Article
Ischemic heart disease and acute cardiac care
REC Interv Cardiol. 2019;1:21-25
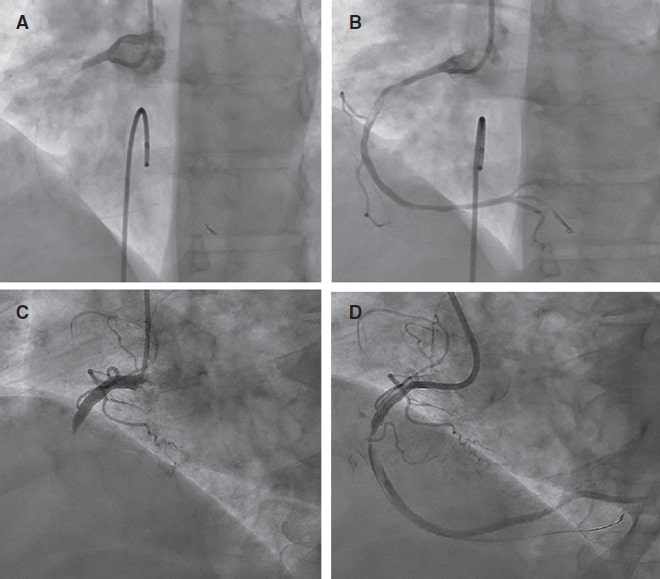
Access to side branches with a sharply angulated origin: usefulness of a specific wire for chronic occlusions
Acceso a ramas laterales con origen muy angulado: utilidad de una guía específica de oclusión crónica
Servicio de Cardiología, Hospital de Cabueñes, Gijón, Asturias, España

ABSTRACT
Introduction and objectives: The use of coronary physiology is essential to guide revascularization in patients with stable coronary artery disease. However, some patients without significant angiographic coronary artery disease will experience cardiovascular events at the follow-up. This study aims to determine the prognostic value of the global plaque volume (GPV) in patients with stable coronary artery disease without functionally significant lesions at a 5-year follow-up.
Methods: We conducted a multicenter, observational, and retrospective cohort study with a 5-year follow-up. A total of 277 patients without significant coronary artery disease treated with coronary angiography in 2015 due to suspected stable coronary artery disease were included in the study. The 3 coronary territories were assessed using quantitative flow ratio, calculating the GPV by determining the difference between the luminal volume and the vessel theoretical reference volume.
Results: The mean GPV was 170.5 mm3. A total of 116 patients (42.7%) experienced major adverse cardiovascular events (MACE) at the follow-up, including cardiac death (11%), myocardial infarction (2.6%), and unexpected hospital admissions (38.1%). Patients with MACE had a significantly higher GPV (231.6 mm3 vs 111.8 mm3; P < .001). The optimal GPV cut-off point for predicting events was 44 mm3. Furthermore, in the multivariate analysis conducted, plaque volume, diabetes, hypertension, age, dyslipidemia, smoking, age, and GPV > 44 mm3 turned out to be independent predictors of MACE.
Conclusions: GPV, calculated from the three-dimensional reconstruction of the coronary tree, is an independent predictor of events in patients with stable coronary artery disease without significant lesions. A GPV > 44 mm3 is an optimal cut-off point for predicting events.
Keywords: Coronary artery disease. Coronary atherosclerosis. Coronary angiography. Global plaque volume. Coronary physiology. Quantitative flow ratio.
RESUMEN
Introducción y objetivos: La fisiología coronaria es fundamental para guiar la revascularización en los pacientes con enfermedad coronaria estable. Sin embargo, algunos pacientes sin enfermedad coronaria significativa en la angiografía presentarán eventos cardiovasculares posteriormente. Este estudio pretende determinar el valor pronóstico del volumen global de placa (VGP) en pacientes con enfermedad coronaria estable sin lesiones funcionalmente significativas durante 5 años de seguimiento.
Métodos: Se realizó un estudio observacional multicéntrico de cohortes retrospectivo con seguimiento a 5 años, que incluyó 277 pacientes sin enfermedad coronaria significativa intervenidos mediante coronariografía en 2015 por sospecha de enfermedad coronaria estable. Se evaluaron los 3 territorios coronarios mediante el cociente de flujo cuantitativo, calculando el VGP como la diferencia entre el volumen luminal y el volumen teórico de referencia del vaso.
Resultados: El VGP medio fue de 170,5 mm3. Durante el seguimiento, 116 pacientes (42,7%) presentaron eventos cardiovasculares mayores (MACE), que incluyeron muerte de causa cardiaca (11%), infarto de miocardio (2,6%) y hospitalizaciones no programadas (38,1%). Los pacientes con MACE tenían un VGP significativamente mayor (231,6 frente a 111,8 mm3, p < 0,001). El punto de corte óptimo del VGP para predecir eventos fue de 44 mm3. En el análisis multivariado, que consideró volumen de placa, diabetes, hipertensión, edad, dislipemia y tabaquismo, la edad y un VGP > 44 mm3 fueron predictores independientes de MACE.
Conclusiones: El VGP calculado mediante reconstrucción tridimensional del árbol coronario es un predictor independiente de eventos en pacientes con enfermedad coronaria estable sin lesiones significativas. Un VGP > 44 mm3 es el punto de corte óptimo para predecir eventos.
Palabras clave: Enfermedad coronaria. Ateroesclerosis coronaria. Angiografía coronaria. Volumen global de placa. Fisiología coronaria. Cociente de flujo cuantitativo.
Abbreviations
GPV: global plaque volume. MACE: major adverse cardiovascular events. QFR: quantitative flow ratio. ROC: receiver operating characteristic curve.
INTRODUCTION
Coronary artery disease is the leading cause of mortality worldwide.1 Despite the safety involved in deferring invasive treatment in patients with stable coronary artery disease without functionally significant lesions,2 a percentage of patients experience cardiovascular events at the long-term follow-up.3 It has been reported that cardiovascular events not only depend on the degree of coronary obstruction assessed by intracoronary physiology4-5 but also on the global atherosclerotic burden and its vulnerability assessed by intracoronary imaging modalities.6-8
The new era of coronary physiology is based on predicting fractional flow reserve by reconstructing the coronary tree using angiography and computational fluid dynamics.9-10 Estimating quantitative flow ratio (QFR) is the most validated method of the ones currently available.
QFR—which predicts fractional flow reserve10-11—has proven to be a better tool than angiography alone to guide the need for lesion revascularization12 and shown long-term prognostic value13. Furthermore, it provides quantitative information out of the 3D reconstruction of the coronary tree, including minimum diameter and area, reference diameters, luminal volume, and atherosclerotic plaque volume in the studied vessel. However, the prognostic value of this quantitative analysis has not been sufficiently studied.
The main aim of this study was to determine the prognostic value of global plaque volume (GPV) in patients with stable coronary artery disease without functionally significant lesions at a 5-year follow-up.
METHODS
We conducted a retrospective observational study on a cohort of patients from 6 tertiary referral centers.
Study population
Patients who underwent coronary angiography from January through December 2015 for suspected stable coronary artery disease were included. Each participant center retrospectively enrolled all patients who underwent coronary angiography for suspected stable coronary artery disease and met the inclusion criteria. Patients with chronic total coronary occlusions, prior coronary artery bypass graft surgery, or inadequate angiographic quality for analysis were excluded. Additionally, patients whose angiographic analysis revealed a positive QFR study (< 0.80) in any coronary territory were excluded. The principal investigator conducted a retrospective follow-up at each center within the next 5 years following the index procedure. Baseline and procedural characteristics, and events at the follow-up were collected by local investigators. The study fully complied the good clinical practice principles and regulations set forth in the Declaration of Helsinki for research with human subjects. The study protocol was approved by the ethics committee of the reference hospital (Hospital Clínico Universitario de Valladolid) and the institutional review boards, including informed consent obtained from participants or, alternatively, approval for retrospective data analysis under ethical committee supervision.
Angiographic analysis
A blinded angiographic analysis of diagnostic coronary angiograms was performed by trained analysts at a centralized imaging unit (Icicorelab, Valladolid) using specialized software (QAngio XA 3D QFR, Medis Medical Imaging System, The Netherlands). A 3D reconstruction of the 3 major coronary vessels was performed using 2 different projections with > 25° of separation. For the right and left circumflex coronary arteries, the proximal marker was manually placed at the vessel ostium, while for the left anterior descending coronary artery, it was placed at the left main coronary artery ostium. The distal marker was placed at the end of the coronary artery. Plaque volume was estimated by calculating the difference between the theoretical reference vessel volume in the absence of atherosclerotic disease and the estimated vessel volume in angiography using QFR software via quantitative analysis. Reference diameters, minimum diameter, and minimum area were obtained for each vessel. Considering contrast flow through the coronary tree, QFR was calculated according to FAVOR II standards for the physiological significance of coronary lesions. Patients with functionally significant disease (QFR < 0.80) were excluded.
Statistical analysis
Categorical variables are expressed as totals and percentages, and continuous ones as means and standard deviations. GPV was estimated as the sum of plaque volume across 3 coronary territories.
The primary endpoint—major adverse cardiovascular events (MACE)—was a composite of cardiac death, acute myocardial infarction, or all-cause unplanned hospital admission.
An optimal GPV cutoff as a predictor of MACE was determined using the receiver operating characteristic (ROC) curve as the value with the maximum Youden index. Multivariate logistic regression models were used to calculate the odds ratio and 95% confidence interval as independent predictors for MACE. Variables with P < .20 in the univariate analysis were included in the multivariate model as covariates.
Event-free survival was compared using Kaplan-Meier and Mantel-Haenszel analyses. All probability values were two-tailed, and P < .05 was considered statistically significant. Statistical analysis was performed using Stata (16.1, StataCorp, College Station, United States).
RESULTS
Descriptive population analysis
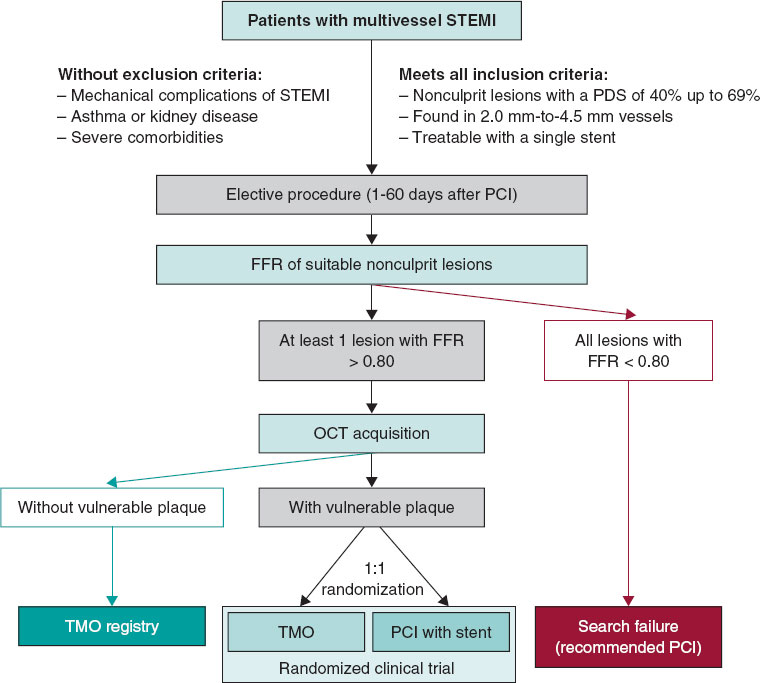
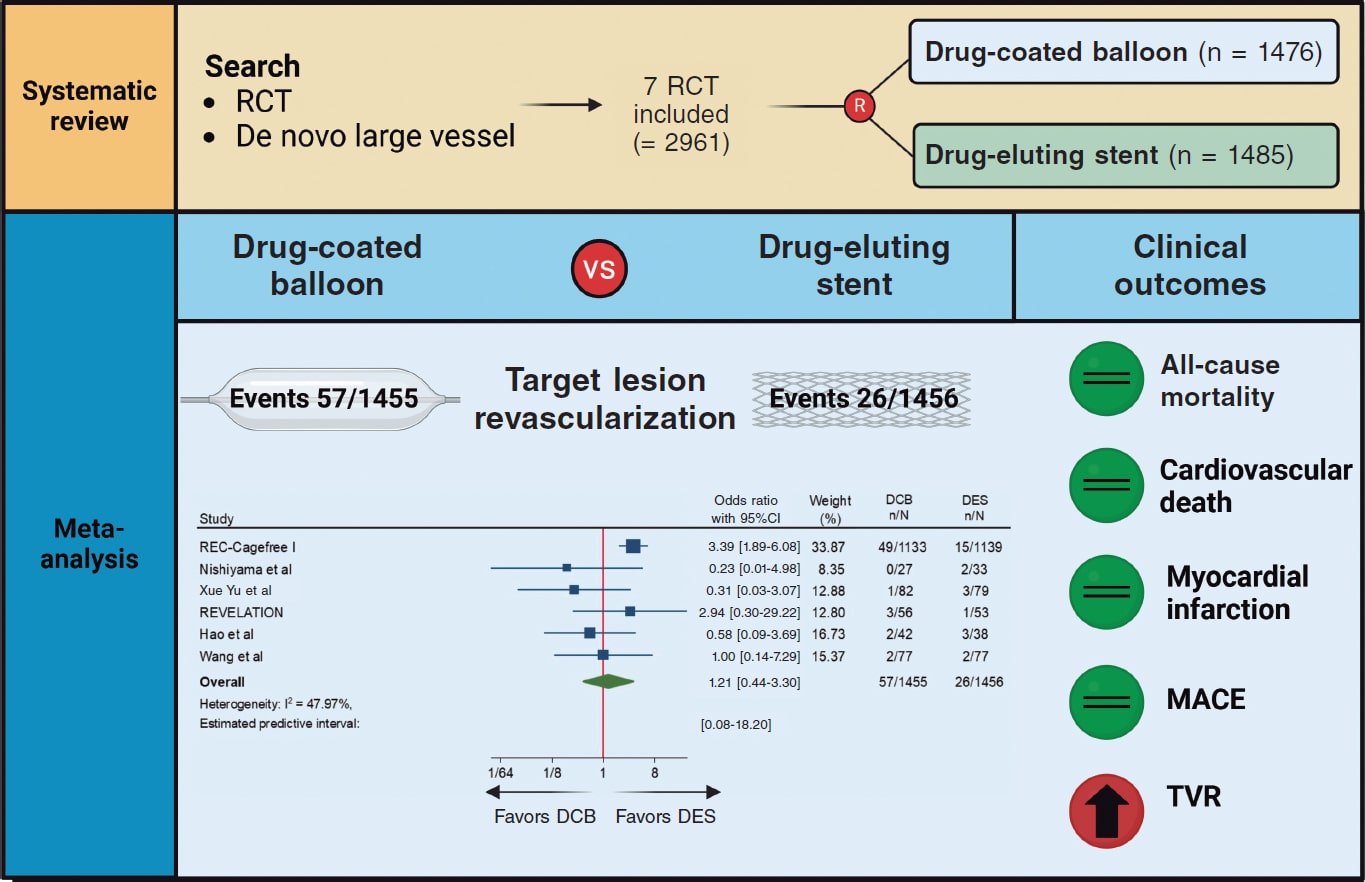
A total of 803 patients were evaluated for inclusion in the registry, 122 of whom (15.2%) were excluded due to chronic occlusions in ≥ 1 coronary territory, 17 (2.12%) due to previous surgical myocardial revascularization, and 159 (19.2%) due to inadequate angiographic analysis in, at least, 1 coronary territory. Among the remaining patients, 228 (45.1%) had significant coronary artery disease (QFR < 0.80) in, at least, 1 coronary territory, which left a final cohort of 277 patients. Patient flowchart is shown in figure 1.

