (24/60, 40%) followed by the normal study (14/60, 23%). In 20% of the patients (12/60), microvascular dysfunction overlapped with epicardial vasospasm while in 17% of the patients (10/60) isolated epicardial vasospasms were seen.
Figure 3. A: endotype-based classification. Values are expressed as absolute number and percentage.B: mean values of absolute flow and microvascular resistance grouped by endotypes. Q, absolute coronary blood flow; R, microvascular resistance.
* P < .05 with respect to normal study.
Table 4 shows how the mechanisms of vasomotor and microvascular dysfunction overlap in many cases.
Table 4.Results of the acetylcholine test and bolus thermodilution study (N = 60)
| Epicardial spasm | Microvascular spasm | IMR ≥ 25 | CFR < 2 | Endotype | Cases |
|---|---|---|---|---|---|
| − | − | − | − | Normal | 14 (23.3%) |
| + | − | − | − | Epicardial vasospasm | 10 (16.7%) |
| − | + | − | − | Microvascular dysfunction | 9 (15.0%) |
| − | − | + | − | Microvascular dysfunction | 5 (8.3%) |
| − | − | − | + | Microvascular dysfunction | 5 (8.3%) |
| − | + | + | − | Microvascular dysfunction | 3 (5.0%) |
| − | + | − | + | Microvascular dysfunction | 1 (1.6%) |
| − | + | + | + | Microvascular dysfunction | 1 (1.6%) |
| + | − | + | − | Mixed disorder | 6 (10.0%) |
| + | + | + | − | Mixed disorder | 2 (3.3%) |
| + | − | + | + | Mixed disorder | 2 (3.3%) |
| + | − | − | + | Mixed disorder | 1 (1.6%) |
| + | + | + | + | Mixed disorder | 1 (1.6%) |
|
Data are expressed as no. (%) |
|||||
The association between epicardial vasospasm and structural microvascular dysfunction (IMR ≥ 25) was the most prevalent combination in cases of mixed disorder (11/12). In turn, this endotype, in continuous thermodilution measurements, showed significant differences compared to the normal pattern, with reduced absolute flow values and elevated resistances (figure 3B) indicative of more serious structural and functional damage.
Concordance among the different indices of microvascular function
The ROC curve analysis of absolute coronary blood flow (Q) with respect to CFR < 2 determined an optimal cut-off value of 170 mL/min (a 64% sensitivity, and a 52% specificity) with an area underthe curve of 0.50 (95% confidence interval [95%CI], 0.33-0.66;P = .97), therefore showing no diagnostic utility.
Given the recent proposal to consider the cut-off value of CFR < 2.57,7 the analysis was performed using this threshold as the reference. In addition, no significant concordance was seen (area under the curve of 0.45 [95%CI, 0.30-0.61;P = .56]).
Regarding R with respect to IMR, an area under the curve of 0.67 (95%CI, 0.51-0.82;P = .04) was obtained, which was indicative of a weak yet significant diagnostic concordance (figure 4). The estimated optimal cut-off value was 435 WU, which was consistent with an 81% sensitivity and a 57% specificity. A total of 66% of cases with IMR ≥ 25 were categorized correctly using this index.
Figure 4. Analysis of the R cut-off value > 435 WU to predict IMR ≥ 25, and scatter plot showing the correlation between IMR and R. IMR, index of microvascular resistance; R, microvascular resistance.
The absence of an association between Q and CFR was confirmed in correlation tests (Spearman’s rho correlation coefficient= -0.02; 95%CI, -0.24-0.25;P = .99). However, a weak yet significant correlation was seen between Q and hyperemic MTT (Spearman’s rho= -0.28; 95%CI, -0.01-0.51;P = .04), and between R and IMR (Spearman’s rho= 0.28; 95%CI, 0.04-0.51;P = .03).
Complications
While the vasoreactivity test was being performed, 3 cases of transient bradycardia (5%) without clinical repercussions and 2 episodes of atrial fibrillation (4%) were reported, 1 of them self-limited while the other required sedation and electrical cardioversion. After the administration of regadenoson, most patients experienced some degree of discomfort, which was well-tolerated and reversed with the administration of 100 mg of intravenous theophylline. No other complications or adverse effects were reported.
DISCUSSION
This study confirms that a high percentage of patients with symptoms or signs of INOCA show microvascular dysfunction or vasospasm in invasive functional testing, and that it is feasible and safe to perform (figure 5).
Figure 5. Study design, endotype-based classification, and analysis using the ROC curve. AUC, area under the curve; CFR, coronary flow reserve; IMR, index of microvascular resistance; INOCA, ischemia with nonobstructive coronary artery disease; Q, absolute coronary blood flow; R, microvascular resistance.
* P < .05.
The percentage of patients with microcirculation or vasomotility alterations found in our study (77%) is consistent with former studies of patients with angina without obstructive coronary artery disease (64% to 89%8-11).
Vasoreactivity test
Some groups systematically use a dose of 200 µg of intracoronary acetylcholine to perform the vasoreactivity test; in our study, the high dose was established at 100 µg according to the COVADIS group, the CorMicA protocol, and the technical document of the Spanish Society of Cardiology Working Group on Cardiac Catheterization and Interventional Cardiology, which highlights its high sensitivity and specificity rates (90% and 99%, respectively).12 As a matter of fact, the high prevalence of positive results seen in our study in the acetylcholine test (60%) is similar to that reported in other series (57% to 71%13-15). In a recent study of 110 patients, Feenstra et al.11 revealed that 62% of the patients had a pathological acetylcholine test that confirmed the presence of epicardial vasospasm and microvascular spasm (36% and 26%, respectively).
In our study, the complications associated with the vasoreactivity test in our study are not very many: 2 cases of atrial fibrillation (4%), which is consistent with the incidence rate reported by the CorMIcA trial (5%).9
Prevalence of endotypes
The most common endotype in our patients was isolated microvascular dysfunction (40%), but not as much as in the CorMicA trial (52%). These differences could be explained by the discrepancy seen in the percentage of completely normal angiographies (22% in the CorMicA vs 63% in our study) due to the possible association between non-obstructive atheromatous disease and microvascular dysfunction.16,17
The prevalence of the remaining endotypes is similar to that reported in the CorMicA trial: isolated epicardial vasospasm (17% vs 17%), and mixed disorder (20% vs 21%). A recent meta-analysis that included 14 427 patients with INOCA also shows similar percentages.18
Indices of microvascular function obtained through bolus thermodilution
The analysis of the MTT obtained with this technique (figure 2), a parameter that correlates inversely with the direct measurement of coronary flow,19 reveals an interesting finding that is consistent with the data published by Nardone et al.20: patients with low CFR have 2 differentiated phenotypes based on the IMR. On the one hand, cases with reduced CFR and elevated resistances have normal baseline flow and low hyperemic flow, which would be indicative of an insufficient vasodilation response. However, in patients with normal resistances, a reduced CFR would be indicative of an abnormally elevated resting flow with hyperemic flow in the normal range. This phenomenon can also be observed in the analysis of patients with high Q (table 3) in whom a reduced CFR can be attributed to elevated baseline flow instead of an insufficient hyperemic response.
Therefore, this subgroup probably shows inefficient or dysregulated baseline myocardial flows. This characteristic, of indeterminate cause, could have important therapeutic implications like a lack of response to vasodilator drugs.
Indices of microvascular function obtained by continuous thermodilution
The continuous thermodilution technique has evolved to the point of quantifying Q and R with a microcatheter and specific software in a simple and precise fashion. The main advantages of this method are its independence from an operator, reproducibility, and induction of hyperemia with a physiological saline solution without the need for pharmacological agents.21-24 However, its main limitation is the lack of normal reference values.
In our study, the lack of a correlation between Q and CFR could be justified by the variations described of baseline myocardial flow. Estimating the CFR requires estimating the baseline coronary flow while Q is a measurement that is representative of hyperemic flow.
The weak concordance seen in this study between Q and hyperemic MTT and between R and IMR shows how difficult it is to establish valid cut-off values for patient comparison with these indices.
With an optimal cut-off value of R in our study of 435 WU (an 81% sensitivity, and a 57% specificity), a total of 66% of cases with IMR ≥ 25 were properly categorized with this index. This value is somewhat lower compared to the one shown by Rivero et al.,25 who analyzed 120 patients and found that an R > 500 WU properly categorized 80% of the cases with IMR ≥ 25.Konst et al.26 studied 84 patients with INOCA using both thermodilution techniques only to find no correlation between the Q-R combo and IMR.
The differences seen may be explained by the fact that the quantitative variability of Q and R values among individuals mostly depends on myocardial mass. However, in positron emission tomography studies, considerable ranges were seen even after adjusting for flow and resistance values for myocardial mass. Therefore, it has been speculated that the most plausible hypothesis is the natural variation of hyperemic myocardial perfusion among individuals.27
Therefore, indices like CFR estimated by continuous thermodilution and microvascular resistance reserve are currently in the pipeline. They correlate the absolute values of flow and resistance seen during hyperemia with those obtained at rest. Nonetheless,these new parameters will still need validation in futurestudies.28,29
Limitations
The data presented here should be interpreted while understanding that this is an observational, single-center study with a small sample size. Therefore, results may be biased by confounding factors associated with a study of this nature.
The left anterior anterior descending coronary artery was considered as the pre-specified target vessel. However, in the routine clinical practice, it may be appropriate to assess other arteries in the presence of negative tests and high clinical suspicion.1
The optimal sequence in invasive functional studies has not been established yet.1 In our case, we chose to perform the acetylcholine test first to minimize the instrumentation of the artery and avoid further guidewire-induced vasoreactivity. However, the spasm and symptoms seen during the provocation test, although transient, could interfere with subsequent measurements of microvascular function. The possibility of determining CFR by continuous thermodilution was established at the beginning of our study, and it was assumed that a comparison of the CFRs obtained with both techniques would have been more appropriate.
In most bolus thermodilution studies, intravenous adenosine is used to induce hyperemia. However, we chose regadenoson because it is easy to use, following our previous experience, and because evidence says it is equivalent to adenosine.30,31
Finally, we should not overlook that this is an invasive study so potential risks associated with the examination should be weighed in. To this date, however, conducting this study has not impacted prognosis.
CONCLUSIONS
The invasive study of coronary vasoreactivity and microcirculation is feasible and safe. These studies allow us to easily recognize different endotypes of patients with INOCA and help us optimize their treatment.
The analysis of CFR, IMR, and Q combined can unmask a subtype of microvascular dysfunction characterized by an abnormally high baseline coronary flow.
The new indices obtained by continuous thermodilution show low concordance with respect to the reference indices. Therefore, future studies will be required to determine the utility of this technique.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
All the authors contributed substantially to the study idea, design, and data mining process. In addition, all approved the manuscript final version for publication.
CONFLICTS OF INTEREST
None reported.
WHAT IS KNOWN ABOUT THE TOPIC?
- The invasive diagnosis of microvascular dysfunction and coronary vasospasm have proven useful to improve the quality of life of patients without obstructive coronary artery disease on the coronary angiography.
