ABSTRACT
Introduction and objectives: Complete and secure venous access closure is essential at the end of large-bore catheter-based procedures. The aim of this study is to evaluate the safety and efficacy profile of a new simplified purse-string suture with 3-way stopcock to improve venous hemostasis in structural heart procedures (the SUTURE-3 study).
Methods: We conducted a multicenter, non-randomized and prospective study including all consecutive patients undergoing any interventional procedures with a ≥ 10-Fr femoral venous access in whom a simplified purse-string suture with 3-way stopcock for venous hemostasis was used. Exclusion criteria were < 10-Fr diameters or ipsilateral artery access. We evaluate the safety profile according to the Valve Academic Research Consortium-3 criteria, efficacy (correct hemostasis), a pain-score (visual analog scale [VAS]) and a clinical 1 month-follow up.
Results: A total 222 patients were included. Mean age was 73.6 ± 9.7 years (58%, men; 85%, hypertensive; 44%, diabetics; 91%, had a past medical history of atrial fibrillation). We found significantly high CHA2DS2-VA (4.1 ± 1.8) and HAS-BLED (3.5 ± 1.6) scores with a higher proportion of previous major bleeding (62%). A total of 54% of patients were on oral anticoagulation and 25%/6% on single/dual antiplatelet therapy, respectively. The most common procedure was left atrial appendage closure (n = 154, 69%) and mitral valve transcatheter edge-to-edge repair (n = 41, 18%). Mean diameter access was 15.6 ± 5-Fr; 91%, echo-guided; compression time, 4 ± 1.7 h; pain in VAS, 0.6 ± 1.5. Hemostasis was instantaneous with a 100% success rate, and only 2 minor bleeding events being reported (0.9%). No access complications (including infections) were recorded after discharge at the 1-month follow-up.
Conclusions: The simplified purse string suture with 3-way stopcock to improve venous hemostasis in structural heart procedures is a simple, inexpensive, safe and effective procedure.
Keywords: Femoral vein closure. Large-bore venous sheath. Purse-string suture.
RESUMEN
Introducción y objetivos: El cierre adecuado del acceso venoso es esencial tras procedimientos transcatéter de gran diámetro. El objetivo fue evaluar la seguridad y la eficacia de una nueva sutura en jareta simplificada con llave de paso de 3 vías para la hemostasia venosa en el intervencionismo cardiaco estructural (estudio SUTURE-3).
Métodos: Estudio multicéntrico, prospectivo y no aleatorizado que incluyó pacientes consecutivos con acceso venoso femoral ≥ 10 Fr en los que se empleó esta sutura simplificada ajustada con llave de 3 pasos. Se excluyeron los accesos < 10 Fr y los accesos arteriales homolaterales. La seguridad se evaluó conforme a los criterios Valve Academic Research Consortium-3, y la eficacia (hemostasia correcta) y el dolor con la escala visual analógica (EVA). Se realizó un seguimiento clínico a 1 mes.
Resultados: Se incluyeron 222 pacientes con una edad media de 73,6 ± 9,7 años, el 58% varones, el 85% hipertensos, el 44% diabéticos y el 91% con antecedentes de fibrilación auricular. Las puntuaciones CHA2DS2-VA (4,1 ± 1,8) y HAS-BLED (3,5 ± 1,6) fueron elevadas, con alta proporción de hemorragias mayores previas (62%). El 54% de los pacientes estaban tomando anticoagulantes orales, y el 24% y el 6% recibían antiagregación simple o doble, respectivamente. Los procedimientos más realizados fueron el cierre percutáneo de la orejuela izquierda (n = 154, 69%) y la terapia de borde a borde mitral (n = 41, 18%). El diámetro medio del acceso fue de 15,6 ± 5 Fr, en el 91% ecoguiado, con un tiempo de compresión de 4 ± 1,7 h y dolor en la retirada de 0,6 ± 1,6 en la EVA. El tiempo de hemostasia fue instantáneo, con una tasa de éxito del 100%, y solo 2 hemorragias menores (0,9%). A 1 mes de seguimiento no se registraron complicaciones del acceso (incluidas infecciones).
Conclusiones: La sutura en jareta simplificada con llave de 3 vías para la hemostasia venosa de accesos ≥ 10 Fr en el intervencionismo cardiaco estructural es un procedimiento sencillo, económico, seguro y efectivo.
Palabras clave: Cierre venoso femoral. Introductor venoso de gran diámetro. Sutura en jareta.
INTRODUCTION
In recent decades, structural heart and electrophysiology procedures have grown exponentially in both volume and indications1. With this increase, the profession is under pressure to process patients quickly and mobilize and discharge them just within hours following the procedure.
Transfemoral venous access using large-bore introducer sheaths is common for the treatment of various conditions (percutaneous left atrial appendage closure, transcatheter edge-to-edge repair, etc.).2-6 This access is ideal due to its large caliber, few anatomical variations, and ability to accommodate 27-Fr outer-diameter introducer sheaths with ease. Other alternative access sites are less suitable for these procedures for various reasons: the jugular approach is uncomfortable for both patients and operators, the subclavian or axillary access sites carry a risk of hemothorax or pneumothorax, and the arm veins are not wide enough.7
Traditionally, hemostasis via venous access has been achieved by manual compression, which is still effective for small-caliber venous sheaths and is the standard against which other venous hemostasis methods are compared.7 Even with smaller sheaths, compression-based hemostasis can take up to 30 minutes to achieve, which is uncomfortable for the patient and inconvenient for health care personnel.6,8-10 Additionally, the immobilization period (4-8 hours) following compression increases cost and patient discomfort, and presents a real risk of deep vein thrombosis, in addition to the hemorrhagic risks associated with incomplete hemostatic control or vascular injury at the access site, leading to hematoma, arteriovenous fistula, or pseudoaneurysm.6,10 In recent years, using vascular ultrasound for puncture has helped increase effectiveness and, more importantly, safety.
Alternatives to compression for achieving immediate and safe venous hemostasis are increasingly important and include subcutaneous suture techniques and vascular closure devices, such as the VASCADE (Cardiva Medical, United States), MYNX (Cardinal Health, United States), the MANTA (Teleflex/Essential Medical, United States), and the ProGlide/ProStyle (Abbott Vascular, United States).7
The figure-of-eight suture has been used as a hemostasis method in various studies, primarily in the field of electrophysiology, achieving faster compression, reducing the length of stay, and with fewer complications compared with manual compression.5,6,8,12-22 The purse-string suture is an alternative method that has proven safe and effective, with results similar to the figure-of-eight suture compared with manual compression across various studies. Moreover, it is associated with shorter hemostasis time, less pain or discomfort, and shorter immobilization times.23-26
A simplified purse-string suture technique has been developed, with only 2 subcutaneous needle trajectories instead of 4, which maintains tension due to a 3-way stopcock enabling continuous tension adjustment compared with a fixed knot. Our objective was to evaluate the safety and efficacy profile of this new suture technique in patients undergoing transfemoral structural heart procedures using large-bore introducer sheaths (≥ 10-Fr).
