ABSTRACT
Introduction and objectives: Transcatheter aortic valve implantation (TAVI) has become the treatment of choice for the management of symptomatic severe aortic stenosis. As it happens with all procedures, the safety and effectiveness of TAVI must be monitored. To this end, we assessed the data available from the Spanish National Health Service from 2014 through 2017.
Methods: The study included patients aged > 50 years treated with TAVI and registered in the Activity Registry of Specialized Health Care from 2014 through 2017 from public and private hospitals in compliance with the National Health System. Multivariate logistic regression analyses were performed to identify factors associated with mortality and complications, and negative binomial models for the mean hospital length of stay (LoS). Standardized rates were used to discriminate both the effectiveness and safety among regions with higher and lower levels of implementation of the technique using the national median as the threshold, (37 implants x 105 habitants) in the 4-year period.
Results: A total of 5454 TAVIs were analyzed. The in-hospital mortality rate dropped from 4.89% in 2014 to 2.7% in 2017. The LoS decreased from 13.1 days in 2014 to 11.3 days in 2017. No differences in mortality were observed among the regions. However, the LoS of regions with a high volume of implants was significantly lower (OR, 0.88; 95%CI, 0.86-0.91; P < .01), as well as the risk of infections (OR, 0.54; 95%CI, 0.32-0.9; P = .02), and pacemaker implantation (OR, 0.77; 95%CI, 0.65-0.91; P < .01).
Conclusions: The use of TAVI in Spain is safe and has grown progressively with improved outcomes regarding morbidity and mortality. Differences among regions have been highlighted regarding patient access to TAVI. This heterogeneity was not associated with mortality but with differences in the morbidity rates.
Keywords: Aortic Stenosis. Surgical aortic valve replacement. Valvular heart disease. Activity Registry of Specialized Health Care. Transcatheter aortic valve implantation. TAVI.
RESUMEN
Introducción y objetivos: El implante percutáneo de válvula aórtica (TAVI) se ha consolidado como tratamiento de la estenosis aórtica grave. Como toda intervención, su seguridad y su efectividad deben monitorizarse en condiciones de práctica real. Para ello, se han analizado los datos del Sistema Nacional de Salud disponibles entre los años 2014 y 2017.
Métodos: Se evaluaron todos los pacientes mayores de 50 años con TAVI por vía transfemoral incluidos en el Registro de Actividad de Atención Especializada entre 2014 y 2017 procedentes de centros públicos o concertados. Se hicieron análisis de regresión logísticos para evaluar los factores asociados con la mortalidad y las complicaciones, y modelos binomiales negativos para la estancia media hospitalaria. Se usaron tasas estandarizadas para ajustar diferencias en las variables de efectividad y seguridad entre las diferentes comunidades autónomas, de acuerdo con el alto y bajo volumen de implantación de la técnica, considerando como referencia la mediana nacional (37 implantes por 105 habitantes) durante el periodo de tiempo analizado.
Resultados: En total se analizaron 5.454 casos de TAVI. La mortalidad intrahospitalaria pasó del 4,89% en 2014 al 2,7% en 2017. La estancia media hospitalaria descendió de 13,1 días en 2014 a 11,3 en 2017. A pesar de no observar diferencias en la mortalidad entre comunidades autónomas, aquellas con mayor volumen de implantes tuvieron una menor estancia media hospitalaria (OR = 0,88; IC95%, 0,86-0,91; p < 0,01), menor riesgo de infecciones (OR = 0,54; IC95%, 0,32-0,9; p = 0,02) y menor necesidad de marcapasos permanente (OR = 0,77; IC95%, 0,65-0,91; p < 0,01).
Conclusiones: El uso de TAVI crece progresivamente en España con resultados cada vez mejores respecto al éxito del implante y la morbimortalidad perioperatoria. La variabilidad interregional en las tasas de implante no se relaciona con la mortalidad observada, pero sí con la morbilidad y la estancia hospitalaria.
Palabras clave: Estenosis aórtica. Reemplazo de la válvula aórtica. Registro de Actividad de Atención Especializada. Enfermedad de las válvulas cardiacas. Implante percutáneo de válvula aórtica. TAVI.
Abbreviations
LoS: length of stay. PPI: permanent pacemaker implantation. RAE-CMBD: Activity Registry of Specialized Health Care. TAVI: transcatheter aortic valve implantation.
INTRODUCTION
Aortic stenosis is the most common acquired valvular heart disease whose prevalence is around 3% in the population ≥ 65 years up to 7.4% in older ages (≥ 85 years).1 It is also the leading cause of valvular surgery in the adult population.2 Age and sex are among the risk factors (higher incidence rate in men).3,4 After symptom onset, mortality rate is high (up to 50%) during the following years.5,6 Due to the continuing growth of the elderly population in our country,7 a significant burden of disease associated with aortic stenosis is expected8 with the corresponding challenge that this poses to the healthcare system.
Until 14-15 years ago the only therapeutic option was surgical aortic valve replacement. In patients considered inoperable due to their age or comorbidities, the therapeutic alternative was only aimed at symptom control. Transcatheter aortic valve implantation (TAVI) created a new option of therapeutic opportunity for inoperable or high-risk patients at the beginning followed by intermediate-risk patients, and eventually for low-risk patients.9-13 Despite this, the rate of treatment with TAVI in Spain is significantly lower compared to other European countries.14 Although there are numerous reports and registries in Europe on the clinical results of TAVI, the evidence available in Spain on the local results from public ownership sources is scarce although 10 years have already gone by since the first TAVI was performed back in April 2007.14
This study presents the current evidence on the real-world clinical practice available in our country on the use of TAVI including a description of the profile of cases treated, the results obtained in terms of mortality, complications, and length of stay (LoS). Also, the main factors associated are analyzed from the perspective of the Spanish National Health System using the Activity Registry of Specialized Health Care (RAE-CMBD) as the main source of information.
