Article
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España
ABSTRACT
Introduction and objectives: Distal embolization and no-reflow are common complications in primary angioplasty and the information available on the role played by the deflation speed of the stent delivery system is scarce. Our aim is to analyze how the deflation speed of the stent delivery system impacts the results of primary angioplasty.
Methods: From December 2016 through February 2019, all consecutive patients with ST-segment elevation myocardial infarction undergoing urgent coronary angiography at our institution and who were eligible for thrombectomy, IIB-IIIA inhibitors, and direct stenting were randomized in a 1:1 ratio to rapid (group 1, n = 103) or slow deflation of the stent delivery system, at 1 atm/second, (group 2, n = 107). Pre- and postdilatation was not allowed per protocol. The primary outcomes were myocardial blush ≥ 2 and ST-segment resolution ≥ 70% while the size of myocardial damage, the ejection fraction both at discharge and at the 12-month follow-up, and the overall and 12-month cardiovascular mortality rates were the secondary outcomes.
Results: The study was stopped prematurely with 50% of the estimated sample size due to futility. Myocardial blush ≥ 2 occurred in 77 (74.7%) vs 79 (75.2%) of the patients, P = .93, and ST-segment resolution ≥ 70% occurred in 54 (53.9%) vs 59 (55.5%) of the patients, P = .75 in groups 1 and 2, respectively without any differences being reported in any of the secondary endpoints.
Conclusions: In our series, the deflation speed of the stent delivery system in primary angioplasty did not modify the myocardial blush ≥ 2, the ST-segment resolution ≥ 70% or impacted the clinical outcomes, the size of myocardial infarction according to the biomarkers or the ejection fraction.
Keywords: Primary angioplasty. ST-segment-elevation myocardial infarction. No-reflow. ST-segment resolution. Myocardial blush.
RESUMEN
Introducción y objetivos: La embolización distal y el fenómeno de no-reflow son complicaciones frecuentes de la angioplastia primaria. La información disponible sobre la influencia de la velocidad de desinflado del sistema de liberación del stent es escasa. Nuestro objetivo es analizar la influencia de este factor en los resultados de la angioplastia primaria.
Métodos: Entre diciembre de 2016 y febrero de 2019, todos los pacientes consecutivos con infarto de miocardio con elevación del segmento ST sometidos a coronariografía urgente en nuestro centro y que eran susceptibles de trombectomía, inhibidores de IIB-IIIA e implante directo de stent fueron aleatorizados 1:1 a un desinflado rápido del sistema de liberación (grupo 1, n = 103) o a un desinflado lento a 1 atm/s (grupo 2, n = 107). Por protocolo, no se permitió la predilatación previa ni posterior. Los objetivos primarios fueron el grado de blush miocárdico ≥ 2 y la resolución del segmento ST ≥ 70%. Los objetivos secundarios fueron el tamaño del infarto, la fracción de eyección al alta y a los 12 meses, y las mortalidades total y cardiovascular a los 12 meses.
Resultados: El estudio se detuvo prematuramente con el 50% del tamaño muestral calculado por futilidad. Se encontró blush ≥ 2 en 77 (74,7%) frente a 79 (75,2%) pacientes (p = 0,93) y resolución del segmento ST ≥ 70% en 54 (53,9%) frente a 59 (55,5%) pacientes (p = 0,75) en los grupos 1 y 2, respectivamente, sin diferencias en ninguno de los objetivos secundarios.
Conclusiones: En nuestra serie, la velocidad de desinflado del sistema de liberación del stent en la angioplastia primaria no modificó el blush miocárdico ni la resolución del segmento ST, y tampoco demostró tener influencia en los resultados clínicos, el tamaño del infarto según los biomarcadores ni la fracción de eyección.
Palabras clave: Angioplastia primaria. Infarto con elevacion del segmento ST. No-reflow. Resolucion del segmento ST. Blush miocardico.
Abbreviations
MB: myocardial blush. pPCI: primary percutaneous coronary intervention. STR: ST-segment resolution. STEMI: ST-segment elevation myocardial infarction. TIMI: Thrombolysis in Myocardial Infarction.
INTRODUCTION
Distal embolization and slow coronary flow often limit the success of primary percutaneous coronary angioplasty (pPCI). In 25% to 50% of the cases, despite satisfactory flow restoration, poor microvascular reperfusion can be seen, which leads to worse prognoses.1 This is a field of ongoing discussion because strategies that initially showed positive results have later been questioned like direct stenting,2 thrombus aspiration,3,4 and the administration of beta-blockers,5 and IIB-IIIA inhibitors.6
It has been confirmed that aggressive balloon dilatation with a high balloon-to-artery ratio may favor the presence of no-reflow and it has been speculated that the deflation speed of the stent delivery system may impact the results too, although the information available on this regard is scarce.
No-reflow may be due to different pathophysiological factors such as distal embolization, ischemia-reperfusion injury, and the susceptibility of coronary microcirculation to injury.7,8 Rapid stent balloon deflation may trigger the so-called siphon effect and rapid changes in coronary hemodynamics that can be associated with distal embolization, and microcirculatory dysfunction.9 As part of a published report, the investigators built an in vitro experimental study and combined it with a computer model to eventually find that the wall shear stress due to the different balloon deflation strategies used triggered differences in the flow final velocity as well.
Our objective is to analyze the impact of the deflation speed of the stent delivery system on myocardial blush (MB), and the ST-segment resolution (STR) in the acute phase, as well as the prognosis and ejection fraction at the 12-month follow-up.
METHODS
Patients
A randomized, parallel, single-center study was conducted with a 24-hour program of pPCI including 440 000 patients. Recruitment was carried out by convenience sampling and eligible patients were all consecutive subjects with ST-segment elevation myocardial infarction (STEMI) referred to receive a pPCI who had a culprit lesion eligible for direct stenting. Patients should have ST-segment elevations ≥ 0.1 mV in 2 contiguous leads or new left bundle branch block.
Exclusion criteria were contraindications to acetylsalicylic acid, clopidogrel or IIB-IIIA inhibitors, impossibility to complete the follow-up, life expectancy < 12 months, lesion not amenable to direct stenting, culprit lesions located at grafts or in-stent thrombosis, and previous oral anticoagulation.
After performing the coronary angiography, the patients who met the inclusion criteria and had no exclusion criteria gave their initial oral consent and were allocated by simple randomization through a computer-generated list that would create individual codes. These codes were inserted one by one in identical envelopes—prepared by personnel not involved in the study—that were thick enough so the codes could not be seen. All patients were asked to confirm their participation by giving their written informed consent within 24 hours. The study protocol was designed in full compliance with the ethical guidelines of the 1975 Declaration of Helsinki as shown in a prior approval granted by the center human research committee.
Parallel groups were created by a) direct stenting with fast deflation of the stent delivery system after 20 seconds of balloon inflation (group 1), or b) direct stenting with slow deflation at 1 atm/second after the same period of inflation (group 2).
Procedure
Patients and outcome evaluators were blind to the procedure. To minimize variability and any potential confounders the protocol was strict and included the administration of 250 mg of acetylsalicylic acid followed by 600 mg of clopidogrel at the first medical contact (according to the myocardial infarction protocol of our unit), 70 mg/kg of IV heparin, and IV abciximab or tirofiban at the beginning of the procedure for a 12-hour administration course. Manual thrombectomy and posdilatation of the stent were performed systematically, but implantation of a second stent was not allowed per protocol. Intention-to-treat and per protocol analyses were performed. The former dictated the main analysis. The volume of contrast per injection was 6 mL administered for 3 seconds into the left main coronary artery followed by 4 mL administered for 2 seconds into the right coronary artery using the ACIST device (ACIST Medical Systems Inc., United States). Intracoronary nitroglycerine (100 µg to 200 µg) was administered before the final injection to assess MB. Myocardial blush was studied in the right anterior oblique 20-degree projection with 20-degree caudal angulation, and in the left anterior oblique 45-degree projection with 20-degree cranial angulation regarding the left main coronary artery, and in the anteroposterior projection with 20-degree cranial angulation regarding the right coronary artery. Recordings were acquired at 30 images/second without image magnification with a prolonged duration until the venous phase of the myocardial circulation was completed.
Within the first 30 minutes upon arrival to the coronary care unit, patients underwent a 12-lead electrocardiogram and blood samples were obtained for troponin I assessment 6 and 24 hours after the procedure, as well as additional measurements until a reduction in the levels reported was confirmed.
Optimal medical management according to guidelines was recommended with statins, beta-blockers, or renin-angiotensin system blockers. Also, dual antiplatelet therapy was indicated for 12 months. Switching to ticagrelor during admission was also recommended in the absence of significant risk of bleeding.
Outcomes
The 2 primary endpoints were how the deflation speed of the stent delivery system impacted MB at the end of the procedure, and the STR. The final MB was analyzed blindly by an external core laboratory in a different region and the variable analyzed was the percentage of MB grade ≥ 2 vs < 2 between both groups by visual assessment. Two interventional cardiologists with > 10 years of experience grading MBs10 were involved in the evaluation and, in case of disagreement, a third opinion was requested. The STR was analyzed by evaluators not involved in the study who were blind to the procedure. The J-point was manually identified with respect to the nearest 0.5 mm in all leads except in the aVR lead. Using the TP segment as the isoelectric baseline interval, the extent of the ST-segment elevation with respect to the nearest 0.05 mV was measured 80 ms after the J-point. The STR was estimated by a reduction in the sum of the ST-segment elevation in all leads except in the aVR from the baseline ECG compared to the ECG performed upon arrival at the coronary care unit. The variable was a binary outcome, the ≥ 70% resolution of the sum of millimeters of ST-elevation between both recordings.
The secondary endpoints were: a) size of the myocardial damage comparing the maximum levels of troponin I; b) ejection fraction at discharge; c) ejection fraction at 12 months; d) all-cause mortality rate at 12 months; and e) 12-month cardiovascular mortality rate.
Definitions
Angiographic thrombus burden was defined according to Sianos’ classification11 while collateral supply was defined according to Rentrop classification.12
Quantitative coronary angiography
The Medis Suite XA system (Medis Medical Imaging, Israel) was used for the analysis according to the experts’ standards.13 Lesion length was measured once the vessel flow had been restored after thrombectomy. The diameter parameters were taken at the end of the procedure after the stent was deployed due to the difficulties reported while performing analyses in thrombotic vessels. The following data were used: reference vessel diameter (the average lumen diameter assumed without atherosclerotic disease), minimal lumen diameter, postoperative stenosis, and the stent-to-artery ratio.
Sample size calculations
Based on a primary endpoint of STR of 50% in the control group14,15 and an increase up to 62.5% in the procedural group following, the principle of minimum clinically significant difference between treatments of 25%,16 and a dropout rate of 10%, 420 patients, 210 per group, were needed.
Interim analysis
Given the uncertainty of the results and the lack of data available on the medical literature, an interim futility analysis was planned after recruiting 50% of the sample size.
Statistical analysis
Quantitative variables with normal distribution were expressed as means and standard deviation, and those without a normal distribution as median and interquartile range. Categorical variables were expressed as absolute values and percentages. The mean comparison was carried out using the Student t test in normal distribution or the Mann-Whitney U test when that assumption was not met. The chi-square test or Fisher’s exact test were used to compare proportions. Two-tailed tests were used to analyze all studies. P values ≤ .05 were considered statistically significant. A logistic regression analysis was performed to adjust for possible imbalances and measure how the deflation speed rate of the stent delivery system impacted each of the 2 primary endpoints. The variables that met the 2 criteria of a reasonable association with the outcomes and P values < .20 in the univariate analysis were tested in the multivariate analysis. The calculations were performed using the SPSS 27.0.0.0 statistical software (IBM Corp, United States).
RESULTS
Baseline
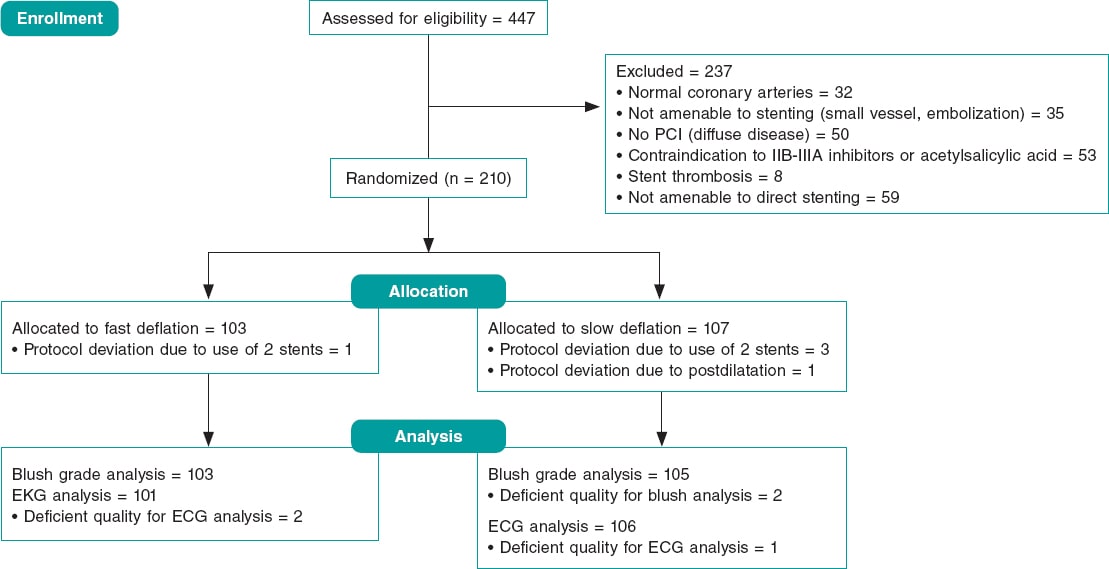
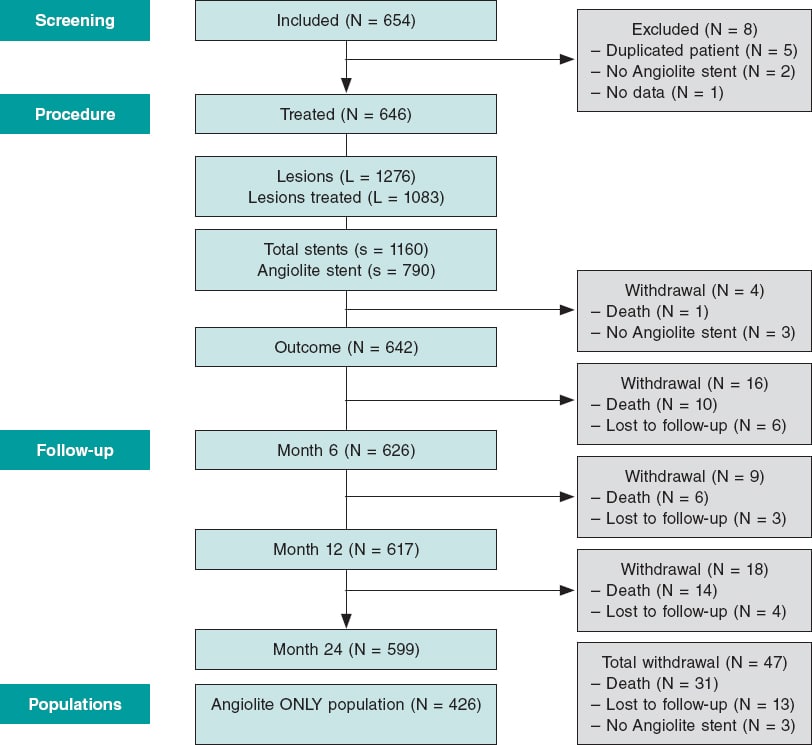
From December 2016 through February 2019 a total of 447 patients were referred to our cath lab with a diagnosis of STEMI (figure 1, flow diagram). A total of 237 (53%) were not eligible for randomization and the remaining 210 (47%) were allocated to fast (103, 49%) or slow balloon deflation (107, 51%). The initially calculated sample size was 420 patients but, after an interim analysis with 50% of the sample recruited, the study was terminated early due to futility. There was 1 protocol violation in the first group and 4 in the second group. The intention-to-treat analysis is seen in this section and the per protocol analysis on tables 3 to 5 of the supplementary data. The baseline and procedural characteristics of the study cohort are shown on table 1 and table 2. There were no statistical differences between both groups although, despite the randomization process, there was a non-significant trend towards a larger vessel diameter in the slow deflation group. All cases were performed with 6-Fr guiding catheters.
Figure 1. Study flow-chart. ECG, electrocardiogram; PCI, percutaneous coronary intervention.
Table 1. Baseline clinical characteristics
| Fast deflation N = 103 | Slow deflation N = 107 | P | |
|---|---|---|---|
| Age | 59.73 (10.56) | 59.33 (10.71) | .78 |
| Sex (female) | 26 (25.2) | 20 (18.7) | .25 |
| Diabetes | 14 (13.6) | 21 (19.6) | .24 |
| Hypertension | 40 (38.9) | 48 (44.8) | .37 |
| Hypercholesterolemia | 37 (35.9) | 45 (42.1) | .36 |
| Smoking | 65 (63.1) | 71 (66.3) | .62 |
| Previous myocardial infarction | 4 (3.9) | 6 (5.6) | .75 |
| Previous percutaneous coronary intervention | 3 (2.9) | 4 (3.7) | 1.00 |
| Previous coronary artery bypass graft | 0 (0) | 1 (0.1) | 1.00 |
| Previous stroke | 1 (0.1) | 0 (0.0) | .49 |
| Creatinine clearance levels < 60 mL/min | 14 (13.6) | 22 (20.5) | .18 |
| Blood pressure at admission | 123.4 (30.8) | 129.6 (28) | .13 |
| Shock | 4 (3.9) | 1 (0.09) | .21 |
| Radial access | 103 (100) | 105 (98.1) | .50 |
| Number of diseased vessels | 1.38 (0.61) | 1.45(0.66) | .42 |
| Total ischemic time | 192 (125-295) | 169 (120-260) | .21 |
| First medical visit to balloon time | 87 (66-130) | 80 (65-114) | .22 |
| ST elevation before procedure (mm) | 11.40 (6.74) | 12.63 (8.06) | .24 |
|
Quantitative variables with normal distribution are expressed as means and standard deviation (SD), variables with non-normal distribution as median and interquartile range, and categorical variables are expressed as absolute values and percentages. |
|||
Table 2. Characteristics of the procedure
| Fast deflation N = 103 | Slow deflation N = 107 | P | |
|---|---|---|---|
| Vessel | .60 | ||
| Left anterior descending coronary artert | 44 (42.7) | 40 (37.4) | |
| Left circumflex artery | 13 (12.6) | 18 (16.8) | |
| Right coronary artery | 46 (44.7) | 49 (45.8) | |
| Preoperative TIMI ≥ grade 2 flowa | 10 (9.7) | 17 (15.9) | .21 |
| Rentrop ≥ 2 | 15 (14.6) | 19 (17.8) | .53 |
| Thrombus grade score ≥ 4 | 46 (44.6) | 50 (46.7) | .76 |
| Drug-eluting stent | 100 (97.1) | 101 (94.4) | .50 |
| Percent diameter stenosis | 99.28 (3.43) | 98.89 (6.48) | .58 |
| RVDb | 2.74 (0.42) | 2.86 (0.47) | .07 |
| Lesion length | 14.07 (5.94) | 13.44 (4.71) | .39 |
| Stent diameter | 3.23 (0.47) | 3.32 (0.57) | .17 |
| Maximum inflation pressure | 14.68 (1.48) | 14.77 (1.69) | .67 |
| MLDc | 2.89 (0.38) | 3.00 (0.49) | .06 |
| Minimum lumen diameter | 2.63 (0.39) | 2.67 (0.48) | .48 |
| Postoperative stenosis | 8.92 (4.75) | 11.20 (6.25) | .01 |
| Stent-to-artery ratio | 1.05 (0.07) | 1.05 (0.08) | .95 |
|
Quantitative variables with normal distribution are expressed as means and standard deviation (SD), variables with non-normal distribution as median and interquartile range, and categorical variables are expressed as absolute values and percentages. a TIMI, Thrombolysis in Myocardial Infarction risk score. b RVD, reference vessel diameter after the procedure. c MLD, maximum lumen diameter after the procedure. |
|||
| Fast deflation N = 103 | Slow deflation N = 107 | P | |
|---|---|---|---|
| Myocardial blush ≥ 2 | 77 (74.7) | 79 (75.2) | .93 |
| Postoperative ST-segment elevation (mm) | 4.26 (5.19) | 4.03 (4.69) | .73 |
| ST-segment elevation resolution (mm) | 7.03 (6.99) | 8.56 (8.11) | .15 |
| Percentage of resolution (%) | 64.97 (33.35) | 65.40 (34.69) | .92 |
| Resolution ≥ 70 % | 54 (53.4) | 59 (55.6) | .75 |
| TIMI grade flow after the procedure | .38 | ||
| 0 | 1 | 0 | |
| 1 | 0 | 1 | |
| 2 | 5 | 9 | |
| 3 | 97 | 97 | |
| Maximum troponin-I levels | 47.84 (14-129) | 72 (29.7-144.75) | .14 |
| Ejection fraction at discharge | 53.9 (8.58) | 54.62 (8.71) | .55 |
| Ejection fraction at 12 months | 57.43 (8.20) | 57.75 (6.48) | .76 |
| In-hospital mortality rate | 1 (0.9) | 2 (1.8) | 1.00 |
| Overall mortality rate at 12 months | 3 (2.9) | 3 (2.8) | 1.00 |
| Cardiovascular mortality rate at 12 months | 2 (1.9) | 3 (2.8) | 1.00 |
| Myocardial infarction | 1 (0.9) | 1 (0.9) | 1.00 |
| Target vessel revascularization | 0 | 1 (0.9) | 1.00 |
|
Quantitative variables with normal distribution are expressed as means and standard deviation (SD), variables with non-normal distribution as median and interquartile range, and categorical variables are expressed as absolute values and percentages. TIMI, Thrombolysis in Myocardial Infarction risk score. |
|||
Table 4. Predictors of myocardial blush ≥ 2 and ST-segment resolution
| OR | 95%CI | P | |
|---|---|---|---|
| Predictors of myocardial blush ≥ 2 | |||
| Systolic blood pressure at admission | 1.02 | 1.02-1.03 | .011 |
| Creatinine clearance levels <60 mL/min | 0.29 | 0.13-0.66 | .003 |
| Postoperative maximum lumen diameter | 3.08 | 1.24-7.63 | .015 |
| Hypertension | 0.52 | 0.26-1.06 | .074 |
| Predictors of ST-segment resolution | |||
| Diabetes | 0.16 | 0.06-0.43 | < .001 |
| Previous myocardial infarction | 13.54 | 1.47-124.91 | .022 |
| Left anterior descending coronary artery | 0.46 | 0.24-0.91 | .025 |
| Preoperative TIMI grade flow ≥ 2 | 3.95 | 1.36-11.46 | .011 |
| Postoperative TIMI grade 3 flow | 7.10 | 1.76-28.68 | .006 |
| Rentrop grade ≥ 2 collateral circulation | 0.31 | 0.13-0.75 | .010 |
|
Quantitative variables with normal distribution are expressed as means and standard deviation (SD), variables with non-normal distribution as median and interquartile range, and categorical variables are expressed as absolute values and percentages. 95%CI, 95% confidence interval; OR, odds ratio; TIMI, Thrombolysis in Myocardial Infarction risk score. |
|||
Endpoints
The primary endpoint, MB grade ≥ 2compared to < 2, occurred in 77 (74.7%) vs 79 (75.2%), P = .93, of the patients, and STR ≥ 70% in 54 (53.9%) vs 59 (55.5%), P = .75, of the patients from the rapid and slow deflation groups, respectively. Also, there were no differences in any of the secondary endpoints regarding the size of myocardial damage, the ejection fraction at discharge, the ejection fraction at 12 months, the overall mortality rate at 12 months or in the cardiovascular mortality rate at 12 months (table 3).
