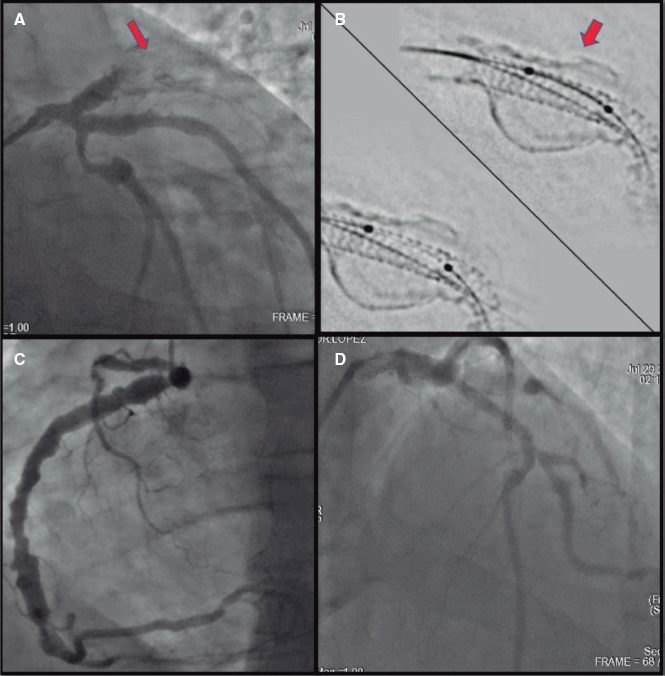
HOW WOULD I APPROACH IT?
This is the case of a massive hemoptysis requiring orotracheal intubation where the origin of the hemorrhage can be clearly identified in a fistula from the left bronchial artery—with fistulous trajectory—to the left pulmonary artery upper branch.
In view of the situation, and once the patient has been stabilized with right selective intubation, discussion starts on how to save the lung from blood content.
Currently, interventional radiology in Spain is available in almost every PCI-capable center meaning that our participation as interventional cardiologists is not required anymore. However, thanks to the «infarction code» network, the geographic availability of cath labs is even greater compared to vascular interventional radiology suites, as it happened to us in our beginnings.
In this case, the source of bleeding has already been found, which means we can attempt the catheterization of the bronchial artery right away. Access via pulmonary artery is indicated for cases when the bronchial artery cannot be catheterized. In these cases, it’d be closed with coils as selectively as possible to prevent massive pulmonary infarction from happening.1
Cardiologists have a great variety of devices available to catheterize arteries originate at the aorta like the coronary or bronchial arteries. «Visceral» catheters like the COBRA one work great to catheterize this kind of ostia while highly «torqueable» and harmless hydrophilic catheters are used to study coronary ostia (figure 1).

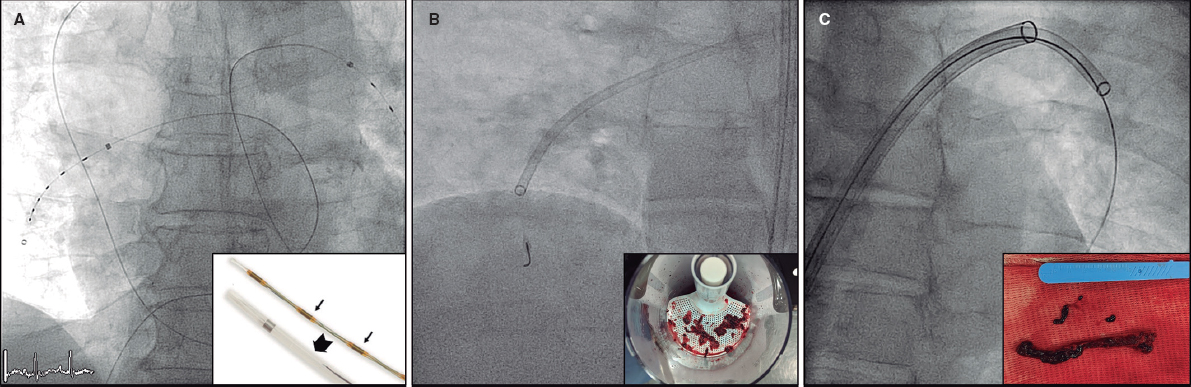
Figure 1. Visceral catheters used to study the branches of both the ascending and the descending aorta.