METHODS
Study design
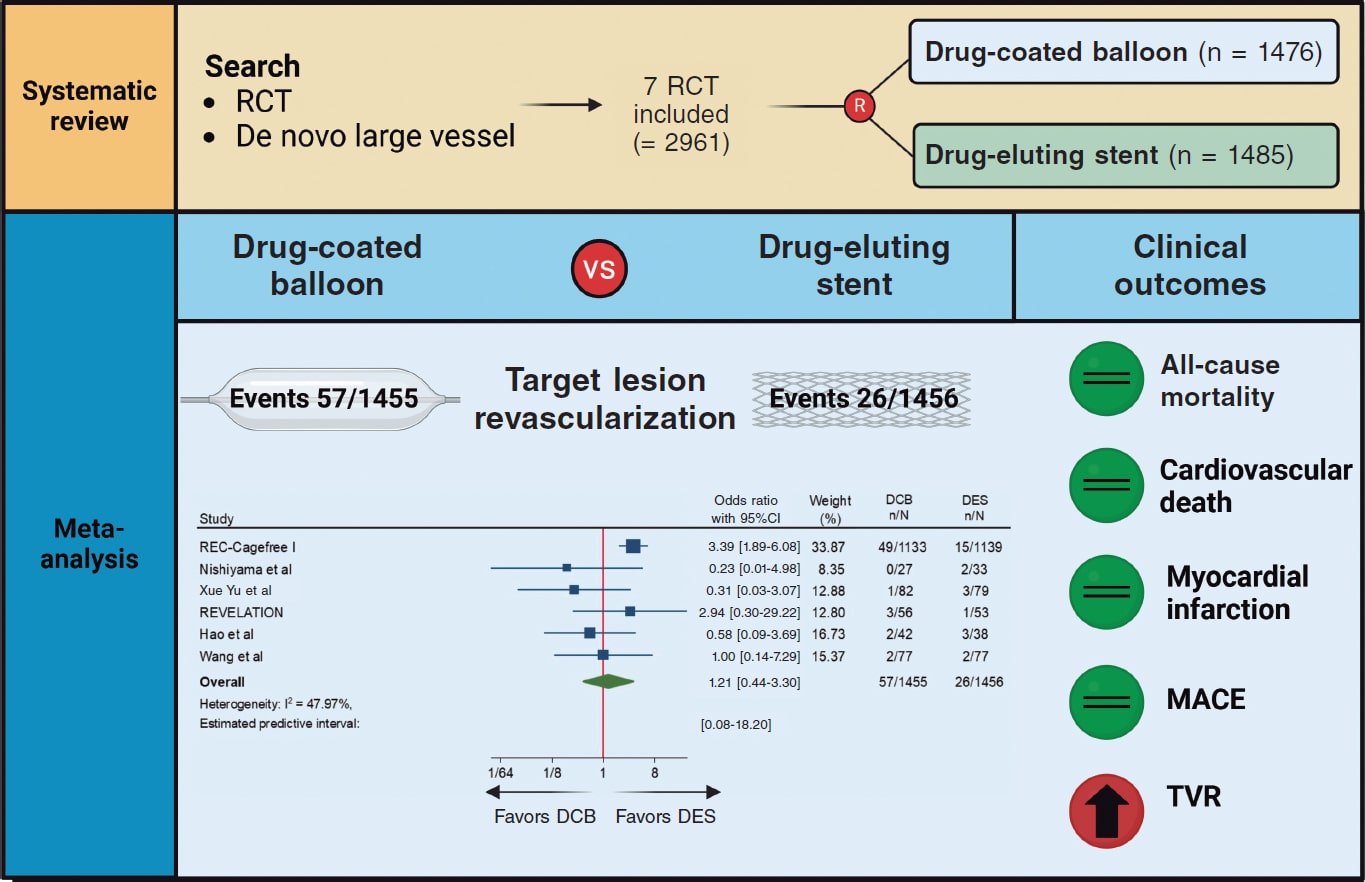
The SUTURE-3 study is a prospective, non-randomized, multicenter registry that included consecutive patients who, after ≥ 10-Fr ultrasound-guided femoral venous access for structural heart procedures, received a simplified purse-string suture using a 3-way stopcock for tension adjustment (figure 1). Closure was performed by the interventional cardiologist or interventional nurse.
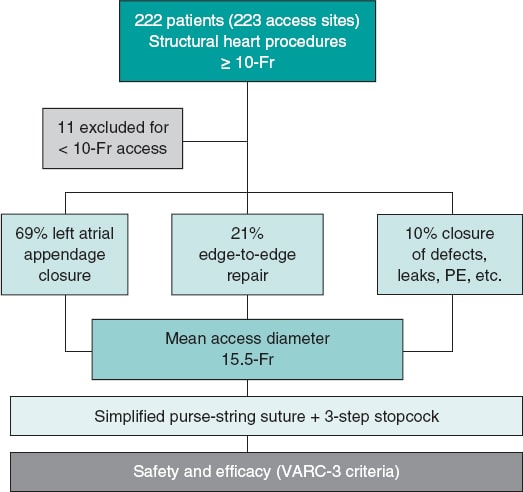
Figure 1. Central illustration. Flowchart of the SUTURE-3 study. PE, pulmonary embolism; VARC-3, Valve Academic Research Consortium 3.
