To the Editor,
This is the case of a young man with a postmyocardial infarction large ventricular septal defect (VSD) surgically repaired 10 days after venoarterial extracorporeal membrane oxygenation (VA-ECMO) therapy. The patient still had a large residual VSD that triggered a situation of refractory congestion due to pulmonary hyperflow that was successfully treated with percutaneous closure. The patient gave his informed consent so this case could be published anonymously.
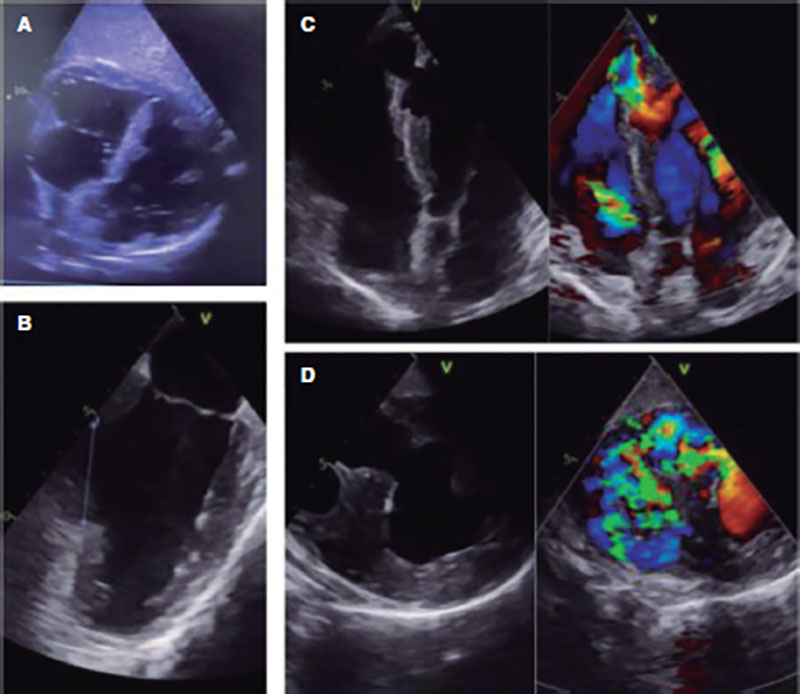
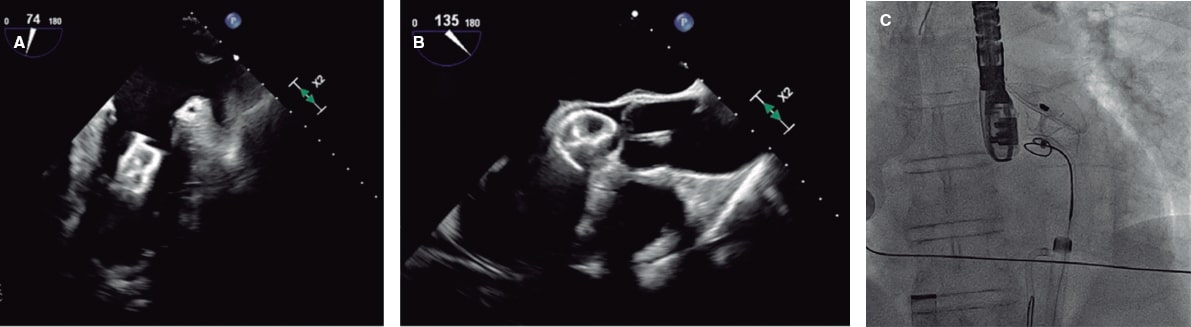
This is the case of a 46-year-old man without a past medical history and inferior wall myoscardial infarction and Killip class I. Cardiac catheterization confirmed the presence of multivessel disease. The culprit lesion found at the proximal right coronary artery [TIMI grade-0 flow (Thrombolysis in Myocardial Infarction)] was revascularized with a drug-eluting stent. The patient was admitted to the coronary care unit, and progressed into cardiogenic shock. Several transthoracic and transesophageal echocardiographic studies revealed the presence of severe biventricular dysfunction and a large, basal inferoseptal VSD (50 mm) of anfractuous non-restrictive trajectory (Qp/Qs ratio of 3) (figure 1).
Figure 1. Large inferoseptal ventricular septal defect up to the apical segments as seen on the transthoracic echocardiography (A) with a 50 mm maximum diameter as seen on the transesophageal echocardiography. (B) The long (C) and short (D) axes seen on the transesophageal echocardiography reveal the presence of a non-restrictive left-to-right shunt.
The patient was intubated, treated with VA-ECMO and with an intra-aortic balloon pump. He required amines with fast stabilization. Direct heart transplantation was suggested due to the high surgical risk involved, but eventually delayed surgical repair was used.
The patient remained stable and without heart failure. After 9 days, he showed signs of hemolysis due to thrombosis of the ECMO filter with acute kidney injury and pulmonary edema that required continuous venovenous hemodiafiltration. Emergency surgery was decided with double coronary artery bypass graft and VSD closure with a pericardial surgical patch. The patient entered a state of deep shock due to severe ventricular dysfunction (left ventricular ejection fraction < 10%) during postoperative period. Afterwards, the patient improved gradually with decannulation and extubation 5 and 7 days, respectively after the procedure.