Since these catheters are often unavailable at the cath lab, the Amplatz guide catheter with apex curve can be used as if we were looking to perform a bypass, and even an EBU guide catheter to modify the curve with another guidewire through the straight end. Once the artery has been catheterized, although not selectively, passing a 0.14 in guidewire can be relatively easy to do to bring stability to the whole surgical kit. Using the radial or femoral access is something that’s completely irrelevant. However, since location is in the thoracic aorta, the femoral access could be one we’re most comfortable with in intubated patients (figure 2).
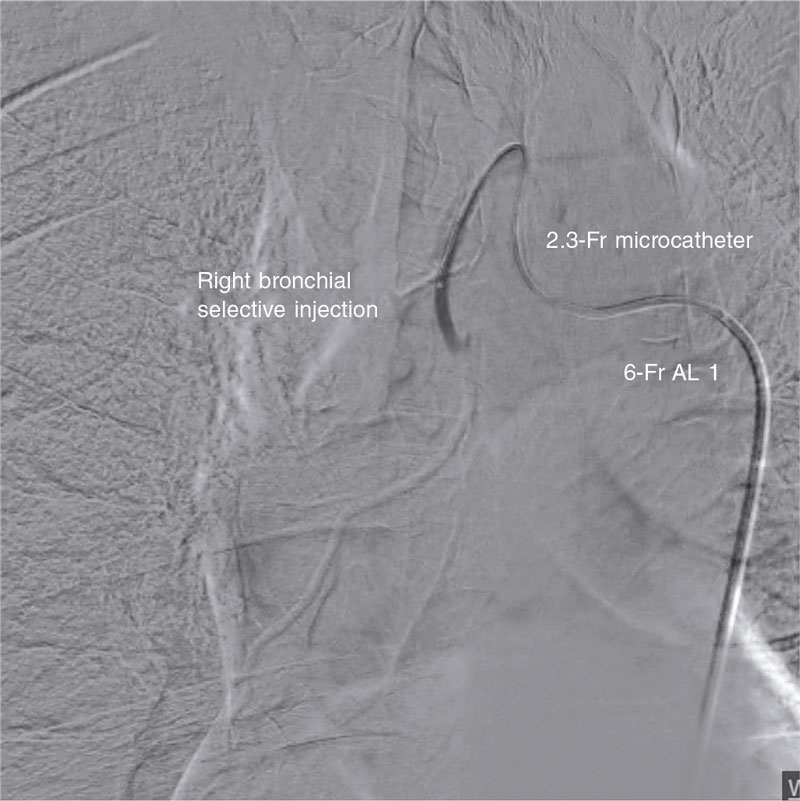
Figure 2. Selective injection into right bronchial artery. After catheterization with the Amplatz 1 guide catheter, a microcatheter is mounted over a 0.14 in guidewire.
A 2.4-Fr microcatheter can be mounted over this guidewire for 0.18 in coils or even a 4-Fr catheter to inject embolization material by thoroughly selecting the target area as much as possible.
I would rather use 0.18 in coils as the method of choice to release them before ever reaching the fistulous trajectory. Afterwards, I’d check whether it has been properly sealed and no collaterals have been recruited prior to the fistulous trajectory (figure 3).