- Indices of microvascular dysfunction obtained by continuous thermodilution offer potential advantages since are they are independent from the operator, reproducible, and do not require pharmacologically induced hyperemia.
WHAT DOES THIS STUDY ADD?
- Invasive coronary functional diagnosis is feasible and safe and highlights the high prevalence of microcirculation and vasomotility alterations in patients without obstructive coronary artery disease.
- The combined analysis of the different indices may be useful to characterize cases with decreased CFR.
- Future studies are needed to establish the utility of microvascular function measurements obtained by continuous thermodilution.
REFERENCES
1. Kunadian V, Chieffo A, Camici PG, et al. An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. Eur Heart J. 2020;41:3504-3520.
2. Camici PG, Crea F. Coronary microvascular dysfunction. N Engl J Med. 2007;356:830-840.
3. Knuuti J, Wijns W, Saraste A, et al. ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41:407-477.
4. Ford TJ, Ong P, Sechtem U, et al. COVADIS Study Group. Assessment of Vascular Dysfunction in Patients Without Obstructive Coronary Artery Disease: Why, How, and When. JACC Cardiovasc Interv. 2020;13:1847-1864.
5. Xaplanteris P, Fournier S, Keulards DCJ, et al. Catheter-Based Measurements of Absolute Coronary Blood Flow and Microvascular Resistance: Feasibility, Safety, and Reproducibility in Humans. Circ Cardiovasc Interv. 2018;11:e006194.
6. Beltrame JF, Crea F, Kaski JC, et al., On Behalf of the Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for vasospastic angina. Eur Heart J. 2015;38:
2565-2568.
7. Demir OM, Boerhout CKM, de Waard GA, et al. Comparison of Doppler Flow Velocity and Thermodilution Derived Indexes of Coronary Physiology. JACC Cardiovasc Interv. 2022;15:1060-1070.
8. Suda A, Takahashi J, Hao K, et al. Coronary functional abnormalities in patients with angina and nonobstructive coronary artery disease. J Am Coll Cardiol. 2019;74:2350-2360.
9. Ford TJ, Stanley B, Good R, et al. Stratified medical therapy using invasive coronary function testing in angina: the CorMicAtrial. J Am Coll Cardiol. 2018;72:2841-2855.
10. Sara JD, Widmer RJ, Matsuzawa Y, Lennon RJ, Lerman LO, Lerman A. Prevalence of Coronary Microvascular Dysfunction Among Patients With Chest Pain and Nonobstructive Coronary Artery Disease. JACC Cardiovasc Interv. 2015;8:1445-1453.
11. Feenstra RGT, Boerhout CKM, Woudstra J, et al. Presence of Coronary Endothelial Dysfunction, Coronary Vasospasm, and Adenosine-Mediated Vasodilatory Disorders in Patients With Ischemia and Non obstructive Coronary Arteries. Circ Cardiovasc Interv. 2022;15:e012017.
12. Gutiérrez E, Gómez-Lara J, Escaned J, et al. Valoración de la función endotelial y provocación de vasoespasmo coronario mediante infusión intracoronaria de acetilcolina. Documento técnico de la ACI-SEC. REC Interv Cardiol. 2021;3:286-296.
13. Aziz A, Hansen HS, Sechtem U, Prescott E, Ong P. Sex-Related Differences in Vasomotor Function in Patients With Angina and Unobstructed Coronary Arteries. J Am Coll Cardiol. 2017;70:2349-2358.
14. Seitz A, Gardezy J, Pirozzolo G, et al. Long-Term Follow-Up in Patients With Stable Angina and Unobstructed Coronary Arteries Undergoing Intracoronary Acetylcholine Testing. J Am Coll Cardiol Interv. 2020;13:1865-1876.
15. Ong P, Athanasiadis A, Borgulya G, et al. Clinical usefulness, angiographic characteristics, and safety evaluation of intracoronary acetylcholine provocation testing among 921 consecutive white patients with unobstructed coronary arteries. Circulation. 2014;129:1723-1730.
16. Melikian N, Vercauteren S, Fearon WF, et al. Quantitative assessment of coronary microvascular function in patients with and without epicardial atherosclerosis. EuroIntervention. 2010;5:939-945.
17. Sharaf B, Wood T, Shaw L, et al. Adverse outcomes among women presenting with signs and symptoms of ischemia and no obstructive coronary artery disease: findings from the National Heart, Lung, and Blood Institute-sponsored Women’s Ischemia Syndrome Evaluation (WISE) angiographic corelaboratory. Am Heart J. 2013;166:134-141.
18. Mileva N, Nagumo S, Mizukami T, et al. Prevalence of Coronary Microvascular Disease and Coronary Vasospasm in Patients With Nonobstructive Coronary Artery Disease: Systematic Review and Meta-Analysis. J Am Heart Assoc. 2022;11:e023207.
19. De Bruyne B, Pijls NH, Smith L, et al. Coronary thermodilution to assess flow reserve: experimental validation. Circulation. 2001;104:2003-2006.
20. Nardone M, McCarthy M, Ardern CI, et al. Concurrently Low Coronary Flow Reserve and Low Index of Microvascular Resistance Are Associated With Elevated Resting Coronary Flow in Patients With Chest Pain and Nonobstructive Coronary Arteries. Circ Cardiovasc Interv. 2022;15:e011323.
21. Van’t Veer M, Adjedj J, Wijnbergen I, et al. Novel monorail infusion catheter for volumetric coronary blood flow measurement in humans: invitro validation. EuroIntervention. 2016;12:701-707.
22. Rivero F, Bastante T, Cuesta J, García-Guimaraes M, Maruri-Sánchez R, Alfonso F. Volumetric Quantification of Coronary Flow by Using a Monorail Infusion Catheter: Initial Experience. Rev Esp Cardiol. 2018;71:1082-1084.
23. Everaars H, de Waard GA, Schumacher SP, et al. Continuous thermodilution to assess absolute flow and microvascular resistance: validation in humans using [15O] H2O positron emission tomography. Eur Heart J. 2019;40:2350-2359.
24. Keulards DCJ, Van’t Veer M, Zelis JM, et al. Safety of absolute coronary flow and microvascular resistance measurements by thermodilution. EuroIntervention. 2021;17:229-232.
25. Rivero F, Gutiérrez-Barrios A, Gomez-Lara J, et al. Coronary microvascular dysfunction assessed by continuous intracoronary thermodilution: A comparative study with index of microvascular resistance. Int J Cardiol. 2021;333:1-7.
26. Konst RE, Elias-Smale SE, Pellegrini D, et al. Absolute Coronary Blood Flow Measured by Continuous Thermodilution in Patients With Ischemia and Nonobstructive Disease. J Am Coll Cardiol. 2021;77:728-741.
27. Fournier S, Keulards DCJ, van’t Veer M, et al. Normal values of thermodilution-derived absolute coronary blood flow and microvascular resistance in humans. EuroIntervention. 2021;17:e309-e316.
28. Gutiérrez-Barrios A, Izaga-Torralba E, Rivero Crespo F, et al. Continuous Thermodilution Method to Assess Coronary Flow Reserve. Am J Cardiol. 2021;141:31-37.
29. De Bruyne B, Pijls NHJ, Gallinoro E, et al. Microvascular Resistance Reserve for Assessment of Coronary Microvascular Function: JACC Technology Corner. J Am Coll Cardiol. 2021;78:1541-1549.
30. Federico P, Martínez L, Castelló T, Pomar F, Peris E. Regadenoson intravenoso frente a adenosina intracoronaria para la medida de la reserva fraccional de flujo. REC Interv Cardiol. 2019;1:77-82.
31. Gill GS, Gadre A, Kanmanthareddy A. Comparative efficacy and safety of adenosine and regadenoson for assessment of fractional flow reserve: A systematic review and meta-analysis. World J Cardiol. 2022;14:
319-328.
ABSTRACT
Introduction and objectives: Coronary calcification is one of the leading factors that affect negatively the safety and effectiveness of percutaneous coronary intervention. Several calcium modification techniques exist. However, there is a lack of randomized evidence on the therapy of choice in this scenario.
Methods: The ROLLERCOASTR is a prospective, multicenter, randomized clinical trial designed to compare the safety and efficacy profile of 3 plaque modification techniques in the moderate-to-severe coronary calcification setting: rotational atherectomy (RA), excimer laser coronary angioplasty (ELCA), and intravascular lithotripsy (IVL). The study primary endpoint is stent expansion evaluated by optical coherence tomography. An intention-to-treat analysis will be conducted with an alpha coefficient of 0.05 between the reference group (RA) and the remaining 2 groups (ELCA and IVL). An analysis of the study primary endpoint per protocol will be conducted for consistency purposes. If the non-inferiority hypothesis is confirmed, a superiority 2-sided analysis will be conducted. Both the clinical events committee and the independent core laboratory will be blinded to the treatment arm. Assuming an α error of 0.05, an β error of 0.2 (80% power), a margin of irrelevance (ε) of 7, and losses of 10% due to measurement difficulty or impossibility to complete the intervention, we estimate a sample size of 56 cases per group. The study secondary endpoints are device success, procedural success, crossover rate among the different techniques used, and the occurrence of major adverse cardiovascular events at 1-year follow-up.
Conclusions: The ROLLERCOASTR trial will evaluate and compare the safety and effectiveness of 3 plaque modification techniques: RA, ELCA, and IVL in patients with calcified coronary stenosis. This trial was registered at clinicaltrials.gov with identifier NCT04181268.
Keywords: Percutaneous coronary intervention. Calcified plaques. Laser. Lithotripsy. Rotational atherectomy. Optical coherence tomography.
RESUMEN
Introducción y objetivos: La calcificación coronaria es uno de los principales factores que inciden negativamente en la seguridad y la eficacia del intervencionismo coronario percutáneo. Existen varias técnicas de modificación del calcio, pero falta evidencia de estudios aleatorizados sobre la terapia de elección en este escenario.
Métodos: El ROLLERCOASTR es un estudio prospectivo, multicéntrico y aleatorizado, diseñado para comparar la seguridad y la eficacia de 3 técnicas de modificación de la placa en el contexto de calcificación coronaria moderada o grave: aterectomía rotacional (AR), aterectomía coronaria con láser láser excimer (ACLE) y litotricia intracoronaria (LIC). El objetivo primario es la expansión del stent evaluada mediante tomografía de coherencia óptica. Su análisis se hará por intención de tratar, con un α de 0,05 entre el grupo de referencia (AR) y cada uno de los otros grupos (ACLE y LIC). Se realizará también un análisis del objetivo primario por protocolo para mantener la coherencia. Si se confirma la hipótesis de no inferioridad, se realizará un análisis bilateral de superioridad. El comité de eventos clínicos y el laboratorio central independiente no conocerán la rama de tratamiento. Asumiendo un error α de 0,05, un error β de 0,2 (80% de potencia), un margen de irrelevancia (ε) del 7% y un 10% de pérdidas por dificultad de medición o imposibilidad de completar la intervención, se estima un tamaño de muestra de 56 casos en cada grupo. Los objetivos secundarios son el éxito del dispositivo, el éxito del procedimiento, la tasa de cruce entre técnicas y la presentación de eventos cardiovasculares adversos importantes al año de seguimiento.
