ABSTRACT
Introduction and objectives: The optimal time to perform a diagnostic coronary angiography in patients admitted due to non-ST-segment elevation acute coronary syndrome (NSTEACS) and start pretreatment with dual antiplatelet therapy is controversial. Our study aims to identify the current diagnostic and therapeutic approach, and clinical progression of patients with NSTEACS in our country.
Methods: The IMPACT-TIMING-GO trial (Impact of time of intervention in patients with myocardial infarction with non-ST segment elevation. Management and outcomes) is a national, observational, prospective, and multicenter registry that will include consecutive patients from 24 Spanish centers with a clinical diagnosis of NSTEACS treated with diagnostic coronary angiography and with present unstable or causal atherosclerotic coronary artery disease. The study primary endpoint is to assess the level of compliance to clinical practice guidelines in patients admitted due to NSTEACS undergoing coronary angiography in Spain, describe the use of antithrombotic treatment prior to cardiac catheterization, and register the time elapsed until it is performed. Major adverse cardiovascular events will also be described like all-cause mortality, non-fatal myocardial infarction and non-fatal stroke, and the rate of major bleeding according to the BARC (Bleeding Academic Research Consortium) scale at 1- and 3-year follow-up.
Results: This study will provide more information on the impact of different early management strategies in patients admitted with NSTEACS in Spain, and the degree of implementation of current recommendations into the routine clinical practice. It will also provide information on these patients’ baseline and clinical characteristics.
Conclusions: This is the first prospective study conducted in Spain that will be reporting on the early therapeutic strategies—both pharmacological and interventional—implemented in our country in patients with NSTEACS after the publication of the 2020 European guidelines, and on the clinical short- and long-term outcomes of these patients.
Keywords: Acute coronary syndrome. Acute myocardial infarction. Non-ST-segment elevation acute coronary syndrome. Dual antiplatelet therapy. Pretreatment. Early invasive strategy. ESC guidelines. Diabetes mellitus. Hemorrhage. Revascularization.
RESUMEN
Introducción y objetivos: El momento óptimo para la realización de un cateterismo diagnóstico en pacientes con síndrome coronario agudo sin elevación del segmento ST (SCASEST) y la necesidad de pretratamiento con doble antiagregación son motivo de controversia. Este estudio pretende conocer el abordaje diagnóstico y terapéutico actual, así como la evolución clínica de los pacientes con SCASEST en España.
Métodos: El estudio IMPACT of Time of Intervention in patients with Myocardial Infarction with Non-ST seGment elevation. ManaGement and Outcomes (IMPACT-TIMING-GO) es un registro nacional observacional, prospectivo y multicéntrico, que incluirá pacientes consecutivos con diagnóstico de SCASEST tratados con coronariografía diagnóstica y que presenten enfermedad coronaria aterosclerótica inestable o causal en 24 centros españoles. El objetivo primario del estudio es conocer el grado de cumplimiento de las recomendaciones de las guías de práctica clínica en pacientes que ingresan por SCASEST tratados con coronariografía en España, describir el uso del tratamiento antitrombótico antes del cateterismo y determinar el tiempo hasta este en la práctica clínica real. Se describirán también los eventos adversos cardiovasculares mayores: mortalidad por cualquier causa, infarto no fatal e ictus no fatal, y también la incidencia de hemorragia mayor según la escala BARC (Bleeding Academic Research Consortium) durante el seguimiento a 1 y 3 años.
Resultados: Este registro permitirá mejorar el conocimiento en relación con el abordaje terapéutico inicial en pacientes que ingresan por SCASEST en España. Contribuirá a conocer sus características basales y su evolución clínica, así como el grado de adherencia y cumplimiento de las recomendaciones de las que se dispone actualmente.
Conclusiones: Se trata del primer estudio prospectivo realizado en España que permitirá conocer las estrategias terapéuticas iniciales, tanto farmacológicas como intervencionistas, que se realizan en nuestro país en pacientes con SCASEST tras la publicación de las guías europeas de 2020, y la evolución clínica de estos pacientes a corto y largo plazo.
Palabras clave: Síndrome coronario agudo. Infarto agudo de miocardio. Síndrome coronario agudo sin elevación del segmento ST. Doble antiagregación plaquetaria. Pretratamiento. Coronariografía precoz. Guía ESC. Diabetes mellitus. Hemorragia. Revascularización.
Abbreviations IMPACT-TIMING-GO: Impact of Time of Intervention in patients with Myocardial Infarction with non-ST segment elevation ManaGement and Outcomes. SCA: síndrome coronario agudo. SCASEST: síndrome coronario agudo sin elevación del segmento ST.
