To the Editor,
Extracorporeal membrane oxygenation (ECMO) has been used as invasive mechanical respiratory support for almost 40 years. Venovenous ECMO makes full use of cardiopulmonary bypass to take over the lungs respiratory function. Venoarterial ECMO provides circulatory support plus oxygenation and is useful to treat cardiogenic shock or refractory cardiopulmonary arrest.1 The technical advances made on ECMO devices have boosted its use through a fully percutaneous approach, often performed by interventional cardiologists trained in large bore percutaneous access. This, in turn, has led to quicker setups and has reduced time in critical situations and increased the chances of survival.2
Since the beginning of the novel coronavirus disease (COVID-19) pandemic, different Chinese centers have reported a high incidence of acute respiratory distress syndrome (ARDS) (17% to 29%) and critical illness (23% to 32%) among hospitalized patients.3 The management of ARDS includes lung-protective ventilation, adequate fluid strategy, and early initiation of pronation.4 If these strategies are insufficient, the use of ECMO to treat refractory hypoxemia becomes the only alternative available.5 Also, important delays in the application of reperfusion therapies in the context of ST-segment elevation myocardial infarction have been observed during the current pandemic, leading to higher mortality rates, mechanical complications, and cardiogenic shock.6 Finally, patients with advanced chronic heart failure have a greater risk of developing severe COVID-19 symptoms.
ARDS, acute cardiogenic shock, and advanced chronic heart failure are the triple threat of ECMO candidates with COVID-19.
This is a multicenter retrospective registry of percutaneous implantation of ECMO between March 15 and May 15, 2020 in 4 Spanish centers during the COVID-19 pandemic. The official number of COVID-19 cases admitted to the centers that participated in the study period was ~4500. A total of 14 infected patients (0.3%) were treated with an ECMO device. The main baseline characteristics of the patients included in the study are shown on table 1. Ten patients developed ARDS (71.4%), 2 ARDS added to decompensated advanced chronic heart failure (14.3%), and 2 more developed cardiogenic shock due to acute coronary syndrome in the context of SARS-CoV-2 infection (14.3%).
Table 1. Baseline characteristics, treatment and outcomes of patients with percutaneous ECMO implantation during the COVID-19 pandemic
| Total (n = 14) | |
|---|---|
| Baseline characteristics | |
| Age (years) | 48.79 ± 7.13 |
| Weight (kg) | 85.79 ± 18.03 |
| Height (m) | 1.73 ± 0.07 |
| BMI (kg/m2) | 28.32 ± 5.02 |
| Female sex | 3 (21.4) |
| Hypertension | 4 (28.6) |
| Diabetes mellitus | 2 (14.3) |
| Dyslipidemia | 5 (35.7) |
| Smoker (former or current) | 6 (42.8) |
| Atrial fibrillation | 1 (7.1) |
| Coronary artery disease | 2 (14.3) |
| Congestive heart failure | 1 (7.1) |
| Previous stroke | 0 (0) |
| Chronic liver disease | 0 (0) |
| Chronic kidney disease | 0 (0) |
| Apnea-hypopnea respiratory syndrome | 2 (14.3) |
| Previous cancer | 1 (7.1) |
| Treatment | |
| Hydroxychloroquine | 13 (92.9) |
| Azythromycin | 12 (85.7) |
| Glucocorticoids | 12 (85.7) |
| Lopinovir/Ritonavir | 12 (85.7) |
| Tozilizumab | 9 (64.3) |
| Remdesvir | 4 (28.6) |
| Antibacterial co-treatment | 14 (100) |
| Cephalosporins | 7 (50) |
| Piperacillin tazobactam | 6 (42.9) |
| Vancomycin | 3 (21.4) |
| Colistin | 3 (21.4) |
| Daptomycin | 1 (7.1) |
| Meropenem | 8 (57.1) |
| Linezolid | 7 (50) |
| Outcomes | |
| Days on ECMO | 12 [9-14] |
| Days on mechanical ventilation | 34 [18-51] |
| Days at the ICU/CCU | 44.5 [19.0-56.0] |
| Length of hospital stay (total, in days) | 62 [27-66] |
| Death | 4 (28.6) |