METHODS
Minimum Data Set
This study is based on information included in the RAE-CMBD of the Spanish Ministry of Health, Consumer Affairs, and Social Welfare. It provides detailed information on the demographic characteristics of hospitalized patients, administrative variables, and clinical variables associated with the diagnoses and procedures of both the patient and type of healthcare received. The diagnoses and procedures registered are coded according to the International Classification of Diseases, Ninth Revision – Clinical Modification (ICM-9-CM) for 2014-2015, and the International Classification of Diseases, Tenth Revision – Clinical Modification (ICM-10-CM) for 2016-2017 that is more specific with diagnoses.
This analysis included all episodes registered from 2014 through 2017 of patients > 50 years of age hospitalized in centers of public or public-private partnership after performing the following procedures: “Endovascular replacement of aortic valve” (ICM-9-CM code: 35.05), and “Replacement of aortic valve with zooplastic tissue, percutaneous approach” (ICM-10-CM code: 02RF38Z). Cases treated with extracorporeal circulation were excluded (ICM-9-CM code: 39.61, and ICM-10-CM code: 5A1221Z) for considering that TAVI does not need this technique, which would, therefore, be a coding mistake of the episode. Similarly, episodes of rehospitalization were excluded to complete the patient’s recovery.
The safety analysis included all complications coded in chapter 17 of ICM-9-CM, and in particular, with codes from category 996: “Complications peculiar to certain specified procedures” including complications not classified under other concepts, and in the use of artificial substitutes involving internal device implantation, among others. On the other hand, in the most recent ICM-10-CM classification, this type of complications can be found under category T82 “Complications of cardiac and vascular prosthetic devices, implants, and grafts”. Also, due to their potential association with TAVI and great interest from the clinical standpoint events such as acute kidney injury, acute myocardial infarction, aortic dissection, stroke, sepsis, and permanent pacemaker implantation (PPI) were considered as well.
Statistical analysis
A descriptive analysis of the study variables was conducted. Continuous variables were expressed as means and standard deviations. The categorical ones as absolute and relative frequencies. The differences between 2 independent groups were compared using the Student t test or the U Mann-Whitney-Wilcoxon test based on their distribution (parametric or nonparametric, respectively). The chi-square test or Fisher’s exact test were used with the categorical variables. Multivariate models were implemented to identify factors associated with the risk of in-hospital mortality, significant complications (logistic regression), and LoS (negative binomial regression). Demographic and clinical variables were examined as explanatory variables: age, sex, rate of implantation in the autonomous community where the procedure was performed, complications, and level of severity of each case based on the RAE-CMBD classification (stratified depending on the characteristics of each patient, diagnoses, and procedures) and categorized into a 4-level scale:15,16 mild, moderate, major or extreme according to the severity-adjusted Diagnosis Related Groups (DRG).
To analyze the possible differences among autonomous communities in the volume of TAVIs performed, the rates of implantation standardized per 100 000 inhabitants (×105) were estimated. The seasonal population of each region in the period adjusted by age group (50-74, 75-84, ≥ 85), and sex was taken as the reference point.17 Discrimination between high- and low-volume regions was made by categorizing the rates of implantation. Rates above the national average during the study period were considered high-volume regions (37 procedures × 105 inhabitants). Differences in the baseline characteristics of patients treated in high- and low-volume autonomous communities were taken into consideration during the adjustment of multivariate models.
The independent variables of all logistic regression and negative binomial models were reviewed by clinical experts to guarantee their clinical sense and then selected in such a way that the resulting model would minimize the Akaike information criterion.18 Once the best model was determined in each case, the odds ratios (OR) and their 95% confidence intervals (95%CI) were estimated to determine whether a certain factor was associated with a higher risk (OR > 1) in the presence of a given result and then compare the size of several factors.
Regarding the multivariate analysis of complications, when the number of cases registered was low and statistical power of contrast was limited, all major complications were grouped following the clinical criterion once again. This is how factors associated with the risk of bleeding and accidental puncture or laceration (ICM-9-CM: 998.11, and 998.2 | ICM-10-CM: I97.4, I97.6, and I97.5*), acute myocardial infarction (ICM-9-CM: 410.*1 | ICM-10-CM: I21*), PPI (ICM-9-CM: 37.8 | ICM-10-CM: 5A1223Z), sepsis, and infections (ICM-9-CM: 995.91, 995.92, and 998.5 | ICM-10-CM: T81.4XXA, and A41*); acute kidney injury (ICM-9-CM: 584 | ICM-10-CM: N17*); and stroke (ICM-9-CM: 997.02, 434, and 435 | ICM-10-CM: I97.8*0, I66, I63.3, I63.4, I63.5, G45*, and I67.82) were identified and assessed.
All statistical contrasts were bilateral, and differences with P values > .05 were considered statistically significant. Statistical analysis was conducted using the statistical software package R (version 3.6.1).
RESULTS
A total of 5454 cases with transcatheter aortic valve implantation via transfemoral access were accounted for: 763 cases reported in 2014, 1171 in 2015, 1477 in 2016, and 2043 back in 2017. Implantation was more common in women (52.2%), mostly in patients between 81 and 85 years of age, both men (37%) and women (44%). Mean age was 81.12 ± 6.43 years during the entire period. Overall, the most common diagnoses that triggered the patients’ hospitalization were aortic valve disease (79.7%, ICM-9-CM, and ICM-10-CM codes: 424.1, and I35, respectively) followed by mitral valve regurgitation, and rheumatic stenosis (3.1%, ICM-9-CM, and ICM-10-CM codes: 396.2, and T82.0*XA, respectively), and congestive heart failure (2.9%, ICM-9-CM, and ICM-10-CM codes: 428.0, and I08.0, respectively).
Rates of transfemoral TAVI, mortality, and hospital stay
The number of TAVIs performed remained variable across the years, and grew gradually with significant increases reported between 2014 and 2017 both in the number of centers where this procedure was conducted and in the overall number of TAVIs performed. Therefore, the national implantation rate (× 105 inhabitants) doubled, and the mean during the study period was 33 procedures × 105 inhabitants (table 1).