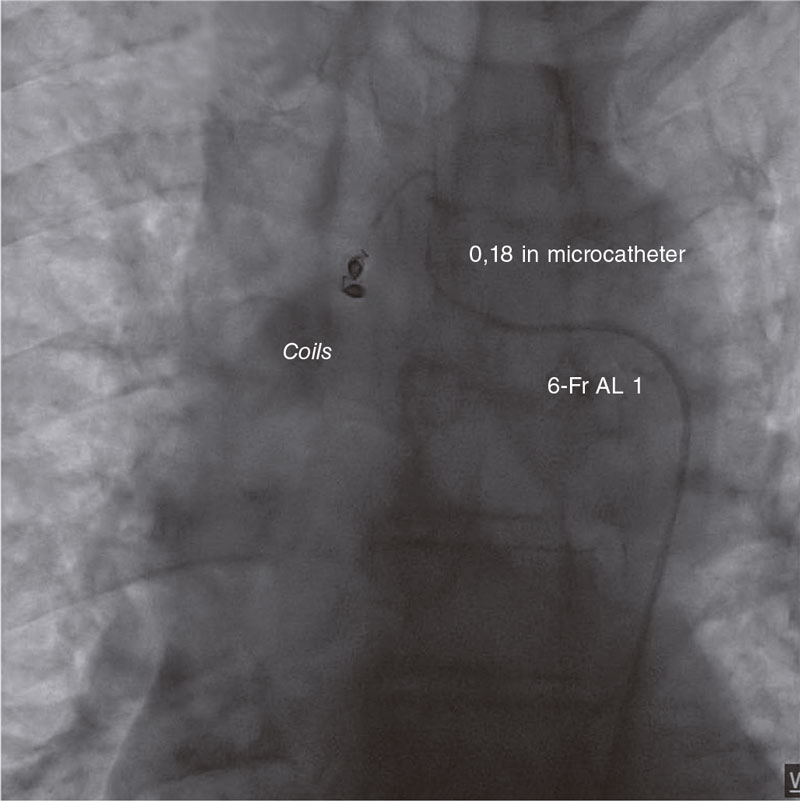
Figure 3. Embolization of the right bronchial artery with 0.18 in coils.
In no coils are available, I’d probably use embolization material like Spongostan gelatin sponge to create gel particles that can stop the bleeding when they can’t get through. It is mixed from 2 syringes containing serum, contrast, and small «confetti» layers of Spongostan that are linked through a 3-way stopcock.2 The content from one syringe is passed on to the next until it has the consistency of small pieces of gel. I would gently inject it through the microcatheter, and if the contrast agent still couldn’t make it through the fistulous trajectory, I’d keep on injecting at low pressure. This can stop the bleeding and help achieve the effect obtained with the coils when we’ve ran out of them, and passage is still not under control. I would never use particles because they migrate through the fistulous trajectory and cause massive pulmonary infarction.
Finally, if the bleeding cannot be stopped, I would leave a balloon inflated to gain time to evacuate the patient to thoracic surgery or the interventional radiology suite.
The takeaway message is that we need to be prepared one way or another3 to solve emergency complications that can be solved using interventional radiology techniques in places where an angiographer—for the lack of an experienced radiologist—is available.
FUNDING
None whatsoever.
CONFLICTS OF INTEREST
J. Portales Fernández received fees for his collaboration for Medtronic, and Boston Scientific.
ACKNOWLEDGEMENTS
We wish to thank Dr. Sergio Moyano Calvente, interventional radiologist at Hospital Universitario de Cáceres, Cáceres, Spain for years of collaboration and mutual work.
REFERENCES
1. Tamashiro A, Miceli MH, Rando C, et al. Pulmonary artery access embolization in patients with massive hemoptysis in whom bronchial and/or nonbronchial systemic artery embolization is contraindicated. Cardiovasc Intervent Radiol. 2008;31:633e7.
2. Abadal JM. Spongostan preparation for Endovascular Embolization. Available at: https://youtu.be/zhq0hYQ8iGs. Accessed 30 Nov 2022.
3. Sopko DR, Smith TP. Bronchial Artery Embolization for Hemoptysis. Semin Intervent Radiol. 2011;28:48-62.
* Corresponding author.
E-mail address: portales70@hotmail.com (J. Portales Fernández).