CASE PRESENTATION
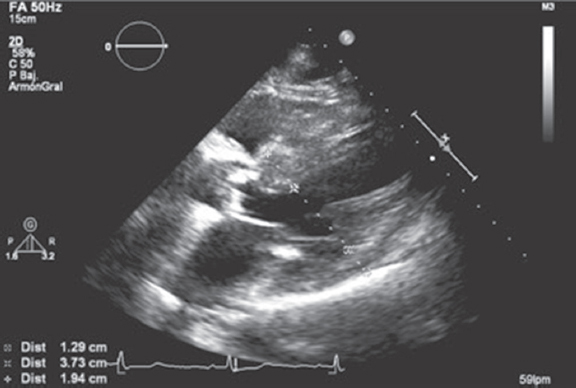
Eighty-four-year-old woman with hypertension, dyslipidemia, a past medical history of bronchial asthma, stroke in the left middle cerebral artery territory without negative side effects, and moderate chronic kidney disease (glomerular filtration rate, 42 mL/min/1.73 m2 according to the Chronic Kidney Disease Epidemiology Collaboration [CKD-EPI] equation). Also, the patient has a past medical history of non-ST-segment elevation acute myocardial infarction with revascularization of the left circumflex artery proximal segment with a conventional stent back in 2006 without an impaired ventricular function. The patient also showed moderate aortic stenosis for which she was regularly monitored by her cardiologist with a rate of progression of valvular heart disease in ranges of severity (peak gradient, 110 mmHg; mean gradient, 74 mmHg; continuity equation valve area, 0.75 cm2 and indexed, 0.45 cm2/m2) with preserved ventricular function (left ventricular ejection fraction of 67% measured using Simpson’s method). The echocardiography (figure 1) revealed the presence of left ventricular hypertrophy (a 19 mm interventricular septum and a 13 mm posterior wall) and a reduced ventricular cavity (a 38 mm end-diastolic diameter).
Figure 1. Parasternal long-axis view. Hypertrophy of septal predominance with small ventricular cavity. Severe aortic calcification.
The patient remained asymptomatic with New York Heart Association functional class III, which is why the case was brought to the heart team. Given the high surgical risk involved (logistic EuroSCORE, 26.2%, Surgeon Thoracic Score, 7.3%) it was decided to proceed with transcatheter aortic valve implantation (TAVI). The study was completed with a coronary angiography (patent stent without de novo lesions), a transesophageal echocardiography, and a computed tomography scan that confirmed the presence of a suitable caliber to use the iliofemoral access and a borderline distance between the aortic annulus and the left main coronary artery of 10 mm with a suitable diameter of both the valvular sinuses and the aortic annulus (22.1 mm). The patient gave her verbal consent for the dissemination of the clinical case for teaching and scientific purposes.
Since the patient was eligible for this procedure, a 26 mm CoreValve Evolut self-expanding aortic valve (Medtronic; Minneapolis, MN, United States) was implanted via transfemoral access after valvuloplasty with a 17 mm balloon. Femoral pre-closure as performed using a Prostar XL 10 device (Abbott Vascular; Reedwood City, CA, United States). Given the left main coronary artery maximum height and the possibility of a coronary occlusion, the heart team decided to use an angioplasty guidewire allocated in the left anterior descending coronary artery through a 3.5-Fr EBU XB guiding catheter (Medtronic, United States) for protection purposes. The procedure was uneventful and without immediate complications with proper penetration of the borders and a trivial degree of aortic regurgitation as seen on the transesophageal echocardiography.
