ABSTRACT
Introduction and objectives: Chronic total coronary occlusion (CTO) of coronary arteries is frequent in heart transplant recipients (HTR). It is usually managed with medical therapy due to the suboptimal results shown by the percutaneous coronary intervention (PCI). Although the PCI of native CTOs is performed in experienced centres, in HTR we do not know PCI results or clinical efficacy. This is the first study ever to actually analyse the PCI-Target Lesion Procedure Success of CTO PCI and its angiographic and clinical follow-up in HTR. In addition, we compared the clinical follow-up of CTO vs non-CTO PCI in HTR.
Methods: We retrospectively analysed the baseline characteristics, procedural outcomes and clinical events during the follow-up of HTR with CTO undergoing PCI between January 1, 2006 and December 31, 2016 in 2 centres with an ongoing CTO program. Over the same period, we also compared clinical events during the follow-up of these patients vs PCI on non-CTO stenosis in HTR at one of the centres.
Results: PCI was successful in 13 out of 14 patients. A systematic follow-up angiography was conducted at centre 1 (n = 10). Two patients showed in-stent restenosis (20%), and a new PCI was performed successfully in both cases. Mortality rate was 28.5%, after a median follow-up of 33.5 months [interquartile range, 20-50]. We found no statistical differences in the clinical events after the PCI of CTO lesions vs non-CTO lesions in HTR.
Conclusions: The PCI of CTO in HTR is feasible in experienced centres and selected patients, with a high success rate and low rate of intraprocedural complications.
Keywords: Chronic total coronary occlusion. Heart transplantation. Coronary angioplasty. Cardiac allograft vasculopathy. Results.
RESUMEN
Introducción y objetivos: Las oclusiones coronarias crónicas totales (OTC) son frecuentes en los pacientes receptores de trasplante cardiaco (PTC) y suelen tratarse con tratamiento médico debido a los resultados subóptimos de la intervención coronaria percutánea (ICP). A pesar de que la ICP de OTC en corazones nativos se lleva a cabo en centros experimentados, no se conocen sus resultados ni la eficacia clínica en PTC. Este es el primer estudio que analiza la tasa de éxito de la ICP sobre OTC en PTC, así como el seguimiento clínico y angiográfico. Así mismo, se comparan los eventos clínicos durante el seguimiento de los PTC tratados con ICP sobre OTC frente a PTC con ICP sobre estenosis no oclusivas.
Métodos: Se analizaron retrospectivamente las características clínicas basales, los resultados del procedimiento y los eventos clínicos durante el seguimiento de los PTC con OTC en quienes se realizó una ICP entre el 1 de enero de 2006 y el 31 de diciembre de 2016 en 2 centros con un programa específico de OTC. Además, se compararon los eventos clínicos durante el seguimiento de estos pacientes con los de PTC tratados con ICP sobre una estenosis coronaria no oclusiva en uno de los centros durante el mismo periodo de tiempo.
Resultados: La ICP resultó exitosa en 13 de los 14 pacientes. Se realizó un seguimiento angiográfico sistemático en el centro 1 (n = 10). Dos pacientes presentaron reestenosis en el interior del stent (20%), por lo que se realizó una nueva ICP sobre ellas que tuvo éxito en ambos casos. La mortalidad fue del 28,5% con una mediana de seguimiento de 33,5 meses [rango intercuartílico, 20-50]. No se encontraron diferencias estadísticamente significativas entre los eventos clínicos tras la ICP sobre una OTC y tras la ICP sobre una estenosis no oclusiva en PTC.
Conclusiones: La ICP sobre OTC en PTC es factible en centros con experiencia y en pacientes seleccionados, con una alta tasa de éxito y una baja frecuencia de complicaciones periprocedimiento.
Palabras clave: Angioplastia coronaria. Enfermedad vascular del injerto. Oclusión total crónica. Resultados. Trasplante cardiaco.
Abbreviations: CAV: cardiac allograft vasculopathy. CTO: chronic total coronary occlusion. HTR: heart transplant recipient. PCI: percutaneous coronary intervention.
INTRODUCTION
Nowadays the orthotopic cardiac transplantation is the endgame for many patients with end-stage heart failure in developed countries.1 After the third year of heart transplantation, chronic rejection is one of the leading causes of morbidity and mortality.2 One of the main manifestations of chronic rejection is cardiac allograft vasculopathy (CAV) that affects nearly 50% of transplanted hearts at 5 years.3 CAV is characterized by diffuse intimal thickening that leads to progressive coronary luminal narrowing, with similar consequences to native heart atherosclerotic disease.4 It typically shows diffuse lesions in the distal territories with more focal stenosis in the proximal segments.5 Although the main treatment of CAV is based on titrating immunosuppressive therapy, a PCI is usually conducted here.