Materials used
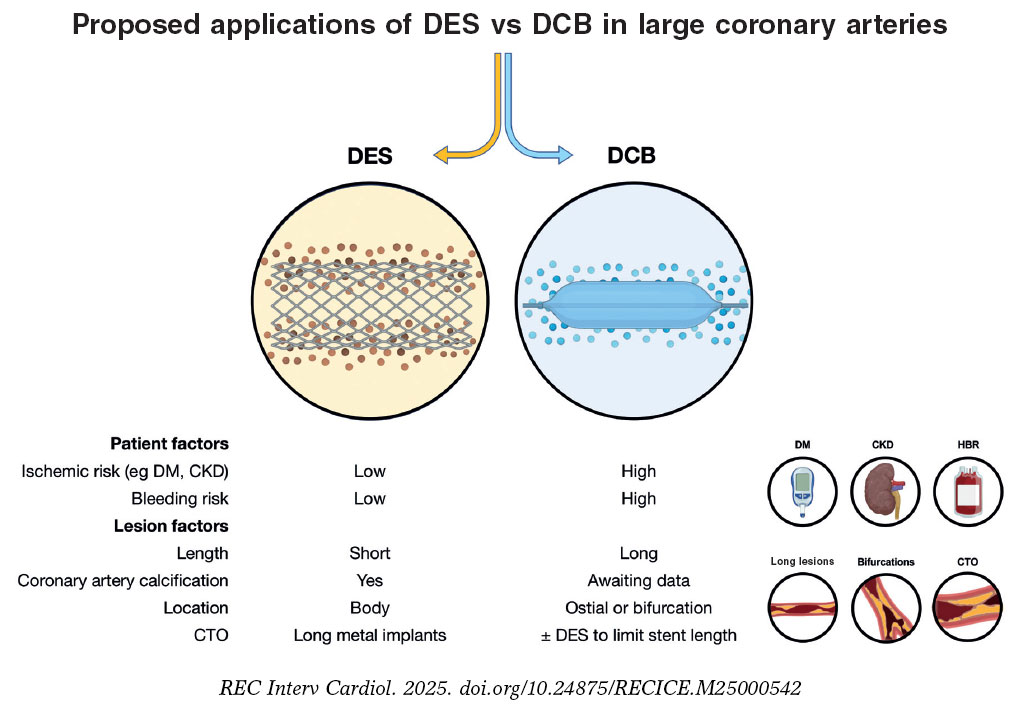
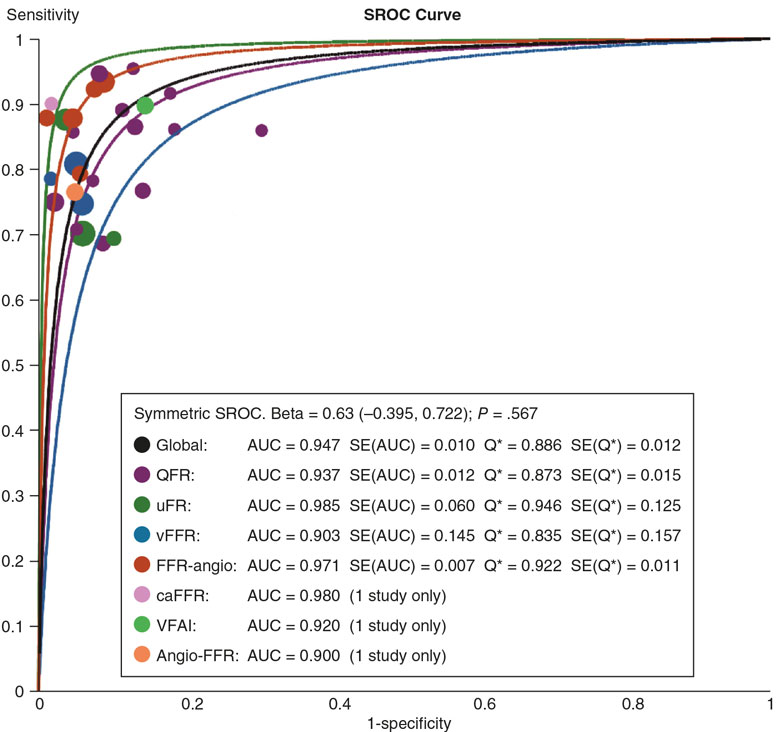
A needle holder was used to place a purse-string suture with a 38-48 mm triangular-tipped curved needle (chosen by the operator based on the diameter of the introducer sheath and vascular puncture depth; for deeper punctures the 48 mm needle was used), and absorbable braided synthetic suture material (Polysorb, CL-928, size 1, GS-13 cutting). A simple 3-way stopcock was used to adjust and maintain suture tension, as shown in figure 2 and figure 3. The total estimated cost of the material was €1.45.
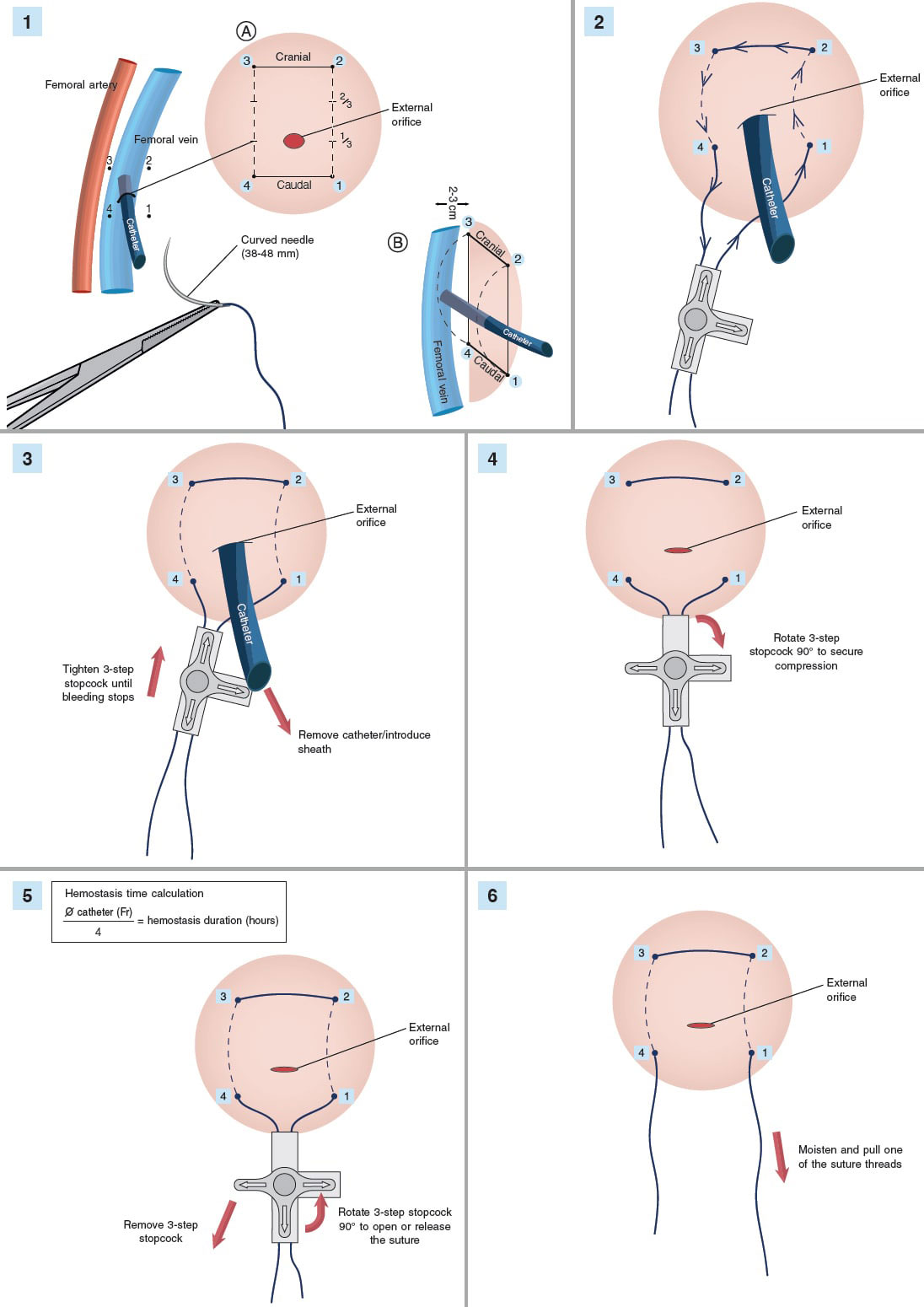
Figure 2. Step-by-step illustration of the simplified purse-string suture with 3-step stopcock.
