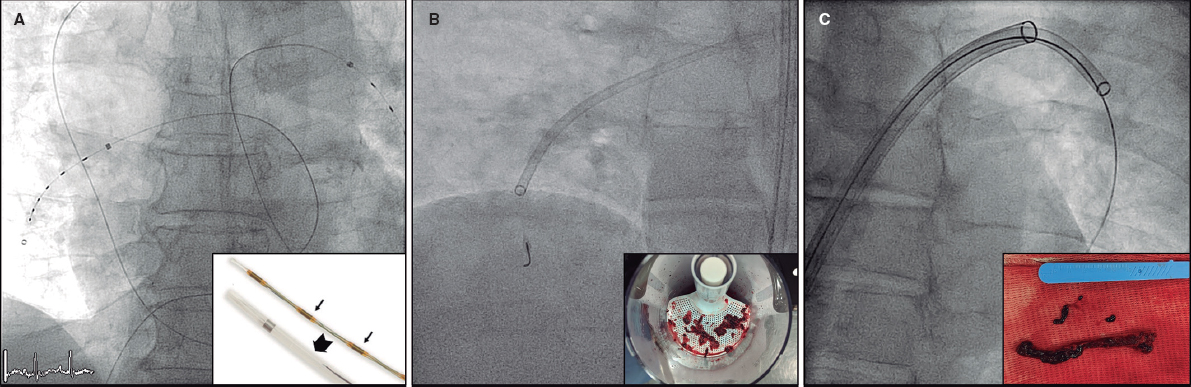
QUESTION: Briefly, what is the evidence behind the percutaneous left atrial appendage closure (LAAC) regarding oral anticoagulation (OA)? Is there any evidence on direct-acting oral anticoagulants (DAOAs)?
ANSWER: The LAAC has been compared to OA regarding the prevention of thromboembolic events in patients with nonvalvular atrial fibrillation (NVAF) in 3 randomized clinical trials: the PROTECT AF, the PREVAIL, and the PRAGUE-17. The first 2 ones randomized 1114 patients with NVAF and CHADS2 ≥ 1 on a 2:1 ratio to receive the Watchman device (Boston Scientific, United States) or warfarin. In an intention-to-treat meta-analysis of aggregate individual data from the 2 studies with 5-year follow-ups no differences were seen between both strategies in the primary composite endpoint of stroke, systemic embolism, and cardiovascular or unexplained death (hazard ratio [HR], 0.82; 95% confidence interval [95%CI], 0.58-1.17; P = .27).1 In the secondary endpoint analysis a higher—though not statistically significant—rate of ischemic stroke or systemic embolism was reported in the device group (HR, 1.71; 95%CI, 0.94-3.11; P = .08). On the contrary, the LAAC was associated with a lower rate of hemorrhagic (HR, 0.20; 95%CI. 0.07-0.56; P = .002) or incapacitating stroke (HR, 0.45; 95%CI, 0.21-0.94; P = .034), major hemorrhage unrelated to the procedure (HR = 0.48; 95%CI 0.32-0.71; P < 0.001), cardiovascular or unexplained death (HR, 0.59; 95%CI, 0.37-0.94; P = .027), and all-cause mortality (HR, 0.73; 95%CI, 0.54-0.98; P = .035).
The results of the PRAGUE17 clinical trial have been recently published. This study randomized 402 patients on a 1:1 ratio with NVAF and an indication for OA to LAAC with the Amplatzer Amulet (Abbott Vascular, United States) (61.3%) or the Watchman device (38.7%) or to a DAOA, mainly apixaban (95.5%).2 This trial only included patients considered of high risk with some of the following criteria: history of bleeding that required intervention or hospitalization, past medical history of embolism despite treatment with OA or CHA2DS2-VASc scores ≥ 3 with HAS-BLED ≥ 2. During the mean follow-up of 20.8 months ± 10.8 months no differences were seen in the composite primary endpoint (stroke, transient ischemic attack, systemic embolism, cardiovascular death, major or clinically relevant bleeding or procedural or device-related complications) reaching criteria of non-inferiority (HR, 0.84; 95%CI, 0.53-1.31; P = .44; P for non-inferiority = .004). No differences between both groups were seen either in any of the events included in the composite primary endpoint separately.
Currently, there are 4 ongoing different randomized clinical trials that will be assessing the benefits of the LAAC with the Watchman FLX device (CHAMPION-AF [NCT04394546]), the Amplatzer Amulet device (CATALYST [NCT04226547]) or with either one of the 2 (OCCLUSION-AF [NCT03642509], CLOSURE-AF [NCT03463317]) vs DAOA therapy in patients with NVAF.
Q.: Which are the current indications for the LAAC? Should the clinical guidelines most recently published be changed somehow?
