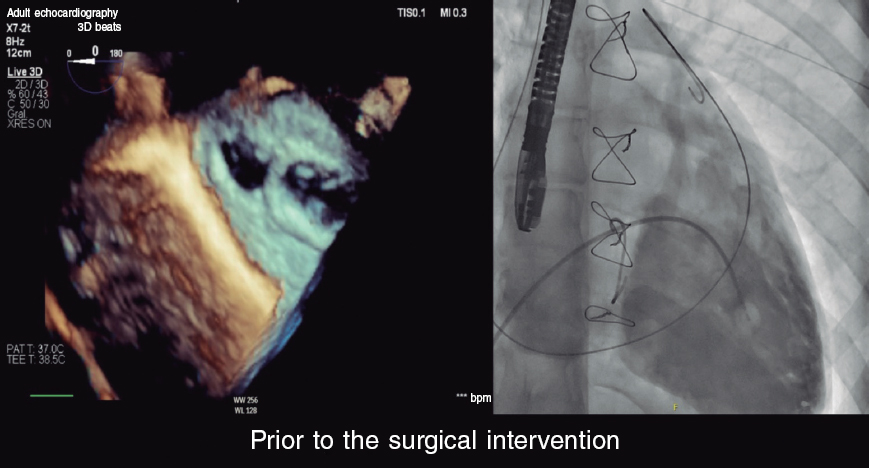
CASE RESOLUTION
Traditionally, the percutaneous closure of this type of defects has been performed using coil embolization procedures and ductus arteriosus closure devices. However, since the appearance of the Amplatzer Vascular Plug (Abbott, United States) its effectiveness in the embolization of certain types of collaterals and fistulae in congenital heart diseases has been recognized.1 Amplatzer Septal Occluders devices (Abbott, United States)—designed for septal defect closure—can be used in certain situations to close non-septal defects, among them, pulmonary systemic fistulae2 and, in general, communications between systemic and pulmonary circulation.
Once the fistula between the pulmonary artery trunk and Fontan circulation was diagnosed, the procedure was performed under general anesthesia, and fluoroscopy and transesophageal echocardiography guidance. Cardiac catheterization was performed via right femoral vein (6-Fr) and right femoral artery (5-Fr).
The retrograde ascending aortic route was used. The left ventricle was accessed and a 0.014 in moderate support guidewire (PT2) (Boston, United States) was used to pass antegradely the subpulmonary stenosis, and the pulmonary trunk until the superior vena cava passing through the fistula. After capturing the border of the guidewire using a 20 mm Gooseneck snare (EV3), an arteriovenous loop was created to facilitate the placement of an 0.035 in Emerald guidewire (CORDIS, United States) and the ascent of a 180º 6-Fr Amplatzer TorqVue sheath (Abbott, United States) via venous access (figure 1).When the border of the sheath was placed inside the left ventricle, a 10 mm muscular Amplatzer VSD occluder device (Abbott, United States) was implanted at subvalvular pulmonary stenosis level followed by a 10 mm Amplatzer Septal Occluder that was successful and uneventfully implanted in the communication between the pulmonary trunk and the pulmonary branch (figure 2) reducing Fontan pressures down to 14 mmHg without flow obstructions towards the branches (videos 1-3 of the supplementary data). The muscular Amplatzer VSD occluder device implanted at subvalvular pulmonary stenosis level was selected for its greater consistency to withstand the hemodynamic stress at that area. The Amplatzer Septal Occluder implanted between the pulmonary trunk and the pulmonary branch was selected for its proper size and morphology regarding the defect. Both devices were implanted so that the jet of pulmonary stenosis would not dilate the pulmonary trunk or triggered the formation of thrombi.