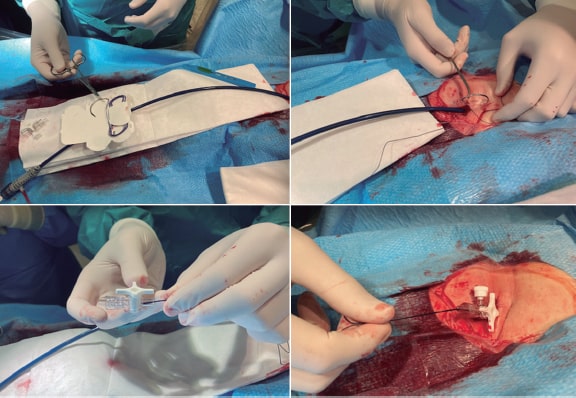
Figure 3. Simplified purse-string suture tightened with 3-step stopcock.
Estimated hemostasis time
An approximate minimum compression time in hours was calculated using the following formula:
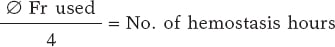
The structural heart procedure followed the standard technique and local protocol in each case via femoral venous access. Patients on antiplatelet therapy (acetylsalicylic acid, clopidogrel, ticagrelor, or prasugrel) continued their regimen before and during the procedure. Patients on oral anticoagulation (vitamin K antagonists or direct oral anticoagulants) received their last dose 12-24 hours prior to the procedure, with no heparin bridging. All patients received perioperative heparin, with an activated clotting time (measured at 30-minute intervals throughout the procedure and at the end before sheath removal) target of 250-300 seconds. No postoperative protamine was administered, even in cases with an activated clotting time ≥ 300 seconds at the end of the procedure.
In the absence of major access complications, anticoagulation therapy was restarted 4-6 hours after the intervention. The postoperative antiplatelet and anticoagulant therapy was based on the patient and procedural characteristics and was not influenced by the type of access closure. For suture removal, it was recommended to moisten the threads with saline solution before extraction, and pain was assessed using a visual analog scale (VAS). Pain during suture placement could not be assessed, as many patients were sedated or anesthetized depending on the procedure.
Inclusion and exclusion criteria
Inclusion criteria were age ≥ 18 years, undergoing a transcatheter procedure via right or left femoral access with an introducer sheath or catheter outer diameter ≥ 10-Fr, and having signed the informed consent form.
Exclusion criteria were ipsilateral arterial access, femoral venous access with an introducer sheath or catheter outer diameter < 10-Fr, more than 1 ipsilateral venous puncture (eg, electrophysiology studies), and lack of consent to participate.
Study endpoints
Primary endpoint
The primary endpoint was to evaluate the efficacy profile of the suture, defined as complete hemostasis of the access site, as well as the safety profile, defined as the absence of major access complications according to the Valve Academic Research Consortium (VARC-3) criteria,27 including vascular injury at the access site (dissection, stenosis, perforation, rupture, arteriovenous fistula, pseudoaneurysm, hematoma, irreversible nerve damage, compartment syndrome, or hemostatic device failure) leading to patient death, major or life-threatening bleeding, visceral ischemia, or neurological injury.
Secondary endpoint
The secondary endpoint was to assess the presence of minor access complications according to the VARC-3 criteria,27 including the already mentioned vascular injuries but not leading to death, life-threatening or major bleeding, visceral ischemia, or neurological injury. Pain at suture removal was assessed using the VAS, and hospital readmissions or access-related events were monitored at the 1-month follow-up.
Ethical considerations
The study fully complied with the privacy policies of each participant center, including rules and regulations on the appropriate use of patient data in research. The study was approved by the Drug Research Ethics Committee of the coordinating center and conducted in full compliance with the criteria set forth in the Declaration of Helsinki. All patients gave their written informed consent form before the procedure and prior to being recruited.
Statistical analysis
As a descriptive cohort study, the statistical analysis was limited to descriptive statistics. Data are expressed as mean ± standard deviation or interquartile range [25-75], and categorical variables as number and percentage. The analysis was conducted using SPSS software (version 25, SPSS Inc., United States).
RESULTS
A total of 222 consecutive patients (223 sutures) were included in the study. Eleven patients were excluded for having vascular accesses < 10-Fr, although similar outcomes were observed regarding safety and efficacy. The study flowchart is shown in figure 1. Baseline characteristics are shown in table 1, illustrating a patient profile of very high thrombotic and hemorrhagic risk. The patients’ mean age was 73.6 ± 9.7 years, with 130 (58%) men, 190 (85%) hypertensive patients, 99 (44%) with diabetes, 143 (64%) with dyslipidemia, 101 (45%) with chronic kidney disease (defined as an estimated glomerular filtration rate < 60 mL/min/m2), and 203 (91%) with a past medical history of atrial fibrillation or flutter. High CHA2DS2-VA (4.1 ± 1.8) and HAS-BLED (3.5 ± 1.6) scores were observed, along with a high prevalence of prior major bleeding events (n = 139, 62%) and ischemic strokes (n = 62, 28%). Regarding concomitant perioperative medication, 119 patients (54%) were on anticoagulation therapy, 53 (24%) on single antiplatelet therapy and 13 (6%) on dual antiplatelet therapy. Preoperative lab test results showed a mean platelet count of 195 350 ± 73 000 and hemoglobin levels of 12.1 ± 2.0 g/dL.
