ABSTRACT
Introduction and objectives: The ROLLER COASTR-EPIC22 trial randomized 171 patients with moderate-to-severe calcified coronary lesions to undergo percutaneous coronary intervention with rotational atherectomy, intravascular lithotripsy, or excimer laser. This trial and its 1-year follow-up demonstrated no significant differences among the 3 arms in terms of procedural success, complications and clinical outcomes. This subanalysis aimed to describe age-related outcomes among patients undergoing percutaneous coronary intervention with plaque-modification techniques.
Methods: Of 171 patients (118 < 75 years; 53 ≥ 75 years), we compared final stent expansion by optical coherence tomography using an intention-to-treat analysis. Secondary endpoints included minimum stent area, angiographic, procedural, and clinical success, in-hospital complications, 1-year all-cause mortality, and major adverse cardiovascular events (MACE), including cardiac death, target vessel myocardial infarction, target lesion revascularization, and stent thrombosis.
Results: Baseline characteristics and treatment allocation were similar among groups. Final stent expansion was 85 ± 17.1 % in patients < 75 vs 84 ± 18.9 % in those ≥ 75 (P = .76). Minimum stent area, procedural success, and in-hospital complications were comparable. At 1 year, the ≥ 75 cohort had significantly higher MACE (11.3% vs 1.7%; P = .01) and all-cause mortality (9.4% vs 0.9%; P = .01).
Conclusions: Elderly patients achieved similar procedural outcomes as younger patients, but higher rates of MACE and all-cause mortality at 1-year follow-up.
Keywords: Rotational atherectomy. Intravascular lithotripsy. Excimer laser coronary angioplasty. Coronary calcification. Optical coherence tomography. Elderly patients.
RESUMEN
Introducción y objetivos: En el ensayo ROLLER COASTR-EPIC22 se aleatorizó a 171 pacientes con lesiones coronarias con calcificación moderada a grave para ser tratados con intervención coronaria percutánea mediante aterectomía rotacional, litotricia intravascular o láser de excímeros (ELCA). El ensayo fundamental y su seguimiento a 1 año demostraron que no había diferencias significativas entre los 3 grupos en cuanto a éxito del procedimiento, complicaciones y eventos clínicos. Este subanálisis tuvo como objetivo describir los resultados estratificados por edad en pacientes sometidos a intervención coronaria percutánea con técnicas de modificación de placa.
Métodos: En un total de 171 pacientes (118 < 75 años y 53 ≥ 75 años) se comparó la expansión final del stent mediante tomografía de coherencia óptica utilizando un análisis por intención de tratar. Los objetivos secundarios fueron el área mínima del stent, el éxito angiográfico, de procedimiento y clínico, las complicaciones intrahospitalarias, la mortalidad por cualquier causa al año y los eventos cardiovasculares adversos mayores (MACE), incluida la muerte de causa cardiovascular, el infarto de miocardio del vaso tratado, la revascularización de la lesión tratada y la trombosis del stent.
Resultados: Las características basales y la asignación del tratamiento fueron similares entre los grupos. La expansión final del stent fue del 85 ± 17,1% en los pacientes < 75 años y del 84 ± 18,9% en aquellos ≥ 75 años (p = 0,76). Al año, la cohorte ≥ 75 años presentó una tasa de MACE significativamente más alta (11,3 frente a 1,7%; p = 0,01), así como de mortalidad por todas las causas (9,4 frente a 0,85%; p = 0,01).
Conclusiones: Los pacientes ancianos lograron resultados procedimentales similares a los más jóvenes, pero con tasas más altas de MACE y de mortalidad por cualquier causa en el seguimiento a 1 año.
Palabras clave: Aterectomía rotacional. Litotricia intravascular. Aterectomía coronaria con láser de excímeros. Calcificación coronaria. Tomografía de coherencia óptica. Pacientes ancianos.
Abbreviations
ELCA: excimer laser coronary atherectomy. IVL: intravascular lithotripsy. MACE: major adverse cardiovascular events. OCT: optical coherence tomography. PCI: percutaneous coronary intervention. PMT: plaque modification technique.
INTRODUCTION
Coronary artery calcification constitutes a major challenge during percutaneous coronary intervention (PCI), as it reduces procedural success and increases the risk of complications. It limits optimal stent expansion, which predicts restenosis, stent thrombosis, and the need of target lesion revascularization.1-3 Several plaque modification techniques (PMT) have been developed to improve clinical outcomes in this setting. Rotational atherectomy has long been considered the standard approach for managing resistant calcified lesions. However, the development of newer technologies such as intravascular lithotripsy (IVL), together with increasing interest in excimer laser coronary atherectomy (ELCA), has expanded the range of available therapeutic options.4-7
The ROLLER COASTR-EPIC22 trial was the first randomized trial comparing rotational atherectomy, IVL, and ELCA in patients with moderate-to-severe calcified coronary lesions. Procedural success, safety and 1-year clinical outcomes were similar across all techniques.8
Elderly patients (≥ 75 years), who often present with increased comorbidity and complex coronary anatomy, remain underrepresented in clinical trials and may experience higher procedural risk and higher long-term mortality.9 In this post-hoc analysis of the ROLLER COASTR-EPIC22 trial, we assessed the impact of age on outcomes of PCI for moderately to severely calcified coronary stenosis treated with contemporary plaque-modification techniques. Specifically, we compared stent expansion, procedural success, in-hospital complications and 1-year clinical outcomes between patients < 75 and ≥ 75 years of age. Exploratory analyses of individual PMT within age strata were performed but were not powered for formal between-technique comparisons.
METHODS
Study design and population
ROLLER COASTR-EPIC22 trial was a multicenter randomized trial (NCT04181268) conducted at 8 high-volume centers in Spain.10 Complete inclusion and exclusion criteria, as well as the randomization process and procedural details, have been previously described.11 Briefly, eligible patients had moderate-to-severe calcification estimated by coronary angiography, and culprit lesions of ST-segment elevation acute coronary syndromes were excluded. All patients gave therir prior written informed consent before participation. Included patients were randomized 1:1:1 to rotational atherectomy, IVL, or ELCA from July 2019 through December 2023.10,11
Stratification and endpoints
This study is a post-hoc age-stratified analysis. Patients were stratified by age in 2 groups: < 75 years (n = 118) and ≥ 75 years (n = 53). Comparisons within age subgroups are exploratory and should be viewed as descriptive rather than confirmatory.
The primary endpoint was stent expansion assessed by optical coherence tomography (OCT), calculated as minimal stent area divided by the mean reference lumen area, analyzed according to the intention-to-treat principle.11
The secondary endpoints included angiographic success (final Thrombolysis in Myocardial Infarction grade-3 flow, assessed at both < 20% and < 30% residual stenosis), procedural success (angiographic success without major perioperative complications), clinical success (procedural success without in-hospital major adverse cardiovascular events [MACE]), device success (defined as achievement of adequate plaque modification with the initial device without the need for a second advanced PMT), minimal stent area by OCT, and in-hospital complications.11
Major perioperative complications were defined as death, perforation, flow-limiting dissection, abrupt vessel closure, or stent thrombosis. MACE was defined as the composite of cardiac death, target vessel myocardial infarction, target lesion revascularization, and definite/probable stent thrombosis.11
Procedures and imaging
Procedures have been previously described and followed latest European guideline recommendations.10 Pre- and post-PMT OCT imaging were recommended to characterize the lesion. Predilation with low-profile balloons was permitted to allow OCT catheter crossing prior to PMT. After plaque modification, drug-eluting stent implantation and optimization were performed, followed by the final mandatory OCT. OCT image acquisition and stent optimization protocols have been previously described.11
Rotational atherectomy, IVL and ELCA were performed according to current recommendations and device instructions, as previously described.6,12,13
Statistical analysis
The principal analyses were conducted in the intention-to-treat population. An additional analysis was performed in an as-treated fashion, based on the last PMT used. Continuous variables are expressed as mean ± standard deviation if normally distributed or median [IQR] otherwise, and categorical variables as counts and percentages. Between-group comparisons used the chi-square test or Fisher’s exact test for categorical variables and Student t test or Mann–Whitney U test for continuous variables. Noninferiority of IVL and ELCA vs rotational atherectomy for stent expansion was tested with a 1-sided t test against a prespecified noninferiority margin of −7%. This margin was adopted from the pivotal ROLLER COASTR-EPIC22 trial and was defined a priori by the steering committee as the smallest absolute difference in stent expansion considered clinically relevant, based on previous studies.11,14,15 Statistical significance was set at P < .05.10,11
Because this was an exploratory, post-hoc age-stratified analysis with a relatively small sample size in the elderly subgroup, no formal adjustment for multiple comparisons was applied. Given the large number of comparisons (2 age groups, 3 PMT and several procedural and clinical endpoints), conservative corrections such as Bonferroni would have markedly reduced statistical power and increased the risk of type II error. Instead, all effect estimates, 95% confidence intervals and exact P values are reported to allow readers to interpret the results with appropriate caution, and all subgroup findings are considered hypothesis-generating rather than confirmatory.
Given the low number of events, survival was assessed with Kaplan–Meier curves and log-rank tests. All statistical analyses were conducted using R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Patient allocation and baseline characteristics
A total of 171 patients were enrolled, of whom 118 (69%) were younger than 75 years (mean age, 66.8 ± 6.4 years; 79.5% male) and 53 (31%) were aged ≥ 75 years (mean age, 79.7 ± 3.7 years; 73.6% male). In patients < 75 years (n = 118), age was 66.8 ± 6.4 years (median, 68; IQR 64–71; range, 44–74), whereas in those ≥ 75 years (n = 53) age was 79.7 ± 3.7 years (median, 79; IQR 77–82; range, 75–89) (table S1 and figure S1).
Baseline clinical characteristics were broadly comparable between age groups, including rates of diabetes, hypertension, and prior revascularization. However, older patients had a lower prevalence of active smoking (15.1% vs 33.6%; P = .02), lower body mass index (27.0 ± 3.8 vs 28.5 ± 4.3 kg/m²; P = .04), and lower haemoglobin (12.8 ± 1.8 vs 13.5 ± 2.0 g/L; P = .04). No significant differences were observed in left ventricular ejection fraction, basal creatinine or clinical presentation, although elderly patients showed a trend towards a more frequent presentation as acute coronary syndrome (table 1).
Table 1. Baseline clinical characteristics
| Variable | < 75 years (n = 118) | ≥ 75 years (n = 53) | P |
|---|---|---|---|
| Age | 66.8 ± 6.4 | 79.7 ± 3.7 | < .01 |
| Male | 93 (79.5) | 39 (73.6) | .51 |
| BMI (kg/m2) | 28.5 ± 4.3 | 27.0 ± 3.8 | .04 |
| Current smoker | 39 (33.1) | 8 (15.1) | .02 |
| Diabetes mellitus | 57 (48.3) | 27 (50.9) | 92 |
| Hypertension | 93 (78.8) | 43 (81.1) | .97 |
| Dyslipidemia | 86 (72.9) | 40 (75.5) | .93 |
| Prior PCI | 40 (33.9) | 16 (30.2) | .73 |
| Prior CABG | 4 (3.4) | 2 (3.8) | 1 |
| LVEF | 54.8 ± 10.9 | 54.9 ± 10.7 | .96 |
| Serum creatinine (mg/dL) | 1.2 ± 1.2 | 1.2 ± 1.0 | .95 |
| Hemoglobin (g/L) | 13.5 ± 2.0 | 12.8 ± 1.8 | .04 |
| Clinical presentation | .08 | ||
| CCS | 77 (65.3) | 29 (54.7) | |
| Unstable angina | 12 (10.2) | 10 (18.8) | |
| NSTEMI | 18 (15.2) | 12 (22.6) | |
| STEMI | 8 (6.8) | 0 (0) | |
|
BMI, body mass index; CABG, coronary artery bypass graft; CCS, chronic coronary syndrome; LVEF, left ventricular ejection fraction; NSTEMI, non-ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction. Data are expressed as No. (%) or mean ± standard deviation. All angiographic parameters were measured at a central core laboratory blinded to the treatment arm. |
|||
Angiographic and procedural data
Target vessels and lesion characteristics, including severity and length of calcification, were comparable across age strata and treatment arms. Most procedures were performed via radial access (> 80%), and treatment allocation was well balanced across age strata. Procedural duration, sheath size, and use of ad hoc PCI did not differ by age. Angiographic and procedural details are summarized in table S2.
OCT findings
The optional pre-plaque modification OCT was performed in 39 of 118 patients < 75 years (33.1%) and in 20 of 53 patients ≥ 75 years (37.7%), with no significant difference between age groups (P = .67).
Final OCT runs were obtained in more than 75% of patients. OCT parameters of calcification, assessed by an independent central core laboratory, consistently demonstrated a heavy calcium burden across all cases, with no significant differences between age groups or treatment modalities. OCT findings are presented in table S3.
Primary and secondary endpoints
No significant differences in terms of final stent expansion, minimal stent area, or final residual stenosis were observed between groups (table 2, figure S2). Angiographic, procedural, and clinical success rates exceeded 90% (considering < 30% residual percent diameter stenosis) in both age groups and did not differ by technique (table 2, figure 1).
Table 2. Procedural outcomes and complications
| Procedural outcomes | < 75 years (n = 118) | ≥ 75 years (n = 53) | P |
|---|---|---|---|
| Success considering < 30% residual percent diameter stenosis | |||
| Angiographic | 112 (94.9%) | 49 (92.5%) | .78 |
| Procedural | 108 (91.5%) | 49 (92.5%) | 1.00 |
| Clinical | 108 (91.5%) | 48 (90.6%) | 1.00 |
| Success considering < 20% residual percent diameter stenosis | |||
| Angiographic | 87 (73.7) | 42 (79.2) | .56 |
| Procedural | 83 (70.3) | 42 (79.2) | .30 |
| Clinical | 83 (70.3) | 41 (77.4) | .44 |
| Final TIMI grade flow | .63 | ||
| 0 | 1 (0.8) | 0 | |
| 1 | 0 | 0 | |
| 2 | 1 (0.8) | 0 | |
| 3 | 116 (98.3) | 53 (100) | |
| Final diameter diameter stenosis (%) | 14.7 ± 11.5 | 12.8 ± 10.9 | .30 |
| Final SE (%) | 85.0 ± 17.1 | 84.0 ± 18.9 | .76 |
| Severe procedural complications | 4 (3.4) | 0 (0) | .42 |
| Death | 0 (0) | 0 (0) | – |
| Perforation | 4 (3.4) | 0 (0) | .42 |
| Flow-limiting dissection | 0 (0) | 0 (0) | – |
| Abrupt vessel closure | 0 (0) | 0 (0) | – |
| Stent thrombosis | 0 (0) | 0 (0) | – |
| Dissection | |||
| A | 3 (2.5) | 1 (1-9) | 1.00 |
| B | 0 (0) | 2 (3.8) | .18 |
| D | 1 (0.8) | 1 (1.9) | 1.00 |
| Slow flow/No-reflow | 1 (0.8) | 1 (1.9) | 1.00 |
| Side branch occlusion | 0 (0) | 1 (1.9) | .68 |
| Complications during admission | 0 (0) | 2 (3.8) | .18 |
| Mortality | 0 (0) | 1 (1.9) | .68 |
| Cardiac death | 0 (0) | 1 (1.9) | .68 |
| Target lesion revascularization | 0 (0) | 0 (0) | – |
| Stent thrombosis | 0 (0) | 0 (0) | – |
| Stroke | 0 (0) | 0 (0) | – |
| BARC grade ≥ 3b bleeding | 0 (0) | 1 (1.9) | .68 |
|
BARC, Bleeding Academic Research Consortium; SE, stent expansion; TIMI, Thrombolysis in Myocardial Infarction. Data are expressed as No. (%) or mean ± standard deviation. Severe procedural complications were defined as death, perforation, flow-limiting dissection, abrupt vessel closure or stent thrombosis. |
|||
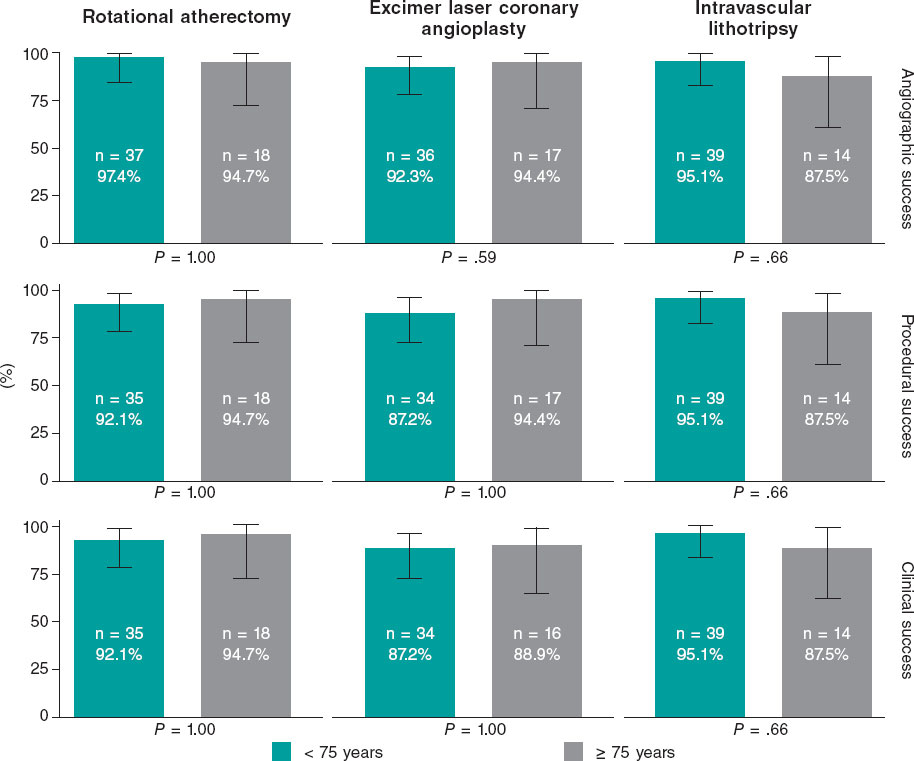
Figure 1. Angiographic, procedural, and clinical success of each technique. Angiographic success defined as final Thrombolysis in Myocardial Infarction grade-3 flow with < 30% residual percent diameter stenosis. Procedural success was considered angiographic success without major perioperative complications. Clinical success was defined as procedural success without in-hospital major adverse cardiovascular events.
Among patients ≥ 75 years, mean stent expansion values were broadly similar across the 3 PMT, and no statistically significant between-technique differences were detected; however, 95%CI, were wide because of the small sample size in each subgroup (table S3). Exploratory analyses of mean differences in stent expansion between techniques yielded results that were directionally consistent with the overall ROLLER COASTR-EPIC22 trial but were clearly underpowered in this age-stratified cohort and should be interpreted as hypothesis-generating only (figure 2 and table S4).
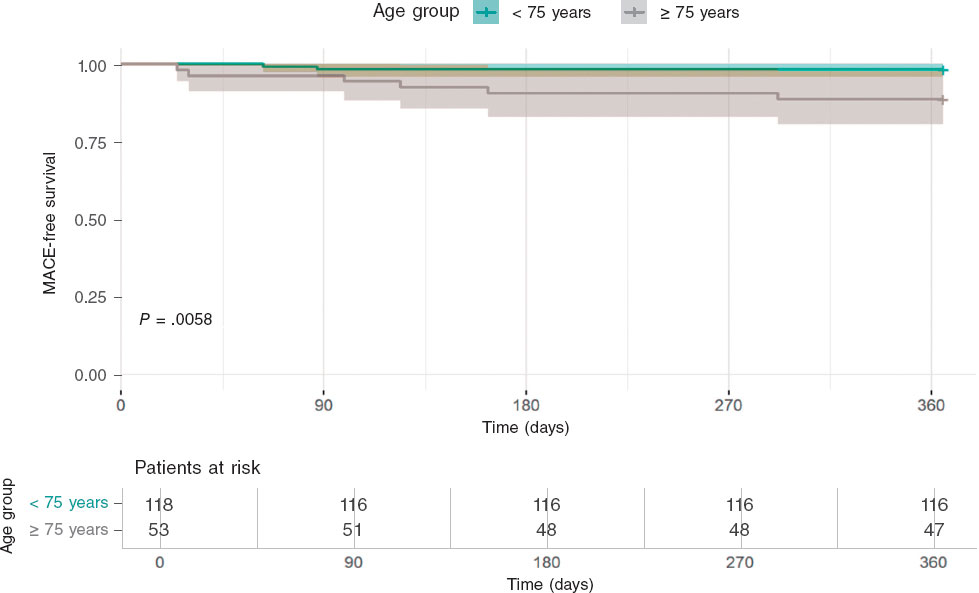
Figure 2. One-year MACE-free survival. Kaplan-Meier curves showing 1-year freedom from MACE in the intention to-treat population. MACE was defined as the composide endpoing of cardiac death, target vessel myocardial infarction, target lesion revascularization, stent thrombosis. MACE, major adverse cardiovascular events.
Overall, device success rate was 88.3% overall and was similar by age: < 75 (89.7%) vs ≥ 75 years (85.7%); P = .76. The causes of crossover differed by device: as in the pivotal trial, in the rotational atherectomy arm most crossovers were prompted by inadequate lesion dilation after atherectomy, whereas in the IVL and ELCA arms the primary driver was inability to cross the lesion with the dedicated balloon or laser catheter (figure S3 and table S5). In-hospital complication rates (perforations, bleeding, death) were low and balanced by age (table 2).
In 1-year follow-up (table 3, figure 2), patients aged ≥ 75 years had a markedly higher rate of MACE than those < 75 years (11.3% vs 1.7%; P = .01). All-cause mortality was also greater in the older cohort (9.4% vs 0.9%; P = .01).
Table 3. One-year event rates
| Event | < 75 years | ≥ 75 years | P |
|---|---|---|---|
| MACE | 2 (1.7%) | 6 (11.3%) | .01 |
| All-cause mortality | 1 (0.9%) | 5 (9.4%) | .01 |
| Cardiac death | 0 (0.00%) | 2 (3.8%) | .10 |
| TVMI | 0 (0.00%) | 2 (3.8%) | .10 |
| TLR | 1 (0.9%) | 2 (3.8%) | .23 |
| TVR | 2 (1.7%) | 4 (7.6%) | .08 |
| ST | 0 (0.00%) | 2 (3.8%) | .10 |
|
MACE, major adverse cardiovascular events; ST, stent thrombosis; TLR, target lesion revascularization; TVMI, target vessel myocardial infarction; TVR, target vessel revascularization. Data are expressed as No. (%). MACE was defined as a composite endpoint of cardiac death, target vessel acute myocardial infarction, target lesion revascularization, and stent thrombosis. P values have been calculated as log-rank p-values from Kaplan-Meier comparisons. |
|||
Although less frequent, each individual component of the composite endpoint was numerically higher in patients ≥ 75 years without reaching statistical significance: cardiac death (11.3% vs 0%; P = .1), target vessel myocardial infarction (3.8% vs 0%; P = .1), target lesion revascularization (3.8% vs 0.8%; P = .23), target vessel revascularization (7.5% vs 1.7%; P = .08) and stent thrombosis (3.8% vs 0%; P = .10).
In a multivariable logistic regression including age ≥ 75 years, anemia, acute coronary syndrome presentation and renal dysfunction, age ≥ 75 years remained the strongest predictor of 1-year endpoints. For MACE, age ≥ 75 years was associated with an adjusted OR, 7.4 (95%CI, 1.5–53.9; P = .02), whereas none of the other covariates showed a clear independent association with the composite endpoint. For all-cause mortality, the adjusted OR for age ≥ 75 years was 10.7 (95%CI, 1.5–218.7; P = .04) and none of the other covariates demonstrated a clear independent effect. These findings are presented in table S6 and figure S4.
DISCUSSION
In this post-hoc age-stratified analysis of the ROLLER COASTR-EPIC22 trial and its 1-year follow-up, we observed that the overall safety and efficacy of PMT appeared consistent across age groups. However, at 1 year older patients experienced significantly higher rates of MACE (11.3% vs 1.7%; P = .01) and all-cause mortality (9.4% vs 0.9%; P = .01) despite comparable in-hospital complication rates.
After adjustment for anemia, acute coronary syndrome presentation, and renal dysfunction, age ≥ 75 years remained associated with higher adjusted odds of both 1-year MACE and all-cause mortality. None of the adjustment covariates demonstrated a clear independent effect, suggesting that unmeasured factors closely related to aging, such as frailty, are likely to account for much of the excess risk observed in older patients.
Randomized evidence directly comparing contemporary PMT in calcified coronary disease remains scarce. The ROLLER COASTR-EPIC22 trial was the first trial to randomize patients among rotational atherectomy, IVL, and ELCA.10 Previous device-specific studies and registries have demonstrated the feasibility of each technique, but none had evaluated all 3 within a single randomized framework.16-18
Coronary calcium is independently associated with long-term mortality risk, and age is not only a key predictor of coronary calcification but also an independent predictor of MACE, regardless of calcium burden.19 Nevertheless, elderly patients are underrepresented in PMT studies, leaving limited evidence to guide the choice of the optimal PMT strategy. Regarding rotational atherectomy, there is little evidence in elderly patients, but retrospective data suggests similar success and in-hospital complication rates compared with younger individuals.20 Similarly, the BENELUX-IVL prospective registry demonstrated the safety profile and technical success of IVL in patients aged ≥ 80 years.21 ELCA, however, has not been specifically studied in the elderly population.
Selecting elderly patients for revascularization requires balancing benefits and risks. Comorbidities (anemia, chronic kidney disease, acute coronary syndrome) and limitations in physiological reserve increase complexity and may reduce revascularization uptake in this population.22,23 Our findings support that chronological age alone should not preclude revascularization, as PMT appeared effective and safe when performed by experienced operators, regardless of age. These results, however, should be interpreted cautiously given the limited sample size and exploratory, post-hoc nature of the age-stratified analysis. The importance of operator experience in PMT for complex scenarios has been highlighted by the CRATER trial program, which evaluated rotational atherectomy in patients with chronic kidney disease and severely calcified lesions and reported high procedural success but also substantial perioperative and long-term event rates, largely reflecting the extreme baseline risk of this population.24,25
Notably, our analysis suggests no excess perioperative complications in older adults, although the study was not powered for subgroup comparisons and 95%CI, were wide, particularly in the elderly ELCA subgroup. We advocate for meticulous patient selection, based on preoperative risks and comorbidities; however, PMT should be used when necessary for adequate lesion preparation to maximize stent expansion and avoid suboptimal implantation, a known driver of adverse clinical events.
In our cohort, elderly patients achieved similar procedural success, stent expansion and in-hospital safety outcomes vs younger patients yet experienced a markedly higher rate of 1-year MACE and all- cause mortality. This pattern is consistent with the e-ULTIMASTER and MOSCA FRAIL trials, in which frailty, rather than age per se, emerged as the main determinant of adverse outcomes in older adults undergoing PCI.26,27 Taken together, these data support that revascularization decisions in elderly patients should not rely exclusively on chronological age, but should incorporate assessment of frailty, functional capacity and comorbidity to individualize the benefit–risk balance of an invasive strategy. The lack of formal frailty indices in our study therefore represents an important limitation. Future trials in elderly patients undergoing complex PCI should prospectively integrate standardized frailty tools to better guide patient selection and tailor treatment.
In the subset of patients with available baseline OCT, we did not observe relevant differences in calcium burden or morphology nor in angiographic severity or extent of disease, between patients < 75 and ≥ 75 years. This suggests that the underlying calcific process is biologically similar across age groups, even if it becomes clinically manifest at different ages, and is consistent with the comparable postoperative stent expansion and minimal stent area observed in both strata. Nevertheless, despite similar stent optimization, the ≥ 75-year group showed a numerically higher rate of definite/probable stent thrombosis and cardiovascular death at 1 year (3.8%), which is clinically meaningful in this context. As underexpansion alone is unlikely to fully explain these events, age-related factors such as greater comorbidity and frailty, a prothrombotic milieu and a reduced myocardial reserve, may have contributed to the excess risk observed in older patients, even after apparently adequate angiographic and OCT-guided results.
Contemporary randomized PMT trials (such as ROTAXUS, PREPARE-CALC, ISAR-CALC 2 or ECLIPSE ) enrolled patients with a mean age around 70 years, but none of them reported age-specific outcomes for those ≥ 75 years, limiting its applicability to older populations.28-32 Our analysis contributes to fill this knowledge gap suggesting procedural success and safety of PMT in the elderly. However, due to the nonprespecified nature of our analysis, these results should be considered hypothesis-generating. Future randomized studies should integrate objective frailty and functional measures and include sufficient follow-up to assess both procedural and long-term outcomes in this growing population.
Study limitations
This study has several limitations to consider. First, it was a post-hoc analysis and the findings should be regarded as hypothesis- generating rather than definitive. Second, because age stratification was not prespecified in the trial design, the study was not statistically powered for subgroup comparisons based on age, and the relatively small number of patients aged ≥ 75 years may limit the generalizability of the results.
Third, frailty status and functional capacity –both known to strongly influence outcomes in elderly patients– were not assessed, which limits the clinical granularity of the findings.
Fourth, the study was not powered to detect differences in clinical events and results should be interpreted cautiously due to the limited number of events.
Fifth, posttreatment OCT images were not available in slightly more than 20% of patients, with a higher proportion of missing studies in the ≥ 75-year group. This pattern of missing data may introduce selection bias, as patients without analyzable postoperative OCT could differ systematically from those with complete imaging, potentially limiting the internal validity and generalizability of the stent expansion findings, particularly in the elderly subgroup.
Sixth, randomization was based on angiographic calcification alone, without prior evaluation of lesion crossability or balloon dilatability, which could have impacted the need for secondary PMT. Indeed, some patients required a second PMT, complicating the interpretation of the intention-to-treat analysis, although complementary as-treated and per-protocol analyses were performed to mitigate this limitation. Additionally, not all patients underwent complete pre- and post-PCI OCT, which may have influenced both device selection and the assessment of stent expansion metrics.
Seventh, the 1-year follow-up provides only mid-term outcome data and does not address long-term endpoints such as stent durability or the need for target lesion revascularization. Lastly, the trial was conducted in high-volume centres by experienced operators, which may limit the applicability of these results to lower-volume settings or institutions with less experience in complex PCI and PMT.
CONCLUSIONS
In this age-stratified analysis of the ROLLER COASTR-EPIC22 trial, elderly patients undergoing PCI for calcified coronary lesions achieved similar procedural success, stent expansion, and in-hospital safety outcomes as younger patients. However, patients aged ≥ 75 years experienced significantly higher rates of MACE and all-cause mortality at 1 year (figure 3).
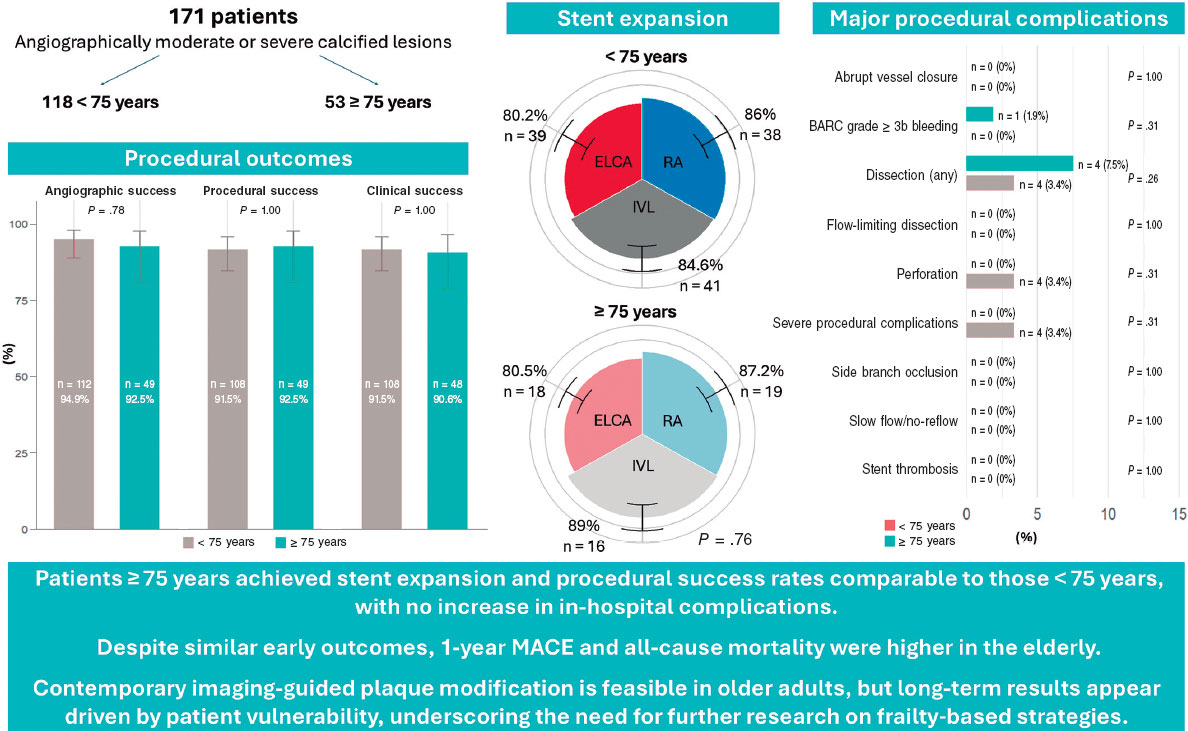
Figure 3. Central illustration. Senior-ROLLER: age-related outcomes of plaque modification in coronary stenosis. Procedural success, stent expansion, and major procedural complications were similar in patients < 75 and those ≥ 75 years; however, the 1-year MACE and all-cause mortality rates were higher in the elderly, which highlights the need for careful patient selection and individualized treatment strategies. BARC, Bleeding Academic Research Consortium; ELCA, excimer laser coronary atherectomy; IVL, intravascular lithotripsy; MACE, major adverse cardiovascular events; RA, rotational atherectomy.
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
FUNDING
The ROLLER COASTR-EPIC22 trial was supported by an unrestricted grant from Fundación EPIC.
ETHICAL CONSIDERATIONS
This is a subanalysis of the ROLLER COASTR-EPIC22 trial (NCT04181268), which was approved by the institutional ethics committee at each participant center and conducted in full compliance with the principles outlined in the Declaration of Helsinki. The authors confirm that written informed consent was obtained from all participants before enrolment.
Sex and gender aspects were considered according to the Sex and Gender Equity in Research (SAGER) guidelines.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
Artificial intelligence was not used in this manuscript.
AUTHORS’ CONTRIBUTIONS
J. Vila-García and M. Basile contributed equally to the manuscript and share first authorship. J. Vila-García and M. Basile contributed equally to several phases of the study, including conception and design, and acquisition, analysis and interpretation of the data. In addition, J. Vila-García contributed to the drafting of the original manuscript and to the writing and revision of the final version, while M. Basile was primarily responsible for the critical review of the article. A. Gómez-Menchero, J. Caballero-Borrego, B. Rivero- Santana, G. Galeote, I.J. Amat-Santos, S. Jiménez-Valero, G. Miñana, S. Ojeda, A. Gonzálvez-García, D. Tébar-Márquez, S. Camacho- Freire, J. Zubiaur, R. Ocaranza-Sánchez and A. Domínguez participated in the acquisition of the data and in the review and editing of the final text. A. Jurado-Román and R. Moreno participated in the conception and design of the study, in the acquisition, analysis and interpretation of the data, in the drafting, review, editing and approval of the final manuscript, and in the overall supervision of the project. A. Jurado-Román was responsible for funding and administration. All authors approved the final version of the manuscript.
CONFLICTS OF INTEREST
S. Ojeda and R. Moreno are associate editors of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. A. Jurado-Román is a proctor for Abbott, Boston Scientific, World Medica, and Philips; has received consulting fees from Boston Scientific and Philips; and has received speaker fees from Abbott, Boston Scientific, Shockwave Medical, Philips, and World Medica. S. Ojeda has received consulting fees from Medtronic and Edwards Lifesciences; and has received speaker fees from Abbott, Boston Scientific, Philips, and World Medical. All other authors report no relationships relevant to the contents of this article to disclose.
ACKNOWLEDGMENTS
We thank the ROLLER COASTR-EPIC22 trial investigators, coordinators, and participating patients for their dedication.
WHAT IS KNOWN ABOUT THE TOPIC?
- Contemporary PMT (rotational atherectomy, IVL, and ELCA) have demonstrated efficacy in managing heavily calcified coronary lesions across all age groups. However, elderly patients remain significantly underrepresented in randomized controlled trials of interventional cardiology, limiting the evidence base for clinical decision-making in this growing population. The ROLLER COASTR-EPIC22 Trial trial was the first randomized study to directly compare all 3 main PMT in a single cohort, establishing their procedural safety and efficacy profile in younger patients. Prior data suggest that age alone may not be an absolute contraindication to contemporary interventional strategies; however, age-stratified outcomes for elderly patients specifically remain sparse in the literature.
WHAT DOES THIS STUDY ADD?
- This analysis provides the first randomized age-stratified evaluation of PMT. Our findings demonstrate that procedural success and stent expansion are comparable between elderly and younger cohorts, with no excess in-hospital complications in older adults. Despite similar early procedural outcomes, elderly patients experienced significantly higher rates of MACE and all-cause mortality at 1-year follow-up, highlighting the importance of patient vulnerability and frailty over chronological age alone. These results suggest that age should not be an exclusion criterion for contemporary PMT when clinically indicated, but rather that individualized risk assessment and targeted follow-up strategies are essential for optimizing outcomes in elderly populations undergoing complex percutaneous coronary intervention.
REFERENCES
1. Généreux P, Madhavan MV, Mintz GS, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes: Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trials. J Am Coll Cardiol. 2014;63:1845-1854.
2. Ma W, Wang Q, Wang B, et al. Novel predictors of stent under-expansion regarding calcified coronary lesions assessed by optical coherence tomography. Catheter Cardiovasc Interv. 2022;99(S1):1473-1481.
3. Ng P, Maehara A, Kirtane AJ, et al. Management of Coronary Stent Underexpansion. J Am Coll Cardiol. 2025;85:625-644.
4. Shah M, Najam O, Bhindi R, Silva KD. Calcium Modification Techniques in Complex Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2021;14:E009870.
5. Barbato E, Gallinoro E, Abdel-Wahab M, et al. Management strategies for heavily calcified coronary stenoses: an EAPCI clinical consensus statement in collaboration with the EURO4C-PCR group. Eur Heart J. 2023;44:4340-4356.
6. Barbato E, CarriéD, Dardas P, et al. European expert consensus on rotational atherectomy. EuroIntervention. 2015;11:30-36.
7. Cobarro L, Jurado-Román A, Tébar-Márquez D, et al. Excimer laser coronary atherectomy in severely calcified lesions: time to bust the myth. REC Interv Cardiol. 2023;6:33-40.
8. Basile M, Gómez-Menchero A, Rivero-Santana B, et al. Rotational Atherectomy, Lithotripsy, or Laser for Calcified Coronary Stenosis: One-Year Outcomes From the ROLLER COASTER-EPIC22 Trial. Catheter Cardiovasc Interv. 2025;106:702-710.
9. Kuna C, Bradaric C, Koch T, et al. Age-related ten-year outcomes after percutaneous coronary intervention of in-stent restenosis. Int J Cardiol. 2025;428:133109.
10. Jurado-Román A, Gómez-Menchero A, Rivero-Santana B, et al. Rotational Atherectomy, Lithotripsy, or Laser for Calcified Coronary Stenosis: The ROLLER COASTR-EPIC22 Trial. JACC Cardiovasc Interv. 2025;18:606-618.
11. Jurado-Román A, Gómez-Menchero A, Amat-Santos IJ, et al. Design of the ROLLERCOASTR trial: rotational atherectomy, lithotripsy or laser for the management of calcified coronary stenosis. REC Interv Cardiol. 2023;5:279-286.
12. Rawlins J, Din JN, Talwar S, O'Kane P. Coronary Intervention with the Excimer Laser: Review of the Technology and Outcome Data. Interv Cardiol Rev. 2016;11:27.
13. Riley RF, Patel MP, Abbott JD, et al. SCAI Expert Consensus Statement on the Management of Calcified Coronary Lesions. J Soc Cardiovasc Angiogr Interv. 2024;3:101259.
14. de Waha S, Allali A, Büttner HJ, et al. Rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: Two-year clinical outcome of the randomized ROTAXUS trial. Catheter Cardiovasc Interv. 2016;87:691-700.
15. Maehara A, Ben-Yehuda O, Ali Z, et al. Comparison of Stent Expansion Guided by Optical Coherence Tomography Vs Intravascular Ultrasound: The ILUMIEN II Study (Observational Study of Optical Coherence Tomography [OCT] in Patients Undergoing Fractional Flow Reserve [FFR] and Percutaneous Coronary Intervention). JACC Cardiovasc Interv. 2015;8:1704-1714.
16. Jurado-Román A, Gonzálvez A, Galeote G, Jiménez-Valero S, Moreno R. RotaTripsy: Combination of Rotational Atherectomy and Intravascular Lithotripsy for the Treatment of Severely Calcified Lesions. JACC Cardiovasc Interv. 2019;12:e127-e129.
17. Hill JM, Kereiakes DJ, Shlofmitz RA, et al. Intravascular Lithotripsy for Treatment of Severely Calcified Coronary Artery Disease. J Am Coll Cardiol. 2020;76:2635-2646.
18. Shibui T, Tsuchiyama T, Masuda S, Nagamine S. Excimer laser coronary atherectomy prior to paclitaxel-coated balloon angioplasty for de novo coronary artery lesions. Lasers Med Sci. 2021;36:111-117.
19. McClelland RL, Chung H, Detrano R, Post W, Kronmal RA. Distribution of coronary artery calcium by race, gender, and age: Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2006;113:30-37.
20. Sharma V, Abdul F, Haider ST, et al. Rotablation in the Very Elderly –Safer than We Think?Cardiovasc Revasc Med. 2021;22:36-41.
21. Oliveri F, García PV, Oort MJH van, et al. Intravascular lithotripsy for the treatment of calcified coronary lesions in individuals of advanced age: a post-hoc analysis of the multicentre, prospective BENELUX-IVL study. eClinicalMedicine. 2025;85:103342.
22. Tisminetzky M, Miozzo R, Gore JM, et al. Trends in the magnitude of chronic conditions in patients hospitalized with a first acute myocardial infarction. J Multimorb Comorbidity. 2021;11:2633556521999570.
23. Nanna MG, Sutton NR, Kochar A, et al. Assessment and Management of Older Adults Undergoing PCI, Part 1: A JACC: Advances Expert Panel. JACC Adv. 2023;2:100389.
24. Galeote G, Zubiaur J, Jurado?Román A, et al. Coronary Rotational Atherectomy Elective Vs Bailout in Patients With Severely Calcified Lesions and Chronic Renal Failure (CRATER) Trial. Catheter Cardiovasc Interv. 2025;106:1702-1712.
25. Zubiaur J, Galeote G, Jurado-Román A, et al. Coronary Rotational ATherectomy Elective Vs Bailout in Patients With Severely Calcified Lesions and Chronic Renal Failure: Long-Term Outcomes of CRATER Trial. Catheter Cardiovasc Interv. 2026;107:431-438.
26. Saada M, Kobo O, Kauer F, et al. Prognosis of PCI in the Older Adult Population: Outcomes From the Multicenter Prospective e-ULTIMASTER Registry. J Soc Cardiovasc Angiogr Interv. 2022;1:100442.
27. Sanchis J, Bueno H, Miñana G, et al. Effect of Routine Invasive vs Conservative Strategy in Older Adults With Frailty and Non–ST-Segment Elevation Acute Myocardial Infarction: A Randomized Clinical Trial. JAMA Intern Med. 2023;183:407-415.
28. Abdel-Wahab M, Richardt G, Büttner HJ, et al. High-speed rotational atherectomy before paclitaxel-eluting stent implantation in complex calcified coronary lesions: The randomized ROTAXUS (Rotational Atherectomy Prior to Taxus Stent Treatment for Complex Native Coronary Artery Disease) trial. JACC Cardiovasc Interv. 2013;6:10-19.
29. Mankerious N, Richardt G, Allali A, et al. Lower revascularization rates after high-speed rotational atherectomy compared to modified balloons in calcified coronary lesions: 5-year outcomes of the randomized PREPARE-CALC trial. Clin Res Cardiol. 2024;113:1051-1059.
30. Rheude T, Rai H, Richardt G, et al. Super high-pressure balloon vs scoring balloon to prepare severely calcified coronary lesions: the ISAR-CALC randomised trial: Balloon-based techniques for calcific CAD. EuroIntervention. 2021;17:481.
31. Abdel-Wahab M, Toelg R, Byrne RA, et al. High-speed rotational atherectomy vs modified balloons prior to drug-eluting stent implantation in severely calcified coronary lesions: The randomized prepare-CALC trial. Circ Cardiovasc Interv. 2018;11:e007415.
32. Kirtane AJ, Généreux P, Lewis B, et al. Orbital atherectomy vs balloon angioplasty before drug-eluting stent implantation in severely calcified lesions eligible for both treatment strategies (ECLIPSE): a multicentre, open-label, randomised trial. Lancet. 2025;405:1240-1251.















