HOW WOULD I APPROACH IT?
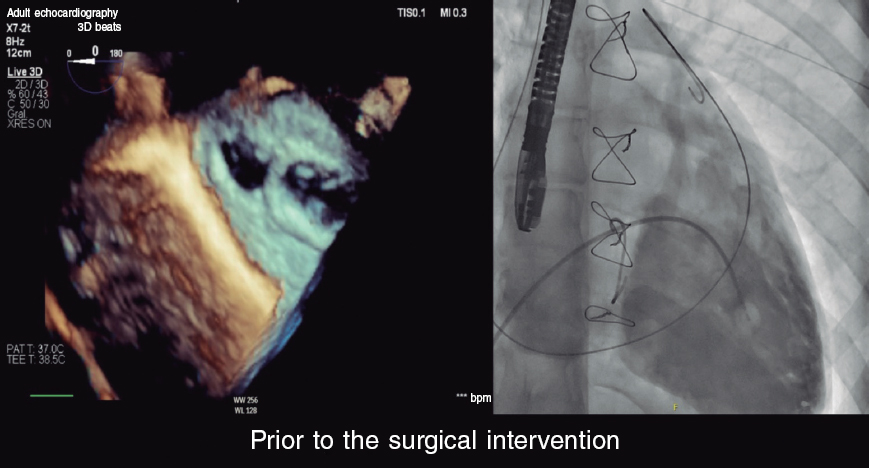
This is a case of transcatheter aortic valve implantation (TAVI) (26 mm CoreValve Evolut). During the implant, the patient developed severe hypotension while at the cath lab. Both the angiography and the echocardiography performed discarded the common causes of hypotension after TAVI:
– Tamponade (the most common one, due to the pacemaker). There is no pericardial effusion.
– Aortic rupture: it is caused during the valvuloplasty before and after the dilatations with self-expanding valves. There is no pericardial effusion or suggestive images on the echocardiography.
– Coronary occlusion: it is discarded by injecting the left main coronary artery (LMCA). When the LMCA needs to be protected, the best thing to do is to leave a stent all the way into the left anterior descending coronary artery that should be long enough to provide coverage from before the LMCA bifurcation until above the leaflets of the implanted valve.
– Vascular complications with hemorrhage. They can be discarded on an angiography.
– Severe aortic regurgitation due to paravalvular leak or leaflet dysfunction of the transcatheter aortic valve. In this case only non-significant aortic regurgitation is seen.
– Other less common causes of hypotension such as mitral regurgitation due to chordal or papillary muscle ruptures and acute thrombosis of the valve can be discarded on the echocardiography.
Once the most common causes have been discarded, a rare complication called suicide left ventricle (SLV) remains.
Patients with aortic stenosis (AS) develop an adaptation of the LV to pressure overload that induces hypertrophy and impaired diastolic relaxation. Within the first few hours following surgical aortic valve replacement, changes in the dynamics of the LV can occur that can trigger the appearance of dynamic intraventricular gradients in up to 15% of the patients.1 For this reason, at times, aortic valvular replacement and septal myectomy are performed.
The appearance of dynamic intraventricular gradients is associated with small LVs, asymmetric hypertrophy, high ejection fractions, and high valvular gradients.1 This case has a gradient of 110 mmHg, left ventricular hypertrophy (a 19 mm septum and a 13 mm posterior wall), and a reduced ventricular cavity (a 38 mm end-diastolic diameter). We should remember that up to 10% of all aortic stenoses are accompanied by asymmetric septal hypertrophy.
Also, some patients with AS may have underlying hypertrophic cardiomyopathy. That is why we should think of this entity in the presence of a fast gradient that does not match the natural progression of a calcified AS (reduced aortic valve area > 0.12 cm2 to 0.19 cm2 per year).
Patients with AS related hypertrophic cardiomyopathy have worse in-hospital results (regarding mortality, cardiogenic shock, and kidney disease) after TAVI.
The echocardiography reveals the presence of severe left ventricular hypertrophy with ventricular collapse and end-systolic left ventricular outflow tract obstruction (LVOTO). Also, it shows a mosaic pattern that affects the entire LVOT compatible with pathological flow acceleration. No systolic anterior motion (SAM) of the mitral valve was reported.