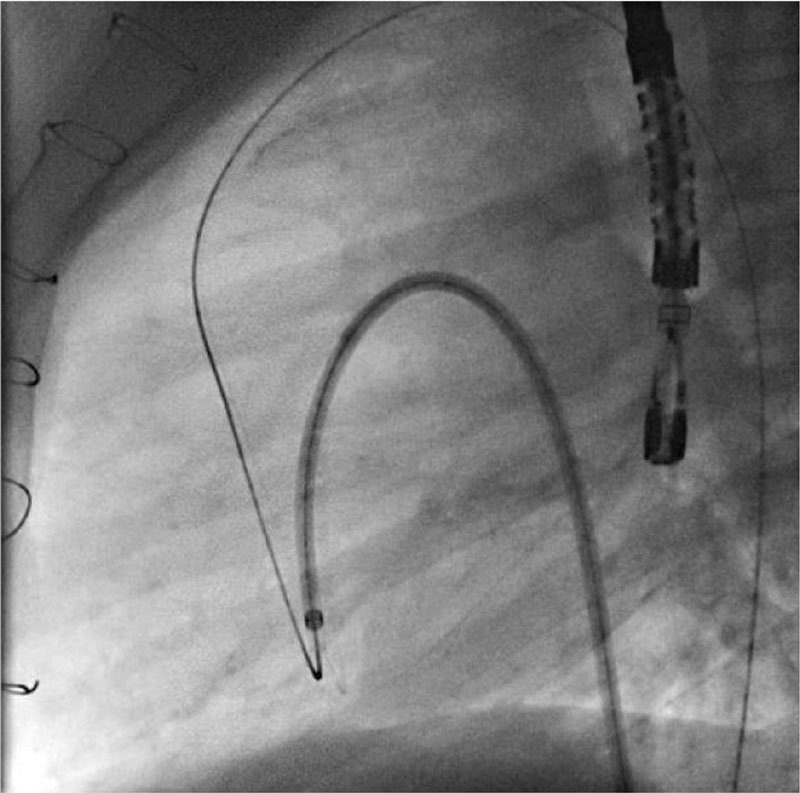
Figure 1. Ascent of the 180º 6-Fr Amplatzer TorqVue sheath towards the left ventricle.
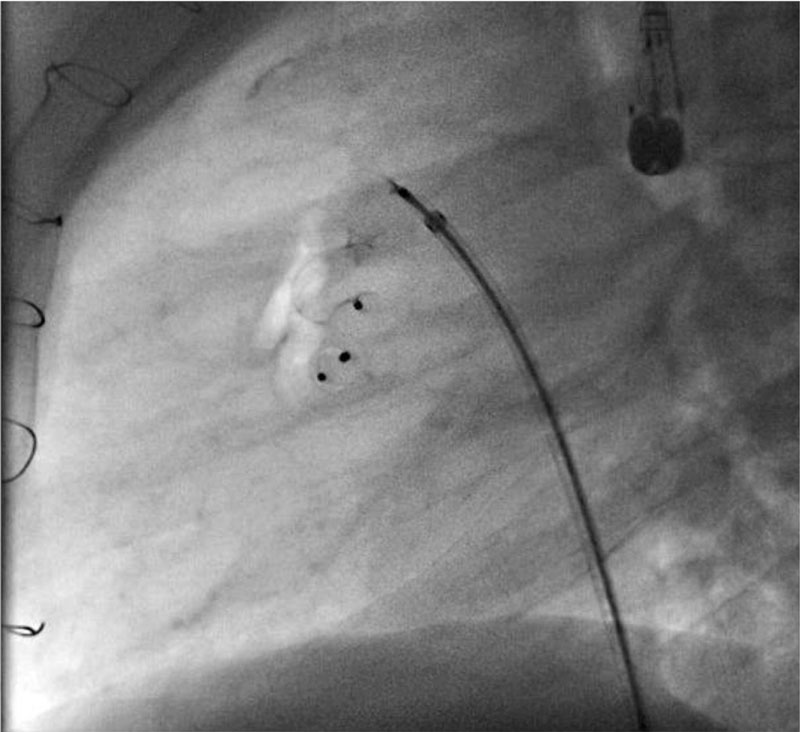
Figure 2. Muscular Amplatzer VSD occluder device implanted at subvalvular pulmonary stenosis level, and Amplatzer Septal Occluder implanted in the communication between the pulmonary trunk and the pulmonary branch.