Table 1. Implantation rate per 100 000 inhabitants, mean hospital stay, and in-hospital mortality rate per autonomous community
| Overall IR for the 2014-2017 period | Hospital stay | Mortality | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adjusted | 2014 | 2015 | 2016 | 2017 | Overall for the study period | Rate reported during the study period | ||||||||||
| Autonomous community | TAVI (n) | Seasonal population | Est (x 105 inhabitants) | 95%CI LL | 95%CI UL | LoS | SD | LoS | SD | LoS | SD | LoS | SD | LoS | SD | |
| 1 | 945 | 2,805,282 | 37 | 34.6 | 39 | 14.8 | 11.9 | 13.2 | 12.7 | 12.1 | 10.3 | 11.7 | 11.3 | 12.7 | 11.5 | 4.2 |
| 2 | 62 | 520 240 | 11 | 8.2 | 14 | 24.6 | 34.2 | 14.6 | 9.9 | 20.8 | 20 | 12.4 | 6.4 | 16.3 | 16.9 | 1.6 |
| 3 | 238 | 486 637 | 45 | 39.5 | 51 | 9.8 | 7.6 | 10.9 | 9.2 | 11.1 | 10.3 | 8.6 | 5.9 | 10 | 8.4 | 5.5 |
| 4 | 49 | 357 975 | 16 | 11.6 | 21 | 14.5 | 9.5 | 11.7 | 10.3 | 8.2 | 3.5 | 12 | 8.9 | 11.4 | 8.4 | 6.1 |
| 5 | 112 | 699 707 | 20 | 16.3 | 24 | 17.9 | 15.3 | 20.1 | 15.7 | 16.3 | 19.5 | 23.2 | 22.7 | 20.4 | 19.5 | 2.7 |
| 6 | 204 | 232 793 | 85 | 73.5 | 97 | 12.7 | 9.6 | 11.2 | 5.7 | 10.6 | 6.5 | 9.1 | 4 | 10.7 | 6.5 | 1.5 |
| 7 | 114 | 720 493 | 14 | 11.7 | 17 | 15.7 | 9.3 | 11.2 | 8.1 | 15 | 14 | 13 | 10 | 13.4 | 10.6 | 2.6 |
| 8 | 413 | 1,067,664 | 32 | 29.0 | 35 | 12 | 6.5 | 13.9 | 14.5 | 9.7 | 7.5 | 11.8 | 9.7 | 11.6 | 9.9 | 2.9 |
| 9 | 671 | 2,656,228 | 25 | 23.3 | 27 | 12.9 | 11.7 | 14.2 | 11.8 | 15.4 | 13.5 | 13.8 | 13.7 | 14.2 | 13.2 | 3.7 |
| 10 | 251 | 1,798,755 | 15 | 13.1 | 17 | 8.6 | 5.3 | 9.3 | 7.8 | 10.4 | 9.4 | 7.2 | 7.8 | 8.9 | 8.1 | 3.6 |
| 11 | 69 | 418 558 | 15 | 12.0 | 19 | 11.4 | 6.2 | 15 | 14.6 | 12.6 | 6.8 | 9.7 | 5.2 | 11.7 | 8.4 | 7.2 |
| 12 | 673 | 1,153,586 | 50 | 46.6 | 54 | 10.6 | 9.2 | 10.2 | 10.4 | 10.5 | 11.9 | 11.1 | 9.5 | 10.7 | 10.2 | 3.7 |
| 13 | 1126 | 2,206,886 | 53 | 49.9 | 56 | 14.8 | 16.3 | 12.6 | 17 | 12.2 | 12.8 | 10.1 | 7.9 | 11.9 | 13 | 2.9 |
| 14 | 211 | 456 332 | 49 | 42.9 | 57 | 8.2 | 7.1 | 6 | 4.9 | 6.7 | 6.2 | 7.8 | 9.9 | 7.1 | 7.5 | 3.3 |
| 15* | 91 | 236 596 | 37 | 29.4 | 45 | 11 | 7.1 | 13.6 | 12.7 | 10.9 | 7.6 | 9.9 | 8.1 | 11 | 8.7 | 5.5 |
| 16* | 225 | 890 671 | 25 | 21.5 | 28 | 13.7 | 14.8 | 9.6 | 6.1 | 11.1 | 7.2 | 10.2 | 8.2 | 10.9 | 9.2 | 3.1 |
| National | 5454 | 16,708,405 | 33 | 31.8 | 34 | 13.1 | 12.5 | 12.1 | 12.6 | 11.9 | 11.3 | 11.3 | 10.6 | 11.9 | 11.5 | 3.6 |
|
95%CI, 95% confidence interval; IR, implantation rate; LL, lower limit; LoS, length of stay (days); SD, standard deviation; TAVI, transcatheter aortic valve implantation; UL, upper limit. * Due to the peculiarities described in the rendering of the service in these regions, the implantation rate could take a detour with respect to the routine clinical practice in these regions. The highest mortality rates are due to the negative results reported within the first years (eg, in autonomous community #3, the mortality rate in 2014 was 16.7% but dropped to 3.3% in 2017; in autonomous community #4, the rate in 2014 was 25% but dropped to 7.4% in 2017; in autonomous community #11, the rate in 2014 was 14.3% but dropped to 6.7% in 2017). In all cases, a clearly positive tendency towards a lower procedural mortality rate was reported. |
||||||||||||||||
The overall mortality rate during the entire period was 3.6% with an obvious decrease from 2014 (4.8%) through 2017 (2.7%) despite the fact that the severity profile assigned to the patients remained constant across the 4 years studied (in 70% of the patients the severity-adjusted DRG score was extreme).
Regarding the length of hospital stay associated with the procedure, the LoS reported in 2014 was 13.1 ± 12.5 days dropping gradually down to 11.3 ± 10.6 days in 2017 with great variability among the different autonomous communities (table 1). When extreme cases were eliminated (defined as cases with stays > 2 standard deviations with respect to the mean), the mean national hospital stay was 8.9 ± 4.69 days, and differences among autonomous communities were reduced from 6.07 ± 4.8 to 11.35 ± 4.31 days; see table 1.
Complications associated with TAVI
Table 2 describes the complications associated with TAVI. Same as it happened with mortality, a gradual reduction in the rate of the most significant complications was reported from 2014 through 2017. The need for PPI was 12.2%, also with great variability of this complication among the different autonomous communities.
