QUESTION: How often do you see nonrevascularizable patients with refractory angina today?
ANSWER: Refractory angina is defined as chronic angina that cannot be controlled even with the use of optimal medical therapy and where all revascularization options have failed.1,2 It is a common entity whose prevalence is on the rise due to the ageing of population and the improved prognosis of ischemic heart disease. It is estimated that between 5% and 10% of the patients with chronic ischemic heart disease can develop refractory angina. Its annual incidence rate is between 50 000 and 100 000 cases in the United States and between 30 000 and 50 000 cases in Europe.1,2 On the other hand, we know that around 10% of all coronary angiographies performed indicate that it is not possible to give efficient options of revascularization. According to the activity registry in interventional cardiology in Spain from 2019,3 this figure would be up to 15 000 patients each year, and it is reasonable to think that a significant percentage can suffer refractory angina.
These patients often remain asymptomatic which negatively impacts their quality of life and. Actually, to this day, we still do not have effective therapeutic options to offer. Multidisciplinary approach is crucial here (cardiology, family medicine, cardiac rehabilitation, psychology, pain management units, etc.). Also, we should consider the use of non-pharmacological therapies like spinal cord or subcutaneous stimulation, external counterpulsation or use the coronary sinus reducer device as well as keep looking into other alternatives like new drugs, cardiac shock wave therapy or use of progenitor cells.1,2
Q.: Please explain to us briefly the diagnostic approach used for screening etiologies different from ischemia due to epicardial coronary artery disease such as microvascular ischemia, vasospasm or even noncardiac causes.
A.: The diagnosis of refractory angina is mainly clinical, but it is important to show that there is a correlation between symptoms and ischemia. That is why it is essential to confirm the presence of myocardial ischemia, if possible, through imaging modalities like stress echocardiography, pharmacological stress echocardiography, single-photon emission tomography or magnetic resonance imaging. If no ischemia is seen on these imaging modalities, other possible causes for the symptoms should be considered like esophageal spasm or osteomuscular etiology. In the presence of ischemia, we need to know the coronary anatomy preferably through a coronary angiography before discussing any revascularization options. Information should be analyzed by the heart team including experienced interventional cardiologists and cardiac surgeons to determine the possibilities of percutaneous or surgical revascularization.
There are 2 groups of patients without revascularization options: the largest one, with advanced coronary disease (diffuse disease, small-caliber distal beds, chronic total coronary occlusions non-eligible for percutaneous coronary intervention, coronary artery bypass graft deterioration…); and that of patients with angina and myocardial ischemia without obstructive coronary lesions. In these patients it would be good to perform a functional study with microvascular dysfunction and coronary spasm testing (coronary reserve, index of microvascular resistance, absolute coronary flow, acetylcholine test) to facilitate targeted therapies with better symptom control.
Q.: What evidence do we have today on the Reducer device?
A.: It is a new option for patients with refractory angina with a different mechanism of action over the cardiac venous system. This mechanism developed over 60 years ago by Claude S. Beck4 is based on creating coronary sinus (CS) stenosis to generate a pressure gradient that is transmitted retrogradely to venules and capillaries, which translates into an improved subendocardial perfusion, probably due to collateral recruitment through the venous plexus and Thebesian veins.
The first experience with humans was reported back in 2007. In 2015 the COSIRA multicenter, randomized, double-blind, sham-controlled clinical trial5 was published. It included a total of 104 patients with refractory angina and functional class III or IV according to the Canadian Cardiovascular Society (CCS) with ischemia seen on the dobutamine stress echocardiography and without any revascularization options after assessment by the medical team assessment. The primary endpoint was to reduce, at least, 2 CSS functional class degrees of severity of angina at the 6-month follow-up. The Reducer device was superior to the sham procedure (35% vs 15%; P = .024), improved, at least, 1 CSS functional class degree of angina in 71% of the patients vs 42%, and improved parameters like duration of exercise or quality of life.
Also, there is evidence of the results of the REDUCE registry6—similar to those reported by the COSIRA trial—in the routine clinical practice on the improvement of, at least, 1 CSS functional degree of angina in 81% of the patients, better parameters of quality of life, greater distances covered in the six-minute walk test, and lower the need for antianginal drugs. The REDUCER-1 registry results of 195 patients presented at Euro-PCR 2019 showed that clinical benefit remained at the 2-year follow-up.7
Overall, in all the studies published it was effective in up to 75% of the patients.8 The reasons why some patients remain unresponsive to therapy are still not known, but they could be associated with lack of device endothelization or with an alternative venous drain through other territories.