| Multiorgan failure | 1 (25) |
| Withholding of live-sustaining therapies | 3 (75) |
BMI, body mass index; CCU, coronary care unit; ECMO, extracorporeal membrane oxygenation; ICU, intensive care unit. Data are expressed as no. (%), mean ± standard deviation or median [interquartile range]. | |
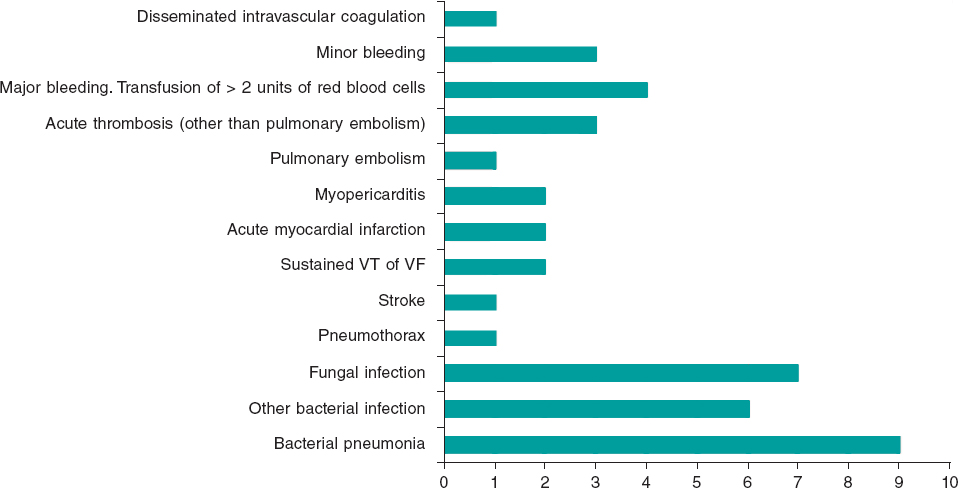
Nine patients (64.3%) were initially admitted to conventional wards and 5 (35.7%) were immediately transferred to the intensive care unit. During admission, different treatments were used as shown on table 1. An echocardiography was performed in all cases; the median left ventricular ejection fraction of patients without previous cardiovascular disease affected by ARDS (10 patients [71.4%]) was 58% [interquartile range (IQR), 51% to 64%] and the tricuspid annular plane systolic excursion was 18.5 mm [17.7 mm to 19.3 mm]. The ECMO device was implanted a median of 7 days from admission through fully percutaneous approach in all cases but one. Based on the ECMO modality used, 2 patients received a venoarterial ECMO (14.3%) and 12 received a venovenous ECMO (85.7%). In 2 patients of the latter group VAV (venoarterial-venous) ECMO conversion was performed (13.3%). Two patients (14.3%) also needed a left ventricular assist device that in both cases was an intra-aortic balloon pump. The complications of these patients are shown on figure 1. Although coexisting infections were the most common problem, we should mention the very high-risk of presenting with thrombotic and bleeding events (4 [28.6%] and 8 [57.1%], respectively) likely due to the ECMO device per se but also to the inflammatory and coagulation disorders due to COVID-19. All patients showed, at least, 1 complication. The median time connected to the ECMO device was 12 days [9-14] and the median length of the hospital stay was 62 days [27-66]. Four patients died (28.6%), 1 (25%) due to multiorgan failure and 3 (75%) after withholding limitation of live-sustaining therapies or limitation of the therapeutic effort.
Figure 1. Complications during extracorporeal membrane oxygenation assistance and frequency. VF, ventricular fibrillation; VT, ventricular tachycardia.
In conclusion, this relatively small registry is nothing but a glimpse of the most advanced therapies used during the current COVID-19 pandemic. It shows that 3 out of 4 patients who required ECMO survived until hospital admission despite the high rate of related complications. We should mention that the rapid and minimally invasive implantation of ECMO devices in critically ill patients has become more and more popular among interventional cardiologists.
REFERENCES
1. Combes A, Hajage D, Capellier G, et al. Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome. N Engl J Med. 2018;378:1965-1975.
2. Napp LC, Kühn C, Hoeper MM, et al. Cannulation strategies for percutaneous extracorporeal membrane oxygenation in adults. Clin Res Cardiol. 2016;105:283?296.
3. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020. https://doi.org/10.1001/jama.2020.1585.
4. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China:a single-centered, retrospective, observational study. Lancet Respir Med. 2020. https://doi.org/10.1016/S2213-2600(20)30079-5.
5. Bartlett RH, Ogino MT, Brodie D, et al. Initial ELSO Guidance Document:ECMO for COVID-19 Patients with Severe Cardiopulmonary Failure. ASAIO J. 2020;66:472?474.
6. Rodríguez-Leor O, Cid-Álvarez B, Ojeda S, et al. Impacto de la pandemia de COVID-19 sobre la actividad asistencial en cardiología intervencionista en España. REC Interv Cardiol. 2020;2:82-89.
Corresponding author: Departamento de Cardiología, Hospital Clínico Universitario, Avda. Ramón y Cajal 3, 47003 Valladolid, Spain.
E-mail address: ijamat@gmail.com (I.J. Amat-Santos).