HOW WOULD I APPROACH IT?
Authors present here an incidental finding of an unusual yet interesting case of a partial anomalous pulmonary venous connection (PAPVC) in a 56-year-old man after a study of moderate exertional dyspnea and angina.
PAPVC is a congenital malformation that can be associated with interatrial septum defects (it has been reported in 10% to 15% of the cases) or happen in isolation. It can be unilateral or bilateral. If found on the right side, the PAPVC often drains into the superior vena cava (sometimes directly into the right atrium or the inferior vena cava), and its association with sinus venosus interatrial communication is a common finding. If on the left side, the left upper pulmonary vein (LUPV) often drains directly into the left innominate vein through an anomalous vertical vein (VV). The presence of 1 bilateral PAPVC is a rarer finding.
In many patients the diagnosis of this entity is merely incidental since patients are often asymptomatic especially for the lack of other congenital cardiac defects. Based on the degree of left-right shunt, the right cavities can be dilated and even in some cases, pulmonary hypertension due to vascular pulmonary vascular bed remodeling can occur. Traditionally, the management of this entity requires surgery to redirect pulmonary flow towards the left cavities through the creation of an intracardiac baffle or the reimplantation of pulmonary vein into the left atrium. Results are favorable in experienced centers since many of these procedures are performed during childhood.1
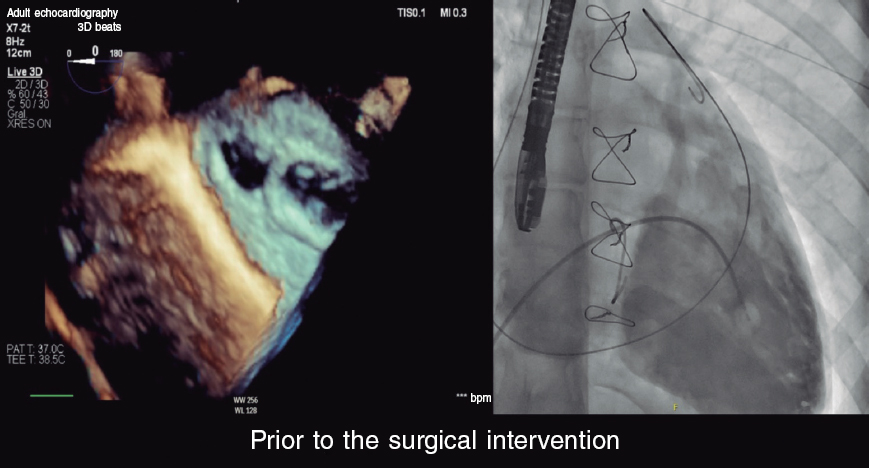
In this case, there is an anatomical particularity conditioning the therapeutic approach: the PAPVC of the LUPV is dual. What this means is that, on the one hand it drains correctly into the left atrium while on the other hand it communicates with the left innominate vein through the VV as confirmed on the images and reconstructions obtained from the multislice computed tomography (CT). In addition to these anatomical findings, we have the information obtained on the echocardiogram showing right cavity dilatation, additional biventricular dysfunction, and traces of severe pulmonary hypertension.
In this context it is advised to perform complete cardiac catheterization to complete the study and assess pulmonary pressures and resistances to discard the presence of significant coronary artery disease in this individual patient.
In patients with an indication for surgical repair due to PAPVC, the European Society of Cardiology clinical practice guidelines of 2020 recommend estimating pulmonary vascular resistances.2 If under 5 Wood units, the procedure is safe and improves functional class while lowering pulmonary pressures. The criterion of pulmonary systolic pressures < 50% of systemic pressure can also be used, as long as pulmonary resistances are less than one third compared to systemic resistances.
PAPVC with dual LUPV drainage is unusual. Its peculiarity is that it allows us to perform a percutaneous procedure to redirect the flow of the LUPV towards the left atrium by occluding the conduit that communicates with systemic venous circulation (usually the VV).3 This allows us to eliminate the left-right shunt and normalize the LUPV return flow thus preventing the setbacks and potential complications associated with heart surgery. In the case presented here, surgical risk is extremely high due to biventricular dysfunction, the presence of severe pulmonary hypertension, and severe 3-vessel coronary artery disease, which could be revascularized in a second surgical act. Therefore, the percutaneous option seems highly attractive.
From a technical point of view, venous access is needed to reach the left innominate vein and access the inside of left atrium through the VV and the LUPV. A femoral vein or left internal jugular vein can be used. Access through the VV can be performed using a long 260 cm 0.035 in hydrophilic guidewire while mounted on a Judkins right or mammary artery specific curve coronary catheter. Once the catheter has been advanced through the left atrium, the hydrophilic guidewire is exchanged for a high-support 260 cm 0.035 in guidewire on which a release system of a proper caliber with respect to the device selected is advanced.
The procedure can be performed without general anesthesia and with sedation only since transesophageal ultrasound is not required. However, it is important to have a good previous radiographic study where the CCTA plays an important role.
Selective angiographies of the VV even with the possibility of temporarily occluding the LUPV with an Amplatzer type of balloon to prevent rapid contrast washout allow us to choose the right device for proper anchoring purposes (with certain compression) avoiding embolization. For its special design and multiple sizes available the Amplatzer Vascular Plug-II device (Abbott, United States) would be my first option. An alternative to it would be the Amplatzer Duct Occluder (ADO)-I device whose design also makes it suitable for different anatomies.
Follow-up after the procedure should include an echocardiogram at 3-6 months to assess the reverse remodeling of right cavities and pulmonary systolic pressure. Performing a thoracic CCTA at 6 months is advised to confirm the complete occlusion of the shunt, and the correct position of the device into the VV.
FUNDING
None whatsoever.
CONFLICTS OF INTEREST
None reported.
REFERENCES
1. Alsoufi B, Cai S, Van Arsdell GS, Williams WG, Caldarone CA, Coles JG. Outcomes after surgical treatment of children with partial anomalous pulmonary venous connection. Ann Thorac Surg. 2007;84:2020-2026.
2. Baumgartner H, De Backer J, Babu-Narayan SV, Budts W, Chessa M, Diller GP, et al. ESC Scientific Document Group. 2020 ESC Guidelines for the management of adult congenital heart disease. Eur Heart J. 2021;42:563-645.
3. Al-Muhaya MA, Alkodami AA, Khoshhal S, Najjar AHA, Al-Mutairi M, Abdelrehim AR. Transcatheter occlusion of the vertical vein in a partial anomalous pulmonary venous connection with dual drainage, case series with literature review. Int J Cardiol Heart Vasc. 2021;37:100889.