An important conclusion we can draw from the accumulated experience is that the implantation of the Reducer device is viable and safe5-8 with serious complications (like CS dissection or perforation or device embolization) in less than 2% of the cases. The 10-plus-year follow-up of the early patients has confirmed its patency and lack of structural alterations in the long-term. No CS thrombosis has ever been reported after implantation.
Magnetic resonance imaging study supports the hypothesis of an increased subendocardial perfusion as the mechanism of action by showing that in ischemic areas the correlation between the myocardial perfusion reserve index and the endocardial reserve index—that remains low at baseline—increases after implantation. Also, myocardial perfusion improves when reducing the myocardial ischemic burden, improving the longitudinal and circumferential strain and systolic function without microstructural alterations or effect on the diastolic function.9
With the accumulated evidence, the last clinical practice guidelines on the management of chronic coronary occlusions of 2019 established by the European Society of Cardiology (ESC) included, for the very first time, the implantation of the Reducer device in patients with refractory angina with the same level of recommendation (IIb, level of evidence B) than other previous non-pharmacological options like external counterpulsation or spinal cord stimulation.10
Q.: How is the selection process of the most eligible candidates for this technique?
A.: In my opinion, most patients with symptomatic refractory angina are potential candidates despite the optimal medical therapy without percutaneous or surgical revascularization options after assessment by the heart team and with myocardial ischemia in the left main coronary artery territory as seen on the imaging modalities.6 Due to vein drain anatomy of the right coronary artery territory through the middle and small cardiac veins that drain into the CS very close to its ostium, this drainage would not be affected by the implant. For this reason, the Reducer device would not be the right option for patients with ischemia in the right coronary artery territory only. Thanks to its mechanism of action, it may be an interesting option for patients with ischemia due to microvascular disease without lesions in the epicardial coronary arteries. Although information is still limited in this context information, the early data are positive.11
One contraindication would be the presence of left ventricular pacing electrodes through the CS for resynchronization purposes, which is why its indication should be carefully studied in patients with ventricular dysfunction who may be candidates to such therapy.
Patients with refractory angina can also be candidates to other non-pharmacological therapies like external counterpulsation or neurostimulation. Although they have been available for decades, these techniques have been underused for different reasons like the perception of the lack of efficacy or placebo effect or lack of solid scientific evidence. Randomized clinical trials are needed to compare these different techniques and establish the best way to approach the management of these patients.
Q.: Can you please give us a brief technical description of this procedure?
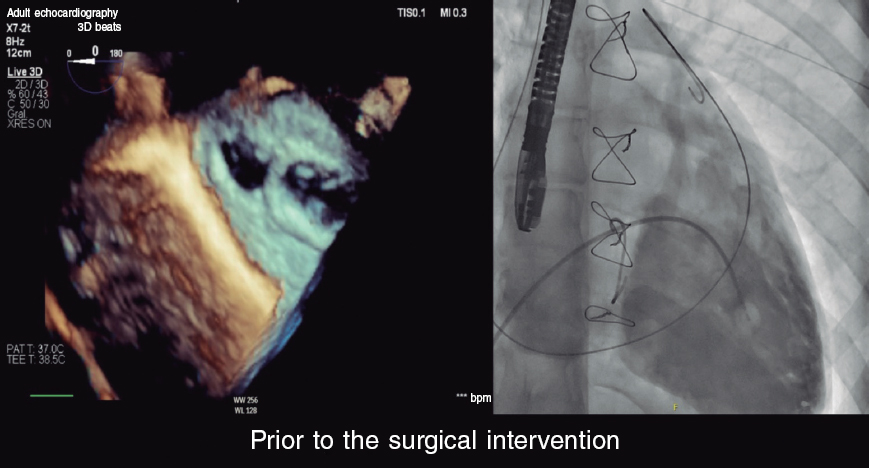
A.: The implantation of the Reducer device is easy on the technical level12,13 and successful in over 95% of the cases. The device is implanted under local anesthesia via right jugular vein as the access of choice and preferably under vascular ultrasound guidance. A 6-Fr introducer sheath is used followed by the advancement of a multipurpose catheter until the right atrium while keeping the mean pressure < 15 mmHg (with higher pressures the implantation is ill-advised since the pressure of the CS is already high at the baseline level). Afterwards, the CS undergoes selective catheterization by carefully advancing the multipurpose catheter and avoiding small-caliber branches like the left marginal ones or the vein of Marshall. There are times that a highly developed or fenestrated Thebesian valve can make selective catheterization difficult. A venography of CS is performed through the multipurpose catheter to see its size, the origin of lateral branches, and the presence of valves (like Vieussens valve that can complicate the implant).
