To the Editor,
Transcatheter aortic valve implantation (TAVI) is a common therapeutic option in patients with degenerative severe aortic stenosis. In our setting, the need for definitive pacemaker implantation after TAVI is around 14%,1 which is an additional cause for morbidity and longer length of stay. In elderly patients both aspects can be especially relevant. Micra leadless pacemaker (Medtronic, United States) is a recent alternative to traditional endocavitary pacemakers. The lack of electrodes or need for subcutaneous bag to carry the generator added to femoral venous access implantation reduce some of the complications associated with conventional pacemakers (especially pneumothorax, hematoma, bag and electrode-related infections). Although initially available for VVI pacing mode only, the new Micra AV (Medtronic, United States) has appeared recently. It maintains atrioventricular synchrony in patients in sinus rhythm by detecting atrial mechanical contraction and the corresponding ventricular pacing.
After over 120 Micra implantations including an early favorable experience after TAVI,2 and widening the indication to patients in sinus rhythm too, we thought of the possibility of implanting both devices at the same procedure. In this work we present the very first series of patients who, after TAVI, were implanted with the Micra leadless pacemaker as permanent pacing therapy at the same procedure. Patients gave their informed consent to analyze and publish the results.
A total of 3 patients treated with TAVI due to symptomatic severe aortic stenosis developed an advanced atrioventricular conduction disorder during the procedure. Table 1 shows the characteristics of patients and procedures. Once the hemostasis of the femoral arterial access used for valve implantation was achieved, the Micra leadless pacemaker via femoral venous access was implanted (figure 1). The procedure went on for an average 28 minutes (19 to 36 min interval), and it was completed successfully in all 3 patients.
Table 1. Characteristics of patients and procedures.
| Patient #1 | Patient #2 | Patient #3 | |
|---|---|---|---|
| Sex | Woman | Man | Man |
| Age | 85 | 82 | 89 |
| Baseline rhythm | Paroxysmal AF | Sinus rhythm | Sinus rhythm |
| Baseline AV conduction | normal | First-degree AV block | First-degree AV block |
| Baseline QRS (ms) | 168 | 149 | 133 |
| Conduction disorder | RBBB | RBBB | RBBB |
| Permanent anticoagulation | Apixaban | no | no |
| LVEF (%) | 60 | 60 | 60 |
| TAVI arterial access site | Right femoral | Right femoral | Right femoral |
| TAVI | 26 mm Corevalve | 29 mm Corevalve | 34 mm Corevalve |
| AV conduction after implantation | Complete AV block | Complete AV block | Complete AV block |
| Type of pacemaker | VVI Micra pacing | AV Micra pacing | AV Micra pacing |
| Micra venous access | Right femoral | Right femoral | Right femoral |
| R-wave (mV) | Non-measurable | 14 | 14 |
| Impedance (Ohm) | 1.023 | 950 | 710 |
| Threshold (V x 0.24 ms) | 1 | 0.25 | 0.38 |
| Overall cath lab time (min) | 152 | 189 | 164 |
| Micra implantation time, cath lab (min) | 19 | 31 | 36 |
| Length of stay (days) | 6 | 5 | 3 |
| Pacing at follow-up (%) | 72 | 96 | 20 |
|
AV, atrioventricular; RBBB, right bundle branch block; AF, atrial fibrillation; LVEF, left ventricular ejection fraction; TAVI, transcatheter aortic valve implantation. |
|||
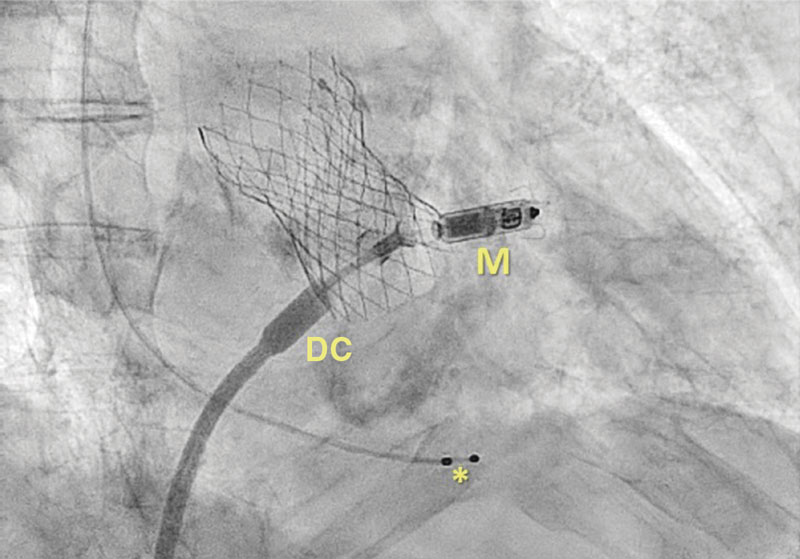
Figure 1. Transcatheter aortic valve already implanted plus Micra (M) device at the moment of delivery. * Indicative of temporary ventricular electrodes. DC, delivery catheter.