Table 2. Complications associated with TAVI
| Complication | Total | ICM-9-CM | 2014 | 2015 | ICM-10-CM | 2016 | 2017 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | N | % | N | % | |||
| Mechanical heart valve prosthesis complication | 218 | 4 | 996.02 | 56 | 7.3 | 47 | 4 | T82.0* XA | 49 | 3.3 | 66 | 3.2 |
| Other complication of heart valve implantation | 143 | 2.6 | 996.71 | 13 | 1.7 | 27 | 2.3 | T82.8*7A, T82.9XXA | 39 | 2.6 | 64 | 3.1 |
| Dissection of aorta | 15 | 0.3 | 441 | 2 | 0.3 | 3 | 0.3 | I71.0* | 3 | 0.2 | 7 | 0.3 |
| Bleeding or iatrogenic stroke | 40 | 0.7 | 997.02 | 4 | 0.5 | 9 | 0.8 | I97.8*0 | 11 | 0.7 | 16 | 0.8 |
| Cerebral artery occlusion | 46 | 0.8 | 434 | 8 | 1 | 11 | 0.9 | I66, I63.3, I63.4, I63.5 | 11 | 0.7 | 16 | 0.8 |
| Transient cerebral ischemia | 27 | 0.5 | 435 | 3 | 0.4 | 6 | 0.5 | G45*, I67.82 | 9 | 0.6 | 9 | 0.4 |
| Acute myocardial infarction | 87 | 1.6 | 410.*1 | 14 | 1.8 | 14 | 1.2 | I21* | 25 | 1.7 | 34 | 1.7 |
| Permanent pacemaker implantation | 663 | 12.2 | 37.8 | 146 | 19.1 | 149 | 12.7 | 5A1223Z | 155 | 10.5 | 213 | 10.4 |
| Acute kidney injury | 441 | 8.1 | 584 | 74 | 9.7 | 90 | 7.7 | N17* | 119 | 8.1 | 158 | 7.7 |
| Postoperative shock | 55 | 1 | 998 | 10 | 1.3 | 14 | 1.2 | T81.1* XA | 16 | 1.1 | 15 | 0.7 |
| Bleeding complicating the procedure | 314 | 5.8 | 998.11 | 54 | 7.1 | 63 | 5.4 | I97.4*, I97.6* | 90 | 6.1 | 107 | 5.2 |
| Accidental puncture or laceration during the procedure | 156 | 2.9 | 998.2 | 31 | 4.1 | 45 | 3.8 | I97.5* | 36 | 2.4 | 44 | 2.2 |
| Infection following a procedure | 45 | 0.8 | 998.5 | 11 | 1.4 | 13 | 1.1 | T81.4XXA | 12 | 0.8 | 9 | 0.4 |
| Sepsis | 28 | 0.5 | 995.91, 995.92 | 6 | 0.8 | 6 | 0.5 | A41* | 8 | 0.5 | 8 | 0.4 |
|
TAVI transcatheter aortic valve implantation. Codes (ICM-10-CM), T82.0 * XA, Other mechanical complication of heart valve prosthesis (initial encounter); T82.8*7A, Other specified complications of cardiac and vascular prosthetic devices, implants and grafts (initial encounter); T82.9XXA, Unspecified complication of cardiac and vascular prosthetic device, implant and graft (initial encounter); I71.0*, Dissection of aorta; I97.8*0, Intraoperative and postprocedural complications and disorders of circulatory system (cardiac surgery); I66, Occlusion and stenosis of cerebral arteries; I63.3, Cerebral infarction due to thrombosis of cerebral arteries; I63.4, Cerebral infarction due to embolism of cerebral arteries; I63.5, Cerebral infarction due to unspecified occlusion or stenosis of cerebral arteries; G45, Transient cerebral ischemic attacks and related syndromes; I67.82, Cerebral ischemia; I21, Acute myocardial infarction; 5A1223Z, Performance of cardiac pacing; N17, Acute kidney failure; T81.1*XA, Shock during or resulting from a procedure (initial encounter); I97.4*, Intraoperative hemorrhage and hematoma of a circulatory system organ or structure complicating a procedure; I97.6*, Postprocedural hemorrhage and hematoma of a circulatory system organ or structure following a procedure; I97.5*, Accidental puncture and laceration of a circulatory system organ or structure during a procedure; T81.4XXA, Infection following a procedure, initial encounter; A41, Sepsis. |
||||||||||||
Factors associated with mortality risk, hospital stay, and complications associated with TAVI
Table 3 describes the profile of cases treated, and the results of the procedures performed in high- and low-volume regions. High-volume autonomous communities treated older patients (80.47 vs 81.91, P < .001) with a rate of extreme risk that was slightly lower (70.3% in low-volume autonomous communities vs 67.1% in high-volume autonomous communities; P = .013). In these autonomous communities, it was reported that the population treated had a lower rate of comorbidities such as diabetes, arterial hypertension, heart failure, chronic obstructive pulmonary disease or smoking (table 3).