Conclusiones: El estudio ROLLERCOASTR evaluará y comparará la seguridad y la eficacia, en pacientes con estenosis coronaria calcificada, de 3 técnicas de modificación de placa: AR, ACLE y LIC. Este ensayo se ha registrado en Clinicaltrials.gov: NCT04181268.
Palabras clave: Intervencionismo coronario percutáneo. Placas calcificadas. Láser. Litotricia. Aterectomía rotacional. Tomografía de coherencia óptica.
Abbreviations
DES: drug-eluting stent. ELCA: excimer laser coronary angioplasty. IVL: intravascular lithotripsy. OCT: optical coherence tomography. PCI: percutaneous coronary intervention. RA: rotational atherectomy.
INTRODUCTION
Percutaneous coronary intervention (PCI) with drug-eluting stent (DES) implantation is the most frequent mode of coronary revascularization.
Calcified coronary lesions pose a challenge to perform successful PCI.1 Coronary calcification impedes PCI by multiple mechanisms like limiting DES lesion crossing, altering the drug elution kinetics, and interfering with optimal stent expansion. In addition, inadequate stent expansion is a powerful predictor of stent thrombosis and restenosis.2-6 Coronary calcification also increases PCI-related procedural complications (dissection, perforation, myocardial infarction), and late adverse clinical outcomes like restenosis, repeat revascularization, stent fracture, and thrombosis.1 The optimal approach for the management of calcified stenosis requires taking into account the characteristics of the lesion, calcium distribution, and the mechanism of action of every plaque-modification device. In this regard, intracoronary imaging techniques such as intravascular ultrasound and optical coherence tomography (OCT) are essential not only to evaluate the severity of calcification and its pattern, but also to optimize stenting.7
Currently, plaque-modification techniques can be categorized into a) balloon-based technologies (cutting/scoring balloons, non-compliant and super high-pressure balloons, and intravascular lithotripsy (IVL), and b) non-balloon-based technologies (rotational atherectomy [RA], orbital atherectomy, and excimer laser coronary angioplasty [ELCA]).8,9
The widespread use of these techniques and devices has been limited due to the risk of complications, the operator’s experience, and the corresponding use of health resources. Over the past few decades, RA has been the therapy of choice for resistant calcified lesions. However, the development of new technologies such as IVL or the improvement of classical therapies such as ELCA has generated uncertainty on the optimal tool to modify calcified plaques as non-randomized comparisons between these techniques have been drawn.
The objective of this randomized trial is to assess the efficacy and safety profile of intensive plaque modification with RA, IVL or ELCA before DES implantation.
METHODS
Patients and study design
The ROLLERCOASTR (Rotational atherectomy, lithotripsy or laser for the treatment of calcified stenosis) is an investigator-initiated, multicenter, prospective, and randomized clinical trial that includes 6 large volume sites. Also, it includes men and women aged ≥ 18 years with a clinical indication for PCI (stable or unstable ischemic heart disease) in vessels with reference diameters ≥ 2.5 and ≤ 4.0 mm and moderate-to-severe calcification estimated by coronary angiography. The main study exclusion criteria are ST-segment elevation acute coronary syndrome as clinical presentation, cardiogenic shock, inability to tolerate dual antiplatelet therapy for, at least, 6 months for those who are not on oral anticoagulation, impossibility to obtain informed consent from the patient or conduct, at least, a 1-year follow-up.
Patients who meet all the inclusion criteria and none of the exclusion ones will be randomized on a 1:1:1 ratio to either lesion preparation with RA, ELCA or IVL. Randomization will on a web-based platform. The complete inclusion and exclusion criteria are shown on table 1 while the study flowchart is described on figure 1.
Table 1. Study inclusion and exclusion criteria
| Inclusion criteria |
|---|
| ≥ 18 years old |
| Diameter stenosis ≥ 70% or fractional flow reserve < 0.8/non-hyperemic indexes < 0.89 |
| Reference vessel diameter ≥ 2.5 and ≤ 4 mm |
| Moderate or severe calcification estimated by coronary angiography |
| Patients with stable coronary artery disease or non-ST-segment elevation acute coronary syndrome |
| Culprit lesions at native vessels or coronary bypasses |
| Exclusion criteria |
| Inability to tolerate a 6-month course of dual antiplatelet therapy in patients naïve to oral anticoagulation |
| ST-segment elevation acute coronary syndrome |
| Cardiogenic shock |
| Impossibility to obtain informed consent from the patient or his legal representative |
| Impossibility to conduct, at least, a 1-year follow-up |
Figure 1. Study flowchart. ELCA, excimer laser coronary angioplasty; EP, endpoint; MACE, major adverse cardiovascular events; OCT, optimal coherence tomography; PCI, percutaneous coronary intervention.
Study primary and secondary endpoints
The objective of this study is to evaluate and compare the results of RA, IVL, and ELCA for the management of calcified coronary lesions. This comparison will be made by assessing the angiographic and OCT findings after the implementation of these plaque modification techniques, and DES implantation and optimization.
The primary endpoint is the comparison between RA (reference group) vs ELCA and RA vs IVL in the percentage of stent expansion measured using OCT. As secondary endpoints we’ll be analyzing the device success (successful stent implantation with minimum stent area ≥ 5.5 mm2, final TIMI grade-3 flow, and no need for another plaque preparation strategy), procedural success (device success and no severe procedural complications like cardiovascular death, perioperative target vessel myocardial infarction, need for new target lesion revascularization, stent thrombosis, stroke or vessel perforation with extravasation [types II or III]), crossover from the assigned plaque modification technique to a different one, and occurrence of major adverse cardiovascular events at 1-year follow-up (cardiovascular death, target vessel myocardial infarction, target lesion revascularization or stent thrombosis). We’ll also be analyzing device success regarding the type of calcified plaque (concentric, eccentric, calcium nodule). The study primary and secondary endpoints are shown on table 2.
| Primary endpoint |
|---|
| Percentage of stent expansion measured by OCT |
| Key secondary endpoints |
| Device success (successful stent implantation with minimum stent area ≥ 5.5 mm2, final TIMI grade-3 flow, and no need for another plaque preparation strategy) |
| Device success depending on the type of the calcific plaque: concentric, eccentric or nodular |
| Procedural success (device success in the absence of procedural severe complications) |
| Crossover from the assigned plaque modification technique to a different one |
| 1 year-MACE (CD, TVMI, TLR or ST) |
|
CD, cardiac death; MACE, major adverse cardiovascular events; OCT, optical coherence tomography; ST, stent thrombosis; TLR, target lesion revascularization; TVMI, target vessel myocardial infarction. |
Devices
- – RA: Rotablator or RotaPro System (Boston Scientific, Unites States).
- – Coronary laser: Coronary laser-emitting device (CVX-300 ELCA System, Spectranetics Inc., United States).
- – Intracoronary lithotripsy: Shockwave System, (Shockwave Medical, United States).
- – OCT system: OCT Imaging system (Abbott Vascular, United States)
- – Stents: new-generation DES are mandatory (those currently being used in participant centers during the inclusion period).
Procedure
The angioplasty will be performed following the recommendations established by the current clinical practice guidelines on the management of coronary revascularization.10 After crossing the lesion with the angioplasty guidewire, a first OCT assessment should be performed. If necessary, balloon dilatation is allowed to cross the OCT catheter. After this first OCT pullback, the use of a plaque modification technique will be required (RA, laser or lithotripsy) on a randomized basis. Afterwards, a second OCT assessment is advised to analyze the effects of the therapy. Finally, the angioplasty will be completed with the implantation of a new-generation DES. Pre or postdilatation will be left to the operator’s criterion. After stenting (in the absence of postdilatation) or after the last postdilatation (if performed), a final OCT pullback will be performed to assess the final stent expansion.
Rotational atherectomy technique
The lesion will be crossed using the RotaWire (Boston Scientific, Unites States) directly or microcatheters or coaxial balloons. The RotaWire type (RotaWire Extra Support and RotaWire Floppy) will be used based on the characteristics of the plaque, the support required, and the operator’s preferences. Afterwards, the rotational atherectomy technique will be used based on the current recommendations.11 A 0.5:0.6 ratio between the burr and the vessel is advised. The rotational speed recommended is between 135 000 rpm and 180 000 rpm. Decelerations > 5000 rpm should be avoided. The burr should be advanced gradually with easy back-and-forth moves. Rotablation time should be < 20 seconds with pauses in between each cycle. Once rotablation has been performed, the burr should be removed with the Dynaglide mode on.
Intracoronary lithotripsy technique
The Shockwave balloon (Shockwave Medical, Inc., United States) is a 12 mm-long angioplasty balloon with 2.5 mm to 4 mm diameters. It can be mounted over a 0.014 in guidewire. Mechanical energy is transmitted to the lesion when the Shockwave balloon contacts the artery intima layer and cracks superficial and deep calcium layers. Therefore, the Shockwave balloon/reference vessel diameter ratio should be 1:1.12 Performing an OCT assessment prior to selecting the size of the balloon is also advised. Predilatation with balloons of smaller diameters is allowed to facilitate the passage of the lithotripsy balloon.
Once the Shockwave balloon is on the lesion, it is inflated at a pressure of 4 atm . Up to 80 pulses per balloon can be administrated (8 runs of 10 pulses). After every run (≤ 10 pulses), the Shockwave balloon is inflated at 6 atm and, after deflation, a new cycle can be applied if necessary. A minimum of 20 pulses per lesion is advised.
Laser technique
The size of the ELCA catheter will be selected considering the diameter of the target vessel on a 0.5-0.6 ratio with respect to its diameter.13 However, 0.9 mm catheters will be prioritized because of their greater crossing capabilities and capacity to emit laser energy with greater fluence (80 mJ/mm2) at the maximum pulse repetition rate (80 Hz). Regarding the device settings, it is recommended to start by applying a 60 mJ/mm2 fluence and a 60 Hz pulse repetition frequency that can go up to 80 mJ/mm2 and 80 Hz based on the operator’s criterion. Energy pulses will be released while the catheter slowly moves forward through the lesion at a rate of 0.5 mm/s, thus allowing proper energy absorption and plaque modification. Retrograde application is also feasible, especially in severe lesions with antegrade resistance. Saline-infusion technique is advised. Both blood and iodinated contrast contain non-aqueous cellular macromolecules like proteins that absorb most of the energy released by the laser creating microbubbles that increase the chances of traumatic dissection.14 On the contrary, the saline solution facilitates the passage of light from the tip of the catheter to the tissue without interferences or microbubbles at that level. Therefore, the saline solution infusion technique is used to safely control the energy that is being released, and minimize the risk of dissection.15 In order to wash out the blood from the catheter-based tissue interface the catheter needs to be properly intubated and the saline solution properly infused during laser application. The application of laser to blood or contrast is allowed in selected cases of uncrossable or undilatable lesions and left to the operator’s criterion.16 At the end of the procedure, parameters like the number of pulses administered, the time of therapy, fluence, and repetition rate will need to be collected.