If the suspected cardiogenic shock is due to a dynamic intraventricular gradient, the following measures should be implemented:
– Serotherapy to keep left ventricular filling pressures higher than normal.
– Withdraw positive inotropic drugs and use medication to increase postload (phenylephrine in infusion to maintain systemic vascular resistances).
– Monitor heart rate with cardioselective beta-blockers without vasodilator effect (metoprolol).
– Consider the use of disopyramide that has proven effective to reduce the dynamic gradient.
– In case of atrioventricular block or sinus bradycardia, implant a dual chamber pacemaker to keep atrioventricular synchrony and recover the contribution of the atrial systole to ventricular filling. This is especially important in the presence of mid-ventricular obliteration with flow acceleration in the LVOT.2
– In patients with SAM, if cardiogenic shock persists with the use of the optimal medical treatment, we would still have more therapeutic options available: alcohol septal ablation, lower implantation of TAVI into the LVOT, emergency surgical myectomy, and implantation of MitraClip.
– Alcohol septal ablation. Using a guidewire in the left anterior descending coronary artery, the injection of alcohol in the septal branches would be pretty fast. The final outcome of septal ablation won’t be seen for another 6 months after the procedure. However, promising results have been reported in the emergency management of LVOTO after the transcatheter implantation of mitral valves,3 and also in a few cases of obstruction after TAVI. If the ablation of a septal branch does not do any good, another branch may be ablated with alcohol (especially in cases with very diffuse septal hypertrophy). Cases of ablation after TAVI have been reported at the follow-up as being asymptomatic. In patients showing LVOT dynamic obstructions and requiring TAVI, performing alcohol septal ablation between 3 and 6 months after the implantation is advised.
– New deeper TAVI to reduce the LVOTO. Very few cases have been published, all with CoreValve, in an attempt to avoid SAM (reported after TAVI and transcatheter implantation of mitral valves). A case of CoreValve collapse due to dynamic obstruction treated with a new CoreValve implanted a little deeper has been reported.
– MitraClip to treat SAM (reported in some of the cases published). This is difficult to perform in an emergency procedure.
– Emergency surgical myectomy: given the patient’s age and past medical history, it does not seem indicated.
Although rare, dynamic obstructions following TAVI can be a complication not easy to solve. The cases with echocardiographic criteria of suspicion should be prevented by implementing the following measures:1
– Proper hydration prior to the implant. Avoid diuretics.
– Use rigid guidewires with not very small curves and place them in the middle of the ventricle.
– Cardioselective IV beta-blockers without vasodilator effects (metoprolol) and keep a low heart rate.
– Avoid positive inotropic drugs.
– Pacing should be performed with a pacemaker in the right cavities and pacing with the guidewire in the LV should be avoided. Atrioventricular sequential pacing should be ready for use.
In the case presented here, if shock persists with the use of the optimal medical treatment and atrioventricular sequential pacing even in the absence of systolic anterior motion (SAM) of the mitral valve, the septal ablation of 1 or several septal branches should be taken into consideration.
FUNDING
No funding was received for this work.
CONFLICTS OF INTEREST
M. Larman is proctor for Edwards Lifesciences and Boston Scientific.
REFERENCES
1. Lopez Ayerbe J, Ewangelista Masid A, Armada Romero E, et al. Predictive factors of abnormal dynamic interventricular gradient after valve replacement in severe aortic stenosis. Rev Esp Cardiol. 2002;55:127-134.
2. Ibrahim H, Barker CM, Reardon MJ, et al. Suicide Left Ventricle Due to Conduction Disturbance Following Transcatheter Aortic Valve Replacement and Reversal With Restoration of Sinus Rhythm:Is There Life After Death?J Invasive Cardiol. 2015;27:E107-109.
3. Guerrero M, Wang DD, HimbertD, et al. Short term results of alcohol septal ablation as a bail-out strategy to treat severe left ventricular outflow tract obstruction after transcatheter mitral valve replacement in patients with severe mitral annular calcification. Catheter Cardiovasc Interv. 2017;90:1220-1226.
Corresponding author: Sección de Hemodinámica, Policlínica Gipuzkoa, Paseo de Miramón 174, 20009 San Sebastián, Guipúzcoa, Spain.
E-mail address: mariano.larman@gmail.com (M. Larman).