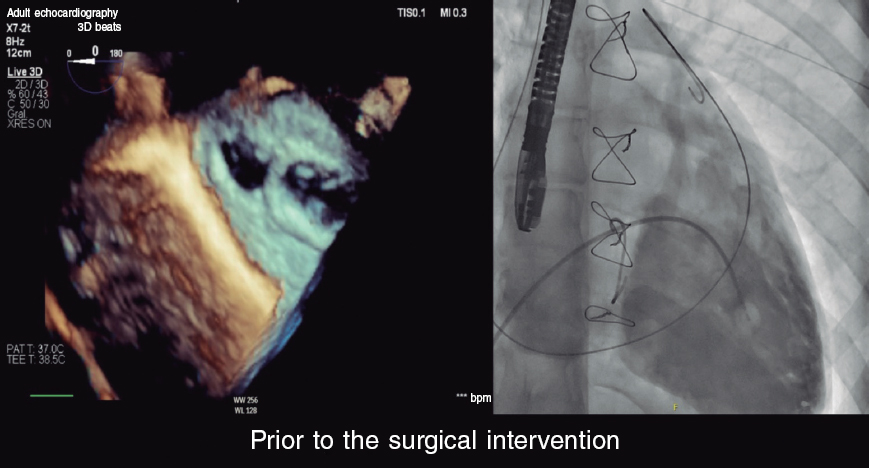
CASE RESOLUTION
Bicuspid aortic valve (BAV) is the most common congenital cardiac anomaly since it affects approximately 1% of the population. Transcatheter aortic valve implantation (TAVI) has become the treatment of choice of aortic stenosis in high to moderate risk surgical patients.1 Initially, the presence of BAV was considered as an exclusion criterion for TAVI. However, with the development of new generation devices, TAVI for BAV has become a feasible treatment strategy given its good clinical outcomes.2 Nevertheless, BAV still represents a challenging clinical scenario the scientific community is still learning about. Therefore, heart team evaluation is key to decide the most suitable valve replacement strategy in patients with BAV.
To solve the challenge previously described a second procedure was scheduled with general endotracheal anesthesia. The patients’ informed consent was obtained. Under biplanar fluoroscopic and transesophageal guidance, transseptal puncture was performed using a 7-Fr Mullins Sheath (Medtronic, Inc., United States). A long stiff J-tip guidewire (Terumo, Japan) and a 5-Fr Judkins Right JR 4 catheter (Boston Scientific, United States) were advanced towards the left atrium, left ventricle, and stenotic aortic valve. The antegrade approach was used to access the descending thoracic aorta.
The Terumo guidewire was snared using a 35 mm Gooseneck snare and then externalized from the left common femoral arterial sheath creating an arteriovenous loop (figure 1). The exchange guidewire was then extracted via right femoral arterial sheath, and an Amplatz Left AL1 catheter (Boston Scientific, United States) was advanced to facilitate the insertion of the Safari guidewire (Boston Scientific, United States) into the left ventricle.
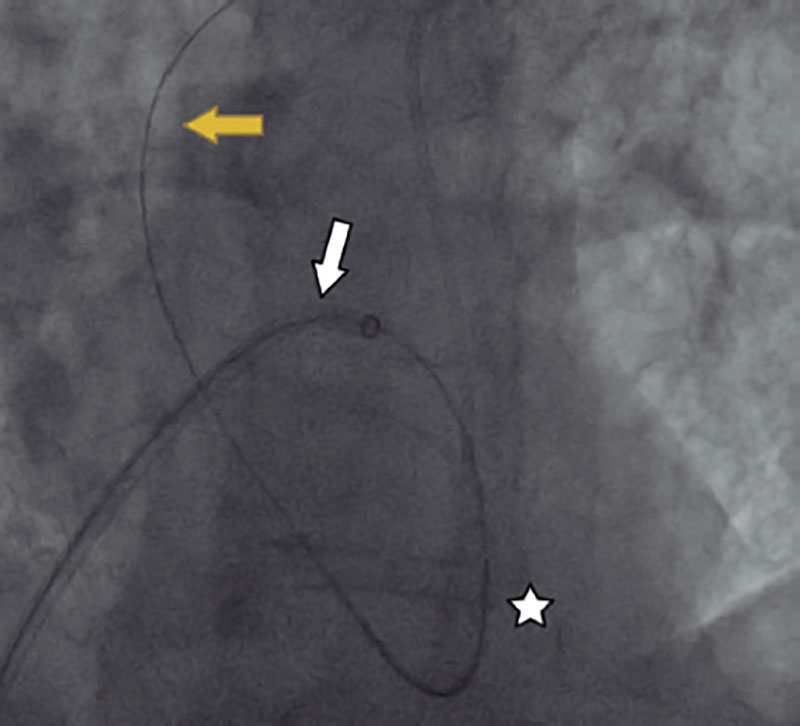
Figure 1. Arteriovenous loop (from the right femoral vein, through atrial septum, left atrium, left ventricle, and aorta, into the left femoral artery). White arrow (Mullins Sheath), yellow arrow (Terumo guidewire), and white star (5-Fr Judkins Right JR 4).