Crossover
Combination of several plaque modification techniques is permitted as they have shown to be complementary in some cases.17,18 If a different plaque preparation technique is required, the technique should be changed based on why the first technique failed (table 3). This switch is consistent with the routine clinical practice. All the material and techniques used will be registered for further analysis.
Table 3. Crossover of plaque modification techniques
| Failed early technique | Reason for failure | 2nd technique |
|---|---|---|
| Rotational atherectomy | Uncrossable lesion with the rotablation olive-shaped burr | ELCA |
| Undilatable lesion (suboptimal balloon expansion after rotablation) | Lithotripsy | |
| Lithotripsy | Uncrossable lesion with Shockwave balloon (despite predilatation, if necessary) | Rotational atherectomy |
| Undilatable lesion (suboptimal balloon expansion after lithotripsy) | ELCA | |
| ELCA | Uncrossable lesion with ELCA | Rotational atherectomy |
| Undilatable lesion (suboptimal balloon expansion after ELCA) | Lithotripsy | |
|
ELCA, excimer laser coronary angioplasty. |
||
Optical coherence tomography image acquisition and stent optimization protocol
Intravascular OCT is performed using a commercially available system (the ILUMIEN OPTIS, OPTIS Integrated, OPTIS Mobile systems, OPTISIntegrated Next, OPTISMobile Next Abbott Vascular) that incorporates a rapid exchange catheter (Dragonfly OPTIS, Dragonfly OpStar Imaging Catheter; Abbott Vascular) and an integrated pullback system (18-36 mm/s). It acquires images at high axial resolution (~15 μm) with blood displacement. A total of 3 pullbacks are advised before and after using the plaque modification technique (to describe the calcified lesion and the effects of each modality over it, respectively), and optimizing the DES implanted. The automated OCT-angiography co-registration (where available) will be used, and recommendations for PCI guidance with OCT19 will be left to the operator’s criterion. Stent expansion can be estimated using 2 methods (figure 2): 1) dual method: it identifies the stented region and splits it in half. Minimum lumen expansion in the stented area (EXP) is estimated for each half (minimum stent area in each segment divided by the proximal or distal reference area x 100). The center point can be moved by the user (both the minimum stent area and the EXP recalculate automatically); 2) tapered mode: reference lumen profile is estimated based on the distal and proximal reference frame mean diameter and side branch mean diameter in between. The software automatically displays the minimum stent area and identifies the frame with the minimum lumen expansion in the stented area (EXP). A colored expansion indicator automatically pops up when a stent is detected. Automatic detection: minimum stent area frame/Automatic detection of minimum expansion frame (EXP).
Figure 2. Stent expansion estimate by optical coherence tomography. EXP, stented area. MSA, minimum stent area. Modified with permission from Abbott Vascular from User Manual of Ultreon 1.0, and User Instructions of AptiVue Software.
With stent lengths > 50 mm, the dual method is preferred. With stent lengths < 50 mm the tapered method is often used. If the dual method is used, the stent expansion percentage of both segments is recorded being considered for analysis the lowest of the 2.
Follow-up and clinical definitions
In-hospital and follow-up outcomes were prespecified in the online database, complied with the requirements set forth by the Spanish Data Protection Act, and were only accessible to participant operators and study coordinators.
After each PCI, electrocardiographic and cardiac biomarker seriation will be performed. Clinical assessment will be conducted 1, 6, 12 months after PCI. Angiographic follow-up will be only clinically driven in patients with new symptoms, ventricular function worsening or new ischemia in non-invasive tests.
Calcification is defined as moderate if radiopacities are noted only during the cardiac cycle before contrast injection, and severe if radiopacities are noted without cardiac motion before contrast injection often compromising both sides of the arterial lumen.
Device success is defined as successful stent implantation with minimum stent areas ≥ 5.5 mm2 by OCT, final TIMI grade-3 flow, and no need for another plaque preparation strategy.
Procedural success is defined as device success and no severe procedural complications: cardiovascular death, perioperative target vessel myocardial infarction, need for new target lesion revascularization, stent thrombosis, stroke or vessel perforation with extravasation [types II or III]).
Other procedural complications included ventricular arrhythmias or hemodynamic instability during PCI, major bleeding (bleeding requiring transfusion, vasopressors, surgery or percutaneous intervention), and flow limiting dissection.
Major cardiovascular adverse events include cardiovascular death, target vessel myocardial infarction, stent thrombosis or target lesion revascularization. All deaths were considered cardiac unless other specific causes were documented. Myocardial infarction was defined according to the current recommendations made,20 and only those associated with the targer lesion, perioperative or at follow-up were considered. Target lesion revascularization or stent thrombosis were defined according to the criteria established by the Academic Research Consortium.21
Primary outcome assessment will be conducted in a central core laboratory by looking at the OCT imaging after stenting. All medical data will be codified anonymously and stored, and confidentiality will be protected at any time in observance of the current legislation. Both the clinical events committee and the independent core laboratory will be blinded to the treatment arm.
Secondary outcome assessment will be performed by assessing both the angiography and the OCT in a central core laboratory and through on-site or phone clinical follow-up sessions with the patients.
Statistical considerations
Sample size determination
This is a non-inferiority study. We expect to obtain similar outcomes regarding stent expansion using rotational atherectomy, laser, and intracoronary lithotripsy. The sample size was estimated based on the design of the trial and the results of former studies.22-24 There are no standard criteria to define stent expansion in the routine clinical practice. In a recent expert consensus document, stent expansion > 80%19 was considered appropriate. However, most former studies did not reach this threshold. In the ILUMIEN II trial, the mean stent expansion measured by OCT was 72.8% with a standard deviation of 12.6.24 To calculate the size of the sample, we assume an α error of 0.05 and a β error of 0.2 (80% power), a margin of irrelevance (ε) of 7, and losses of 10% due to measurement difficulty or impossibility to complete the intervention. With these parameters we estimate a sample size of 56 cases per group.
Statistical analysis
The study primary endpoint analysis will be conducted by lesion and intention-to-treat with a 1-sided Student t test and an alpha coefficient of 0.05 between the reference group and the other groups (ELCA, and IVL). An analysis of the primary endpoint per protocol will be conducted and presented for consistency purposes. If the hypothesis of non-inferiority is confirmed, a 2-sided superiority analysis will be conducted. Clinical endpoints will be analyzed by patient.
Quantitative variables following a normal distribution will be expressed as median ± standard deviation. Those not following such distribution will be expressed as median and minimum and maximum values. Qualitative variables will be expressed as absolute values and frequencies.
P values < .05 will be considered statistically significant, and the 95% confidence interval of the study variables will be estimated. The Kolgomorov-Smirnov test will be used to confirm the adjustment of variables to normal distribution. Regarding mean comparisons, the Student t test or the non-parametric Mann-Whitney U test (in case of qualitative dichotomous variables), and the ANOVA test or the non-parametric Kruskal Wallis test (in case of qualitative non-dichotomous variables) will be used. Regarding the bivariate analysis of qualitative variables, the chi-square test or Fisher’s exact test will be used. If necessary, the linear correlation among the different quantitative variables will be performed using Pearson correlation coefficient or Spearman’s correlation.
Regarding the multivariate analysis, the Cox regression analysis with forward, stepwise selection will be used drawing event-free survival curves using the Kaplan-Meier estimator. Variables will be considered potential predictors of risk in the multivariate model in the presence of a statistically significant correlation in the univariate analysis or a trend towards significance. The SPSS statistical software (version 20.0, SPSS Inc) will be used for all the estimates.
Organization and ethical concerns
The study protocol has been approved at each participant center by its internal ethics committee. All patients will have to give their informed written consent prior to their participation. The study is an investigator-initiated trial and follows the good clinical practice guidelines applicable to epidemiological studies. The rights and integrity of participants shall be guaranteed at all time while data confidentiality shall be safeguarded in observance of EU directives, the Declaration of Helsinki, as well as local rules and regulations. The ROLLERCOASTR trial is registered at clinicaltrials.gov wit identifier NCT04181268. The study promoter is Fundación EPIC. The study is supported by unrestricted grants from Fundación EPIC. The steering committee is the trial main decision-making committee and has final word on the medical and scientific approach to the trial. The clinical events committee includes interventional cardiologists who don’t participate in the trial and are blinded to the randomized therapy. The clinical events committee will be responsible for developing specific criteria for the adjudication of the study clinical events and endpoints as per protocol. All members of the clinical events committee will be blinded to the study primary outcomes.
DISCUSSION
At least a third of all coronary lesions requiring PCI show significant calcification.9 As a matter of fact, this is probably one of the greatest challenges interventional cardiologists face to this date. Different tools are available to prepare calcified plaques. These techniques are increasingly used in the routine clinical context based on the operator’s experience or availability25 since there are barely any comparative studies on this regard.
The role of rotational atherectomy is to facilitate stenting in calcified non-dilatable lesions. The technology has evolved for over 20 years now, and lots of patients have been treated with it. The setback is that it has a longer learning curve compared to other plaque modification techniques and requires a specific guidewire. The evidence available on RA in the calcified lesion setting shows higher procedural success rates compared to conventional or modified balloons with almost the same clinical outcomes. However, even the most recent trials have important limitations as a limited use of intracoronary imaging techniques and new-generation DES.22,23
The arrival of laser to treat atherosclerosis goes back to the 1980s to treat lower limb ischemia at the beginning, and then coronary artery disease.26 However, both catheters and the techniques were rudimentary, and complications were a common thing. The early randomized clinical trials that compared ELCA to RA or balloon angioplasty (before the stent era) did not show favorable outcomes.27 The refinement of this technology followed by the introduction of safe laser-based techniques has improved its results. However, no direct comparisons have been drawn over the past few years. Although, traditionally, severe calcification has been a non-favorable scenario for ELCA, this technique has repeatedly obtained good results in settings in which calcium is a common finding: balloon failure (uncrossable or undilatable lesions), in-stent restenosis, underexpanded stents or chronic total coronary occlusions.13 Excimer laser releases energy in the UV range in very short pulses (nanoseconds). Billions of molecules per pulse are broken. Absorption depth is 50 µm, thus reducing the risk of collateral tissue damage (compared to previous infrared lasers). Laser ablates the atherosclerotic material mediated by 3 different mechanisms: photochemical (fracture of molecular bonds): the UV light pulse hits the plaque and is highly absorbed with each photon generated carrying sufficient energy to break molecular bonds; photothermal (tissue vaporization): molecular bonds also vibrate during the absorption process resulting in heat. Intracellular water is vaporized leading to cell rupture and the creation of a vapor bubble, and photokinetic (clearance of byproducts): the rapid expansion and collapse of the vapor bubble further breaks down the plaque, but it also helps clear byproducts of ablation like water, gases, and small particles. Laser effect is amplified especially when it acts directly on blood or a contrast agent. Therefore, to reduce the risk of coronary artery dissection, laser ablation is often performed during the continuous infusion of saline solution.13 One advantage of laser is its short learning curve. It can be used through conventional 0.014 in guidewires in a rapid-exchange fashion and conventional 6-Fr guiding catheters. In addition, most of these particles are small enough to be cleared by the reticuloendothelial system, thus minimizing the risk of distal microembolization (1 more advantage compared to other plaque modification techniques).13
Lithotripsy is the latest technology that has become available to treat heavily calcified lesions. It emits pulsatile mechanical waves through emitters integrated in a semi-compliant balloon that is initially inflated at 4 atm. Afterwards, energy pulses are applied, and the vibrations produced interact with the atherosclerotic plaque breaking down both the superficial and deep calcium deposits.9 This effect on deep calcium deposits is one of the greatest advantages of lithotripsy over other techniques. Also, this technique learning curve is short since it’s based on a well-known coronary balloon technology. The DISRUPT CAD trials12 have demonstrated the safety and efficacy profile of this technique treating heavily calcified lesions and its use has grown exponentially ever since. The main limitation of this technique is that, as it is a balloon-based technology with a smaller diameter of 2.5 mm, extremely tight stenoses can hamper its use as a first-line therapy, thus needing predilatation with lower profile balloons, and even with RA17 or laser18 combined to overcome this problem.