Finally, left ventriculography revealed the absence of contrast passage towards the pulmonary trunk and Fontan circulation, and the presence of a pseudoaneurysm already present in a former study (figure 3) with a 24 mm × 26 mm orifice opening and a 32 mm × 36 mm saccular formation with calcification in its anterior side. The patient remained on the same treatment prior to the procedure (acetylsalicylic acid, and enalapril), and antiplatelet therapy was reinitiated 24 hours later. The imaging modalities performed at the follow-up confirmed the absence of pseudoaneurysm development, which is why a conservative approach has been adopted ever since.
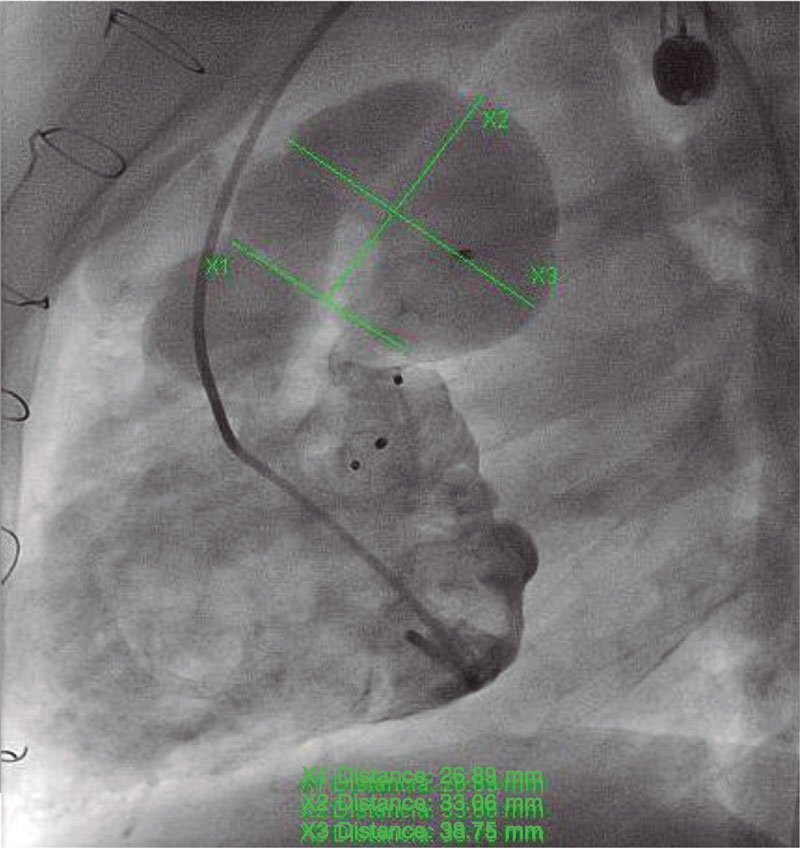
Figure 3. Ventriculography after device implantation showing the aneurysm of pulmonary trunk with visualization of the pseudoaneurysm.
The patient’s corresponding informed consents—that remain on file—were obtained to conduct this study.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
J.M. Blanco Borreguero drafted and reviewed the manuscript. I. Guillén Rodríguez assisted the patient and supervised the manuscript. L. Marcos Fuentes assisted the patient and provided the images. A. Capilla Miranda drafted and reviewed the manuscript. J.F. Coserria Sánchez supervised the manuscript, assisted the patient, and provided the images.
CONFLICTS OF INTEREST
None reported.
SUPPLEMENTARY DATA
Vídeo 1. Blanco Borreguero JM. DOI: 10.24875/RECICE.M22000302
Vídeo 2. Blanco Borreguero JM. DOI: 10.24875/RECICE.M22000302
Vídeo 3. Blanco Borreguero JM. DOI: 10.24875/RECICE.M22000302
REFERENCES
1. Healan S, Varga P, Shah A. Percutaneous transcatheter clousure of a large systemic to pulmonary venous fistulae un an adult patient after extracardiac Fontan. Catheter Cardiovasc Interv. 2015;86:472-475.
2. Awasthy N, Tomar M, Radhakrishnan S, Shrivastava S. Unconventional uses of septal occlude devices: Our experience reviewed. Indian Heart J. 2015;67:128-135.
* Corresponding author.
E-mail address: jblancobo@icloud.com (J.M. Blanco Borreguero).