Predictors of myocardial blush
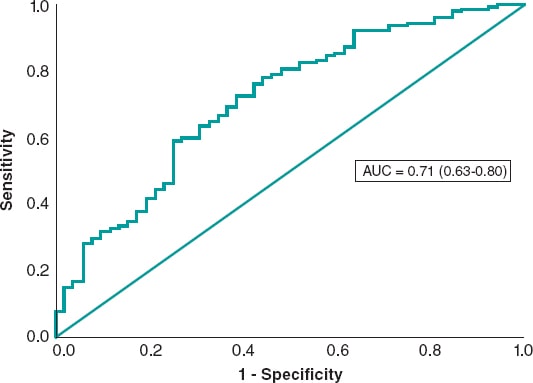
The univariate analysis was performed with the variables shown on table 1 of the supplementary data. The variables age, creatinine clearance levels < 60 mL/min, postoperative maximum lumen diameter, past medical history of hypertension, systolic blood pressure at admission, Rentrop grade ≥ 2 collateral circulation, and the first medical contact to balloon time were tested using a logistic regression model. Systolic blood pressure at admission, creatinine clearance levels < 60 mL/min, and the postoperative maximum lumen diameter were predictors of blush ≥ 2 while in the final model hypertension remained with P values = .074 (table 4). The predictive power was moderate with an area under the ROC curve of 0.71 (0.63-0.80) (figure 2).
Figure 2. Receiver operating characteristic curve of the logistic regression model for myocardial blush prediction.
Predictors of ST-segment resolution ≥ 70%
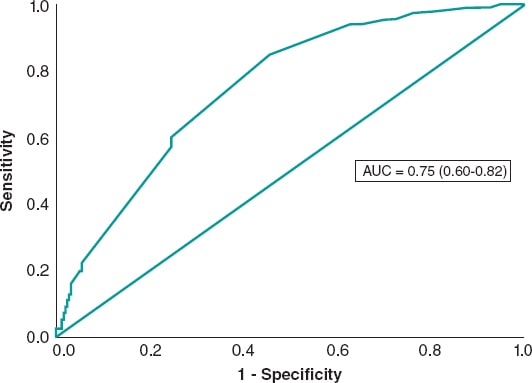
The univariate analysis was performed with the variables listed on table 2 of the supplementary data. The variables tested in the multivariate analysis were sex, diabetes, hypercholesterolemia, smoking, shock, left anterior descending coronary artery, previous myocardial infarction, preoperative TIMI grade ≥ 2 flow, postoperative TIMI grade 3 flow, and Rentrop grade ≥ 2 collateral circulation, number of millimeters of ST elevation before the procedure, and creatinine clearance levels < 60 mL/min. The logistic regression model included diabetes, previous myocardial infarction, left anterior descending coronary artery, preoperative TIMI grade ≥ 2 flow, postoperative TIMI grade 3 flow, and Rentrop grade ≥ 2 collateral circulation as predictors of ST-segment resolution ≥ 70% (table 4). The area under the ROC curve was 0.75 (0.68-0.82) (figure 3).
Figure 3. Receiver operating characteristic curve of the logistic regression model for ST-segment resolution.
Per protocol analysis
Protocol deviation was seen in 5 patients. In the rapid deflation group 2 stents were needed in 1 patient. In the slow deflation group 3 patients received 2 stents followed by 1 postdilatation (figure 1). Tables 3, 4 and 5 of the supplementary data show the per protocol analysis without any significant differences compared to the intention-to-treat analysis.
Missing values
In 2 patients from the slow deflation group, the quality of the angiogram did not allow us to perform a proper analysis. Regarding the electrocardiogram, suboptimal quality was recorded in 2 patients from the rapid deflation group and in 1 patient from the slow deflation group. All of them may be considered as missing values completely at random, which means that the randomization balance was never affected.
DISCUSSION
In this randomized study we assessed how the deflation speed of the stent delivery system impacted myocardial blush ≥ 2, and ST-segment resolution ≥ 70%. The most important findings are: a) the study was stopped with 50% of the predefined sample sized due to futility and neither MB nor STR were modified by the intervention; b) no differences were seen in the size of myocardial damage, ejection fraction at 12 months and discharge or in the all-cause and 12-month cardiovascular mortality rates; c) systolic blood pressure at admission, creatinine clearance levels < 60 mL/min, and postoperative maximum lumen diameter played a role in MB while the past medical history of hypertension would have probably been included in the final model if the sample size would have been larger; and d) STR was influenced by diabetes, previous myocardial infarction, left anterior descending coronary artery, preoperative TIMI grade ≥ 2 flow, postoperative TIMI grade 3 flow, and Rentrop grade ≥ 2 collateral circulation.
The data available on the medical literature on this research topic is significantly scarce and, to our knowledge, only 1 group has provided information. Gu et al.17 also studied the association of balloon deflation during stent deployment with coronary flow and clinical outcomes regarding pPCI in a series of 211 patients. They found that slow deflation led to favorable coronary flow and infarct size compared to conventional rapid deflation. These contradictory results may be justified by the remarkable differences seen between both cohorts. Former studies have reported on the role of balloon inflation,18 thrombectomy,19,20 and IIB-IIIA inhibition21 in the management of MB. In our series, we designed a strict protocol to control these potential confounders, which is why pre- and postdilatation was not allowed, and both thrombus aspiration and IIB-IIIA inhibitors were essential components of the procedure. The study conducted by Gu et al. allowed both pre- and postdilatation while the use of thrombectomy, and IIB-IIIA inhibitors was left to the operator’s discretion. Indeed, predilatation was performed in > 80% of the patients from both groups, postdilatation in roughly 40%, thrombus aspiration in only 20%, and IIB-IIIA inhibitors were administered in 70% of the patients. Undoubtedly, the approach conducted by Gu et al. favored external validity although, in our opinion, the influence of these 4 factors may have influenced the results deeply, mainly when no adjustment was performed through a multivariate analysis. Finally, although closely related, the TIMI frame count and MB are not the same endpoint, and the ST-segment resolution was not assessed in the study conducted by Gu et al. Regarding the clinical endpoints, no differences were seen between the 2 strategies in any of the 2 studies.
As we mentioned, we were not able to show that the deflation speed of the stent delivery system impacted MB. In the multivariate analysis performed, blood pressure levels at admission, creatinine clearance levels, and the postoperative maximum lumen diameter were all predictors of MB while a past medical history of hypertension would have probably reached statistical significance with a larger sample size. Former reports have underlined how blood pressure impacts MB during the procedure.22 Also, patients with hypertension due to an increased microvascular resistance have shown an impaired flow.22 In addition, it has been reported that the adverse event of renal function regarding cardiovascular events may be mediated by an increased microvascular resistance.23 Time to treatment has impacted MB in previous studies.24 In our cohort, there were significant differences in the univariate analysis, but in the last step of the multivariate analysis it was removed from the final model, although it would have probably been present with a larger sample size. However, in the comparison of our series with the aforementioned study, we tested the vessel size as a predictor of MB while this variable was not analyzed in Luca’s study, but it had played a role in previous cohorts.25
Consistent with this, the deflation speed of the stent delivery system did not seem to play a role in STR. We found up to 6 factors that proved its impact on the ST-segment resolution, most of them already described in former studies. As it leads to a lower ST-segment elevation, collateral circulation reduces the impact of pPCI in STR.26 Anterior infarctions with culprit lesion in the left anterior descending coronary artery also led to lower ST-segment recoveryies in previous cohorts.27-29 This was also seen with preoperative TIMI grade < 2 flow, and final TIMI grade flow < 3,14,27,28,30 and diabetes.14,28 In our series, previous myocardial infarction was a predictor of STR, although we found no explanation for this finding.
Limitations
The study was stopped in the interim analysis based on the criterion of futility. However, we do not expect the results of primary endpoints to have been any different with the whole sample size. We could have probably found more predictors and a higher predictive power of the MB and STR models, but this was not the endpoint of our study. The risk profile of the patients was low because the inclusion criteria of direct stenting, use of IIB-IIIA inhibitors, and thrombectomy focused the study on lesions more frequently associated with younger patients with a low bleeding risk and less calcification, which are features associated with better outcomes. This limits the external validity of the study because, as shown on figure 1, roughly 50% of the patients were ineligible to enter the study. This may have also played a role in the lack of differences seen between the study groups. However, as we have already explained, the purpose of our study was to avoid any confounders. Clopidogrel was the P2Y12 inhibitor at the first medical visit according to the protocol of the regional myocardial infarction network of our area. This may also limit the external validity of the results. Myocardial blush was visually assessed and, although it was performed by 2 experienced operators, certain degree of subjectivity cannot be ruled out. The predictive power for both MB and STR was low, but it has also occurred in former series28 being the concordance between those factors described as moderate.31 Finally, we could not find any explanations for the role of previous myocardial infarction predicting STR as this factor was not present in former series.
CONCLUSIONS
In our series, the deflation speed of the stent delivery system in primary angioplasty did not change myocardial blush or ST-segment resolution and no influence was seen on the clinical outcomes, size of myocardial infarction assessed by biomarkers, and ejection fraction at discharge and after 12 months.
FUNDING
The study has been supported by a research grant from Abbott Laboratories.
AUTHORS’ CONTRIBUTIONS
B. Vega, J. M. Vegas, J. Rondan, E. Segovia, and Í. Lozano: design, data mining, manuscript drafting, and manuscript revision. A. Pérez de Prado, C. Cuellas-Ramon, M. López-Benito, T. Benito-González, and F. Hernández-Vázquez: blush measurements, and manuscript revision.
CONFLICTS OF INTEREST
The authors declared no conflict of interests whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Distal embolization and slow coronary flow frequently reduce the success of primary angioplasty.
- Several interventions have been tested but it is a field of ongoing debate because the strategies that showed positive results at the beginning have now been questioned such as direct stenting, thrombus aspiration, and use of beta-blockers and IIB-IIIA inhibitors.
- It has been demonstrated that aggressive balloon dilatation with a high balloon to artery ratio may favor the presence of no-reflow. Also, it has been speculated that the deflation speed of the stent delivery system may impact the results, although the information available on this regard is scarce.
WHAT DOES THIS STUDY ADD?
- Our objective is to analyze how the deflation speed of the stent delivery system impacts myocardial blush, ST-segment resolution in the acute phase, prognosis, and the ejection fraction at 12months.
- The study was prematurely stopped due to futility because the speed of deflation of the stent delivery system did not change the primary outcomes or impacted the size of the infarction, prognosis or the ejection fraction at 12 months whatsoever.
REFERENCES
1. Ito H, Tomooka T, Sakai N, et al. Lack of myocardial perfusion immediately after successful thrombolysis. A predictor of poor recovery of left ventricular function in anterior myocardial infarction. Circulation. 1992;85:1699-1705.
2. Mahmoud KD, Jolly SS, James S, et al. Clinical impact of direct stenting and interaction with thrombus aspiration in patients with ST-segment elevation myocardial infarction undergoing percutaneous coronary intervention:Thrombectomy Trialists Collaboration. Eur Heart J. 2018;39:2472-2479.
3. Frobert O, Lagerqvist B, Olivecrona GK, et al. Thrombus aspiration during ST-segment elevation myocardial infarction. N Engl J Med. 2013;369:1587-1597.
4. Jolly SS, Cairns JA, Yusuf S, et al. Outcomes after thrombus aspiration for ST elevation myocardial infarction:1-year follow-up of the prospective randomised TOTAL trial. Lancet. 2016;387:127-135.
5. Roolvink V, Ibanez B, Ottervanger JP, et al. Early Intravenous Beta-Blockers in Patients With ST-Segment Elevation Myocardial Infarction Before Primary Percutaneous Coronary Intervention. J Am Coll Cardiol. 2016;67:2705-2715.
6. Ellis SG, Tendera M, de Belder MA, et al. Facilitated PCI in patients with ST-elevation myocardial infarction. N Engl J Med. 2008;358:2205-2217.
7. Konijnenberg LSF, Damman P, Duncker DJ, et al. Pathophysiology and diagnosis of coronary microvascular dysfunction in ST-elevation myocardial infarction. Cardiovasc Res. 2020;116:787-805.
8. Dong M, Mu N, Guo F, et al. The beneficial effects of postconditioning on no-reflow phenomenon after percutaneous coronary intervention in patients with ST-elevation acute myocardial infarction. J Thromb Thrombolysis. 2014;38:208-214.
9. Li R, Zijlstra JG, Kamps JA, van Meurs M, Molema G. Abrupt reflow enhances cytokine-induced proinflammatory activation of endothelial cells during simulated shock and resuscitation. Shock. 2014;42:356-364.
10. Perez de Prado A, Fernandez-Vazquez F, Cuellas-Ramon JC, Iglesias-Garriz I. Coronary clearance frame count:a new index of microvascular perfusion. J Thromb Thrombolysis. 2005;19:97-100.
11. Sianos G, Papafaklis MI, Daemen J, et al. Angiographic stent thrombosis after routine use of drug-eluting stents in ST-segment elevation myocardial infarction:the importance of thrombus burden. J Am Coll Cardiol. 2007;50:573-583.
12. Rentrop KP, Cohen M, Blanke H, Phillips RA. Changes in collateral channel filling immediately after controlled coronary artery occlusion by an angioplasty balloon in human subjects. J Am Coll Cardiol. 1985;5:587-592.
13. Suzuki N, Asano T, Nakazawa G, et al. Clinical expert consensus document on quantitative coronary angiography from the Japanese Association of Cardiovascular Intervention and Therapeutics. Cardiovasc Interv Ther. 2020;35:105-116.
14. Farkouh ME, Reiffel J, Dressler O, et al. Relationship between ST-segment recovery and clinical outcomes after primary percutaneous coronary intervention:the HORIZONS-AMI ECG substudy report. Circ Cardiovasc Interv. 2013;6:216-223.
15. Fabris E, van 't Hof A, Hamm CW, et al. Clinical impact and predictors of complete ST segment resolution after primary percutaneous coronary intervention:A subanalysis of the ATLANTIC Trial. Eur Heart J Acute Cardiovasc Care. 2019;8:208-217.
16. Fregni F. Sample Size Calculation. Clinical Thinking in Clinical Research:Applied Theory and Practice Using Case Studies. New York:Oxford University Press. 2018:225-242.
17. Gu J, Zhuo Y, Liu TJ, et al. Balloon Deflation Strategy during Primary Percutaneous Coronary Intervention in Acute ST-Segment Elevation Myocardial Infarction:A Randomized Controlled Clinical Trial and Numerical Simulation-Based Analysis. Cardiol Res Pract. 2020;2020:4826073.
18. Loubeyre C, Morice MC, Lefevre T, Piechaud JF, Louvard Y, Dumas P. A randomized comparison of direct stenting with conventional stent implantation in selected patients with acute myocardial infarction. J Am Coll Cardiol. 2002;39:15-21.
19. Lemesle G, Sudre A, Bouallal R, et al. Impact of thrombus aspiration use and direct stenting on final myocardial blush score in patients presenting with ST-elevation myocardial infarction. Cardiovasc Revasc Med. 2010;11:149-154.
20. Sardella G, Mancone M, Nguyen BL, et al. The effect of thrombectomy on myocardial blush in primary angioplasty:the Randomized Evaluation of Thrombus Aspiration by two thrombectomy devices in acute Myocardial Infarction (RETAMI) trial. Catheter Cardiovasc Interv. 2008;71:84-91.
21. G DEL, Bellandi F, Huber K, et al. Early glycoprotein IIb-IIIa inhibitors in primary angioplasty-abciximab long-term results (EGYPT-ALT) cooperation:individual patient's data meta-analysis. J Thromb Haemost. 2011;9:2361-2370.
22. Marra MP, Corbetti F, Cacciavillani L, et al. Relationship between myocardial blush grades, staining, and severe microvascular damage after primary percutaneous coronary intervention a study performed with contrast-enhanced magnetic resonance in a large consecutive series of patients. Am Heart J. 2010;159:1124-1132.
23. Bajaj NS, Singh A, Zhou W, et al. Coronary Microvascular Dysfunction, Left Ventricular Remodeling, and Clinical Outcomes in Patients With Chronic Kidney Impairment. Circulation. 2020;141:21-33.
24. De Luca G, van 't Hof AW, de Boer MJ, et al. Time-to-treatment significantly affects the extent of ST-segment resolution and myocardial blush in patients with acute myocardial infarction treated by primary angioplasty. Eur Heart J. 2004;25:1009-1013.
25. Ng VG, Lansky AJ, Toro S, et al. Prognostic utility of myocardial blush grade after PCI in patients with NSTE-ACS:Analysis from the ACUITY trial. Catheter Cardiovasc Interv. 2016;88:215-224.
26. Bottner RK, Morea CJ, Green CR, Renzi RH, Kent KM, Krucoff MW. Quantitation of ischemia during total coronary occlusion with computer-assisted high resolution ST-segment monitoring:effect of collateral flow. J Electrocardiol. 1987;20 Suppl:104-106.
27. Brodie BR, Stuckey TD, Hansen C, et al. Relation between electrocardiographic ST-segment resolution and early and late outcomes after primary percutaneous coronary intervention for acute myocardial infarction. Am J Cardiol. 2005;95:343-348.
28. Verouden NJ, Haeck JD, Kuijt WJ, et al. Clinical and angiographic predictors of ST-segment recovery after primary percutaneous coronary intervention. Am J Cardiol. 2010;105:1692-1697.
29. Lefevre T, Garcia E, Reimers B, et al. X-sizer for thrombectomy in acute myocardial infarction improves ST-segment resolution:results of the X-sizer in AMI for negligible embolization and optimal ST resolution (X AMINE ST) trial. J Am Coll Cardiol. 2005;46:246-252.
30. De Luca G, Ernst N, van 't Hof AW, et al. Preprocedural Thrombolysis in Myocardial Infarction (TIMI) flow significantly affects the extent of ST-segment resolution and myocardial blush in patients with acute anterior myocardial infarction treated by primary angioplasty. Am Heart J. 2005;150:827-831.
31. Brener SJ, Dizon JM, Mehran R, et al. Complementary prognostic utility of myocardial blush grade and ST-segment resolution after primary percutaneous coronary intervention:analysis from the HORIZONS-AMI trial. Am Heart J. 2013;166:676-683.

ABSTRACT
Introduction and objectives: Patients with left main coronary artery (LMCA) stenosis have been excluded from the trials that support the non-inferiority of the instantaneous wave-free ratio (iFR) compared to the fractional flow reserve (FFR) in the decision-making process of coronary revascularization. This study proposes to prospectively assess the concordance between the two indices in LMCA lesions and to validate the iFR cut-off value of 0.89 for clinical use.
Methods: National, prospective, and observational multicenter registry of 300 consecutive patients with intermediate lesions in the LMCA (angiographic stenosis, 25% to 60%. A pressure gudiewire study and determination of the RFF and the iFR will be performed: in the event of a negative concordant result (FFR > 0.80/iFR > 0.89), no treatment will be performed; in case of a positive concordant result (FFR ≤ 0.80/iFR ≤ 0.89), revascularization will be performed; In the event of a discordant result (FFR> 0.80/iFR ≤ 0.89 or FFR ≤ 0.80/iFR> 0.89), an intravascular echocardiography will be performed and revascularization will be delayed if the minimum lumen area is > 6 mm2. The primary clinical endpoint will be a composite of cardiovascular death, LMCA lesion-related non-fatal infarction or need for revascularization of the LMCA lesion at 12 months.
Conclusions: Confirm that an iFR-guided decision-making process in patients with intermediate LMCA stenosis is clinically safe and would have a significant clinical impact. Also, justify its systematic use when prescribing treatment in these potentially high-risk patients.
Registered at ClinicalTrials.gov ( Identifier: NCT03767621).
Keywords: iFR. FFR. Left main coronary artery.
RESUMEN
Introducción y objetivos: Los pacientes con estenosis en el tronco coronario izquierdo (TCI) han sido excluidos de los ensayos que apoyan la no inferioridad del cociente de presiones en el índice diastólico instantáneo sin ondas (iFR) respecto a la reserva fraccional de flujo (RFF) en la toma de decisiones sobre revascularización coronaria. El presente estudio propone valorar de manera prospectiva la concordancia entre los dos índices en lesiones del TCI y validar el valor de corte del iFR de 0,89 para su uso clínico.