Intracoronary imaging modalities allow more accurate assessments of coronary artery disease compared to conventional angiography and give us essential information for PCI planning. This is particularly relevant during the management of calcified and complex lesions impacting the results of the angioplasty and the patient’s prognosis28 by optimizing DES implantation, thus leading to better stent expansion, vessel wall apposition, and eventually a greater luminal area. The OCT has greater spatial resolution9 compared to the intracoronary ultrasound and has proven useful showing the effect of plaque modification therapies and stent optimization. All these reasons and the lack of use of intracoronary imaging techniques in previous plaque modification techniques has led us to using OCT to assess the study primary endpoint: percentage of stent expansion.
The ROLLERCOASTR trial will compare the 3 strategies most used in the routine clinical practice to treat lesions with moderate-to-severe calcifications. In addition, it will provide us with information on the effect of each of these strategies and the specific settings where they can be more useful. To this end, an intracoronary imaging study with an OCT will be performed to know the specific substrate of calcification and the type of plaque on which the therapy is performed as well as the effects this therapy will have. The study hypothesis is that the 3 modalities complement each other and have different effects depending on the characteristics of the lesion. At manuscript submission, a total of 135 patients have been included.
CONCLUSIONS
The ROLLERCOASTR is a prospective, multicenter, randomized clinical trial designed to compare the safety and efficacy profile of 3 plaque modification techniques in the moderate-to-severe coronary calcification setting: RA, ELCA, and IVL. The study primary endpoint is stent expansion evaluated by OCT. The secondary endpoints are device success, procedural success, crossover rate among techniques, and the occurrence of major adverse cardiovascular events at 1-year follow-up (cardiac death, target vessel myocardial infarction, need for new target lesion revascularization or stent thrombosis). We will also be describing the effects of the 3 imaging modalities in calcified lesions with OCT. Enrollment will end in 2023.
FUNDING
The study is supported by unrestricted grants from Fundación EPIC.
AUTHORS’ CONTRIBUTIONS
A. Jurado-Román: conceptualization, original draft, review, and editing. A. Gómez-Menchero, I.J. Amat-Santos, J. Caballero-Borrego, S. Ojeda, and R. Ocaranza-Sánchez: drafting, review, and editing. S. Jiménez-Valero, G. Galeote, and R. Moreno: conceptualization, drafting, review, and editing.
CONFLICTS OF INTEREST
S. Ojeda and R. Moreno are associate editors of REC: Interventional Cardiology. The journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. S. Ojeda has received consulting fees and participated on Medtronic and Edwards Lifesciences Data Safety Monitoring Board or Advisory Boards, and payment or honoraria for lectures, presentations, speakers bureaus, manuscript drafting or educational events organized by Philips, Biomenco, and World Medica. R. Moreno has received payment or honoraria for lectures, presentations, speakers bureaus, manuscript drafting or educational events organized by Medtronic Inc, Boston scientific, Abbott vascular, Biosensors, Biotronik, Edwards Lifesciences, AMGEN, Astra Zeneca, Daiichi Sankyo New Vascular Therapies, and Biosensors. A. Jurado-Román has received payment or honoraria for lectures, presentations, speakers bureaus, manuscript drafting or educational events organized by Boston Scientific, Shockwave, Philips, Biotronik, Biomenco, Abbott, and Medtronic. A. Gómez-Menchero, J. Caballero-Borrego, R. Ocaranza, G. Galeote, and S. Jiménez-Valero declared no conflicts of interest whatsoever. I. Amat-Santos has received payment or honoraria for lectures, presentations, speakers bureaus, manuscript drafting or educational events organized by Boston Scientific.
WHAT IS KNOWN ABOUT THE TOPIC?
- Coronary calcification worsens the safety and efficacy of percutaneous coronary intervention.
- Several calcium modification techniques are currently available. However, there is a lack of randomized evidence on the therapy of choice in this scenario.
WHAT DOES THIS STUDY ADD?
- The ROLLERCOASTR is a multicenter randomized study that compared 3 advanced plaque modification techniques in the coronary calcification setting: rotational atherectomy, excimer laser, and lithotripsy.
- The study primary endpoint is stent expansion evaluated by optical coherence tomography.
- Secondary endpoints are device success (overall and depending on the type of calcific plaque), procedural success, crossover rate, and the occurrence of major adverse cardiovascular events at 1-year follow-up.
REFERENCES
1. Bourantas CV, Zhang YJ, Garg S, et al. Prognostic implications of coronary calcification in patients with obstructive coronary artery disease treated by percutaneous coronary intervention: a patient-level pooled analysis of 7 contemporary stent trials. Heart Br. 2014;100:1158-1164.
2. Mori S, Yasuda S, Kataoka Y, Morii I, Kawamura A, Miyazaki S. Significant association of coronary artery calcification in stent delivery route with restenosis after sirolimus-eluting stent implantation. Circ J. 2009;73:1856-1863.
3. Tzafriri AR, Garcia-Polite F, Zani B, et al. Calcified plaque modification alters local drug delivery in the treatment of peripheral atherosclerosis. J Control Release. 2017;264:203-210.
4. Wiemer M, Butz T, Schmidt W, Schmitz KP, Horstkotte D, Langer C. Scanning electron microscopic analysis of different drug eluting stents after failed implantation: from nearly undamaged to major damaged polymers. Catheter Cardiovasc Interv. 2010;75:905-911.
5. Kobayashi Y, Okura H, Kume T, et al. Impact of target lesion coronary calcification on stent expansion. Circ J. 2014;78:2209-2214.
6. Lee MS, Shah N. The Impact and Pathophysiologic Consequences of Coronary Artery Calcium Deposition in Percutaneous Coronary Interventions. J Invasive Cardiol. 2016;28:160-167.
7. di Mario C, Koskinas KC, Räber L. Clinical Benefit of IVUS Guidance for Coronary Stenting: The ULTIMATE Step Toward Definitive Evidence? J Am Coll Cardiol. 2018;72:3138-1341.
8. Barbato E, Shlofmitz E, Milkas A, Shlofmitz R, Azzalini L, Colombo A. State of the art: evolving concepts in the treatment of heavily calcified and undilatable coronary stenoses - from debulking to plaque modification, a 40-year-long journey. EuroIntervention. 2017;13:696-705.
9. De Maria GL, Scarsini R, Banning AP. Management of Calcific Coronary Artery Lesions: Is it Time to Change Our Interventional Therapeutic Approach? JACC Cardiovasc Interv. 2019;12:1465-1478.
10. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
11. Barbato E, Carrié D, Dardas P, et al. European expert consensus on rotational atherectomy. EuroIntervention. 2015;11:30-36.
12. Kereiakes DJ, Di Mario C, Riley RF, et al. Intravascular Lithotripsy for Treatment of Calcified Coronary Lesions: Patient-Level Pooled Analysis of the Disrupt CAD Studies. JACC Cardiovasc Interv. 2021;14:1337-1348.
13. Rawlins J, Din JN, Talwar S, O’Kane P. Coronary Intervention with the Excimer Laser: Review of the Technology and Outcome Data. Interv Cardiol. 2016;11:27-32.
14. Baumbach A, Haase KK, Rose C, Oberhoff M, Hanke H, Karsch KR. Formation of pressure waves during in vitro excimer laser irradiation in whole blood and the effect of dilution with contrast media and saline. Lasers Surg Med. 1994;14:3-6.
15. Tcheng JE. Saline infusion in excimer laser coronary angioplasty. Semin Interv Cardiol SIIC. 1996;1:135-41.
16. Latib A, Takagi K, Chizzola G, et al. Excimer Laser LEsion modification to expand non-dilatable stents: the ELLEMENT registry. Cardiovasc Revasc Med. 2014;15:8-12.
17. Jurado-Román A, Gonzálvez A, Galeote G, Jiménez-Valero S, Moreno R. RotaTripsy: Combination of Rotational Atherectomy and Intravascular Lithotripsy for the Treatment of Severely Calcified Lesions. JACC Cardiovasc Interv. 2019;12:e127-129.
18. Jurado-Román A, García A, Moreno R. ELCA-Tripsy: Combination of Laser and Lithotripsy for Severely Calcified Lesions. J Invasive Cardiol. 2021;33:E754-755.
19. Räber L, Mintz GS, Koskinas KC, et al. Clinical use of intracoronary imaging. Part 1: guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention. 2018;14:656-677.
20. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol. 2018;72:2231-2264.
21. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation. 2007;115:2344-2351.
22. de Waha S, Allali A, Büttner HJ, et al. Rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: Two-year clinical outcome of the randomized ROTAXUS trial. Catheter Cardiovasc Interv. 2016;87:691-700.
23. Abdel-Wahab M, Toelg R, Byrne RA, et al. High-Speed Rotational Atherectomy Versus Modified Balloons Prior to Drug-Eluting Stent Implantation in Severely Calcified Coronary Lesions. Circ Cardiovasc Interv. 2018;11:e007415.
24. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of Stent Expansion Guided by Optical Coherence Tomography Versus Intravascular Ultrasound: The ILUMIEN II Study (Observational Study of Optical Coherence Tomography [OCT] in Patients Undergoing Fractional Flow Reserve [FFR] and Percutaneous Coronary Intervention). JACC Cardiovasc Interv. 2015;8:1704-1714.
25. Romaguera R, Ojeda S, Cruz-González I, Moreno R. Spanish Cardiac Catheterization and Coronary Intervention Registry. 30th Official Report of the Interventional Cardiology Association of the Spanish Society of Cardiology (1990-2020) in the year of the COVID-19 pandemic. Rev Esp Cardiol. 2021;74:1095-1105.
26. Bittl JA, Sanborn TA, Tcheng JE, Siegel RM, Ellis SG. Clinical success, complications, and restenosis rates with excimer laser coronary angioplasty. The Percutaneous Excimer Laser Coronary Angioplasty Registry. Am J Cardiol. 1992;70:1533-1539.