Table 3. Comparative analysis between high- and low-volume autonomous communities regarding TAVI
| Low-volume (N = 3002) | High-volume (N = 2452) | P | |||
|---|---|---|---|---|---|
| Characteristics of the patient | |||||
| Age; mean (SD) | 80.47 | 6.48 | 81.91 | 6.29 | < .001* |
| Age group; n (%) | |||||
| 50-74 | 455 | 15.2 | 275 | 11.2 | < .001* |
| 75-84 | 1749 | 58.3 | 1228 | 50.1 | |
| ≥ 85 | 798 | 26.6 | 949 | 38.7 | |
| Sex (% women), n (%) | 1520 | 50.6 | 1327 | 54.1 | .011* |
| Level of severity, n (%) | |||||
| Mild | 13 | 0.4 | 20 | 0.8 | .013* |
| Moderate | 44 | 1.5 | 18 | 0.7 | |
| Major | 835 | 27.8 | 768 | 31.3 | |
| Extreme | 2110 | 70.3 | 1646 | 67.1 | |
| Diabetes mellitus; n (%) | 1059 | 35.3 | 706 | 28.8 | < .001* |
| Hypercholesterolemia; n (%) | 1365 | 45.5 | 1070 | 43.6 | .185 |
| Obesity; n (%) | 381 | 12.7 | 273 | 11.1 | .085 |
| Arterial hypertension; n (%) | 1742 | 58.0 | 1234 | 50.3 | < .001* |
| Atrial fibrillation; n (%) | 1037 | 34.5 | 904 | 36.9 | .079 |
| Heart failure; n (%) | 611 | 20.4 | 357 | 14.6 | < .001* |
| COPD; n (%) | 330 | 11.0 | 207 | 8.4 | .002* |
| Chronic kidney injury; n (%) | 634 | 21.1 | 482 | 19.7 | .194* |
| Smoking; n (%) | 450 | 15.0 | 283 | 11.5 | < .001* |
| Use of anticoagulants; n (%) | 511 | 17.0 | 485 | 19.8 | .01* |
| Procedural results | |||||
| Death; n (%) | 113 | 3.8 | 81 | 3.3 | .401 |
| Hospital stay (days); mean (SD) | 12.72 | 11.82 | 10.85 | 11.08 | < .001* |
| Puncture/laceration; n (%) | 89 | 3.0 | 67 | 2.7 | .667 |
| Hemorrhage complicating the procedure; n (%) | 156 | 5.2 | 158 | 6.4 | .056 |
| AMI; n (%) | 56 | 1.9 | 31 | 1.3 | .098 |
| Permanent pacemaker implantation; n (%) | 400 | 13.3 | 263 | 10.7 | .004* |
| Sepsis and infectious events; n (%) | 47 | 1.6 | 21 | 0.9 | .026 |
| Acute kidney injury; n (%) | 215 | 7.2 | 226 | 9.2 | .007 |
| Stroke; n (%) | 54 | 1.8 | 41 | 1.7 | .801 |
|
AMI, acute myocardial infarction; COPD, chronic obstructive pulmonary disease; SD, standard deviation. * Statistically significant differences between high- and low-income autonomous communities. High-volume of implants defined as regions with implantation rates > national mean of 37 procedures × 105 inhabitants. Level of severity according to the RAE-CMBD case classification. |
|||||
Procedural results also varied between high- and low-volume autonomous communities: in high-volume regions, the duration of the LoS was shorter and the risk of certain complications like need for PPI, sepsis or infections was lower too. However, these regions reported a higher risk of acute kidney injury in the bivariate analysis (table 3).
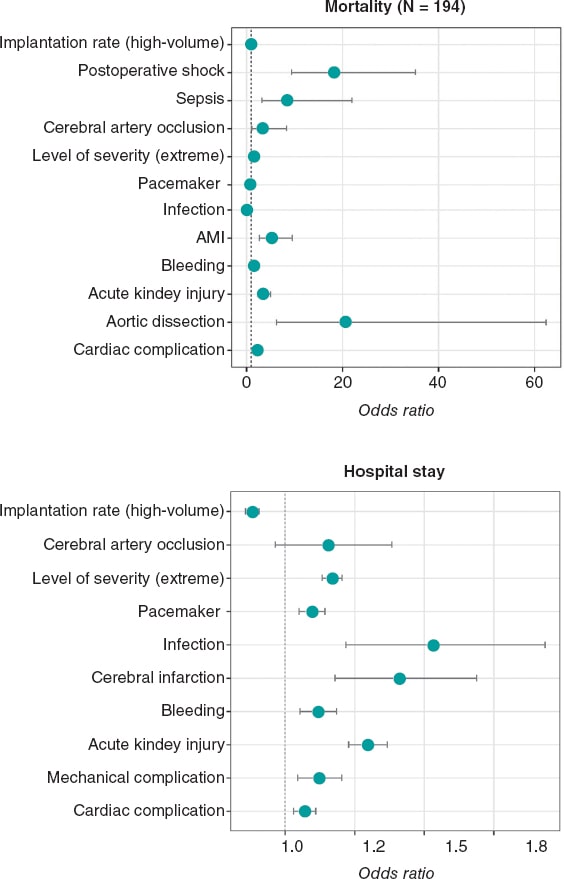
Due to these differences, age, sex, and the level of severity of each case were included in the programmed multivariate models to adjust the analyses of the explanatory variables associated with the in-hospital mortality rate. Figure 1 shows that aortic dissection (OR, 20.58; 95%CI, 6.27-62.40; P < .01), and postoperative shock (OR, 18.16; 95%CI, 9.43-35.16; P < .01) were significantly associated with the postoperative mortality rate. The explanatory weight of other complications like acute myocardial infarction, acute kidney injury, cerebral artery occlusion or heart complications was significantly lower. Mortality differences between high- and low-risk autonomous communities did not reach statistical significance in the overall period studied (figure 1).
Figure 1. Mortality and hospital stay registered based on the rate of implantation and occurrence of procedural complications. Figures represent the impact that the independent variables selected in the logistic multivariate (mortality) or negative binomial models (hospital stay) have on every complication. The following were always among the candidate variables considered: sex, age, region-adjusted volume of TAVIs performed, and level of severity (RAE-CMBD). Axes in the coordinate plane vary based on the sizes of the odds ratio presented. AMI, acute myocardial infarction.