The use of PCIs for the management of CAVs has been reported to have success rates above 90% but with long-term restenosis rates of up to 36%.6 On the other hand, as a result of denervation following transplantation and subsequent incomplete reinnervation, most patients are asymptomatic or show atypical symptoms, despite silent progression to advanced stages of the disease. Therefore, angiographic findings of a CTO in this population are not rare.
Due to the high rate of restenosis associated with these procedures7 and the lack of solid evidence of a clinical benefit, medical treatment is advised in these patients especially when it comes to CTOs. Nevertheless, CTO recanalization has experienced a significant boost due to new techniques and technological advances made over the last few years. Therefore, in highly experienced centres performing PCIs of CTOs, this kind of procedures can be an alternative.
We know from registries published in recent years that the success rates of PCIs on CTOs, in non-transplanted patients are between 60% and 80% in the United States, Canada, and Europe.8,9 However, there are no studies of the success rates and results of PCIs on CTOs in patients in whom CAV can play an important role. We don’t have data on short and long-term clinical benefits either.
Consequently, the main goal of our study is to evaluate the characteristics of this population, the feasibility of PCI in these patients and its clinical results. In addition, we will make a comparison with heart transplant recipients (HTR) who underwent a PCI on lesions without CTO criteria.
METHODS
Definitions
For the purpose of this paper, the main conditions are defined as follows. According to the EuroCTO definition, chronic total coronary occlusion (CTO) is defined as the presence of Thrombolysis in Myocardial Infarction 0 flow within the occluded coronary segment with an estimated occlusion duration of > 3 months.10 Percutaneous coronary intervention (PCI)-target lesion procedure success is defined as the achievement of < 30% residual diameter stenosis of the target lesion as assessed by visual inspection or quantitative coronary angiography, without an in-hospital major adverse cardiac event (death, acute myocardial infarction, or repeated coronary revascularization of the target lesion).11 In-stent restenosis is the re-narrowing of a stent implanted at a lesion site to treat a prior stenosis, to an in-stent diameter stenosis of > 50%, including the original treated site plus the adjacent vascular segments 5 mm proximal and 5 mm distal to the stent.11 Regarding PCI-related myocardial infarction related (and according to the 4th universal definition of myocardial infarction), stand-alone post-procedural increases of cardiac troponin values are enough to establish a diagnosis of procedural myocardial injury but not for the diagnosis of a type 4a myocardial infarction. Type 4a myocardial infarction requires the elevation of cardiac troponin values greater than 5 times the 99th percentile URL in patients with normal baseline values or patients with elevated pre-procedural cardiac troponin in whom the cardiac troponin levels are stable (≤ 20% variation) or dropping. The post-procedural cardiac troponin needs to rise > 20% to an absolute value more than five times the 99th percentile upper reference limit. In addition, there should be evidence of new myocardial ischaemia, either from electrocardiogram changes, imaging evidence, or procedural-related complications associated with reduced coronary blood flow such as coronary dissections, occlusions of a major epicardial artery or side branch occlusion/thrombi, collateral flow disruptions, slow flows or no-reflow, or distal embolizations.12
Patients and data analysis
First, we performed an analysis of the incidence of CTO in the context of coronary angiography screening of CAV in one of our centres. We also show the proportion of patient who underwent PCIs. Secondly, we conducted a retrospective analysis of all HTRs who underwent PCIs on a coronary CTO between January 1, 2006 and December 31, 2016 in 2 centres with an ongoing CTO program. Data from both centres were used for the analysis of the CTO PCI procedure characteristics, immediate results, clinical events and coronary angiography during follow-up. Also, we analysed clinical events during follow-up of the same previous HTR with PCI on a CTO and compared it to HTRs with PCI on a non-CTO lesion. The clinical endpoints analysed were these: readmission for heart failure or acute myocardial infarction, sustained ventricular arrhythmias, cardiovascular death, and all-cause mortality. Patient demographics and PCI-related data have been extracted from hospital databases. Digital images have been mined from dedicated storage servers. Transthoracic echocardiography images were obtained before and after CTO recanalizations to assess variations in the left ventricular ejection fraction using the Simpson method. Diagnostic coronary angiographies were analysed to assess the CTO characteristics prior to the PCI. Short- and long-term complications were obtained from medical records. In addition, follow-up coronary angiographies were performed in most patients, according to the protocols of the different centres.
This study was approved by the clinical trials committee of the Hospital Universitario Puerta de Hierro de Majadahonda, Madrid, Spain, in full compliance with the principles of the Declaration of Helsinki.
Statistical methods
For the assessment of the differences in the baseline demographic characteristics between the CTO group and the non-CTO group, the qualitative variables were expressed in percentages and analysed using the chi-square test. The quantitative variables were expressed as mean ± standard deviation and analysed using the Student t test. The quantitative variables without normal distribution were expressed as median ± interquartile range and analysed using the Wilcoxon test.