27. Appelman YE, Piek JJ, Strikwerda S, et al. Randomised trial of excimer laser angioplasty versus balloon angioplasty for treatment of obstructive coronary artery disease. Lancet. 1996;347:79-84.
28. Ahn JM, Kang SJ, Yoon SH, et al. Meta-analysis of outcomes after intravascular ultrasound-guided versus angiography-guided drug-eluting stent implantation in 26,503 patients enrolled in three randomized trials and 14 observational studies. Am J Cardiol. 2014;113:1338-1347.
ABSTRACT
Introduction and objectives: Percutaneous coronary interventions (PCI) of chronic total occlusions (CTO) are long procedures where many patients suffer moderate-to-high level anxiety and pain. Virtual reality (VR) has proven capable of reducing procedural pain and anxiety in many medical procedures. The objective of this study is to demonstrate that the use of VR during CTO PCI reduces anxiety and pain compared to conventional routine clinical practice.
Methods: Randomized, controlled, open-label, superiority trial clinical trial with 2 parallel arms including 58 patients with a scheduled CTO PCI randomized on a 1:1 ratio to VR during the procedure or conventional management. In both arms, the administration of anxiolytic drugs will be left to the lead operator’s discretion and based on the degree of anxiety o pain perceived. The remaining actions for the management of pre- and perioperative anxiety will be identical in both arms. The primary endpoint will be the maximum level of anxiety perceived by the patient. Secondary endpoints will be the level of patient-perceived pain, the need for intraoperative anxiolytic drug therapy, dose of drug administered, and satisfaction with the VR goggles.
Results: The results of this study will add significant knowledge on the utility of VR regarding anxiety reduction in CTO PCIs.
Conclusions: The ReViCTO trial is the first randomized clinical trial to use VR during a PCI CTO. Its results will show the utility of this technology to reduce anxiety and pain in PCIs performed on CTOs.
Diseño del ensayo registrado en ClinicalTrials.gov (identificador: NCT05458999).
Keywords: Chronic total coronary occlusion Virtual reality Anxiety
RESUMEN
Introducción y objetivos: Las intervenciones coronarias percutáneas (ICP) sobre oclusiones totales crónicas (OTC) son procedimientos largos en los que muchos pacientes sufren ansiedad y dolor. La realidad virtual ha demostrado reducir el dolor y la ansiedad en muchos procedimientos médicos. Nuestro objetivo es demostrar que el uso de la realidad virtual durante la ICP de OTC reduce la ansiedad y el dolor en comparación con la práctica convencional.
Métodos: Ensayo clínico aleatorizado, controlado, abierto y de superioridad con 2 grupos paralelos en el que 58 pacientes con una ICP de OTC programada serán aleatorizados 1:1 al uso de realidad virtual frente al tratamiento convencional. La administración de fármacos ansiolíticos será a criterio del operador principal y en función del grado de ansiedad o dolor percibido. El resto de las acciones para el tratamiento de la ansiedad serán idénticas en ambos grupos. El objetivo primario será el nivel máximo de ansiedad percibido por el paciente. Los objetivos secundarios serán el nivel de dolor percibido por el paciente, la necesidad de tratamiento farmacológico ansiolítico, la dosis de fármaco administrada y la satisfacción con la realidad virtual.
Resultados: Los resultados de este estudio añadirán conocimientos importantes sobre la utilidad de la realidad virtual en la reducción de la ansiedad en los procedimientos de ICP de OTC.
Conclusiones: El ensayo ReViCTO es el primer ensayo clínico aleatorizado que utiliza la realidad virtual durante la ICP en OTC. Sus resultados mostrarán la utilidad de esta tecnología para reducir la ansiedad y el dolor en esta intervención.
Diseño del ensayo registrado en ClinicalTrials.gov (identificador: NCT05458999).
Palabras clave: Oclusión total crónica Realidad virtual Ansiedad
Abbreviations
CTO: chronic total coronary occlusion. PCI: percutaneous coronary intervention. VAS: visual analogue scale. VASA: visual analogue scale of anxiety. VASP: visual analogue scale of pain. VR: virtual reality.
INTRODUCTION
Chronic total coronary occlusions (CTO) are diagnosed in up to 15% of patients with coronary artery disease undergoing coronary angiography.1 Percutaneous coronary interventions (PCI) of CTOs are one of the greatest challenges we face in interventional cardiology due to the complexity of these procedures and the increased risk of complications.2 Over the past few decades, advances in techniques and devices have made it possible to obtain better results while reducing the associated complications.3-5 Anxiety and pain during these procedures are often treated with oral benzodiazepines plus opioids or IV benzodiazepines upon request during the procedure. The possibility of performing these procedures without anesthesia or sedation avoids the risks associated with these therapies. On the contrary, it submits the patient to pain and anxiety during the procedure. Several factors such as long procedures, patient immobility (especially in biradial access), and monotonous and hostile environments (operating rooms or cath labs) influence patient anxiety. Virtual reality (VR) has been successfully used in several clinical settings such as transcatheter aortic valve implantation6 or atrial fibrillation ablation7 to reduce intraoperative anxiety. There is no evidence on the use of VR reducing perioperative patient anxiety during PCI, specifically in CTO PCI. Compared to standard PCI, this procedure could benefit even further from VR due to its longer duration, use of double arterial access, and possibility of triggering ischemia and chest pain.
The objective of this study is to determine whether the use of a VR system in PCIs on CTOs decreases the level of anxiety and pain during CTO procedures compared to conventional management.
METHODS
Overall study design
The Decreasing patient anxiety during revascularization of chronic total coronary occlusions using virtual reality glasses (ReViCTO) trial (ClinicalTrials.gov Identifier: NCT05458999) was designed as a randomized, controlled, open-label, superiority clinical trial with 2 parallel arms (procedural use of VR goggles vs conventional management) with a primary endpoint of maximum level of anxiety perceived by the patient measured through the visual analogue scale of anxiety (VASA).
The study will be conducted in full compliance with the principles set forth in the Declaration of Helsinki (1996) and the International Conference on Harmonization Good Clinical Practice Guideline. The study protocol was approved by the Clinical Research Ethics Committee (CREC) of Hospital Clínico Universitario de Valencia, Spain. All patients signed an informed consent form. The study is registered at clinicaltrials.gov (NCT05458999). The World Health Organization minimum standard list of items for clinical trials are listed in table 1 of the supplementary data. This protocol follows the SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials.8
Study setting and eligibility criteria
The trial will be conducted at Hospital Clínico Universitario de Valencia, Spain, a reference teaching hospital on interventional cardiology that treats nearly 800 000 patients both from rural and metropolitan areas. Since this is a preliminary study it is designed as a single-center trial. All procedures will be performed by a team of 2 interventional cardiologists experienced in CTO revascularization. Patient enrolment started back in December 2021. On Dec. 25th 2022, 25 patients had already been enrolled in the study (43% of the target population). Patients with visual impairment, dementia, language barriers or any situations that would prevent the use of VR glasses will be excluded. Inclusion and exclusion criteria are listed in table 1. All patients must meet all the inclusion criteria and none of the exclusion criteria.
Table 1. Inclusion and exclusion criteria
| Inclusion criteria |
| Age > 18 years |
| Elective percutaneous coronary intervention on chronic total coronary occlusion |
| Physical and mental ability to wear virtual reality glasses |
| Exclusion criteria |
| Unable to or unwilling to give informed consent |
| Visual impairment |
| Dementia |
| Language barrier (unable to communicate fluently in Spanish or English) |
| Any other situations that would prevent the use of virtual reality glasses |
Assignment of interventions
Each patient will be randomized on a 1:1 ratio to the intervention (use of VR goggles during the CTO procedure) or the control arm (routine clinical practice). Given the small sample size estimated, permuted block randomization was used to guarantee an equal number of participants per arm.9 Random sequence was computer generated using blocks with a size unknown to the investigators until the end of recruitment. Patient enrolment and arm assignment will be performed by the investigators. Allocation concealment will be ensured using a web application that assigns a unique identification number and the assigned arm once the patient has been recruited for the trial. This system prevents changes to the identification number or arm deleting patients after randomization. Because of the nature of the trial no masking or blinding will be applied at any level.
Participant timeline
Since there is no follow-up, this study has a very simple timeline. Upon arrival to the cath lab, all patients scheduled for elective CTO PCI will be screened and checked to see if they meet all the inclusion criteria and none of the exclusion criteria. If they don’t meet these criteria, they will be considered a screening failure and will not participate in the study. If all criteria are met by the patients, the investigators will need to obtain their written informed consent right before patients arrive at the cath lab. The functioning of the trial will be explained orally and reading of the informed consent will be offered allowing enough time, if necessary. Patients will be randomized to wear VR goggles or to the control arm. During the PCI, all measures regarding anxiety will be applied regardless of the allocation arm. Also, all drug therapies administered will be registered by the study nurse. After the procedure, the patient’s perceived anxiety and pain will be assessed by the study nurse, this being the end of the trial for the patient (figure 1).
Figure 1. Central illustration. Trial flowchart.
Data collection
Demographics, the past medical history, preoperative (indication for revascularization, blood tests, left ventricular ejection fraction…), and perioperative variables [arterial access, radiation dose, maximum level of anxiety (VASA), and visual analogue scale of pain (VASP) perceived by the patient measured through visual analogue scale, nausea, and dizziness during the procedure] will be collected (table 2 of the supplementary data).
Clinical variables will be collected from the local and regional electronic clinical data system and asked directly to the patient when lacking. Blood test results will be collected from the local laboratory system using the last available determination. Echocardiographic and magnetic resonance imaging data will be collected from the local electronic clinical data system. The Seattle Angina Questionnaire, VASA, VASP, the presence of nausea or dizziness, overall satisfaction with the procedure, and overall satisfaction will be assessed by the study nurse and included in a dedicated form (table 3 of the supplementary data). All these data will be transferred to a dedicated database in 1 single local computer. This database is designed with range check for numerical variables to prevent erroneous data entry. Also, the database will check for duplicates when entering the hospital identification number. All data will be stored in a database kept in a dedicated computer with no Internet connection to avoid unwanted leaks or stole information. The investigators will have access to this database only.
Trial intervention
Eligible patients will be randomized to the intervention (VR goggles) or the control arm (routine clinical practice).
Virtual reality goggles
A commercial Oculus Quest 2 VR goggle system (Meta Platforms, Inc., United States) will be used. The viewing consisted of using the capabilities of the VR goggles to recreate a 2D playback that simulates the size of a large-format movie screen. Using Netflix video streaming system (Netflix Inc. United States), the documentary series “Our Planet” [Silverback Films, United Kingdom]10 will be played for all patients starting with chapter 1, and sequentially and automatically playing the following chapters. Before the procedure, the patient will be informed on the VR goggle system-based operation, possible side effects (nausea or dizziness), and the possibility to remove it at any time. Before the arterial puncture, the VR goggle system will be put on and checked for proper functioning. It will be removed before removing arterial introducers, when the patient wishes to do so or if serious complications occur. During the procedure, the patient’s general condition will be checked every 30 min.