Métodos: Registro multicéntrico nacional, prospectivo, observacional, con la inclusión de 300 pacientes consecutivos con lesiones intermedias (estenosis angiográfica 25-60%) en el TCI. Se realizará un estudio con guía de presión y determinación de RFF e iFR. En caso de resultado concordante negativo (RFF > 0,80 / iFR > 0,89), no se realizará tratamiento; en caso de resultado concordante positivo (RFF ≤ 0,80 / iFR ≤ 0,89), se realizará revascularización; en caso de resultado discordante (RFF > 0,80 / iFR ≤ 0,89 o RFF ≤ 0,80 / iFR > 0,89), se realizará estudio con ecocardiografía intravascular y se considerará diferir la revascularización si el área luminal mínima es > 6 mm2. El criterio de valoración clínico primario será la incidencia del combinado de muerte cardiovascular, infarto no mortal relacionado con la lesión del TCI o necesidad de revascularización de la lesión del TCI a los 12 meses.
Conclusiones: La demostración de la seguridad clínica en la toma de decisiones del iFR en pacientes con lesiones intermedias en el TCI tendría un impacto clínico importante y justificaría su uso sistemático para la decisión del tratamiento en estos pacientes de potencial alto riesgo.
Registrado en ClinicalTrials.gov (identificador: NCT03767621).
Palabras clave: iFR. RFF. Tronco coronario izquierdo.
Abbreviations
MLA: minimum lumen area. FFR: fractional flow reserve. iFR: instantaneous wave-free ratio. IVUS: intravascular ultrasound. LMCA: left main coronary artery.
INTRODUCTION
Assessing functional severity of coronary stenoses at left main coronary artery (LMCA) level through coronary angiography has serious limitations.1 To treat angiographically intermediate stenoses (25% to 60% diameter) the use of invasive (ultrasound or optical coherence tomography) or functional imaging modalities (determining fractional flow reserve [FFR] to indicate the need for revascularization) has been proposed.2 Patients with LMCA stenosis have traditionally been excluded from randomized clinical trials that assessed the prognostic capabilities of the functional assessment of coronary stenoses through the use of FFR.3-5 The use of FFR to assess LMCA stenoses is backed by a limited number of non-randomized clinical trials that confirmed that FFR values > 0.80 is associated with a low risk of events if no revascularization is performed in patients with intermediate LMCA stenoses.6
The instantaneous wave-free ratio (iFR) is a new, easier-to-use, and cost-effective invasive index to assess the coronary function compared to FFR since there is no need to induce maximum coronary hyperemia to estimate it.7 Although a non-inferior prognostic value of iFR compared to the FFR has recently been confirmed in patients with intermediate lesions in 2 large trials, the presence of LMCA lesions was largely anecdotal or inexistent in both indices.8,9 However, a non-randomized clinical trial has been published with a similar design to those previously conducted with the FFR that provides encouraging data on the value of iFR in the decision-making process regarding the LMCA. However, in such trial, the FFR—the most widely used index to assess intermediate LMCA stenoses—was not determined at the same time, which means that the results of this registry cannot be put into context.10 Also, there are signs that the location of the LMCA lesion is a predictor of worse concordance between both indices.11
Proving the clinical safety of iFR in patients with intermediate LMCA lesions would have a major clinical impact and justify its systematic use in the decision-making process regarding the management of these high-risk patients.
The objective of this study is to assess the concordance between 2 physiological indices—the FFR and the iFR—in the assessment of intermediate LMCA lesions. Also, to validate prospectively the clinical safety profile of a revascularization strategy based on an iFR cut-off value of 0.89.
METHODS
Study design
National, prospective, observational, and multicenter registry including 300 consecutive patients with intermediate LMCA lesions (25% to 60% angiographic stenosis). A study will be conducted in all patients using intracoronary guidewire pressures. Also, both the FFR and the iFR values will be determined distal to the LMCA. Per protocol it is advised that the indication for revascularization should be decided based on the result of the iFR in such a way that:
-
– In patients with iFR and FFR values in the LMCA lesion > 0.89 and > 0.80, respectively clinical follow-up without LMCA lesion revascularization is indicated. In the presence of other lesions outside the LMCA with percutaneous revascularization criteria, the revascularization of these other lesions is indicated.
-
– In patients with iFR and FFR values in the LMCA lesion ≤ 0.89 and ≤ 0.80, respectively the revascularization of the LMCA lesion is indicated (percutaneous through a drug-eluting stent or surgical). In the presence of other lesions outside the LMCA with revascularization criteria (whether percutaneous or surgical), the revascularization of these other lesions is indicated.
-
– In case of discrepancy between the FFR and the iFR (positive vs negative or vice versa with 2 or more points above or below the respective cut-off value) an intravascular ultrasound (IVUS) should be performed to decide whether to indicate revascularization or not; with minimum lumen areas (MLA) > 6 mm2 revascularization is ill-advised.
Patients whose management is not consistent with what the iFR value recommends will not be addressed for the strategy safety analysis, and clinical outcomes will be assessed separately.
Figure 1 shows the decision-making algorithm based on FFR and iFR results. IVUS is indicated in controversial cases, and recommended in the remaining cases to determine the correlation between the MLA and the iFR.
Figure 1. Decision-making algorithm based on the FFR and iFR results. DES, drug-eluting stent; FFR, fractional flow reserve; iFR, instantaneous wave-free ratio; IV, intravenous; IVUS, intravascular ultrasound; LMCA, left main coronary artery; MACE, major adverse cardiovascular events, MLA, minimum lumen area.
In patients eligible for percutaneous treatment, IVUS is highly recommended, and its utility will be assessed prospectively during the planning and optimization of the procedure.
Clinical follow-up is advised from 12 months to 5 years to determine the prognostic primary endpoint by assessing a composite endpoint of cardiovascular death, LMCA lesion-related non-fatal infarction or need for LMCA revascularization at the 12-months and 5-year follow-up.
Notifications
The study has been approved by the reference ethics committee and notified to the local ethics committee of all participant centers. The study has been registered in Clinicaltrials.gov with registration number NCT03767621. Devices with CE marking have only been used, and only for the indications already approved. The study observes the principles established by the Declaration of Helsinki. All patients gave their prior written informed consent to participate in the study.
Study population
Patients with suspected or confirmed ischemic heart disease on whom a coronary angiography is performed that detects intermediate angiographic LMCA stenoses (between 25% and 60%). Also, patients in whom intracoronary pressure guidewires are used to determine the iFR and the FFR in the LMCA lesion to decide on the indication for myocardial revascularization—whether percutaneous with a DES or surgical—based on the indication considered more appropriate.
Inclusion and exclusion criteria are shown on table 1. In cases of severe lesions at left anterior descending coronary artery or left circumflex artery level, the patient will not be included in the study unless the LMCA lesion is assessed after the percutaneous treatment of these lesions while taking into account that, if the LMCA lesion is significant, treatment will be percutaneous.
Table 1. Inclusion and exclusion criteria of the iLITRO-EPIC-07 trial
| Inclusion criteria |
| Patients with intermediate LMCA lesions (25% to 60% angiographic stenosis on visual estimations) eligible for a pressure guidewire study to determine the iFR |
| Patients aged ≥ 18 years |
| Patients capable of giving their informed consent |
| Exclusion criteria |
| Patients with an indication for coronary artery bypass graft regardless of the significance of the LMCA lesion |
| Patients with LMCA lesions showing ulceration, dissection or thrombus |
| Patients with lesions in a previously non-dysfunctional arterial or venous graft in the territory irrigated by the LMCA (protected LMCA) |
| Patients with acute coronary syndrome with potentially culprit lesion in the LMCA |
| Patients incapable of giving their informed consent |
|
iFR, instantaneous wave-free ratio; LMCA, left main coronary artery. |
Study endpoints
The iLITRO-EPIC 07 trial has 2 primary endpoints:
-
1) To establish concordance before indicating revascularization between 2 invasive functional assessment indices through intracoronary pressure guidewire in intermediate LMCA lesions with FFR and iFR cut-off values ≥ 0.80 (with IV adenosine) and ≥ 0.89 to delay treatment.
-
2) To validate prospectively the safety profile associated with the decision-making process regarding the revascularization of intermediate LMCA stenoses based on an iFR cut-off value of 0.89 measured using an intracoronary pressure guidewire to decide whether to revascularize or not based on the number of patients with delayed LMCA revascularization of the composite endpoint of cardiovascular death, LMCA lesion-related non-fatal infarction or need for LMCA revascularization at the 12-month follow-up.
Secondary endpoints are to determine the correlation between the iFR value in these lesions and the MLA determined by the IVUS and the utility of IVUS for the planning and optimization of LMCA lesions (table 2).
Table 2. Secondary endpoints of the iLITRO-EPIC-07 trial
| Correlation between the assessment obtained through pressure guidewire (iFR) and the minimum lumen area measured through IVUS |
| Role of IVUS in the planning of treatment in the subgroup of patients treated with percutaneous therapy |
| Role of IVUS in the optimization of treatment in the subgroup of patients treated with percutaneous therapy |
| All-cause mortality at 12 months and 5 years |
| Cardiovascular death at 12 months and 5 years |
| Non-fatal infarction at 12 months and 5 years |
| LMCA lesion-related non-fatal infarction at 12 months and 5 years |
| Revascularization at 12 months and 5 years |
| Myocardial infarction associated with the revascularization of the LMCA (whether percutaneous or surgical) |
| Thrombosis of 1 or several stents in the LMCA at 12 months and 5 years |
| Restenosis of 1 or several stents in the LMCA at 12 months and 5 years |
| New target lesion revascularization in the LMCA (whether percutaneous or surgical) at 12 months and 5 years |
|
iFR, instantaneous wave-free ratio; LMCA, left main coronary artery; IVUS: intravascular ultrasound. |
Study procedure
Figure 2 shows the procedure methodology on a flowchart.
Figure 2. Study protocol and procedures. ACS, acute coronary syndrome; CABG, coronary artery bypass graft; FFR, fractional flow reserve; iFR, instantaneous wave-free ratio; IVUS, intravascular ultrasound; LAD, left anterior descending coronary artery; LCX, left circumflex artery; LMCA, left main coronary artery; MLA, minimum lumen area; PCI, percutaneous coronary intervention; QCA, quantitative coronary angiography.
Protocol to perform a study using a pressure guidewire
The patient is eligible for functional assessment in the presence of intermediate LMCA stenoses with visual estimations on the coronary angiography between 25% and 60%.
After catheterization using a guide catheter, at least, 200 µg of intracoronary nitroglycerin should be administered to keep coronary reactivity under control. Afterwards, the intracoronary guidewire should be advanced with the sensor placed in the ostium of the guide catheter; also, pressure curves should be brought back to normal for 5 to 10 heart beats. if the lesion has an ostial location, normalization will occur by removing the guide catheter from the coronary artery and placing the guidewire into the aorta. Afterwards, the guidewire should be removed from the catheter, and coronary catheterization performed to advance the guidewire.
The pressure guidewire should be advanced until, at least, 3 times the diameter of the vessel beyond the most distal stenosis to be able to measure the iFR according to the standard protocol.
After measuring the iFR, the guidewire should be removed with pressure curve monitorization until the inside of the guide catheter. At this point, the presence of the pressure calibration loss phenomenon (drifting) should be discarded. In case of overt drift (Pd/Pa measured on the catheter tip < 0.98 or > 1.02) measures should be taken again.
Afterwards, the FFR will be determined during hyperemia through the administration of adenosine in continuous IV infusion at doses ≥ 140 µg/kg/min for, at least, 2 minutes or an IV bolus of 0.4 mg of regadenoson.
After measuring the FFR, the guidewire should be removed with pressure curve monitorization until the inside of the guide catheter. At this point, the presence of drifting should be discarded. In case of overt drift (Pd/Pa measured on the catheter tip < 0.98 or > 1.02) measures should be taken again.
In case of discrepancy between the results of the FFR and the iFR (FFR ≤ 0.80 with iFR ≥ 0.90 or FFR ≥ 0.81 with iFR ≤ 0.89) IVUS will be performed, and the MLA determined. Revascularization will be indicated with MLAs < 6 mm2 based on the results from the LITRO trial.12
Protocol to conduct IVUS studies
IVUS studies will be mandatory if the FFR and the iFR disagree. In patients eligible for percutaneous treatment of their LMCA lesions, the IVUS is highly recommended to guide the procedure. In the remaining patients (when iFR-guided medical therapy or surgical revascularization is decided) the IVUS is recommended to establish the correlation between the iFR value and the MLA measured on the LMCA whenever possible. The IVUS system used can be mechanical or rotational with resolutions between 20 MHz and 60 MHz.
An 0.014 in intracoronary guidewire will be advanced to perform the IVUS study (it can be the same pressure guidewire used to determine the iFR) towards the left anterior descending or left circumflex coronary arteries. After the administration of 200 µg of intracoronary nitroglycerin, the IVUS catheter will be advanced distal to the LMCA bifurcation. Afterwards, the catheter will be manual or automatically removed until the ascending aorta. It is essential that the guide catheter should remain outside the coronary artery to study the left main coronary artery entirely including its ostial region. The catheter will be placed in the left anterior descending coronary artery (preferably) or left circumflex artery or both (to conduct 2 studies with MLA determination from these positions and eventually pick the one with the lowest values).
In cases of catheter backward jump, even on manual mode (with calcified angulation) it is recommended to move the catheter forward from the aorta to acquire images of the region of interest that had not been properly assessed.
Technical aspects of the assessment of left main coronary artery lesions through fractional flow reserve
The study of LMCA lesions using pressure guidewires has some particularities that should be addressed when conducting the study.
Location of the lesion
A total of 3 different possible lesion locations can be anatomically distinguished on the LMCA depending on whether there is damage to the ostium, body or distal portion (bifurcation). The location of the lesion inside the LMCA has implications when conducting the study with the pressure guidewire. When the lesion is found in the ostium or the body, catheterization should be coaxial. Non-coaxial catheterization involves contact of the catheter lumen with the vessel wall to the extent that it can dampen the aortic pressure and artificially elevate the value of the FFR. For this reason, non-selective catheterization is advised when equalizing or normalizing the catheter and guidewire pressures when the latter is placed distal to the lesion to measure the FFR during maximum hyperemia. When the lesion is found in the LMCA distal portion and there is damage to its origin and main branches, both the distal LMCA and each one of its branches should be treated as 1 functional unit regardless of the degree of damage to these branches. To estimate the FFR, measurements are taken from the left anterior descending and left circumflex coronary arteries. The LMCA lesion is considered functionally significantly when the measurements of either one of the 2 main vessels is < 0.80.
Induction of hyperemia
In the assessment of LMCA lesions the use of an intracoronary bolus of adenosine is ill-advised because, since the non-selective catheterization of the left coronary artery is required, part of the drugs administered never reach this coronary artery, which is why the induction of hyperemia can be suboptimal. For this reason, the IV administration of drugs whether adenosine (infusions of 140 µg/kg/min for, at least, 2 minutes) or regadenoson (doses of 0.4 mg in IV bolus) is advised.13
Presence of left anterior descending or left circumflex coronary artery lesions
The presence of 1 isolated LMCA lesion is not rare. A series of all-comers treated with diagnostic coronary angiography proved that, in patients with damage to the LMCA, only 9% had 1 single LMCA lesion, 17% had 1 LMCA lesion plus damage to 1 vessel, 35% had 1 LMCA lesion plus damage to 2 vessels, and 38% had LMCA disease plus damage to 3 vessels.14
Statistical analysis
Demographic, clinical, hemodynamic, and procedural data will be presented for the entire group. Continuous variables will be expressed as mean, and standard deviation (or if the distribution of the values do not follow a normal, as median, and interquartile range). Categorical variables will be expressed as frequencies and percentages. The data obtained will be studied using the unilateral analysis of variance (ANOVA) for the continuous variables, and Fisher’s exact test or the chi-square test for the categorical variables, when appropriate. When appropriate, non-parametric tests will be used with variables without a normal distribution or when normalization is not possible. The Kaplan-Meier survival curves will be presented for the previously specified criteria. The concordance analyses will be conducted using Cohen’s kappa coefficient. Also, sensitivity, specificity, positive and negative predictive values, and the area under the receiver operating characteristic (ROC) curve will be estimated.
Data curation and monitorization
Clinical, angiographic, physiological, and IVUS data will all be saved in a safe electronic CRD managed by Fundación EPIC, the promotor of the study. Clinical data at both the 12-month and 5-year follow-up, as well as the presence of cardiovascular events at the follow-up will also be saved in the same electronic CRD.
DISCUSSION
The iLITRO-EPIC 07 trial has a double primary endpoint: on the one hand, to establish the concordance between 2 intracoronary physiological indices, the FFR and the iFR, when assessing the severity of intermediate LMCA lesions; on the other hand, to study the use of a predetermined iFR value to indicate the revascularization of intermediate LMCA lesions with an up to 5-year clinical follow-up.
Left main coronary artery disease. Implications for the interventional cardiologist
Significant LMCA disease, understood as a stenosis in its greater diameter > 50%, is associated with a poor mid-term prognosis. Studies prior to coronary revascularization confirmed survival rates < 40% at the 4-year follow-up after diagnosis.15
The limitations of the angiographic assessment of the severity of LMCA lesions are well established.16-18 Before suggesting revascularization in a patient with LMCA lesions, in particular ostial lesions, it is important to know whether the lesion really needs to be revascularized, that is, whether it is hemodynamically significant. LMCA stenoses are found in between 4% to 9% of all diagnostic coronary angiographies.1 Due to their anatomical location, catheter-induced artifacts or to the severity of distal lesions, among other factors, interpreting LMCA lesions is associated with the highest intra- and inter-observer variability compared to lesions found in other parts of the coronary tree.16 When stenoses ≥ 50% were found in the CASS registry,19 a second observer confirmed that the stenosis was not significant in 19% of the cases.
Several former studies have confirmed that the prognosis of patients with functionally insignificant LMCA lesions is favorable.6 Also, that the surgical revascularization of hemodynamically insignificant lesions is associated with a high rate of early graft failure.20
The LITRO trial, led by the Spanish Society of Cardiology Working Group on Intracoronary Diagnostic Techniques, was a multicenter and prospective study. It proved that, in patients with angiographically intermediate LMCA lesions, the presence of a MLA ≥ 6 mm2 measured on the IVUS allows us to delay revascularization in a safely manner.12
Evidence to guide the revascularization of the left main coronary artery through functional assessment
To this date, no definitive data on the prognostic value of iFR measurements in intermediate LMCA stenoses have been published. The presence of a significant stenosis (> 70%) on the coronary angiography was an exclusion criteria in the DEFER, FAME, and FAME II clinical trials, as well as in the DEFINE FLAIR trial. Only the IFR SWEDEHEART trial included 30 patients with significant LMCA stenoses (1.6% of all the patients included).3-5,8,9 An observational and retrospective study of 314 patients confirmed that delaying the revascularization of the LMCA using a iFR cut-off value of 0.89 as the guide was safe at the 30-month clinical follow-up.10 However, in this observational registry the FFR, a widely validated index in the LMCA, was not obtained at the same time. This means that the results reported by this registry cannot be put into context and the concordance between both indices cannot be analyzed either.
The data available that support the use of the FFR in LMCA lesions come from several studies shown on table 1. The cut-off values used in these studies go from 0.75 to 0.80. In the study that has included, to this date, the highest number of patients with intermediate angiographic lesions, 213, only patients with FFR values < 0.80 were treated. However, in patients with higher values a conservative manage was used. No differences in the mortality or severe cardiovascular event rates were reported at the 5-year follow-up.6 Therefore, the reference FFR value for LMCA lesions, as well as the remaining lesions, is < 0.80.
A metanalysis that included data from 8 landmark studies found no differences in the primary endpoint of death, non-fatal myocardial infarction or revascularization. However, the need for revascularization was greater in the group on medical therapy: whether this was primarily due to the revascularization of the LMCA is still under discussion.21
A recent study that assessed the correlation between the FFR and the iFR values based on the location of the lesion studied revealed that such correlation was weaker when the lesion was found on the LMCA or in the proximal left anterior descending coronary artery compared to other locations. This was attributed to a greater amount of vessel-dependent myocardium in these proximal lesions. Taking the FFR value and an iFR cut-off value ≥ 0.89 as a reference, both the false positives (21.9%) and the false negatives (26.7%) were more evident when the lesion was found on the LMCA or the proximal left anterior descending coronary atery.11 Some studies have suggested that resting indices like the iFR could provide better measurements of coronary flow during hyperemia compared to the FFR.22,23 This means that using the FFR as the gold standard could be questionable in this setting. Also, the scientific evidence available indicates that the discrepancies seen between the iFR and the FFR are not associated with a worse prognosis.24 This means that the present study could clarify whether the iFR is associated with a weaker indication for revascularization in intermediate LMCA lesions with the exact same clinical safety compared to the FFR.
CONCLUSIONS
The iLITRO-EPIC 07 trial is the first prospective study to assess the concordance between the FFR and the iFR in intermediate LMCA lesions. Also, that it is safe to guide the indication for revascularization based on an iFR cut-off value of 0.89.
FUNDING
The promoter of the study, Fundación EPIC, has received an institutional research grant from Phillips Volcano (The Netherlands) to pay for the design and maintenance costs of the electronic CRD. Philips Volcano has not been involved in the design of the study or protocol whatsoever. Philips Volcano has not been involved in the development of the study whatsoever including recruitment, follow-up, data curation, result analysis and interpretation, writing or final approval of both the protocol and this manuscript. The authors are solely responsible for the study design, writing, edition, and final version of the manuscript.
AUTHORS’ CONTRIBUTIONS
All the authors are lead investigators of the iLITRO-EPIC07 trial at their corresponding working centers, collaborated in the writing of the study protocol, and in the recruitment of the patients. The manuscript was written by O. Rodríguez-Leor, J.M. de la Torre-Hernández, and A. Pérez de Prado; the remaining authors reviewed the manuscript.
CONFLICTs OF INTEREST
A. Pérez de Prado declared to have received fees from iVascular, Boston Scientific, Terumo, B. Braun, and Abbott Vascular. José M. de la Torre Hernández is the editor-in-chief of REC: Interventional Cardiology. F. Alfonso, and J. Sanchis are associate editors of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed.
WHAT IS KNOWN ABOUT THE TOPIC?
- In intermediate LMCA stenoses (25% to 60% diameter) the use of invasive (ultrasound or optical coherence tomography) or functional imaging modalities (by measuring the FFR) has been proposed to eventually indicate the need for revascularization. Patients with LMCA stenoses were excluded from randomized clinical trials that assessed the prognostic capabilities of the functional assessment using the FFR.3 However, its use has been backed by several non-randomized clinical trials that confirmed that values > 0.80 are indicative of a low risk of events if revascularization is eventually spared. The iFR is a new physiological index that does not require hyperemia to be determined, which simplifies the whole process. There are still no data on the concordance between both indices in LMCA lesions or the safety of this new index in the assessment of these patients.