Table 1. Baseline characteristics
| Characteristics | n (%) |
|---|---|
| Age (years) | 73.6 ± 9.7 |
| Male | 130 (58.3%) |
| Body mass index | 28.4 ± 5.5 |
| Smoking status | |
| No | 113 (50.7%) |
| Current smoker | 31 (13.9%) |
| Former smoker | 79 (35.4%) |
| Hypertension | 190 (85.2%) |
| Diabetes mellitus | 99 (44.4%) |
| Dyslipidemia | 143 (64.1%) |
| CKD | 101 (5.3%) |
| Ischemic heart disease | |
| No | 166 (74.4%) |
| PCI | 56 (25.1%) |
| CABG | 1 (0.4%) |
| Stroke | |
| No | 151 (67.4%) |
| Ischemic | 62 (27.8%) |
| Hemorrhagic | 10 (4.5%) |
| Bleeding history | 139 (62.3%) |
| Atrial fibrillation/flutter | 203 (91%) |
| CHA2DS2-VASc score | 4.1 ± 1.8 |
| HAS-BLED score | 3.5 ± 1.6 |
| Anticoagulation | |
| None | 103 (46.2%) |
| VKA | 32 (14.3%) |
| DOAC | 82 (36.8%) |
| Heparin | 6 (2.7%) |
| Antiplatelet therapy | |
| Single | 53 (23.8%) |
| Dual | 13 (5.8%) |
| Baseline blood tests | |
| Hemoglobin (g/dL) | 12.1 ± 2.0 |
| Platelets (per mL) | 195,350 ± 73,000 |
|
DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; CABG, coronary artery bypass grafting; PCI, percutaneous coronary intervention; CKD, chronic kidney disease (estimated glomerular filtration rate < 60 mL/min). |
|
Procedural characteristics (table 2) showed that nearly all access procedures were ultrasound-guided (n = 203, 91%). The most common intervention was transcatheter left atrial appendage closure (n = 154, 69%), followed by mitral valve transcatheter edge-to-edge repair (n = 41, 18%), septal defect closure (n = 10, 4%), and less frequent procedures such as transcatheter mitral valvuloplasty (n = 5, 2.2%). The mean access diameter was 15.6 ± 5.1-Fr, with a median access time of 1.2 minutes [1.6-1.1], and a mean compression time of 4.0 ± 1.7 hours. In 2 complex cases—1 involving a transcatheter left atrial appendage closure and the other MitraClip implantation requiring a new transseptal puncture due to an undersized diameter of the introducer sheath—the suture effectively controlled hemostasis, allowing repositioning of the introducer sheath back to its original diameter. Upon suture removal, the mean pain score assessed via VAS was very low (0.6 ± 1.5). Hemostasis was immediate in all cases (100% success rate), with only 2 minor bleeding events (0.9%) being reported in patients in whom the suture was removed prematurely by inexperienced staff who did not follow the above-mentioned recommendations (see Methods). Despite the option of retightening the suture using the 3-way stopcock, manual compression was chosen and successfully achieved hemostasis. Postoperative lab test results revealed a significant anemia in the patients (preoperative hemoglobin, 12.1 ± 2.0; postoperative hemoglobin, 11.1 ± 2.0; P < .05), likely associated with drugs (frail patients with prior bleeding and, therefore, with an indication for transcatheter left atrial appendage closure) and procedural blood loss. Although one-third of patients (n = 88, 39.6%) required red blood cell transfusion during hospitalization, none did so due to bleeding at the venous puncture site. There were no complications associated with the venous access site at the 1-month follow-up (including infections). Long-term follow-up showed relatively low rates of overall (n = 29, 13%) and cardiovascular mortality (n = 6, 2.7%), considering the high-risk of the study population, with no events at the puncture site.
Table 2. Procedural characteristics
| Characteristics | n (%) |
|---|---|
| Type of procedure | |
| Left atrial appendage closure | 154 (69.1%) |
| Patent foramen ovale | 5 (2.2%) |
| Atrial septal defect | 5 (2.2%) |
| Mitral valvuloplasty | 5 (2.2%) |
| Mitral valve edge-to-edge repair | 41 (18.4%) |
| Paravalvular leak | 3 (1.3%) |
| Pulmonary embolism | 2 (0.9%) |
| Tricuspid edge-to-edge repair | 5 (2.2%) |
| Leadless pacemaker | 1 (0.4%) |
| Access | |
| Right femoral vein | 223 (100%) |
| Ultrasound-guided puncture | 203 (91%) |
| Use of transesophageal echocardiography | 221 (99.1%) |
| Transseptal needle | |
| None | 11 (4.9%) |
| Brockenbrough | 100 (44.8%) |
| Radiofrequency | 111 (49.8%) |
| Access diameter (Fr) | 15.5 ± 5.0 |
| Fluoroscopy time (min) | 21.8 ± 18.5 |
| Contrast volume (mL) | 86 ± 57 |
| Time to perform suture (min) | 0.6 ± 1.5 |
| Compression time (h) | 4.0 ± 1.7 |
| Procedural success | 221 (99.1%) |
| Pain at removal (VAS) | 0.6 ± 1.6 |
| Postoperative hemoglobin (g/dL) | 11.1 ± 2.0 |
| Postoperative red blood cell transfusion | 88 (39.6%) |
| Minor events (VARC-3) at 30 days | |
| Minor bleeding | 2 (0.9%) |
| Others | 0 (0%) |
| Major events (VARC-3) at 30 days | 0 (0%) |
| Long-term follow-up events | |
| All-cause mortality | 29 (13%) |
| Cardiovascular mortality | 6 (2.7%) |
|
VARC-3, Valve Academic Research Consortium 3; VAS, Visual Analogue Scale. |
|
DISCUSSION
The main findings of this study demonstrate that, in a large and contemporary cohort of consecutive patients undergoing transcatheter structural heart procedures with venous access diameters ≥ 10-Fr, the use of this new simplified purse-string suture tightened with a 3-way stopcock was safe, effective, and fast, achieving 100% immediate hemostasis with a very low rate of associated bleeding. Additionally, this method is convenient for both patients and health care staff, as it causes minimal discomfort during removal and allows for short compression times, while facilitating early ambulation. Furthermore, the materials are widely available in any hospital setting and are very low-cost.
In recent years, the number and indications for structural heart procedures have grown exponentially.1 Despite this development, vascular access complications are still the most common finding at the cath lab.28 With the expansion of structural heart procedures, due to the size of the devices or their mechanisms, it is necessary to use large-bore introducer sheaths being the diameter of the access site one of the most important predictors of vascular complications.29 Transfemoral venous access with large-bore introducer sheaths is widely used in various conditions.2-6 Although ultrasound-guided puncture is recommended due to its superior safety and efficacy profile, there are no clinical practice guidelines specifying the optimal method or device for achieving hemostasis. Traditional methods such as manual compression and weight-based techniques exist, along with more state-of-the-art approaches like subcutaneous sutures (figure-of-eight, purse-string) or arterial closure devices repurposed for venous use (both suture-based and collagen plugs).7 Table 3 provides a comparative summary of subcutaneous suture techniques reported in the literature.