When it comes to the size of the sample, a comparative analysis of the clinical outcomes between CTO and non-CTO group was conducted using Fisher’s exact statistical test. To compare mortality between the CTO with the non-CTO group we used the Kaplan-Meier survival analysis. For all the tests, a P values < .05 were considered statistically significant. The statistical analysis was performed using the software SPSS package (V 21.0.0.0).
RESULTS
Incidence of chronic total coronary occlusion in heart transplant recipients
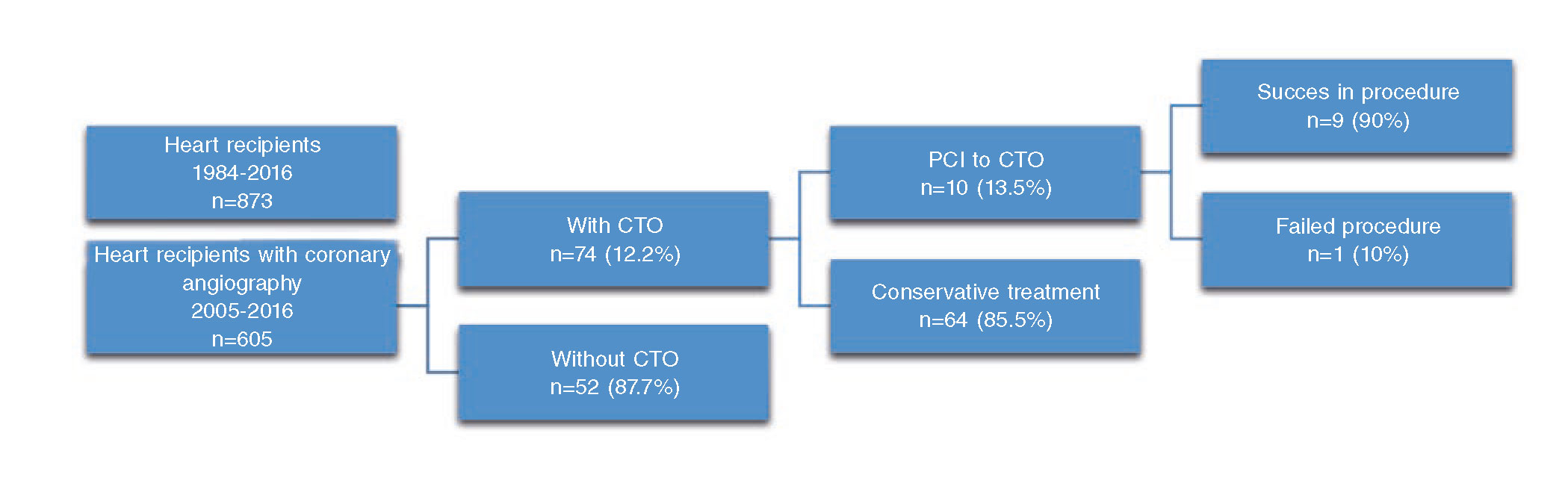
On the incidence of CTOs, only patients from Hospital Universitario Puerta de Hierro were studied. During the analysed period, 605 coronary angiographies were performed in HTRs. Among these, 74 patients (12%) had a CTO according to the EuroCTO criteria. Of these patients, PCIs were performed in only 10 (13%), leaving the remaining 64 patients under medical treatment (figure 1).
Figure 1. Study flowchart. CTO, chronic total coronary occlusion; PCI, percutaneous coronary intervention.