WHAT DOES THIS STUDY ADD?
- The iLITRO-EPIC07 trial is an attempt to prospectively assess the concordance between the FFR and the iFR, as well as the safety profile of an iFR-guided revascularization strategy.
REFERENCES
1. Lindstaedt M, Spiecker M, Perings C, et al. How good are experienced interventional cardiologist at predicting the functional significance of intermediate or equivocal left main coronary stenosis?Int J Cardiol. 2007;120:254-261.
2. Windecker S, Kohl P, Alfonso F, et al. 2014 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2014;35:2541-2619.
3. Pijls NH, van Schaardenburgh P, Manoharan G, et al. Percutaneous coronary intervention of functionally nonsignificant stenosis:5-year follow-up of the DEFER Study. J Am Coll Cardiol. 2007;49:2105-2111.
4. Tonino PA, De Bruyne B, Pijls NH, et al. Fractional flow reserve vs. angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213-224.
5. De Bruyne B, Pijls NH, Kalesan B, et al. Fractional flow reserve-guided PCI vs. medical therapy in stable coronary disease. N Engl J Med. 2012;367:991-1001.
6. Hamilos M, Muller O, Cuisset T, et al. Long-term clinical outcome after fractional flow reserve-guided treatment in patients with angiographically equivocal left main coronary artery stenosis. Circulation. 2009;120:1505-1512.
7. Sen S, Escaned J, Malik IS, et al. Development and validation of a new adenosine-independent index of stenosis severity from coronary wave-intensity analysis:results of the ADVISE (ADenosine Vasodilator Independent Stenosis Evaluation) study. J Am Coll Cardiol. 2012;59:1392-1402.
8. Davies JE, Sen S, Dehbi HM, et al. Use of the instantaneous wave-free ratio or fractional flow reserve in PCI. N Engl J Med. 2017;376:1824-1834.
9. Götberg M, Christiansen EH, Gudmundsdottir IJ, et al. iFRSWEDEHEART Investigators. Instantaneous free-wave ratio versus fractional flow reserve to guide PCI. N Engl J Med. 2017;376:1813-1823.
10. Warisawa T, Cook CM, Rajkumar C, et al. Safety of Revascularization Deferral of Left Main Stenosis Based on Instantaneous Wave-Free Ratio Evaluation. JACC Cardiovasc Interv. 2020;13:1655-1664.
11. Kobayashi Y, Johnoson NP, Berry C, et al. The influence of lesion location on the diagnostic accuracy of adenosine-free coronary pressure wire measurements. J Am Coll Cardiol Interv. 2016;9:2390-2399.
12. de la Torre Hernandez JM, Hernandez F, Alfonso F, et al. Prospective application of pre-defined intravascular ultrasound criteria for assessment of intermediate left main coronary artery lesions results from the multicenter LITRO study. J Am Coll Cardiol. 2011;58:351-358.
13. Nair PK, Marroquin OC, Mulukutla SR, et al. Clinical utility of regadenoson for assessing fractional flow reserve. JACC Cardiovasc Interv. 2011;1085-1092.
14. Ragosta M, Dee S, Sarembock IJ, et al. Prevalence of unfavorable angiographic characteristics for percutaneous intervention in patients with unprotected left main coronary artery disease. Catheter Cardiovasc Interv. 2006;68:357-362.
15. Cameron A, Kemp HG Jr, Fisher LD, et al. Left main coronary artery stenosis:angiographic determination. Circulation. 1983;68:484-489.
16. Fisher LD, Judkins MP, Lesperance J, et al. Reproducibility of coronary arteriographic reading in the coronary artery study (CASS). Catheter Cardiovasc Diagn. 1982;8:565-575.
17. Arnett EN, Isner JM, Redwood DR, et al. Coronary artery narrowing in coronary heart disease:comparison of cineangiographic and necropsy findings. Ann Intern Med. 1979;91:350-356.
18. Lenzen MJ, Boersma E, Bertrand ME, et al. Management and outcome of patients with established coronary artery disease:the Euro Heart Survey on coronary revascularization. Eur Heart J. 2005;26:1169-1179.
19. Kandzari DE, Colombo A, Park SJ, et al. Revascularization for unprotected left main disease:evolution of the evidence basis to redefine treatments standards. J Am Coll Cardiol. 2009;54:1576-1588.
20. Botman CJ, Schonberger J, Koolen S, et al. Does stenosis severity of native vessels influence bypass graft patency?A prospective FFR-guided study. Ann Thorac Surg. 2007;83:2093-2097.
21. Mallidi J, Atreya AR, Cook J, et al. Long term outcomes following fractional flow reserve guided treatment of angiographically ambiguous left main coronary artery disease:a meta-analysis of prospective cohort studies. Catheter Cardiovasc Interv. 2015;86:12-18.
22. Petraco R, van de Hoef TP, Nijjer S, et al. Baseline instantaneous wave-free ratio as a pressure-only estimation of underlying coronary flow reserve:results of the JUSTIFY-CFR Study (Joined Coronary Pressure and Flow Analysis to Determine Diagnostic Characteristics of Basal and Hyperemic Indices of Functional Lesion Severity Coronary Flow Reserve). Circ Cardiovasc Interv. 2014;7:492-502.
23. Nijjer SS, de Waard GA, Sen S, et al. Coronary pressure and flow relationships in humans:phasic analysis of normal and pathological vessels and the implications for stenosis assessment:a report from the Iberian-Dutch-English (IDEAL) collaborators. Eur Heart J. 2016;37:2069-2080.
24. Lee JM, Shin ES, Nam CW, et al. Clinical outcomes according to fractional flow reserve or instantaneous wave-free ratio in deferred lesions. JACC Cardiovasc Interv. 2017;10:2502-2510.
ABSTRACT
Introduction and objectives: Former studies have associated the severity of calcified plaques (CP) on the invasive coronary angiography (ICA) with a limited number of optical coherence tomography (OCT) measurements. The objective of this study was to describe the correlation between an extended and comprehensive set of OCT measurements and the severity of calcifications as seen on the ICA.
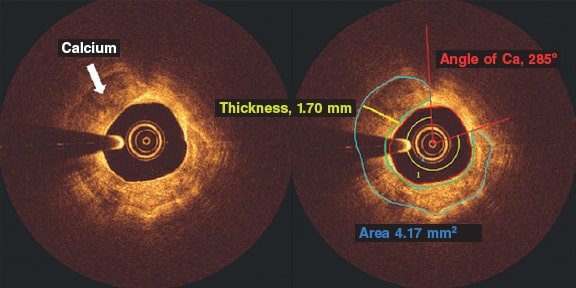
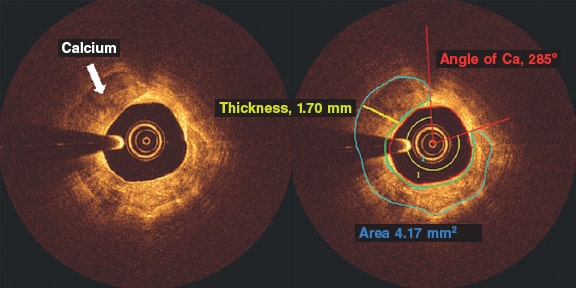
Methods: We retrospectively studied 75 patients (75 lesions) who underwent ICA and, concurrently, OCT imaging at a single institution. The OCT was performed before the percutaneous coronary intervention and after the administration of intracoronary nitroglycerine. The coronary artery calcium was scored using a three-tier classification system on the ICA. Maximum calcium angle, area, maximum thickness, length of calcium, and calcium depth were assessed on the OCT.
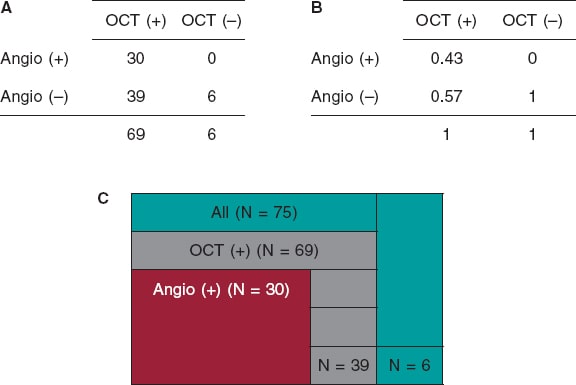
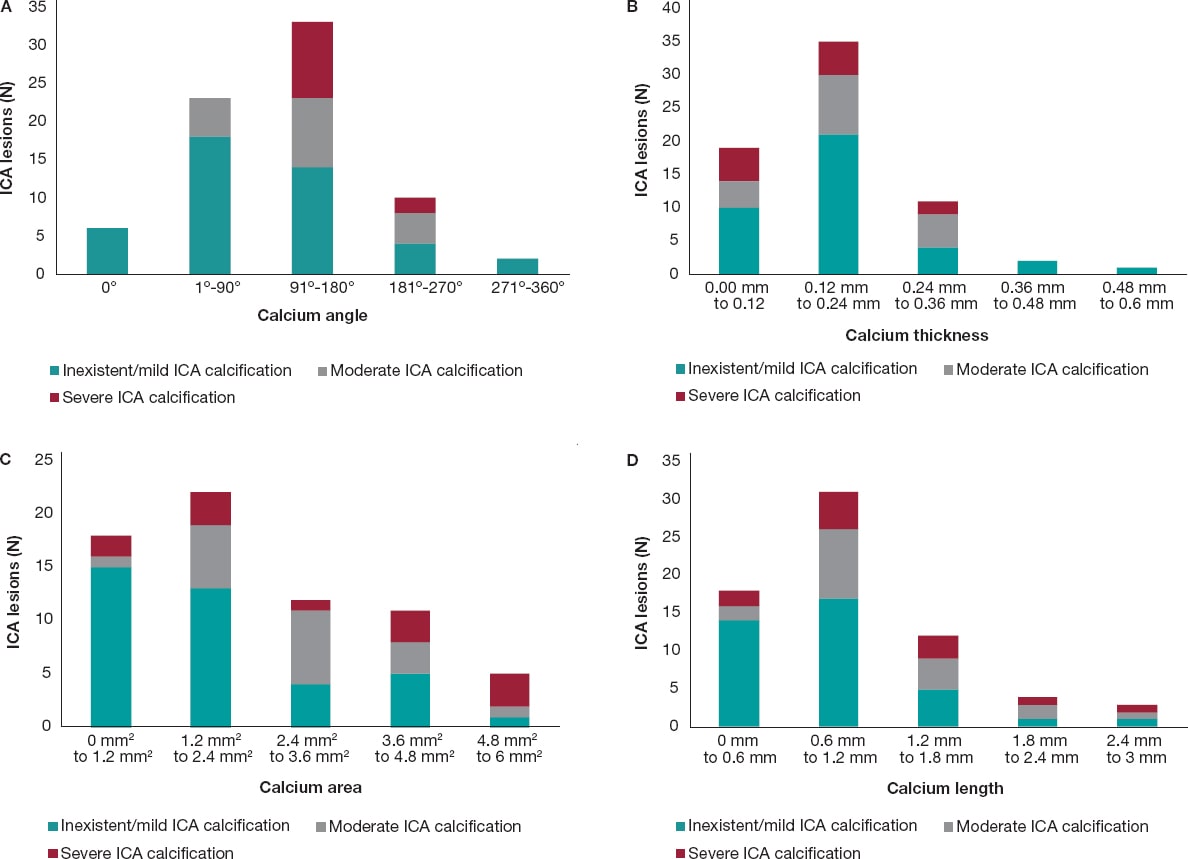
Results: The ICA detected fewer CP lesions compared to the OCT (N = 69; 92%), all cases of positive ICA were detected by the OCT (N = 30; 100%). The OCT did not find any positive lesions in negative angiographic lesions (N = 6; 100%). The sensitivity of the ICA was 43.5% (95%CI, 0.32-0.56) and its specificity, 100% (95%CI, 0.52-1.0). In most cases, as calcium angle, thickness, and area increased on the OCT so did the calcium severity of the lesions on the angiography.
Conclusions: Compared to the OCT, the ICA has a low sensitivity and a high specificity in the detection of calcified plaques. As calcium angle, thickness, area, and length increased on the OCT so did the number of angio-defined lesions of severe CP.
Keywords: Tomography. Optical coherence tomography. Invasive coronary angiography. Percutaneous coronary intervention. Calcification.
RESUMEN
Introducción y objetivos: Estudios previos han asociado la gravedad de la calcificación de las lesiones coronarias evaluadas con angiografía coronaria invasiva (ACI) con un número limitado de medidas obtenidas con tomografía de coherencia óptica (OCT). El objetivo de este estudio es analizar la correlación de una amplia y exhaustiva serie de medidas de OCT con la gravedad de la calcificación estimada por ACI.
Métodos: Se estudiaron retrospectivamente 75 pacientes (75 lesiones) de un único centro a quienes se realizaron simultáneamente ACI y OCT. La OCT se llevó a cabo tras la administración de nitroglicerina intracoronaria antes del intervencionismo coronario. En la ACI, la calcificación coronaria se valoró utilizando un sistema de clasificación en tres grados. Con OCT se evaluaron el máximo ángulo, el área, el grosor máximo, la longitud y la profundidad del calcio.
Resultados: La ACI detectó menos lesiones calcificadas que la OCT (n = 69; 92%) y todos los casos detectados por ACI fueron identificados con OCT (n = 30; 100%). La OCT no encontró calcio en ninguna de las lesiones sin calcio en la ACI (n = 6; 100%). La sensibilidad de la ACI fue del 43,5%, (IC95%, 0,32-0,56) y la especificidad del 100% (IC95%, 0,52-1,0). A medida que se incrementaron el ángulo, el grosor y el área del calcio por OCT también aumentó la gravedad del calcio determinada por ACI en la mayoría de los casos.
Conclusiones: La ACI tiene una baja sensibilidad, pero una alta especificidad, para la detección de lesiones calcificadas en comparación con la OCT. Al incrementarse el ángulo, el grosor, el área y la longitud del calcio en la OCT aumenta el número de lesiones con calcificación grave en la ACI.
Palabras clave: Tomografia. Coherencia optica. Angiografia coronaria invasiva. Intervencion coronaria percutanea. Calcificacion.
Abbreviations
CP: calcified plaque. OCT: optical coherence tomography. ICA: invasive coronary angiography.
INTRODUCTION
Coronary artery disease is very prevalent in the United States and is associated with high cardiovascular mortality rates.1 The management of advanced coronary artery disease (eg, calcified lesions) is often the percutaneous coronary intervention, but the use of the PCI alone in calcified plaques (CP) is associated with poor procedural outcomes.2-5 This is mainly due to the lack of information on the spread of calcification and its appropriate management before stenting. Therefore, intravascular imaging modalities are necessary for the characterization of calcium inside the vessel and better guide the interventional cardiologist.6-9
The optical coherence tomography (OCT) is a high-resolution cross-sectional imaging modality with an unparalleled axial resolution of around 4-20 microns.10 The OCT allows more accurate measurements of the CP over other invasive imaging modalities like the invasive coronary angiography (ICA) and the intravascular ultrasound (IVUS).11
Prior studies have associated the severity of the CP on the ICA with a limited number of measurements on the OCT.6,12-14 Our study aimed to further describe the correlation between an extended and comprehensive set of OCT measurements and the severity of calcification as seen on the ICA.
METHODS
Study population
We retrospectively studied 75 patients who underwent ICA and concurrently had OCT imaging acquired at the St. Francis Hospital, Roslyn, NY, United States, from November 2018 through April 2019. A total of 109 lesions were identified in these patients on the ICA. An OCT plus an ICA analysis were performed on 75 of these lesions deemed primary lesions while 34 lesions were excluded from the analysis (no OCT available). All primary lesions were lesions seen on the OCT images, not on the target lesion that received the stent during the procedure. No severely calcified plaques that could not be catheterized were excluded. All the lesions excluded were secondary or tertiary lesions that were deemed non-primary based on the lower calcification burden. No lesions required preparation or ablation before the OCT imaging. All the calcified spots in the population were not thick enough so as to cast a shadow. An institutional review board waiver was obtained because of the retrospective nature of this study. Patient consent was obtained for both the ICA and the OCT.
Optical coherence tomography acquisition
The OCT was performed before the percutaneous coronary intervention and after the administration of intracoronary nitroglycerine (100 µg-200 µg) using the frequency-domain OCT ILUMIEN OPTIS system (Abbott Vascular, United States) and a 2.7-Fr OCT imaging catheter (C7 Dragonfly, Dragonfly Duo or Dragonfly OPTIS; Abbott Vascular, United States). An OCT catheter was advanced distally to the lesion. Also, contrast media was injected manually through the guiding catheter with automatic pullback at a rate of 20 mm/sec for an average pullback distance of 75 mm ± 12.2 mm.
Imaging definition and analysis
The ICA and the OCT imaging were co-registered with respect to each other based on each patients’ anatomical landmarks. Afterwards, the co-registered ICA and OCT imaging had all identifiers removed. Both the ICA and the OCT measurements were assessed independently by two experienced angiography evaluators who were blind to the patients’ information except for the data on the anatomical location of the lesion on the ICA that was assessed on 2 different projections to secure increased accuracy when looking at the vessel. The evaluators then scored the degree of calcium based on the three-tier classification system: minimal or no calcification; calcium covering ≤ 50% of the vessel circumference was classified as “moderate calcification”; calcium covering between 50% and 100% of the vessel circumference was classified as “severe calcification” according to Mintz et al. classification.9 In case of discrepancy between the evaluators, a third evaluator blind to the information of both the patient and the independent reviewers’ assessment was invited to grade the degree of calcification.
The OCT calcium analysis was performed in the pre-percutaneous coronary intervention iFR-pullbacks. All the OCT analyses of the CP were performed using the QIVUS 3.1 validation utility tool (Medis Medical Imaging, The Netherlands) based on a standardized operating procedure at the core lab (MedStar Cardiovascular Research Network). The CP was analyzed on the area of maximum severity and defined by heterogenous areas of low signal attenuation and sharply demarcated borders. We assessed all pullbacks at lesion site level: the maximum calcium angle, maximum thickness, and length of calcium (number of frames with calcium). The angle of calcium was determined using the center of the lumen as the vertex (figure 1, red rays) as it extended from one clearly delineated border of the calcium plaque to the other. Automatic software detection was used to identify the fibrous cap overlying the calcium area and the maximum and minimum depths of calcium (figure 1, area in green). We tracked down the area of calcium determined by border delineation of the heterogenous calcium plaque. Calcium thickness (figure 1, yellow line) was analyzed on the slice with the maximum angle (figure 1). The length of calcium was derived by the total number of calcium-containing slices and then multiplied by the frame interval.
Figure 1. Optical coherence tomography frames showing a calcified plaque. The angle of calcium was determined using the center of the lumen as the vertex (red rays) and extending from one clearly delineated border of the calcium plaque to the other. Automatic software detection was used to identify the cap of the calcium plaque and the maximum and minimum depths of calcium (area in green). Calcium thickness (yellow line) was analyzed on the slice with the maximum angle after tracking down the area of calcium determined by the delineated borders of the heterogenous calcium plaque. Ca, calcium.
Intra- and inter-rater observer reproducibility
The intra-rater variability of the ICA and the OCT imaging analysis was assessed by evaluating 24 randomly selected images of primary lesions deemed inexistent/mild, moderate, and severe by 2 independent evaluators on both the ICA and the OCT. All OCT measurements including angle, thickness, length, and area were also measured. The same 2 evaluators analyzed the same 24 ICA and OCT images 4 weeks after the early evaluation.
The inter-rater variability of the ICA and the OCT imaging analysis was assessed by evaluating 50 randomly selected images of primary lesions deemed inexistent/mild, moderate, and severe by the same 2 independent evaluators on both the ICA and the OCT. All OCT measurements including angle, thickness, length, and area were also measured. The independent evaluator analyses were then compared. Both the inter and Intra-rater reproducibility were analyzed using Cohen’s kappa coefficient.
Statistical method
The comparison of all categorical variables (presented as counts and percentages) was performed using the chi-square test or Fisher’s exact test. Continuous data were compared used the Student t test. Continuous data were expressed as mean ± standard deviation for normally distributed variables or as median (interquartile range) for non-normally distributed variables. The sensitivity and specificity of the ICA with respect to the OCT were determined using standard 2 x 2 tables. Logistic regression determined the relationship between severity as seen on the angiography and the OCT measurements. The receiver operating characteristic (ROC) analysis established the optimal cut-off values using the area under the curve and Youden’s index.
RESULTS
Intra- and inter-rater observer reproducibility analysis
There was a 96% agreement (23/24; k = 0.92) on the intra-rater agreement between the analysts. This was indicative of an almost perfect inter-analysis agreement. There was only 1 case of disagreement between moderate calcification vs inexistent/mild calcification.
There was a 94% agreement (47/50; k = 0.72) on the inter-rater agreement between the analysts. This was indicative of substantial inter-rater agreement. There was disagreement between the analysts in 2 cases of moderate vs inexistent/mild calcification and in 1 case of moderate vs severe calcification.
Population
The baseline clinical characteristics of our patients are shown on table 1. Patient population was predominantly male with ages from 56.3 to 75.5. Most patients presented with unstable angina. Comorbidities were present in most of the patients being hypertension the most prevalent of all closely followed by hyperlipidemia. Smokers comprised over half of the patient population. The most common vessel imaged on the OCT was the left anterior descending coronary artery.