Table 3. Summary of major studies on hemostasis with subcutaneous suture
| Authors and year | n | Age (years) | Type of hemostasis | Study design | Main procedures | Mean diameter of introducer sheath | Ultrasound | Time to hemostasis (min) | Time to ambulation (h) | Length of stay (h) | Major Complications | Minor Complications | Comments |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Camacho et al., 2025* SUTURE-3 | 222 | 74 | Simplified purse-string suture + 3-way stopcock | Multicenter, observational, prospective | LAAC TMVR PFO ASD | 16-Fr 10-26-Fr | Yes | < 0.5 min | 4 h | – | 0% | 0.9% | – |
| Geis et al., 201530 | 80 | 71 | FOES + MC 12 h (n = 40) ProGlide (n = 40) | Single-center, observational, prospective | TMVR (MitraClip) | 24-Fr | – | – | – | – | 0% 2.5% | 22% 15% | – |
| Steppich et al., 201813 | 277 | 74 | FOES (n = 150) ProGlide (n = 127) | Single-center, retrospective, registry-based | TMVR (MitraClip) | 24-Fr | – | – | – | – | 2.7% 3.1% NS | 15.3% 15.7% NS | – |
| Pracon et al., 20176 | 86 | 62 | MC (n = 30), Parallel Z-suture (n = 60) | Single-center, open-label, 2:1 randomized | LAAC PMC ASD | 10-22-Fr 14-Fr | – | 12 min < 1 min | 16 h 7 h | – | – | 37% 13% | Modified parallel Z-suture |
| Mohammed et al., 20225 VACCAR | 434 | 64 | MC (n = 156) FOES (n = 203) ProGlide (n = 75) | Single-center, observational, prospective | AF ablation | 8-15-Fr | Yes | 20 min 7 min 9 min | 6.5 h 2.2 h | 29 h 27.5 h | 1% 0% 0% | 2.6% 1.5% 4.0% | Pain reported |
| Traullé et al., 201615 | 124 | 58 | FOES | Single-center, observational | AF cryoablation | 15-Fr | – | – | – | – | – | 2.4% | – |
| Aytemir et al., 201617 | 200 | 55 | MC (n = 100) FOES (n = 100) | Single-center, observational, retrospective | AF cryoablation | 15-Fr | – | 14 min 0 min | – | – | – | 4% 0% | – |
| Issa and Amr, 201522 | 376 | 65 | MC (n = 253) FOES (n = 123) | Single-center, observational | AF ablation | 11.5-Fr | Yes | – | – | – | 4.7% 2.4% | 7.5% 7.3% | – |
| Velagic et al., 202116 | 190 | 60 | MC (n = 100) FOES (n = 90) | Unicéntrico Retrospectivo | AF cryoablation | 15-Fr | Yes | – | – | – | 0% 0% | 8.8% 7% | Longer electrophysiology lab times |
| Jensen et al., 202019 | 290 | 66 | MC (n = 132) FOES (n = 158) | Single-center, retrospective | Procedures EP LAAC ASD | 14.9-Fr 14.8-Fr | – | – | – | – | – | 6.8% 1.3% | Longer device exchange times |
| Yorgun et al., 201914 | 150 | 64 | FOES (n = 75) FOES + 3-way stopcock (n = 75) | Single-center, observational, prospective | AF cryoablation | 15-Fr | – | 1.66 ± 0.32 0.78 ± 0.24 | 4 h 4 h | 1.3 ± 0.6 1.2 ± 0.4 | 2.7% rebleeding, 2.7% infection 0% | 12% 0% | – |
| Joshua et al., 202131 | 4 | FOES + Flowstasis (Inari) | Single-center, case series | Thrombectomy in DVT/PE, TIPS, leadless pacemaker | 16-27-Fr | – | – | – | – | – | – | – | |
| Kumar et al., 20198 | 749 | 13 | MC (n = 365) + FOES + Fisherman’s knot (n = 384) | Single-center, randomized | PMC, ASD, PFO, pulmonary valvuloplasty | 10-14-Fr 12-Fr | Yes | 14.3 min 1.1 min | 18.9 h 3.3 h | 36.8 h 24.6 h | – | 1.6% 0.2% | – |
| Morgan et al., 201220 | 26 | 7.8 | MC (n = 30) S8 (n = 26) | Single-center, retrospective registry | Pediatric interventions | 6-22-Fr 9.1-Fr | – | 13.6 min 10 min | – | – | – | 6.1% 7.7% | – |
| Zhou et al., 201421 | 104 | 8.1 | FOES | Single-center, observational | ASD Pulmonary stenosis | 7-14-Fr | – | – | – | 5.6 ± 2.2 d | – | 1.9% | – |
| Jackson et al., 201926 GITAR | 200 | 62 | MC Purse-string suture | Randomized | AF ablation | 8.5-15-Fr | – | 10.4 ± 2.2 min 0.45 ± 2.0 min | – | – | – | – | Pain reported 29% 15% |
| Kottmaier et al., 201725 | 784 | 63 | MC (n = 322) Purse-string suture (n = 462) | Single-center, observational | AF ablation | 8 + 11.7-Fr | Yes | – | 10 h 12 h | – | 0% 0% | 24% 27% | Ultrasound later |
| Akkaya et al., 202024 | 41 | 80 | Purse-string suture | Single-center, observational | TMVR (MitraClip) | 24-Fr | – | – | – | – | 0% | 14.6% | Without protamine |
| Kypta et al., 201623 | 77 | 80 | Double purse-string suture | Single-center, observational | Leadless pacemaker | 18-23-Fr | – | – | – | – | – | 3.9% (2.6% hematoma, 1.3% fistula) | – |
|
AF, atrial fibrillation; ASD, atrial septal defect; DVT, deep vein thrombosis; EP, electrophysiology; FOES, figure-of-eight suture; LAAC, left atrial appendage closure; MC, manual compression; NS, non-significant; PFO, patent foramen ovale; PMC, percutaneous mitral commissurotomy; PTE, pulmonary embolism; TIPS, transjugular intrahepatic portosystemic shunt; TMVR, transcatheter mitral valve repair. * Current study. |
|||||||||||||
The proposed mechanism by which this simplified purse-string suture with a 3-way stopcock achieves hemostasis is similar to the mechanism of other figure-of-eight techniques.18 The suture passes medial and lateral to the access site, engaging sufficient skin and subcutaneous tissue to generate a mechanical tamponade around the venous puncture site. In the study by Cilingiroglu et al.,18 there was no extravasation after suture placement, and only mild venoconstriction was noted on phlebography; however, at 24 hours, ultrasound confirmed normal compressibility in the absence of stenosis, thrombosis, or other complications. Further studies have not found this stenosis using ultrasound either.6
Figure-of-eight suture has been used for venous hemostasis in various studies proving superior to conventional compression: faster compression, a reduced length of stay, fewer complications, etc.5,6,8,12-21 However, most of these studies were single-center and primarily focused on electrophysiology; only a few addressed hemostasis in structural heart procedures.6,8,30
The purse-string suture is an even easier alternative than the figure-of-eight, and uses a large non-absorbable braided suture on a curved needle that is passed in-and-out at 4 points around the introducer sheath to form a square. Pulling and tightening the suture compresses the subcutaneous tissue and exerts pressure on the puncture. Although the evidence is more limited, 4 studies have demonstrated that it is safe and effective procedures, with similar outcomes compared with the figure-of-eight suture and improved results compared with manual compression, including shorter hemostasis and immobilization times and less discomfort.23-26 Furthermore, this evidence includes one of the few randomized trials to date (albeit 2-center and in electrophysiology) showing excellent results and evaluating pain compared with manual compression.26 In our study, discomfort during suture removal was minimal, as reflected in VAS scores, which is an important consideration in promoting the wider adoption of the technique.