A.: The European guidelines on the management of AF published in 2020 suggest that the LAAC could be considered in patients with NVAF and a contraindication to OA (IIb level of recommendation, C level of evidence).3 Regarding this indication, I think a few considerations should be made. In the first place, the evidence available on the LAAC is limited basically because of the non-inferiority design of the clinical trials available with a limited number of patients. Also, due to the great heterogeneity of the antithrombotic therapy indicated post-LAAC, that could be conditioning the applicability of the results obtained. Secondly, the guidelines were written before the results of the PRAGUE-17 trial came out, that are consistent with the efficacy and safety profile of the procedure, also vs DAOAs (although in a limited 2-year follow-up) that is currently the treatment of choice to prevent thromboembolic events in patients with AF with, at least, moderate risk. Thirdly, since it is a «prophylactic» therapy, procedural success should be high, and the rate of related complications should be low. In this sense, in the largest registry published to this date of over 38 000 patients, the rate of procedural success was 98.3% and the rate of major complications, 2.16%.4 Finally, the lack of compliance to medical therapy and under or overdosing are common problems in our routine clinical practice, which has a significant impact on the efficacy of OA, which is probably reported in the randomized clinical trials as well.
Q.: Which is the common practice at your center?
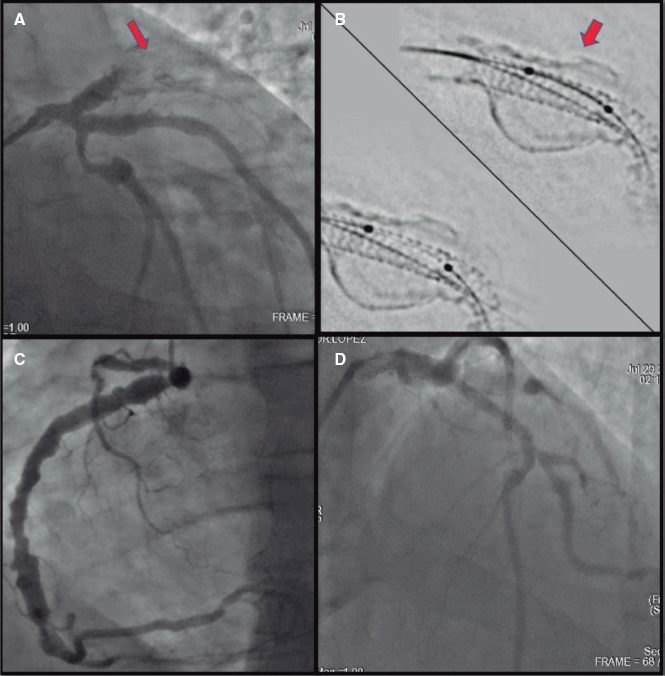
A.: The patient’s profile eligible for this procedure is that of an elderly patient with an indication for OA due to NVAF with a previous episode of intracranial or gastrointestinal bleeding. Other less common cases are patients with chronic anemia or high bleeding risk with an indication for OA or dual or triple antithrombotic therapy. Finally, more and more patients with stroke despite being on OA are being reported. We perform a transesophageal echocardiogram (TEE) prior to the procedure in all the cases. We often perform implantation with mild sedation and TEE guidance with microprobe. Antithrombotic therapy upon discharge is based on every particular patient and followed-up at the specific structural heart disease consultation within the first year including TEE sometime within the first 3 months after hospital discharge.
Q.: How valuable are imaging modalities in the selection of patients and procedural guidance?
A.: The preprocedural study of left atrial appendage aims at determining the anatomical feasibility for implantation purposes, providing the right dimensions for a proper sizing of the device, and excluding the existence of thrombi. To this end, a 3D imaging modality should be used, usually a TEE or a computed tomography scan. The selection of one or the other will depend on its accessibility, experience of every center, and certain characteristics of the patient like the presence of kidney disease. Regarding the computed tomography scan, a 2-stage acquisition protocol with sensitivity and specificity rates close to 100% for the detection of thrombi should be conducted.
In most cases, the LAAC is performed with TEE guidance. However, this imaging modality has complications: on the one hand, the prolonged use of the probe can cause non-negligible oropharyngeal lesions. On the other hand, general anesthesia is required in many patients with the corresponding complications and increased procedural duration and costs. In this sense, as we have been gaining experience on device implantation, new options have come up to minimize these limitations like the TEE microprobe and the intracardiac echocardiography. The microprobe provides slightly lower quality of image compared to conventional TEE but is better tolerated by the patient and requires no general anesthesia. The drawback is that no 3D images can be acquired, which means that a previous study capable of providing 3D images is necessary. Regarding intracardiac echocardiography, the latest machines available provide better quality of images, some even the possibility of 3D reconstructions (still not on the market). They do not require the presence of an imaging specialist at the cath lab or use of a transesophageal probe either. On the other hand, both the cost of the device and its disposable nature reduce its utility in the routine clinical practice. Several ongoing prospective registries will be shedding light on the safety, feasibility, and possible technical advantages.