Table 1. Patient demographics and angiographic findings
| N = 75 | |
|---|---|
| Age, years | 65.9 ± 9.6 |
| Male | 55 (73.3) |
| Body height, cm | 171.6 ± 11.6 |
| Body weight, kg | 92.4 ± 20.3 |
| Creatinine levels, mg/dL | 1.12 ± 0.95 |
| Diabetes | 28 (37.33) |
| Hypertension | 59 (78.67) |
| Hyperlipidemia | 57 (76) |
| Smoker | 40 (53.33) |
| Hemodialysis | 2 (2.67) |
| Peripheral artery disease | 4 (5.33) |
| Previous myocardial infarction | 11 (14.67) |
| Previous coronary artery bypass graft | 4 (5.33) |
| Clinical presentation | |
| ST-elevation myocardial infarction | 0 (0) |
| Non-ST-elevation myocardial infarction | 7 (9.33) |
| Unstable angina | 43 (57.33) |
| Silent ischemia | 4 (5.33) |
| Angiographic findings | |
| Percutaneous coronary intervention | 61 (81.33) |
| Femoral access site | 63 (84) |
| Catheter Size, French | 6 |
| Target vessel | |
| Left main coronary artery | 1 (1.33) |
| Left anterior descending coronary artery/Diagonal branches | 61 (81.33) |
| Left circumflex artery/Ramus intermedius branch/Obtuse marginal | 10 (13.33) |
| Right circumflex artery/Posterior descending artery | 7 (9.59) |
| Lesion location | |
| Proximal | 40 (57.14) |
| Mid | 26 (37.14) |
| Distal | 4 (5.71) |
| Lesion and stent parameters | |
| Lesion length, mm | 25.84 ± 13.47 |
| Lesion stenosis | 74.74 ± 15.27 |
| Stent diameter, mm | 3.11 ± 0.53 |
| Stent length, mm | 24.62 ± 8.84 |
| Pullback distance, mm | 75 ± 12.2 |
|
Data are expressed as no. (%) or mean ± standard deviation. |
|
Angiographic severity and optical coherence tomography parameters
We examined a total of 75 lesions. The detection of CP lesions on the angiography in relation to the OCT is shown on figure 2. The angiography detected fewer CP lesions compared to the OCT that detected positive lesions (n = 69; 92%). All cases of positive angiography were detected by the OCT (n = 30; 100%). The OCT did not find any positive lesions in negative angiographic lesions (n = 6; 100%). A total of 43% of the lesions were both OCT positive and ICA positive. The ICA sensitivity was 95%CI, 0.32-0.56, and the ICA specificity, 95%CI, 0.52-1.0.
Figure 2. Calcified plaques lesions as seen on the angiography in relation to the OCT. A: OCT positive and negative values on the x-axis, and angio positive and negative values on the y-axis. The 4 x 4 table shows the correlation between the OCT and the angio measurements by primary lesion number. B: OCT positive and negative values on the x-axis, and angio positive and negative values on the y-axis. The 4 x 4 table shows the correlation between the OCT and the angio measurements by primary lesion percentage. C: total primary lesion numbers (in green color); the partition on the green color represents the primary lesions not found on the OCT or the angio. All OCT positive primary lesions are represented (in gray color); the partition on the gray color represents the OCT primary lesions not found on the angio. All angio positive lesions are shown (in red color); these lesions were all detected by the OCT. Angio, angiography; OCT, optical coherence tomography.
In most of cases, as the calcium angle (figure 3A), thickness (figure 3B), area (figure 3C), and length (figure 3D) increased on the OCT so did the calcium severity of the lesions on the angiography. The association between calcium severity as seen on the angiography and calcium length as seen on the OCT is shown on figure 3D. On the OCT, the severity of CP lesions run parallel to the increasing length seen on OCT.
Figure 3. A: angiographic lesions graded by severity inside the OCT angle measurements. All values are expressed in frequencies. Angles are in ranges of equal proportions based on the degrees seen. The most severe OCT lesions were found in the 91º-180° range followed by the 181º-270° OCT angle measurement. No severe CP lesions were found in the 0°, 1º-90° OCT angle measurements. B: angiographic lesions graded by severity inside the OCT thickness. All lesions are expressed as frequencies. The OCT thickness is expressed in mm and distributed in ranges that go from minimum to maximum values. The highest degree of angiographic calcium score-severe CP-was equally found in 0 mm-0.12 mm, and 0.12 mm-0.24 mm thicknesses on the OCT. C: angiographic lesions graded by severity inside the OCT area. All lesions are expressed as frequencies. The OCT area is expressed in mm2 and distributed in ranges that go from minimum to maximum values. The highest degree of angiographic calcium score-severe CP-was found from 4.8 mm2 to 6 mm2. D: angiographic lesions graded by severity inside the OCT length. All lesions are expressed as frequencies. The OCT length is expressed in mm and distributed in ranges that go from minimum to maximum values. The highest degree of angiographic calcium score- severe CP- was detected from 0.6 mm to 1.2 mm. CP, calcified plaques; ICA, interventional coronary angiography; OCT, optical coherence tomography.
DISCUSSION
The main findings of our study are: a) compared to the OCT, the ICA has a low sensitivity and a high specificity for the detection of calcium; b) as calcium angle, thickness, area, and length increased on the OCT so did the number of angio-defined severe CP lesions.
The ICA provides 2D real-time imaging with in-vivo characteristics of the lumen profile.15 Conversely, the invasive 3D-OCT imaging modality has the highest resolution to characterize variations in the composition of the plaque.11,16 The ICA detection of angiographic lesions has been used for decades. However, studies have shown that the ICA capabilities to detect calcified plaques in the arterial wall are poor.6,11,17 Some studies have compared the ICA characterization and quantification of plaque to the coronary computed tomography angiogram and the intravascular ultrasound, but few have looked into ICA plaque characterization and quantification with the OCT.6 Our study examined the sensitivity and specificity of ICA compared to the OCT. We examined 75 lesions and found that ICA sensitivity and specificity were 32%-56%, and 52%-100% with a 95%CI, respectively. Sensitivity was lower compared to former studies that showed a 50.9% sensitivity and a 95.1% specificity.6 The sensitivity of ICA is low because it only provides a 2D projection of the lesion and its resolution compared to the OCT is worse.18,19
The OCT detected all CPs present on the ICA (n = 30) and, also, lesions that were not present on the ICA (n = 39). On the angiography, the presence of CP is indicative that calcification has large CP characteristics on the OCT (eg, angle, thickness, and area). Our study concluded that severe calcifications on the ICA are seen with higher calcium angles on the OCT as the study conducted by Wang et al proved.6 The clinical implication of this is that when the ICA detects a calcified lesion, whether moderate or severe, the clinician can be sure that this calcification is, actually, present. The OCT would be the logical next step for a better characterization of the CP. Determining the morphology of the calcified lesion (eg, superficial, deep, or nodular) on the OCT allows selecting the optimal lesion preparation strategy. Also, the OCT detected calcifications that the ICA simply could not find, indicative that the ICA alone is not reliable to detect CPs. Therefore, with suspected lesions, the OCT should be the next step for a comprehensive assessment of these lesions.
The OCT measurements of a calcified lesion thickness, length, and area are unique to this technology because the OCT is the only invasive imaging modality capable of measuring these values.6 Thicknesses > 0.5 mm are associated with stent underexpansion.7,20 We did not explore this in our population since not all lesions received percutaneous coronary intervention. We did expand, however, the OCT analysis to include the depth, and area of calcium on the OCT. We found that as the area increased on the OCT so did the number of severe lesions on the ICA. We found that most severe CP lesions were in the 4.8-6 mm2 range. Perhaps, calcium areas > 5 mm2 may be the fourth “5” in the OCT-based “rule of five” that identifies the CP features associated with poor stent expansion.7
Study limitations
This was a retrospective observational study with its inherent limitations. The sample size was relatively small.
CONCLUSIONS
Invasive coronary angiography has a low sensitivity and a high specificity for the detection of calcified plaques compared to the OCT. As calcium angle, thickness, area, and length increased on the OCT so did number of angio-defined severe CP lesions.
FUNDING
None.
AUTHORS’ CONTRIBUTIONS
C. McGuire: study idea, data mining, manuscript draft, and analysis; E. Schlofmitz: study idea, data mining, critical review of the manuscript; G. D. Melaku, K. O. Kuku, and Y. Kahsay: data mining, critical review of the manuscript; R. Schlofmitz, and A. Jeremias: writing, critical review of the manuscript; H. M. Garcia-Garcia: study idea, data analysis, data mining, preparation, and critical review of the manuscript.
CONFLICTS OF INTEREST
H.M. Garcia-Garcia declared having received institutional grant support from Biotronik, Boston Scientific, Medtronic, Abbott, Neovasc, Shockwave, Phillips, and Corflow. The remaining authors declared no conflicts of interest.
WHAT IS KNOWN ABOUT THE TOPIC?
- Percutaneous coronary interventions rely on angiography to inform most of the clinical decisions on lesion preparation; however, the extent of calcium is poorly assessed on the angiography.
- The relation between the ICA and the OCT regarding the severity of CP was examined using thickness and angle measurements on the OCT.
- No examination has been conducted of all OCT measurements and their relation to the severity of CP as seen on the ICA.
WHAT DOES THIS STUDY ADD?
- Compared to the OCT, the ICA has a low sensitivity but a high specificity to detect severely calcified plaques.
- As calcium increased on the OCT measurements regarding area, length, thickness, and angle so did the number of angio-defined severe CP lesions, which is indicative that all OCT measurements can be used to detect severely calcified lesions.
- The OCT offers a feasible alternative to the angiography regarding calcium assessment; it extends calcium characterization by providing detailed information to shed light on the use of dedicated calcium debulking therapies for lesion preparation.
REFERENCES
1. Miao Benjamin, Hernandez Adrian V., Alberts Mark J., Mangiafico Nicholas, Roman Yuani M., Coleman Craig I. Incidence and Predictors of Major Adverse Cardiovascular Events in patients With Established Atherosclerotic Disease or Multiple Risk Factors. J Am Heart Assoc. 2020;9:e014402.
2. Guedeney P, Claessen BE, Mehran R, et al. Coronary Calcification and Long-Term Outcomes According to Drug-Eluting Stent Generation. JACC Cardiovasc Interv. 2020;13:1417-1428.
3. Costa JR, Sousa A, Moreira AC, et al. Incidence and Predictors of Very Late (?4 Years) Major Cardiac Adverse Events in the DESIRE (Drug-Eluting Stents in the Real World)-Late Registry. JACC Cardiovasc Interv. 2010;3:12-18.
4. Conway C, McGarry JP, Edelman ER, McHugh PE. Numerical Simulation of Stent Angioplasty with Predilation: An Investigation into Lesion Constitutive Representation and Calcification Influence. Ann Biomed Eng. 2017;45:2244-2252.
5. Waters DD, Azar RR. The Curse of Target Lesion Calcification. J Am Coll Cardiol. 2014;63:1855-1856.
6. Wang X, Matsumura M, Mintz GS, et al. In Vivo Calcium Detection by Comparing Optical Coherence Tomography, Intravascular Ultrasound, and Angiography. JACC Cardiovasc Imaging. 2017;10:869-879.
7. Fujino A, Mintz G, Matsumura M, et al. TCT-28. A New Optical Coherence Tomography-Based Calcium Scoring System to Predict Stent Underexpansion. J Am Coll Cardiol. 2017;70 (18, Supplement):B12-B13.
8. Lee T, Mintz GS, Matsumura M, et al. Prevalence, Predictors, and Clinical Presentation of a Calcified Nodule as Assessed by Optical Coherence Tomography. JACC Cardiovasc Imaging. 2017;10:883-891.
9. Mintz GS, Popma JJ, Pichard AD, et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation. 1995;91:1959-1965.
10. Brezinski ME, Tearney GJ, Bouma BE, et al. Imaging of coronary artery microstructure (in vitro) with optical coherence tomography. Am J Cardiol. 1996;77:92-93.
11. Wang Ying, Osborne Michael T., Tung Brian, Li Ming, Li Yaming. Imaging Cardiovascular Calcification. J Am Heart Assoc. 2018;7:e008564.
12. Oosterveer TTM, van der Meer SM, Scherptong RWC, Jukema JW. Optical Coherence Tomography: Current Applications for the Assessment of Coronary Artery Disease and Guidance of Percutaneous Coronary Interventions. Cardiol Ther. 2020;9:307-321.
13. Gharaibeh Y, Prabhu DS, Kolluru C, et al. Coronary calcification segmentation in intravascular OCT images using deep learning: application to calcification scoring. J Med Imaging (Bellingham). 2019;6:045002.
14. Kume T, Akasaka T, Kawamoto T, et al. Assessment of Coronary Intima - Me-dia Thickness by Optical Coherence Tomography. Circ J. 2005;69:903-907.
15. Ryan Thomas J. The Coronary Angiogram and Its Seminal Contributions to Cardiovascular Medicine Over Five Decades. Circulation. 2002;106:752-756.
16. Kubo T, Imanishi T, Takarada S, et al. Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J Am Coll Cardiol. 2007;50:933-939.
17. Tuzcu EM, Berkalp B, De Franco AC, et al. The dilemma of diagnosing coronary calcification: Angiography versus intravascular ultrasound. J Am Coll Cardiol. 1996;27:832-838.
18. Lee CH, Hur S-H. Optimization of Percutaneous Coronary Intervention Using Optical Coherence Tomography. Korean Circ J. 2019;49:771-793.
19. Park S-J, Kang S-J, Ahn J-M, et al. Visual-functional mismatch between coronary angiography and fractional flow reserve. JACC Cardiovasc Interv. 2012;5:1029-1036.
20. Fujino A, Mintz GS, Lee T, et al. Predictors of Calcium Fracture Derived From Balloon Angioplasty and its Effect on Stent Expansion Assessed by Optical Coherence Tomography. JACC Cardiovasc Interv. 2018;11:1015-1017.
* Corresponding author: Division of Interventional Cardiology, MedStar Washington Hospital Center, 100 Irving St, NW Washington D.C. 20010, United States.
E-mail addresses: ; (H.M. Garcia-Garcia)
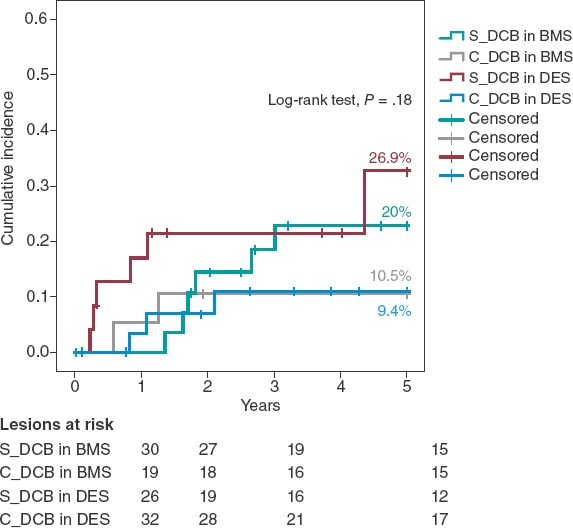
ABSTRACT
Introduction and objectives: Drug-eluting balloon (DEB) angioplasty is an effective technique to treat in-stent restenosis (ISR). Neointimal modification with cutting balloon (CB) or scoring balloon (SB) enhances the angiographic results of DEB, but with no benefits have been reported in the clinical endpoints at the mid-term. There is lack of information on the clinical long-term results of this strategy. We aim to compare very long-term results of CB before DEB vs standard DEB to treat real-world patients with ISR.
Methods: Retrospective cohort registry of DEB PCIs to treat ISR defined by the use of CB. The primary endpoint was clinically driven target lesion revascularization (TLR) at 5 years. The secondary endpoints were based on the ARC-2 criteria.
Results: From January 2010 to December 2015, 107 ISRs were treated with DEB in 91 patients. CBs were used in 51 lesions (46 patients). Both cohorts were well balanced regarding clinical, lesion, and procedural characteristics. Compared to standard DEBs, CBs showed lower, although statistically non-significant rates, of TLR at 5 years (9.8% vs 23.6%, OR, 0.36; 95% confidence interval 0.19 to 1.09 P = .05). The Kaplan-Meier cumulative incidence of time until TLR showed similar results (log-rank test P value = .05) with similar rates of TLR at 1 year (3.9% vs 7.1%, P = .68) as curve separation in the long-term. There were no differences in the secondary endpoints. No stent thrombosis was reported.
Conclusions: In a real-world setting, neointimal modification with CB before DEB vs standard DEB to treat ISR shows lower, although statistically non-significant rates of TLR at 5 years. This benefit has been confirmed in the long-term and is consistent with bare-metal and drug-eluting stents.
Keywords: Drug-eluting balloon. In-stent restenosis. Cutting/scoring balloon.
RESUMEN
Introducción y objetivos: El uso de balón farmacoactivo (BFA) es una estrategia efectiva en el tratamiento de la reestenosis de stents coronarios (RIS). La modificación neointimal con balón de corte (BC) o incisión junto con BFA se asocia a mejores resultados angiográficos, aunque sin impacto en eventos clínicos a medio plazo. Los resultados clínicos de esta estrategia a muy largo plazo en la vida real son desconocidos. Se evaluó la eficacia de BC junto con BFA frente a BFA estándar en un registro de pacientes de la vida real con RIS a muy largo plazo (5 años).
Métodos: Registro retrospectivo de 2 cohortes de pacientes con RIS tratados con BFA, definidas por el uso de BC. El evento primario fue la tasa de revascularización clínicamente indicada de la lesión tratada a 5 años. Se valoraron eventos secundarios según los criterios ARC-2.
Resultados: Entre enero de 2010 y diciembre de 2015 se usó BFA en 107 RIS en 91 pacientes. En 51 lesiones (46 pacientes) se utilizó BC. Ambas cohortes presentaron similares características clínicas y de procedimiento. Respecto al uso estándar de BFA, el BC consiguió una reducción numérica, pero no significativa, en la tasa de revascularización de la lesión tratada a 5 años (9,8% frente a 23,6%; odds ratio = 0,36; intervalo de confianza del 95%, 0,19-1,09; p = 0,05). El análisis de incidencia acumulada de Kaplan-Meier mostró resultados parecidos (log-rank, p = 0,05), con similar tasa de eventos a 1 año (3,9% frente a 7,1%; p = 0,68), y separación de las curvas con el tiempo. No se evidenciaron diferencias en los eventos secundarios. No hubo trombosis de stent en la cohorte.
Conclusiones: En una cohorte de la vida real, la modificación neointimal de la RIS con BC junto con BFA, en comparación con BFA estándar, logra una reducción numérica, pero no significativa, en la tasa de revascularización de la lesión tratada a 5 años. El beneficio de esta estrategia se evidencia a largo plazo y es consistente entre RIS de stent convencional y de stent farmacoactivo.
Palabras clave: Balon farmacoactivo. Reestenosis. Balon de corte.
Abreviaturas
BC: balón de corte o incisión. BFA: balón farmacoactivo. RIS: reestenosis de stent coronario. RLT: revascularización de la lesión tratada. SFA: stent farmacoactivo. SM: stent convencional.
INTRODUCTION
In-stent restenosis (ISR) is a common problem in the routine clinical practice regarding percutaneous coronary intervention (PCI), and its management is associated with high rates of target lesion revascularization (TLR).1 Together with the implantation of a new everolimus drug-eluting stent, the PCI with drug-coated balloon (DCB) is the strategy of choice to treat ISR after bare-metal stent (BMS) and drug-eluting stent (DES) implantation, and has a class I indication after confirmation that it can reduce the rate of TLR at the follow-up without having to implant a new layer of metal into the artery.2-5 Despite of this, TLR is still high in the long-term (up to 20% at 3 years),6-11 which is suggestive that new strategies may be needed to improve these results.
The cutting balloon (CB) consists of small blades or nitinol bands on its surface to optimize the predilatation of coronary lesions by performing controlled fractures of the atheromatous plaque. Compared to the plain old balloon angioplasty, its use for the management of ISR is associated with structural changes of the neointima and acute improvements of the lumen area,12 although no angiographic or clinical benefit has been reported in the mid-term.13,14
The efficacy of the DCB depends on the transfer of drug from the surface of the balloon to the tissue where it exerts it antiproliferative effect.15 Theoretically speaking, greater the neointimal disarrays are associated with more effective transfers and smaller issue thickness. As a matter of fact, preclinical studies have suggested a greater effect of DCB inhibiting neointimal growth.16 This greater disarray and reduction of the neointima can be achieved using a CB before the DCB.
Although this hypothesis has not been confirmed in animal models in the short-term,17 the strategy has shown better angiographic results in the mid-term (6 to 8 months) (significant reduction of binary restenosis), but no effect on TLR or clinical events at the 1-year follow-up.18 No long-term results have been published on the use of this strategy.
Our objective was to assess the very long-term results of the use of CB plus DCB to treat ISR.
METHODS
Retrospective registry of cohorts of real-world patients with, at least, 1 ISR treated with DCB at a single high-volume PCI center (> 800/year) and a 5-year follow-up. Two different cohorts were defined based on the use of CB prior to the PCI with DCB (C_DCB) or standard DCB (S_DCB). The C_DCB cohort was defined by the use of, at least, 1 cutting balloon (Flextome Cutting Balloon, Boston Scientific, United States) or 1 scoring balloon (ScoreFlex, OrbusNeich, China). The use of the CB was left to the operator’s discretion. The ISR was defined as an angiographic stenosis > 50% in 2 different orthogonal radiographic projections inside the stent or < 5 mm from its borders plus symptoms of angina or objective confirmation of myocardial ischemia or fractional flow reserve/positive instantaneous wave-free ratio. Lesions were treated with 2 types of drug-coated balloons based on their availability at the time: the SeQuent Please (B. Braun Surgical, Germany) or the Pantera Lux (Biotronik, Switzerland). Data on the long-term progression of patients with ISR treater with the SeQuent Please DCB in this cohort regardless of the use of CB were reported beforehand.19
Exclusion criteria were cardiogenic shock or cardiac arrest in the index event, the presence of ≥ 3 layers of metal in the lesion with ISR and a contraindication to dual antiplatelet therapy with acetylsalicylic acid and a P2Y12 inhibitor for, at least, a month.
The clinical and procedural characteristics were obtained from the center and the cath lab databases. The coronary study of the lesions was performed with the Xcelera system (Philips, The Netherlands) using the projection with the highest degree of stenosis. The Mehran classification of ISR was used to categorize the lesions.20 The strategy of the procedure including the use and type of CB was left to the operator’s criterion. DCB dilatation lasted for, at least, 60 seconds at nominal pressure. The PCI, management, and previous and later treatment of the patients was performed based on the routine clinical practice.