Conventional retrograde aortic valvuloplasty with a 25 mm balloon (Edwards Lifesciences, United States) under pacing at 180 bpm was performed. However, the crossing of the prosthesis through the aortic valve was unsuccessful (video 1 of the supplementary data) due to the valve severe calcification. Therefore, using the same transseptal access (the transseptal sheath was left into the left atrium if the antegrade system was eventually needed), an additional valvuloplasty with a 25 mm noncompliant Atlas balloon (Bard Peripheral Vascular, United States) was performed. This caused massive aortic regurgitation with hemodynamic impairment. However, it allowed crossing the prosthesis (overexpanded SAPIEN 3 Ultra 29 mm by adding 4 mL of extra volume due to an extremely large annulus) that was successfully implanted (video 2 of the supplementary data). After releasing the valve, the ascending aortogram demonstrated optimal valve position with mild paravalvular leak (video 3 of the supplementary data). Immediate clinical improvement was observed after TAVI. Postoperative echocardiography confirmed the presence of mild paravalvular leak and normal transvalvular flow (Vmax, 1.8 m/s).
Progressive clinical improvement was observed during hospitalization. However, on day 21 after TAVI the patient developed aspiration pneumonia that led to his death.
The importance of this case is that given this progressive expansion towards younger and lower-risk patients,3 those with BAV are often treated with TAVI. However, the singular anatomy of the BAV adds complexity to the technical challenges associated with TAVI.
As it was shown in the case presented here, patients with BAV disease undergoing TAVI represent a challenging scenario compared to tricuspid aortic disease. Uncrossable aortic valve during TAVI is not a common clinical scenario with an incidence rate of 0.37% in our case series. The presence of a highly calcified and asymmetric raphe between the noncoronary and the right cusp added to the presence of an extremely large annulus resulted in a technically arduous procedure. A hybrid transseptal strategy using the antegrade crossover technique ought to be considered in selected cases to improve the rate of technical success in patients with challenging anatomies in whom retrograde valve crossing is not possible.
FUNDING
None reported.
AUTHORS’ CONTRIBUTIONS
J. Martínez-Sole, S. Lozano-Edo, and J. Sanz-Sánchez designed, drafted the manuscript, and were involved in the manuscript critical review and final approval. F. Ten-Morro, L. Andrés-Lalaguna, and J.L. Díez-Gil designed the study, conducted the manuscript critical review, and approved its final version for publication.
CONFLICTS OF INTEREST
None whatsoever.
SUPPLEMENTARY DATA
Vídeo 1. Martínez-Sole J. DOI: 10.24875/RECICE.M22000312
Vídeo 2. Martínez-Sole J. DOI: 10.24875/RECICE.M22000312
Vídeo 3. Martínez-Sole J. DOI: 10.24875/RECICE.M22000312
REFERENCES
1. Leon MB, Smith CR, Mack M, et al. PARTNER Trial Investigators. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363:1597-1607.
2. Yoon SH, Bleiziffer S, De Backer O, et al. Outcomes in transcatheter aortic valve replacement for bicuspid versus tricuspid aortic valve stenosis. J Am Coll Cardiol. 2017;69:2579-2589
3. Braghiroli J, Kapoor K, Thielhelm TP, et al. Transcatheter aortic valve replacement in low risk patients: a review of PARTNER 3 and Evolut low risk trials. Cardiovasc Diagn Ther. 2020;10:59-71.
* Corresponding author.
E-mail address: sjorge4@gmx.com (J. Sanz Sánchez)