Although limited to very short series, the experience with leadless pacemakers in patients treated with TAVI is satisfactory.2-4 Retrospectively, shorter length of stay, less tricuspid regurgitation, and lack of complications like pneumothorax, bleeding or bag-related infections have been reported compared to conventional pacemaker implantation.3,4
There are certain risks involved in conventional pacemaker implantation within the same procedure after TAVI: a) anticoagulation needed to perform the valvular procedure increases the risk of bleeding in the vascular access site (subclavian/cephalic). Therefore, the site becomes less controllable and compressible than the femoral access site needed for Micra implantation, and b) pneumothorax, especially serious in elderly patients who, at times, are on mechanical ventilation. In contrast, Micra implantation requires sedoanalgesia to advance the introducer sheath (23-Fr) through the inferior vena cava, Therefore, implantation after TAVI requires extending sedoanalgesia only. In addition to the potential benefits of performing both techniques simultaneously, it is not required to leave temporary ventricular electrodes until definitive pacemaker implantation. The risk of displacement (with loss of ventricular capture), the need for relocation, ventricular perforation, and endovascular infection can also be prevented with the strategy presented here.
The limitations of Micra pacemaker implantation include costs—higher compared to conventional pacemakers (compensated by a shorter length of stay)—and longer cath lab time (though not as long as the time it takes to start a new procedure, transfer the patient, administer new sedation, etc.). The durability of the device also could be considered a limitation (around 10 years). Therefore, in the post-TAVI setting, the Micra device is spared for patients ≥ 80 years. Because of the preliminary nature of our work further studies will be necessary to confirm the benefits of this new therapeutic strategy.
In conclusion, this was the very first series of patients treated with TAVI and Micra at the same procedure. Results are favorable and confirm not only that post-TAVI Micra implantation is an appealing strategy to reduce the complications associated with conventional pacemakers, but also that it is feasible at the same procedure. It reduces substantially the length of stay and the risks associated with temporary ventricular electrodes. The current possibility of implanting the Micra device while keeping atrioventricular synchrony widens the indications for patients in sinus rhythm.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
T. Bastante: drafted the manuscript; F. Alfonso: critical review. All the authors: they contributed substantially to the study design, data curation, analysis and interpretation, and final approval of the version that would be published.
CONFLICTS OF INTEREST
F. Alfonso is an associate editor of REC: Interventional Cardiology; the journal’s editorial procedure to ensure impartial handling of the manuscript has been followed. The authors declared no conflicts of interest whatsoever.
REFERENCES
1. Jiménez-Quevedo P, Muñoz-García A, Trillo-Nouche R, et al. Evolución temporal en el tratamiento transcatéter de la estenosis aórtica: análisis del registro español de TAVI. REC Interv Cardiol. 2020;2:98-105.
2. Cuesta J, Bastante T, Alfonso F. Marcapasos sin cables Micra tras implante de prótesis valvular aórtica percutánea. Med Clin (Barc). 2020;154:239-240.
3. Moore S, Chau K, Chaudhary S, et al. Leadless pacemaker implantation: A feasible and reasonable option in transcatheter heart valve replacement patients. Pacing Clin Electrophysiol. 2019;42:542-547.
4. Okuyama K, Izumo M, Sasaki K, et al. Comparison in clinical outcomes between leadless and conventional transvenous pacemaker following transcatheter aortic valve implantation. J Invasive Cardiol 2020;32:400-404.