INTRODUCTION
Ischemic heart disease is the leading cause of mortality in developed countries.1 The rate of acute coronary syndrome (ACS), specially non-ST-segment elevation ACS (NSTEACS), has increased over the last few years, in part, due to the ageing of the population.2-3 Given the underlying pathophysiology4 patients receive specific antithrombotic treatment, and invasive approach is used in most of the cases.1-3 The new guidelines published by the European Society of Cardiology (ESC) on the management of NSTEACS1 include changes compared to the guidelines published back in 2016. The most significant ones include antithrombotic treatment, the revascularization strategy, and several controversial innovations.
In the guidelines published in 2020, early cardiac catheterization within the first 24 hours after admission was advised (level of evidence IA) in patients diagnosed with acute myocardial infarction with GRACE scores (Global Registry of Acute Coronary Events) > 140 or dynamic electrocardiographic changes suggestive of ischemia.1 Also, the previous window of recommendation of 0 to 72 hours for moderate risk patients is now gone.4 On the other hand, the systematic use of pretreatment at admission with an P2Y12 inhibitor antiplatelet drug (ticagrelor, prasugrel or clopidogrel) in patients to be treated with an early invasive strategy is now ill-advised.1
The objective of the IMPACT registry (Time of intervention in patients with myocardial infarction with non-ST segment elevation, management and outcomes [IMPACT-TIMING-GO]) is to get the big picture on the current treatment of NSTEACS, in Spain, in association with catheterization times, use of pretreatment in these patients, and describe the possible prognostic implications of the different strategies used in real life.
METHODS
Study design and population
This is an observational, prospective, multicenter, and nationwide registry that will include all consecutive patients admitted with a diagnosis of NSTEACS to the different participant centers, treated with diagnostic coronary angiography, and with unstable or causal atherosclerotic disease regardless of further treatment administered by the heart team. The baseline characteristics of the patients included, and their clinical progression regarding in-hospital events will be studied. Patients will undergo a 1-and-3-year clinical follow-up period.
This registry has been promoted by the Spanish Society of Cardiology Young Cardiologists Working Group with scientific support from the Spanish Society of Cardiology Research Agency. Also, it has been approved by different Research Ethics Committees with drugs from all the participant hospitals. Finally, it has been designed according to the STROBE checklist for observational studies.
The list of centers that will eventually participate in the registry is shown on figure 1. Inclusion and exclusion criteria are shown on table 1. The presence of elevated markers of myocardial damage or electrocardiographic changes is not mandatory. Patients with a clinical diagnosis of unstable angina can be included as long as coronary angiography confirms the clinical diagnosis.
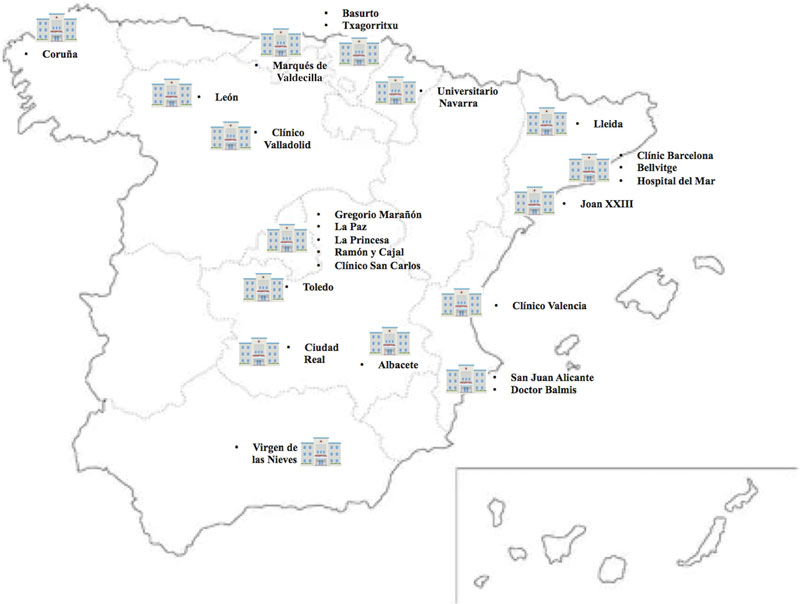
Figure 1. Map with the Spanish participant centers in the IMPACT-TIMING-GO registry.