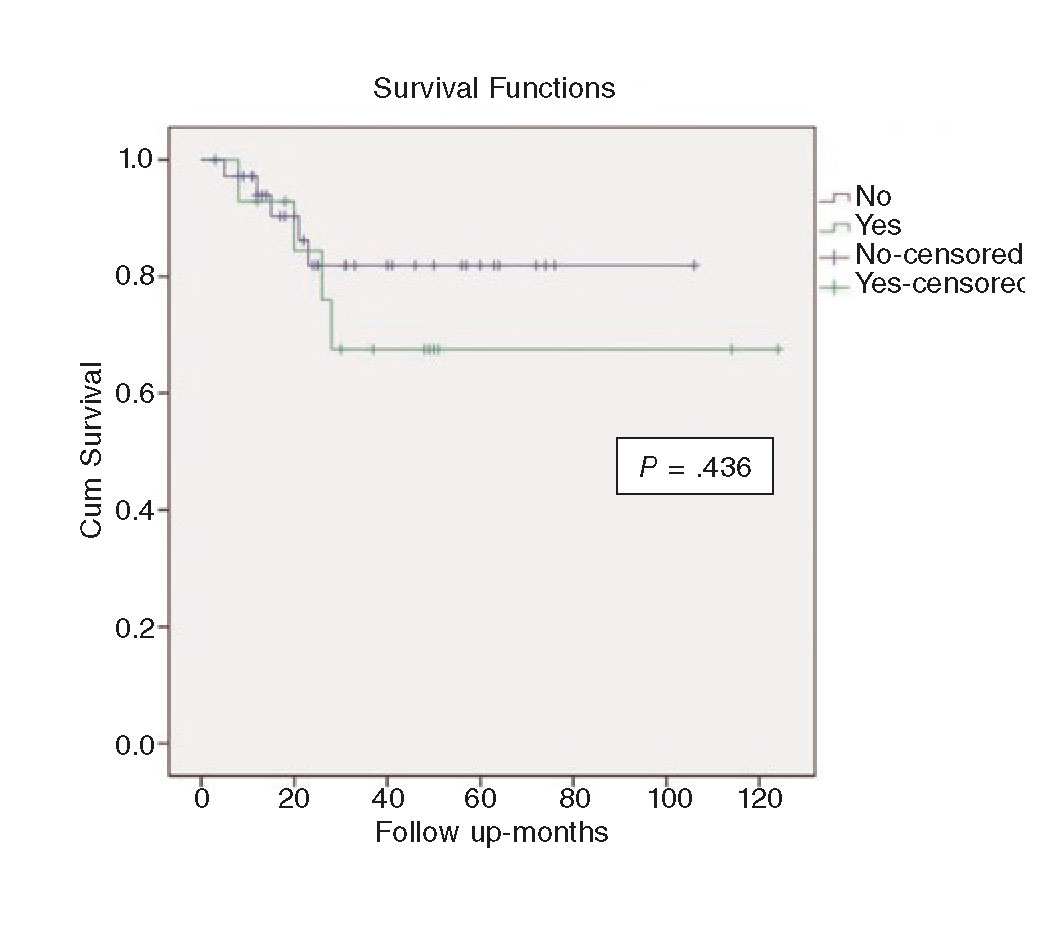
Figure 2. Kaplan-Meier survival analysis comparing chronic total coronary occlusion (green) vs non-chronic total coronary occlusion (blue) percutaneous coronary intervention.
Clinical characteristics. Procedural outcomes and follow-up
Clinical characteristics
Ten patients patients from Hospital Universitario Puerta de Hierro and 4 patients from Hospital Clinic de Barcelona, Barcelona, Spain were included in the study.
The baseline clinical characteristics and follow-up of each patient are shown on table 1. Eighty-five percent were males with an average age of 57 years [46.2-66] at the time of the PCI.
Table 1. Clinical variables and follow-up
| Patient | Age, y | HT | DM | DL | Clinical presentation prior to CTO diagnosis | Time from heart transplantation to PCI, ears | Time from CTOs PCI to FUCA, days | ISR in follow-up | LVEF prior PCI | LVEF post-PCI | Follow-up post-PCI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 73 | Yes | No | No | Asymptomatic | 14 | 238 | No | 59% | 52% | Alive at 4 and 1 months |
| 2 | 64 | No | No | No | HF | 8 | 192 | No | 30% | 45% | Alive at 4 and 4 months |
| 3 | 25 | No | No | No | HF | 5 | 1122 | Yes | 35% | 40% | Alive at 10 and 4 months |
| 4 | 31 | Yes | No | Yes | Asymptomatic | 8 | 175 | - | 50% | 55% | Alive at 9 and 6 months |
| 5 | 60 | Yes | No | Yes | Angina | 16 | 210 | No | Unknown | Unknown | Death at 1 and 8 months due to ruptured iliac artery aneurysm. |
| 6 | 57 | Yes | No | Yes | Asymptomatic | 10 | No FUCA | Unknown | 40% | 60% | Alive at 4 and 2 months |
| 7 | 53 | Yes | No | Yes | Asymptomatic | 12 | No FUCA | Unknown | 60% | Unknown | Sudden death at 2 and 4 months |
| 8 | 32 | No | Yes | No | Asymptomatic | - | No FUCA | Unknown | 35% | 40% | Alive at 3 and 1 months |
| 9 | 73 | Yes | Yes | No | Asymptomatic | 10 | 161 | No | 50% | 65% | Death due to metastatic pancreatic cancer at 2 and 3 months |
| 10 | 63 | Yes | No | No | Angina | 6 | No FUCA | Unknown | 60% | 55% | Sudden death at 267 d |
| 11 | 57 | No | No | No | Dyspnea | 14 | 134 | No | 60% | Unknown | Alive at 1 and 6 months |
| 12 | 51 | No | No | No | Asymptomatic | 10 | 246 | No | 55% | Unknown | Alive at 2 and 6 months |
| 13 | 53 | Yes | No | Yes | Dyspnea | 18 | 90 | Yes | 55% | Unknown | Alive at 1 y |
| 14 | 72 | No | No | Yes | Asymptomatic | 18 | 1439 | No | 65% | 63% | Alive at 4 y |
|
CTO, chronic total coronary occlusion; DL, dyslipidemia; DM, diabetes mellitus; FUCA, follow-up coronary angiography; HF, heart failure; HT, hypertension; ISR, in-stent restenosis; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention. |
|||||||||||
On the clinical manifestations when the CTO was diagnosed, 4 patients had angina or angina-like symptoms (28%), 2 patients required hospitalization for decompensated heart failure (14%) and the remaining 8 (57%) were asymptomatic. In asymptomatic patients, the diagnosis of ischemia was achieved by studying regional wall motion abnormalities in the follow-up echocardiograms (62%) and electrocardiographic changes suggestive of ischemia (12%). In the remaining patients (26%), the diagnosis was achieved based on the CAV screening coronary angiography.