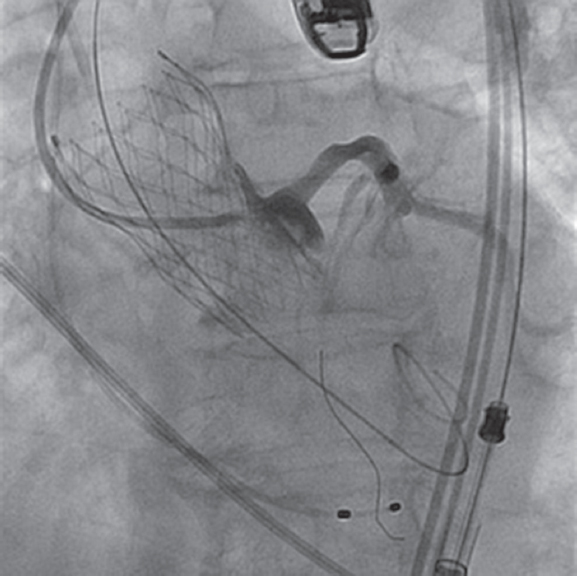
After implantation and while at the cath lab, the patient showed early and sudden arterial hypotension (60/40 mmHg). Competence was confirmed between the rhythm paced by a temporary pacemaker and sinus rhythm without atrioventricular conduction disorders or apparent signs of acute ischemia. Taking into consideration the borderline distance to the left main coronary artery, the injection occurred through the guiding catheter inserted (figure 2). Similarly, the angiography performed discarded any vascular access complications.
Figure 2. Screening of complications: diagnostic angiography on left coronary artery.
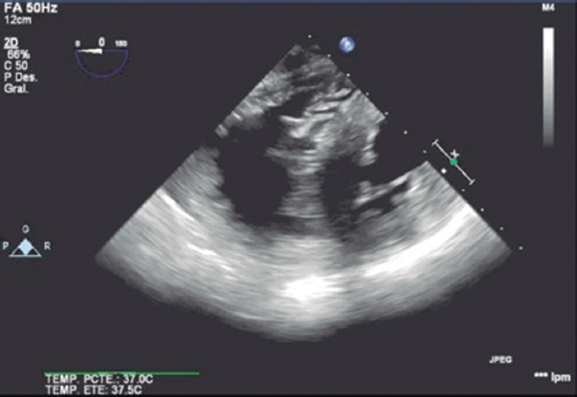
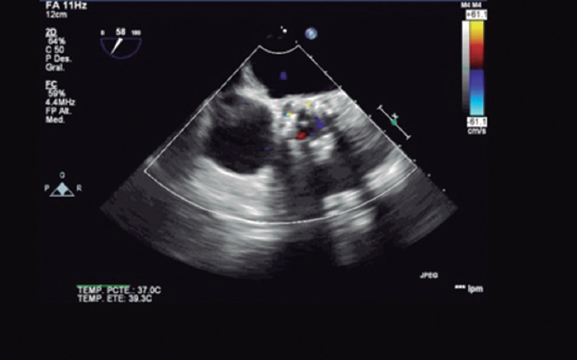
The rapid echocardiographic evaluation performed discarded pericardial effusion (figure 3) and confirmed the integrity of the mitral valvular apparatus and lack of compromised aortic annulus without aortic dissection or rupture; the good positioning and proper functioning of the valve were confirmed without significant aortic regurgitation (figure 4) and no changes with respect to the immediate study following the implant.
Figure 3. Screening of complications: transesophageal echocardiography on the transgastral view.
Figure 4. Screening of complications: transesophageal echocardiography, short-axis view.
Ventricular function proved to be hyperdynamic with flow acceleration (videos 1 and 2 of the supplementary data).
FUNDING
No funding.
AUTHORS' CONTRIBUTION
All authors prepared and revised the article.
CONFLICTS OF INTEREST
None declared.
SUPPLEMENTARY DATA
Vídeo 1. Barrionuevo Sánchez M. I. DOI: 10.24875/RECICE.M20000159
Vídeo 2. Barrionuevo Sánchez M. I. DOI: 10.24875/RECICE.M20000159
* Corresponding author: Servicio de Cardiología, Hospital Universitari de Bellvitge, Feixa Llarga s/n, 08907, L’Hospitalet de Llobregat, Barcelona, Spain.
E-mail address: bsanchezmarisa@gmail.com (M.I. Barrionuevo Sánchez).