The system will be prepared following these steps: 1) drawing the security perimeter with the controller; 2) starting the Netflix application; 3) selecting the “Void Theater” option; 4) searching for the documentary series “Our Planet” and playing the first episode; 5) adjusting the screen size with the controller; 6) selecting travel mode; 7) putting the VR goggles on the patient (figure 2); 8) asking the patient if he can watch and hear correctly. If not, the VR goggles should be repositioned.
Figure 2. Real patient wearing virtual reality goggles during a chronic total coronary occlusion revascularization procedure using double radial artery access.
Control arm
The comparator chosen is the current clinical practice with no VR goggles. A possible comparator using a VR goggle with no content was discarded because of the high chances of claustrophobia or mental discomfort.
Both arms will receive drugs upon request to reduce perceived pain and anxiety. In both arms, anxiolytic drugs (morphine chloride or midazolam at 1 mg boluses) will be administered by the circulating nurse if the patient explicitly expresses the need for such treatment or if external signs of anxiety or pain (agitation, complaints...) are observed. The last decision on treatment administration will be left to the lead operator. The remaining actions for the management of pre- and perioperative anxiety will be identical in both arms. Preoperative anxiolytic treatment was not routinely administered to all patients, only upon the patient’s request.
Endpoints
The primary endpoint will be to assess changes to the maximum level of anxiety perceived by the patient during the procedure. Secondary endpoints will be to assess a) changes to the maximum level of pain perceived by the patient during the procedure; b) differences in the need for intraoperative anxiolytic drug therapy or doses of anxiolytic drugs (midazolam or morphine chloride) administered during the procedure; and c) the overall satisfaction experienced with the VR goggles.
Both the primary (anxiety) and secondary endpoints of pain will be measured through the VASA11 and pain (VASP)12 that go from 1 to 10. Both VASA and VASP will be collected by a specialized nurse right after the end of the procedure before leaving the room through a specific survey on the maximum level of anxiety or pain perceived during the procedure (table 3 of the supplementary data).
The VAS will be used to quantify the patients’ responses objectively. The VAS eliminates the examiner influence or bias that often comes with verbal questioning and is a more appealing method of evaluation for participants. Although this method is not perfect, it remains a common way to assess anxiety and pain.13,14 The patient will also be asked if he’d like VR to be used in other similar settings. Total doses of benzodiazepines (midazolam) or opioids (morphine) administered during the procedure will be registered in total milligrams.
Sample size estimate
The sample size was estimated based on the primary endpoint. In former studies that assessed anxiety during catheterization the standard deviation of VASA was 2.715,16 (σ). VASA > 2 (μ1 – μ2) was descriptive of clinically significant differences. To detect differences ≥ 2 in VASA assuming a normal distribution, alpha and beta risks of 0.05 (α) and 0.2 (β) in bilateral contrast in a sample size of 58 patients (29 in each arm) were estimated.
Statistical analysis
Quantitative variables will be expressed as mean ± standard deviation when they follow a normal distribution and as median [interquartile range] if they don’t. Qualitative ones will be expressed as percentages (absolute value). Fisher’s exact test or the chi-square test will be used to compare qualitative variables. Also, the Student t test or the Mann-Whitney U test will be used if quantitative variables don’t follow a normal distribution.
The primary endpoint (VASA), VASP, and dosage of drugs will be compared in both arms using the Student t test. The use or non-use of drugs during the procedure, the presence or absence of dizziness or nausea will be compared using Fisher’s exact test or the chi-square test. Subgroup analyses will be performed based on sex, age, and previous experience with new technologies.
All statistical tests will be bilateral and considered significant if P < .05. Statistical analyses will be performed with R Core Team (2020) statistical software package (R Foundation for Statistical Computing, Austria).
DISCUSSION
CTOs are present in up to 20% of the patients with coronary artery disease. These numbers increase parallel to age (up to 40% in diabetics or patients with heart failure).17,18 In the past, most of these patients were referred for revascularization surgery due to poorly successful PCIs in this kind of lesions. Over the past few decades, several advances have been made regarding devices and technical materials, organization, and concentration of complex procedures in reference centers. Also, increased operator experience has led to a high success rate of 90%, and a very low rate of severe complications19,20 with the corresponding increase in the number indications for PCI CTO.
Although PCIs are a common and relatively low risk procedure, many patients undergoing these treatments experience anxiety (up to 37% in some populations).21-23 Anxiety involves feelings of fear, tension or panic or the prospect that something unpleasant is about to happen. State anxiety may be more clinically relevant for patients undergoing PCI because it is transient in nature and amenable to clinical procedures. Patients undergoing PCI have multiple sources of anxiety including their own concerns. These concerns can include fear of discomfort, uncertainty, and fear associated with survival that can be more distressing than chest pain itself.24
PCI CTO creates more anxiety for the patients compared to other interventional procedures for several reasons. In the first place, double access with high-calibre sheaths is frequently used, even biradial. Repeated access punctures with consequently an increased pain and limited patient mobility contributes to more discomfort and higher anxiety levels. Secondly, the duration of the procedure is long, and can be up to 3 to 4 hours or more in some special scenarios. Being exposed to immobility in a monotone and hostile scenario for such a long time is a reasonable cause for anxiety. Thirdly, cath labs are often strange environments for the patient with machinery and equipment that may be frightening for him at first. Furthermore, discussion with the treating team, the use of terms unfamiliar to the patient or the existence of beeps and alarms can make the patient think that something bad might happen to him, thus increasing the levels of anxiety. Fourthly, patients undergoing elective PCI CTO usually undergo, at best, at least, 1 invasive coronary angiography, and commonly up to several coronary interventions including failed CTO revascularization attempts. Previous procedures could be remembered as painful or stressful and anticipation anxiety could appear. Stress and anxiety associated with needle-related procedures may lead to needle phobia,25 which could also contribute to a high level of anxiety. Fifthly, chest pain is an important factor of procedural anxiety during CTO PCI, and it occurs in a large number of patients. For example, with retrograde approaches, the flow of collateral branches on which the CTO-related myocardial territory is completely dependent is interrupted due to their occupation by the guidewire or microcatheter, thus causing ischemia and pain). Therefore, there is a potential high anxiety level in patients undergoing CTO PCI that depends on multiple factors and mechanisms that feed from one another.
At the end of the 20th century, it was noted that both behavioral and pharmaceutical interventions should be used to manage pain during medical procedures.26 Distraction techniques may be effective reducing the patients’ pain during various invasive procedures because pain involves both physical stimuli and emotional responses. Studies have shown that various distraction techniques like music, massage, breathing exercises, and behavioral therapy can effectively reduce the feeling of pain and stress symptoms during painful procedures.27,28
VR is a computer-generated simulation of the physical world that allows people to experience it in a realistic way. VR goggles achieve visual and auditory semi-isolation that, together with the images projected, evade the patient while act on environmental and emotional factors of anxiety. VR has proven superior to other distraction methods such as television, listening to music or playing games.29,30 VR has been used to relieve anxiety and pain in patients undergoing several kinds of procedures as needle-related interventions,31 burn wound debridement,32,33 physical therapy,34 dental procedures,35 colonoscopy,36 minor surgical procedures,37 nasal endoscopy38 or chemotherapy.39 A total of 4 randomized clinical trials have been conducted to study the level of anxiety experienced by adults undergoing different medical procedures like hysteroscopy,40 labor,41 and colonoscopy.42 The studies used various measurement tools such as the VAS scale from 0 to 10, the 5-point Likert scale 0-5, and the State-Trait Anxiety Inventor.
Experiences with VR in interventional cardiology during procedures are scarce. Back In 2020, Bruno et al.6 used a randomized clinical trial to prove the that the use of a VR-based system was safe and feasible during TAVI and that VAS score was reduced by 3 points with the use of VR without impacting nausea or vomiting. Almost all patients said they would use this technology in a similar setting. It is remarkable that this study population was an old population (mean age, 83 years) without previous experience with VR and limited experience with new technologies. This shows that even in a population not used to new technologies that could be expected to reject or not tolerate VR goggles, its use was tolerated and effective. Moreover, this study found that it was important not only that patients accepted the new technology, but also that interventional cardiologists approved it. At first, their reaction went from full support to slight rejection. Those who hesitated to use this new approach thought that their interaction with the patient during the procedure might be limited. However, over time, when they saw that this was not the case, acceptance increased. Similarly, Roxburgh et al.7 tested the utility of VR in patients undergoing atrial fibrillation ablation in an observational study of 48 patients. They showed that VR reduced perceived pain during the procedure and that VR can be easily incorporated into the standard procedure workflow. As far as we know, no studies have ever been conducted on the use of VR during PCI CTO.
Most studies on VR technology have been observational and not standardized, which complicates comparing results and drawing solid conclusions. Additionally, currently, no guidelines or consensus have ever been published on how to incorporate VR technology to cardiac procedures.43 To address these challenges, an expert taskforce from the international scientific community may be useful to identify evidence gaps, set priorities, standardize research protocols, and create guidelines to implement VR technology in heart procedures.
Limitations
Some limitations should be taken in account in this clinical trial. First, the open-label nature of the trial could lead to bias favorable to the RV group due to the lack of blinding and potential for patients and investigators to influence the outcomes. The entire staff will be warned on this possible bias and advised to prevent it before each procedure. Second, some uncontrolled confounding factors may play a role in the differences seen in the administration of drug therapy between RV and the control groups. We will try to prevent or, at least, mitigate this treatment only if the patient explicitly wishes to do so or if outward signs of anxiety or pain are observed. Third, the results of this trial should be interpreted and applied with caution to other scenarios due to the single-center nature of this trial. Fourth, the primary and secondary endpoints of anxiolytic treatment needed could be closely correlated. Our objective is to determine whether the use of VR goggles decreases anxiety. However, it could happen that both groups have similar levels of anxiety and a greater need for anxiolytic treatment (which is a surrogate of increased anxiety, on the one hand, that could also expose patients to a higher risk of adverse effects, on the other). The choice of the primary and secondary endpoints of anxiety and need for anxiolytic treatment allows us to explore this possibility. Finally, the drug therapy of anxiety was not protocolized but left to the operator’s discretion.
CONCLUSIONS
The ReViCTO trial is the first randomized clinical trial ever designed to evaluate the use of VR during CTO PCI. Results will show the utility of this technology reducing anxiety and pain in PCI CTO.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
A. Fernández-Cisnal, and G. Miñana: study idea or design, data curation, analysis, and interpretation, drafting, and final approval of the version submitted for publication. B. Silla, J.M. Ramón, E. Valero, and S. García-Blas: data curation, analysis, and interpretation, revision of the manuscript regarding significant intellectual content, and final approval of the version submitted for publication. J. Núñez, V. Bodí, and J. Sanchis: data analysis and interpretation, revision of the manuscript regarding significant intellectual content, and final approval of the version submitted for publication. A. Fernández-Cisnal, and G. Miñana agree to take full responsibility for all aspects of the manuscript, and investigate and resolve all questions regarding the accuracy and truthfulness of the study as a whole.