The patient showed pulmonary congestion and required venovenous hemodiafiltration followed by IV diuretics. The transthoracic and transesophageal echocardiographic follow-up studies confirmed the presence of a novel non-restrictive residual VSD. After a negative fluid balance, cardiac catheterization revealed these values: aortic pressure, 90/60 mmHg; pulmonary arterial pressure, 26/16/8 mmHg; pulmonary capillary wedge pressure; 7 mmHg, right atrial pressure, 4 mmHg, and a Qp/Qs ratio of 1.7.
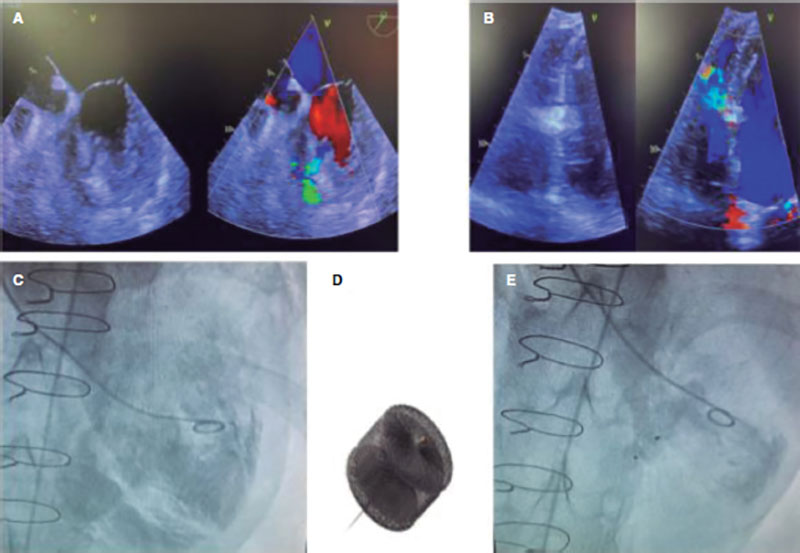
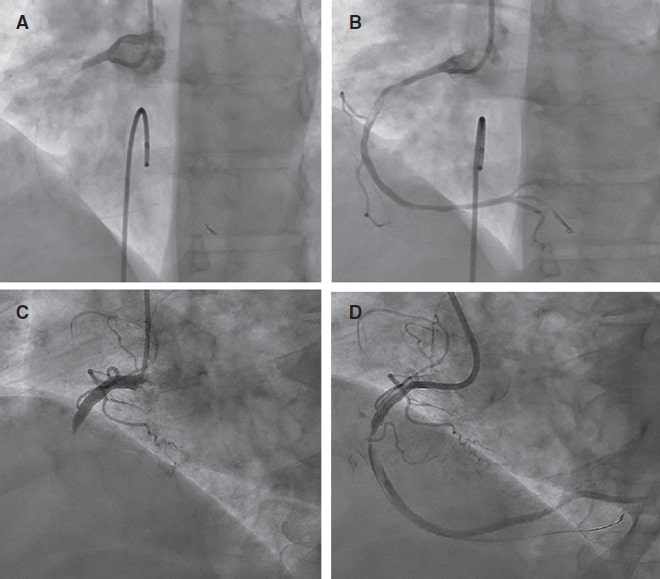
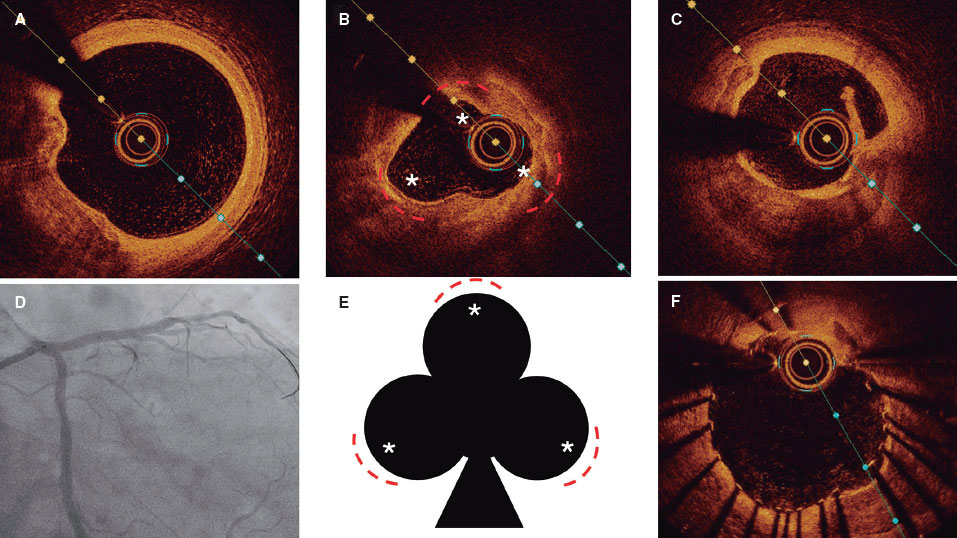
Given the presence of residual VSD with congestion due to hyperflow, closure was indicated. Due to the high surgical risk involved (myopathy, renal failure, ventricular dysfunction), the percutaneous approach was used. VSD was closed via femoral vein using a 12 mm Amplatzer AVPII device (Abbott, United States) that resulted in the overt reduction of the angiographic shunt with a restrictive intra-device residual shunt (figure 2).
