HOW WOULD I APPROACH IT?
Authors present an exceptional case of early formation of 1 ventricular pseudoaneurysm after surgical mitral valve replacement complicated with the rupture of the atrioventricular groove that was repaired surgically. The excellent images provided by the computed tomography scan reveal a large multilobulated irregular pseudoaneurysm with an anatomy eligible for percutaneous treatment.
For quite a few years, the closure of pseudoaneurysms has been described in the medical literature as case reports and case series with satisfactory results,1 which is why it is currently considered a therapeutic alternative to surgery.
In this case, surgery does not seem like a good option due to the patient’s critical condition, and the technical complexity involved in the repair, which is suggestive of percutaneous closure through occluder device implantation. The images provided reveal characteristics associated with location and morphology favorable to the use of percutaneous treatment: saccular morphology of the pseudoaneurysm, circular neck, and relatively small size (< 10 mm), apparently demarcated borders (muscular) in the defect, and distance from mitral annulus without the possibility of a compromised prosthetic function.
The excellent images provided by the computed tomography scan allow us to plan the procedure and select the type and size of the device that should be used. Since the transesophageal echocardiogram can lose quality due to the interference from the mechanical mitral valve and in an attempt to prevent the morbidity associated with anesthesia, I would perform the procedure guided by angiography and transthoracic echocardiography only.
Theoretically speaking, the closure of a left ventricular pseudoaneurysm can be performed through 3 different accesses: transseptal, transapical, and aortic retrograde. In this case, transseptal access is not viable due to the presence of a mechanical mitral valve. Transapical access offers the advantages of an easy access to the defect thanks to its proximity and the possibility of using large caliber introducer sheaths when performed surgically. However, it is a more traumatic access that I would spare for cases with mechanical aortic valves involved or when other accesses have already failed. Aortic retrograde access—often via femoral artery but equally feasible from a different arterial access—seems, in my opinion, like the most appealing of all as first-line therapy considering the patient’s status and the location of the defect, which anticipates proper coaxiality in the delivery system. One additional access that has been described is to directly puncture the pseudoaneurysm. But, in this case, it was not a viable option either given the location of the pseudoaneurysm.
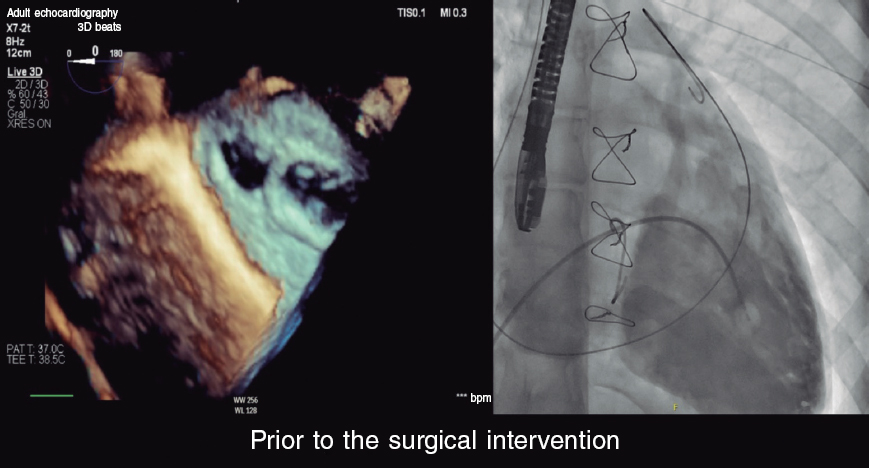
Via retrograde access, one of the sensitive parts of the procedure is accessing the cavity and introducing a delivery catheter of the caliber required. Although multipurpose catheters have traditionally been used, in this case, to access the cavity, the use of coronary catheters with JL, AL or extra-backup L tips can help in our way towards the lateral wall. Guide catheter extension systems are also useful. Although the ideal thing to do would be to place a high-support guidewire once inside the defect, there is this risk of neck laceration and rupture of the pseudoaneurysm. Although the use of a 0.35 in standard Teflon-coated guidewire is feasible, I believe that the use of a 260 cm 0.35 in hydrophilic guidewire could provide greater support because given the size of the aneurysm several loops inside of it would be possible. Afterwards, I would carefully advance the delivery system until it reaches the cavity. If it were a straight introducer, I would pre-form the tip with a Mullins-type curve. Given the deep location of the defect, and depending on the size of the patient, the usual delivery catheters via femoral access can sometimes be a little small. We should make sure we have longer introducer systems (≥ 90 cm) of a caliber that matches the device we’re going to use. For the lack of it, an alternative is to use brachial access to gain over 20 cm of catheter length.
On the occluder device that should be used there are different options available, but specific designs for ventricular septal defects (VSD) and interatrial shunts (IAS) seem like the most suitable ones. The standard device used to close the ductus arteriosus does not have a double retention disc, which does not seem like a good option. «Vascular plug» devices provide good occlusion capabilities, but they require significant oversizing to have enough radial strength. Both VSD and IAS devices provide proper and balanced radial strength, and anatomic conformability of the self-centering waist. Also, the retention discs are large too. I would personally use a muscular VSD occluder device because it comes with symmetrical retention discs and provides great radial strength, particularly in small and intermediate sizes. Regarding size I would choose a slightly oversized device (≈20%), which is why a muscular VSD device of 10 mm or 12 mm would be enough and compatible with 7-Fr or 8-Fr introducer sheaths.
FUNDING
None whatsoever.
CONFLICTS OF INTEREST
None reported.
REFERENCES
1. Dudiy Y, Jelnin V, Einhorn BN, Kronzon I, Cohen HA, Ruiz CE. Percutaneous closure of left ventricular pseudoaneurysm. Circ Cardiovasc Interv. 2011;4:322-326.
* Corresponding author:
E-mail address: drgmarti@googlemail.com (G. Martí Aguasca).