Table 1. Inclusion and exclusion criteria of the IMPACT-TIMING-GO registry
| Inclusion criteria |
|---|
| NSTEACS with in-hospital invasive treatment regardless of when it is performed. |
| Evidence of causal or unstable atherosclerotic disease. |
| Age ≥ 18 years. |
| Capacity to give informed consent. |
| Exclusion criteria |
| Minors and those who withdraw their consent to be included or followed at any time during the study. |
| Assessment of myocardial damage markers associated with type 2 myocardial infarction. |
| Patients without any signs of coronary artery disease including those with myocarditis, Prinzmetal angina, takotsubo syndrome or MINOCA. |
| Patients diagnosed with spontaneous coronary artery dissection. |
| Patients with complete left bundle branch block or pacemaker rhythm on the electrocardiogram performed at admission. |
| Patients with a valve heart disease eligible for surgery. |
| Patients with a known past medical history of diffuse coronary artery disease noneligible for revascularization. |
| Patients with known or confirmed allergy to some antiplatelet drug. |
IMPACT-TIMING-GO, IMPACT of time of intervention in patients with myocardial infarction with non-ST segment elevation. Management and outcomes; MINOCA, Myocardial infarction with non-obstructive coronary artery disease; NSTEACS, non-ST-segment elevation acute coronary syndrome. |
Endpoints
The study primary endpoint is to know the degree of compliance of the recommendations included in the clinical practice guide-lines in patients admitted due to NSTEACS treated with coronary angiography, in Spain, describe the use of antithrombotic treatment before cardiac catheterization, and the time elapsed until it is performed in the real-world clinical practice.
The secondary endpoints are:
- – To describe the baseline, clinical, and epidemiological characteristics of the study population.
- – To study the rates of cardiovascular mortality, new revascularization, stent thrombosis, and hospitalizations due to heart failure during admission and at the 1-and-3-year follow-up.
- – To describe major cardiovascular adverse events of all-cause mortality, non-fatal stroke, non-fatal infarction, and the rate of major bleeding grades 3, 4, and 5 according to the BARC scale (Bleeding Academic Research consortium.5) Data will be analyzed during admission and at the 1-and-3-year follow-up.
- – To know the medical treatment at discharge and at follow-up of patients discharged in Spain after NSTEACS.
- – To know the degree of control of the different cardiovascular risk factors associated with the endpoints defined in the ESC guidelines 2021 on prevention of cardiovascular disease in the routine clinical practice.6
Data curation and definitions
Data will be collected prospectively by trained medical investigators from each participant center in a specific standard form. Demographic data, the baseline clinical characteristics, and all analytical, electrocardiographic, and echocardiographic data will be included as well.
Similarly, data on disease progression and the in-hospital stay, indication for coronary angiography and when it is be performed, type of treatment received (conservative, stent implantation or revascularization surgery), and the in-hospital complications occurred (hemorrhages and severity, heart failure or shock, reinfarction, stroke, confusional state, mechanical and arrhythmic complications, infectious complications requiring antibiotic therapy, and mortality causes) will be collected. Finally, the medical treatment at hospital discharge and level of compliance of the current recommendations based on the clinical practice guidelines will be studied too.
The definitions of the variables are shown on table 2.7-8
Table 2. Definitions of target variables
| Variable | Definition |
|---|---|
| All-cause mortality | All deaths regardless of their cause. |
| Cardiovascular death | All deaths of vascular causes both cardiac (heart failure/shock; malignant arrhythmias; myocardial infarction) and non-coronary vascular including cerebrovascular disease, pulmonary embolism, aneurysms/aortic dissections, acute ischemia of lower limbs, etc. All sudden deaths of unknown causes will be adjudicated as cardiovascular death. |
| Non-cardiac death | All deaths that do not meet the previous definition like deaths due to infections, cancer, pulmonary diseases, accidents, suicide or trauma. |
| Myocardial infarction | It is defined based on the criteria established in the 4th and current Universal definition.4 Therefore, patients with type 2 infarction, extracardiac causes or without elevated markers of myocardial damage were excluded. |
| Stroke/Transient ischemic attack | New-onset neurological, focal or global deficit due to ischemia or hemorrhage, and as long as it is part of diagnostic judgement at hospital discharge. |
| Stent thrombosis | Defined based on the Academic Research Consortium of randomized clinical trials with stents.7 |
| New revascularization | All unscheduled revascularizations performed after hospital discharge, whether surgical or percutaneous, including target vessel failure and target lesion failure. |
| Admission due to heart failure | Unscheduled hospital admission > 24 hours with a primary diagnosis of heart failure based on the current defintion.8 |
Follow-up
Clinical follow-up to detect events will be conducted by medical investigators through on-site visits, health record reviews or phone calls with the patient, family members or treating physician at 1 and 3 years. Clinical variables, functional class, and additional variables (analytical, electrocardiographic, and echocardiographic, and treatment received) will be included. The overall mortality rate and its causes, need of emergency hospitalization (duration > 24 hours) and its causes, and the rates of non-fatal infarction and stroke will be collected as well. All deaths due to myocardial infarction, sudden death or heart failure will be considered cardiovascular deaths.