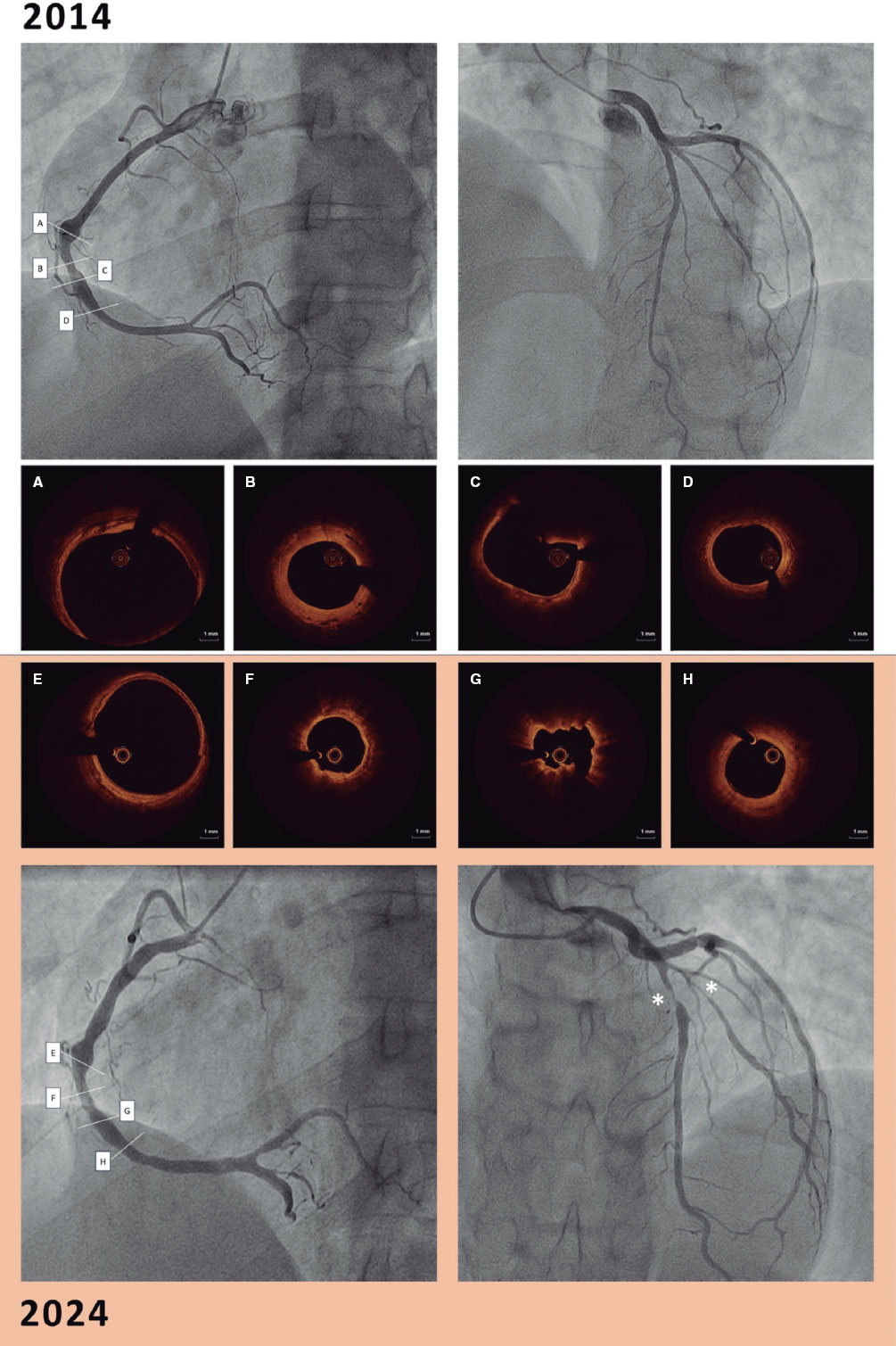
Figure 1. Flowchart of the patient selection process for inclusion in the study. CABG, coronary artery bypass graft; CTO, chronic total coronary occlusion; QFR, quantitative flow ratio.
The mean age of the population was 65.8 years (most were hypertensive [74.4%] men [66.1%]). Table 1 illustrates the baseline characteristics of the population. The median follow-up was 69 months, during which time 5 patients were lost to follow-up.
Table 1. Baseline characteristics of the included population
| Variable | n/mean | Proportion/SD |
|---|---|---|
| Female Sex | 94 | 33.9% |
| Hypertension | 206 | 74.3% |
| Diabetes mellitus | 106 | 38.2% |
| Dyslipidemia | 188 | 67.9% |
| Smoking | 121 | 43.7% |
| Chronic kidney disease | 21 | 7.6% |
| Peripheral arterial disease | 14 | 5.1% |
| Previous ischemic heart disease | 105 | 37.9% |
| Age (years) | 65.8 | 12.2 |
| Weight (kg) | 78.0 | 15.0 |
| Height (cm) | 156.2 | 36.8 |
| Left ventricular ejection fraction (%) | 57.4 | 9.3 |
|
SD, standard deviation. |
||
Angiographic analysis
Mean plaque volume in the study population was 170.5 mm3 (± 16.5); mean QFR was 0.95. Table 2 illustrates the overall means from the angiographic analysis according to the coronary territory studied. Plaque volume was independently analyzed for each coronary territory and was significantly higher in the right (243 mm3) vs the left anterior descending (161.4 mm3) and left circumflex coronary arteries (172.9 mm3). Data on this analysis by coronary territories are shown in table 1 and figure 1 of the supplementary data.
Table 2. Characteristics of the angiographic analysis performed in the 3 coronary territories using quantitative flow ratio
| Variable | Mean | SD | 95%CI |
|---|---|---|---|
| QFR | 0.95 | 0.37 | 0.95-0.96 |
| Length | 76.99 | 13.21 | 75.22-78.77 |
| Proximal diameter | 3.18 | 0.47 | 3.11-3.24 |
| Distal diameter | 1.99 | 0.34 | 1.95-2.04 |
| Reference diameter | 2.69 | 0.42 | 2.58-2.70 |
| Minimum lumen diameter | 1.76 | 0.34 | 1.72-1.81 |
| Percent diameter stenosis | 33.81 | 6.44 | 32.95-34.68 |
| Stenosis area (%) | 38.72 | 9.59 | 37.43-40.01 |
| Minimum lumen area | 3.53 | 1.30 | 3.35-3.70 |
| Lumen volume | 295.5 | 242.25 | 262.83-328.12 |
| Plaque volume | 170.54 | 240.24 | 138.17-202.91 |
|
SD, standard deviation; 95%CI, 95% confidence interval; QFR, quantitative flow ratio. |
|||
Prognostic value of global plaque volume
The primary event (MACE) occurred in 116 patients, which amounts to 42.7% of the cohort at the follow-up. Among these patients, 11% died, 2.6% suffered an acute myocardial infarction, and 38.1% required unplanned hospitalization. Patients who developed MACE had a significantly higher GPV (231.6 vs 111.8 mm3; P < .001), as well as those with a higher mortality rate (255.2 mm3 vs 154.3 mm3; P = .04) or unplanned hospitalizations (235.0 mm3 vs 125.4 mm3; P < .001). However, there were no significant differences in patients who experienced acute myocardial infarction (235.1 mm3 vs 169.3 mm3; P = .51).
The optimal GPV cutoff to predict events was set at 44 mm3 based on ROC curve analysis (sensitivity, 64%; specificity, 65.8%; LR+, 1.9; LR–, 0.6).
Table 3 illustrates the study of the main determinants of the primary event. Variables with a significance level of P < .10 were included in the multivariate analysis. In the final model, age and GPV were independent predictors. A GPV > 44 mm3 was associated with a 2.8-fold higher risk of events at the follow-up (figure 2).
Table 3. Uni- and multivariate analysis of determinants of the main event
| Determinants of the main event | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|
| OR | 95%CI | OR | 95%CI | |
| Sex, female | 1.09 | 0.66-1.81 | ||
| Age* | 1.03 | 1.01-1.10 | 1.03 | 1.00-1.07 |
| Hypertension* | 2.26 | 1.26-4.07 | 1.70 | 0.82-3.53 |
| Diabetes mellitus | 1.18 | 0.72-1.93 | ||
| Dyslipidemia | 1.04 | 0.62-1.73 | ||
| Smoking | 1.01 | 0.72-1.42 | ||
| Chronic kidney disease | 1.00 | 0.41-2.46 | ||
| Peripheral arterial disease | 1.37 | 0.47-4.01 | ||
| Previous ischemic heart disease* | 1.52 | 0.93-2.50 | 1.46 | 0.80-2.68 |
| LVEF | 0.98 | 0.96-1.01 | ||
| GPV (> 44 mm3)* | 1.93 | 1.17-3.18 | 2.80 | 1.51-5.21 |
| Reference vessel diameter* | 2.20 | 1.12-4.35 | 1.62 | 0.75-3.50 |
|
* P values < .10 were included in the multivariate analysis. 95%CI, 95% confidence interval; GPV, global plaque volume; LVEF, left ventricular ejection fraction; OR, odds ratio. |
||||

Figure 2. Kaplan-Meier curve showing the patients’ event-free survival based on their global plaque volume.
DISCUSSION
The main finding of this study is that GPV quantification emerged as an independent prognostic factor in patients without functionally significant coronary artery disease, which demonstrated that those with a higher GPV experienced more events at the follow-up. The optimal GPV cutoff for event prediction was set at 44 mm3. This study emphasizes the importance of anatomically characterizing coronary arteries without significant lesions.
Despite the absence of significant coronary artery obstructions, some patients still experience events during follow-up.14 In patients with a negative QFR functional study, it has been reported that the 5-year rate of events—cardiac death, target vessel myocardial infarction—is 11.6%,3 similar to our findings, where mortality rate was 11% and acute myocardial infarction occurred in 2.6% of patients. Determining the difference between the actual vessel diameter and the estimated diameter obtained through 3D reconstruction from QFR-based angiography has been used in other studies.15 This estimation—previously derived from coronary computed tomography16-17—has demonstrated the prognostic significance of plaque volume differences between normal and non-obstructive coronary arteries. These differences have also been confirmed using invasive imaging modalities such as intravascular ultrasound.18 Although angiography-derived percent luminal stenosis shows poor concordance with myocardial ischemia,19 a greater degree of coronary stenosis (percent diameter stenosis > 50%) is associated with a higher event rate at the 2-year follow-up in patients without functionally significant coronary lesions.20 The present study takes a step further into the minimally invasive characterization of atherosclerotic burden using easy-to-implement 3D coronary tree reconstruction technology as an independent prognostic factor in patients without functionally significant coronary lesions. In this regard, this study is consistent with recent studies which demonstrated that subclinical atherosclerosis burden—measured by vascular ultrasound for carotid plaque quantification and computed tomography for coronary calcium scoring—in asymptomatic individuals is independently associated with all-cause mortality.21
Based on these findings, GPV measurement enables the identification of patients who, despite having no significant coronary lesions, are at risk of developing events within the next 5 years, allowing for intensified treatment and cardiovascular risk factor control. However, this study has limitations, including its retrospective design for patient inclusion and recruitment, the use of indirect methods—such as QFR—to estimate plaque volume, and the inability of this method to describe plaque characteristics, or potential lipid plaque vulnerability. Of note, the estimated plaque volume in each coronary artery was not specifically correlated with events in that territory but rather with overall adverse cardiovascular events. Therefore, further studies are needed to confirm or refute this hypothesis.
CONCLUSIONS
Plaque volume, calculated by 3D coronary tree reconstruction, is an independent predictor of events in patients with suspected stable ischemic heart disease without significant coronary artery disease. The optimal GPV cutoff for event prediction is 44 mm3.
FUNDING
C. Cortés received funding through the Río Hortega contract CM22/00168 and Miguel Servet CP24/00128 from Instituto de Salud Carlos III (Madrid, Spain).
ETHICAL CONSIDERATIONS
The present study was conducted in full compliance with clinical practice guidelines set forth in the Declaration of Helsinki for clinical research and was approved by the ethics committees of the reference hospital (Hospital Clínico Universitario de Valladolid) and other participant centers. Possible sex- and gender-related biases were also considered.
DECLARATION ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the writing of this text.
AUTHORS’ CONTRIBUTIONS
C. Cortés and J. Ruiz-Ruiz participated in study design, data analysis, manuscript drafting, and critical review. C. Fernández and M. García participated in data collection and result analysis. F. Rivero and R. López-Palop assisted in data collection. S. Blasco and A. Freites contributed to statistical analysis. L. Scorpiglione and M. Rosario Ortas Nadal collaborated in data interpretation. O. Jiménez participated in manuscript preparation and initial review. J.A. San Román Calvar and I.J. Amat-Santos conducted the final review and approved the version for publication.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- Global plaque volume has already been identified as an independent risk factor for the occurrence of new coronary events at the follow-up of patients without significant coronary lesions. However, this risk was determined using coronary computed tomography and imaging modalities such as intravascular ultrasound.
WHAT DOES THIS STUDY ADD?
- This article is the first study to only use the patient’s own angiography and minimally invasive coronary physiology techniques, such as quantitative flow ratio to determine plaque volume and its relationship with major cardiovascular events at a 5-year follow-up in patients without significant coronary artery disease. This approach simplifies the implementation of this technique and enhances prevention strategies for patients at higher risk of cardiovascular events.
REFERENCES
1. Laslett LJ, Alagona PJ, Clark BA 3rd, et al. The worldwide environment of cardiovascular disease:prevalence, diagnosis, therapy, and policy issues:a report from the American College of Cardiology. J Am Coll Cardiol. 2012;60:S1-49.
2. Zimmermann FM, Ferrara A, Johnson NP, et al. Deferral vs. of percutaneous coronary intervention of functionally non-significant coronary stenosis:15-year follow-up of the DEFER trial. Eur Heart J. 2015;36:3182-3188.
3. Kuramitsu S, Matsuo H, Shinozaki T, et al. Five-Year Outcomes After Fractional Flow Reserve-Based Deferral of Revascularization in Chronic Coronary Syndrome:Final Results From the J-CONFIRM Registry. Circ Cardiovasc Interv. 2022;15:E011387.
4. De Bruyne B, Pijls NHJ, Kalesan B, et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012;367:991-1001.
5. Ciccarelli G, Barbato E, Toth GG, et al. Angiography versus hemodynamics to predict the natural history of coronary stenoses:Fractional flow reserve versus angiography in multivessel evaluation 2 substudy. Circulation. 2018;137:1475-1485.
6. Mortensen MB, Dzaye O, Steffensen FH, et al. Impact of Plaque Burden Versus Stenosis on Ischemic Events in Patients With Coronary Atherosclerosis. J Am Coll Cardiol. 2020;76:2803-2813.
7. Shan P, Mintz GS, McPherson JA, et al. Usefulness of Coronary Atheroma Burden to Predict Cardiovascular Events in Patients Presenting With Acute Coronary Syndromes (from the PROSPECT Study). Am J Cardiol. 2015;116:1672-1677.
8. Prati F, Romagnoli E, Gatto L, et al. Relationship between coronary plaque morphology of the left anterior descending artery and 12 months clinical outcome:the CLIMA study. Eur Heart J. 2020;41:383-391.
9. Tu S, Westra J, Yang J, et al. Diagnostic Accuracy of Fast Computational Approaches to Derive Fractional Flow Reserve From Diagnostic Coronary Angiography:The International Multicenter FAVOR Pilot Study. JACC Cardiovasc Interv. 2016;9:2024-2035.
10. Westra J, Andersen BK, Campo G, et al. Diagnostic Performance of In?Procedure Angiography?Derived Quantitative Flow Reserve Compared to Pressure?Derived Fractional Flow Reserve:The FAVOR II Europe?Japan Study. J Am Heart Assoc. 2018;7:009603.
11. Cortés C, Carrasco-Moraleja M, Aparisi A, et al. Quantitative flow ratio —Meta-analysis and systematic review. Catheter Cardiovasc Interv. 2021;97:807-814.
12. Xu B, Tu S, Song L, et al. Angiographic quantitative flow ratio-guided coronary intervention (FAVOR III China):a multicentre, randomised, sham-controlled trial. Lancet. 2021;398:2149-2159.
13. Cortés C, Fernández-Corredoira PM, Liu L, et al. Long-term prognostic value of quantitative-flow-ratio-concordant revascularization in stable coronary artery disease. Int J Cardiol. 2023;389:131176.
14. Wang TKM, Oh THT, Samaranayake CB, et al. The utility of a “non-significant“coronary angiogram. Int J Clin Pract. 2015;69:1465-1472.
15. Kolozsvári R, Tar B, Lugosi P, et al. Plaque volume derived from three-dimensional reconstruction of coronary angiography predicts the fractional flow reserve. Int J Cardiol. 2012;160:140-144.
16. Huang FY, Huang BT, Lv WY, et al. The Prognosis of Patients With Nonobstructive Coronary Artery Disease Versus Normal Arteries Determined by Invasive Coronary Angiography or Computed Tomography Coronary Angiography:A Systematic Review. Medicine (Baltimore). 2016;95:3117.
17. Khajouei AS, Adibi A, Maghsodi Z, Nejati M, Behjati M. Prognostic value of normal and non-obstructive coronary artery disease based on CT angiography findings. A 12 month follow up study. J Cardiovasc Thorac Res. 2019;11:318-321.
18. Lee JM, Choi KH, Koo BK, et al. Prognostic Implications of Plaque Characteristics and Stenosis Severity in Patients With Coronary Artery Disease. J Am Coll Cardiol. 2019;73:2413-2424.
19. Tebaldi M, Biscaglia S, Fineschi M, et al. Evolving Routine Standards in Invasive Hemodynamic Assessment of Coronary Stenosis. JACC Cardiovasc Interv. 2018;11:1482-1491.
20. Ciccarelli G, Barbato E, Toth GG, et al. Angiography versus hemodynamics to predict the natural history of coronary stenoses:Fractional flow reserve versus angiography in multivessel evaluation 2 substudy. Circulation. 2018;137:1475-1485.
21. Fuster V, García-Álvarez A, Devesa A, et al. Influence of Subclinical Atherosclerosis Burden and Progression on Mortality. J Am Coll Cardiol. 2024;84:1391-1403.

ABSTRACT
Introduction and objectives: There is limited data on the impact of the culprit vessel on very long-term outcomes after ST-elevation myocardial infarction (STEMI). The aim was to analyze the impact of the left anterior descending coronary artery (LAD) as the culprit vessel of STEMI on very long-term outcomes.
Methods: We analyzed patients included in the EXAMINATION-EXTEND study (NCT04462315) treated with everolimus-eluting stents or bare-metal stents after STEMI (1498 patients) and stratified according to the culprit vessel (LAD vs other vessels). The primary endpoint was the patient-oriented composite endpoint (POCE), including all-cause mortality, myocardial infarction (MI) or revascularization at 10 years. Secondary endpoints were individual components of POCE, device-oriented composite endpoint and its individual components and stent thrombosis. We performed landmark analyses at 1 and 5 years. All endpoints were adjusted with multivariable Cox regression models.
Results: The LAD was the culprit vessel in 631 (42%) out of 1498 patients. The LAD-STEMI group had more smokers, advanced Killip class and worse left ventricular ejection fraction. Conversely, non-LAD-STEMI group showed more peripheral vascular disease, previous MI, or previous PCI. At 10 years, no differences were observed between groups regarding POCE (34.9% vs 35.4%; adjusted hazard ratio [HR], 0.95; 95% confidence interval [95%CI], 0.79-1.13; P = .56) or other endpoints. The all-cause mortality rate was higher in the LAD-STEMI group (P = .041) at 1-year.
Conclusions: In a contemporary cohort of STEMI patients, there were no differences in POCE between LAD as the STEMI-related culprit vessel and other vessels at 10 years follow-up. However, all-cause mortality was more common in the LAD-STEMI group within the first year after STEMI.
Keywords: Acute myocardial infarction. STEMI. Angiography. Coronary. Percutaneous coronary intervention.
RESUMEN
Introducción y objetivos: Existen datos limitados sobre el impacto a muy largo plazo del vaso culpable después de un infarto de miocardio con elevación del segmento ST (IAMCEST). El objetivo fue analizar el efecto de la arteria descendente anterior (DA) como vaso culpable en el IAMCEST en los resultados a muy largo plazo.
Métodos: Se analizaron los pacientes incluidos en el estudio EXAMINATION-EXTEND (NCT04462315) que recibieron stents liberadores de everolimus o stents metálicos después de un IAMCEST (1.498 pacientes) y se estratificaron según el vaso culpable (DA frente a otros vasos). El objetivo primario fue el objetivo combinado orientado al paciente (POCE) que incluyó muerte por cualquier causa, infarto agudo de miocardio (IAM) o revascularización a los 10 años. Los objetivos secundarios fueron los componentes individuales del POCE, el evento compuesto orientado al dispositivo y sus componentes individuales, así como la trombosis del stent. Se realizaron análisis de puntos de referencia a 1 y 5 años. Todos los objetivos fueron ajustados mediante modelos de regresión de Cox multivariantes.
Resultados: De los 1.498 pacientes, la DA fue el vaso culpable en 631 (42%). El grupo IAMCEST-DA mostró mayor proporción de fumadores, una clase Killip más avanzada y una peor fracción de eyección del ventrículo izquierdo. En cambio, el grupo sin IAMCEST-DA mostró mayor prevalencia de enfermedad vascular periférica, IAM previo y angioplastia coronaria previa. A los 10 años no se observaron diferencias entre los grupos para el POCE (34,9 frente a 35,4%; hazard ratio, 0,95; intervalo de confianza del 95%, 0,79-1,13; p = 0,56) ni para otros objetivos. Hubo una mayor mortalidad por cualquier causa en el grupo IAMCEST-DA (p = 0,041) al primer año.
Conclusiones: En una cohorte contemporánea de pacientes con IAMCEST no hubo diferencias en cuanto al POCE entre la DA como vaso culpable en el IAMCEST y los otros vasos a los 10 años de seguimiento. Sin embargo, en el primer año después del IAMCEST, la mortalidad por cualquier causa fue más común en el grupo IAMCEST-DA.
Palabras clave: Infarto agudo de miocardio. IAMCEST. Angiografía. Coronaria. Intervención coronaria percutánea.
Abbreviations
LAD: left anterior descending coronary artery. LVEF: left ventricular ejection fraction. MI: myocardial infarction. PCI: percutaneous coronary intervention. POCE: patient-oriented composite endpoint. STEMI: ST−segment elevation myocardial infarction.
INTRODUCTION
Percutaneous coronary intervention (PCI) is the first-line therapy in patients with ST-segment-elevation myocardial infarction (STEMI).1 The STEMI-related culprit vessel is usually considered as one of the most important prognostic factors in STEMI patients.2,3 This assumption comes from previous studies –conducted in the pre-reperfusion or thrombolysis era– which showed that left anterior descending artery (LAD)-related STEMIs were associated with worse clinical outcomes compared with right coronary (RCA) and left circumflex artery (LCX)-related lesions.4-9
However, in the contemporary era of primary PCI there are limited data about the prognostic impact of LAD as the STEMI-related culprit vessel especially in a very long follow-up.10,11
Therefore, the aim of this study was to investigate the impact of the LAD as the STEMI-related culprit vessel on very long-term clinical outcomes in STEMI patients undergoing primary PCI enrolled in the EXAMINATION-EXTEND study (10-year follow-up of the EXAMINATION trial).
METHODS
Study design and patients
The EXAMINATION trial (NCT00828087) was an all-comer, multicenter, prospective, 1:1 randomized, 2-arm, single-blind, controlled trial conducted at 12 centers across 3 countries to assess the superiority of EES (Xience V) vs BMS (Multilink Vision, Abbott Vascular) in STEMI patients regarding the primary endpoint of all-cause mortality, any myocardial infarction, and any revascularization at 1 year. The study had broad inclusion criteria and few exclusion criteria to ensure an all-comer STEMI population representative of the routine clinical practice. The study outcomes have been reported up to the year 5.12,13 After that, it was reinitiated as the EXAMINATION- EXTEND study to evaluate patient- and device-oriented composite endpoints at 10 years. The latter is registered at ClinicalTrials.gov (NCT04462315) as an investigator-driven extension of follow-up of the EXAMINATION trial. An independent study monitor (ADKNOMA, Barcelona, Spain) verified the adequacy of the extended follow-up and events reported. All events were adjudicated and classified by an independent event adjudication committee blinded to the therapy groups (Barcicore Lab, Barcelona, Spain). The 10-year primary endpoint results of the EXAMINATION-EXTEND study have been previously published.14 For the aim of this study, baseline, procedural characteristics and outcomes were stratified according to the STEMI-related culprit vessel (LAD vs others). All centers participating in the EXAMINATION trial received the approval of their Medical Ethics Committee, and all enrolled patients who had already signed their written informed consent forms. Medical ethics committee approval for EXAMINATION- EXTEND was granted at the institutions of the principal investigators (Hospital Clínic and Hospital Bellvitge, Barcelona, Spain), and the requirement to obtain informed consent to gather information on 10- year events was waived. The study complied with the Declaration of Helsinki.
Study endpoints
The primary endpoint of this study was the patient-oriented composite endpoint of all-cause mortality, any myocardial infarction, or any revascularization at 10 years. Secondary endpoints were each individual components of the primary endpoint, device-oriented composite endpoint (cardiac death, target-vessel myocardial infarction, target lesion revascularization), its individual components and stent thrombosis. Detailed descriptions of the study endpoints and definitions have been published previously.15
Statistical analysis
Continuous variables are expressed as median (interquartile range; IQR), and categorical variables as absolute and relative frequencies (percentages).
Baseline clinical, angiographic, and procedural characteristics were compared between the groups stratified by the STEMI-related artery (LAD vs other vessels) using the Wilcoxon rank sum test, the chi-square, or Fisher’s exact test, where appropriate.
Time-to-event curves for POCE and all-cause death were plotted using the one minus the Kaplan-Meier estimate and the cumulative incidence function for other outcomes. The incidence of events at the follow-up was compared between groups using log-rank or Grey’s test. Landmark analyses were also performed, setting landmark points at 1 and 5 years.
The association between LAD as a STEMI-related culprit vessel and events was analyzed in univariable and multivariable cause-specific Cox regression models. Covariates were added to the multivariable model in 2 blocks. The first model included all clinically relevant baseline characteristics variables with P < .1 in the between-groups comparison (LAD vs other vessels), i.e., sex, smoking status, peripheral vascular disease, previous PCI, previous CABG, previous MI, and Killip class. The second model (expanded adjustment) included both the baseline characteristics and the left ventricular ejection fraction (LVEF) at discharge.
Two-tailed P-value < .05 was considered statistically significant. All statistical analyses were performed using R (R Core Team (2022). R: a language for statistical computing. R Foundation for Statistical Computing, Austria) with the following packages: survival, tidycmprsk, jskm, and gtsummary.
RESULTS
Patient characteristics
In 631 (42%) out of the 1498 STEMI patients included in the EXAMINATION EXTEND trial, the LAD was the culprit vessel (LAD-STEMI group), whereas in 867 patients (58%) it was not (non- LAD-STEMI group). Patients’ inclusion flowchart is shown in figure 1.