The time elapsed from cardiac transplantation to coronary CTO PCI procedure varies from a minimum of 5 years to a maximum of 18 years (median 10 years).
Baseline coronary angiography and procedure
The angiographic characteristics of CTOs and details of each patient’s procedure can be found on table 2 and table 3.
Table 2. Chronic total coronary occlusion angiographic characteristics
| Patient | CTO location | J-CTO score | Blunt entry | Calcification | Occlusion length > 2 cm | Bending > 45º | Re-try lesion |
|---|---|---|---|---|---|---|---|
| 1 | Proximal RCA | 0 | - | - | - | - | - |
| 2 | Proximal LAD | 0 | - | - | - | - | - |
| 3 | First OM | 2 | + | - | - | + | - |
| 4 | Mid LAD | 1 | - | - | + | - | - |
| 5 | Mid RCA | 2 | - | - | + | + | - |
| 6 | Proximal LAD | 1 | - | - | + | - | - |
| 7 | Mid LAD | 1 | - | - | - | + | - |
| 8 | Distal RCA | 1 | - | - | - | + | - |
| 9 | Mid LAD | 0 | - | - | - | - | - |
| 10 | Second OM | 1 | + | - | - | - | - |
| 11 | Mid RCA | 2 | - | + | - | + | - |
| 12 | Mid LAD | 0 | - | - | - | - | - |
| 13 | Mid LAD | 2 | - | + | + | - | - |
| 14 | Mid LAD | 1 | + | - | - | - | - |
|
CTO, chronic total coronary occlusion; LAD, left anterior descending artery; OM, obtuse marginal artery; RCA, right coronary artery. |
|||||||
Table 3. Variables related to the percutaneous coronary intervention
| Patient | Access | Guide catheter | Guidewires | Successful guidewire | Microcatheter | Stent | Total length treated with stent | Successful intervention | Contrast |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Femoral | AR 2-6 Fr | 2 | Miracle 3g | Yes | CYPHER | 33 mm | Yes | 241 mL |
| 2 | Femoral-radial | EBU 3.5-6 Fr | 2 | PT Graphics | No | CYPHER SELECT | 23 mm | Yes | 218 mL |
| 3 | Radial | AL 2-6 Fr | 2 | Miracle Bros 3 | No | CYPHER | 33 mm | Yes | 117 mL |
| 4 | Femoral-radial | EBU 4-8 Fr | 3 | Miracle 6 | Yes | TAXUS Liberté x3 | 96 mm | No | 132 mL |
| 5 | Femoral-radial | JR 4-6 Fr | 2 | Miracle 3 | No | TAXUS Liberté XIENCE V x2 | 83 mm | Yes | 300 mL |
| 6 | Femoral | EBU 3.5-7 Fr | Unknown | Pilot 50 | Yes | CYPHER x3 Vision x1 | 66 mm | Yes | 468 mL |
| 7 | Femoral | JL 4-7 Fr | Unknown | Miracle 3 | Yes | CYPHER | 33 mm | Yes | 158 mL |
| 8 | Femoral-radial | JR 4-6 Fr | Unknown | Fielder FC | Yes | CYPHER | 18 mm | Yes | 182 mL |
| 9 | Femoral | EBU 3.5-6 Fr | 2 | Fielder XT | Yes | XIENCE V Prime x2 | 43 mm | Yes | 225 mL |
| 10 | Femoral-radial | Hockey S | Unknown | Fielder XT | Yes | Xience xpedition | 22 mm | Yes | 240 mL |
| 11 | Femoral | JR 4-6 Fr | 1 | Gaia third | Yes | Resolute Onyx | 38 mm | Yes | 224 mL |
| 12 | Radial | EBU 4-6 Fr | 1 | Fielder XT | No | Resolute Integrity | 22 mm | Yes | 154 mL |
| 13 | Radial | EBU 4-7 Fr | 2 | Fielder XT | Yes | BioMatrix x2 | 53 mm | Yes | 129 mL |
| 14 | Femoral-femoral | EBU 4-8 Fr | 2 | Fielder XT | Yes | PROMUS Element | 18 mm | Yes | 200 mL |
The occluded artery was the left anterior descending artery in 8 patients (57%), the right coronary artery in 4 patients (29%) and the left circumflex artery in 2 patients (14%). Only in one case (patient 8) a distal occlusion was treated, while the remaining patients showed proximal or mid segment occlusions. No patient had more than 1 CTO.