In general, when 2 methods yield similar results, the easier or less invasive one is considered superior. The study of this new simplified purse-string technique reveals an excellent safety (0.9% minor bleeding) and efficacy profile (immediate hemostasis, completion time of 1-2 minutes, maintenance time < 6 hours in all cases). Moreover, it provides some potential advantages over other techniques:
- –Only 2 instead of 4 needle punctures and subcutaneous trajectories are needed.
- –The 2 trajectories run parallel to the introducer sheath, theoretically reducing any puncturing risks.
- –Unlike traditional knots, tension is adjustable at any time using a 3-way stopcock. This allows intraoperative suture placement when downsizing the introducer sheath or catheter (eg, repositioning, new transseptal puncture, etc.) and when bleeding needs to be avoided.
- –Unlike more complex closure devices, the learning curve is not steep and is easily manageable by trained cath lab staff.
- –Availability and cost of this type of hemostasis make it ideal for use in developing countries or to contain health care spending, especially compared with devices such as the ProStyle or the MANTA.
Limitations
The main limitations stem from the study design. Although multicenter, it was not randomized and did not include a comparison group using other hemostasis techniques. The study lacks a control group, and therefore, while comparisons can be drawn with historical studies and registries, data should be interpreted with caution. Furthermore, the study does not evaluate procedures with multiple venous punctures, such as the ones typically used in electrophysiology procedures, even though the utility of this type of suture in such procedures has been widely confirmed. Additionally, the study will not be conducted in patients with an ipsilateral transcatheter arterial line inserted at the same time, as this could be considered a confounding factor if any events occur.
CONCLUSIONS
The simplified purse-string suture with a 3-way stopcock for venous hemostasis in structural heart procedures with ≥ 10-Fr access is a simple, safe, effective, and low-cost procedure, comfortable for both the patient and health care personnel, which achieves immediate hemostasis with a short compression time.
FUNDING
None declared.
ETHICAL CONSIDERATIONS
The study followed the privacy policies of each participant center, including rules and regulations on the appropriate use of data derived from patient research. The study was approved by the Drug Research Ethics Committee of the coordinating center and conducted in full compliance with the principles set forth in the Declaration of Helsinki. All patients signed a specific informed consent form before the procedure and prior to being recruited. The SAGER guidelines regarding potential sex or gender bias were followed.
STATEMENT ON THE USE OF ARTIFICIAL INTELLIGENCE
The authors declare that artificial intelligence was not used at any point for drafting, data analysis, or knowledge extraction.
AUTHORS’ CONTRIBUTIONS
S.J. Camacho Freire: conception and design of the suture and the study; patient enrollment; data acquisition, analysis, and interpretation; article drafting; creation of figures and tables. M.A. Montilla Garrido and E. Izaga Torralba: patient enrollment; data acquisition, analysis, and interpretation; critical review of the article. J. León Jiménez, P. Gómez Fernández, I. Sánchez Fernández, J. Roa Garrido, F.J. Landero García, O. Lagos De Grande, M.E. Gracia Hiraldo, C. Lluch Requerey, J.F. Díaz Fernández, and A.E. Gómez Menchero: patient enrollment; data acquisition; critical review of the article. F. Navarro Roldán: critical review of the article.
CONFLICTS OF INTEREST
None declared.
ACKNOWLEDGMENTS
To Elisa Muñoz Navarro, for digitalization and illustration development.
WHAT IS KNOWN ABOUT THE TOPIC?
- Various sutures (purse-string and figure-of-eight) used for hemostasis in transcatheter large-bore venous procedures are safe and effective, offering advantages over conventional compression methods and specific vascular closure devices.
WHAT DOES THIS STUDY ADD?
- A new simplified suture technique for large-bore femoral venous hemostasis had safety and efficacy outcomes similar to former techniques. Additionally, it has several advantages over other sutures and vascular closure devices: it is easier to use, requires fewer punctures and subcutaneous trajectories, avoids trajectories perpendicular to the catheter, can be adjusted at any time with a 3-way stopcock (if catheter downsizing is required), and is readily available and low-cost.
REFERENCES
1. Ojeda S, Romaguera R, Cruz-González I, Moreno R. Spanish Cardiac Catheterization and Coronary Intervention Registry. 29th Official Report of the Interventional Cardiology Association of the Spanish Society of Cardiology (1990-2019). Rev Esp Cardiol. 2020;73:927-936.
2. Hosseini SM, Rozen G, Saleh A, et al. Catheter Ablation for Cardiac Arrhythmias:Utilization and In-Hospital Complications, 2000 2013. JACC Clin Electrophysiol. 2017;3:1240-1248.
3. Ali M, Shreenivas SS, Pratt DN, Lynch DR, Kereiakes DJ. Percutaneous Interventions for Secondary Mitral Regurgitation. Circ Cardiovasc Interv. 2020;13:008998.
4. Reynolds D, Duray GZ, Omar R, et al. A Leadless Intracardiac Transcatheter Pacing System. N Engl J Med. 2016;374:533-541.