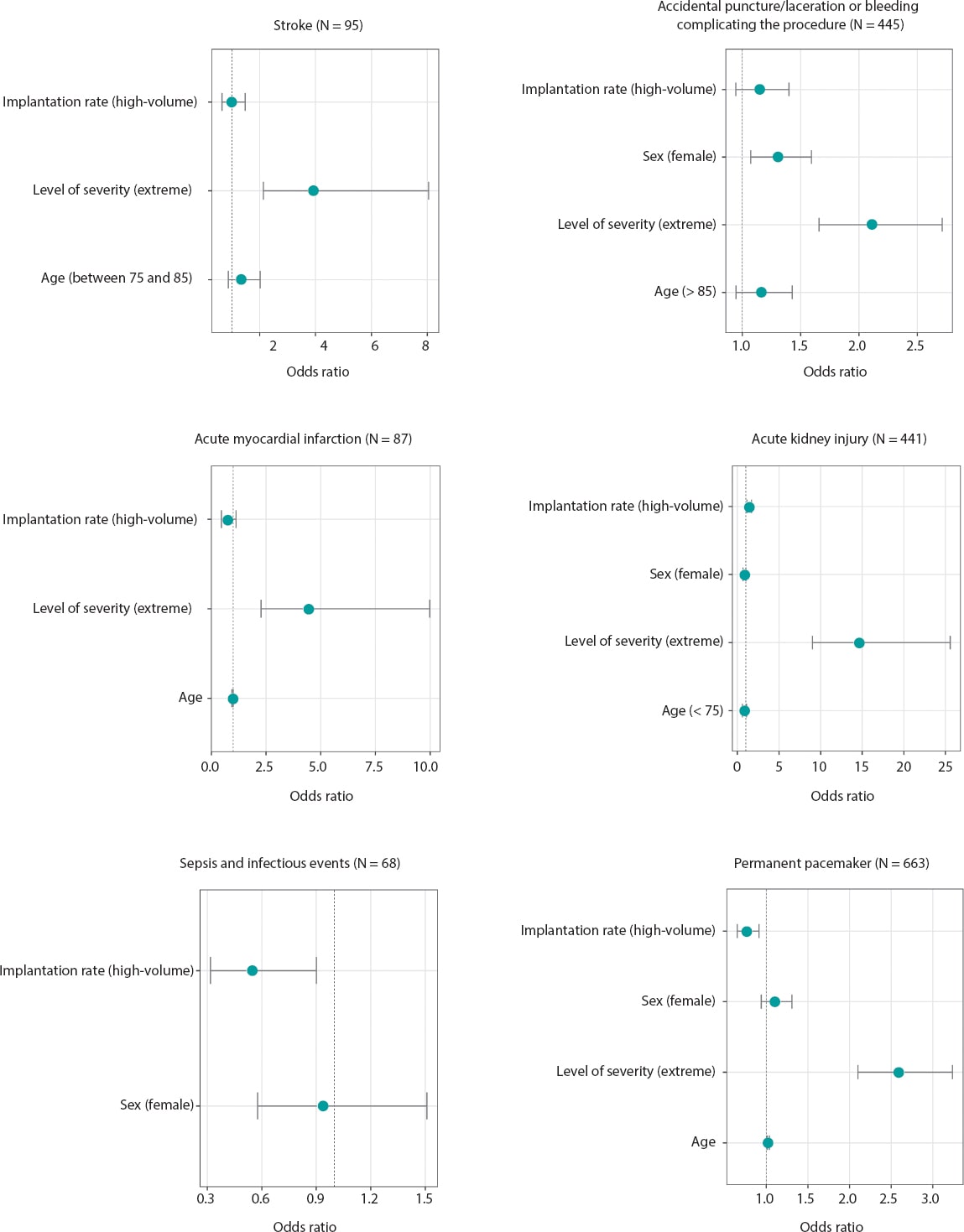
On the other hand, the higher severity of the cases, and the appearance of some complications contributed to a significant increase in the duration of the LoS (figure 1). The level of severity was also the factor that kept a stronger correlation with the occurrence of complications such as bleeding and accidental puncture or laceration, acute myocardial infarction, PPI, acute kidney injury, and stroke (figure 2). In high-volume autonomous communities, the risk of sepsis or infections and need for PPI was significantly lower compared to low-volume autonomous communities (OR, 0.77; 95%CI, 0.65-0.91; P < .01, and OR, 0.54; 95%CI, 0.32-0.9; P = .02, respectively). Finally, we should mention that high-volume autonomous communities also had a significantly shorter LoS that was 12% shorter compared to low-volume autonomous communities (OR, 0.88; 95%CI, 0.86-0.91; P < .01) as shown on figure 1.
Figure 2. Results of the logistic regression analysis on the risk of clinical complications. Figures represent the impact that the independent variables selected in the multivariate models (genetic algorithm selection following the Akaike information criterion) have on every complication. The following were always among the candidate variables considered: sex, age, region-adjusted volume of TAVIs performed, and level of severity (RAE-CMBD). Axes in the coordinate plane vary based on the sizes of the odds ratio presented.
DISCUSSION
Our study main finding was that transcatheter aortic valve implantation via transfemoral access is safe and effective in Spain. Secondly, with the growing number of procedures performed each year and the experience gained, the rates of non-lethal complications, and mortality (2.7% over the last year studied) have reduced gradually.
It is undeniable that TAVI is a safe and effective procedure according to the scientific evidence available from clinical trials,12,13,19 and meta-analyses.20,21 That is why its indication has probably widened from inoperable high-risk patients to intermediate and low-risk patients. This study includes evidence from sources from the Spanish Ministry of Health, Consumer Affairs, and Social Welfare including data from the real-world routine clinical practice in our country.
In Spain, data from the National Registry of Activities in Interventional Cardiology of the Spanish Society of Cardiology Working Group on Hemodynamics and Interventional Cardiology22 and from RAE-CMBD show a considerable increase in the number of cases treated in the 2014-2017 period22 in all autonomous communities. Even so, in our country, the rate of TAVI is still significantly lower compared to other countries.14 We should mention the ongoing improvement reported in the rates of mortality (56.25% reduction in the study period), non-lethal complications, and LoS despite the high rates (around 70%) of cases reported of extreme severity according to the codification of cases in the RAE-CMBD.
Unlike previous studies that showed a worse prognosis in men after implantation data do not show any significant differences based on sex.23 Indeed, the factors that seem to be associated with a higher mortality rate are postoperative shock, sepsis, aortic dissection, and myocardial infarction. In this case, the patient’s risk ratio and the volume of implantation are clearly associated with a different mortality rate, which may have to do with the fact that more experience has been gained with this procedure in the entire country. Consistent with this, data from the CMBD have shown a better mortality rate after conventional surgical aortic valve replacement of 3.3% in 2017 (3.6% in patients between 70 and 80 years of age, and 4.3% in patients > 80 years of age).24
The rate of PPI found was similar to the one reported by other authors in our country.22 In this case, coding the severity of the cases treated was actually associated with a higher risk. The need for PPI after TAVI has been associated, above all, with the type of valve used (this need is greater with self-expanding valves).25 In our analysis, it is impossible to distinguish the type of valve used because this information is not on the data provided by the RAE-CMBD. Both the need for postoperative PPI and the appearance of infectious processes and sepsis after the procedure were inversely associated with a higher number of procedures being performed. This association between the volume of procedures performed and fewer complications has already been described in different settings such as after coronary revascularization26 or after conventional surgical aortic valve replacement,24 among others.
Added to its clinical benefits, TAVI has consolidated as a cost-effective alternative to conventional surgical aortic valve replacement. The fact that the procedural results of TAVI have become safer and more effective gradually with lower mortality and morbidity rates and shorter LOSs is probably associated with the experience gained by the surgeons, the volume of procedures performed, and the technical and technological advances made.24
The growing elderly population in our country and the growing number of indications are the reason why the number of TAVI-eligible patients has been growing.1,11 In this sense, it is important to add new evidence to contribute to the assessment of the health outcomes of these procedures27 to guarantee homogeneous quality services in our National Health System. Also, to provide assessment mechanisms to the strategic lines defined in cardiovascular health28 since TAVI has consolidated a cost-effective option compared to conventional surgical aortic valve replacement.29,30
Limitations
The use of an administrative database to obtain information has obvious pros and cons. On the one hand, it allows us to draw a great deal of information from the national census thanks to the obligatory nature of this registry. Also one of its strengths is the high data standardization.27 However, the administrative nature of the RAE-CMBD whose clinical variables are based on the discharge summary, the thoroughness of coding, and the possible inconsistencies among centers when implementing the codes can impact the accuracy of the results. However, since the period analyzed is a 4-year period, we could say that there are no substantial changes in coding capable of impacting the results significantly. Still, we should mention that the specific codes of complications of ICM-9-CM may not include all the complications that can occur during TAVI. Despite of this, all major clinical complications for the analysis of procedural results were studied. On the other hand, the analysis conducted took into consideration data from the RAE-CMBD until 2017, the most recent ones collected to this date. Also, it would be good to analyze data collected over the last years to see how results evolved. We should expect these results to be even better given the increased number of TAVIs performed, the surgeons’ greater experience, and the improved technology available. This should be analyzed by future studies.
Added to this, we should mention that the information collected in this analysis comes from procedures registered in public or public centers with shared activity. Therefore, discrepancies can be found with the data published by the National Registry of Activities in Interventional Cardiology of the Spanish Society of Cardiology Working Group on Hemodynamics and Interventional Cardiology in absolute terms.22 Still, both sources barely differ in the percentage of cases seen that require PPI (overall difference of 2% in 2016, and 0.1% in 2017 being the data from the Registry of the Spanish Society of Cardiology Working Group on Hemodynamics and Interventional Cardiology) greater.
One of the last limitations of this study is that only the episode that triggered the implantation was analyzed, and no long-term results were obtained, which would provide quality information to assess the extent and cost of healthcare. However, the results from this study are interesting to the extent that they provide key information on aspects to go deeper in the generation of arguments for quality healthcare management.
CONCLUSIONS
TAVI is a safe and effective procedure whose implantation rate is on the rise. Still, there is a huge variability between different autonomous communities and hospitals in Spain. This procedure is mostly performed in patients > 75 years of age with low morbidity and mortality rates that can be compared absolutely to those of conventional surgery. Rates have been going down over the 4 years studied (2014-2017) with gradual reductions in the postoperative LoS, especially in autonomous communities with higher implantation rates.
FUNDING
This study has been funded by Edwards Lifesciences S.L.
AUTHORS’ CONTRIBUTIONS
M. Álvarez-Bartolomé, and J. Cuervo conducted both the validation and statistical management of data. All authors contributed to the study design, conducted the critical review of the manuscript, and gave their final approval. Also, they take full responsibility in all aspects of the study by guaranteeing its integrity and accuracy.
CONFLICT OF INTERESTS
B. Martí-Sánchez is a member of Edwards Lifesciences S.L., the sponsor of this study. J. Cuervo is a member of Axentiva Solutions and received fees for his scientific consulting work for Edwards Lifesciences S.L.
WHAT IS KNOWN ABOUT THE TOPIC?
- The rate of TAVI via transfemoral access is significantly lower in Spain compared to other European countries. Also, even though a decade has passed since the first implantation was performed, there is still scarce evidence on the use and results of this procedure in our country.
WHAT DOES THIS STUDY ADD?
- The study provides solid and precise information on the safety, effectiveness, and results of the use of TAVI in the Spanish population.
- There is inter-territory variability in the use of TAVI. Still, the results show low mortality and morbidity rates and a gradual reduction of the mean hospital stay in the study period.
- The rate of complications and mortality seems to go down as more and more TAVIs are performed.
- Higher implantation rates were associated with shorter hospital stays, and a lower risk of permanent pacemaker implantation and infections during the hospital stay.
BIBLIOGRAFÍA
1. Ferreira-González I, Pinar-Sopena J, Ribera A, et al. Prevalence of calcific aortic valve disease in the elderly and associated risk factors:A population-based study in a Mediterranean area. Eur J Preventive Cardiol. 2013;20:1022-1030.
2. Salinas P, Moreno R, Calvo L, et al. Seguimiento a largo plazo tras implante percutáneo de válvula aórtica por estenosis aórtica grave. Rev Esp Cardiol. 2016;69:37-44.
3. Izquierdo-Gómez MM, Hernández-Betancor I, García-Niebla J, Marí-López B, Laynez-Cerdeña I, Lacalzada-Almeida J. Valve Calcification in Aortic Stenosis:Etiology and Diagnostic Imaging Techniques. BioMed Research International. 2017;5178:1-12.
4. Stewart BF, Siscovick D, Lind BK, et al. Clinical Factors Associated With Calcific Aortic Valve Disease fn1fn1This study was supported in part by Contracts NO1-HC85079 through HC-850086 from the National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, Maryland. J Am Coll Cardiol. 1997;29:630-634.
5. Ramaraj R, Sorrell VL. Degenerative aortic stenosis. BMJ (Clinical research ed). 2008;336:550-555.
6. Otto CM, Lind BK, Kitzman DW, Gersh BJ, Siscovick DS. Association of Aortic-Valve Sclerosis with Cardiovascular Mortality and Morbidity in the Elderly. N Engl J Med. 1999;341:142-147.
7. INE. Nota de prensa 20 de octubre de 2016. Proyecciones de Población 2016-2066. 2016;2066. Available online:https://www.ine.es/prensa/np994.pdf. Accessed 29 June 2020.
8. Thaden JJ, Nkomo VT, Enriquez-Sarano M. The Global Burden of Aortic Stenosis. Prog Cardiovasc Dis. 2014;56:565-571.
9. Leon MB, Smith CR, Mack M, et al. Transcatheter Aortic-Valve Implantation for Aortic Stenosis in Patients Who Cannot Undergo Surgery. N Engl J Med. 2010;363:1597-1607.
10. Duncan A, Ludman P, Banya W, et al. Long-term outcomes after transcatheter aortic valve replacement in high-risk patients with severe aortic stenosis:the U.K. Transcatheter Aortic Valve Implantation Registry. JACC Cardiovasc Interv. 2015;8:645-653.
11. Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017;38:2739-2791.
12. Mack MJ, Leon MB, Thourani VH, et al. Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients. N Engl J Med. 2019;380:1695-1705.
13. Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter Aortic-Valve Replacement with a Self-Expanding Valve in Low-Risk Patients. N Engl J Med. 2019;380:1706-1715.
14. Biagioni C, Tirado-Conte G, Nombela-Franco L, et al. Situación actual del implante transcatéter de válvula aórtica en España. Rev Esp Cardiol. 2017;70(Supl1):6-8.
15. Ministerio de Sanidad, Consumo y Bienestar Social. Grupos Relacionados de Diagnóstico APR-GRD V32. Norma Estatal 2017.;2019. Available online https://www.mscbs.gob.es/estadEstudios/estadisticas/docs/CMBD/Nota_difus_Norma_Estatal_2017.pdf. Accessed 3 Jul 2020
16. Subdirección General de Información Sanitaria e Innovación. Ministerio Sanidad, Servicios Sociales e Igualdad. Norma Estatal RAE-CMBD 2016 –Nota Informativa.;2018. Available online http://pestadistico.inteligenciadegestion.msssi.es/. Accessed 29 Jun 2020
17. Instituto Nacional de Estadística. Tablas de Mortalidad Por Año, Comunidades y Ciudades Autónomas, Sexo, Edad y Funciones. Available online https://www.ine.es/jaxiT3/Tabla.htm?t=27154&L=0. Accessed 29 Jun 2020.
18. Calcagno V, de Mazancourt C. glmulti:An R Package for Easy Automated Model Selection with (Generalized) Linear Models. J Statistical Software. 2010;34:1-29.
19. Castrodeza J, Amat-Santos IJ, Blanco M, et al. Propensity score matched comparison of transcatheter aortic valve implantation versus conventional surgery in intermediate and low risk aortic stenosis patients:A hint of real-world. Cardiol J. 2016;23:541-551.
20. Sardar P, Kundu A, Chatterjee S, et al. Transcatheter versus surgical aortic valve replacement in intermediate-risk patients:Evidence from a meta-analysis. Catheter Cardiovasc Interv. 2017;90:504-515.
21. Khan SU, Lone AN, Saleem MA, Kaluski E. Transcatheter vs surgical aortic-valve replacement in low- to intermediate-surgical-risk candidates:A meta-analysis and systematic review. Clin Cardiol. 2017;40:974-981.
22. Sección de Hemodinámica y Cardiología Intervencionista. Sociedad Española de Cardiología. Registro Nacional de Actividad En Cardiología Intervencionista 2017.;2018. Available online https://www.hemodinamica.com/wp-content/uploads/2018/11/PRESENTACIO%CC%81N-DEFINITIVA-REGISTRO-2017.pdf Accessed 27 Jun 2020.
23. European Society of Cardiology. Women less likely to die after TAVI than men. Published online 2013. Available online https://www.escardio.org/The-ESC/Press-Office/Press-releases/Women-less-likely-to-die-after-TAVI-than-men. Accessed 29 Jun 2020.
24. Carnero-Alcázar M, Maroto-Castellanos LC, Hernández-Vaquero D, et al. Isolated aortic valve replacement in Spain:national trends in risks, valve types, and mortality from 1998 to 2017. Rev Esp Cardiol. 2021;74:700-707.
25. Deharo P, Bisson A, Herbert J, et al. Impact of Sapien 3 Balloon-Expandable Versus Evolut R Self-Expandable Transcatheter Aortic Valve Implantation in Patients With Aortic Stenosis:Data From a Nationwide Analysis. Circulation. 2020;141:260-268.
26. Goicolea Ruigómez FJ, Elola FJ, Durante-López A, Fernández Pérez C, Bernal JL, Macaya C. Coronary artery bypass grafting in Spain. Influence of procedural volume on outcomes. Rev Esp Cardiol. 2020;73:488-494.
27. Íñiguez Romo A, Bertomeu Martínez V, Rodríguez Padial L, et al. The RECALCAR Project. Healthcare in the Cardiology Units of the Spanish National Health System, 2011 to 2014. Rev Esp Cardiol. 2017;70:567-575.
28. Ministerio de Sanidad. El Ministerio de Sanidad traslada a las CCAA el borrador de la Estrategia en Salud Cardiovascular del SNS. Available online https://www.mscbs.gob.es/gabinete/notasPrensa.do?id=4993. Accessed 20 Oct 2020.
29. Ribera A, Slof J, Andrea R, et al. Transfemoral transcatheter aortic valve replacement compared with surgical replacement in patients with severe aortic stenosis and comparable risk:Cost–utility and its determinants. International J Cardiol. 2015;182:321-328.
30. Baron SJ, Wang K, House JA, et al. Cost-Effectiveness of Transcatheter Versus Surgical Aortic Valve Replacement in Patients With Severe Aortic Stenosis at Intermediate Risk. Circulation. 2019;139:877-888.