Fifty percent of the patients (7 patients) had 1 vessel disease, 28% (4 patients) 2 vessels disease and 21% (3 patients) 3 vessels disease. Other non-CTO severe lesions were treated before the CTO procedure in those patients with multivessel disease. The mean J-CTO13 score was 1 (± 0.78).
In 11 patients (79%), the femoral artery was used as the main access, in 5 of these patients the radial approach was used simultaneously. In the remaining 3 patients (21%), only radial approach was used. Contralateral injections were used in 6 interventions (3%).
In all cases, the anterograde strategy was used. An average of 1.9 guidewires were used per procedure and an intravascular ultrasound was performed in 3 of the procedures. In 10 patients (71%), at least one dedicated microcatheter was used. In every case, a guidewire escalation approach was performed, starting with guidewires with lower tip load and penetration capacity to guides with higher tip load and penetration capacity.
In every case, drug-eluting stents were deployed with an average of 1.57 stents per patient and an average stent length of 41.5 mm.
The amount of contrast used in the procedures went from 117 to 468 mL with a median of 209 mL. However, no events of contrast-induced nephropathy were reported.
The PCI was successful in 13 patients (92.8%). The only failed attempt (patient 4) was a mid-segment left anterior descending artery CTO with a J-CTO score of 1. Three drug-eluting stents were deployed but final Thrombolysis in Myocardial Infarction flow was 1. Follow-up coronary angiography showed no improvement in coronary flow. No further attempts were made to recanalize the vessel.
In-hospital results
Regarding cardiovascular events during hospitalisation after the PCI, only 1 patient had a procedural myocardial injury, with a significant increase of myocardial necrosis markers (troponin I peak of 9 µg/dL for a 99th percentile upper reference limit of 0.06 µg/dL) but without haemodynamic impairment or new regional wall motion abnormality. No radiodermatitis was reported.
Clinical and angiographic follow-up
During a median follow-up of 33.5 months [20-50] mortality rate was around 28.5% (4 out of 14 CTO patients). Among these, 2 deaths were due to sudden cardiac death, 1 to advanced stage pancreatic carcinoma and 1 death was due to suspected ruptured right iliac artery aneurysm (unrelated to the procedure).
The improvement of left ventricular ejection fraction measured using the Simpson method was confirmed in 6 of the 9 patients who underwent an echocardiogram both before and after the CTO procedure, with 5.8% (± 0.87) of global mean improvement (P = NS).
All patients from centre #1 had a coronary angiography during follow-up. The median time from the CTO PCI to the follow-up angiography was 201 days (161-246). Two patients had in-stent restenosis (20%) after 3 and 37.4 months, respectively (one first-generation drug-eluting stent and one second-generation stent). In both cases reintervention was successful, and new drug-eluting stents were deployed with no further events at follow-up.
CTO versus non-CTO PCI in heart transplant recipients
We compared the results of 14 HTRs in whom one CTO lesion PCI was performed and 36 HTRs with non-CTO lesion PCI over the same period of time.
The long-term follow-up of patients was 100% (CTO and non-CTO patients), with median follow-up of 27 months [14.7-50.2], a minimum of 3, and a maximum of 124 months.
No statistically significant differences were found when the clinical and demographic baseline variables were compared in both groups (table 4).
Table 4. Demographic characteristics of heart recipient patients with percutaneous coronary intervention. Period 2005-2016
| Variable | CTO | Non-CTO stenosis | P |
|---|---|---|---|
| Patient | 14 | 36 | - |
| Mean age, y [IQR] | 57 [46.2-66] | 62 [47.5-68.7] | .552 |
| Woman (%) | 14.3 | 22.2 | .538 |
| HT (%) | 57.1 | 63.9 | .667 |
| DM (%) | 21.4 | 22.2 | .953 |
| DL (%) | 50 | 52.8 | .863 |
| Actual smoker/former smoker (%) | 50 | 23 | .685 |
| CKD (%) | 90 | 74.3 | .303 |
| Statins (%) | 90 | 82.4 | .572 |
| Median age at the time of heart transplant in years [IQR] | 44 [37.5-58] | 46 [35.7-54] | .709 |
| Drug-eluting stent (%) | 100 | 100 | 1 |
| Acute rejection (%) | 70 | 51.4 | .259 |
| Mean LVEF (%) (standard deviation) | 50 (11.4) | 52.7 (10.2) | .558 |
|
CKD, chronic kidney disease; CTO, chronic total coronary occlusion; DL, dyslipidemia; DM, diabetes mellitus; HT, hypertension; IQR, interquartile range; LVEF, left ventricular ejection fraction. |
|||
During follow-up, there were no statistically significant differences in the all-cause mortality rate and cardiovascular mortality rate between both groups (table 5). The mortality rate was 28% (4 out of 14) in the CTO group and 14% (5 out of 36) in the non-CTO group (P = .245). The cardiovascular mortality rate was 21% (3 out of 14) in the CTO group and 8% (3 out of 36) in the non-CTO group (P = .331).