The study was conducted in observance of the criteria established at the Declaration of Helsinki and the International Council on Harmonization Good Clinical Practice guidelines (ICH-GCP). Also, it was authorized by Hospital Clínico Lozano Blesa (Zaragoza, Spain) management and ethics committee. No informed consents were needed given the retrospective nature of the study. A 5-year long follow-up period was arranged. Every follow-up was performed by checking the electronic database of the regional healthcare system where all the patient’s clinical events were thoroughly detailed. Data were anonymized through internal numerical identification at the cath lab.
All events were defined in a standard way according to the ARC-2 consensus.21 The primary endpoint was the need for TLR with a clinically indicated DCB at 5 years and estimated on the overall number of all target lesions. Clinically indicated TLR was defined as a new-onset ISR > 70% or > 50% of the target lesion in the presence of ischemic symptoms, a positive inducible ischemia on stress testing dependent on the vessel or fractional flow reserve values ≤ 0.80 or instantaneous wave-free ratio values ≤ 0.89.
Secondary endpoints were the presence or lack of target vessel revascularization, and target vessel myocardial infarction (according to the universal definition22), all-cause mortality, death due to cardiac causes (acute myocardial infarction, severe arrhythmia, heart failure, unwitnessed or unknown death) or cardiovascular death (cardiac or stroke induced or due to other cardiovascular processes), BARC type ≥ 3 bleeding, stroke (new neurologic deficit > 24 h duration) or a composite endpoint of target lesion failure (TLR + target vessel myocardial infarction + cardiovascular death), target vessel failure (target vessel revascularization + target vessel myocardial infarction + cardiovascular death) or patient-oriented composite endpoint (any revascularization + acute myocardial infarction + stroke + overall death). These endpoints were estimated on the overall number of patients. Definitive or probable stent thrombosis was also defined based on the ARC-2 criteria and estimated on the overall number of lesions.
Data mining and analysis were performed using the SPSS 19.0. statistical software (IBM, United States). Quantitative variables were expressed as mean and standard deviation. Qualitative variables were expressed as relative percentage. The cumulative incidence of the endpoints at the follow-up was also estimated. The variables and the group endpoints studied were compared on a bivariate analysis using the chi-square test (or Fisher’s exact test, when appropriate) or the Student t test regarding the quantitative variables. Cox regression analysis was performed to estimate the primary endpoint predictors (including the variables associated with P values < .1). Survival was analyzed using the Kaplan-Meier method to build the cumulative incidence curve of time to the primary endpoint based on the strategy of treatment used. P values < .05 were considered statistically significant.
RESULTS
A total of 107 ISRs were treated with DCBs in 95 procedures performed on 91 patients from January 2010 through December 2015 (in 4 patients the PCI with DCB was repeated at the follow-up, in 1 case using a different DCB on the same previously treated lesion). A total of 51 lesions (42 patients) were treated with a PCI plus CB + DCB (C_DCB), and 56 lesions (49 patients) with standard DCB (S_DCB). A total of 53 lesions were treated with the SeQuent Please device, and 54 with the Pantera Lux. The cutting balloon and the scoring balloon were used in 36 and 15 lesions, respectively.
The study cohorts were similar regarding the clinical characteristics (table 1), and the lesion and procedural characteristics (table 2). Some of the differences reported in the C_DCB group where that radial access was more common, and the size of the stent and minimum lumen diameter were greater, although with a similar percent diameter stenosis of the lesion before and the after the PCI. Patients had a high prevalence of cardiovascular risk factors including diabetes in 35% of the cases. A total of 47 new coronary angiographies were performed at the follow-up. In 29 of these the target lesion had good results. The rate of new coronary angiography was similar in both groups (44.6% vs 41.2% in the C_DCB group. P = .71). A total of 18 TLRs were performed at the follow-up (16.8%) of which 17 were treated with a PCI (16 stent-in-stent), and 1 with coronary artery bypass graft. The rate of TLR was numerically lower in the C_DCB group at 1 (3.9% vs 7.1%; P = .68) and 3 years (9.8% vs 17.9%; P = .23). Compared to the S_DCB strategy, the use of the C_DCB reduced the 5-year rate of TLR although not statistically significant (9.8% vs 23.2%; OR, 0.36; 95% confidence interval [95%CI], 0.19-1.09; P = .05). The Kaplan-Meier analysis of the cumulative incidence curve revealed the differences seen at the 5-year follow-up (log-rank test, P = .05) with a similar 1-year event rate and curve separation consistent with the passing of the follow-up period (figure 1).
Table 1. Baseline characteristic of the patients
| S_DCB | C_DCB | P | |
| N = 49 patients/ 56 lesions | N = 42 patients/ 51 lesions | ||
| Age | 68.9 ± 11.3 | 67.7 ± 10 | .58 |
| Male | 85.7% (35) | 83.3% (35) | .75 |
| Arterial hypertension | 26.8% (14) | 23.8% (10) | .6 |
| Dyslipidemia | 46.9% (23) | 28.6% (12) | .7 |
| Smoking | 61.2% (30) | 57.1% (24) | .69 |
| Diabetes | 37.5% (21) | 35.3% (18) | .81 |
| AF in oral anticoagulants | 22.4% (11) | 19% (8) | .38 |
| Previous myocardial infarction | 55.1% (27) | 50% (21) | .62 |
| Previous coronary artery bypass graft | 6.1% (3) | 4.8% (2) | 1 |
| CKD (GFR < 60mL/min) | 32.7% (16) | 33.3% (14) | .94 |
| LVEF (%) | 54 ± 10 | 55 ± 9 | .51 |
|
AF, atrial fibrillation; CKD, chronic kidney disease; GFR, glomerular filtration rate; LVEF, left ventricular ejection fraction. |
|||
Table 2. Lesion and procedural characteristics
| S_DCB | C_DCB | P | S_DCB | C_DCB | P | |||
|---|---|---|---|---|---|---|---|---|
| N = 49 patients/ 56 lesions | N = 42 patients/ 51 lesions | N = 49 patients/ 56 lesions | N = 42 patients/ 51 lesions | |||||
| Procedural characteristics | Lesion characteristics | |||||||
| Clinical signs | .87 | Location of ISR | .35 | |||||
| Stable angina | 55.4% (31) | 56.9% (29) | LAD | 53.6% (30) | 45.1% (23) | |||
| Unstable angina/NSTEACS | 41.1% (23) | 41.2% (21) | LCX | 23.2% (13) | 15.7% (8) | |||
| STEACS | 3.6% (2) | 2% (1) | RCA | 16.1% (9) | 31.4% (16) | |||
| Radial access | 55.4% (31) | 78.4% (40) | .01 | LMCA | 5.4% (3) | 3.9% (2) | ||
| DCB caliber (mm) | 3.03 ± 0.37 | 3.15 ± 0.42 | .13 | Coronary artery bypass graft | 1.8% (1) | 3.9% (2) | ||
| DCB length (mm) | 20.2 ± 5.8 | 19.5 ± 4.7 | .53 | Mehran's angiographic classification of ISR pattern | .42 | |||
| DCB inflation pressure (atm) | 14 ± 3 | 14 ± 3 | .81 | IA | 1.8% (1) | 3.9% (2) | ||
| CB caliber (mm) | N/A | 2.93 ± 0.45 | IB | 3.6% (2) | 0% (0) | |||
| CB length (mm) | N/A | 8 ± 3 | IC | 41.1% (23) | 49% (25) | |||
| CB inflation pressure (atm) | N/A | 14 ± 3 | ID | 1.8% (1) | 3.9% (2) | |||
| NCB | 53.6% (30) | 70.6% (36) | .07 | II | 21.4% (12) | 27.5% (14) | ||
| NCB caliber (mm) | 3.12 ± 0.42 | 3.28 ± 0.43 | .14 | III | 21.4% (12) | 11.8% (6) | ||
| NCB length (mm) | 13.2 ± 3.1 | 12.6 ± 3.8 | .65 | IV | 8.9% (5) | 3.9% (2) | ||
| NCB inflation pressure (atm) | 18 ± 4 | 18 ± 3 | .74 | ISR based on type of stenting | .4 | |||
| Intracoronary imaging | 8.9% (5) | 5.9% (3) | .55 | BMS | 53.6% (30) | 37.3% (19) | ||
| Multivessel disease | 62.7% (32) | 47.7% (21) | .14 | DES | 33.9% (19) | 45.1% (23) | ||
| Complete revascularization | 82.4% (42) | 93.2% (41) | .13 | DES in BMS | 8.9% (5) | 11.8% (6) | ||
| P2Y12 inhibitor | .64 | DES in DES | 3.6% (2) | 5.9% (3) | ||||
| Clopidogrel | 88.2% (45) | 81.6% (36) | Time from implantation | 4.1 ± 4.8 | 3.8 ± 5 | .69 | ||
| Prasugrel | 3.9% (2) | 4.5% (2) | Bifurcation | 32.1% (18) | 23.5% (12) | .32 | ||
| Ticagrelor | 7.8% (4) | 13.6% (6) | Stent caliber (mm) | 2.96 ± 0.43 | 3.1 ± 0.56 | .02 | ||
| Duration of dual antiplatelet therapy | .27 | Stent length (mm) | 22.4 ± 6.5 | 22.8 ± 7.1 | .75 | |||
| 1 month | 3.9% (2) | 2.3% (1) | Reference diameter (mm) | 2.98 ± 0.48 | 3.12 ± 0.53 | .16 | ||
| 3 months | 21.6% (11) | 9.1% (4) | Minimum lumen diameter (mm) | 0.73 ± 0.51 | 0.68 ± 0.5 | .67 | ||
| 6 months | 21.6% (11) | 34.1% (15) | Length (mm) | 13.2 ± 5.6 | 11.7 ± 5.3 | .18 | ||
| 12 months | 52.9% (27) | 54.5% (24) | Stenosis (%) | 72 ± 18 | 75 ± 16 | .3 | ||
| Minimum lumen diameter post-PCI (mm) | 2.43 ± 0.46 | 2.77 ± 0.62 | .002 | |||||
| Acute lumen gain (mm) | 1.7 ± 0.64 | 2.08 ± 0.83 | .01 | |||||
| Stenosis post-PCI (%) | 14 ± 5 | 14 ± 6 | .45 | |||||
| Final TIMI grade 3 flow | 98.2% (55) | 100% (51) | 1 | |||||
|
BMS, bare-metal stent; CB, cutting balloon; DCB, drug-coated balloon; DES, drug-eluting stent; ISR, in-stent restenosis; LAD, left anterior descending coronary artery; LCX, left circumflex artery; LMCA, left main coronary artery; NCB, non-compliant balloon; NSTEACS, non-ST-segment elevation acute coronary syndrome; PCI, percutaneous coronary intervention; RCA, right coronary artery; STEACS, ST-segment elevation acute coronary syndrome; TIMI, Thrombolysis in Myocardial Infarction. |
||||||||
Figure 1. Kaplan-Meier analysis of the 5-year cumulative incidence of target lesion revascularization. DCB, drug-coated balloon.
The 5-year cumulative incidence of secondary endpoints is shown on table 3. The incidence rate of target vessel-related composite endpoints (target lesion failure and target vessel failure) was numerically lower in the C_DCB group although not statistically significant. No differences were found in the remaining secondary endpoints. The overall mortality rate at the follow-up was 31.8% (n = 19) being neoplasms the most common cause (n = 7). The incidence rates of stroke and patient-oriented composite endpoint were high (10.9% and 51.6%, respectively), which was consistent with an old cohort with high cardiovascular risk. No cases of definitive or probable stent thrombosis were seen at the follow-up.
Table 3. 5-year cumulative incidence of primary and secondary endpoints
| S_DCB | C_DCB | P | |
|---|---|---|---|
| N = 49 patients/ 56 lesions | N = 42 patients/ 51 lesions | ||
| Primary endpoint | |||
| TLR (clinically justified) | 23.2% (13/56) | 9.8% (5/51) | .05 |
| Secondary endpoints | |||
| Target vessel revascularization | 28.6% (16/56) | 17.6% (9/51) | .18 |
| Any revascularization | 28.6% (14/49) | 26.2% (11/42) | .8 |
| Target vessel myocardial infarction | 7.1% (4/56) | 5.9% (3/51) | .79 |
| Myocardial infarction | 18.3% (9/49) | 7.2% (3/42) | .19 |
| Death due to cardiac causes | 4.1% (2/49) | 4.8% (2/42) | 1 |
| Cardiovascular death | 16.3% (8/49) | 11.9% (5/42) | .54 |
| Overall mortality | 36.7% (18/49) | 26.2% (11/42) | .28 |
| Stroke | 10.2% (5/49) | 11.9% (5/42) | .55 |
| BARC type 3-5 bleeding | 7.1% (4/49) | 3.9% (2/42) | .68 |
| Target lesion failure | 37.5% (21/56) | 25.5% (13/51) | .18 |
| Target vessel failure | 41.1% (23/56) | 25.5% (13/51) | .08 |
| POCE | 53.1% (26/49) | 50% (21/42) | .77 |
|
BARC, Bleeding Academic Research Consortium; DCB, drug-coated balloon; POCE, patient-oriented composite endpoints; TLR, target lesion revascularization. |
|||
A Kaplan-Meier subanalysis based on ISR after BMS or DES implantation showed that the C_DCB strategy consistently reduced the 5-year rate of TLR in half with both types of stent although not statistically significant (figure 2).
Figure 2. Kaplan-Meier analysis of the 5-year cumulative incidence of target lesion revascularization based on whether the stent is made out of metal or is drug-eluting. BMS, bare-metal stent; DCB, drug-coated balloon, DES, drug-eluting stent.
Aside from the C_DCB no association was found between the variables and the 5-year rate of TLR except for the location of ISR that was 100% in cases found in coronary artery bypass graft stents (3 cases) compared to 14.4% in cases found in the native coronary tree (P = .003). The 5-year rate of TLR was similar in diabetic patients (17.9% vs 16.2%; P = .81) in the ISR of DESs (17.2% vs 16.3%; P = .9) and in stents < 3 mm (12.9% vs 18.4%; P = .58) without any differences based on the type of DCB used (Sequent, 20.4% vs Pantera, 13.2%; P = .32). In the Cox regression analysis, the use of the C_DCB was not an independent predictor of TLR at 5 years being the ISR of a coronary artery bypass graft the only independent predictor (OR, 5.4; 95%CI, 1.5-19.8; P = .01).
DISCUSSION
As far as we know, the study presented here is the first one to confirm:
-
- The use of a CB in connection with a DCB in the ISR setting shows a tendency to reduce the rate of TLR.
-
- The benefit of this strategy is evident in the long-term.
-
- The benefit seems to be consistent in ISR after BMS and DES implantation.
-
- The strategy is safe and there are no traces of stent thrombosis when a CB is used.
Compared to the plain old balloon angioplasty for the management of ISR, the CB achieves greater lumen areas because it breaks down the elastic and fibrotic continuity of the neointima by reducing its integrity and resistence.12 However, this acute angiographic improvement is not associated with lower but high rates of TLR (18% to 29%) at the 1-year follow-up.13,14 Similarly, in our series, the use of the CB is associated with a significant increase of minimum lumen diameter and acute gain after the procedure (table 2) despite the fact that the caliber of non-compliant balloons and DCBs was similar between both groups. Although stent diameter was slightly larger in the C_DCB group, the final percent stenosis did not change significantly between both groups; still, this may be an important piece of information in our results since the size of the vessel has been described as an independent predictor of new restenosis.23
The use of the DCB to treat ISR is something common after several meta-analyses revealed that, together with DES implantation with in-stent everolimus, this strategy is the most effective one to avoid new revascularizations.2-4 Afterwards, in the RIBS IV (with DES) and RIBS V (with BMS) clinical trials Alfonso et al. proved the long-term superiority of DES implantation with in-stent everolimus.8,9,24 However, the philosophy of not adding a new metal layer (or delay it through time) and questions associated with its long-term safety10,11 have turned DCB implantation into a common practice to treat ISR. Added to the RIBS IV-V studies, other trials have reported on the long-term effectiveness of DCB (PEPCAD7 with BMS, and PEPCAD-DES6 and ISAR-DESIRE 310 with DES). Overall, in these 5 studies, a total of 94 TLRs were reported in 524 ISRs treated with DCB, which is a 3-year rate of TLR of 17.9%. These results are accurately reproduced in our S_DCB cohort with rates high enough to justify looking into ways to improve the efficacy of DCBs.
The efficacy of DCBs is based on a transfer of the drug to the neointima of ISR where it exerts its antiproliferative effect. The proper preparation of the lesion by reducing neointimal thickening and increasing the surface of contact with the balloon is the key to achieve successful DCB implantations.15 Preclinical studies suggest that greater neointimal disarrays can increase the release and retention of the drug into the tissue, thus increasing its effects.16 Considering the greater acute lumen gain and controlled disarray that the CB provides, results can improve if used together with the DCB. This hypothesis was put to the test, but not proven, in a preclinical trial. The reason was that the use of the CB was not associated with a lower neointimal volume or acute lumen loss. Nonetheless, this assessment was made was very early (28 days).17
The synergistic effects of CB plus DCB were also confirmed by Scheller et al.25 in the PATENT-C trial. They took a different angle and studied the addition of an antiproliferative drug (paclitaxel) to the scoring balloon that reduced the 1-year rate of TLR significantly (3% vs 32%; P = .004). This information is consistent with the 1-year rate of TLR of 3.9 seen in our C_DCB cohort. From a new and different angle too, while still observing the philosophy of not leaving any material behind in the long-term after the PCI, Alfonso et al. conducted the RIBS VI Scoring trial and analyzed the impact of a CB before bioresorbable scaffold implantation to treat ISR. However, the 1-year rate of TLR was not reduced (9.8 vs 11.1%).26
Two randomized clinical trials have assessed the effect of CB implantation before DCB implantation to treat ISR. Aoki et al.27 found no angiographic differences at the 8-month follow-up in the ELEGANT trial. However, this was a comparative study vs a non-compliant balloon. Kufner et al.18 specifically tested the effects of CB implantation in the ISAR-DESIRE 4 trial. The primary endpoint was an angiographic result that confirmed that this strategy effectively reduced binary ISR at the 6 to 8-month follow-up. However, no differences were seen when the clinical events or TLR were assessed at the 1-year follow-up (16.2% vs 21.8%; P = .26). Qualitatively speaking, these results are consistent with what our series described because, although long-term benefits were reported, the 1-year rate of TLR did not change between our groups. No long-term data have ever been published so our cohort cannot be compared to corroborate the benefits described. Quantitatively speaking, we saw differences in the 1-year rate of TLR, much lower in our study (3.9% vs 7.1%). Three may be the reasons for this. In the first place, the scheduled angiographic assessment of the ISAR-DESIRE 4 trial because if we look at the Kaplan-Meier analysis of the TLR, in this study more clinical events were reported at the 6 to 8-month follow-up (when the angiographic assessment occurred). This is suggestive of a TLR guided by angiographic criteria (the so-called oculodilatory reflex) and not clinically justified as it was the case in our series. Secondly, the exclusive use of the scoring balloon vs the predominant use of the CB in our series since the CB achieves greater neointimal disarray and larger residual lumen diameters, thus increasing the efficacy of the DCB. Thirdly, the exclusive management of ISR after DES implantation vs ISR after any other type of stent implantation (BMS or DES) of our series since different authors have proposed the lower efficacy of the DCB to treat ISR after DES implantation.11,28 Based on this previous knowledge a subanalysis of the C_DCB strategy based on the type of stent used was conducted (figure 2). A consistent efficacy both in BMSs and DESs was seen with a similar 5-year rate of TLR in both subgroups (10.5% and 9.4% respectively)
Treating ISR with DCBs is a safe strategy associated with very low rates of stent thrombosis (around 1%) at the long-term follow-up.11 The role that a greater CB-induced neointimal tissue disarray plays in the appearance of thrombotic phenomena on the lesion is unknown. Consistent with the mid-term results of theISAR-DESIRE 4 trial, in our series, long-term target lesion thrombosis is null, which is a guarantee that the use of C_DCB is safe.
Limitations
Our study has several limitations. It is a retrospective, observational, and single-center study. Although the use of the DCB is the treatment of choice for the management of ISR in our center, it is possible that patients with more unfavorable ISR may have been excluded for having been treated with a DES. The use of intracoronary imaging was limited and the characterization of ISR could have given relevant information on the therapeutic strategy used and its long-term results. The size of the sample was not big enough to obtain powerful evidence. A larger sample size and longer follow-up is, therefore, guaranteed.
CONCLUSIONS
In a real-world cohort, changing the neointima of ISR with CB plus DCB vs standard DCB reduces the 5-year rate of TLR although not statistically significant. The benefit of this strategy is evident in the long-term and consistent between ISR after BMS and DES implantation.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
J.A. Linares Vicente: study design, data mining, and analysis and writing of the manuscript. J.R. Ruiz Arroyo: data and critical review of the manuscript. A. Lukic, B. Simó Sánchez, and O. Jiménez Meló: data mining. A. Riaño Ondiviela, P. Morlanes Gracia, and P. Revilla Martí: data mining.
CONFLICTS OF INTEREST
None reported.
WHAT IS KNOWN ABOUT THE TOPIC?
- The use of CB to treat ISR with DCB has been associated with better angiographic results although with no impact on the mid-term clinical events. The clinical outcomes of this long-term strategy are still unknown.
WHAT DOES THIS STUDY ADD?
- The use of CB plus DCB to treat ISR is associated with lower rates of TLR. The benefit of this strategy has been reported in the long-term. This benefit seems to be consistent with both ISR after BMS and DES implantation.
REFERENCES
1. Alfonso F, Byrne RA, Rivero F, Kastrati A. Current Treatment of In-Stent Restenosis. J Am Coll Cardiol. 2014;63:2659-2673.
2. Giacoppo D, Gargiulo G, Aruta P, Capranzano P, Tamburino C, Capodanno D. Treatment strategies for coronary in-stent restenosis: systematic review and hierarchical Bayesian network meta-analysis of 24 randomised trials and 4880 patients. BMJ. 2015;351:h5392.
3. Siontis GCM, Stefanini GG, Mavridis D, et al. Percutaneous coronary interventional strategies for treatment of in-stent restenosis: a network meta-analysis. Lancet. 2015;386:655-664.
4. Sethi A, Malhotra G, Singh S, Singh PP, Khosla S. Efficacy of Various Percutaneous Interventions for In-Stent Restenosis. Circ Cardiovasc Interv. 2015;8:e002778.