Q.: Are there any significant differences among the different devices available?
A.: Despite the technical advances made in the procedure, there are still pending challenges or, at least, room for improvement: a) great anatomical variability in the left shape, orientation, and size of left atrial appendage; b) risk of perforation with pericardial effusion; c) device embolization; d) persistent residual leak after implantation; and e) rate of device related thrombosis. For all these reasons, there are many devices to perform LAAC available today or in the pipeline that try to improve some of these aspects. Generally speaking, 2 important types of design can be distinguished here: tamponade-like devices like the Watchman, and anchor-disc shaped devices like the Amplatzer Amulet. In principle, the latter can be more versatile in complex anatomies while the first one has a growing body of evidence. Regarding procedural results, the observational data obtained do not suggest differences in the implantation success rate or in the rate of serious complications, device thrombosis or significant residual leak reported between the 2 most widely used devices to this date. However, the results from comparative studies AMULET IDE [NCT02879448] and SWISS-APERO [NCT03399851]5 will bring us more data on this regard.
There are many other devices available with scarce evidence and limited used, but with technical advances and very promising designs: extreme sizes, antithrombotic expanded polytetrafluoroethylene covering, self-adaptable configuration to the left atrial appendage ostium, small anchor with large disc configuration, highly flexible or articulated devices…Given the great anatomical variability of the left atrial appendage, maybe some devices will be complementary in the future. Therefore, if the volume of cases allows it, it can make sense to use more than just 1 type of device in every patient, the one more suitable to his anatomy.
Q.: Please tell us about any relevant ongoing clinical trials. In your professional opinion, what type of study should be conducted?
A.: There is no doubt that the most significant randomized clinical trials that are being conducted today are those comparing the LAAC to DAOAs. These trials will eventually determine the actual benefit of this procedure. Another very important aspect to elucidate is the optimal antithrombotic therapy to prevent device thrombosis. In this sense, the following randomized clinical trials should be highlighted: a) the SAFE-LAAC trial [NCT03445949] will assess the 6-month efficacy profile of dual antiplatelet therapy vs a 1-month course of dual antiplatelet therapy plus 5 months of single antiplatelet therapy; b) the ANDES trial [NCT03568890] will analyze the DAOA strategy vs an 8-week course of dual antiplatelet therapy; c) the FADE-DRT trial [NCT04502017] will be comparing the efficacy profile of 3 strategies consisting of a 6-week course of OA followed by a 6-month course of dual antiplatelet therapy, half-dose DAOA plus genotype-guided dual targeted therapy (acetylsalicylic acid + clopidogrel or half-dose DAOA depending on clopidogrel resistance or not); and d) the ASPIRIN-LAAO trial [NCT03821883] that will be analyzing the benefit of keeping single antiplatelet therapy starting 6 months after the procedure.
Another particularly interesting randomized clinical trial is the OPTION [NCT03795298] that will be randomizing 1600 patients treated with ablation due to AF to receive the Watchman FLX device or OA. Finally, from a theoretical point of view, evidence is still scarce in some potentially LAAC-eligible patients as it is in patients with recurring thromboembolic events despite OA or with NVAF and chronic kidney disease on dialysis. Both groups are characterized by a very high thrombotic risk. There is lack of consensus regarding the most adequate antithrombotic strategy that is empirically administered in most of the cases. Also, patients on dialysis have a high hemorrhagic risk and, unlike the general population, OA does not seem to benefit them. Some observational studies suggest that the LAAC can be safe and effective in these subgroups of patients. However, to this date, no randomized clinical trial has studied this therapy compared to others.
FUNDING
The author received no financial support for this work.
CONFLICTS OF INTEREST
None declared.
REFERENCES
1. Reddy VY, Doshi SK, Kar S, et al. 5-Year Outcomes After Left Atrial Appendage Closure:From the PREVAIL and PROTECT AF Trials. J Am Coll Cardiol. 2017;70:2964-2975.
2. Osmancik P, Herman D, Neuzil P, et al. Left Atrial Appendage Closure Versus Direct Oral Anticoagulants in High-Risk Patients With Atrial Fibrillation. J Am Coll Cardiol. 2020;75:3122-3135.
3. Hindricks G, Potpara T, Dagres N, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2020:ehaa612.
4. Freeman JV, Varosy P, Price MJ, et al. The NCDR Left Atrial Appendage Occlusion Registry. J Am Coll Cardiol. 2020;75:1503-1518.
5. Basu Ray I, Khanra D, Shah S, et al. Meta-Analysis Comparing WatchmanTM and Amplatzer Devices for Stroke Prevention in Atrial Fibrillation. Front Cardiovasc Med. 2020;7:89.
* Corresponding author: Servicio de Cardiología, Complejo Asistencial Universitario de León, Altos de Nava s/n, 24017 León, Spain.
E-mail address: tomasbenito@outlook.com (T. Benito-González).