Table 5. Major cardiac events. Period 2005-2016
| Variable | CTO | Non-CTO | P Fisher´s exact test |
|---|---|---|---|
| HF or AMI hospital admissions- n (%) | 4 (28.6%) | 11 (31.4%) | .844 |
| Death-n (%) | 4 (28.6%) | 5 (13.9%) | .245 |
| Cardiovascular death-n (%) | 3 (21.4%) | 3 (8.3%) | .331 |
| Sustained ventricular arrhythmias-n (%) | 0 (0%) | 0 (0%) | - |
|
AMI, acute myocardial infarction; CTO, chronic total coronary occlusion; HF, heart failure. |
|||
Regarding the 3 patients with cardiovascular death in the non-CTO group, all had severe CAV and advanced chronic kidney disease: 2 of them died due to refractory heart failure and 1 due to humoral rejection. The causes of death in the CTO group are described in the dedicated section.
The rate of readmission for heart failure or acute myocardial infarction during follow-up was 28.6% (4 of 14) in the CTO group and 31.4% (11 of 35) in the non-CTO group (P = .844).
No ventricular tachycardias were reported at follow-up in any of the groups.
DISCUSSION
There is little experience in the percutaneous management of CAVs. Until recently, CAV was considered a progressive and irreversible process with few therapeutic options. Since treatment with m-TOR inhibitors to delay the progression of the disease is effective in many cases, the PCI has emerged as an adjunctive treatment in these patients with good results.14 However, the results of the CTO PCI in this context are largely unknown. Considering CAV as a diffuse vessel disease, the clinical and angiographic outcomes of CTO recanalization in transplanted hearts are difficult to extrapolate based on the results observed in atherosclerotic coronary occlusions.
As far as we know, this is the very first systematic study on the prevalence of CTOs in HTRs. Also, it is the very first analysis of PCI results of CTOs in this population.
We have found a PCI-target lesion procedure success rate very similar to that of studies published in CTO secondary to atherosclerotic coronary heart disease (92.8%). The rate of intrastent restenosis is similar to that found in large series of PCI in CTO in non-transplanted patients. Although the procedures were performed over an extended period of time, there were no differences in success rate between older and recent procedures in witch we cannot exclude an inclusion bias.
Regarding the safety of PCI, there has only been one procedural myocardial injury with elevation of cardiac necrosis markers, although there has been no haemodynamic involvement or impact on the ejection fraction of the left ventricle.
Despite the fact that our patients were treated with immunosuppressants, we found no contrastinduced nephropathy events, which may reflect that an optimization in contrast volume, adequate prophylaxis and the correct selection of cases can decrease the rate of renal impairment.
Unexpectedly, given the progressive nature of CAV, only 2 of the cases developed in-stent restenosis that was successfully treated in both cases with no other restenosis during long-term follow-up.
Consistent with the poor clinical prognosis of CAV in series already published, in our registry there is a high mortality rate during the long-term follow-up (28.5%). Of the 3 patients who died of cardiovascular death, 2 of them developed sudden death (271 and 856 days after PCI to CTO) and 1 patient died due to hypovolemic shock secondary to the suspected and unrelated to the procedure ruptured iliac artery aneurysm.
Although statistically irrelevant, there is an apparent higher mortality rate in HTRs with CTOs compared to HTRs with nonocclusive stenosis, which may have to do with a more advanced stage of CAV or with more associated comorbidities.
We should mention here that in our study, the CTO recanalization produces a statistically nonsignificant improvement of the ejection fraction.
Accordingly, our study suggests that following an adequate selection of cases, centres experienced in the management of CTOs can feasibly handle PCIs on CTOs in transplanted hearts with a high PCI-target lesion procedure success and low periprocedural complications. However, probably due to the underlying disease, medium and long-term results are still poor with a high mortality rate and a significant rate of restenosis.
Limitations
Although it should be noted here that this is the first study ever on this subject, there are several limitations. First of all, its retrospective nature. Secondly, the number of heart transplant patients is limited despite combining the experience of 2 high volume HTR centres. Nevertheless, we should keep in mind that the overall experience with these patients is very scarce considering their special characteristics.