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
6. Rittger H, Waliszewski M, Brachmann J, et al. Long-Term Outcomes After Treatment With a Paclitaxel-Coated Balloon Versus Balloon Angioplasty. JACC Cardiovasc Interv. 2015;8:1695-700.
7. Unverdorben M, Vallbracht C, Cremers B, et al. Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis: the three-year results of the PEPCAD II ISR study. EuroIntervention. 2015;11:926-934.
8. Alfonso F, Pérez-Vizcayno MJ, Cuesta J, et al. 3-Year Clinical Follow-Up of the RIBS IV Clinical Trial. JACC Cardiovasc Interv. 2018;11:981-991.
9. Alfonso F, Pérez-Vizcayno MJ, García del Blanco B, et al. Long-Term Results of Everolimus-Eluting Stents Versus Drug-Eluting Balloons in Patients With Bare-Metal In-Stent Restenosis. JACC Cardiovasc Interv. 2016;9;1246-1255.
10. Kufner S, Cassese S, Valeskini M, et al. Long-Term Efficacy and Safety of Paclitaxel-Eluting Balloon for the Treatment of Drug-Eluting Stent Restenosis. JACC Cardiovasc Interv. 2015;8:877-884.
11. Giacoppo D, Alfonso F, Xu B, et al. Drug-Coated Balloon Angioplasty Versus Drug-Eluting Stent Implantation in Patients With Coronary Stent Restenosis. J Am Coll Cardiol. 2020;75:2664-2678.
12. Ahmed JM, Mintz GS, Castagna M, et al. Intravascular ultrasound assessment of the mechanism of lumen enlargement during cutting balloon angioplasty treatment of in-stent restenosis. Am J Cardiol. 2001;88:1032-1034.
13. Albiero R, Silber S, di Mario C, et al. Cutting balloon versus conventional balloon angioplasty for the treatment of in-stent restenosis. J Am Coll Cardiol. 2004;43:943-949.
14. Park S-J, Kim K-H, Oh I-Y, et al. Comparison of Plain Balloon and Cutting Balloon Angioplasty for the Treatment of Restenosis With Drug-Eluting Stents vs Bare Metal Stents. Circ J. 2010;74:1837-1845.
15. Byrne RA, Joner M, Alfonso F, Kastrati A. Drug-coated balloon therapy in coronary and peripheral artery disease. Nat Rev Cardiol. 2014;11:13-23.
16. Radke P, Joner M, Joost A, et al. Vascular effects of paclitaxel following drug-eluting balloon angioplasty in a porcine coronary model: the importance of excipients. EuroIntervention. 2011;7:730-737.
17. Kong J, Hou J, Ma L, et al. Cutting balloon combined with paclitaxel-eluting balloon for treatment of in-stent restenosis. ArchCardiovasc Dis. 2013;106:79-85.
18. Kufner S, Joner M, Schneider S, et al. Neointimal Modification With Scoring Balloon and Efficacy of Drug-Coated Balloon Therapy in Patients With Restenosis in Drug-Eluting Coronary Stents. JACC Cardiovasc Interv. 2017;10:1332-1340.
19. Linares Vicente JA, Ruiz Arroyo JR, Lukic A, et al. 5 year-effectiveness of paclitaxel drug-eluting balloon for coronary in-stent restenosis in a real-world registry. REC Interv Cardiol. 2019;2:92-98.
20. Mehran R, Dangas G, Abizaid AS, et al. Angiographic Patterns of In-Stent Restenosis. Circulation. 1999;100:1872-1878.
21. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials: The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
22. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction (2018). Circulation. 2018;138:e618-e651.
23. Cassese S, Xu B, Habara S, et al. Incidence and predictors of reCurrent restenosis after drug-coated balloon Angioplasty for Restenosis of a drUg-eluting Stent: The ICARUS Cooperation. Rev Esp Cardiol. 2018;71:620-627.
24. Alfonso F, Pérez-Vizcayno MJ, García del Blanco B, et al. Comparison of the Efficacy of Everolimus-Eluting Stents Versus Drug-Eluting Balloons in Patients With In-Stent Restenosis (from the RIBS IV and V Randomized Clinical Trials). Am J Cardiol. 2016;117:546-554.
25. Scheller B, Fontaine T, Mangner N, et al. A novel drug-coated scoring balloon for the treatment of coronary in-stent restenosis: Results from the multi-center randomized controlled PATENT-C first in human trial. Catheter Cardiovasc Interv. 2016;88:51-59.
26. Alfonso F, Cuesta J, García del Blanco B, et al. Scoring balloon predilation before bioresorbable vascular scaffold implantation in patients with in-stent restenosis: the RIBS VI 'scoring' study. Coron Artery Dis. 2021;32:96-104.
27. Aoki J, Nakazawa G, Ando K, et al. Effect of combination of non-slip element balloon and drug-coating balloon for in-stent restenosis lesions (ELEGANT study). J Cardiol. 2019;74:436-442.
28. Habara S, Kadota K, Shimada T, et al. Late Restenosis After Paclitaxel-Coated Balloon Angioplasty Occurs in Patients With Drug-Eluting Stent Restenosis. J Am Coll Cardiol. 2015;66:14-22.
ABSTRACT
Introduction and objectives: After the positive pre-clinical and clinical results with Angiolite, a cobalt-chromium sirolimus-eluting stent, we decided to analyze its performance in a non-selected, real-world population: the RANGO registry.
Methods: We conducted an observational, prospective, multicenter registry of patients with different clinical indications. All consecutive patients treated with percutaneous coronary intervention with, at least, 1 Angiolite stent and who gave their informed consent were included. The registry primary endpoint was the occurrence of target lesion failure (TLF) at 6, 12, and 24 months defined as cardiovascular death, myocardial infarction (MI) related to target vessel, and clinically driven target lesion revascularization. The secondary endpoints were the individual components of the primary endpoint, major adverse cardiovascular events (MACE: all-cause mortality, any MI, or any revascularization), and stent thrombosis. We describe the 2-year clinical results of the RANGO study in the entire population, in those who only received Angiolite stents, and in 2 predefined subgroups: diabetics and patients with small-vessels (≤ 2.5mm).
Results: 646 patients (426 of them only received Angiolite stents) with a high-risk profile were recruited: prevalence of previous MI (18.4%), previous coronary revascularization (23.4%), clinical presentation as ST-segment elevation MI (23.1%), and multivessel disease (47.8%). At the 2-year follow-up, the rates of TLF, MACE, and stent thrombosis were 3.4%, 9.6%, and 0.9%, respectively. Similar results were observed among patients treated with Angiolite stents only: TLF, 3.1%; MACE, 8.0%; thrombosis, 0.7%. The rates were not significantly different for the diabetic (TLF, 3.0%; MACE, 14.1%; thrombosis, 1.0%), and small-vessel subgroups (TLF, 4.3%; MACE, 12.1%; thrombosis, 0%).
Conclusions: In conclusion, the results of this observational registry on the use of Angiolite in a real-world population, including a high-risk population, corroborate the excellent results observed in previous studies, up to a 2-year follow-up. An extended 5-year follow-up is planned to discard the occurrence of late events.
Keywords: Sirolimus-eluting-stent. Durable fluoropolymer. Observational study. Efficacy. Safety. Stent thrombosis.
RESUMEN
Introducción y objetivos: Para confirmar los resultados observados en análisis preclínicos y clínicos del stent liberador de sirolimus Angiolite se diseñó el registro observacional de vida real RANGO.
Métodos: El registro prospectivo multicéntrico incluyó pacientes con distintas indicaciones clínicas que recibieron al menos 1 stent Angiolite para tratar su enfermedad coronaria y que dieron su consentimiento informado. El objetivo primario fue la incidencia de fracaso del tratamiento de la lesión (FTL) a 6, 12 y 24 meses, definido como muerte de causa cardiaca, infarto de miocardio en relación con el vaso tratado o nueva revascularización de la lesión tratada. Los objetivos secundarios fueron los componentes individuales del objetivo primario y las incidencias de eventos cardiacos mayores (MACE) y de trombosis del stent. Se presentan los resultados del registro RANGO a 2 años en la población global, en los pacientes que recibieron stent Angiolite y en 2 subgrupos predefinidos de diabéticos y vasos pequeños (≤ 2,5 mm).
Resultados: Se seleccionaron 646 pacientes (426 solo recibieron stents Angiolite) con un perfil de riesgo elevado: infarto previo (18,4%), revascularización coronaria previa (23,4%), presentación clínica como infarto agudo con elevación del segmento ST (23,1%) y enfermedad multivaso (47,8%). A los 2 años, la incidencia de FTL en el grupo global fue del 3,4%, la de MACE fue del 9,6% y la de trombosis del stent fue del 0,9%. En el grupo tratado solo con stents Angiolite, los resultados fueron similares (FTL 3,1%, MACE 8,0% y trombosis 0,7%). Los resultados no fueron significativamente diferentes en los diabéticos (FTL 3,0%, MACE 14,1% y trombosis 1,0%) y en los pacientes con vasos pequeños (FTL 4,3%, MACE 12,1% y trombosis 0%).
Conclusiones: Los resultados del registro observacional RANGO a los 2 años en población de vida real con perfil de riesgo elevado confirman los excelentes resultados del stent Angiolite observados en estudios previos. Se plantea un seguimiento clínico a 5 años para descartar eventos muy tardíos.
Palabras clave: Stent liberador de sirolimus. Fluoropolimero estable. Estudio observacional. Eficacia. Seguridad. Trombosis del stent.
Abbreviations
DES: drug-eluting stents. MACE: major adverse cardiovascular events. PCI: percutaneous coronary intervention. TLF: target lesion failure. TLR: target lesion revascularization. TVR: target vessel revascularization.
INTRODUCTION
Drug-eluting stents (DES) are one of the greatest advances in the percutaneous treatment of coronary artery disease. These devices have consistently shown lower rates of revascularization of the treated vessel in a wide range of clinical situations, and have become the treatment of choice.1 However, the risk of late and very late stent thrombosis arose with first-generation DES,2 and, to this date, it is still a matter of concern.3 This phenomenon has been associated with side effects to the drug (impairing the proliferation of new endothelial cells), the polymer, the stent platform or a combination of them on the vessel wall, leading to delayed or incomplete endothelialization, persistent inflammatory reactions, and the development of neo-atherosclerosis. New DES have been developed with superior efficacy in terms of abolishing the need for revascularization, but with the reassurance of much lower rates of stent thrombosis, the most dreadful clinical manifestation of suboptimal vessel healing. The Angiolite stent (iVascular, Spain) is a thin-strut cobalt-chromium sirolimus-eluting stent with biostable coating made of 3 layers: acrylate to ensure adhesion to the metal surface, fluoroacrylate loaded with sirolimus (1.4 µg/mm2), and a top layer of fluoroacrylate for drug release control (> 75% elution within the first month).
The Angiolite stent was initially tested in a pre-clinical model with very promising results,4 with an equivalent antiproliferative response, and a better healing pattern compared to the XIENCE stent (Abbott Vascular, United States). Subsequently, a first-in-human study5 (ANCHOR study) proved a powerful inhibition of neointimal hyperplasia as seen on the OCT: The Angiolite stent efficiently inhibited the proliferative response (vessel area stenosis, 4.4% ± 11.3%), in- stent late lumen loss at 6 months (0.07 mm ± 0.37 mm), and had a low rate of strut malapposition (1.1% ± 6.2%). Finally, the ANGIOLITE study,6 a randomized clinical trial, compared the Angiolite stent to the XIENCE stent in 223 patients (randomization with a 1:1 allocation ratio). In this study, the primary endpoint, the 6-month in-stent late lumen loss, was non-inferior in the Angiolite group (0.04 mm ± 0.39 mm) compared to the XIENCE group (0.08 mm ± 0.38 mm). The stent received the CE marking (Conformité Européenne) for its routine use. Therefore, we designed the present observational, prospective, registry to endorse the previous results in the routine clinical practice, with wider indications for use.
METHODS
Study design
The EPIC02-RANGO study was designed as a prospective, single-arm, multicenter, observational registry for the evaluation of the safety and efficacy profile of the Angiolite stent in unselected patients representative of the routine clinical practice. The study design was approved by all investigators and the sponsor as well. A reference ethics committee approved the protocol and the informed consent forms; local ethics committees were informed that this study would be conducted in their centers in compliance with the national legislation. The study was conducted and monitored by an independent contract research organization. The authors of this original manuscript independently conducted the data final analysis, interpreted the study results, and drafted/wrote this original manuscript. The sponsor was informed on the status of the study and the final results, but had no further participation.
Selection of the study population
To be enrolled in the study, subjects should met all the 3 following inclusion criteria: ≥ 18 years-old; treated with percutaneous coronary intervention (PCI) with at least 1 Angiolite stent; and have received proper information and signed the corresponding informed consent.
To guarantee a real-world population, non-stringent exclusion criteria were applied. Subjects were only excluded from the study if they met any of the following exclusion criteria: contraindication to dual antiplatelet therapy; established cardiogenic shock; unlikely to complete the scheduled follow-up; or formal refusal to participate in the study.
The PCI (predilatation, invasive imaging, postdilatation, planning, and final performance) was left at the discretion of the operator, and was indicative of the real-world use of the stents. Medical treatment during and after the procedure, including antiplatelet regime and duration, also followed the standard local practices; however, we suggested the investigators to follow the guidelines available on the management of these patients.1,7
Endpoints
The primary endpoint was target lesion failure (TLF) at 6, 12, and 24 months defined as cardiovascular death, target vessel myocardial infarction or clinically driven target lesion revascularization.
-
The secondary endpoints were:
-
– Target vessel failure defined as cardiovascular death, target vessel myocardial infarction or target vessel revascularization.
-
– Major adverse cardiovascular events (MACE) defined as all-cause mortality, any myocardial infarction or any target vessel revascularization.
-
– Stent thrombosis (definite or probable, as defined by the ARC criteria8).
In all cases, myocardial infarction refers to spontaneous infarction only. Two subgroups were predefined: patients with diabetes, and patients with Angiolite stents placed in small vessels (stent diameter ≤ 2.5 mm).
Sample size calculation
We conducted an exploratory analysis that rendered a population of 640 patients (with an estimated loss to follow-up of 10%). This sample size produces a 2-sided 95% confidence interval with a precision equal to 1.75% when the TLF rate is 4.86%. This value was obtained from the data published from different contemporary stents9-17 (table 1 of the supplementary data).
Table 1. Baseline and clinical characteristics
| Total N = 646 | Angiolite only population N = 426 | |
|---|---|---|
| Age (years old) | 66.41 ± 11.93 | 65.72 ± 11.98 |
| Male sex | 495 (76.6%) | 320 (75.1%) |
| Cardiovascular risk factors & history | ||
| Hypertension | 402 (62.2%) | 254 (59.6%) |
| Dyslipidemia | 385 (59.6%) | 251 (58.9%) |
| Diabetes mellitus* | 199 (30.8%) | 119 (27.9%) |
| Current smoker | 182 (28.2%) | 127 (29.8%) |
| Chronic kidney disease | 46 (7.1%) | 25 (5.9%) |
| Peripheral vascular disease* | 44 (6.8%) | 23 (5.4%) |
| Previous stroke | 28 (4.3%) | 17 (4.0%) |
| Previous myocardial infarction | 119 (18.4%) | 73 (17.1%) |
| Previous coronary surgery | 20 (3.1%) | 13 (3.1%) |
| Previous PCI | 131 (20.3%) | 78 (18.3%) |
| Atrial fibrillation | 34 (5.3%) | 20 (4.7%) |
| Heart failure | 46 (7.1%) | 32 (7.5%) |
| Valvular heart disease ≥ grade III | 16 (2.5%) | 7 (1.6%) |
| PCI indication | ||
| NSTEMI | 220 (34.1%) | 141 (33.1%) |
| STEMI | 149 (23.1%) | 112 (26.3%) |
| Stable angina | 120 (18.6%) | 68 (16.0%) |
| Unstable angina (negative biomarkers) | 72 (11.1%) | 51 (12.0%) |
| Silent myocardial ischemia | 32 (5.0%) | 19 (4.5%) |
| Other | 53 (8.2%) | 35 (8.2%) |
|
NSTEMI, non-ST-elevation acute myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction. * Significant differences between patients with the Angiolite stent only vs patients with any stents in addition to the Angiolite, P < .05. Data are expressed as no. (%) or mean ± standard deviation. |
||
Population analysis
The primary safety and efficacy analysis considered all patients who received the Angiolite stent only except for those who withdrew their consent. The secondary analysis was performed on all patients included in the study who received, at least, 1 Angiolite stent plus another different stent except for those who withdrew their consent.
Clinical events committee
An independent data and safety monitoring board reviewed the cumulative safety data to safeguard the well-being of the participants. All events were remotely monitored by a contract research organization. The clinical events committee reviewed, adjudicated, and classified all adverse events. The 5 members of the clinical events committee were not affiliated to the centers that participated in the study.
A total of 90 random patient audits (14% of the global population) were conducted at 4 centers, including the top 3 recruiters. The result of these audits detected 9 unreported events, most of them corresponded to scheduled procedures that required admission (non-cardiac surgeries and 2 scheduled PCI cases). None of the events associated with these audits corresponded to events classified as primary or secondary endpoints.
Descriptive statistics
All continuous variables were summarized using the following descriptive statistics: n (based on the number of recorded data values for each parameter), mean, standard deviation, 95% confidence interval for the mean, median, interquartile range [Q1, Q3], maximum, and minimum. The frequency and percentages (based on the number of recorded data values for each parameter) of the observed values are reported for all categorical measures. In general, all data are listed, and sorted by study site, and subject.
Statistical methods
Regarding the continuous variables, results were expressed as mean ± standard deviation. Variables were compared using an independent t test or the Mann-Whitney test, when applicable. Categorical variables are expressed as counts and percentages and compared using the chi-square test or Fisher’s exact test. Variables were compared between patients with only the Angiolite stent versus patients with other stents in addition to the Angiolite one. The clinical variables at 6, 12, and 24 months were expressed as counts and percentages. Time-to-event hazard curves were expressed as Kaplan-Meier estimates.
These methods were applied for the entire cohort and the 2 predefined subgroups, when appropriate: patients with diabetes, and patients with small vessel lesions (stent diameter ≤ 2.5 mm).
The statistical software SAS Version 9.4 was used for all statistical analyses, listings, tabulations, and figures.
RESULTS
A total of 654 patients were recruited from 16 academic medical centers in Spain and Portugal from June 2017 through July 2018. A total of 8 patients were excluded for not meeting the selection criteria (2 in whom the Angiolite stent was not intented to be used, 5 duplicated patients with staged, planned, procedures, and 1 patient without any data available). Therefore, the population analyzed consisted of 646 patients (figure 1); a total of 426 patients were treated with Angiolite stents only (primary analysis).
Figure 1. Flow chart of the study.
The baseline characteristics and clinical data, as well as the angiographic and procedural features are shown on table 1 and table 2, respectively. Noteworthy, the population has a high-risk profile with a remarkable prevalence of previous myocardial infarction (18.4%), previous coronary revascularization (23.4%), clinical presentation as ST-segment elevation myocardial infarction (23.1%), and multivessel disease (47.8%).
Table 2. Angiographic and procedural features
| Total N = 646 | Angiolite only population N = 426 | |
|---|---|---|
| Coronary angiography | ||
| Radial approach | 585 (90.6%) | 396 (93.0%) |
| Extension of the disease | ||
| No. of diseased vessels* | ||
| 1 | 337 (52.2%) | 289 (67.8%) |
| 2 | 198 (30.7%) | 92 (21.6%) |
| 3 | 111 (17.1%) | 45 (10.6%) |
| Left main coronary artery* | 29 (4.5%) | 12 (2.8%) |
| Proximal LAD disease | 179 (27.7%) | 110 (25.8%) |
| Diffuse disease* | 128 (19.8%) | 63 (14.8%) |
| No. of lesions per patient* | 1.98 ± 1.24 | 1.51 ± 0.90 |
| No. of treated lesions per patient* | 1.68 ± 0.95 | 1.25 ± 0.53 |
| No. of stents per patient* | 1.80 ± 1.11 | 1.24 ± 0.55 |
| Index procedure | ||
| Revascularization | ||
| Complete | 489 (75.7%) | 331 (77.7%) |
| Functional | 84 (13.0%) | 51 (12.0%) |
| Intravascular imaging | ||
| IVUS | 15 (2.3%) | 5 (1.2%) |
| OCT | 12 (1.9%) | 7 (1.6%) |
| Staged revascularization* | 85 (13.2%) | 26 (6.1%) |
|
IVUS, intravascular ultrasound; LAD, left anterior descending coronary artery; OCT, optical coherence tomography. * Significant differences between patients with the Angiolite stent only vs patients with any stents in addition to the Angiolite, P < .05. Data are expressed as no. (%) or mean ± standard deviation. |
||
The mean ± standard deviation number of lesions per patient was 1.98 ± 1.2, the mean number of treated lesions per patient was 1.68 ± 0.9 with a mean number of stents per patient of 1.80 ± 1.1. These numbers were significantly lower among patients treated with the Angiolite stent and consistent with the different patient profile. Table 3 summarizes the characteristics and treatment of each individual lesion. Interestingly, Angiolite stents were more frequently used to treat the infarct-related artery compared to other stents in our population. Subsequently, lesions with thrombus were more common in the group treated with Angiolite stents only while severe calcification was more prevalent in the entire group. Procedural complications occurred in 10 patients, 7 of them associated with Angiolite stents: 1 uncrossable lesion, 1 guidewire-related distal perforation, 1 severe no-reflow phenomenon, and 4 cases of dissection, 2 of them treated with additional stents. The procedural and device success rates were 99.7% and 99.2%, respectively. In more complex anatomic scenarios, specifically lesions with moderate/severe calcification, the procedural and device success rates stayed high (99.6% and 99.3%, respectively). Those rates were 100% in the subgroup of lesions at bifurcations or at left main coronary artery level.