Sample size estimate
Taking the events seen in previous studies with a population of similar characteristics as the reference,9-14 a sample size of 800 patients will be enough to know the baseline characteristics of the study population, and the therapeutic approach currently used in Spain in our routine clinical practice. Patients lost to follow-up will be handled by multiple imputation.
Statistical analysis
Categorical variables will be expressed as number and percentage. Quantitative variables will be expressed as mean ± standard deviation. Quantitative variables with normal distribution will be expressed as median and interquartile range [25%-75%]. The normal distribution of quantitative variables will be assessed using the Kolmogorov-Smirnoff test. Regarding the reference variables, Student t test will be used to compare quantitative variables, and the chi-square test or Fisher’s exact test, if applicable, to compare categorical variables. Statistical analysis will be performed using the SPSS statistical software version 22.0 (IBM Corp., Armonk, United States).
Specific studies on subgroups of special interest will be conducted: feminine sex, patients ≥ 75 years, those with GRACE scores > 140, diabetic patients, those with a past medical history of renal failure, with an indication for chronic oral anticoagulation, with multivessel disease, acute myocardial infarction, ventricular dysfunction according to the current clinical practice guidelines and based on the day of admission (holiday vs working day), and patients who require transfer to tertiary centers to receive a coronary angiography.
Ethical principles
Inclusion in the study will not imply changes to the patients’ treatment. Instead, it will follow the routine clinical practice and the recommendations set forth by the current clinical practice guidelines. Therefore, antithrombotic treatment and additional examinations including the need for a coronary angiography and the time it is performed will all be decided by the heart team based on the routine clinical practice. Coronary angiography, vascular access, antithrombotic treatment during the procedure, and the material and devices used will all be decided by the treating operator in charge of the case. All patients will sign a written informed consent form before being included in the study that will be conducted in full compliance with the Declaration of Helsinki. This study will also observe all legal regulations applicable to this type of studies and follow the good clinical practice rules while being conducted.
DISCUSSION
The IMPACT-TIMING-GO registry will give us information on the current real-world management of patients with NSTEACS with invasive treatment and causal coronary artery disease, which will allow us to assess the degree of implementation of the current recommendations of ESC guidelines 2020 on cardiac catheterizations performed within the first 24 hours and no pretreatment with P2Y12 inhibitors. Similarly, different prognostic differences that early invasive treatment and no pretreatment could have in the real life of patients diagnosed with NSTEACS could be suggested.
Despite the clinical practice guidelines recommendations on the invasive treatment of patients with NSTEACS, the main clinical trials published to this date have been unable to demonstrate any clear benefits from systematic early invasive treatment.9-14 The VERDICT trial,9 published in 2018, randomized 2147 patients with NSTEACS on a 1:1 ratio to receive early (< 12 hours) or delayed (48 to 72 hours) cardiac catheterization. No significant differences were found in the composite endpoint of major cardiovascular events at 4-year follow-up. However, in the subgroup of patients with GRACE scores > 140 statistically significant differences were seen favorable to the early strategy regarding major adverse cardiovascular events (hazard ratio, 0.81; 95% confidence interval, 0.67-1.01; P = .023). Consistent with this, the TIMACS clinical trial10 published in 2008 of 3031 patients with NSTEACS found no differences in the primary endpoint when early invasive strategy (< 24 hours) and delayed approach (> 36 hours) were compared, except for, once again, in patients with GRACE scores > 140. Other randomized clinical trials with fewer patients show contradictory results11-14 some without significant differences.15 Also, in many cases, the results favorable to the early strategy are associated with refractory ischemia, not with hard endpoints like cardiovascular mortality or non-fatal myocardial infarction. In Spain, evidence on the management of NSTEACS is prior to the current clinical practice guidelines,16-17 and the most recent registry is retrospective, which is suggestive of a possible mortality benefit in patients with GRACE scores > 140.18 Over the last 2 decades, in our country, the use of an invasive strategy in patients with NSTEACS has increased significantly from 20% in the MASCARA registry in 200516 up to 80% in the DIOCLES study from 2012.17 However, evidence is scarce on catheterization times, our capacity to adapt to current recommendations (the median time of the DIOCLES trial was 3 days), the possible impact this time reduction can have, and on the consequencies from not starting antiplatelet pretreatment in patients who don’t meet the times recommended.