In addition, the proportion of HTRs in our centres who undergo CTO PCI is low. Considering the CAV inner nature, it is not rare to find CTO of distal vessels or diffuse distal disease making any PCI attempts futile if not impossible. In fact, most of our patients who underwent PCIs showed CTOs in proximal segments with a good distal vessel. We could conclude that our procedures were performed in highly selected patients. This would be a bias that would be favoring a high success rate by selecting less complex cases (medium J-CTO score 1).
When it comes to the statistical analysis, we should mention here that it has not been possible to perform more powerful analyses such as propensity score analyses due to the low number of patients included in the registry.
However, despite these limitations, we believe that our study is relevant because it shows the experience accumulated over the years in 2 high heart transplant volume centres and because it is the very first study on this subject with a long-term follow-up of the patients.
CONCLUSIONS
Coronary CTO is a common condition in HTRs. PCI is feasible in centres with extensive experience conducting CTO procedures and in selected patients, with a PCI-target lesion procedure success and periprocedural complications rate similar to global CTO procedures and non-CTO PCIs. The rate of in-stent restenosis in HTRs is similar to that found in large series of CTO PCIs in non-transplanted patients. There is a non-significantly higher all-cause and cardiovascular mortality in HTRs undergoing a CTO PCIs compared to those with non-CTO PCIs. This may have to do with more advanced stages of the CAV.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- CTOs are frequent in the context of allograft vasculopathy in heart recipient patients, being usually managed with medical treatment due to the technical difficulty and the poor results of PCIs in this population.
WHAT DOES THIS STUDY ADD?
- In this study we concluded that performing PCIs of CTOs in selected patients with heart transplantations is feasible in experienced centres, with a PCI-target lesion procedure success rates and hospital complications similar to that those of heart recipients with non-CTO lesions.
REFERENCES
1. Behrendt, D, Ganz, P, Fang, J. Cardiac allograft vasculopathy. Curr Opin Cardiol. 2000;15:422-429.
2. Suzuki J, Ogawa M, Hirata Y, Nagai R, Isobe M. Effects of immunoglobulin to prevent coronary allograft vasculopathy in heart transplantation. Expert Opin Ther Targets. 2012;16:783-789.
3. Daly KP, Seifert ME, Chandraker A, et al. VEGF-C, VEGF-A and related angiogenesis factors as biomarkers of allograft vasculopathy in cardiac transplant recipients. J Heart Lung Transplant. 2013;32:120-128.
4. Costello JP, Mohanakumar T, Nath DS. Mechanisms of chronic cardiac allograft rejection. Tex Heart Inst J. 2013;40:395-399.
5. Tuzcu EM, De Franco AC, Goormastic M, et al. Dichotomous pattern of coronary atherosclerosis 1 to 9 years after transplantation:insights from systematic intravascular ultrasound imaging. J Am Coll Cardiol. 1996;27:839-846.
6. Tanaka K, Li H, Curran PJ, et al. Usefulness and safety of percutaneous coronary interventions for cardiac transplant vasculopathy. Am J Cardiol. 2006;97:1192-1197.
7. Morino Y, Kimura T, Hayashi Y, et al. In-hospital Outcomes of Percutaneous Coronary Intervention in patients with Chronic Total Occlusion. Insights from the J-CTO registry. JACC Cardiovasc Interv. 2010;3:143-151.
8. Brilakis ES, Banerjee S, Karmpaliotis D, et al. Procedural outcomes of chronic total occlusion percutaneous coronary intervention:a report from the NCDR (National Cardiovascular Data Registry). JACC Cardiovasc Interv. 2015;8:245-253.
9. Konstantinidis NV, Werner GS, Deftereos S, et al Temporal Trends in Chronic Total Occlusion Interventions in Europe. Circ Cardiovasc Interv. 2018;11:e006229.
10. Di Mario C, Werner GS, Sianos G, et al. European perspective in the recanalisation of Chronic Total Occlusions (CTO):consensus document from the EuroCTO Club. EuroIntervention. 2007;3:30-43.
11. Hicks KA, Tcheng JE, Bozkurt B, et al. 2014 ACC/AHA Key Data Elements and Definitions for Cardiovascular Endpoint Events in Clinical Trials:A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (Writing Committee to Develop Cardiovascular Endpoints Data Standards). J Am Coll Cardiol. 2015;66:403-469.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40:237-269.
13. Morino Y, Abe M, Morimoto T et al. Predicting successful guidewire crossing through chronic total occlusion of native coronary lesions within 30 minutes:the J-CTO (Multicenter CTO Registry in Japan) score as a difficulty grading and time assessment tool. JACC Cardiovasc Interv. 2011;4:213-221.
14. Benza RL, Zoghbi GJ, Tallaj J et al. Palliation of allograft vasculopathy with transluminal angioplasty:a decade of experience. J Am Coll Cardiol. 2004;43:1973-1981.