5. Mohammed M, Ramirez R, Steinhaus DA, et al. Comparative outcomes of vascular access closure methods following atrial fibrillation/flutter catheter ablation:insights from VAscular Closure for Cardiac Ablation Registry. J Interv Card Electrophysiol. 2022;64:301-310.
6. Pracon R, Bangalore S, Henzel J, et al. A randomized comparison of modified subcutaneous “Z“-stitch versus manual compression to achieve hemostasis after large caliber femoral venous sheath removal. Catheter Cardiovasc Interv. 2018;91:105-112.
7. Bin Waleed K, Leung LW, Akhtar Z, Sohal M, Zuberi Z, Gallagher MM. New approaches to achieving hemostasis after venous access in cardiovascular patients. Kardiol Pol. 2022;80:750-759.
8. Kumar P, Aggarwaal P, Sinha SK, et al. Efficacy and Safety of Subcutaneous Fellow's Stitch Using “Fisherman's Knot“Technique to Achieve Large Caliber ( >10 French) Venous Hemostasis. Cardiol Res. 2019;10:303-308.
9. Shaw JA, Dewire E, Nugent A, Eisenhauer AC. Use of suture-mediated vascular closure devices for the management of femoral vein access after transcatheter procedures. Catheter Cardiovasc Interv. 2004;63:439-443.
10. Barbetta I, van den Berg JC. Access and hemostasis:femoral and popliteal approaches and closure devices —why, what, when, and how?Semin Intervent Radiol. 2014;31:353-360.
11. KupóP, Pap R, Sághy L, et al. Ultrasound guidance for femoral venous access in electrophysiology procedures-systematic review and meta-analysis. J Interv Card Electrophysiol. 2020;59:407-414.
12. Lakshmanadoss U, Wong WS, Kutinsky I, Khalid MR, Williamson B, Haines DE. Figure-of-eight suture for venous hemostasis in fully anticoagulated patients after atrial fibrillation catheter ablation. Indian Pacing Electrophysiol J. 2017;17:134-139.
13. Steppich B, Stegmüller F, Rumpf PM, et al. Vascular complications after percutaneous mitral valve repair and venous access closure using suture or closure device. J Interv Cardiol. 2018;31:223-229.
14. Yorgun H, Canpolat U, Ates AH, et al. Comparison of standard vs modified “figure-of-eight“suture to achieve femoral venous hemostasis after cryoballoon based atrial fibrillation ablation. Pacing Clin Electrophysiol. 2019;42:1175-1182.
15. TraulléS, Kubala M, Doucy A, Quenum S, Hermida JS. Feasibility and safety of temporary subcutaneous venous figure-of-eight suture to achieve haemostasis after ablation of atrial fibrillation. Europace. 2016;18:815-819.
16. Velagic V, Mugnai G, Pasara V, et al. Use of figure of eight suture for groin closure with no heparin reversal in patients undergoing cryoballoon ablation for atrial fibrillation. J Interv Card Electrophysiol. 2021;60:433-438.
17. Aytemir K, Canpolat U, Yorgun H, et al. Usefulness of 'figure-of-eight'suture to achieve haemostasis after removal of 15-French calibre femoral venous sheath in patients undergoing cryoablation. Europace. 2016;18:1545-1550.
18. Cilingiroglu M, Salinger M, Zhao D, Feldman T. Technique of temporary subcutaneous “Figure-of-Eight“sutures to achieve hemostasis after removal of large-caliber femoral venous sheaths. Catheter Cardiovasc Interv. 2011;78:155-160.
19. Jensen CJ, Schnur M, Lask S, et al. Feasibility of the Figure-of-8-Suture as Venous Closure in Interventional Electrophysiology:One Strategy for All?Int J Med Sci. 2020;17:965-969.
20. Morgan GJ, Waragai T, Eastaugh L, Chaturvedi RC, Lee KJ, Benson L. The fellows stitch:large caliber venous hemostasis in pediatric practice. Catheter Cardiovasc Interv. 2012;80:79-82.
21. Zhou Y, Guo Z, Bai Y, et al. Femoral venous hemostasis in children using the technique of “figure-of-eight“sutures. Congenit Heart Dis. 2014;9:122-125.
22. Issa ZF, Amr BS. Venous hemostasis postcatheter ablation of atrial fibrillation while under therapeutic levels of oral and intravenous anticoagulation. J Interv Card Electrophysiol. 2015;44:97-104.
23. Kypta A, Blessberger H, Lichtenauer M, et al. Subcutaneous Double “Purse String Suture“—A Safe Method for Femoral Vein Access Site Closure after Leadless Pacemaker Implantation. Pacing Clin Electrophysiol. 2016;39:675-679.
24. Akkaya E, Sözener K, Rixe J, et al. Venous access closure using a purse-string suture without heparin antagonism or additional compression after MitraClip implantation. Catheter Cardiovasc Interv. 2020;96:179-186.
25. Kottmaier M, Bourier F, Reents T, et al. Safety and Feasibility of Subcutaneous Purse-String Suture of the Femoral Vein After Electrophysiological Procedures on Uninterrupted Oral Anticoagulation. Am J Cardiol. 2017;119:1781-1784.
26. Jackson N, McGee M, Ahmed W, et al. Groin Haemostasis With a Purse String Suture for Patients Following Catheter Ablation Procedures (GITAR Study). Heart Lung Circ. 2019;28:777-783.
27. Généreux P, Piazza N, Alu MC, et al. Valve Academic Research Consortium 3:Updated Endpoint Definitions for Aortic Valve Clinical Research. J Am Coll Cardiol. 2021;77:2717-2746.
28. Wiley JM, White CJ, Uretsky BF. Noncoronary complications of coronary intervention. Catheter Cardiovasc Interv. 2002;57:257-265.
29. Moussa Pacha H, Al-Khadra Y, Soud M, Darmoch F, Moussa Pacha A, Alraies MC. Percutaneous devices for left atrial appendage occlusion:A contemporary review. World J Cardiol. 2019;11:57-70.
30. Geis NA, Pleger ST, Chorianopoulos E, Müller OJ, Katus HA, Bekeredjian R. Feasibility and clinical benefit of a suture-mediated closure device for femoral vein access after percutaneous edge-to-edge mitral valve repair. EuroIntervention. 2015;10:1346-1353.
31. Joshua CH, Jeffrey BH, Elliott MG. Achievement of rapid venous hemostasis following large-bore catheter procedures. CathLab Digest. 2021. Available at:https://www.hmpgloballearningnetwork.com/site/cathlab/achievement-rapid-venous-hemostasis-following-large-bore-catheter-procedures. Accessed 1 Feb 2025.