On the other hand, the current formal recommendation from the clinical practice guidelines of not pretreating systematically with a P2Y12 inhibitor (level of recommendation IIIA1) patients on early invasive treatment is mainly based on 3 clinical trials and their meta-analysis.19 In the ACCOAST trial, pretreatment with prasugrel did not reduce thrombotic events in patients with NSTEMI. However, cardiac surgery-related and potentially fatal hemorrhages increased.20 We should mention that the median time elapsed since the prasugrel loading dose until the coronary angiography was performed was 4 hours. In the ISAR-REACT 5 trial published in 2019, a non-pretreatment strategy with prasugrel in patients with ACS vs pretreatment with ticagrelor proved superior regarding the primary endpoint of thrombotic events with a tendency towards fewer hemorrhagic events.21 We should mention that the intrinsic effect of the drug used should not be obviated or else the fact that the median time elapsed since randomization until the prasugrel loading dose was received in the non-pretreatment group was 60 minutes. Finally, the first study that compared 2 different pretreatment strategies vs the intraoperative administration of ticagrelor did not show any clear benefits regarding thrombosis or a deleterious effect of pretreatment regarding bleeding.22 Once again, the median time elapsed until the cardiac catheterization was performed was < 24 hours since hospital admission (23 hours). Surprisingly, clinical practice guidelines leave the door opened to a weak level of recommendation (IIbC) regarding pretreatment of patients in whom early catheterization < 24 hours is not possible.1
In conclusion, current recommendations on early invasive treatment and no antiplatelet pretreatment in patients with NSTEACS are controversial and can also be difficult to implement in the routine clinical practice in our setting. The ultimate objective of the IMPACT-TIMING-GO registry is to shed light on the current management of NSTEACS in Spain. After the impact that the COVID-19 pandemic has had on the general structure of the healthcare system and the drop in the number of interventional procedures performed in 2020,23 we should expect to see pre-pandemic numbers in 2022 and cath labs and cardiac surgery intensive care units going back to normal. Therefore, moment seems ripe to conduct a real-world registry.
CONCLUSIONS
The IMPACT-TIMING-GO registry is the first prospective study ever conducted in Spain that will be giving us information on the early therapeutic strategies—both pharmacological and interventional—performed in our country in patients with NSTEACS after the publication of the ESC guidelines 2020, and the impact of these and other measures indicated in these patients at follow-up.
FUNDING
This unfunded study has been promoted by the Spanish Society of Cardiology Young Cardiologists Working Group with scientific endorsement from the Spanish Society of Cardiology.
AUTHORS’ CONTRIBUTIONS
Study design, data curation and review, statistical analysis, and manuscript drafting: P. Díez-Villanueva, F. Díez-Delhoyo, and M.T. López-LLuva. All the authors participated in the manuscript review and approval process.
CONFLICTS OF INTEREST
None reported.
ACKNOWLEDGEMENTS
We wish to thank the Spanish Society of Cardiology Young Cardiologists Working Group for their drive to engage the youth in medical research.
WHAT IS KNOWN ABOUT THE TOPIC?
- The management of patients with NSTEACS includes dual antiplatelet therapy with a P2Y12 inhibitor and, in most cases, invasive approach through cardiac catheterization for further revascularization. The current ESC clinical practice guidelines recommend early invasive approach (<24 hours) and no pretreatment systemically though both aspects are still controversial.
- The degree of implementation of such recommendations in the routine clinical practice, in Spain, is still unknown.
WHAT DOES THIS STUDY ADD?
- This study will improve our knowledge on early therapeutic approach, and its prognostic impact in patients admitted due to NSTEACS in Spain.
- Also, it will bring us information on the characteristics and clinical evolution of these patients in association with the recommendations and therapeutic targets we have today.
REFERENCES
1. Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42:1289-1367.
2. Díez-Villanueva P, Méndez CJ, Alfonso F. Non-ST elevation acute coronary syndrome in the elderly. J Geriatr Cardiol JGC. 2020;17:9-15.
3. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the management of patients with non-ST-elevation acute coronary syndromes: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;130:2354-2394.