Figure 1. Study flowchart. A total of 1498 patients were initially recruited. At 10 years, clinical follow-up was obtained in 95.2% of the patients. LAD, left anterior descending artery; STEMI, ST-elevation myocardial infarction.
LAD-STEMI group had a higher incidence of active smokers, advanced Killip class and more depressed LVEF vs the non-LAD-STEMI group, which, however, exhibited a higher incidence of peripheral vascular disease, previous MI and previous PCI (table 1). Also, although non-statistically significant, the frequency of late comers and bailout PCI was numerically higher in the LAD-STEMI group.
Table 1. Baseline clinical characteristics
| Clinical characteristics | Overall (N = 1498)a | LAD-STEMI (N = 631)a | Non-LAD-STEMI (N = 867)a | |
|---|---|---|---|---|
| 61 [51-71] | 61 [51-71] | 61 [51-70] | .778 | |
| 1,244 (83%) | 512 (81%) | 732 (84%) | .094 | |
| 415 (28%) | 197 (31%) | 218 (25%) | .009 | |
| 655 (44%) | 268 (43%) | 387 (45%) | .419 | |
| 725 (48%) | 307 (49%) | 418 (48%) | .843 | |
| 55 (3.7%) | 14 (2.2%) | 41 (4.7%) | .011 | |
| 31 (2.1%) | 14 (2.2%) | 17 (2.0%) | .726 | |
| 80 (5.3%) | 23 (3.7%) | 57 (6.6%) | .013 | |
| 61 (4.1%) | 18 (2.9%) | 43 (5.0%) | .042 | |
| 10 (0.7%) | 1 (0.2%) | 9 (1.0%) | .052 | |
| .126 | ||||
| 1,268 (85%) | 520 (82%) | 748 (86%) | ||
| 98 (6.5%) | 51 (8.1%) | 47 (5.4%) | ||
| 34 (2.3%) | 14 (2.2%) | 20 (2.3%) | ||
| 97 (6.5%) | 46 (7.3%) | 51 (5.9%) | ||
| < .001 | ||||
| I | 1,337 (90%) | 525 (83%) | 812 (94%) | |
| II | 115 (7.7%) | 76 (12%) | 39 (4.5%) | |
| III | 23 (1.5%) | 20 (3.2%) | 3 (0.3%) | |
| IV | 18 (1.2%) | 8 (1.3%) | 10 (1.2%) | |
| 52 (45, 58) | 46 [40-55] | 55 [50-60] | < .001 | |
| 1.38 (0.70, 3.00) | 1.27 [0.67-3.00] | 1.47 [0.75-3.00] | .353 | |
|
CABG, coronary artery bypass graft; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention. a>Median [interquartile range] or frequency (%). bWilcoxon rank sum test; Pearson’s chi-squared test; Fisher’s exact test. |
||||
Regarding procedural data, LAD-STEMI group received smaller stent diameter (3.12 mm vs 3.26 mm; P = .001) and had a lower incidence of ST-segment resolution than the non-LAD-STEMI group (73%, vs 50%; P = .001) (table 2). The use of GP IIb/IIIa inhibitors was numerically lower in the LAD-STEMI group, although the differences between groups were not statistically significant. Of note, almost half of the patients (46%) with LAD-STEMI had the lesion in the proximal LAD compared with 44% of them who had it in the mid/distal LAD.
Table 2. Angiographic and procedural characteristics
| Procedural characteristics | Overall (N = 1498)a | LAD-related STEMI (N = 631)a | Non-LAD-related STEMI (N = 867)a | |
|---|---|---|---|---|
| N/A | ||||
| LAD | 631 (42) | 631 (100) | 0 (0) | |
| LMCA | 3 (0.2) | 0 (0) | 3 (0.3) | |
| RCA | 650 (43) | 0 (0) | 650 (75) | |
| LCx | 207 (14) | 0 (0) | 207 (24) | |
| SVG | 7 (0.5) | 0 (0) | 7 (0.8) | |
| 188 (13) | 72 (11) | 116 (13) | .256 | |
| 3.9 [2.7-6.8] | 4.0 [2.7-7.3] | 3.9 [2.7-6.3] | .366 | |
| 976 (65) | 405 (64) | 571 (66) | .502 | |
| 785 (52) | 312 (49) | 473 (55) | .051 | |
| 885 (60) | 390 (63) | 495 (59) | .113 | |
| .312 | ||||
| DES | 751 (50) | 326 (52) | 425 (49) | |
| BMS | 747 (50) | 305 (48) | 442 (51) | |
| 1.39 (0.65) | 1.37 (0.63) | 1.40 (0.66) | .428 | |
| 23 (18-35) | 23 (18-33) | 23 (18-35) | .154 | |
| 3.20 (0.45) | 3.12 (0.40) | 3.26 (0.47) | < .001 | |
| 221 (15) | 97 (15) | 124 (14) | .564 | |
| .607 | ||||
| 0 | 26 (1.7) | 9 (1.4) | 17 (2.0) | |
| 1 | 12 (0.8) | 5 (0.8) | 7 (0.8) | |
| 2 | 59 (4.0) | 29 (4.6) | 30 (3.5) | |
| 3 | 1396 (94) | 584 (93) | 812 (94) | |
| 852 (63) | 285 (50) | 567 (73) | < .001 | |
|
BMS, bare metal stent; CABG, coronary artery bypass graft. DES, drug-eluting stent; LAD, left anterior descending coronary artery, LCx, left circumflex artery; LMCA, left main coronary artery; PCI, percutaneous coronary intervention; RCA, right coronary artery; STEMI: ST-segment elevation myocardial infarction; SVG, saphenous venous graft; TIMI, thrombolysis in myocardial infarction. aMedian [interquartile range], mean (standard deviation) or frequency (%). bFisher’s exact test; Pearson’s chi-squared test; Wilcoxon rank sum test. |
||||
Ten-year outcomes
At the 10-year follow-up, POCE did not differ between LAD-STEMI and non-LAD-STEMI group (adjusted HR, 0.95; 95%CI, 0.79-1.13; P = .56) (figure 2). Moreover, no differences were found in terms of each individual component of POCE (all-cause mortality, MI, any revascularization) (figure 3) and other secondary endpoints (figure 1 of the supplementary data). Furthermore, when the expanded adjustment was performed and LVEF was included in the multivariable analysis, there were no inter-group differences between (table 3).

Figure 2. Central illustration. Outcomes of patients with ST-segment elevation myocardial infarction according to the culprit vessel at the 10-year follow-up. LAD, left anterior descending coronary artery; STEMI: ST-segment elevation myocardial infarction; POCE: patient-oriented composite endpoint.

Figure 3. Time-to-event curves for the patient-oriented composite endpoint (A), all-cause mortality (B), myocardial infarction (C), and any revascularization (D) in patients stratified according to the culprit vessel. LAD, left anterior descending coronary artery; MI, myocardial infarction; STEMI, ST-segment elevation myocardial infarction; POCE, patient-oriented composite endpoint.
Table 3. Ten-year outcomes
| 10-year outcomes | LAD-related STEMI (N = 631) | Non-LAD-related STEMI (N = 867) | Unadjusted HR (95%CI) | Adjusted HR (95%CI) | Expanded adjusted HR (95%CI) | |||
|---|---|---|---|---|---|---|---|---|
| Patient-oriented composite endpointc | 220 (34.9) | 307 (35.4) | 0.99 (0.83-1.17) | .87 | 0.95 (0.79-1.13) | .56 | 0.98 (0.78-1.23) | .86 |
| All-cause mortalityd | 131 (21.6) | 179 (21.2) | 1.02 (0.82-1.28) | .84 | 0.93 (0.74-1.18) | .56 | 0.81 (0.59-1.09) | .17 |
| Any myocardial infarctione | 33 (5.5) | 53 (6.3) | 0.86 (0.56-1.33) | .50 | 0.93 (0.60-1.45) | .76 | 1.14 (0.67-1.93) | .61 |
| Any revascularization | 108 (17.4) | 161 (18.8) | 0.93 (0.73-1.18) | .55 | 0.96 (0.75-1.22) | .72 | 1.12 (0.83-1.52) | .45 |
| Device-oriented composite endpointf | 94 (14.3) | 132 (14.2) | 0.98 (0.75-1.28) | .88 | 0.91 (0.70-1.20) | .50 | 0.95 (0.67-1.35) | .77 |
| Cardiac death | 72 (9.8) | 95 (10.0) | 1.06 (0.78- 1.44) | .71 | 0.89 (0.65- 1.23) | .49 | 0.71 (0.47-1.09) | .12 |
| Target vessel myocardial infarction | 16 (2.6) | 36 (4.2) | 0.62 (0.34-1.11) | .10 | 0.69 (0.38-1.25) | .22 | 0.87 (0.43-1.77) | .71 |
| Target lesion revascularization | 44 (7.0) | 63 (7.3) | 0.97 (0.66-1.43) | .89 | 1.01 (0.68-1.49) | .96 | 1.20 (0.76-1.93) | .43 |
| Definite/probable stent thrombosisg | 17 (2.7) | 28 (3.3) | 0.84 (0.46-1.54) | .57 | 0.83 (0.45-1.55) | .57 | 0.80 (0.38-1.73) | .58 |
|
95%CI, 95% confidence interval; HR, hazard ratio; LAD, left anterior descending artery, STEMI: ST-elevation myocardial infarction. Data are expressed as no. (%). aCause-specific Cox regression model adjusted for sex, smoking status, peripheral vascular disease, previous percutaneous coronary intervention, previous coronary artery bypass graft, previous myocardial infarction, and Killip class. bCause-specific Cox regression expanded model, adjusted for baseline comorbidities and left ventricular ejection fraction at discharge. cComposite endpoint of all-cause death, any recurrent myocardial infarction, and any revascularization. dDeath was adjudicated according to the Academic Research Consortium definition. eMyocardial infarction was adjudicated according to the World Health Organization extended definition. fComposite endpoint of cardiac death, target vessel myocardial infarction, target lesion revascularization, and stent thrombosis. gStent thrombosis was defined according to the Academic Research Consortium definition. |
||||||||
Landmark analyses
POCE landmark analysis showed no differences between the 2 groups across different time points. (figure 4A). Looking specifically at the various POCE individual components, the LAD-STEMI group exhibited a higher rate of all-cause mortality within the first year vs the non-LAD-STEMI group (p = 0.041), but this difference disappeared thereafter (figure 4B). Between years 0 and 1, there was also a trend toward a lower rate of myocardial infarction in the LAD-STEMI group vs the non-LAD-STEMI group (p = 0.081), which disappeared after year 1 (figure 4C). No differences were ever found regarding any revascularization (figure 4D) or other secondary endpoints between the 2 groups (figure 2 of the supplementary data).