Figure 2. Postoperative non-restrictive residual ventricular septal defect, (A) and presence of a small residual ventricular septal defect after closure with the Amplatzer device (B) as seen on the transthoracic echocardiography. Angiography shows the presence of a significant left-to-right shunt (C) significantly reduced after percutaneous closure (E) with an Amplatzer-AVPII device (D).
Venovenous hemodiafiltration and diuretics were removed after closure. Neurohormonal blockade was initiated, and the patient was discharged from the hospital after achieving euvolemic state with good functional class.
Postmyocardial infarction VSD is a rare mechanical complication. Its incidence rate has dropped (1%-3% down to 0.1%-0.3%) in the era of percutaneous revascularization. It often appears 3 to 5 days after infarction although it can occur within the first 24 hours or later. In the anterior acute myocardial infarction setting, VSD is often apical and has a simple trajectory. In the inferior wall acute myocardial infarction setting, however, VSD is often basal, large, and has an anfractuous and non-restrictive trajectory with worse prognosis due to the presence of a larger shunt and right ventricular damage. Definitive treatment is surgical repair, but it has a high mortality rate (up to 40%). The best time to perform surgery is still controversial: clinical practice guidelines recommend emergency surgery. However, experienced centers prefer delayed surgeries when the appearance of scar tissue allows proper suture.1 In the series published, the mortality rate associated with early surgeries is higher compared to delayed surgeries beyond the first week. However, selection bias can occur since the most severe patients are operated on early. While waiting, the use of mechanical support devices can prevent hemodynamic deterioration.2 However, the risk of complications associated with treatment is higher with longer waiting times. Regarding the device that should be selected, evidence here is based on small observational studies. Intra-aortic balloon pump can be an option, but it is insufficient in the presence of established shock; the Impella device (Abiomed, United States) allows proper left ventricular discharge. Setback here is the possibility of reversing the shunt causing arterial desaturation. VA-ECMO has been successfully used and reverses the situation of shock as a bridging therapy to surgery or, in cases of very large VSD, as a bridging therapy to heart trasplantation.3 Total artificial heart has also been used in this setting yet experience is limited on this regard. In experimental models no device has been able to normalize the hemodynamic situation or balance the Qp/Qs ratio. However, it seems that the combination of VA-ECMO plus Impella/intra-aortic balloon pump is the most favorable option.4 A special situation is the presence of pulmonary edema due to pulmonary hyperflow following left-to-right shunt. It looks like optimizing the left ventricular discharge could improve this situation by reducing the Qp/Qs ratio. However, management is still controversial. We have been gaining experience with percutaneous closure and it has been used as the definitive treatment in the management of small VSDs, and as a bridging therapy to surgery with larger VSDs although with risk of failure and embolization involved. Its use has also been reported in residual VSDs after cardiac surgery.5
In conclusion, the management of postmyocardial infarction VSD is controversial. Surgery is the treatment of choice, and it seems like delaying surgery increases the chances of success. However, the optimal waiting time is still unknown. The use of mechanical support can prevent hemodynamic deterioration being VA-ECMO an attractive therapeutic option. Percutaneous closure can be an alternative in certain settings. Finally, evidence on this regard is scarce and based on observational studies only and questions still abound.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
All the authors made their contributions during the patient’s entire healthcare process while drafting and reviewing the case.
CONFLICTS OF INTEREST
None reported.
REFERENCES
1. Ronco D, Matteucci M, Kowalewski M, et al. Surgical Treatment of Postinfarction Ventricular Septal Rupture. JAMA Netw Open. 2021;4:e2128309.
2. Hussain S, Pillarella J, Pauwaa S, et al. Management of Post Infarction Ventricular Septal Rupture in Contemporary Era. J Card Fail. 2020;26(10 Suppl):S106.
3. Rob D, Špunda R, Lindner J, et al. A rationale for early extracorporeal membrane oxygenation in patients with postinfarction ventricular septal rupture complicated by cardiogenic shock. Eur J Heart Fail. 2017;19:97-103.
4. Pahuja M, Schrage B, Westermann D, Basir MB, Reshad Garan A, Burkhoff D. Hemodynamic effects of mechanical circulatory support devices in ventricular septal defect. Circ Heart Fail. 2019;12:e005981.
5. Faccini A, Butera G. Techniques, Timing, and Prognosis of Transcatheter Post Myocardial Infarction Ventricular Septal Defect Repair. Curr Cardiol Rep. 2019;21:59.