CONFLICTS OF INTEREST
J. Núñez received fees for participating in the advisory boards and educational activities from Astra Zeneca, Boehringer-Ingelheim, NovoNordisk, Bayer, and Novartis. J. Sanchis received speaker fees from Abbott Vascular, and Prosmedica. G. Miñana received speaker fees from Abbott Vascular, and Teleflex, and support for attending meetings from Medtronic and World Medical. The remaining authors declared no other conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- CTO PCI is one of the greatest challenges for interventional cardiology due to the complexity of these procedures and the increased risk of complications.
- Several factors like long procedures, patient immobility, and the presence of a monotonous and hostile environment influence patient anxiety, which is usually treated with benzodiazepines and opioids upon request during the procedure.
- Virtual reality has been successfully used in several clinical settings reducing intraoperative anxiety. There is no evidence that the use of VR reduces perioperative patient anxiety during CTO PCI.
WHAT DOES THIS STUDY ADD?
- The ReViCTO trial is the first randomized clinical trial ever conducted to use VR during PCI CTO. Its results will show the utility of this technology reducing anxiety and pain in PCI CTO.
- Primary endpoint will be to assess changes to the maximum level of anxiety perceived by the patient.
- Secondary endpoints will be a) changes to the maximum level of pain during the procedure; b) differences in the need for intraoperative anxiolytic drug therapy; and c) overall satisfaction with the VR goggles.
REFERENCES
1. Tajti P, Burke MN, Karmpaliotis D, et al. Update in the Percutaneous Management of Coronary Chronic Total Occlusions. JACC Cardiovasc Interv. 2018;11:615-625.
2. Tajti P, Brilakis ES. Chronic Total Occlusion Percutaneous Coronary Intervention: Evidence and Controversies. J Am Heart Assoc. 2018;7:e006732.
3. Patel VG, Brayton KM, Tamayo A, et al. Angiographic success and procedural complications in patients undergoing percutaneous coronary chronic total occlusion interventions: a weighted meta-analysis of 18,061 patients from 65 studies. JACC Cardiovasc Interv. 2013;6:128-136.
4. Brilakis ES, Banerjee S, Karmpaliotis D, et al. Procedural outcomes of chronic total occlusion percutaneous coronary intervention: a report from the NCDR (National Cardiovascular Data Registry). JACC Cardiovasc Interv. 2015;8:245-253.
5. Azzalini L, Karmpaliotis D, Santiago R, et al. Contemporary Issues in Chronic Total Occlusion Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2022;15:1-21.
6. Bruno RR, Lin Y, Wolff G, et al. Virtual reality-assisted conscious sedation during transcatheter aortic valve implantation: a randomised pilot study. EuroIntervention. 2020;16:e1014-e1020.
7. Roxburgh T, Li A, Guenancia C, et al. Virtual Reality for Sedation During Atrial Fibrillation Ablation in Clinical Practice: Observational Study. J Med Internet Res. 2021;23:e26349.
8. Chan AW, Tetzlaff JM, Altman DG, et al. SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials. Ann Intern Med. 2013;158:200.
9. Schulz KF, Grimes DA. Unequal group sizes in randomised trials: guarding against guessing. Lancet. 2002;359:966-970.
10. Our Planet. Silverback Films, UK. Available at: https://www.netflix.com/es/title/80049832. Accessed 02 Feb 2023.
11. Williams VS, Morlock RJ, Feltner D. Psychometric evaluation of a visual analog scale for the assessment of anxiety. Health Qual Life Out. 2010;8:57.
12. Carlsson AM. Assessment of chronic pain. I. Aspects of the reliability and validity of the visual analogue scale. Pain. 1983;16:87-101.
13. Aubrun F, Paqueron X, Langeron O, Coriat P, Riou B. What pain scales do nurses use in the postanaesthesia care unit? Eur J Anaesth. 2003;20:745-749.
14. Kindler CH, Harms C, Amsler F, Ihde-Scholl T, Scheidegger D. The Visual Analog Scale Allows Effective Measurement of Preoperative Anxiety and Detection of Patients’ Anesthetic Concerns. Anesth Analg. 2000;90:706-712.
15. Delewi R, Vlastra W, Rohling WJ, et al. Anxiety levels of patients undergoing coronary procedures in the catheterization laboratory. Int J Cardiol. 2017;228:926-930.
16. Vlastra W, Delewi R, Rohling WJ, et al. Premedication to reduce anxiety in patients undergoing coronary angiography and percutaneous coronary intervention. Open Hear. 2018;5:e000833.
17. Damluji AA, Pomenti SF, Ramireddy A, et al. Influence of Total Coronary Occlusion on Clinical Outcomes (from the Bypass Angioplasty Revascularization Investigation 2 DiabetesTrial). Am J Cardiol. 2016;117:1031-1038.
18. Tajstra M, Pyka Ł, Gorol J, et al. Impact of Chronic Total Occlusion of the Coronary Artery on Long-Term Prognosis in Patients With Ischemic Systolic Heart Failure Insights From the COMMIT-HF Registry. JACC Cardiovasc Interv. 2016;9:1790-1797.
19. Christopoulos G, Karmpaliotis D, Alaswad K, et al. Application and outcomes of a hybrid approach to chronic total occlusion percutaneous coronary intervention in a contemporary multicenter US registry. Int J Cardiol. 2015;198:222-228.
20. Amat-Santos IJ, Martin-Yuste V, Fernández-Díaz JA, et al. Resultados inmediatos e impacto funcional y pronóstico tras la recanalización de oclusiones coronarias crónicas. Resultados del Registro Ibérico. Rev Esp Cardiol. 2019;72:373-382.
21. Astin F, Jones K, Thompson DR. Prevalence and patterns of anxiety and depression in patients undergoing elective percutaneous transluminal coronary angioplasty. Hear Lung J Acute Critical Care. 2005;34:393-401.
22. Lenzen MJ, Gamel CJ, Immink AW. Anxiety and Well-Being in First-Time Coronary Angioplasty Patients and Repeaters. Eur J Cardiovasc Nur. 2002;1:195-201.
23. Trotter R, Gallagher R, Donoghue J. Anxiety in patients undergoing percutaneous coronary interventions. Hear Lung J Acute Critical Care. 2011;40:185-192.
24. Heikkilä J, Paunonen M, Virtanen V, Laippala P. Fear of patients related to coronary arteriography. J Adv Nurs. 1998;28:54-62.
25. McLenon J, Rogers MAM. The fear of needles: A systematic review and meta-analysis. J Adv Nurs. 2019;75:30-42.
26. Zeltzer LK, Altman A, Cohen D, LeBaron S, Munuksela EL, Schechter NL. American Academy of Pediatrics Report of the Subcommittee on the Management of Pain Associated with Procedures in Children with Cancer. Pediatrics. 1990;86(5 Pt 2):826-831.
27. Manne SL, Bakeman R, Jacobsen PB, Gorfinkle K, Redd WH. An analysis of a behavioral intervention for children undergoing venipuncture. Health Psychol. 1994;13:556-566.
28. Burns-Nader S, Joe L, Pinion K. Computer tablet distraction reduces pain and anxiety in pediatric burn patients undergoing hydrotherapy: A randomized trial. Burns. 2017;43:1203-1211.
29. Hoffman HG, Chambers GT, Meyer WJ, et al. Virtual Reality as an Adjunctive Non-pharmacologic Analgesic for Acute Burn Pain During Medical Procedures. Ann Behav Med. 2011;41:183-191.
30. Malloy KM, Milling LS. The effectiveness of virtual reality distraction for pain reduction: A systematic review. Clin Psychol Rev. 2010;30:1011-1018.
31. Wang Y, Guo L, Xiong X. Effects of Virtual Reality-Based Distraction of Pain, Fear, and Anxiety During Needle-Related Procedures in Children and Adolescents. Front Psychol. 2022;13:842847.
32. Kipping B, Rodger S, Miller K, Kimble RM. Virtual reality for acute pain reduction in adolescents undergoing burn wound care: A prospective randomized controlled trial. Burns. 2012;38:650-657.
33. Khadra C, Ballard A, Déry J, et al. Projector-based virtual reality dome environment for procedural pain and anxiety in young children with burn injuries: a pilot study. J Pain Res. 2018;11:343-353.
34. Soltani M, Drever SA, Hoffman HG, et al. Virtual Reality Analgesia for Burn Joint Flexibility: A Randomized Controlled Trial. Rehabil Psychol. 2018;63:487-494.
35. Lahti S, Suominen A, Freeman R, Lähteenoja T, Humphris G. Virtual Reality Relaxation to Decrease Dental Anxiety: Immediate Effect Randomized Clinical Trial. Jdr Clin Transl Res. 2020;5:312-318.
36. Veldhuijzen G, Klaassen NJM, Wezel RJAV, Drenth JPH, Esch AAV. Virtual reality distraction for patients to relieve pain and discomfort during colonoscopy. Endosc Int Open. 2020;08:E959-E966.
37. Clerc PGB, Arneja JS, Zwimpfer CM, Behboudi A, Goldman RD. A Randomized Controlled Trial of Virtual Reality in Awake Minor Pediatric Plastic Surgery Procedures. Plast Reconstr Surg. 2021;148:400-408.
38. Liu KY, Ninan SJ, Laitman BM, Goldrich DY, Iloreta AM, Londino AV. Virtual Reality as Distraction Analgesia and Anxiolysis for Pediatric Otolaryngology Procedures. Laryngoscope. 2021;131:E1714-E1721.
39. Fabi A, Fotia L, Giuseppini F, et al. The immersive experience of virtual reality during chemotherapy in patients with early breast and ovarian cancers: The patient’s dream study. Frontiers Oncol. 2022;12:960387.
40. Deo N, Khan K, Mak J, et al. Virtual reality for acute pain in outpatient hysteroscopy: a randomised controlled trial. Bjog Int J Obstetrics Gynaecol. 2021;128:87-95
41. Xu N, Chen S, Liu Y, Jing Y, Gu P. The Effects of Virtual Reality in Maternal Delivery: Systematic Review and Meta-analysis. Jmir Serious Games. 2022;10:e36695.
42. Cakir SK, Evirgen S. The Effect of Virtual Reality on Pain and Anxiety During Colonoscopy: A Randomized Controlled Trial. Turk J Gastroenterol. 2021;32:451-547.
43. Mahtab EAF, Egorova AD. Current and future applications of virtual reality technology for cardiac interventions. Nat Rev Cardiol. 2022;19:779-780.
- Regional differences in STEMI care in Spain. Data from the ACI-SEC Infarction Code Registry
- Clinical outcomes of patients undergoing percutaneous coronary intervention treated with colchicine
- High rate of uncovered struts in latest generation drug-eluting stents with durable, biodegradable polymer or lack of it 1 month after implantation
- Impact of time of intervention in patients with NSTEMI. The IMPACT-TIMING-GO trial design
Interviews
An interview with Bruno Scheller
aServicio de Cardiología, Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria de La Princesa (IIS-IP), Universidad Autónoma de Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain