To the Editor,
Excimer laser coronary atherectomy (ELCA) is a plaque-modification technique used in the treatment of heavily calcified coronary lesions.1,2 Although recommendations favor saline infusion, contrast- enhanced ELCA is still used for resistant lesions despite higher dissection and perforation risk.3 It remains unclear whether laser activation in the presence of contrast may also lead to specific and reproducible device-failure events, as existing literature reports safety and complications without linking events to specific hardware failure.2 Therefore, we queried publicly available narratives from the United States Food and Drug Administration (US FDA) Manufacturer and User Facility Device Experience (MAUDE) database for ELCA (product code “LPC”) from 1 January 2016 through 31 May 2025. MAUDE is a passive FDA repository of device adverse events comprising mandatory reports from manufacturers and user facilities as well as voluntary reports from clinicians. We implemented a 2-stage large language model (LLM) workflow to screen narratives and extracted event-action sequences. All analyses used performed using the Gemini 2.5 Pro model with temperature = 0 and top-p = 1 to ensure deterministic outputs. In stage 1, rules-assisted screening with negation handling was used to identify candidate reports describing laser activation in the presence of contrast (eg, “contrast present,” “lased in contrast,” “before flushing”). In stage 2, the full free text of all narratives was analyzed to extract event sequences and device-outcome relationships without predefined labels. Prompts required structured outputs (presence/absence labels and an ordered action-outcome sequence) and mandated inclusion of verbatim supporting phrases from each narrative for all key labels. Two interventional cardiologists independently adjudicated all cases, with disagreements (2 of 120) resolved by consensus. We applied an explicit case definition for distal marker-band detachment. Reports were classified as “explicit detachment” only when the narrative clearly indicated that the marker band had separated, fractured, or become dislodged. “Activation in contrast” was defined as a) activation during active contrast injection, b) activation immediately after contrast delivery without documented saline flushing, or c) activation after incomplete flushingwith probable residual contrast at the catheter tip.
Within MAUDE (16 756 924 total entries across all device categories during the study period, not limited to ELCA procedures), the product-code query (“LPC”) retrieved 120 unique ELCA narratives. As a passive repositoru, MAUDE does not allow inference of incidence or causality. Distal marker-band detachment was reported in 27 of 120 narratives. Seven reports explicitly described laser activation in the presence of iodinated contrast (5.8%), of which 5 reported detachment. Across these 5 detachment narratives with confirmed contrast-time activation, the LLM identified a recurring narrative pattern: recent contrast injection or likely residual contrast at the catheter tip, laser activation before documented saline flushing (off-instructions for use [IFU]), and subsequent report of distal marker-band detachment. Among the remaining 113 narratives without explicit contrast-time activation, detachment was reported in 22. This signal is mechanistically plausible: since contrast media acts as a powerful chromophore, activating ELCA in its presence significantly amplifies local energy absorption and cavitation. This enhanced photomechanical effect, intended to modify resistant plaque, creates an omnidirectional stress wave that may inadvertently exceed the structural tolerance of the catheter’s distal tip assembly, resulting in marker-band detachment. In clinical series, the most frequently described complications of contrast-enhanced ELCA are coronary perforation and dissection, whereas this MAUDE-based analysis highlights a device-component failure—distal marker-band detachment—consistent with tip damage in the setting of incomplete saline flushing4,5 (figure 1).
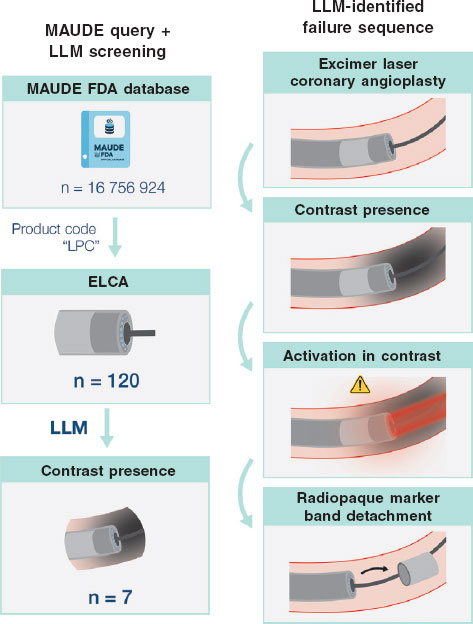
Figure 1. MAUDE query (LPC) with LLM-assisted screening identified 120 ELCA narratives (not a procedural denominator), including 7 with explicit contrast-time activation. A hypothesis-generating signal was observed, characterized by a recurring narrative pattern of recent contrast administration followed by activation before documented saline flushing, culminating in distal marker-band detachment (5 or 7 cases). ELCA, excimer laser coronary atherectomy; FDA, Food and Drug Administration; LLM, large language model; MAUDE, Manufacturer and User Facility Device Experience.
Our findings, grounded in real-world adverse-event narratives, organize these concerns into an explicit hypothesis-generating pattern that may be modifiable through technique and adherence to the saline-flush IFU recommendations. Beyond this specific mechanism, the same LLM-assisted workflow can be extended across intracoronary atherectomy modalities to systematically map associations among lesion substrate, procedural technique, and device-failure modes. Viewing each MAUDE report as a sequence of actions and consequences rather than a single coded term enables comparative signal detection and hypothesis generation for device-specific guardrails and training strategies.
The practical implications of these findings are hypothesis generating and primarily focused on awareness: a) contrast-time ELCA may be associated with distal marker-band detachment through 2 potential mechanisms: first, the chromophore effect that amplifies photomechanical shockwaves; second, the use of contrast may simply signal resistant or ‘uncrossable’ lesions requiring stationary activation, where focal energy concentration compromises the structural integrity of the catheter tip; b) recognition of this potential failure mode may enhance intraoperative vigilance and prompt systematic inspection of the catheter tip; and c) LLM-assisted analysis of MAUDE narratives can identify low-frequency, mechanism- specific device failure patterns that may inform refinement of practice, operator training, and device design.
The principal limitations of this analysis include potential underreporting inherent to the MAUDE database, narrative heterogeneity across reports, and the absence of exposure denominators, which preclude accurate estimation of event incidence. Signal detection relies on the linguistic structure of individual reports, raising the possibility of misclassification, particularly when key procedural details are not documented. Moreover, the LLM-derived event sequences reflect associative rather than causal relationships. MAUDE reports are unverified and narratives may be incomplete or biased by reporting from clinicians or manufacturers.
In conclusion, LLM-assisted analysis of MAUDE narratives revealed a consistent, small signal linking contrast-time laser activation to device-specific failure, most notably distal marker-band detachment. This observation is hypothesis-generating and warrants vaildation in prospective registries and, ideally, bench testing. This workflow illustrates how scalable LLM pipelines can transform passive adverse-event repositories into comparative, technology-agnostic maps of device-specific risk.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
This study analysed publicly available, de-identified reports from the US FDA MAUDE database; therefore, ethics committee approval and informed consent were deemed unnecesary. The SAGER guidelines were considered; however, sex/gender analyses were not feasible because MAUDE narratives do not provide reliable patient-level sex or gender data for analysis.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
During manuscript preparation, the authors used a large language model (Gemini 2.5 Pro) to assist with narrative screening and sequence extraction as described in the manuscript, and minor language edits. All outputs were reviewed and edited by the authors, who take full responsibility for the content.
AUTHORS’ CONTRIBUTIONS
A. Fernández-Cisnal: conceptualization, clinical adjudication, MAUDE query, methodology (LLM workflow), data analysis, drafting of the manuscript. A. Jurado-Román: interpretation, critical revision of the manuscript. G. Miñana: clinical adjudication, interpretation, critical revision of the manuscript. All authors approved the final version.
CONFLICTS OF INTEREST
None declared.
REFERENCES
1. Golino L, Caiazzo G, CalabròP, et al. Excimer laser technology in percutaneous coronary interventions:Cardiovascular laser society's position paper. Int J Cardiol. 2022;350:19-26.
2. Cobarro L, Jurado-Román A, Tébar-Márquez D, et al. Excimer laser coronary atherectomy in severely calcified lesions:time to bust the myth. REC Interv Cardiol. 2023;6:33-40.
3. Sintek M, Coverstone E, Bach R, et al. Excimer laser coronary angioplasty in coronary lesions:Use and safety from the NCDR/CATH PCI registry. Circ Cardiovasc Interv. 2021;14:e010061.
4. Vizzari G, Caminiti R, Ielasi A, et al. Contrast-enhanced excimer laser stepwise approach during PCI for resistant coronary lesions. Catheter Cardiovasc Interv. 2024;104:220-226.
5. Chaturvedi A, Case BC, Hashim HD, et al. Contrast?enhanced excimer laser coronary atherectomy. Catheter Cardiovasc Interv. 2024;104:907-908.