Figure 4. Landmark analysis for the patient-oriented composite endpoint (A), all-cause mortality (B), myocardial infarction (C), and any revascularization (D) in patients stratified according to the culprit vessel. LAD, left anterior descending coronary artery; MI, myocardial infarction; STEMI, ST-segment elevation myocardial infarction; POCE, patient-oriented composite endpoint.
DISCUSSION
The main findings of this study can be summarized as follows: a) STEMI patients with LAD as the culprit vessel have a different baseline clinical profile vs STEMI patients with other culprit vessels; b) in the contemporary era of primary PCI, LAD as the STEMI-related culprit vessel did not bring worse very long-term outcomes compared with other coronary vessels; c) nevertheless, between years 0 and 1 the LAD-STEMI group exhibited a higher all-cause mortality rate, which disappeared thereafter compared with non-LAD-STEMI group.
Cardiology community knows (as reflected by the ESC guidelines on the management of acute coronary syndromes) that STEMI with LAD involvement as culprit vessel is a clinical marker of high risk of further events.1 LAD-related STEMI represents, approximately, 40% up to 50% of all STEMIs,12,16 and its worse prognosis has been related to the large myocardium covered by the LAD flow compared with the myocardium supplied by other coronary vessels. Of note, those studies were performed in the pre-reperfusion4-7 and early thrombolysis/PCI era,8,9 when PCIs were still not widely available. In the PCI era, there are very few studies (with short or mid-term follow-ups ranging from 1 to 3 years) reporting that LAD-STEMI is associated with an increased risk of stroke, heart failure, all-cause mortality10,17 and cardiovascular death11 after the PCI.
In our analysis, conducted in a cohort where the PCI was extensively performed, LAD as the STEMI culprit vessel did not appear to confer a worse prognosis to patients at the 1- or even 10-year follow-up. Of interest, LAD-STEMI patients exhibited the classical clinical features related to LAD, such as advanced Killip class at the time of presentation, lower ST-segment resolution and lower LVEF, which is similar to previous studies.8-11,17 All these unfavorable clinical characteristics are indeed related to the large amount of myocardium damaged in a LAD-STEMI with subsequent heart failure and ventricular arrhythmias.17-19 Nevertheless, this did not translate into a worse, very long-term clinical outcome. Significantly, even after accounting for variations in LVEF (which we addressed separately in our model due to its perceived role in the outcome cascade) the results showed no differences. This observation stands in contrast to earlier evidence, where the higher mortality rate in this cohort had been partially attributed to the subsequent decline in LVEF after STEMI.9,10
Several explanations may be claimed to understand our main finding. It may be hypothesized that worse outcome related to anterior STEMI may have been overcome by the introduction of the PCI with quick myocardial reperfusion. Pharmacological treatment has been also improved from thrombolysis to the PCI era, not only in terms of antiplatelet agents, but also in terms of secondary prevention (high intensity statins and angiotensin converting enzyme inhibitors/angiotensin receptor blockers or angiotensin receptor/neprilysin inhibitors for left ventricular dysfunction).20-23 Furthermore, in our study, the LAD-STEMI group had a higher proportion of active smokers. Smoking cessation remains the most critical preventive measure for coronary artery disease. The relationship between smoking and cardiovascular outcomes has been a matter of discussion, as some studies have suggested improved cardiovascular outcomes, even in the long term, among smokers who experienced STEMI.24 However, many of these studies were observational registries conducted in the pre-PCI era. Recent evidence indicates that smoking is associated with more post-PCI long-term adverse outcomes.25 Therefore, the so-called “smoker’s paradox” might be better explained by factors such as younger age and a lower prevalence of other risk factors among smokers. Indeed, in our study, while the LAD-STEMI group had a higher proportion of smokers, they had a lower prevalence of other risk factors, such as peripheral vascular disease and a history of prior PCI or MI.
Last, but not least, in landmark analysis we found that between years 0 and 1, all-cause mortality was more common in the LAD-STEMI group. Notably, in this period, there was a numerically higher number of cardiac deaths (although not statistically significant, P = .12), a similar finding to other existing evidence that found a higher relatively short-term mortality in the LAD-STEMI group within the first 30 days. In these studies, the elevated short-term mortality was associated with acute sequelae, such as heart failure and was also speculated to be connected to other lethal complications, such as ventricular arrhythmias, cardiogenic shock or mechanical complications.10,11 In our cohort, we found a trend towards a higher rate of reinfarction in the non-LAD-STEMI group (P = .081) that was largely unrelated to TLR, TVMI, or stent thrombosis. This observation contrasts with previous literature that reported a more common occurrence of reinfarction at the follow-up in patients with the SVG as the culprit vessel26 as well as the LAD,8 but not in LCx or the RCA.9-11
Our 10-year follow-up revealed similar clinical event rates between LAD-STEMI and non-LAD-STEMI group, indicating absence of long- term divergence. Previous studies showed a favorable post-acute phase prognosis for LAD-STEMI patients,10,11 which is consistent with our findings. In fact, non-cardiac factors seem to impact long-term mortality more than infarct location does.19 Thus, patients with STEMI should receive uniform management focused on secondary prevention strategies, regardless of the culprit vessel. Unfortunately, insufficient long-term data collection limits deeper insights into these outcomes (such as the presence of heart failure, optimal medical therapy, or other comorbidities).
Limitations
This study presents several limitations. First, this is a non-prespecified post-hoc analysis of the EXAMINATION-EXTEND study and therefore its conclusions must be considered only hypothesis generating. The association between infarction and outcomes may be driven by confounders which have not been recorded in the study. Then, several clinical and procedural characteristics were not available for the analysis, such as specified in-hospital or follow-up clinical data, like optimal medical treatment or compliance to medication at the follow-up.
CONCLUSIONS
In a contemporary cohort of STEMI patients, there were no differences in POCE between LAD as the STEMI-related culprit vessel and other vessels at the 10-year follow-up. However, within the first year after STEMI, all-cause death was more common in the LAD-STEMI group. Our results should be considered as hypothesis-generating. Further studies are needed to specifically assess the relationship between infarction location and outcomes in a contemporary setting where interventional and medical treatments are optimized.
FUNDING
The EXAMINATION-EXTEND study was funded by an unrestricted grant of Abbott Vascular to the Spanish Society of Cardiology (promoter). P. Vidal Calés has been supported by a research grant provided by Hospital Clínic at Barcelona, Spain.
ETHICAL CONSIDERATIONS
The study fully complied with the Declaration of Helsinki and was approved by our Institutional Review Committee. All patients signed a written informed consent form before being included in this study. The clinical ethics committee gave its approval for the analysis of the data collected. In this work, SAGER guidelines regarding sex and gender bias have been followed.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence tools were used during the preparation of this work.
AUTHORS’ CONTRIBUTIONS
The authors declare they meet the full criteria and requirements for authorship and have reviewed and agree with the content of the article. P. Vidal Calés, K. Bujak, R. Rinaldi, A. Salazar Rodríguez, S. Brugaletta and M. Sabaté contributed to conceptualization, design, data analysis and drafting of the manuscript. L. Ortega-Paz, J. Gómez-Lara, V. Jiménez-Diaz, M. Jiménez, P. Jiménez-Quevedo, R. Diletti, P. Bordes, G. Campo, A. Silvestro, J. Maristany, X. Flores, A. De Miguel-Castro, A. Íñiguez, A. Ielasi, M. Tespili, M. Lenzen, N. Gonzalo, M. Tebaldi, S. Biscaglia, R. Romaguera, J.A. Gómez-Hospital and P. W. Serruys reviewed and edited the manuscript.
CONFLICTS OF INTEREST
M. Sabaté declares he has received consulting fees from Abbott Vascular and iVascular outside the submitted work. R. Romaguera is associate editor of REC: Interventional Cardiology. The journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. The rest of the authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THIS TOPIC?
- – In STEMI patients, the culprit vessel is often regarded as a crucial prognostic factor.
- – This assumption is based on earlier studies conducted during the pre-reperfusion or thrombolysis era, which demonstrated that STEMIs involving the left anterior descending coronary artery (LAD) were linked to poorer clinical outcomes vs those involving other vessels.
- – In the current PCI era, there is limited data on the long-term prognostic impact of the LAD as the culprit vessel in STEMI patients.
WHAT DOES THIS STUDY ADD?
- – Patients with LAD as the STEMI-related culprit vessel have a higher all-cause mortality within the first year after STEMI.
- – However, our study found that this difference did not persist beyond the initial year suggesting that the prognostic impact of the culprit vessel might pertain to the immediate post- STEMI period.
- – Moreover, our results support that (irrespective of the location of the infarction) all STEMI patients should receive uniform medical care in the long-term focused on implementing secondary prevention strategies.
REFERENCES
1. Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44:3720-3826.
2. De Luca G, Suryapranata H, van 't Hof AW, et al. Prognostic assessment of patients with acute myocardial infarction treated with primary angioplasty:implications for early discharge. Circulation. 2004;109:2737-2743.
3. Addala S, Grines CL, Dixon SR, et al. Predicting mortality in patients with ST-elevation myocardial infarction treated with primary percutaneous coronary intervention (PAMI risk score). Am J Cardiol. 2004;93:629-632.
4. Thanavaro S, Kleiger RE, Province MA, et al. Effect of infarct location on the in-hospital prognosis of patients with first transmural myocardial infarction. Circulation. 1982;66:742-747.
5. Stone PH, Raabe DS, Jaffe AS, et al. Prognostic significance of location and type of myocardial infarction:independent adverse outcome associated with anterior location. J Am Coll Cardiol. 1988;11:453-463.
6. Hands ME, Lloyd BL, Robinson JS, de Klerk N, Thompson PL. Prognostic significance of electrocardiographic site of infarction after correction for enzymatic size of infarction. Circulation. 1986;73:885-891.
7. Welty FK, Mittleman MA, Lewis SM, Healy RW, Shubrooks SJ, Jr., Muller JE. Significance of location (anterior versus inferior) and type (Q-wave versus non-Q-wave) of acute myocardial infarction in patients undergoing percutaneous transluminal coronary angioplasty for postinfarction ischemia. Am J Cardiol. 1995;76:431-435.
8. Kandzari DE, Tcheng JE, Gersh BJ, et al. Relationship between infarct artery location, epicardial flow, and myocardial perfusion after primary percutaneous revascularization in acute myocardial infarction. Am Heart J. 2006;151:1288-1295.
9. Elsman P, van 't Hof AW, de Boer MJ, et al. Impact of infarct location on left ventricular ejection fraction after correction for enzymatic infarct size in acute myocardial infarction treated with primary coronary intervention. Am Heart J. 2006;151:1239.
10. Entezarjou A, Mohammad MA, Andell P, Koul S. Culprit vessel:impact on short-term and long-term prognosis in patients with ST-elevation myocardial infarction. Open Heart. 2018;5:e000852.
11. Koga S, Honda S, Maemura K, et al. Effect of Infarction-Related Artery Location on Clinical Outcome of Patients With Acute Myocardial Infarction in the Contemporary Era of Percutaneous Coronary Intervention- Subanalysis From the Prospective Japan Acute Myocardial Infarction Registry (JAMIR). Circ J. 2022;86:651-659.
12. Sabate M, Cequier A, Iñiguez A, et al. Everolimus-eluting stent versus bare-metal stent in ST-segment elevation myocardial infarction (EXAMINATION):1 year results of a randomised controlled trial. Lancet. 2012;380:1482-1490.
13. SabatéM, Brugaletta S, Cequier A, et al. Clinical outcomes in patients with ST-segment elevation myocardial infarction treated with everolimus-eluting stents versus bare-metal stents (EXAMINATION):5-year results of a randomised trial. Lancet. 2016;387:357-366.
14. Brugaletta S, Gomez-Lara J, Ortega-Paz L, et al. 10-Year Follow-Up of Patients With Everolimus-Eluting Versus Bare-Metal Stents After ST-Segment Elevation Myocardial Infarction. J Am Coll Cardiol. 2021;77:1165-1178.
15. SabatéM, Cequier A, Iñiguez A, et al. Rationale and design of the EXAMINATION trial:a randomised comparison between everolimus-eluting stents and cobalt-chromium bare-metal stents in ST-elevation myocardial infarction. EuroIntervention. 2011;7:977-984.
16. Nabel EG, Braunwald E. A tale of coronary artery disease and myocardial infarction. N Engl J Med. 2012;366:54-63.
17. Reindl M, Holzknecht M, Tiller C, et al. Impact of infarct location and size on clinical outcome after ST-elevation myocardial infarction treated by primary percutaneous coronary intervention. Int J Cardiol. 2020;301:14-20.
18. Chen ZW, Yu ZQ, Yang HB, et al. Rapid predictors for the occurrence of reduced left ventricular ejection fraction between LAD and non-LAD related ST-elevation myocardial infarction. BMC Cardiovasc Disord. 2016;16:3.
19. Pedersen F, Butrymovich V, Kelbæk H, et al. Short- and long-term cause of death in patients treated with primary PCI for STEMI. J Am Coll Cardiol. 2014;64:2101-2108.
20. Wilt TJ, Bloomfield HE, MacDonald R, et al. Effectiveness of statin therapy in adults with coronary heart disease. Arch Intern Med. 2004;164:1427-1436.
21. Freemantle N, Cleland J, Young P, Mason J, Harrison J. beta Blockade after myocardial infarction:systematic review and meta regression analysis. BMJ. 1999;318:1730-1737.
22. Pfeffer MA, Greaves SC, Arnold JM, et al. Early versus delayed angiotensin-converting enzyme inhibition therapy in acute myocardial infarction. The healing and early afterload reducing therapy trial. Circulation. 1997;95:2643-2651.
23. Mehran R, Steg PG, Pfeffer MA, et al. The Effects of Angiotensin Receptor-Neprilysin Inhibition on Major Coronary Events in Patients With Acute Myocardial Infarction:Insights From the PARADISE-MI Trial. Circulation. 2022;146:1749-1757.
24. Barbash GI, White HD, Modan M, et al. Significance of smoking in patients receiving thrombolytic therapy for acute myocardial infarction. Experience gleaned from the International Tissue Plasminogen Activator/Streptokinase Mortality Trial. Circulation. 1993;87:53-58.
25. Yadav M, Mintz GS, Généreux P, et al. The Smoker's Paradox Revisited:A Patient-Level Pooled Analysis of 18 Randomized Controlled Trials. JACC Cardiovasc Interv. 2019;12:1941-1950.
26. Stone SG, Serrao GW, Mehran R, et al. Incidence, predictors, and implications of reinfarction after primary percutaneous coronary intervention in ST-segment-elevation myocardial infarction:the Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction Trial. Circ Cardiovasc Interv. 2014;7:543-551.

ABSTRACT
Introduction and objectives: Calcified coronary lesions are becoming more prevalent and remain therapeutically challenging. Although a variety of devices can be used in this setting, cutting balloons (CB) and scoring balloons (SB) are powerful and simple tools to treat calcified plaques vs more complex devices. However, there are some drawbacks: these are stiff and bulky balloons that, as a first device, complicate lesion crossing and navigability in the presence of tortuosity, thus making it extremely difficult to recross once the balloon has been inflated. The objective of this study was to evaluate the safety and efficacy profile of the new Naviscore SB designed to overcome these drawbacks.
Methods: The first-in-man Naviscore Registry is a multicenter, prospective trial that included 85 patients with moderate (34%) or severe (66%) de novo calcified coronary lesions located in the native arteries, with stable angina and an indication for percutaneous coronary intervention.
Results: Mean age was 71 ± 11 years, with a high prevalence of comorbidities. Used as the first device, the Naviscore was able to cross 76% of the lesions and was used in 98% of the cases effectively modifying the calcified plaque. Procedural success was achieved in 94% of cases. Basal stenosis of 81 ± 12% decreased to 33 ± 8.5% after Naviscore and to 7.5 ± 2.6% after stent implantation. There were no major adverse cardiovascular events during admission. Perforation, device entrapment or flow-limiting dissections did not occur—only type A/B dissections in 13%—which were fixed with stent implantation. Device performance was deemed superior to the usual SB or CB used by the participant centers.
Conclusions: The Naviscore SB is very effective crossing severely calcified lesions as the first device, with effective plaque modification, stent expansion and an excellent safety profile. The Naviscore improves the behavior of current CB and SB. Due to its simplicity of use and performance, the Naviscore can be the first-choice SB to treat significant calcified lesions.
Keywords: Calcified coronary lesions. Scoring balloon. Plaque modification.
RESUMEN
Introducción y objetivos: Las lesiones coronarias calcificadas son cada vez más prevalentes y suponen un reto terapéutico. Aunque se pueden tratar con distintos dispositivos, los balones de corte (BC) y de scoring (BS) son herramientas potentes y de más fácil uso que otros dispositivos de mayor complejidad. Sin embargo, tienen un alto perfil de cruce, son rígidos y cuesta cruzar la lesión como primer dispositivo; navegan mal y es difícil recruzar cuando ya se ha dilatado el balón. El objetivo del estudio fue evaluar la eficacia y la seguridad del nuevo BS Naviscore, diseñado para soslayar estos inconvenientes.
Métodos: El Registro Naviscore es un estudio por primera vez en humanos, multicéntrico y prospectivo, en 85 pacientes con lesiones coronarias de novo con calcificación moderada (34%) o grave (66%), localizadas en arterias nativas, con angina estable e indicación de angioplastia.
Resultados: La edad media fue de 71 ± 11 años y hubo una alta prevalencia de comorbilidad. Naviscore cruzó como primer dispositivo en el 76% de los casos y se empleó hasta en el 98% para dilatar la lesión. Se logró el éxito del procedimiento en el 94%. La estenosis basal pasó del 81 ± 12 al 33 ± 8,5% después de Naviscore y al 7,5 ± 2,6% después del stent. No se registraron eventos coronarios adversos durante la hospitalización. Tampoco hubo casos de perforación, atrapamiento del dispositivo ni disección limitante del flujo; solo disecciones tipo A/B en el 13%, resueltas tras el stent. El comportamiento de Naviscore se evaluó como superior al de los BC o BS habituales en los centros participantes.
Conclusiones: Naviscore tiene una alta capacidad de cruce de las lesiones como primer dispositivo, una gran eficacia en la modificación de la placa y un excelente perfil de seguridad. Por su facilidad de uso y eficacia, Naviscore podría considerarse como el BS de primera elección en el tratamiento de lesiones calcificadas complejas.
Palabras clave: Lesiones coronarias calcificadas. Balón scoring. Modificación de placa.
Abbreviations:
CB: cutting balloon. PCI: percutaneous coronary intervention. SB: scoring balloon.
INTRODUCTION
Currently, the number of percutaneous coronary interventions (PCI) involving moderate-to-severe calcified plaques is increasing due to a progressively aging population and extending procedural indications into more comorbid patients. The presence of such calcification is extremely relevant as it is strongly associated with worse outcomes, specially by means of stent underexpansion, a potent predictor of stent thrombosis or in-stent restenosis.1-3 Moreover, calcified plaques can make advancing the devices difficult and trigger stent deformation and entrapment, coronary artery dissection, or perforation.1,4-6. Currently, there is a growing interest in the assessment of plaque morphology and its modification prior to stent implantation, which has led to the development of multiple tools such as rotational atherectomy, lithotripsy, orbitational atherectomy, cutting balloons (CB) and scoring balloons (SB).7-11 The latter are easy to use and aim to create a controlled fracture of calcium deposits and plaque dilatation to facilitate stenting.12-14 However, despite their theoretical simplicity, these devices are bulky and stiff, making it difficult to cross the lesion at the first attempt, navigate the vessel, and recross the lesion once inflated. Therefore, there is a need for a more trackable and better-profiled SB to improve the uptake of these devices to treat calcified coronary artery disease.
The newly designed Naviscore SB (iVascular, Spain) seeks to address these drawbacks. Its structure is based on 125-µm thick nitinol laser cut filaments arranged in an axial pattern placed over a semi-compliant high-pressure balloon with a nominal pressure of 8 atm, a rated burst pressure of 20 atm, and a mean burst pressure of 26 atm (figure 1). A nylon compensation tube in the shaft helps to re-wrap during balloon deflation. The mechanical properties of nitinol tend to regain its original shape once the balloon has been deflated. The nylon compensation tube elongates once the balloon has been inflated and due to its elastic properties, it regains its original length when deflated (video 1 of the supplementary data). The 2 mechanisms produce a powerful re-wrapping of the entire system when the balloon has been deflated, regaining its original crossing profile, which allows for easy lesion recross and further dilatations as many times as required. Axial distribution of scoring elements provides a high push against calcified lesions. Nitinol elastic properties provide a better navigability through tortuous calcific vessels compared with rigid scoring elements, such as stainless steel. The durable hydrophilic coating of the Hydrax Plus catheter (iVascular, Spain) significantly reduces its coefficient of friction to 0.04 by increasing slip and navigability. Also, its axial design enables a far larger contact area with the vessel wall compared with other devices with spiral configuration of nitinol filaments such as the AngioSculpt catheter (Philips Healthcare, The Netherlands) (figure 2). In vitro testing (iVascular, Spain) was conducted to measure the crossing profile of different SBs using a non-contact laser meter where the profile is calculated through the shadow that has been created. This allows us to measure the profile without exerting any pressure on the device.15 The Naviscore crossing profile is 5% lower than Angiosculpt, and 31% lower than Wolverine (Boston Scientific, United States). This catheter is available in a wide range of measures from 1.5 mm up to 3.5 mm in diameter and from 6.0 mm up to 15 mm in length, all of them compatible with a 6-Fr guiding catheter.

Figure 1. Structure of the Naviscore SB. MBP, mean burst pressure; RBP, rated burst pressure.

Figure 2. In vitro model assessment of the AngioSculpt scoring surface (upper image) vs the Naviscore (lower image). The Naviscore scoring surface is 6 times larger than that of the AngioSculpt.
The present study aims to demonstrate the safety and efficacy profile associated with crossing and treating calcified coronary lesions with the Naviscore SB.
METHODS
The Naviscore first-in-man study is a multicentric and prospective registry that evaluated the device safety and efficacy profile in the treatment of calcified lesions in 85 patients from 10 centers (9 in Spain and 1 in Portugal), all from the Euro 4C Group, founded in 2018 and focused on the cardiac care of calcified and complex patients. All operators involved in this study were experts in the treatment of calcified coronary lesions and familiar with most tools designed to treat such lesions.
Inclusion criteria were the presence of de novo moderate-to-severe calcified lesions by angiographic criteria in the native coronary tree of patients with chronic coronary syndrome scheduled for a PCI due to symptom persistence despite optimal medical therapy and/or evidence of inducible ischemia. The only exclusion criterion was the presence of the patient’s hemodynamic compromise.
The study was designed to assess the safety and efficacy profile of Naviscore in terms of delivery success when used as the first device to dilate the lesion, plaque modification capabilities, and complications. Consequently, operators were asked to use the Naviscore in all cases as the first device to cross and dilate the lesion. However, in cases of failed lesion crossing, dilatation with a small balloon was recommended with subsequent re-use of the same Naviscore catheter.
Operators involved in the study had little prior experience with the Naviscore in, at least, 3 cases and were asked to include, at least, 5 patients in the study. The operators assessed the performance of the catheter in each procedure in terms of pushability, navigability, crossing, deflation time, re-wrap, recrossing capabilities and ease of retrieval, and made a subjective comparison with their routinely used SB or CB.
The baseline clinical characteristics were recorded prior to the procedure and angiographical and optical coherence tomography (OCT) images were analyzed separately by 2 different operators. Coronary angiography was performed using, at least, 2 orthogonal projections to show stenosis as it is commonly used in the routine clinical practice. The view with the most severe stenosis was selected for the quantitative analysis of the lesion before and after the PCI. Lesion calcification was angiographically categorized as none/mild, moderate (radiopacities were only noted during the cardiac cycle movement prior to contrast injection) or severe (radiopacities noted without cardiac movement prior to contrast injection involving both sides of the arterial lumen).16 Lesions were categorized as A, B1, B2 and C based on the modified ACC/AHA Task Force classification, which is in turn, based on the morphology and potential complexity of the PCI.17 Procedural success was defined as an angiographically residual percent diameter stenosis < 30% after stent implantation, absence of major complications and final Thrombolysis in Myocardial Infarction (TIMI) grade-3 flow.18 OCT analysis was performed as recommended in the routine clinical practice: lesion and proximal and distal references within 5 mm were used to estimate diameters and areas. Calcium cracks were defined as fissures involving a calcified region.19,20
Statistical analysis
Data are expressed as mean ± standard deviation for continuous variables with a normal distribution, median and interquartile range [IQR] for continuous variables with a non-Gaussian distribution, and counts and percentages for categorical data.
Statistical analyses were performed using the Stata software version 16.1 (College Station, TX, United States).
Ethical considerations
Informed consent was obtained from all the patients and the study was approved by the Research Ethics Committee. The authors declare that procedures were followed according to the regulations established by the Clinical Research and Ethics Committee and the Declaration of Helsinki of the World Medical Association.
RESULTS
From November 2021 through February 2022, a total of 85 patients—80% males—with a mean age of 71 ± 11 years were included in the present study. One center included a total of 21 patients and the remaining 9, between 5 and 10 patients each. Baseline patient and lesion characteristics are shown in table 1. Regarding comorbidities, the prevalence of diabetes mellitus, dyslipidemia, hypertension, and chronic kidney disease was 44%, 70%, 75%, and 18% respectively. Prior revascularization was present in 43% of the patients (PCI in 38% and coronary artery bypass graft in 16%). The left anterior descending coronary artery was the most common location of target lesions (41%), followed by the right coronary artery (28%), left circumflex artery (16%) and left main coronary artery (15%). Most lesions (87%) were categorized as type B2/C, 66% were severely calcified and 34% had moderate calcification by angiographic assessment. Chronic total occlusion was reported in 10% of treated lesions. Reference vessel diameter was 3.0 ± 0.5 mm; mean lesion length, 20.3 ± 9.4 mm; and diameter stenosis, 81.4 ± 12%.
Table 1. Baseline clinical and angiographic characteristics
| Clinical and angiographic characteristics (n = 85) | n (%) |
|---|---|
| Age, years | 71 ± 11 |
| Male | 68 (80%) |
| Diabetes | 37 (44%) |
| Dyslipidemia | 59 (70%) |
| Hypertension | 67 (75%) |
| Chronic kidney disease | 15 (18%) |
| Current/former smokers | 53 (62%) |
| Prior PCI/CABG | 37 (43%) |
| Type B2/C lesions | 74 (87%) |
| Severe calcification | 56 (66%) |
| Moderate calcification | 29 (34%) |
| Basal percent diameter stenosis | 81 ± 12% |
| Chronic total occlusion | 8 (10%) |
| Lesion location: Left main coronary artery | 13 (15%) |
| LAD | 35 (41%) |
| RCA | 24 (28%) |
| LCx | 13 (16%) |
|
CABG, coronary artery bypass graft; LAD, left anterior descending coronary artery; LCx, left circumflex artery; PCI, percutaneous coronary intervention; RCA, right coronary artery. |
|
The Naviscore catheter diameters used to dilate the lesions were 2.0 mm (21%), 2.5 mm (38%), 3.0 mm (31%), and 3.5 mm (10%). Mean number of device inflations was 2.7 ± 1.5 times.
The Naviscore crossing performance of is shown in figure 3. Despite the strong recommendation to use Naviscore as the first device, some operators decided to use Rotablator or small balloons first in 10 patients due to severely narrowed and/or calcified vessels. In all those cases, the Naviscore successfully crossed and dilated the lesion after the first attempt. In the 75 patients in whom the Naviscore was used as the first device, the lesions were crossed and treated successfully in 57 (76%) of them. In the remaining 18 (24%) patients, the Naviscore crossed the lesion after pre-dilatation with a small balloon in 16 (89%) patients. Only 2 patients had non-crossable lesions.

Figure 3. Crossing performance of the Naviscore.
PCI results and in-hospital outcomes are shown in table 2. Procedural success was achieved in 94% of cases. The mean lesion percent diameter stenosis decreased from 81.4 ± 12% at baseline to 33.3 ± 8.5% after Naviscore dilatation, with a residual percent diameter stenosis of 7.5 ± 2.6% after stent implantation. There were no in-hospital major adverse cardiovascular events or any cases of perioperative perforation or device entrapment. Coronary dissections occurred in 13% of the cases (all of them type A or B) and resolved after stent implantation.
Table 2. Angiographic and in-hospital results
| Angiographic and in-hospital clinical results (n = 85) | n (%) |
|---|---|
| Procedural success: residual percent diameter stenosis < 30% after stenting, absence of major complications and TIMI grade-3 flow | 80 (94%) |
| Percent diameter stenosis pre-Naviscore | 81 ± 12% |
| Percent diameter stenosis post-Naviscore | 33 ± 8.5% |
| Percent diameter stenosis post-stenting | 7.5 ± 2.6% |
| MACE (in-hospital) | 0% |
| Death, MI, emergency CABG | 0% |
| Perforation | 0% |
| Limiting flow dissection | 0% |
| Type A or B dissection | 11 (13%) |
| Device entrapment | 0% |
|
CABG, coronary artery bypass graft; MACE, major adverse cardiovascular events; MI, myocardial infarction; TIMI, Thrombolysis in Myocardial Infarction. |
|
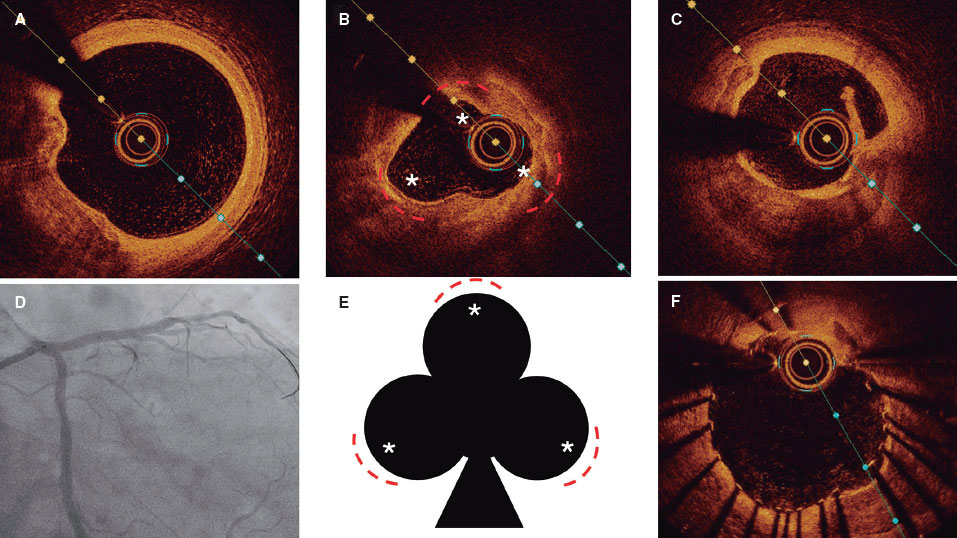
Ten procedures were OCT-guided. Pre-dilatation analysis could only be performed in 5 lesions; the OCT catheter could not cross the remaining lesions. Four of those had Fujino’s scores8 of 4 and in 2 of them the nodules protruded into the lumen. After dilatation, all lesions exhibited dissections that covered the intima and the media. Fractures were seen in all calcified plaques, which were deeper and wider in non-nodular calcified regions. Enlargement of lumen area after treatment with the Naviscore and correct stent apposition and expansion was observed in all imaging-guided cases (figure 4).

Figure 4. Clinical examples of 2 different lesions treated with the Naviscore. Angiography and baseline OCT (A) after dilatation with the Naviscore catheter (B) and post-stent implantation (C). On the left side, panel A shows a severely stenotic fibrocalcific plaque on the left anterior descending coronary artery that Naviscore (B) modifies creating calcium fractures (*) and dissection (arrow) resulting in stent implantation with good apposition and expansion (C). The right side shows a severely calcified plaque on the right coronary artery (A) with an arc of calcium of 180º at its proximal edge (lateral OCT picture) and 360º at its distal edge (central OCT picture) that the Naviscore modifies (B) creating calcium fractures (*) and dissection (arrow) resulting in stent implantation with good apposition and expansion (C). OCT, optical coherence tomography.
Table 3 shows the subjective performance of the Naviscore as evaluated by the operators of the present study. The Naviscore performance including push, navigability, crossing, deflation time, re-wrap, recrossing capabilities and ease of retrieval was deemed superior to the Wolverine, NSE Alpha (Nipro Co. Ltd., Japan), AngioSculpt, and Scoreflex (OrbusNeich, China).
Table 3. Subjective performance of the Naviscore compared with traditional and scoring or cutting balloons of participant centers. Push, capacity to cross the lesion, deflation time, rewrap and recrossing capabilities were the most valued characteristics of the Naviscore
| Parameter | Push | Navigability | Crossing | Friction | Device visibility | Deflation time | Rewrap | Recrossing capability | Ease of retrieving | Global evaluation |
|---|---|---|---|---|---|---|---|---|---|---|
| Better | 60% | 54% | 63% | 52% | 34% | 65% | 65% | 69% | 53% | 78% |
| Equal | 38% | 46% | 35% | 46% | 65% | 34% | 34% | 29% | 46% | 21% |
| Worse | 2% | 0% | 2% | 2% | 1% | 1% | 1% | 2% | 1% | 1% |
DISCUSSION
Findings of this first-in-man registry with the new SB Naviscore in moderately to severely calcified coronary lesions performed in CHIP (complex and high-risk intervention in indicated patients) by highly experienced operators on this field can be summarized as follows: a) the Naviscore was able to cross the lesions as the first device in 3 out of 4 patients in such difficult scenario; b) this device proved to be effective to treat complex coronary lesions, with procedural success rates of 94%; c) the Naviscore was safe as no major dissection, perforation, or device entrapment were observed and d) the performance of the Naviscore SB was better compared with other commercially available SB and CB as subjectively assessed by the experienced operators in this study.
Calcified coronary lesions account for up to 30% of lesions scheduled for PCI and are associated with worse clinical outcomes.1 Furthermore, these lesions are probably the most challenging ones for PCI operators. Thus, it is of paramount importance to develop specific devices for this scenario.21-23 Although several plaque-modification techniques have appeared in recent years, there are not very many head-to-head comparisons, thus complicating the choice between them. In contrast, several treatment combinations and algorithms have been published.24-27 Ablation techniques, such as rotational or orbital atherectomy, are especially indicated in uncrossable or undilatable lesions with balloon catheters. However, since there are more potential complications and a steeper learning curve associated with these therapies, developing new tools with a better crossing profile would be very positive in this scenario. The good crossability of the Naviscore SB showed in the present study is probably related to its unique nitinol structure in an axial configuration. CB such as the Wolverine or the NSE Alpha have a similar axial configuration of their cutting elements. However, the crossing profile of the Naviscore is 31% smaller than the CB. Although a comparative study on the crossing capabilities of those devices is not available, such a different profile favors the superior crossing capabilities of the Naviscore device. In fact, the operators of the present registry highlighted the ability to cross and recross lesions as one of the best features of the device compared with their usual CB or SB. Compared with the AngioSculpt—a SB that shares a nitinol structure with the Naviscore— the helical configuration of nitinol filaments in front of the axial alignment of the Naviscore nitinol filaments can make a difference. Axial alignment adds push to the device through the lesion, while the helical nitinol configuration can deform the structure under friction, thus reducing its navigability and, in some cases, cause device entrapment.28 Furthermore, as shown in figure 2, helical distribution of nitinol significantly reduces the nitinol scoring surface in front of an axial distribution.
The efficacy of the Naviscore balloon has proven to be good in the present study, with a procedural success rate of 94%. Furthermore, quantitative angiographic analysis showed a reduction of basal stenosis from 81% to 31% after Naviscore dilatation and to 7.5 ± 2.6% after stent implantation. Finally, the OCT evaluation confirmed the presence of extensive calcium fractures caused by the scoring filaments (figure 4). As the balloon gradually inflates, the radial forces concentrate along the surface of the nitinol scoring elements, resulting in a more controlled balloon expansion, increasing the force of the nitinol frame filaments to break down the calcified plaques.29 In vitro experiments comparing a simple SB (Scoreflex) with a conventional balloon catheter to dilate concentric tubes of calcium revealed that the inflation pressure required to break down the calcium tubes was consistently lower with SB. Finite element analysis revealed that the first main stress applied to the calcified plaque was, at least, 3-fold higher when inflating the balloon catheter with scoring elements.30 Naviscore has the largest scoring surface in the SB current market, being 6 times more extensive than the AngioSculpt (figure 2). Pressure concentration of the scoring elements is the mechanism of the increased ability of SB to dilate calcified lesions and facilitate stent expansion. Residual stenosis after stent placement was 7.5 ± 2.6% in our study.
Finally, the Naviscore proved to be safe in the present study, which could be justified by the mechanism of action of the device that uses nitinol filaments as the anchor to avoid balloon slippage, and allows balloon controlled expansion, minimizing the risk of barotrauma, coronary dissection, and perforation. Using OCT imaging, SB broke down the calcified lesion without the undesirable dissection of noncalcified segments, thus allowing successful stent implantation with adequate expansion.29,30
Limitations
One limitation of the study is its own design as a registry and therefore, the absence of a randomized comparator. Instead, expert operators in the treatment of calcified coronary lesions were asked to subjectively compare the device at test with their commonly used SB or CB in terms of push, cross/recross, rewrap, navigability and time of deflation. The subjective nature of this assessment, while providing valuable information, could be a limitation.
Another limitation is the sample size of the study, especially the size of the population involved in the OCT imaging analysis. Unfortunately, in our setting, the use of this technique to analyze calcified lesions, although on the rise, is still far from what would be recommended. However, and despite the limitations in terms of number, the cases analyzed with intracoronary imaging homogeneously show us the effect of the device under study—calcium fractures, dissection and increase in luminal area—as well as the optimal stent expansion.
The small sample of patients in this study does not allow for a disaggregated analysis by sex to draw any valid results.
CONCLUSIONS
The Naviscore SB is a step ahead in this field with an innovative design using a nitinol frame with axial distribution of filaments placed over a high-pressure balloon to improve current SB or CB designs. This provides a strong pushing capability and flexibility to cross the most difficult calcified lesions in 3 out of 4 patients and easily navigate through tortuous anatomy. Superior scoring surface provides strong plaque modification capabilities by facilitating calcium fractures and controlled dissections, and ultimately, optimal stent expansion. Uniform and controlled balloon expansion and the anchor effect provided by the nitinol frame minimizes the risk of uncontrolled dissections and distal embolization, thus providing an outstanding safety profile, confirmed in this study by the absence of major adverse cardiovascular events, device entrapment or flow- limiting dissections. Therefore, the Naviscore can be considered as the front-line SB, either alone or in combination with atheroablative techniques in the treatment of moderate-to-severe calcified lesions.
FUNDING
This study was partly funded by iVascular, Barcelona, Spain, who provided the devices to perform the study.
ETHICAL CONSIDERATIONS
Informed consent was obtained from all patients and the study was approved by the Research Ethics Committee. The authors declare that the procedures were followed in full compliance with the regulations set forth by the Clinical Research and Ethics Committee and Declaration of Helsinki of the World Medical Association. In accordance with the regulations of the SAGER guidelines, the small sample of patients in this study does not allow for a disaggregated analysis by sex to draw any valid results.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence has been used in the development of this paper.
AUTHORS’ CONTRIBUTIONS
A. Serra Peñaranda designed the protocol, database and study outline, participated in data collection, coordinated data analysis and interpretation, and drafted the article. E. Fernández Peregrina participated in data collection, data analysis and interpretation and drafted the article. M. Jiménez Kockar participated in data collection, data analysis and interpretation, and performed the statistical analysis. B. García del Blanco, S. Romani, J. Martín-Moreiras, E. Pinar Bermúdez, A. Rodrigues, S. Ojeda, N. Gonzalo López, A. Regueiro and A. Serrador Frutos participated in data collection and critically revised the manuscript. All authors gave their final approval to the last version for publication.
CONFLICTS OF INTEREST
S. Ojeda is an associate editor of REC: Interventional Cardiology. The journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. A. Serra Peñaranda and Ander Regueiro received consulting fees from iVascular, Barcelona, Spain. The remaining authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Calcified coronary lesions are becoming more prevalent in the routine clinical practice and remain therapeutically challenging for interventional cardiologists.
- Careful plaque modification is mandatory prior to stent implantation to achieve optimal results after the PCI.
- Several techniques and devices have been developed in this regard such as rotational and orbital atherectomy, lithotripsy and modified balloons.
- CB and SB are simple devices that do not require a learning curve. However, their design is that of a bulky and stiff device, which complicates lesion crossing, regarding navigation through vessels with some tortuosity and lesion recrossing once dilated.
WHAT DOES THIS STUDY ADD?
- The new Naviscore SB design is highly effective in crossing severely narrowed and calcified coronary lesions at the first attempt and has powerful plaque modification capabilities, while keeping an excellent safety profile.
- This device is a significant improvement over other CB and SB devices currently available in the market and could be selected as the first-choice SB tool to treat moderate-to-severe calcified lesions.
SUPPLEMENTARY DATA
Vídeo 1. Serra Peñaranda A. DOI: 10.24875/RECICE.M24000484
REFERENCES
1. Généreux P, Madhavan MV, Mintz GS, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes:Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trials. J Am Coll Cardiol. 2014;63:1845-1854.
2. Fujii K, Carlier SG, Mintz GS, et al. Stent underexpansion and residual reference segment stenosis are related to stent thrombosis after sirolimus-eluting stent implantation:An intravascular ultrasound study. J Am Coll Cardiol. 2005;45:995-998.
3. Kobayashi Y, Okura H, Kume T, et al. Impact of target lesion coronary calcification on stent expansion –An optical coherence tomography study –. Circulation. 2014;78:2209-2214.
4. Hendry C, Fraser D, Eichhofer J, et al. Coronary perforation in the drug-eluting stent era:Incidence, risk factors, management and outcome:The UK experience. EuroIntervention. 2012;8:79-86.
5. Madhavan M, Tarigopula M, Mintz GS, Maehara A, Stone GW, Généreux P. Coronary artery calcification:Pathogenesis and prognostic implications. J Am Coll Cardiol.2014;63:1703-1714.
6. Moussa I, Ellis SG, Jones M, et al. Impact of coronary culprit lesion calcium in patients undergoing paclitaxel-eluting stent implantation (a TAXUS-IV Sub Study). Am J Cardiol. 2005;96:1242-1247.
7. McInerney A, Escaned J, Gonzalo N. Calcified coronary artery disease:pathophysiology, intracoronary imaging assessment, and plaque modification techniques. REC Interv Cardiol. 2022;4:216-227.
8. Fujino A, Mintz GS, Matsumura M, et al. A new optical coherence tomography-based calcium scoring system to predict stent underexpansion. EuroIntervention. 2018;13:e2182-2189.
9. Vaquerizo B, Serra A, Miranda F, et al. Aggressive plaque modification with rotational atherectomy and/or cutting balloon before drug-eluting stent implantation for the treatment of calcified coronary lesions. J Interv Cardiol. 2010;23:240-248.
10. Moussa I, Di Mario C, Moses J, et al. Coronary stenting after rotational atherectomy in calcified and complex lesions. Angiographic and clinical follow-up results. Circulation. 1997;96:128-136.
11. de Maria GL, Scarsini R, Banning AP. Management of Calcific Coronary Artery Lesions:Is it Time to Change Our Interventional Therapeutic Approach?JACC:Cardiovascular Interventions. 2019;12:1465-1478.
12. Sugawara Y, Ueda T, Soeda T, Watanabe M, Okura H, Saito Y. Plaque modification of severely calcified coronary lesions by scoring balloon angioplasty using Lacrosse non-slip element:insights from an optical coherence tomography evaluation. Cardiovasc Interv Ther. 2019;34:242-248.
13. Barath P, Fishbein MC, Vari S, Forrester JS. Cutting balloon:A novel approach to percutaneous angioplasty. Am J Cardiol. 1991;68:1249-1252.
14. Okura H, Hayase M, Shimodozono S, et al. Mechanisms of acute lumen gain following cutting balloon angioplasty in calcified and noncalcified lesions: An intravascular ultrasound study. Catheter Cardiovasc Interv. 2002;57:429-436.
15. Keyence Telecentric measurement System TM-X5000 series. Available at: https://www.keyence.com/products/measure/micrometer/tm-x5000/. Accessed 12 Sep 2024.
16. Mintz GS, Popma JJ, Pichard AD, et al. Patterns of Calcification in Coronary Artery Disease A Statistical Analysis of Intravascular Ultrasound and Coronary Angiography in 1155 Lesions. Circulation. 1995;91:1959-1965.
17. Ellis SG, Vandormael MG, Cowley MJ, et al. Coronary Morphologic and Clinical Determinants of Procedural Outcome With Angioplasty for Multivessel Coronary Disease Implications for Patient Selection. Circulation. 1990;82:1193-1202.
18. TIMI Study Group. The thrombolysis in myocardial infarction (TIMI) trial. N Engl J Med. 1985;312:932-936.
19. Räber L, Mintz GS, Koskinas KC, et al. Clinical use of intracoronary imaging. Part 1 :guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. Eurointervention. 2018;14:656-677.
20. Fujino A, Mintz G, Lee T, et al. Predictors of calcium fracture derived from balloon angioplasty and its effect on stent expansion assessed by optical coherence tomography. JACC Cardiovasc Interv. 2018;11:1015-1017.
21. Généreux P, Lee AC, Kim CY, et al. Orbital Atherectomy for Treating De Novo Severely Calcified Coronary Narrowing (1-Year Results from the Pivotal ORBIT II Trial). Am J Cardiol. 2015;115:1685-1690.
22. Kereiakes DJ, Di Mario C, Riley RF, et al. Intravascular Lithotripsy for Treatment of Calcified Coronary Lesions:Patient-Level Pooled Analysis of the Disrupt CAD Studies. JACC Cardiovasc Interv. 2021;14:1337-1348.
23. Abdel-Wahab M, Toelg R, Byrne R, et al. High- speed rotational atherectomy versus modified balloons prior to drug-eluting stent implatation in severely calcified coronary lesions. The randomized PREPARE CALC Trial. Circ Cardiovasc Interv. 2018;11:e007415.
24. Jurado-Román A, Gómez-Menchero A, Gonzalo N, et al. Plaque modification techniques to treat calcified coronary lesions. Position paper from the ACI-SEC. REC Interv Cardiol. 2023;5:46-61.
25. Kawashima H, Kyono H, Nakashima M, et al. Cardiovascular Revascularization Medicine Prognostic impact of scoring balloon angioplasty after rotational atherectomy in heavily calcified lesions using second-generation drug- eluting stents:A multicenter registry-based study. CRM. 2020;21:322-329.
26. Tang Z, Bai J, Su SP, et al. Aggressive plaque modification with rotational atherectomy and cutting balloon for optimal stent expansion in calcified lesions. J Geriatr Cardiol. 2016;13:984-991.
27. Amemiya K, Hwa M, Maehara A, et al. Effect of cutting balloon after rotational atherectomy in severely calcified coronary artery lesions as assessed by optical coherence tomography. Catheter Cardiovasc Interv. 2019;94:936-944.
28. Sanchez Recalde A, Galeote G, Martin-Reyes R, Moreno R. Angiosculpt PTCA Balloon entrapment during dilatation of a heavily calcified lesion. Rev Esp Cardiol. 2008;61:1361-1363.
29. Kanai T, Hiro T, Takayama T, et al. Three-dimensional visualization of scoring mechanism of Angiosculpt balloon for calcified coronary lesions using Optical coherence tomography. J Cardiol Cases. 2021;5:e16-e19.
30. Kawase Y, Saito N, Watanabe S, et al. Utility of a scoring balloon for a severely calcified lesion:bench test and finite element analysis. Cardiovasc Interv Therapeutics. 2014;29:134-139.

ABSTRACT
Introduction and objectives: Ultrathin-strut stents (UTS) represent a significant advancement in percutaneous coronary intervention. This study aimed to evaluate the safety and short- to mid-term outcomes of stenting with the thinnest struts on the market (50 μm) using a biodegradable everolimus-eluting polymer (Evermine 50) in real-world patients with coronary artery disease.
Methods: A single-arm, multicenter, prospective study was conducted in real-world patients. A total of 161 patients with de novo lesions who received at least 1 UTS stent were enrolled. The primary safety endpoint was the occurrence of major adverse cardiovascular events, defined as cardiac death, target-vessel myocardial infarction, or the need for revascularization of the target lesion at 12 months. The incidence of stent thrombosis at 12 months was also analyzed.
Results: The study included 161 patients with a mean age of 64 ± 14 years; 79% were male, 34% had diabetes, and 66% had hypertension. The most common indication for intervention was non-ST-segment elevation myocardial infarction (42%), followed by ST-segment elevation myocardial infarction (22%). The procedural success rate was 100%. At 12 months of follow-up, the incidence of MACE was 2.5%, and the definite stent thrombosis rate was 1.3%.
Conclusions: The use of the 50 μm UTS stent with a biodegradable everolimus-eluting polymer demonstrated a favorable safety profile and good clinical outcomes in unselected patients at 1 year of follow-up.
Keywords: Coronary artery disease. Percutaneous coronary intervention. Ultrathin struts.
RESUMEN
Introducción y objetivos: Los stents de struts ultrafinos (SUF) constituyen una mejora en el campo del intervencionismo coronario percutáneo. El objetivo de este estudio fue evaluar la seguridad y los resultados a corto y medio plazo del stent con los struts más finos del mercado (50 μm), con polímero biodegradable y liberador de everolimus (Evermine 50), en pacientes del mundo real con enfermedad coronaria.
Métodos: Se diseñó un estudio prospectivo, multicéntrico, de un solo grupo, en pacientes del mundo real. Se incluyeron 161 pacientes con lesiones de novo en los que se implantó al menos 1 stent de SUF. La variable principal de seguridad fueron los eventos adversos cardiovasculares mayores, compuesto de muerte cardiaca, infarto de miocardio atribuido al vaso diana y necesidad de revascularización de la lesión diana a los 12 meses de seguimiento. También se analizó la incidencia de trombosis del stent a los 12 meses del procedimiento.
Resultados: De los 161 pacientes incluidos (edad media 64 ± 14 años; 79% varones), el 34% eran diabéticos y el 66% eran hipertensos. La indicación más frecuente fue infarto sin elevación del segmento ST (42%), con un 22% de casos en contexto de infarto con elevación del segmento ST. El porcentaje de éxito del procedimiento fue del 100%. A los 12 meses de seguimiento, la incidencia de eventos adversos cardiovasculares mayores fue del 2,5%, con una tasa de trombosis del stent definitiva del 1,3%.
Conclusiones: El uso de stent con SUF de 50 μm, con polímero biodegradable y liberador de everolimus en pacientes no seleccionados mostró unos buenos resultados clínicos, así como un buen perfil de seguridad a 1 año de seguimiento.
Palabras clave: Enfermedad coronaria. Intervencionismo coronario percutaneo. Strut ultrafino.
Abbreviations
MACE: major adverse cardiovascular events. MI: myocardial infarction. PCI: percutaneous coronary intervention. ST: stent thrombosis. STEMI: ST-segment elevation myocardial infarction. UTS: ultra-thin strut.
INTRODUCTION
Percutaneous coronary intervention (PCI) has grown exponentially along with the technological evolution associated with this procedure. The continuous advancement of technology has enabled the development of stents with thinner struts, which offer a series of advantages over stents with thicker struts. One of the advantages of these new stents is the improved device profile—with increased flexibility—providing better navigability and greater lesion crossing capability. On the other hand, ultra-thin struts (UTS) cause fewer disturbances to normal laminar blood flow at target lesion level, due to the reduced protrusion of material into the vascular lumen. This seems to be associated with a lower degree of platelet activation and muscle cell proliferation,—the processes involved in stent failure—in terms of stent thrombosis (ST) and in-stent restenosis.1,2 In lesions located in small caliber vessels (≤ 2.5 mm), the use of UTS could provide additional advantages due to a higher ratio between the size of the struts and the lesion luminal area.3 Furthermore, UTS stents seem to be associated with less acute damage to the vascular endothelium during stent deployment. This reduced initial aggression could diminish the barotrauma-related inflammatory response and, therefore, prevent in-stent restenosis and promote faster device endothelialization.4,5 Studies have indicated that the use of UTS stents could be associated with lower rates of in-stent restenosis and a reduced need for new revascularizations.6,7
The Evermine 50 EES stent (Meril Life Sciences, India) is a UTS (50 μm) stent with CE marking consisting of a cobalt-chromium alloy platform with an everolimus-eluting biodegradable polymer. The aim of this study was to evaluate the 1-year safety and efficacy outcomes after UTS stent deployment in real-world patients with coronary artery disease.
METHODS
We conducted a prospective, non-randomized, multicenter study with patients who underwent UTS stent deployment at 4 different Spanish hospitals (data from the Everythin Registry). To be included in the study, patients had to be older than 18 years, with available coronary angiographies in the context of chronic or acute coronary syndrome, and have, at least, 1 target lesion with a 2 mm up to 4.5 mm reference vessel diameter on visual estimation. Overlapping stents was ill-advised and, if necessary, the overlap length should be ≤ 2 mm. PCI in multiple vessels and lesions during the same surgical act was allowed, and deferred procedures within the first 90 days since the initial procedure were also accepted. In these cases, any further procedures were not coded as an event—i.e. need for new revascularization—but as scheduled procedures. Only 1 case—1 target lesion treated with UTS stent deployment—was counted per patient. Deploying the study UTS stent was not mandatory in any of the other treated lesions, only in the target lesion/vessel.
The study followed the privacy policy of each research center, including regulations for the appropriate use of data from patient research. The study was approved by the Ethics Committee for Drug Research of the coordinating center. Moreover, the study was conducted in full compliance with the terms set forth in the Declaration of Helsinki. All patients signed specific informed consent forms prior to being included in the study.
Study device and procedure
The Evermine 50 EES (Meril Life Sciences, India) is a UTS (50 μm) stent with a cobalt-chromium platform coated with a biodegradable polymer composed of poly-L-lactic acid and poly(lactic-co-glycolic) acid. The Evermine stent—which has a hybrid design with an open cell in its central part and a closed cell at the edges—releases everolimus (1.25 μg/mm²) as the antiproliferative drug. The stent has received the corresponding CE marking and is available in several lengths from 8 mm up to 48 mm with diameters ranging from 2 mm up to 4.5 mm. The main features of the Evermine 50 EES device are illustrated in figure 1.

Figure 1. A: illustrative image of the Evermine 50 stent (Meril Life Sciences, India). B: description of the main characteristics of the stent. C: comparison of the study stent strut thickness vs major competing next-generation stents. PLGA, poly(lactic-co-glycolic acid); PLLA, poly-L-lactic acid (Images courtesy of Meril Life Sciences. Images reproduced with permission from Meril Life Sciences or its affiliates.)
PCI was performed following each center routine practice within the recommendations outlined in the clinical practice guidelines.8 The PSP algorithm (predilation, sizing [stent size selection], and postdilation) was recommended for optimal device implantation. The study protocol recommended postdilation, especially in cases where any degree of underexpansion was identified immediately after device implantation. Although the study protocol recommended the use of intravascular imaging modalities to guide the procedure, this was left to the operator’s discretion. All patients received a 300 mg loading dose of acetylsalicylic acid prior to the intervention followed by a loading dose of a second antiplatelet agent—clopidogrel, prasugrel, or ticagrelor—after the PCI, which was maintained for 3 up to 12 months and left to the discretion of the responsible investigator of the center.
Endpoints and definitions
The primary endpoint of the study was the occurrence of major adverse cardiovascular events (MACE) at 12 months. MACE were defined as the composite of cardiac death, non-fatal target vessel myocardial infarction (MI), or the need for target lesion revascularization. Secondary endpoints included each individual component of the composite endpoint, the overall mortality and ST (both definite and probable) according to the definitions of the Academic Research Consortium9 12 months after implantation. Additionally, the rates of device and procedural success were taken into consideration. Device success was defined as the deployment of the study stent in the target lesion with a final percent diameter residual stenosis < 30% by visual estimation. Procedural success was defined as the success of the device without any in-hospital complications, including death, MI, and target lesion revascularization.
Statistical analysis
Quantitative variables are expressed as mean and standard deviation or as median and interquartile range [IQR], depending on their distribution. Categorical variables are expressed as number and percentage. All analyses were performed using the statistical tool STATA 12 (StataCorp LLC, United States).
RESULTS
Demographic and baseline clinical characteristics
A total of 161 patients were included in the study from November 2020 through April 2022 whose demographic data and clinical characteristics are shown in table 1. The mean age was 64 ± 14 years, and 79% were male. A total of 66% of the patients were hypertensive; 53% had dyslipidemia; 34%, diabetes mellitus, and 59% a history of smoking. A total of 20% of the patients had experienced a prior MI, and 22% a previous PCI. The most common indication for the intervention was the diagnosis of non-ST-segment elevation acute myocardial infarction (42%), followed by ST-segment elevation myocardial infarction (STEMI) (22%) and chronic coronary syndrome (21%).
Table 1. Baseline characteristics of the study population
| Basal characteristics | Patients (n = 161) |
|---|---|
| Age (years) ± SD | 64 ± 14 |
| Male, n (%) | 126 (79) |
| BMI (kg/m²) | 28 ± 3.5 |
| Hypertension, n (%) | 106 (66) |
| Dyslipidemia, n (%) | 86 (53) |
| Diabetes mellitus, n (%) | 55 (34) |
| Smoking status, n (%) | |
| Non-smoker | 65 (40) |
| Former smoker | 49 (30) |
| Current smoker | 47 (29) |
| Previous AMI, n (%) | 33 (20) |
| Previous stroke, n (%) | 2 (1.2) |
| Atrial fibrillation, n (%) | 7 (4.3) |
| Peripheral vascular disease, n (%) | 10 (6.2) |
| Previous coronary angioplasty, n (%) | 36 (22) |
| Previous coronary artery bypass grafting, n (%) | 4 (2.5) |
| COPD, n (%) | 13 (8) |
| Chronic kidney disease, n (%) | 14 (9) |
| Glomerular filtration rate (mL/min/1.73 m²) | 61 ± 10 |
| Left ventricular function, (%) | 55 ± 11 |
| Indication for coronary angiography, n (%) | |
| Chronic coronary syndrome | 34 (21) |
| Unstable angina | 24 (15) |
| NSTEMI | 67 (42) |
| STEMI | 36 (22) |
|
AMI, acute myocardial infarction; BMI, body mass index; COPD, chronic obstructive pulmonary disease; NSTEMI, non-ST-segment elevation acute myocardial infarction; SD, standard deviation; STEMI, ST-segment elevation myocardial infarction. |
|
Angiographic and procedural characteristics
The lesion angiographic characteristics, and the results of the intervention are shown in table 2. Most patients had significant single-vessel disease (71%), being the presence of 2 or 3-vessel disease far less common (20% and 9%, respectively). The most widely treated vessel was the left anterior descending coronary artery (54%), followed by the right coronary artery (27%) and the left circumflex artery (17%). The target lesion median percent diameter stenosis by visual estimation was 90% [IQR, 75-99]. A total of 29% of the target lesions showed some degree of calcification on angiography. Intracoronary imaging modalities (7% optical coherence tomography) were used to guide the PCI in 11% of the cases. The mean number of stents deployed per lesion was 1.04 ± 0.22, with a median stent diameter of 3.0 mm [IQR 2.75-3.5] and a median stent length of 19 mm [IQR 19-24]. Pre- and postdilation were performed in 71% and 39% of the cases, respectively. The device and procedural success rates were 100%, without any procedure-related complications being reported in patients treated during the inpatient period.
Table 2. Angiographic, procedural and clinical follow-up characteristics of the cohort
| Angiographic and procedural characteristics | Patients (n = 161) |
|---|---|
| Radial access, n (%) | 158 (98) |
| Diseased vessels, n (%) | |
| 1-vessel disease | 114 (71) |
| 2-vessel disease | 32 (20) |
| 3-vessel disease | 15 (9) |
| Target lesion location, n (%) | |
| Left main coronary artery | 3 (1.8) |
| Proximal left anterior descending coronary artery | 37 (23) |
| Mid left anterior descending coronary artery | 40 (24.8) |
| Distal left anterior descending coronary artery | 10 (6.2) |
| Proximal left circumflex artery | 10 (6.2) |
| Mid left circumflex artery | 11 (6.8) |
| Distal left circumflex artery | 6 (3.7) |
| Proximal right coronary artery | 13 (8) |
| Mid right coronary artery | 18 (11.2) |
| Distal right coronary artery | 13 (8) |
| Bifurcation lesions, n (%) | 12 (7.5) |
| Calcified lesions, n (%) | 46 (29) |
| Visual percent diameter stenosis, median [IQR] | 90 [75-99] |
| Predilation, n (%) | 114 (71) |
| Postdilation, n (%) | 63 (39) |
| Intracoronary imaging modalities, n (%) | 18 (11) |
| Optical coherence tomography | 11 (7) |
| Intravascular ultrasound | 7 (4) |
| No. of stents deployed, mean ± SD | 1.04 ± 0.22 |
| Stent diameter (mm), median [IQR] | 3.0 [2.75-3.5] |
| Stent length (mm), median [IQR] | 19 [19-24] |
| Device success, n (%) | 161 (100) |
| Procedural success, n (%) | 161 (100) |
| Antiplatelet therapy after PCI, n (%) | |
| Acetylsalicylic acid | 161 (100) |
| Clopidogrel | 78 (48) |
| Ticagrelor | 68 (42) |
| Prasugrel | 15 (9) |
| Clinical follow-up | |
| 12-month follow-up, n (%) | 158 (98) |
| MACE, n (%) | 4 (2.5) |
| Cardiac death | 1 (0.6) |
| Target vessel MI | 2 (1.3) |
| Target lesion revascularization | 2 (1.3) |
| Overall mortality, n (%) | 3 (1.9) |
| Stent thrombosis, n (%) | |
| Definite | 2 (1.3) |
| Probable | 1 (0.6) |
|
IQR, interquartile range; MACE, major adverse cardiovascular events; MI, myocardial infarction; PCI, percutaneous coronary intervention; SD, standard deviation. |
|
Clinical outcomes at the follow-up
The 12-month follow-up was completed in 158 patients (98%). One year after implantation, 4 patients exhibited MACE (2.5%), and 3 patients died (1.9%). The cause of death was cardiac in 1 patient (due to a probable ST 7 days after the procedure) and non-cardiac in the remaining 2 (one due to lung neoplasm and the other to multiple organ failure). There were 2 non-fatal MIs (1.3%), both due to late definite ST (1 occurred 8 months after stent deployment and was associated with the study UTS stent, while the other one occurred 9 months after deployment due to a different thrombosed non-UTS stent implanted in a lesion of the target lesion same vessel. Only 2 patients required target lesion revascularization at the follow-up (1 due to ST and the other one due to in-stent restenosis).
DISCUSSION
The present study prospectively and multicentrically evaluates the safety and efficacy profile of implanting an UTS stent in a real-world population. Its main findings are that the UTS stent demonstrated a high procedural success rate, without in-hospital complications, acceptable midterm clinical outcomes, and a 2.5% rate of MACE 12 months after implantation.
The baseline characteristics of the study population are similar to the ones reported in previous studies that analyzed various stent technologies in patients with atherosclerotic coronary artery disease.10-12 However, it is noteworthy that in this study, 79% of cases were performed in the context of an acute coronary syndrome, including 22% of patients diagnosed with STEMI. In acute coronary syndrome—especially STEMI—there are factors associated with poorer outcomes of the implanted device, both in the short and long term. Firstly, the state of generalized vasoconstriction of the coronary tree and high thrombotic burden can complicate the appropriate selection of the size of the stent, thus leading to the implantation of smaller devices in relation to the actual size of the vessel, a mechanism involved in ST and in-stent restenosis. Furthermore, in the context of acute lesions, there is a higher risk of embolization and no-reflow or slow-flow phenomena, which can sometimes condition suboptimal final outcomes in terms of distal coronary flow, involving a greater risk of further ST. In our study, no ST occurred in patients with an early diagnosis of STEMI. Although it is worth mentioning that the results of the study stent were good—even in demanding contexts such as STEMI—the absolute number of STEMI patients included was low, meaning that data should be contrasted in larger series.
UTS stents provide better navigability, flexibility, and conformity to the vessel geometry. However, there may be doubts on whether the presence of UTS can lead to a reduction of the stent radial strength, which could have further implications for treating more unfavorable lesions, such as calcified lesions. Although, in the present study, 29% of the treated lesions showed some degree of calcification on angiography, the success rate of the stent reached 100%. This demonstrates the good performance of this UTS stent across different scenarios, achieving excellent radial strength even in the most challenging situations, such as calcified coronary lesions. These results are especially relevant in the specific context of the study, where, despite the recommendation for systematic postdilation, the final rate of stent postdilation was relatively low (39%).
Previous studies have consistently shown good clinical follow-up results for UTS stents with low rates of ST.13-15 The reason for this low rate of ST would be strut thickness per se, which would favor early neointimal coverage, thereby reducing the risk of ST (especially late and very ST).4 In the specific case of the study device (Evermine 50 EES), Patted et al.13 described the 6-month follow-up results of 251 patients. In this single-center, prospective experience, the authors describe a 6-month rate of MACE of 0.8%, with no ST at the follow-up. Regarding differences with respect to our series, nearly one-third of the cases were procedures in asymptomatic patients or with silent ischemia. Additionally, the rate of postdilation (57%) was higher than that of our cohort, which may have influenced the ST outcomes. The same group retrospectively described the results of 171 patients treated with the Evermine 50 EES stent,16 with 2-year rates of procedural success and MACE of 100% (same as in our study) and 2.4%, respectively. Again, the authors noted the absence of definite or probable ST at the follow-up. In this single-center cohort, the rate of stent postdilation was not reported, which may have implications for the prevention of MACE, especially ST. A meta-analysis that analyzed various types of UTS stents found no significant differences in the likelihood of stent failure, including ST across different stents with struts < 70 μm.17 In the present study, although the 1-year rate of definite ST after stent deployment was 1.3%, only 1 of these STs was attributed to the study device. The rate of ST is similar to that of other real-world experiences with second and third-generation stents,18-20 which confirms the good performance of the Evermine 50 EES in unselected real-world patients.
Limitations
The main limitations of the study are the relatively low number of patients included, and the absence of a comparator group. Furthermore, although the events reported at the follow-up were reviewed by the principal investigator of the coordinating center based on the case reports submitted by each principal investigator from the collaborating centers, these events were not allocated by an independent event adjudication committee. The fact that, in our cohort, few intracoronary imaging modalities were used to guide the PCI—reflecting real clinical practice—could be interpreted as a limitation of the study.
CONCLUSIONS
With data from a prospective, multicentric study of real-world patients, the PCIs performed with a 50 μm UTS stent, with a biodegradable polymer and everolimus elution had good clinical outcomes and a favorable safety profile at the 12-month follow-up.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The study was approved by the Drug Research Ethics Committee of the coordinating center. The study was conducted in full compliance with the terms outlined in the Declaration of Helsinki. All patients signed specific informed consent forms prior to the intervention and before being included in the study.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used for this work.
AUTHORS’ CONTRIBUTIONS
J. Casanova-Sandoval and M. García-Guimarães participated in the conception and design of the study, analysis and interpretation of results, and drafting the manuscript. G. Miñana Escrivà, E. Bosch-Peligero, J.F. Muñoz-Camacho, D. Fernández-Rodríguez, K. Rivera, A. Fernández-Cisnal, and D. Valcárcel-Paz participated in data acquisition and critically reviewed the content of the manuscript. All authors gave their final approval for the publication of the latest draft of the manuscript.
CONFLICTS OF INTEREST
None declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- The use of UTS stents may be associated, through various mechanisms, with better clinical outcomes compared with thicker-strut stents. Previous studies suggest that UTS stents are associated with less stent failure, preventing in-stent restenosis and ST.
WHAT DOES THIS STUDY ADD?
- In this prospective, multicentric study of real-world patients, the use of a 50 μm UTS stent with a biodegradable polymer and everolimus elution was associated with good clinical outcomes, and a favorable safety profile at the 12-month clinical follow-up.
REFERENCES
1. Duraiswamy N, Schoephoerster RT, Moreno MR, Moore JE. Stented artery flow patterns and their effects on the artery wall. Annu Rev Fluid Mech. 2007;39:357-382.
2. Chiu JJ, Chien S. Effects of disturbed flow on vascular endothelium:Pathophysiological basis and clinical perspectives. Physiol Rev. 2011;91:327-387.
3. Buiten RA, Ploumen EH, Zocca P, et al. Outcomes in Patients Treated with Thin-Strut, Very Thin-Strut, or Ultrathin-Strut Drug-Eluting Stents in Small Coronary Vessels:A Prespecified Analysis of the Randomized BIO-RESORT Trial. JAMA Cardiology. 2019;4:659-669.
4. Miura T, Ueki Y, Senda K, et al. Early vascular response of ultra-thin bioresorbable polymer sirolimus-eluting stents assessed by optical frequency domain imaging:the EVALUATION study. Cardiovasc Interv Ther. 2021;36:281-288.
5. Otaegui Irurueta I, González Sucarrats S, Barrón Molina JL, et al. Can an ultrathin strut stent design and a polymer free, proendothelializing probucol matrix coating improve early strut healing?The FRIENDLY-OCT trial. An intra-patient randomized study with OCT, evaluating early strut coverage of a novel probucol coated polymer-free and ultra-thin strut sirolimus-eluting stent compared to a biodegradable polymer sirolimus-eluting stent. Int J Cardiol. 2022;360:13-20.
6. Kastrati A, Mehilli J, Dirschinger J, et al. Intracoronary stenting and angiographic results:Strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation. 2001;103:2816-2821.
7. Farb A, Weber DK, Kolodgie FD, Burke AP, Virmani R. Morphological predictors of restenosis after coronary stenting in humans. Circulation. 2002;105:2974-2980.
8. Neumann F-J, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
9. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials. Eur Heart J. 2018;39:2192-2207.
10. Nakamura M, Kadota K, Nakagawa Y, et al. Ultrathin, Biodegradable-Polymer Sirolimus-Eluting Stent vs Thin, Durable-Polymer Everolimus-Eluting Stent. JACC Cardiovasc Interv. 2022;15:1324-1334.
11. de la Torre Hernández JM, Ocaranza Sanchez R, Santas Alvarez M, et al. Comparison of One-Year Outcomes Between the ihtDEStiny BD Stent and the Durable-Polymer Everolimus- and Zotarolimus-Eluting Stents:A Propensity-Score-Matched Analysis. Cardiovasc Revasc Med. 2021;31:1-6.
12. De Silva K, Li Kam Wa ME, Wells T, et al. The everolimus eluting Synergy MegatronTM drug-eluting stent platform:Early outcomes from the European Synergy MegatronTM Implanters'Registry. Catheter Cardiovasc Interv. 2023;102:1222-1228.
13. Patted SV, Patted AS, Turiya PK, Thakkar AS. Clinical Outcomes of World's Thinnest (50 µm) Strut Biodegradable Polymer Coated Everolimus-Eluting Coronary Stent System in Real-World Patients. Cardiol Res. 2018;9:370-377.
14. Araujo GN, Machado GP, Moura M, et al. Real-World Assessment of an Ultrathin Strut, Sirolimus-Eluting Stent in Patients with ST-Elevation Myocardial Infarction Submitted to Primary Percutaneous Coronary Intervention (INSTEMI Registry). Arq Bras Cardiol. 2023;120:e20220594.
15. Kasturi S, Polasa S, Sowdagar MA, et al. Safety and Clinical Performance of Biodegradable Polymer-Coated Ultra-Thin Everolimus-Eluting Stents in “Real-World“Patients:A Multicenter Registry (PERFORM-EVER). Anatol J Cardiol. 2022;26:619-628.
16. Patted SV, Thakkar AS. Clinical outcomes of ultrathin strut biodegradable polymer-coated everolimus-eluting stent in patients with coronary artery disease. ARYA Atheroscler. 2020;16:130-135.
17. Marengo G, Bruno F, Scudeler L, et al. Comparison Among Ultra-Thin Coronary Stents:A Network Meta-Analysis. Am J Cardiol. 2024;216:9-18.
18. Park S, Rha S-W, Choi BG, et al. Efficacy and Safety of Sirolimus-Eluting Stent with Biodegradable Polymer UltimasterTM in Unselected Korean Population:A Multicenter, Prospective, Observational Study From Korean Multicenter Ultimaster Registry. Korean Circ J. 2024;54:339-350.
19. Yu HY, Ahn J, Choi BG, et al. Three-Year Clinical Outcomes With the Cilotax Dual Drug-Eluting Stent vs Everolimus-Eluting Stents in Patients With Acute Myocardial Infarction. Texas Hear Inst J. 2024;51:e238271.
20. Nakao S, Ishihara T, Tsujimura T, et al. Two-year real world clinical outcomes after intravascular imaging device guided percutaneous coronary intervention with ultrathin-strut biodegradable-polymer sirolimus-eluting stent. Int J Cardiol. 2024;399:131686.
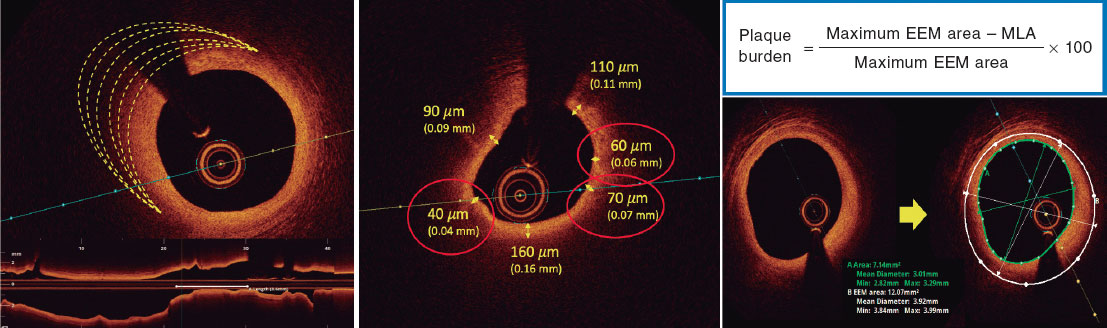
ABSTRACT
Introduction and objectives: The optimal treatment of nonculprit angiographic intermediate lesions (diameter stenosis 40%-69%) in patients with ST-segment elevation myocardial infarction (STEMI) is still unknown. Lesions with fractional flow reserve (FFR) ≤ 0.80 are indicative of ischemia and benefit from revascularization. However, lesions with FFR > 0.80 and optical coherence tomography (OCT) findings of vulnerability have been hypothesized to cause adverse events during follow-up. The study aims to compare the efficacy of a preventive treatment with stent implantation plus optimal medical therapy vs optimal medical therapy alone for nonculprit intermediate lesions with FFR > 0.80 and OCT findings of plaque vulnerability in STEMI patients at 4 years of follow-up.
Methods: This parallel-group, multicenter, controlled, single-blind, and 1:1 randomized trial will enroll a total of 600 STEMI patients with ≥ 1 intermediate nonculprit lesions with FFR > 0.80 and OCT findings of plaque vulnerability. The primary endpoint is target vessel failure, defined as the composite of cardiac death, target vessel myocardial infarction, or target vessel revascularization. The study will include a parallel registry of patients with FFR > 0.80 but without OCT findings of vulnerability. Vulnerable plaques are defined as lipid-rich fibroathermas with plaque burden ≥ 70% and a thin fibrous cap (≤ 80 μm).
Results: The VULNERABLE trial will reveal the role of preventive treatment with stent implantation for nonculprit and functionally nonsignificant vulnerable plaques in STEMI patients.
Conclusions: This is the first randomized trial of OCT-guided treatment of vulnerables plaques. Registered at ClinicalTrials.gov (NCT05599061).
Keywords: Fractional flow reserve. Optical coherence tomography. ST-segment elevation myocardial infarction. Vulnerable plaque.
RESUMEN
Introducción y objetivos: El tratamiento óptimo de las lesiones angiográficas intermedias (diámetro de estenosis 40-69%) no culpables en pacientes con infarto agudo de miocardio con elevación del segmento ST (IAMCEST) está por determinar. La reserva fraccional de flujo (RFF) permite diagnosticar lesiones causantes de isquemia (RFF ≤ 0,80) que se benefician de una revascularización. No obstante, las lesiones con RFF > 0,80 y criterios de vulnerabilidad por tomografía de coherencia óptica (OCT) también se ha hipotetizado que pueden causar eventos adversos en el seguimiento. El objetivo es comparar la eficacia del tratamiento preventivo con implantación de stent más tratamiento médico óptimo de lesiones intermedias no culpables con RFF > 0,80 y características de placa vulnerable frente a solo tratamiento médico óptimo en pacientes con IAMCEST a 4 años de seguimiento.
Métodos: Estudio de grupos paralelos, multicéntrico, controlado, aleatorizado 1:1 y simple ciego. Se incluirán 600 pacientes con IAMCEST y al menos una lesión intermedia no culpable que presenten RFF > 0,80 y características de placa vulnerable por OCT. El objetivo primario se define como fallo del vaso diana, compuesto de muerte cardiaca, infarto del vaso diana y necesidad de revascularización del vaso diana. El estudio incluye un registro paralelo para pacientes con RFF > 0,80 sin características de placa vulnerable. Se define placa vulnerable como fibroateromas lipídicos con carga de placa ≥ 70% y capa fibrosa fina (≤ 80 µm).
Resultados: El estudio VULNERABLE permitirá conocer el papel del tratamiento preventivo con stent de placas vulnerables no culpables funcionalmente no significativas en pacientes con IAMCEST.
Conclusiones: Se trata del primer estudio aleatorizado para el tratamiento de placas vulnerables guiado por OCT. Registrado en ClinicalTrials.gov (NCT05599061).
Palabras clave: Reserva fraccional de flujo. Tomografía de coherencia óptica. Infarto agudo de miocardio con elevación del segmento ST. Placa vulnerable.
Abbreviations
FFR: fractional flow reserve. MLA: minimum lumen area. OCT: optical coherence tomography. OMT: optimal medical therapy. PDE: percent diameter stenosis. STEMI: ST-segment elevation myocardial infarction.
INTRODUCTION
The presence of multivessel disease, defined as angiographic lesions with a percent diameter stenosis (PDS) ≥ 50% by visual estimation in patients with ST-segment elevation myocardial infarction (STEMI), is estimated to be approximately 50%.1 The COMPLETE trial compared angiography-guided preventive revascularization with stent implantation added to optimal medical therapy (OMT) for nonculprit lesions with a PDS ≥ 70% vs OMT alone.2 The trial found that angiography-guided preventive revascularization significantly reduced adverse cardiovascular events at 3 years of follow-up.2 Although the COMPLETE trial required physiological assessment using fractional flow reserve (FFR) for lesions with a PDS between 50% and 69% to guide the decision on revascularization, in practice, it was performed in only a very small percentage of patients.
The FLOWER-MI and FRAME-AMI trials3,4 investigated preventive stenting of FFR-guided nonculprit lesions—obtained through intracoronary pressure wire—compared with angiography-guided complete revascularization (visual estimation). Both trials mainly included intermediate lesions and demonstrated that pressure wire-guided preventive revascularization significantly reduces the need for revascularization, with similar or superior efficacy to angiography-guided complete revascularization.3,4 Despite these findings, clinical practice guidelines based on the COMPLETE trial recommend preventive stenting of nonculprit lesions guided by angiography alone.5,6
It is important to note that FFR is considered the gold standard for detecting myocardial ischemia (FFR ≤ 0.80). However, deferring treatment of nonculprit lesions that do not cause ischemia (FFR > 0.80) through OMT raises concerns in selected cases in which the anatomical features of the lesion suggest signs of vulnerability. In the FLOWER-MI trial, the group of patients randomized to undergo pressure-wire-guided revascularization with an FFR > 0.80 (referred for OMT) had more adverse events than those in the same group with FFR values ≤ 0.80 (referred for percutaneous revascularization).7 Several studies using intravascular imaging modalities have also demonstrated an association between the presence of fibro-lipid plaques with high lipid content and thin fibrous caps—known as vulnerable plaques—and the development of future adverse events due to plaque rupture.8,11
The VULNERABLE trial aims to evaluate the efficacy of a combined strategy using intracoronary physiological techniques and intravascular imaging to guide the treatment of intermediate nonculprit lesions in STEMI patients. The study hypothesis is that preventive stenting—in addition to OMT—in intermediate nonculprit lesions with FFR values > 0.80 and characteristics of vulnerable plaque will be superior to OMT alone. The present article includes the rationale and design of the study.
METHODS
Design
The VULNERABLE trial (NCT05599061) includes 3 groups based on the results obtained during the combined functional and anatomical assessment using pressure wires and optical coherence tomography (OCT). Figure 1 shows the study flowchart, which illustrates the 3 groups: patients with FFR ≤ 0.80 treated with stent (search failures), patients with FFR > 0.80 without vulnerable plaque characteristics (included in the registry group), and patients with FFR > 0.80 and vulnerable plaque characteristics (included in the randomized clinical trial).
This is a multicenter, controlled, prospective, randomized, parallel-group, single-blind study with patients included in the clinical trial group. The study will be conducted in accordance with the recommendations outlined in the Declaration of Helsinki on clinical research and has been approved by the lead ethics committee (Hospital Universitari de Bellvitge) and endorsed by the remaining ethics committees of participating centers. The participating centers and principal investigators are shown in table 1 of the supplementary data.
Table 1. Objectives of the VULNERABLE trial
| Primary endpoint |
|---|
| Compare the percentage of TVF between the 2 groups of patients assigned to the randomized clinical trial (FFR > 0.80 with characteristics of vulnerable plaque by OCT): preventive revascularization with stent + OMT vs OMT alone |
| Key secondary endpoints |
| Compare the percentage of TVF between patients allocated to the registry group (FFR > 0.80 without characteristics of vulnerable plaque by OCT and treated with the OMT) and patients allocated to the randomized OMT group (FFR > 0.80 with characteristics of vulnerable plaque) |
| Other secondary endpoints |
| Compare the rate of all-cause mortality reported between the 2 subgroups of randomized patients |
| Compare the percentage of cardiac deaths reported between the 2 subgroups of randomized patients |
| Compare the percentage of all myocardial infarctions reported between the 2 subgroups of randomized patients |
| Compare the percentage of target vessel myocardial infarctions reported between the 2 subgroups of randomized patients |
| Compare the percentage of target vessel revascularization needs between the 2 subgroups of randomized patients |
| Evaluate the percentage of restenosis and stent thrombosis in the preventive revascularization group with stent + OMT of the randomized clinical trial |
| * Although all objectives are marked with a complete 4-year follow-up, an interim study will be conducted at 2 years. ** All objectives will be calculated on an intention-to-treat basis according to the statistical plan. An exploratory per-protocol analysis will also be conducted based on the assessment by the study’s core imaging laboratory. |
|
FFR: fractional flow reserve; OCT: optical coherence tomograph; OMT: optimal medical treatment; TVF: target vessel failure. |
The study has been entirely designed and initiated by researchers and is sponsored by the Spanish Society of Cardiology Working Group on Intracoronary Diagnostic Techniques, which includes a steering committee, a data and safety monitoring board, and an independent event adjudication committee. The members of these committees are listed in table 2 of the supplementary data. The steering committee and all study investigators are committed to accurate data collection and adherence to the study protocol. The funding entity (Abbott Vascular, United States) plays no role in the study design, data collection, analysis, or the writing of the study results. The study sponsor (Foundation for Education in Interventional Cardiology Procedures [EPIC]), along with the principal investigators, is responsible for data management and confidentiality.
Table 2. Inclusion and exclusion criteria of the VULNERABLE trial
| Inclusion criteria |
|---|
| Patients older than 18 years |
| With STEMI (ST-segment elevation > 1 mm in, at least, 2 contiguous leads or true posterior ST-segment elevation with > 2 mm depression in anterior leads or new onset left bundle branch block) treated with successful revascularization of the culprit lesion within 72 hours from symptom onset |
| Presenting with multivessel disease with, at least, 1 angiographically intermediate lesion (PDS of 40% up to 69% by visual estimation) in a native vessel different from the culprit vessel |
| Planned FFR-guided percutaneous revascularization with a single 2.0 mm-to- 4.5 mm stent |
| Between 1 and 60 days after the index procedure (revascularization of the STEMI culprit vessel) |
| Exclusion criteria |
| Life expectancy < 4 years |
| Women of childbearing age who wish to become pregnant |
| Known intolerance to acetylsalicylic acid, heparin, everolimus, or iodinated contrast |
| Unresolved mechanical complications or infarct-related cardiogenic shock |
| Lesions suitable for the study located in the left main coronary artery, vessels with previous revascularization, in coronary bifurcations with > 2.5 mm side branches, severe angulations, or segments with severe calcification |
| History of severe asthma |
| Chronic kidney disease with glomerular filtration rate < 45 mL/min |
|
FFR: fractional flow reserve; PDS: percent diameter stenosis; STEMI: ST-segment elevation myocardial infarction. |
Endpoints
The primary objective of the VULNERABLE study (NCT05599061) is to compare the efficacy of preventive stenting combined with OMT vs OMT alone for intermediate lesions in noninfarct-related arteries with an FFR > 0.80 and vulnerable plaque characteristics as identified by OCT over a 4-year follow-up period. The primary endpoint of the study is the rate of target vessel failure (TVF), which is defined as a composite of cardiac death, target vessel myocardial infarction, or the need for target vessel revascularization.
The study also aims to evaluate several secondary endpoints, which are summarized in table 1. Among these secondary objectives, a key focus is the comparison of the TVF rate (the primary endpoint) between the registry group (patients with FFR > 0.80 without vulnerable plaque characteristics treated with OMT) and the randomized OMT arm of the clinical trial (patients with FFR > 0.80 and vulnerable plaque characteristics). The study endpoints are defined in table 3 of the supplementary data.12,13
Patient inclusion and exclusion criteria
The inclusion and exclusion criteria for the study are detailed in table 2. In brief, all patients with STEMI who have undergone successful revascularization of the culprit lesion and have at least 1 intermediate lesion (visually defined as having a DS of 40%-69%) in a noninfarct-related artery will be eligible for the study if percutaneous revascularization with a single stent guided by FFR is being considered. The study procedure must be conducted between 1 and 60 days after the revascularization of the culprit lesion. Patients must provide informed consent prior to the elective procedure for evaluating the nonculprit lesion.
Study protocol for nonculprit lesions and randomization
Eligible lesions will first be assessed with a pressure wire following the standard procedures in each center. Lesions with an FFR ≤ 0.80 will be considered search failures, and revascularization will be recommended based on clinical indications.5,6
Lesions with an FFR > 0.80 will be further evaluated with OCT according to the standard acquisition methods to detect vulnerable plaques in each center. The decision on whether a lesion meets the criteria for vulnerable plaque will be made by an accredited local investigator during the study procedure.
Patients with at least 1 lesion with an FFR > 0.80 without vulnerable plaque characteristics on OCT will be included in the registry group of the study. The protocol recommends OMT for all lesions with an FFR > 0.80 without vulnerable plaque characteristics. These patients will receive the same clinical follow-up as those in the randomized clinical trial group.
Patients with at least 1 lesion with an FFR > 0.80 that meets the criteria for a vulnerable plaque on OCT will be included in the clinical trial group. These patients will be randomized 1:1 to either preventive stenting combined with OMT or OMT alone (figure 1). Randomization will be conducted without stratification by center or clinical condition, using telematic algorithms. This process will be carried out online via the data collection platform provided by pInvestiga (Pontevedra, Spain).
Figure 1. Study diagram. FFR, fractional flow reserve; OCT, optical coherence tomography; OMT, optimal medical treatment; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction.
The supplementary data provide additional details on the FFR assessment method, including special situations where the lesion under study could not be fully evaluated, instances of unstable nonculprit plaques, complications related to diagnostic techniques, or patients with more than 1 nonculprit lesion.
Study device and implantation procedure
Patients with an FFR > 0.80 and vulnerable plaque characteristics identified by OCT assigned to the percutaneous coronary intervention group will be treated with an everolimus-eluting stent (Xience, Abbott, United States). According to the protocol, stent implantation must be guided by OCT. The criteria for OCT-guided stent implantation are detailed in table 4 of the supplementary data.
Optimal medical therapy
All patients included in both the randomized clinical trial and the registry must receive treatment in accordance with the European Society of Cardiology guidelines for managing acute coronary syndromes.5 The study protocol emphasizes managing modifiable risk factors—such as diet, smoking, obesity, exercise, and psychological status—as well as nonmodifiable risk factors, with set targets for blood pressure (systolic < 130 mmHg and diastolic < 80 mmHg), low-density lipoprotein cholesterol (< 55 mg/dL), and glycated hemoglobin A1c (< 7%). Pharmacological therapy should include beta-blockers and renin-angiotensin system inhibitors. Dual antiplatelet therapy is also recommended, but only during the first year after the index procedure, at the discretion of each center. As per the protocol, patient treatment details will be reported annually, and 2 lipid profile tests will be conducted throughout the study.
Vulnerable plaque criteria on optical coherence tomography and investigator training
Based on histopathological data, a plaque is defined as vulnerable when it is caused by a fibroatheroma with a large necrotic core composed of cellular debris and a high number of inflammatory cells, covered by a thin fibrous cap (≤ 65 µm).14 The criteria for identifying a vulnerable plaque in the study are adapted from the classic histopathological definition but modified for OCT assessment. These criteria are shown in figure 2.
Figure 2. Vulnerable plaque criteria by optical coherence tomography. EEM, external elastic membrane; minimal lumen area.
According to the protocol, 3 simultaneous criteria are required to define a vulnerable plaque by OCT:
The presence of a fibro-lipid plaque with a necrotic core covering more than 90º of the perimeter of the vessel over a length of more than 5 mm. A necrotic core is defined as a hypointense image with poorly defined borders that attenuates the OCT light beam, preventing visualization of the artery behind the core.
The presence of a thin fibrous cap, defined as ≤ 80 µm (65 + 15 µm axial resolution) in ≥ 3 consecutive images. The fibrous cap is defined as the tissue separating the necrotic core from the vessel lumen. Investigators will be trained to differentiate other findings that could be mistaken for a thin cap on OCT. Figure 3 shows examples of analogous OCT images that may mimic a thin fibrous cap but do not correspond to vulnerable plaques.
Figure 3. Distinction between vulnerable plaques and other findings by optical coherence tomography (OCT). A: plaque with superficial calcium (hypointense core with well-defined margins that do not attenuate the passage of light; arrow) and a thin fibrous cap. B: calcified nodule (arrow) protruding into the lumen and attenuating the signal, despite being composed of calcium. C: tangential signal loss (arrow) due to insufficient light beams caused by the peripheral, noncentral position of the OCT probe. D: superficial accumulation of macrophages (arrow) with a hyperintense appearance relative to the adjacent intima, with signal attenuation behind. E: presence of blood in the lumen due to inadequate flushing (arrow) during image acquisition, which distorts the arterial wall image, creating the appearance of hypointense regions. F: presence of blood between the probe and the OCT catheter (arrow) due to inadequate flushing, which distorts the arterial wall image and mimics hypointense regions.
Investigators will be required to measure a plaque burden of ≥ 70% in the cross-sectional area corresponding to the minimal luminal area (MLA) within the lesion. To perform this assessment, it is necessary to measure the vessel perimeter by delineating the external elastic membrane (EEM). Due to the difficulty of assessing the vessel perimeter in fibro-lipid plaques, especially at the MLA site, investigators will be trained to choose a section as close as possible to the MLA, where at least 60% of the vessel perimeter can be visualized if it is not possible at the same point. This allows for calculation using the following formula (figure 4):
Figure 4. Plaque burden assessment by optical coherence tomography. A: cross-section of the minimal lumen area. B: cross-section where the external elastic membrane (EEM) was measured. Since the EEM cannot usually be assessed in the cross-section corresponding to the MLA, an approximate estimation is made by measuring the EEM within 10 mm proximal or distal to the MLA (preferably distal) in the absence of side branches. The EEM will be assessed in the first cross-section where 60% of the EEM perimeter can be evaluated.
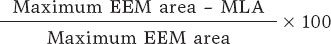
As per protocol, at least 1 local investigator from each participating center must have completed an online training course for the detection and assessment of vulnerable plaques using OCT, following the study criteria. Upon completing this course and passing a specific questionnaire, the investigator will be certified and approved to participate in the study.
Angiographic and optimal coherence tomography quantification analyses
The study includes an independent imaging laboratory for angiographic quantification and OCT analysis (Barcelona Cardiac Imaging Core Laboratory [BARCICORE-Lab]) to monitor adherence to the study criteria for diagnosing vulnerable plaques. A blinded analysis of the study results will be conducted, and patients will be assigned according to the protocol for exploratory analysis. A detailed explanation of the angiographic and OCT analysis conducted by the study laboratory is shown in the supplementary data.
Clinical follow-up and blinding
Patients in both the registry group and the randomized clinical trial group will undergo clinical follow-up for 4 years. Follow-up will include telephone consultations at 1 and 3 years, and in-person visits at 2 and 4 years. Each follow-up will involve an electrocardiogram and blood tests with cholesterol determination.
Patients in the randomized clinical trial group will be blinded to their assigned treatment group (single-blind). The details of blinding and monitoring are specified in the supplementary data.
Sample size calculation
The sample size has been calculated for the randomized clinical trial group. The number of patients included in the registry and search failures will depend on the total number needed to achieve the estimated sample size for the randomized trial.
According to previous studies on patients with acute coronary syndrome, theTVF rate for nonculprit lesions meeting vulnerable plaque criteria treated with OMT is estimated to be around 8% to 10% at 4 years. In similar lesions treated with stenting, the rate is approximately 4%.2,7,9 The studies used for the sample size calculation are summarized in table 5 of the supplementary data. Based on the study hypothesis, preventive stenting in nonculprit lesions with an FFR > 0.80 and vulnerable plaque characteristics is expected to reduce the primary endpoint by 60%. The estimated rate of TVF in the OMT group at 4 years is 10%. Assuming an annual loss to follow-up rate of 1.5% (total 6%), randomizing 600 participants 1:1 to preventive stenting plus OMT vs OMT alone will provide 80% power to demonstrate the superiority of preventive stenting with a 2-sided alpha error of .05.
Statistical analysis plan
The primary and secondary endpoints will be analyzed using the intention-to-treat principle at the 4-year follow-up. Comparisons will estimate event proportions between groups using logistic regression and will be reported as odds ratios with 95% confidence intervals. Only 1 event per patient will be counted for the primary endpoint. P values < .05 will be considered statistically significant for the primary endpoint. Kaplan-Meier curves will be used to visualize the time to the first event between groups.
For primary endpoint composites with missing data, a specific monitoring plan will determine if the missing data are random. In cases where data are adjudicated as missing at random, imputation methods will be used. For nonrandom missing data, sensitivity analyses using worst-case and last observation carried forward methods will be conducted.
Subgroup analyses will be performed for the primary and secondary endpoints, which involves comparing TVF rates between registry patients and those randomized to OMT in the clinical trial. Prespecified subgroups include: age > 75 years, sex, diabetes mellitus, left ventricular ejection fraction ≤ 35% at the time of the procedure, lesions in the proximal or mid-left anterior descending artery, and lesions in vessels with a reference diameter ≤ 2.75 mm.
Additionally, a hypothesis-generating parallel analysis will be conducted according to the study protocol. Patients will be included in the analysis only if the imaging laboratory confirms that their assigned treatment group, as determined by the local investigator, is consistent with the presence of vulnerable plaque identified by OCT. Patients will be excluded if there is a discrepancy between the investigator’s assignment and the imaging laboratory’s findings.
Interim analysis
After 2 years of follow-up, an interim analysis of the data is planned to monitor the primary endpoint in the randomized clinical trial group. Clinical follow-up will be extended if the events observed in the OMT arm of the randomized clinical trial are less than 4%.
DISCUSSION
The VULNERABLE trial aims to investigate the combined use of intracoronary physiology and images to guide the treatment of intermediate nonculprit lesions in STEMI patients.
Several lipid-lowering and anti-inflammatory drugs have been shown to reduce thrombotic events in patients with STEMI, likely by stabilizing functionally nonsignificant vulnerable plaques.15,17 In the PACMAN-AMI trial, treatment with alirocumab in addition to statins significantly reduced atheroma, decreased lipid content, and led to thickening of the fibrous cap compared with placebo in coronary regions with angiographically nonobstructive atherosclerosis (DS, 20%-50%).18 However, it is noteworthy that only 31% of patients in that study exhibited all 3 markers of reduced atherosclerosis, and data on more significant plaques (eg, 40%-69% stenosis with vulnerability criteria) were not specified.19
The use of stents in patients with vulnerable plaques is intended to enhance neointimal healing of the struts, which thickens the fibrous cap and stabilizes the plaque. The randomized PREVENT trial assessed the effectiveness of preventive stenting for functionally nonsignificant vulnerable lesions in patients with chronic coronary syndrome compared with OMT. Vulnerable plaques were identified using various intravascular imaging techniques, with most being guided solely by intravascular ultrasound. The study found that preventive stenting resulted in a statistically significant reduction in the rate of TVF at 2 years of follow-up (0.4% vs 3.4%; P = .0003).11
Finally, several observational trials have demonstrated that OCT is an effective method for detecting vulnerable plaques and monitoring the response to intensive treatments aimed at stabilizing these plaques through fibrous cap thickening.18,20 The PECTUS-obs trial included 438 acute coronary syndrome patients with nonculprit lesions with FFR > 0.80 treated with the OMT alone.10 All lesions were examined using OCT, with criteria similar to those used in the VULNERABLE trial to define vulnerable plaques. In that study, 34% of patients had at least 1 vulnerable lesion, which was associated with a higher risk of adverse events (15.4% vs 8.2% for the composite endpoint of death, myocardial infarction, or revascularization in the groups with and without vulnerable plaques, respectively). The VULNERABLE trial is the first to use OCT to guide the treatment of vulnerable plaques in functionally nonsignificant lesions.
CONCLUSIONS
The VULNERABLE trial aims to evaluate the effectiveness of preventive stenting plus OMT vs OMT alone for vulnerable plaques, as defined by OCT, in functionally nonsignificant intermediate lesions in nonculprit vessels of patients with STEMI. In addition, the study will provide information on the clinical relevance of the presence of vulnerable plaques in nonculprit lesions.
FUNDING
This study has been funded by Abbott Vascular.
ETHICAL CONSIDERATIONS
The study is being conducted following the recommendations outlined in the Declaration of Helsinki on clinical research, has been approved by Hospital Universitari de Bellvitge research ethics committee, and endorsed by the remaining ethics committees of participating centers. Informed consent acceptance and signature are required prior to performing any elective procedures to study the nonculprit lesion. Potential sex and gender biases are considered.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
No artificial intelligence was used in the drafting of this manuscript.
AUTHORS’ CONTRIBUTIONS
J. Gómez-Lara and E. Gutiérrez-Ibañes drafted this document. The remaining signatories reviewed the document, made changes at their discretion, and approved the final text.
CONFLICTS OF INTEREST
J. Gómez-Lara and E. Gutiérrez-Ibañes received a grant from Abbott Vascular for this study. A. Jurado-Román has received fees from Abbott, Boston, and Shockwave. E. Fernández received fees from Abbott and Hexacath. C. Cortés received a Río Hortega Contract from Instituto de Salud Carlos III. S. Brugaletta received fees from Abbott, Microport, and General Electric. T. García-Camarero received fees from Medtronic and Boston. J.A. Linares Vicente received fees from Abbott Vascular, Braun, AstraZeneca, Bayer, and IZASA. O. Rodríguez-Leor received fees from Shockwave, WorlsMedica, and Medtronic. S. Ojeda received fees from Abbott, Boston, WorldMedica, and Biosensors. A. Pérez de Prado received grants and fees from Abbot, Boston, iVascular, and Terumo. H.M. García-García received fees from ACIST, Boston Scientific, Medis, Biotronik, InfraRedx/Nipro, Chiesi, and Cordis. S. Ojeda and A. Pérez de Prado are associate editors of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial processing of the manuscript has been followed. The remaining authors declared no conflicts of interest whatsoever.
WHAT IS KNOWN ABOUT THE TOPIC?
- Thin-cap fibroatheromas, also known as vulnerable plaques, are responsible for most acute coronary syndromes. Approximately 50% of patients with STEMI have additional angiographic lesions beyond the culprit lesion, which are associated with a significant number of adverse ischemic events. Preventive stenting for severe nonculprit lesions (DS ≥ 70%) has been shown to reduce the number of adverse events. However, the effectiveness of preventive stenting for angiographically intermediate nonculprit lesions (SD, 40%-69%) that have characteristics of vulnerable plaques remains to be determined.
WHAT DOES THIS STUDY ADD?
- VULNERABLE is the first randomized trial to evaluate the preventive treatment of angiographically intermediate, nonculprit lesions that exhibit features of vulnerability identified by OCT in patients with STEMI.
REFERENCES
1. Park DW, Clare RM, Schulte PJ, et al. Extent, location, and clinical significance of non-infarct-related coronary artery disease among patients with ST-elevation myocardial infarction. JAMA 2014;312:2019-2027.
2. Mehta SR, Wood DA, Storey RF, et al. Complete Revascularization with Multivessel PCI for Myocardial Infarction. N Engl J Med. 2019;381:1411-1421.
3. Lee JM, Kim HK, Park KH, et al. Fractional flow reserve versus angiography-guided strategy in acute myocardial infarction with multivessel disease:a randomized trial. Eur Heart J. 2023;44:473-484.
4. Puymirat E, Cayla G, Simon T, et al. Multivessel PCI Guided by FFR or Angiography for Myocardial Infarction. N Engl J Med. 2021;385:297-308.
5. Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44:3720-3826.
6. Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization:Executive Summary:A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:4-17.
7. Denormandie P, Simon T, Cayla G, et al. Compared Outcomes of ST-Elevation Myocardial Infarction Patients with Multivessel Disease Treated with Primary Percutaneous Coronary Intervention and Preserved Fractional Flow Reserve of Non-Culprit Lesions Treated Conservatively and of Those with Low Fractional Flow Reserve Managed Invasively:Insights from the FLOWER MI trial. Circ Cardiovasc Interv. 2021;14:011314.
8. Stone GW, Maehara A, Lansky AJ, et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364:226-235.
9. Erlinge D, Maehara A, Ben-Yehuda O, et al. Identification of vulnerable plaques and patients by intracoronary near-infrared spectroscopy and ultrasound (PROSPECT II):a prospective natural history study. Lancet. 2021;397:985-995.
10. Mol JQ, Volleberg R, Belkacemi A, et al. Fractional Flow Reserve-Negative High-Risk Plaques and Clinical Outcomes After Myocardial Infarction. JAMA Cardiol. 2023;8:1013-1021.
11. Park SJ, Ahn JM, Kang DY, et al. Preventive percutaneous coronary intervention versus optimal medical therapy alone for the treatment of vulnerable atherosclerotic coronary plaques (PREVENT):a multicentre, open-label, randomised controlled trial. Lancet. 2024;403:1753-1765.
12. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials:a case for standardized definitions. Circulation. 2007;115:2344-2351.
13. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40:237-269.
14. Virmani R. Pathology of the vulnerable plaque. J Am Coll Cardiol. 2006;47:C13-8.
15. Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med. 2004;350:1495-1504.
16. Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N Engl J Med. 2018;379:2097-2107.
17. Tardif JC, Kouz S, Waters DD, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019;381:2497-2505.
18. Raber L, Ueki Y, Otsuka T, et al. Effect of Alirocumab Added to High-Intensity Statin Therapy on Coronary Atherosclerosis in Patients With Acute Myocardial Infarction:The PACMAN-AMI Randomized Clinical Trial. JAMA. 2022;327:1771-1781.
19. Biccire FG, Haner J, Losdat S, et al. Concomitant Coronary Atheroma Regression and Stabilization in Response to Lipid-Lowering Therapy. J Am Coll Cardiol. 2023;82:1737-1747.
20. Kedhi E, Berta B, Roleder T, et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve:the COMBINE OCT-FFR trial. Eur Heart J. 2021;42:4671-4679.
- Incidence and predictors of radial artery occlusion following transradial coronary procedures
- Distal radial access for coronary procedures in an all-comer population: the first 1000 patients in a prospective cohort
- Safety and efficacy of the Essential Pro paclitaxel drug-eluting balloon for the treatment of coronary in-stent restenosis
- Percutaneous treatment of the left main coronary artery in older adults. Impact of frailty on mid-term results
Editorials
Expanding the role of drug-coated balloons in native large coronary artery disease
aDepartment of Biomedical Sciences, Humanitas University, Pieve Emanuele, Milan, Italy
bCardio Center, IRCCS Humanitas Research Hospital, Rozzano, Milan, Italy
cDivision of Cardiology, Montefiore Medical Center, Bronx, New York, United States
Original articles
Editorials
The role of angiography-derived physiological assessment techniques in the post-FAVOR III Europe era?
aServicio de Cardiología, Hospital General Universitario Gregorio Marañón, Instituto de Investigación Sanitaria Gregorio Marañón, Universidad Complutense de Madrid, Madrid, Spain
bCentro de Investigación Biomédica en Red de Enfermedades Cardiovasculares (CIBERCV), Instituto de Salud Carlos III, Madrid, Spain
Original articles
Review Articles
Interviews
An interview with Camino Bañuelos
aServicio de Cardiología, Hospital Clínico San Carlos, Instituto de Investigación Sanitaria del Hospital Clínico San Carlos (IdISSC), Madrid, Spain
bServicio de Cardiología, Hospital Clínico Universitario de Santiago de Compostela, Santiago de Compostela, A Coruña, Spain