Table 3. Characteristics and treatment of each individual lesion
| Total L = 1083 (84.9% of all lesions) | Angiolite only population L = 531 (82.5% of all lesions) | |
|---|---|---|
| Vessel | ||
| Left anterior descending territory | 459 (42.4%) | 236 (44.4%) |
| Right coronary territory | 327 (30.2%) | 172 (32.4%) |
| Circumflex territory | 273 (24.9%) | 112 (21.2%) |
| Left main coronary artery | 19 (1.8%) | 5 (0.9%) |
| Other | 5 (0.7%) | 6 (1.1%) |
| AHA/ACC Classification* | ||
| A | 95 (8.8%) | 68 (12.8%) |
| B1 | 355 (32.8%) | 193 (36.3%) |
| B2 | 429 (39.6%) | 185 (34.8%) |
| C | 204 (18.8%) | 85 (16.0%) |
| Lesion characteristics | ||
| Thrombus* | 145 (13.4%) | 91 (17.1%) |
| Stent at the infarct-related artery* | 366 (33.8%) | 249 (46.9%) |
| Severe calcification* | 85 (7.8%) | 22 (4.1%) |
| Restenotic lesion treated | 37 (3.4%) | 22 (4.1%) |
| Chronic total coronary occlusion | 37 (3.4%) | 20 (3.8%) |
| Lesion at bifurcation | 108 (10.0%) | 47 (8.9%) |
| Severe tortuosity | 142 (13.1%) | 62 (11.7%) |
| Vessel diameter (mm) | 2.91 ± 0.55 | 2.91 ± 0.53 |
| Lesion length (mm)* | 19.47 ± 9.80 | 17.56 ± 8.26 |
| Pre-dilatation* | 786 (72.6%) | 363 (68.4%) |
| Scoring balloon | 45 (4.2%) | 11 (2.1%) |
| Cutting balloon | 28 (2.6%) | 8 (1.5%) |
| Rotational atherectomy | 27 (2.5%) | 9 (1.7%) |
| Thrombectomy* | 75 (6.9%) | 48 (9.0%) |
| Stents implanted | S = 1160 | S = 529 |
| No. of stents per lesion | 1.07 ± 0.45 | 1.00 ± 0.35 |
| Characteristics of the stent* | ||
| Type = Angiolite stent | 784 (67.6%) | 529 (100.0%) |
| Stent diameter (mm) | 2.99 ± 0.51 | 2.99 ± 0.46 |
| Stent length (mm) | 21.38 ± 8.51 | 20.34 ± 7.03 |
| Maximum pressure (atm) | 14.61 ± 2.48 | 14.69 ± 2.46 |
| Stent crossing the lesion at the 1st attempt | 1067 (98.5%) | 527 (99.2%) |
| Lesions at bifurcation | 104 (96.3%) | 45 (95.7%) |
| Moderate or severe calcification | 268 (97.1%) | 75 (97.4%) |
| Left main coronary artery | 19 (100%) | 5 (100%) |
| Postdilatation | 284 (26.2%) | 149 (28.1%) |
| Balloon diameter (mm) | 3.24 ± 0.62 | 3.25 ± 0.53 |
| Type of balloon, non-compliant | 186 (67.4%) | 112 (76.7%) |
|
ACC, American College of Cardiology; AHA, American Heart Association; L, lesions; S, stents. * Significant differences between patients with the Angiolite stent only vs patients with any stents in addition to the Angiolite, P < .05. Data are expressed as no. (%) or mean ± standard deviation. |
||
The 6-month and 1-year follow-ups were good, with only 9 (1.4%) and 12 (1.9%) patients lost to follow-up, respectively. At the 1-year follow-up, 368 patients (59.6%) were still on dual antiplatelet therapy; this rate dropped to a 15.5% at the 2-year follow-up. During the established follow-up period (2 years for all patients), only 13 patients (2%) were lost. In the global population, at 2 years, the rates of TLF, target vessel failure, and MACE were 3.4%, 4.3%, and 9.6%, respectively. Two of the 9 cases of TLF were not associated with Angiolite stents but with other stents implanted. The rate of definite/probable stent thrombosis was 0.9%; all patients were on dual antiplatelet therapy when the event occurred. Interestingly, 4 cases appeared during the first week of follow-up, 1 case within the first month, and only 1 case of stent thrombosis after 6 months (268 days). Table 4 and figure 2 summarize the individual event rate and timing.
Table 4. Outcomes in the global population
| Total population (N = 646) | 6-month follow-up | 1-year follow-up | 2-year follow-up |
|---|---|---|---|
| Death | 11 (1.7%) | 17 (2.6%) | 31 (4.8%) |
| Cardiovascular death | 6 (0.9%) | 8 (1.2%) | 11 (1.7%) |
| Myocardial infarction | 11 (1.7%) | 16 (2.5%) | 20 (3.1%) |
| Target vessel myocardial infarction | 6 (0.9%) | 8 (1.2%) | 8 (1.2%) |
| Definite/probable device thrombosis | 5 (0.8%) | 6 (0.9%) | 6 (0.9%) |
| Revascularization | 13 (2.0%) | 22 (3.4%) | 32 (5.0%) |
| Target lesion revascularization | 6 (0.9%) | 8 (1.2%) | 9 (1.4%) |
| Target vessel revascularization | 7 (1.1%) | 11 (1.7%) | 15 (2.3%) |
| Non-target vessel revascularization | 6 (0.9%) | 11 (1.7%) | 17 (2.6%) |
| Target lesion failurea | 13 (2.0%) | 18 (2.8%) | 22 (3.4%) |
| Target vessel failureb | 14 (2.2%) | 21 (3.3%) | 28 (4.3%) |
| MACEc | 25 (3.9%) | 41 (6.3%) | 62 (9.6%) |
|
MACE, major adverse cardiovascular events. a Target lesion failure defined as cardiovascular death, target vessel myocardial infarction, and clinically indicated target lesion revascularization. b Target vessel failure defined as cardiovascular death, target vessel myocardial infarction, and target vessel revascularization. c MACE defined as all-cause mortality, any myocardial infarction, any revascularization. |
|||
Figure 2. 2-year cumulative incidence of events in the entire population (N = 646).
In the primary analysis population (patients treated with Angiolite stents only) at 2 years, the rates of TLF, target vessel failure, and MACE were 3.1%, 4.0%, and 8.0%, respectively. The rate of definite/probable stent thrombosis was 0.7%. No cases of stent thrombosis were found beyond the first 6 months. Table 5 and figure 3 summarize the individual event rate and timing.
Table 5. Outcomes in the primary analysis population: patients treated with the Angiolite stent only
| Angiolite only population (N = 426) | 6-month follow-up | 1-year follow-up | 2-year follow-up |
|---|---|---|---|
| Death | 5 (1.2%) | 10 (2.3%) | 18 (4.2%) |
| Cardiovascular death | 3 (0.7%) | 5 (1.2%) | 7 (1.6%) |
| Myocardial infarction | 5 (1.2%) | 5 (1.2%) | 10 (2.3%) |
| Target vessel myocardial infarction | 4 (0.9%) | 4 (0.9%) | 4 (0.9%) |
| Definite/probable device thrombosis | 3 (0.7%) | 3 (0.7%) | 3 (0.7%) |
| Revascularization | 7 (1.6%) | 11 (2.7%) | 18 (4.2%) |
| Target lesion revascularization | 3 (0.7%) | 4 (0.9%) | 5 (1.2%) |
| Target vessel revascularization | 4 (0.9%) | 7 (1.6%) | 9 (2.1%) |
| Non-target vessel revascularization | 3 (0.7%) | 4 (0.9%) | 9 (2.1%) |
| Target lesion failurea | 7 (1.6%) | 10 (2.3%) | 13 (3.1%) |
| Target vessel failureb | 8 (1.9%) | 13 (3.1%) | 17 (4.0%) |
| MACEc | 13 (3.2%) | 22 (5.3%) | 34 (8.0%) |
|
MACE, major adverse cardiovascular events. a Target lesion failure defined as cardiovascular death, target vessel myocardial infarction, and clinically indicated target lesion revascularization. b Target vessel failure defined as cardiovascular death, target vessel myocardial infarction, and target vessel revascularization. c MACE defined as all-cause mortality, any myocardial infarction, any revascularization. |
|||
Figure 3. 2-year cumulative incidence of events in the primary analysis population of patients treated with the Angiolite stent only (N = 426).
The subgroup analysis rendered 2-year results that were slightly worse that those observed in the global population:
-
– The diabetic subgroup showed rates of TLF, target vessel failure, and MACE of 3.0%, 4.5%, and 14.1%, respectively. The rate of stent thrombosis was 1.0%: 2 cases among 199 diabetic patients; only 1 of these cases appeared in the primary analysis of patients treated with the Angiolite stent only. Supplementary data give a description of the event rate (table 2 of the supplementary data).
-
– The patients with stents placed in small vessels (≤ 2.5 mm) showed rates of TLF, target vessel failure, and MACE of 4.3%, 6.0%, and 12.1%, respectively. No stent thrombosis was found. Supplementary data give a description of the event rate (table 3 of the supplementary data).
DISCUSSION
The results of the current real-world registry of the Angiolite coronary stent show an outstanding safety and efficacy profile as the ANCHOR5–first-in-human study–and the ANGIOLITE6 randomized clinical trial comparison with the XIENCE stent showed. The clinical profile shows a relatively high-risk population with a prevalence of diabetes mellitus of 30.8%, 17.6% on anticoagulation with oral drugs, 18.4% of patients diagnosed with previous myocardial infarction, and 23.4% with previous coronary revascularization. Also, a high rate of complex coronary artery disease was found in the recruited population: significant multivessel disease was diagnosed in 47.8%, compromised left main coronary artery in 4.5%, and diffuse coronary artery disease in 19.8% of the patients. Therefore, the mean number of significant lesions (1.98 ± 1.24), treated lesions (1.68 ± 0.95), and stents implanted per patient (1.8 ± 1.11) was relatively high. The ST-segment elevation myocardial infarction clinical setting of the PCI in around a quarter of the cases also shows the all-comer, real-world nature of the study.
The registry was designed to include all the patients in whom an Angiolite stent was intended to be used. Therefore, we may distinguish 2 different populations: those in whom ONLY the Angiolite stent was intended (primary analysis) and those who received different stents to treat other lesions on top of the Angiolite stent (secondary analysis). These populations have some significant differences: Angiolite ONLY-patients were more prone to have single vessel disease, few significant lesions, few treated lesions, and few stents implanted. Reasonably, this population with lower atherosclerotic burden showed less diffuse disease and fewer staged procedures. However, not all the characteristics of this group were so favorable since the presence of thrombus and the target lesion as the infarct-related artery were more common in the Angiolite ONLY stent group.
The primary endpoint, TLF at 1-year was consistently low both in the Angiolite ONLY population (primary analysis), 2.3%, and in the entire population (secondary analysis), 2.8%. Target vessel failure, a wider safety variable, was also noticeably low (3.1% and 3.3%, respectively). To confirm these results, MACE (including all-cause mortality too), a clinically oriented variable, was also very low (5.3% and 6.3%, respectively). An overview of the TLF results of different stents tested in registries and RCTs is shown on table 1 of the supplementary data. In these studies, the TLF mean value at 1-year is 5.4%, higher that the rate seen in this study.
The 2-year follow-up confirmed the very low rate of unfavorable cardiac events seen at the early 1-year period. The rate of new cardiac events, both device- and patient-oriented, within the second follow-up year was about half of the observed rate during the first year.
Both the ANCHOR FIH5 and the ANGIOLITE RCT6 pointed out an extraordinary antiproliferative efficacy of the Angiolite stent, with a mean late luminal loss < 0.05 mm. Consequently, we thought it was mandatory to assess the safety of this stent through the rate of stent thrombosis. The real-world use of the Angiolite stent is associated with a low rate of such a catastrophic complication (0.7% in primary analysis, 0.9% in secondary analysis), which guarantees the safe use of this powerful DES. The studies published showed a mean rate of stent thrombosis from 0.4% to 4.9% at the 2-year follow up (table 1 of the supplementary data). Also, the very low rate of definite/probable stent thrombosis beyond the first week (only 2 cases, 1 within the first month and the other 268 days later) restates this safety profile. We should mention that the use of dual antiplatelet therapy was high in this population (59.6% at the 1-year follow-up), which is indicative of the prevalence of acute coronary syndrome as the patients’ clinical presentation (68.3% of the patients).
The predefined subgroup analysis rendered interesting results. Diabetic patients showed TLF and stent thrombosis rates at 2 years, similar to the overall rate (3.0% vs 3.4%, and 1.0% vs 0.9%, respectively), while the rate of MACE was higher (14.1% vs 9.6%). This finding may show the worse clinical prognosis of diabetic patients, not necessarily associated with the lesion treated but with the remaining coronary artery disease. Our results are consistent with previous data published on the EVOLVE II substudy on diabetes13 that showed a 2-year TLF rate of 11.2% and a definite/probable stent thrombosis of 1.1%.
As expected, the subgroup of small vessel disease (≤ 2.5 mm) showed slightly higher rates of 2-year TLF and MACE (4.3% and 12.1%, respectively) than the global population (3.4% and 9.6%, respectively). The lack of definite/probable stent thrombosis cases could be indicative of detection bias as the thrombosis of these vessels may have a milder clinical expression. The results of this subgroup are usually hard to compare with other data as the definition of small vessel is highly arbitrary, from 2.25 mm to 3.0 mm. However, the results of our study are consistent with those reported in the Basket Small18 trial.
Limitations
The limitations of this study are the well-known issues of real-world observational registries: potential selection bias, reporting biases, and losses to follow-up (not in this case though, with a 98% of the follow-up period completed). However, the results are similar to previously reported data and are consistent with the results of previous studies with this stent. In the global population (patients who received other stents besides Angiolite stents), endpoints like probable stent thrombosis or cardiovascular death cannot be clearly attributed to a certain stent.
To minimize potential errors and reinforce the safety message, the steering committee has decided to extend the follow-up period up to 5 years.
CONCLUSIONS
In conclusion, the results of this observational registry on the use of the Angiolite DES in a real-world population confirm the excellent efficacy and safety profile seen in previous studies at the 2-year follow-up. An extended 5-year follow-up is planned to discard late events.
FUNDING
This study was conducted with financial support from Cardiva S.L. Data management and analysis were performed by an independent CRO. The final draft and the manuscript were wrote by investigators without any participation from the sponsors.
AUTHORS’ CONTRIBUTIONS
Idea and design: A. Pérez de Prado, F. Lozano Ruiz-Poveda, J. Moreu Burgos, B. García del Blanco, E. Pinar, V. Peral, J.R. Rumoroso, and R. Trillo Nouche. Data acquisition: A. Pérez de Prado, R. Ocaranza-Sánchez, F. Lozano Ruiz-Poveda, J. Moreu Burgos, R. Álvarez Ramos, A. Rodrigues, L. Fernández González, P. Aguar, B. García del Blanco, E. Pinar, V. Peral, F. Sainz Laso, J.R. Rumoroso, A. Torres, M. Sabaté, and R. Trillo Nouche. Statistical analysis and manuscript writing: A. Pérez de Prado, F. Lozano Ruiz-Poveda, J.R. Rumoroso, and R. Trillo Nouche. Provision of critical feedback to the manuscript and final content approval: A. Pérez de Prado, R. Ocaranza-Sánchez, F. Lozano Ruiz-Poveda, J. Moreu Burgos, R. Álvarez Ramos, A. Rodrigues, L. Fernández González, P. Aguar, B. García del Blanco, E. Pinar, V. Peral, F. Sainz Laso, J.R. Rumoroso, A. Torres, M. Sabaté, and R. Trillo Nouche.
CONFLICTS OF INTEREST
A. Pérez de Prado, and M. Sabaté received consulting honoraria and research grants from iVascular, and Cardiva S.L. F. Lozano Ruiz-Poveda received honoraria for his lectures from Abbott and Medtronic. All authors have declared payments to their centers from Cardiva S.L.
WHAT IS KNOWN ABOUT THE TOPIC?
- Current DES offer superior efficacy in terms of reducing restenosis with very low rates of stent thrombosis. The Angiolite stent (iVascular, Barcelona, Spain) is a thin-strut cobalt-chromium sirolimus-eluting stent with biostable coating of thrombus-resistant fluoroacrylate loaded with sirolimus. This stent has been comprehensively tested in preclinical studies, in a first-in-human study (ANCHOR study), and in a randomized clinical trial (compared to a cobalt-chromium everolimus- eluting stent) with consistent positive results. We designed an observational, prospective, and registry to endorse the previous results in our daily routine practice.
WHAT DOES THIS STUDY ADD?
- The results of this observational registry on the use of the Angiolite stent in a real-world, high-risk population confirm the excellent results seen in previous studies at the 2-year follow-up. Both the rate of device-related outcomes (target lesion and vessel failure) and patient-related outcomes (MACE) were lower compared to former data.
SUPPLEMENTARY DATA
REFERENCES
1. Neumann F-J, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. European Heart Journal. 2018;40:87-165.
2. Lüscher TF, Steffel J, Eberli FR, Joner M, Nakazawa G, Tanner FC, et al. Drug-eluting stent and coronary thrombosis:biological mechanisms and clinical implications. Circulation. 2007;115:1051-8.
3. Gori T, Polimeni A, Indolfi C, Räber L, Adriaenssens T, Münzel T. Predictors of stent thrombosis and their implications for clinical practice. Nature Reviews Cardiology. 2019;16:243-256.
4. Estevez-Loureiro R, Perez de Prado A, Perez-Martinez C, Cuellas-Ramon C, Regueiro-Purrinos M, Gonzalo-Orden JM, et al. Safety and Efficacy of New Sirolimus-eluting stent Models in a Preclinical Study. Rev Esp Cardiol (Engl Ed). 2015;68:1118-24.
5. Puri R, Otaegui I, Sabate M, Serra-Penaranda A, Puigfel M, Perez de Prado A, et al. Three- and 6-month optical coherence tomographic surveillance following percutaneous coronary intervention with the Angiolite(R) drug-eluting stent :The ANCHOR study. Catheter Cardiovasc Interv. 2018;91:435-443.
6. Moreu J, Moreno-Gomez R, Perez de Prado A, Garcia Del Blanco B, Trillo R, Pinar E, et al. First-in-man randomised comparison of the Angiolite durable fluoroacrylate polymer-based sirolimus-eluting stent versus a durable fluoropolymer-based everolimus-eluting stent in patients with coronary artery disease:the ANGIOLITE trial. EuroIntervention. 2019;15:e1081-e1089.
7. Valgimigli M, Bueno H, Byrne RA, Collet J-P, Costa F, Jeppsson A, et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS:The Task Force for dual antiplatelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). European Heart Journal. 2017;39:213-260.
8. Garcia-Garcia HM, McFadden EP, Farb A, Mehran R, Stone GW, Spertus J, et al. Standardized End Point Definitions for Coronary Intervention Trials:The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
9. Stone GW, Rizvi A, Sudhir K, Newman W, Applegate RJ, Cannon LA, et al. Randomized comparison of everolimus- and paclitaxel-eluting stents. 2-year follow-up from the SPIRIT (Clinical Evaluation of the XIENCE V Everolimus Eluting CoronarystentSystem) IV trial. J Am Coll Cardiol. 2011;58:19-25.
10. Waltenberger J, Hoffmann S, Brachmann J, Van Der Heijden J, Richardt G, Froebert O, et al. Bioflow-III:one year target lesion failure data of an all-comers registry with a drug eluting stent. European Heart Journal. 2013;34:P3036-P3036.
11. Pilgrim T, Heg D, Roffi M, Tüller D, Muller O, Vuilliomenet A, et al. Ultrathin strut biodegradable polymer sirolimus-eluting stent versus durable polymer everolimus-eluting stent for percutaneous coronary revascularisation (BIOSCIENCE):a randomised, single-blind, non-inferiority trial. Lancet. 2014;384:2111-22.
12. Kereiakes DJ, Meredith IT, Windecker S, Lee Jobe R, Mehta SR, Sarembock IJ, et al. Efficacy and safety of a novel bioabsorbable polymer-coated, everolimus-eluting coronary stent :the EVOLVE II Randomized Trial. Circ Cardiovasc Interv. 2015;8.
13. Kereiakes DJ, Meredith IT, Masotti M, Carrie D, Moreno R, Erglis A, et al. Safety and efficacy of a bioabsorbable polymer-coated, everolimus-eluting coronary stent in patients with diabetes:the EVOLVE II diabetes substudy. EuroIntervention. 2017;12:1987-1994.
14. Kim CH, Lee E, Kang J, Han J-K, Yang H-M, Park KW, et al. TCT-754 One-year clinical outcome of patients treated with Resolute Onyx versus Resolute Integrity:A Comparison of the HOST-ONYX and HOST-RESOLINTE Registries. Journal of the American College of Cardiology. 2017;70:B319-B319.
15. Tam CC, Chan K, Lam S, Yung A, Lam YM, Chan C, et al. One-year clinical outcomes of patients implanted with a Resolute Onyx zotarolimus-eluting stent. J Int Med Res. 2018;46:457-463.
16. von Birgelen C, Zocca P, Buiten RA, Jessurun GAJ, Schotborgh CE, Roguin A, et al. Thin composite wire strut, durable polymer-coated (Resolute Onyx) versus ultrathin cobalt–chromium strut, bioresorbable polymer-coated (Orsiro) drug-eluting stents in allcomers with coronary artery disease (BIONYX):an international, single-blind, randomised non-inferiority trial. The Lancet. 2018;392:1235-1245.
17. Wijns W, Valdes-Chavarri M, Richardt G, Moreno R, Iniguez-Romo A, Barbato E, et al. Long-term clinical outcomes after bioresorbable and permanent polymer drug-eluting stent implantation:final five-year results of the CENTURY II randomised clinical trial. EuroIntervention. 2018;14:e343-e351.
18. Jeger RV, Farah A, Ohlow M-A, Mangner N, Möbius-Winkler S, Leibundgut G, et al. Drug-coated balloons for small coronary artery disease (BASKET-SMALL 2):an open-label randomised non-inferiority trial. The Lancet. 2018;392:849-856.
- Long-term (> 12 months) single-center registry of Magmaris implantation in the acute coronary syndrome setting
- Long-term results of a primary angioplasty program in patients over 80 years of age
- Usefulness of physiological coronary assessment with iFR in daily practice and all-comer patients: immediate and follow-up results
- Predictors of postprocedural fractional flow reserve: insights from the FFR-SEARCH study
Interviews
An interview with Bruno Scheller
aServicio de Cardiología, Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria de La Princesa (IIS-IP), Universidad Autónoma de Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain