ABSTRACT
Severe tricuspid regurgitation is independently associated with an increased mortality. Recent studies show that surgery for isolated tricuspid valve (TV) is still associated with the highest surgical risk of all valve procedures. Percutaneous treatments for the management of tricuspid regurgitation have become an alternative to surgery in select patients of high surgical risk. To this day, the most widely used device is MitraClip in the tricuspid position. We reviewed the different devices used today for transcatheter treatments paying special attention to MitraClip. We describe the basic standpoints on transesophageal echocardiography screening and step-by-step procedure for the implantation of the clip. This document stands as a guide to systematize tricuspid valve assessments and the steps involved in device implantation to achieve successful procedures.
Keywords: Tricuspid regurgitation. Transesophageal echocardiography. Transcatheter tricuspid valve repair.
RESUMEN
La insuficiencia tricuspídea se asocia de manera independiente con un aumento de la mortalidad. Investigaciones recientes demuestran que la cirugía aislada de la válvula tricúspide presenta la mortalidad quirúrgica más alta dentro de los procedimientos valvulares. Las terapias percutáneas han surgido como una alternativa a la cirugía en pacientes seleccionados de alto riesgo quirúrgico. El dispositivo MitraClip en posición tricuspídea ha sido el más empleado. El propósito de este artículo es revisar los diferentes dispositivos transcatéter para el tratamiento de la insuficiencia tricuspídea, con especial énfasis en el MitraClip. Se describirán las vistas básicas de la exploración por imagen y el procedimiento paso a paso para la inserción del clip. Este documento pretende ser una guía para la sistematización de la evaluación de la válvula tricúspide y los pasos del implante del dispositivo para asegurar el éxito del procedimiento.
Palabras clave: Insuficiencia tricuspídea. Ecocardiograma transesofágico. Reparación tricuspídea transcatéter.
Abbreviations RV: right ventricle. TTE: transthoracic echocardiogram. TEE: transesophageal echocardiogram. TR: tricuspid regurgitation. TV: tricuspid valve.
INTRODUCTION
Although traditionally tricuspid valve disease has been considered a less serious condition compared to left-sided valvular heart disease, nearly 2 million patients in the United States suffer from moderate tricuspid regurgitation (TR). However, less than 10 000 patients are treated with surgery each year. Functional TR amounts to 90% of all cases of TR and is often due to tricuspid valve (TV) annular dilation (mainly in the anteroposterior diameter) and right ventricle (RV) dilation due to progressive left heart disease.1 TV has been considered “the forgotten valve” for years. This problem may be explained because it was believed that TR was well-tolerated and decreased after treatment of the left-sided valvular heart disease. However, patients with significant TR and heart failure often remain very symptomatic.2 The presence of moderate or severe TR is associated with a higher mortality rate regardless of other variables like the ejection fraction or pulmonary pressures. Mortality rate is > 25% per year.3-5
Current data confirm that repairing the TV while performing left heart cavity surgery is safe. However, reinterventions due to persistent TR are associated with high morbidity and mortality rates.6 Recent studies show that isolated TV surgery is still the valve surgery that is most associated with higher surgical risk and mortality rates between 8.8% and 9.7%.7 Therefore, the percutaneous therapies for the management of TR are an alternative to conventional surgery for patients who, until recently, were eligible for conservative medical treatment only for being high surgical risk patients.
Depending on the repair anatomical target, devices can be divided into coaptation, valve annuloplasty, and replacement devices (whether in the orthotopic or heterotopic position). The most widely used technique is the edge-to-edge repair with the MitraClip device (Abbott Vascular, Santa Clara, United States) in the tricuspid position. The TriValve registry8 included over 650 procedures with the MitraClip device (66% of all percutaneous procedures performed on the TV). This article has 2 objectives: first, to propose a protocol for the echocardiographic assessment of TV in patients with TR to decide on the suitability and feasibility of performing tricuspid repair with this system and guiding the procedure on a step-by-step basis; and second, to review briefly other transcatheter percutaneous devices available today that have already become clinically relevant.
TRICUSPID VALVE IMAGING FOR INTERVENTIONAL PROCEDURES
All patients eligible for tricuspid percutaneous procedures should initially be treated with a transthoracic echocardiogram (TTE). If the valve can be repaired using the edge-to-edge technique, a more advanced assessment using the transesophageal echocardiogram (TEE) should be performed.
TVs that clearly interfere with pacemaker or implantable defibrillator leads, leaflet perforation, severe restriction or significant thickening should not be considered favorable on the TTE. The traditional views for TV assessment through TTE are the parasternal view on the TV modified long axis, the short axis, the modified apical 4-chamber view, and the subcostal view in the short axis.
TEE assessments are crucial to decide whether a procedure using this device is feasible or not. Anatomy should be adequate, yet images should have enough quality to guide the procedure; at times, anatomical variants or the presence of intracardiac prosthetic material can make the procedure unfeasible. The current TEE guidelines established by the American Society of Echocardiography include additional images for TV assessment purposes.9 Multiplane rotations and multiple views are important for correct leaflet identification including adjacent structures used as reference.10
TRICUSPID VALVE ASSESSMENT THROUGH TRANSESOPHAGEAL ECHOCARDIOGRAM
The essential views for a detailed assessment of the TV regarding transcatheter procedures are:
1. Mid-esophageal 4-chamber view at 0º. The septal leaflet (adjacent to the aorta) and the anterior leaflet (adjacent to the RV free wall) can be seen when the transducer is in anteflexion position (figure 1). The posterior leaflet can be seen when it is in the retroflexion position.
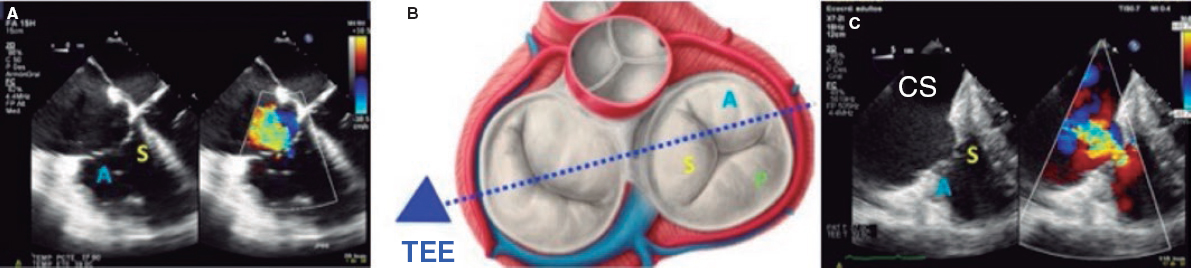
Figure 1. A: mid-esophageal 4-chamber view showing the septal leaflet and the anterior leaflet. B: imaginary view in the 4-chamber view. C: deep transesophageal view. A, anterior; TEE, transesophageal echocardiogram; S, septal; CS, coronary sinus.
To optimize the images of the right heart structures, the transducer should be turned clockwise. The artifacts of the septum and aortic or mitral valves can block the view of the septal leaflet. Tricuspid annulus is measured in this view with open flap-like cusps at the end of the diastole.
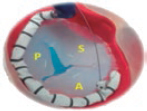
2. Mid-esophageal view of the RV inflow and outflow tract (intercommissural tricuspid view) between 60° and 90°. It shows the imaginary line from the anteroseptal (A-S) commissure up to the posteroseptal (P-S) commissure. The anterior leaflet close to the aorta and the posterior leaflet on the lateral side can be seen (video 1 of the supplementary data). This is the reference view to obtain biplanar images for the assessment of the septal leaflet. By bringing the cursor close to the aorta (X-plane tool), the orthogonal view shows the A-S coaptation and the anterior and septal leaflets. By moving it away from the aorta towards the lateral position, the view shows the P-S coaptation and the posterior and septal leaflets. The same views should be acquired using the Doppler color technique (figure 2) to see whether the origin of the regurgitant jet is of anterolateral or P-S predominance.
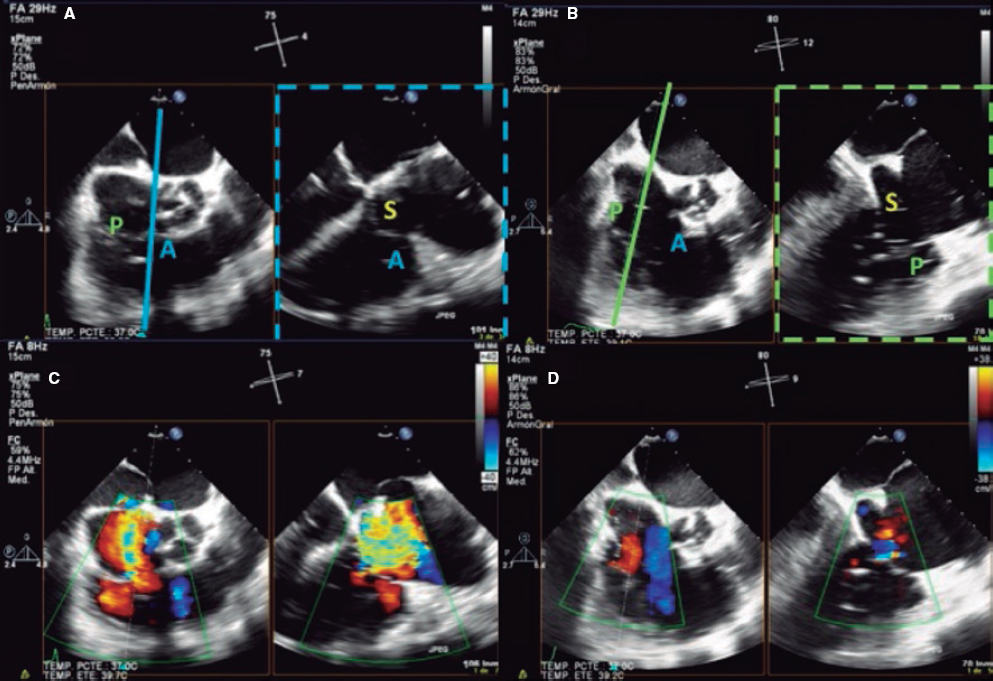
Figure 2. Intercommissural mid-esophageal view. A: when placing the cursor next to the aorta, the orthogonal view shows the anteroseptal coaptation line. B: by moving the cursor towards the most lateral region, the orthogonal view is acquired showing the posteroseptal coaptation line. C and D: color biplanar imaging showing the origin of regurgitation jet. A, anterior; P, posterior; S, septal.
Also, these views are useful to measure the length of the leaflets, see coaptation defects, assess the movement of the leaflets, and see the presence of strings that can make the procedure difficult. The presence of serious restrictions in the movement of the septal leaflet limits the performance of the procedure with the current device.
3. Deep esophageal 4-chamber view at 0°. Since the right inferior border of the heart is close to the diaphragm, the deepest insertion of the TEE transducer reaches the distal esophagus, close to the gastroesophageal junction; it may be that this view will only show the right atrium and coronary sinus and no images of the left atrium (figure 1). This prevents left heart structure-related artifacts from happening like the acoustic shadowing that the mitral prosthetic material can cause on the septal leaflet. This is the optimal view to acquire 3D volumes.
4. Transgastric view. These views are acquired by advancing the transducer towards the stomach. Starting at the baseline transgastric short axis and by rotating the transducer between 20° and 40° and making small clockwise rotations with anteversion, the optimal view to see the 3 tricuspid leaflets is acquired (TV short axis). This is the only 2D (bidimensional) view to see the 3 cusps and commissures simultaneously with the posterior leaflet in the nearby field, the anterior leaflet in the distant field, and the septal one adjacent to the septum (figure 3 and video 2 of the supplementary data). Images need to be optimized to make sure we are positioned on the tip of the leaflets and parallel to the valvular view. To do this we can start with a 2-chamber transgastric view of the RV at around 90º-110°. By using the X-plane tool to place the cursor on the tip of the leaflets, the orthogonal view will be acquired (that will be the TV short axis). This view provides a great deal of information on the TV:
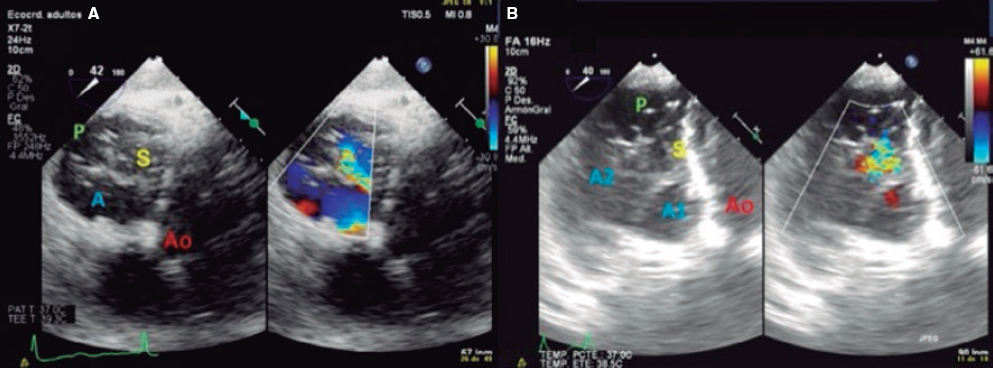
Figure 3. Transgastric view. A: transgastric short axis view of a tricuspid valve with 3 cusps. B: transgastric short axis view of an anatomical variant with 2 scallops in the anterior leaflet. A: anterior; A1, scallop 1 of the anterior leaflet; A2, scallop 1 of the anterior leaflet; Ao, aorta; P, posterior; S, septal.
– Number of leaflets: there are usually 3, but we may find valves with several scallops and even 4 cusps in up to 40% of the patients.11
– Portion of the valve occupying each leaflet: it is very useful to make a graphic representation of what part of the circumference occupies each leaflet.
– Location of the regurgitant jet origin: the leaflets distal edge needs to be approached for a correct visualization of the coaptation defect in the short axis.
The impact of 3D images is not as powerful when performing procedures on the TV compared to the mitral valve. Lang et al.12 suggest a standard image visualization to see a frontal view of the TV by placing the septal leaflet in the inferior position (at the 6:00 hour position). These recommendations were established at the end of the last decade when there were no transcatheter procedures and the surgical view was trying to be replicated. Rotating the image 90° with the aortic valve in the 11:00 hour position is currently the preferred option. This should improve our understanding of the anatomy of the TV by creating a common language between cardiac imaging specialists and interventional cardiologists.
The quantitative analysis of leaflets and annulus is performed through multiplanar reconstructions of 3D views.13 By placing orthogonal views on the tricuspid annulus at the leaflet insertion site, the TV short axis is acquired. After precise adjustment the annular diameters and area dimensions can be obtained (figure 1 of the supplementary data).
The new classification of TR with new degrees of massive and torrential severity can be crucial for the adequate classification of patients and to assess the procedural final results.14
KEY TRANSESOPHAGEAL ECHOCARDIOGRAM VIEWS FOR TRICUSPID VALVE CLIP REPAIR
There are 4 key TEE views to guide the TV clip implantation: 4-chamber, bicommissural, transgastic, and grasping views (capture of tricuspid leaflets inside the device). The former views have already been described; we will now focus on the latter.
Grasping view
This is the view that better shows the leaflets that should be treated and the clip with wide open arms. Finding the best grasping view for clip implantation in the tricuspid position is one of the most complex and important steps during the procedure. The first limitation to find it is that, unlike what happens in the mitral valve, there are no clear anatomical references to guarantee the perpendicularity of the clip with respect to the leaflets. That is why a correct alignment at transgastric level is critical. After finding this alignment, the transducer is removed to look for the grasping view at mid-esophageal level.
The most common strategy is to place a clip between the A and S leaflets and towards the TV most central region. To acquire the grasping view and see the A and S leaflets we should start at the intercommissural view and the multiplanar view, and the grasping view will often be found at around 160° (figure 4). Another option is to perform a TEE transducer sweep from 0° to 180° until finding a direct grasping view to actually see the leaflets and the clip with wide open arms. In any case, every patient should be handled individually since the TV has more anatomical variations than the mitral valve. Also, the angle between the intercommissural view and the coaptation line of the A and S leaflets can be < 90º and mislead the multiplanar assumptions. The real-time 3D TEE can be useful to guide the clip, but not so much with mitral procedures.
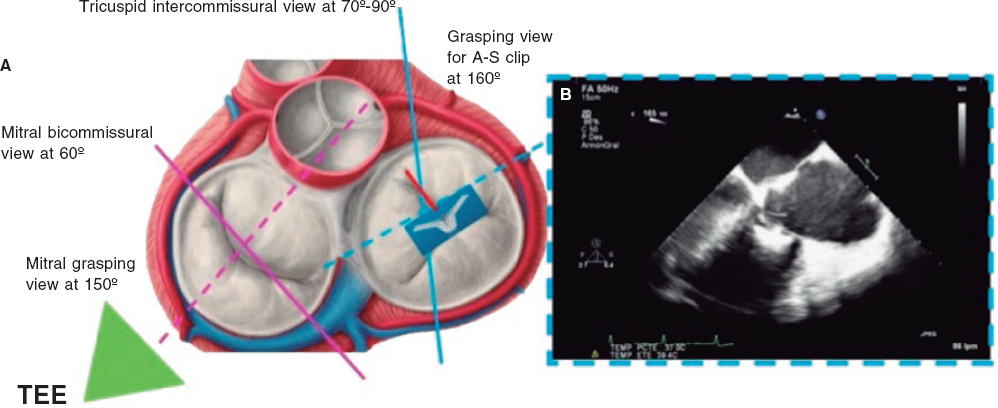
Figure 4. Search for the grasping view for the clip in the anteroseptal (A-S) position with respect to the center of the tricuspid valve. A: grasping view for A-S clip (discontinuous blue line). B: transesophageal echocardiogram (TEE) at 160° showing the clip with wide open arms and the anterior and septal leaflets resting on them.
STEP-BY-STEP PROCEDURE
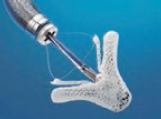
In most cases, the MitraClip implantation in the tricuspid position is performed through femoral access under general anesthesia. It is monitored through fluoroscopy and TEE; the combination of the 2 is essential to achieve optimal results. This is the step-by-step procedure with the key data that should be assessed through imaging modality:
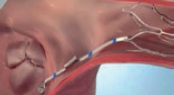
1. Percutaneous access through the right femoral vein using the Seldinger technique. A high-support guidewire is inserted through a 7-Fr introducer sheath towards the superior vena cava (SVC) and after access pre-dilation, the guide catheter (GC) is advanced towards the right atrium.
2. The GC is inserted similar to the way it is done in the mitral valve procedure and the handle is ± rotated approximately 180º in the direction (−). The GC is also rotated 180º counterclockwise to face the anterior region. Once in the right atrium, the dilator and the guidewire are removed, and the catheter is softly aspirated to avoid air embolism. The rotation is then released (−).
3. At this point, the procedure can be performed in 2 different ways. The first is using the handle (+) to increase the GC cuve and point directly at the TV. In this case, the clip catheter is normally inserted into the GC (blue line with blue line) while the GC guarantees the correct positioning of the device to perform leaflet grasping. In the second way, the most widely used, the clip catheter is inserted misaligned (90° counterclockwise or 180°; the authors of this article prefer the 180° misalignment). This technique facilitates the straddling (of the clip radiopaque markers on the GC) of the clip catheter on the GC (this should be done towards the SVC to avoid damaging surrounding structures). After reaching the straddling position, the A or M wheel (depending on the patient’s anatomy) will deflect the tip of the clip towards the target TV region. This technique allows a more versatile range of motion with the device to avoid orientation or distance issues between the tip of the GC and the TV target area. Over the next few steps we will be describing the GC-clip catheter misalignment technique. From the echocardiographic standpoint, these steps are monitored in the 90º-110° bicaval view with biplane trying to not damage any anatomical structures.
4. Once the straddling position has been reached, the A or M wheel (or a combination of the 2) is activated to deflect the clip towards the TV. The wheel selected will depend on the trajectory of the clip. The objective is to achieve the most perpendicular trajectory possible to the TV and avoid, as much as possible, the catheter from coming too close to the interatrial septum (called septal hugging). This circumstance will make the clip point at the TV lateral region. Fluoroscopy in the 45º left, anterior, oblique view is used to see the tip of the clip move towards the left of the screen, away from the septum, pointing towards the operator (figure 2 of the supplementary data). Echocardiographic monitoring will then move from the bicaval to the intercommissural view.
5. When the clip remains perpendicular on the TV, the GC clockwise rotation will move the clip towards the septal position. The GC counterclockwise rotation will move it towards the lateral region. The stabilizer advancement will bring the clip closer to the A-S commissure. After retraction, it will pass to the P-S commissure. This will allow complete range of motion and guarantee the correct location of the grasping (figure 5).
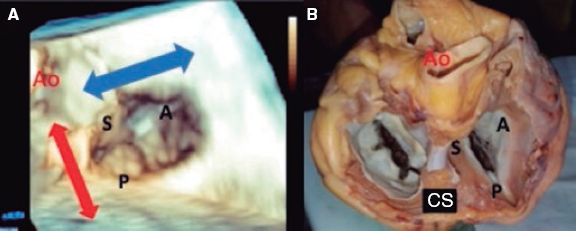
Figure 5. A: transesophageal echocardiogram in 3D zoom mode; periprocedural frontal view. The guide catheter can move inside the right atrium towards the aortic valve by advancing the entire system (red arrow) or towards the posterior leaflet by retracting it; the movement towards the septum (blue arrow) is possible through clockwise rotation of the guide catheter, and towards the right ventricle free wall through counterclockwise rotation. B: explanted heart, anatomic view. A, anterior; Ao, aorta; P, posterior; S, septal; CS, coronary sinus.
6. When the desired position has been achieved, the clip opens and perpendicularity is assessed. This should be done by using a 2D TEE in the transgastric short axis or by 3D zooming in the atrial view. The clip should be rotated smoothly so that it remains completely perpendicular to the desired grasping site (figure 6 and video 3 of the supplementary data).
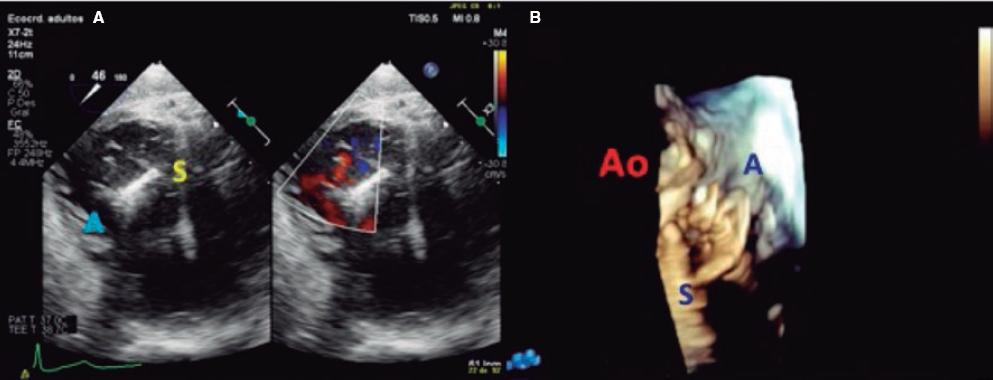
Figure 6. With the clip on the tricuspid annulus the transgastric short axis (A) or 3D zoom (B) should be used to steer the clip. A, anterior; Ao, aorta; S, septal.
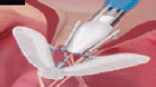
7. Afterwards, the clip closes at 60º and is advanced slowly towards the RV just underneath the leaflets to avoid interfering with RV strings or structures. It is advisable to perform this maneuver through fluoroscopy guidance in a 30º right, anterior, oblique view (RV long axis). This fluoroscopic view will confirm that the clip describes a straight trajectory and maintains its perpendicularity. Fine tuning the rotation of the clip can be made while advancing towards the RV, but it should not be done underneath the leaflets (to avoid string interference). Then, the clip slowly opens at 120º to later retract and guarantee the capture of the leaflets (figure 3 of the supplementary data and video 4 of the supplementary data).
8. Back in the RV, the perpendicularity of the clip with respect to the target coaptation line should be confirmed on the transgastric short axis view. Then, the grasping view should be looked for, that is, the view that better shows the target leaflets and the clip with wide open arms. It is essential to have good ultrasound imaging during grasping to guarantee the correct insertion of the leaflets and the perpendicularity of the arms of the clip. Repeated suboptimal captures of the leaflets should be avoided to not cause excessive damage (TV leaflets are thinner and more fragile compared to the mitral valve leaflets) (figure 7).
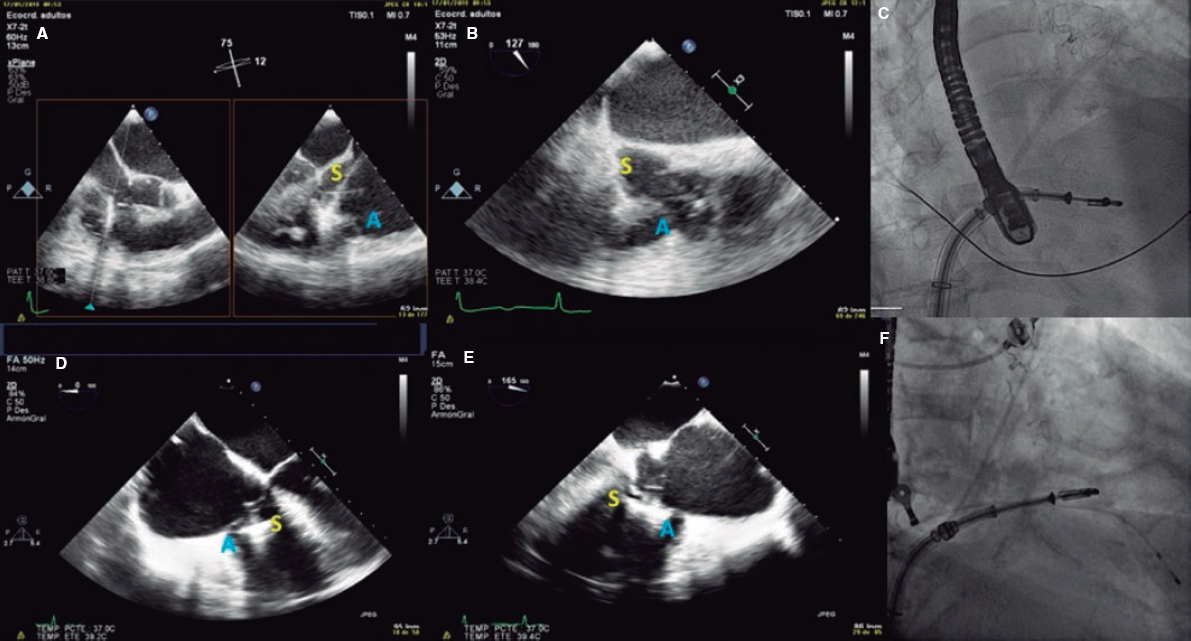
Figure 7. Examples of grasping view. A and D: patient 1, grasping from the intercommissural view at 75° and from the direct grasping view at 125°. B and E: patient 2, direct grasping view acquired at 0° and 160°. C and F: right, anterior, oblique projection to steer the movement of the clip. A, anterior; S, septal.
9. Once the leaflets have been captured, their insertion should be confirmed through multiple 2D TEE views. Also, the presence of tissue bridges should be verified (though 3D TEE views). Multiplane is very useful for assessment purposes. A TTE or an intracardiac echocardiography should be used in cases of uncertain leaflet insertion. The TV mean gradient should be measured to discard stenosis; in general, mean gradients > 3 mmHg are not recommended (figure 8).
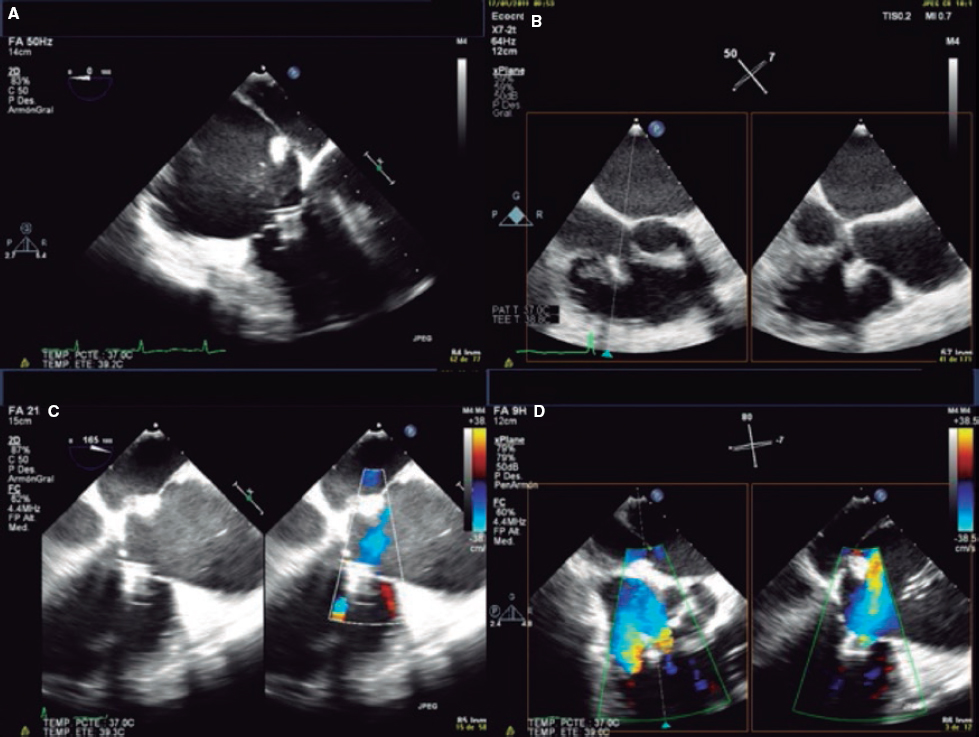
Figure 8. A-D: grasping assessment in multiple views for leaflet insertion assessment and reduction of tricuspid regurgitation.
10. Standard release of the clip. Once it has been completely released, the degree and location of residual TR and need for new clip implantation should be assessed. The position of the new clips will depend on the location and amount of residual TR jets.
Since MitraClip XTR was introduced for the first time, most procedures have been performed using this device. It allows easier captures even when there are coaptation defects among the leaflets. The step-by-step procedure described here refers to the standard MitraClip device used today in mitral procedures. A modification of the Mitraclip device (in the GC) will soon be available to facilitate the steering and positioning of the clip (with excellent results in the 30-day assessment in the TRILUMINATE trial.15) This new version will replace the device mitral version. The steps will change too. Similarly, we will have a new iteration of the system within the next few months (TriClip).
There are several technical possibilities for clip implantation in the TV; the strategy can vary depending on the surface occupied by each leaflet and the location of the regurgitation jet. For the management of A-S coaptation defects, implanting the clip as close as possible to the center of the valve maximizes the reduction of TR. In patients with large coaptation defects, the most common strategy is to treat the A-S coaptation line using several clips and avoid the bicuspidization of the TV (video 5 of the supplementary data). An alternative to this can be to implant a first clip between the anterior and septal leaflets plus a second clip between the posterior and septal leaflets to create a triple-orifice TV (figure 4 of the supplementary data). In this sense, the triple-orifice technique can be superior to the A-S bicuspidization one because it achieves a direct reduction of coaptation defects and counteracts annular dilation by exerting traction forces in the annular anterior and posterior regions. However, the analysis of both strategies did not find any differences between the 2 regarding TR or clinical improvement.16
The selection of patients for tricuspid valve repair should include a combination of clinical, hemodynamic, and anatomical characteristics. Table 1 shows the criteria that define the optimal candidates who are eligible for TR repair using the MitraClip device.17 It should be emphasized that all patients should have symptomatic TR despite receiving optimal medical treatment, high surgical risk, and severe TR.
Table 1. Criteria to identify the ideal candidates for tricuspid repair with the MitraClip device
| Optimal | Possible | Exceptional |
|---|---|---|
| Secondary TR with normal leaflets | Secondary TR with normal appearance of the leaflets or primary TR with valve prolapse | Significant leaflet thickening (rheumatic) or severe valve shortening or destruction or prolapse |
| Small coaptation defect (< 3-4 mm) and good leaflet mobility | Moderate coaptation defect (4-7.2 mm), reduced leaflet mobility | Large coaptation defect (> 7.2 mm) or severe leaflet restriction |
| Good TEE window | Enough echocardiographic window to see the leaflets | Insufficient echocardiographic window to see the leaflets |
| Without PM or ICD leads | Presence of PM or ICD leads without significant leaflet or clip interaction | PM or ICD lead-induced TR |
| Normal-moderately depressed RV function Normal size or moderately dilated RV | Moderately reduced RV function, moderate RV dilation | Severely reduced RV function or severe RV dilation |
| Normal pulmonary systolic pressure | < 60 mmHg-65 mmHg and pulmonary resistances < 4 WU | > 60 mmHg-65 mmHg or pulmonary resistances < 4 WU |
ICD, implantable cardioverter-defibrillator; PM, pacemaker; RV, right ventricle; TEE, transesophageal echocardiogram; TR, tricuspid regurgitation; WU, Wood units. Reproduced with permission from Hausleiter et al.17. | ||
The echocardiographic predictors of success during TR repair with the MitraClip device recently published are: the location of the jet in the A-S coaptation line or central region, and the coaptation defect between leaflets < 7.2 mm are the best predictors with the NTR device.17 Procedural success is also essential because it is a variable independently associated with better prognosis.8,18
OTHER TRANSCATHETER DEVICES TO TREAT TRICUSPID REGURGITATION
Table 2 and table 3 show other transcatheter devices for the management of TR.
Table 2. Transcatheter tricuspid percutaneous repair devices
Table 3. Transcatheter tricuspid valve replacement percutaneous devices
Edge-to-edge repair devices
Pascal
This device is used for edge-to-edge repair by capturing the leaflets of the TV. This device uses clasps (metal blades to capture the leaflets) and paddles (arms that go into the leaflets) to gently grasp the leaflets against a spacer. The clasps move independently for better leaflet capture in the device, while the spacer increases the coaptation surface reducing leaflet stress. The complete system is 22-Fr and the device can be fully elongated favoring its interaction with subvalvular apparatus. The procedure is TEE-guided through transfemoral access and under general anesthesia. Under TEE and fluoroscopy guidance, the device is steered towards the TV and implanted similar to the MitraClip. In a recent communication, the early experience with the Pascal repair system for TR repair was reported. Out of 12 patients treated, 92% reduced their TR in, at least, 1 degree, and all of them improved their functional class at the 30-day follow-up.19 This early experience is encouraging, but more data are needed before using this device in a larger community of patients.
Annuloplasty devices
Trialign
The Trialign device (Mitralign Inc., Tewksbury, Massachusetts) is a transjugular suture-based annuloplasty system to reduce the tricuspid annular diameter through plicated leaflet tissue. During the procedure a pair of polyester sutures are released into the tricuspid annulus next to the anteroposterior and P-S commissures. It is then cinched using a polyester suture that obliterates the tricuspid posterior leaflet and fixates to the atrial side. The results of the SCOUT trial20 have already been published on the successful implantation of a pair of sutures in 14 out of 16 patients (87.5%). The mean reduction after the procedure was 37% in the tricuspid annulus and 59% in the regurgitant orifice area. A pair of sutures can be implanted in the anterior annulus to optimize the final outcome. There was no perioperative mortality and quality of life tests improved at the 1-month follow-up.
Cardioband
The Cardioband system (Edwards LifeSciences, CA, United States) is a percutaneous direct annuloplasty device for TV repair. It is implanted into the tricuspid annulus by a series of anchors that pass through the annulus and into the baseline RV myocardium following the natural shape of the annulus and sparing the septal region where it meets the atrioventricular node. Afterwards, it is cinched using TEE-guidance and the annular septal-lateral diameter is reduced.
The 6-month data of the TRI-REPAIR trial21 have been published. It included 30 patients and procedure technical success was 100%. There was a 9% reduction of the annular septal-lateral diameter, and a 50% reduction of the PISA (proximal isovelocity surface area) at the 6-month follow-up. Eighty-eight percent of the patients had New York Heart Association (NYHA) functional class I-II and improved results in the quality of life tests.
TriCinch
The TriCinch (4Tech Cardio, Galway, Ireland) is an annuloplasty device with a deflectable catheter and a coil system in its distal end that anchors to the tricuspid annulus. This anchoring generates traction in the entire annulus through a band connected to it. The anchor is implanted in the pericardial space to avoid detachment at follow-up. Fixation site is in the middle region of the anterior tricuspid annulus under fluoroscopy and echocardiographic guidance. Then the catheter is tightened to cinch the tricuspid annulus, reduce its A-S dimension, and improve leaflet coaptation. Lastly, a self-expandable nitinol stent is released inside the inferior vena cava (IVC) to secure the system and keep the tension applied. The implantation of the TriCinch device preserves the native anatomy and facilitates other future treatment options. The experience with animals confirms the safety and efficacy profile of this device.22
Valve replacement devices
The transcatheter replacement of the TV can be orthotopic or heterotopic. The first experiences with this technique used aortic valves for percutaneous implantation in the tricuspid position. The “valve-in-valve” or “valve-in-ring” percutaneous implantation of an aortic valve in the tricuspid position is more common. Acceptable short-term results with these procedures have been reported.23 However, the development of transcatheter tricuspid valve replacement systems in native TV is much more interesting.
GATE
The GATE bioprosthesis (NaviGATE Cardiac Structures, Lake Forest, CA, United States) is the only device available today experienced clinically in humans as compassionate use. Also, it is the only device that allows fully orthotopic transcatheter tricuspid valve replacements. It is a xenopericardial leaflet bioprosthesis inserted into a self-expandable nitinol scaffold. The nitinol stent is wider in the ventricular region, giving it a tronco-conical morphology that reduces the transvalvular gradient and minimizes the outflow tract obstruction (since very little material protrudes towards the ventricle). It can be implanted using the jugular approach or the right atrium through minithoracotomy; the femoral access is still under development. The necessary size for the jugular access is ≥ 14 mm because a 42-Fr introducer sheath is used. In both cases, at least 7 cm are necessary from the annular entry site to steer the device (which should be angulated 70º) so that it is perfectly coaxial to the annular plane.
The prosthesis is available in 5 different sizes (36 mm, 40 mm, 44 mm, 48 mm, and 52 mm). A slight oversizing (from 5% to 10%) with respect to the annular dimensions is advisable. To select the device size, it is required to measure the annulus using TEE and computed tomography scan. On the CAT scan, the distance between the annulus to the right coronary artery is a very important feature to avoid damage and have guidance during the procedure. The x-ray projection imaging of the implant is also derived from the CAT scan.
To this day, very few implants have been performed worldwide and always with compassionate use. Recently, the experience of the University of Columbia with 5 consecutive procedures has been published.24 Procedural success was 100% and the route of access was minithoracotomy in all cases. Only 1 patient died within the first 30 days after the procedure. Successful implantation was associated with improved RV remodeling, increased cardiac output, and a better NYHA functional class in most patients.
CAVI
The objective of CAVI (caval valve implantation) is to avoid return flow from TR to the cava veins. This means that the devices per se do not eliminate TR. They improve systemic venous congestion instead. The procedure is performed by implanting valves (in the early experience, non-dedicated balloon-expandable valves) into the origin of the IVC and SVC. The implant can be simple (IVC only) or double depending on the anatomical characteristics of the venous system drainage into the right atrium. There a special self-expandable CAVI device available too, the TricValve (P&F Products Features Vertriebs, Vienna, Austria), that consists of a nitinol stent specifically designed for low-pressure circulation. The largest sizes available are 38 mm and 43 mm for the SVC and IVC, respectively. This means that the optimal diameters of the anchoring area should be ≤ 35 mm (from 28 mm to 43 mm).
Both valves are released using the transfemoral access in a 27-Fr dedicated catheter. The IVC device is released with the portion covered inside the right atrium and the waist inside the diaphragm hiatus to allow the drainage of venous hepatic system. Although it has been the first transcatheter device ever used, it poses some problems because it does not correct TR. The long-term impact of the ventricularization of the right atrium, the persistent overload of the right atrium and RV, the persistent low cardiac output state, and the impact on the function of the RV prevent a more widespread use of this technique. The consequences of thrombotic complications remain unknown too.
Tricento
The Tricento device (NTV AG, Muri, Switzerland) is a sophisticated CAVI concept. It consists of a bicaval, anchored, covered stent with an element of the lateral bicuspid valve made out of a porcine pericardium that only requires a low closing pressure. The nitinol stent has radiopaque markers to allow precise positioning and orientation during implantation. Using the transfemoral approach, the device is released through a 24-Fr catheter. The Tricento is fully repositionable and retrievable until it is completely released. Because of the patients’ great anatomical variability, the stent should be individualized. Once again, the idea is to avoid return flow from TR towards the cava veins. However, all the main setbacks of the CAVI concept are applicable here.
CONCLUSION
There are several transcatheter therapies under development for the management of TR. This article standardized the basic echocardiographic views available for the selection of patients. Also, it stands as a guide for the TV implantation of MitraClip, the most widely used device today. Given the non-stop advance of these devices and cardiac imaging, both imaging and interventional protocols will be jointly developed.
CONFLICTS OF INTEREST
The authors declared no conflicts of interest whatsoever regarding this manuscript.
SUPPLEMENTARY DATA
Video 1. Moñivas Palomero V. DOI: 10.24875/RECICE.M19000091
Video 2. Moñivas Palomero V. DOI: 10.24875/RECICE.M19000091
Video 3. Moñivas Palomero V. DOI: 10.24875/RECICE.M19000091
Video 4. Moñivas Palomero V. DOI: 10.24875/RECICE.M19000091
Video 5. Moñivas Palomero V. DOI: 10.24875/RECICE.M19000091
BIBLIOGRAFÍA
1. Dreyfus GD, Martin RP, Chan KM, Dulguerov F, Alexandrescu C. Functional tricuspid regurgitation:a need to revise our understanding. J Am Coll Cardiol. 2015;65:2331-2336.
2. Taramasso M, Vanermen H, Maisano F, Guidotti A, La Canna G, Alfieri O. The growing clinical importance of secondary tricuspid regurgitation. J Am Coll Cardiol. 2012;59:703-710.
3. Nath J, Foster E, Heidenreich PA. Impact of tricuspid regurgitation on long-term survival. J Am Coll Cardiol. 2004;43:405-409.
4. Topilsky Y, Nkomo VT, Vatury O, et al. Clinical outcome of isolated tricuspid regurgitation. JACC Cardiovasc Imaging. 2014;7:1185-1194.
5. Topilsky Y, Inojosa JM, Benfari G, et al. Clinical presentation and outcome of tricuspid regurgitation in patients with systolic dysfunction. Eur Heart J. 2018;39:3584-3592.
6. Dreyfus GD, Corbi PJ, Chan KM, Bahrami T. Secondary tricuspid regurgitation or dilatation:which should be the criteria for surgical repair? Ann Thorac Surg. 2005;79:127-132.
7. Zack CJ, Fender EA, Chandrashekar P, et al. National Trends and Outcomes in Isolated Tricuspid Valve Surgery. J Am Coll Cardiol. 2017;70:2953-2960.
8. Taramasso M, Alessandrini H, Latib A, et al. Outcomes After Current Transcatheter Tricuspid Valve Intervention:Mid-Term Results From the International TriValve Registry. JACC Cardiovasc Interv. 2019;12:155-165.
9. Hahn RT, Abraham T, Adams MS, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination:recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J Am Soc Echocardiogr. 2013;26:921-964.
10. Hahn RT. State-of-the-art review of echocardiographic imaging in the evaluation and treatment of functional tricuspid regurgitation. Circ Cardiovasc Imaging. 2016;9:e005332.
11. Holda MK, Zhingre Sanchez JD, Bateman MD, Iaizo PA. Right Atrioventricular Valve Leaflet Morphology Redefined:Implications for Transcatheter Repair Procedures. JACC Cardiovasc Interv. 2019;12:169-178.
12. Lang RM, Badano LP, Tsang W, et al. American Society of Echocardiography;European Association of Echocardiography. EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. Eur Heart J Cardiovasc Imaging. 2012;13:1-46.
13. Muraru D, Hanh RT, Soliman OI, et al. 3-Dimensional Echocardiography in Imaging the Tricuspid Valve. JACC Cardiovasc Imaging. 2019;12:500-515.
14. Santoro C, Del Castillo AM, Gonzalez-Gomez A, et al. Mid-term outcome of severe tricuspid regurgitation:are there any differences according to mechanism and severity?Eur Heart J Cardiovasc Imaging. 2019;20:1-8.
15. Nickenig G, Hausleiter J, Hahn R, et al. Percutaneous Edge-to-Edge Repair for Tricuspid Regurgitation:Primary Outcomes from the TRILUMINATE clinical trial. En:EuroPCR;2019 May 21;Paris. Available online: https://abstractbook.pcronline.com/export/pdf/id/110069.
16. Braun D, Orban M, Orban M, et al. Transcatheter Edge-to-Edge Repair for Severe Tricuspid Regurgitation Using the Triple-Orifice Technique Versus the Bicuspidalization Technique. JACC Cardiov Interv. 2018;11:1790-1792.
17. Hausleiter J, Braun D, Orban M, et al. Patient selection, echocardiographic screening and treatment strategies for interventional tricuspid repair using the edge-to-edge repair technique. EuroIntervention. 2018;14:645-653.
18. Besler C, Orban M, Rommel KP, et al. Predictors of Procedural and Clinical Outcomes in Patients With Symptomatic Tricuspid Regurgitation Undergoing Transcatheter Edge-to-Edge Repair. JACC Cardiov Interv. 2018;11:1119-1128.
19. Fam NP. Pascal tricuspid:early experience technology and clinical updates. En:Transcatheter Cardiovascular Therapeutics 2018;2018 Sept 23;San Diego. Available online: https://www.tctmd.com/slide/pascal-tricuspidearly-experience-technology-and-clinical-updates.
20. Hahn RT, Meduri CU, Davidson CJ, et al. Early Feasibility Study of a Transcatheter Tricuspid Valve Annuloplasty:SCOUT Trial 30-Day Results. J Am Coll Cardiol. 2017;69:1795-1806.
21. Nickenig G, Weber M, Schueler R, et al. 6-Month Outcomes of Tricuspid Valve Reconstruction for Patients With Severe Tricuspid Regurgitation. J Am Coll Cardiol. 2019;73:1905-1915.
22. Taramasso M, Latib A, Denti P, et al. Percutaneous repair of the tricuspid valve using a novel cinching device:acute and chronic experience in a preclinical large animal model. EuroIntervention. 2016;12:918-925.
23. McElhinney DB, Cabalka AK, Aboulhosn JA, et al. Transcatheter Tricuspid Valve-in-Valve Implantation for the Treatment of Dysfunctional Surgical Bioprosthetic Valves:An International, Multicenter Registry Study. Circulation. 2016;133:1582-1593.
24. Hahn RT, George I, Kodali SK, et al. Early Single-Site Experience With Transcatheter Tricuspid Valve Replacement. JACC Cardiovasc Imaging. 2019;12:416-429.
Corresponding author: Unidad de Cardiología Intervencionista, Hospital Álvaro Cunqueiro, Clara Campoamor 341, 36312 Vigo, Spain.
E-mail address: roiestevez@hotmail.com (R. Estévez-Loureiro).