If the procedure is continued, heparin is administered, and the guidewire is placed distal to the CS. Then, the introducer sheath is changed for a 9-Fr sheath to advance the guide catheter and deliver the device in the location previously indicated by the venography (2-4 cm from the origin of CS avoiding the jailing of lateral branches). The device consists of a stainless-steel balloon-expandable stent whose balloon has the shape of a sand clock that leaves a central waist of 3 mm. It is implanted through the inflation of a second balloon for 30 to 60 seconds at 4 to 6 atm, adjusting its size to the actual diameters of CS of 9.5 mm to 13 mm. During inflation oversizing is attempted (10% to 20%) to reduce the risk of embolization and facilitate neointimal coverage. Once implanted, the balloon is carefully removed until the guide catheter making sure that the Reducer device remains stable. Lastly, a new venography is performed to check the position of the device and lack of complications. The patient can be discharged early on after the procedure and dual antiplatelet therapy is advised for, at least, a month. The effect is evident with device endothelization, which creates a coronary sinus (CS) stenosis, which is why it is necessary to wait between 4 and 6 weeks to determine its efficacy.
In conclusion, the CS Reducer device is a new technique for the management of refractory angina. It improves myocardial perfusion acting from the cardiac venous system. Clinical results confirm its efficacy and safety profile, the technique is easy to use, and its indication has already been established in the clinical practice guidelines. Clinical evidence still needs to grow with more patients, longer follow-up periods, comparisons to other therapies, and further research on the mechanisms of nonrespondent patients. Nonetheless, the Reducer device is a new tool in the interventional cardiology armamentarium that can be an option for patients with refractory angina without revascularization options.
FUNDING
No funding.
CONFLICTS OF INTEREST
None declared.
REFERENCES
1. Gallone G, Baldetti L, Tzanis G, et al. Refractory Angina:From Pathophysiology to New Therapeutic Nonpharmacological Technologies. JACC Cardiovasc Interv. 2020;13:1-19.
2. Sainsbury PA, Fisher M, de Silva R. Alternative interventions for refractory angina. Heart. 2017;103:1911-1922.
3. Ojeda S, Romaguera R, Cruz-González I, et al. Registro Español de Hemodinámica y Cardiología Intervencionista. XXIX Informe Oficial de la Asociación de Cardiología Intervencionista de la Sociedad Española de Cardiología (1990-2019). Rev Esp Cardiol. 2020;73:927-936.
4. Beck CS, Leighninger DS. Scientific basis for the surgical treatment of coronary artery disease. J Am Med Assoc. 1955;159:1264-1271.
5. Verheye S, Jolicoeur EM, Behan MW, et al. Efficacy of a device to narrow the coronary sinus in refractory angina. N Engl J Med. 2015;372:519-527.
6. Giannini F, Baldetti L, Konigstein M. Safety and efficacy of the reducer:A multi-center clinical registry REDUCE study. Int J Cardiol. 2018;269:40-44.
7. EuroPCR 2019. A proven evidence-based therapy when angina persists. Reducer I Registry. Verheye S. Available online: https://www.pcronline.com/. Accessed 8 Sep 2020.
8. Konigstein MN, Giannini F, Banai S. The Reducer device in patients with angina pectoris:mechanisms, indications, and perspectives. Eur Heart J. 2018;39:925-933.
9. Palmisano A, Giannini F, Rancoita P, et al. Feature tracking and mapping analysis of myocardial response to improved perfusion reserve in patients with refractory angina treated by coronary sinus Reducer implantation: a CMR study. Int J Cardiovasc Imaging. 2020. http://dx.doi.org/10.1007/s10554-020-01964-9.
10. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41:407-477.
11. Giannini F, Baldetti L, Ielasi A, et al. First Experience With the Coronary Sinus Reducer System for the Management of Refractory Angina in Patients Without Obstructive Coronary Artery Disease. JACC Cardiovasc Interv. 2017;10:1901-1903.
12. Giannini F, Aurelio A, Jabbour RJ, et al. The coronary sinus reducer:clinical evidence and technical aspects. Expert Rev Cardiovasc Ther. 2017;15:47-58.
13. Giannini F, Tzanis G, Ponticelli F, et al. Technical aspects in coronary sinus Reducer implantation. EuroIntervention. 2020;15:1269-1277.
Corresponding author: Paseo de la Castellana 261, 28046 Madrid, Madrid, Spain.
E-mail address: sjvcardio@yahoo.es (S. Jiménez Valero).