4. Roffi M, Patrono C, Collet JP, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:267-315.
5. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123:2736-2747.
6. Visseren FLJ, Mach F, Smulders YM, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies With the special contribution of the European Association of Preventive Cardiology (EAPC). Rev Esp Cardiol. 2022;75:429.
7. Garcia-Garcia HM, McFadden EP, Farb A, et al. Standardized End Point Definitions for Coronary Intervention Trials: The Academic Research Consortium-2 Consensus Document. Circulation. 2018;137:2635-2650.
8. McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599-3726.
9. Kofoed KF, Kelbæk H, Hansen PR, et al. Early Versus Standard Care Invasive Examination and Treatment of Patients With Non-ST-Segment Elevation Acute Coronary Syndrome. Circulation. 2018;138:2741-2750.
10. Mehta SR, Granger CB, Boden WE, et al. Early versus delayed invasive intervention in acute coronary syndromes. N Engl J Med. 2009;360:2165-2175.
11. Thiele H, Rach J, Klein N, et al. Optimal timing of invasive angiography in stable non-ST-elevation myocardial infarction: the Leipzig Immediate versus early and late PercutaneouS coronary Intervention triAl in NSTEMI (LIPSIA-NSTEMI Trial). Eur Heart J. 2012;33:2035-2043.
12. Milosevic A, Vasiljevic-Pokrajcic Z, Milasinovic D, et al. Immediate Versus Delayed Invasive Intervention for Non-STEMI Patients: The RIDDLE-NSTEMI Study. JACC Cardiovasc Interv. 2016;9:541-549.
13. Montalescot G, Cayla G, Collet JP, et al. Immediate vs delayed intervention for acute coronary syndromes: a randomized clinical trial. JAMA. 2009;302:947-954.
14. Lemesle G, Laine M, Pankert M, et al. Optimal Timing of Intervention in NSTE-ACS Without Pre-Treatment: The EARLY Randomized Trial. JACC Cardiovasc Interv. 2020;13:907-917.
15. Janssens GN, van der Hoeven NW, Lemkes JS, et al. 1-Year Outcomes of Delayed Versus Immediate Intervention in Patients With Transient ST-Segment Elevation Myocardial Infarction. JACC Cardiovasc Interv. 2019;12:2272-2282.
16. Ferreira-González I, Permanyer-Miralda G, Marrugat J, et al. MASCARA (Manejo del Síndrome Coronario Agudo. Registro Actualizado) study. General findings. Rev Esp Cardiol. 2008;61:803-816.
17. Barrabés JA, Bardají A, Jiménez-Candil J, et al. Prognosis and management of acute coronary syndrome in Spain in 2012: the DIOCLES study. Rev Esp Cardiol. 2015;68:98-106.
18. Álvarez Álvarez B, Abou Jokh Casas C, Cordero A, et al. Early revascularization and long-term mortality in high-risk patients with non-ST-elevation myocardial infarction. The CARDIOCHUS-HUSJ registry. Rev Esp Cardiol. 2020;73:35-42.
19. Dawson LP, Chen D, Dagan M, et al. Assessment of Pretreatment With Oral P2Y12 Inhibitors and Cardiovascular and Bleeding Outcomes in Patients With Non-ST Elevation Acute Coronary Syndromes: A Systematic Review and Meta-analysis. JAMA Netw Open. 2021;4:e2134322.
20. Montalescot G, Bolognese L, Dudek D, et al. Pretreatment with prasugrel in non-ST-segment elevation acute coronary syndromes. N Engl J Med. 2013;369:999-1010.
21. Schüpke S, Neumann FJ, Menichelli M, et al. Ticagrelor or Prasugrel in Patients with Acute Coronary Syndromes. N Engl J Med. 2019;381:1524-1534.
22. Tarantini G, Mojoli M, Varbella F, et al. Timing of Oral P2Y12 Inhibitor Administration in Non-ST Elevation Acute Coronary Syndrome. J Am Coll Cardiol. 2020;76:2450-2459.
23. Romaguera R, Ojeda S, Cruz-González I, collaborators of the ACI-SEC, REGISTRY COLLABORATORS. Spanish Cardiac Catheterization and Coronary Intervention Registry. 30th Official Report of the Interventional Cardiology Association of the Spanish Society of Cardiology (1990-2020) in the year of the COVID-19 pandemic. Rev Esp Cardiol. 2021;74:1095-1105.
* Corresponding author.
E-mail address: felipediezdelhoyo@hotmail.com (F. Díez-Delhoyo).