To the Editor,
Catheter-directed therapy is indicated in high-risk patients (PE-HR) to treat pulmonary embolism (PE) when systemic thrombolysis is contraindicated or has failed (indication IIA, C). Also, as a bailout therapy in intermediate/high-risk patients (PE-IHR) with hemodynamic impairment as an alternative to systemic thrombolysis (indication IIA, C).1 However, clinical practice guidelines do not specify which is the optimal catheter-directed therapy that should be used. It can be local thrombolysis that consists of the direct infusion of variable doses (around 20% of systemic dose) of thrombolytic drugs (alteplase or tenecteplase) into the pulmonary artery (intra-thrombus) or else mechanical thrombectomy with direct aspiration catheter that consists of thrombus extraction (with or without previous fragmentation). Finally, a mechanical-pharmacological strategy with both techniques combined can be used sequentially.2
This is the early experience of mechanical thrombectomy with direct aspiration using the FlowTriever retrieval/aspiration system (Inari, United States) of 4 Spanish and Portuguese centers. The Hospital Clínico San Carlos ethics committee approved the registry and obtained the participants’ written informed consent. The FlowTriever is a 24-Fr thrombus aspiration system with an oversized tube and accessories to optimize thrombus extraction.3,4 These are 3 cases of PE-HR with contraindication for systemic thrombolysis and 7 cases of PE-IHR treated at centers experienced in catheter-directed therapy, but not with this specific device, whose distribution in these countries started back in 2022. Clinical and procedural characteristics are shown on table 1. Procedure was performed on a mean of 25.7 ± 23 hours since diagnosis. Procedures were successful in 100% of the cases (defined as catheter navigation towards the pulmonary artery with extraction of thrombotic material and no procedural deaths or complications reported) with obtention of abundant thrombus as shown on figure 1. Drug therapy consisted of low molecular weight heparin in 4/10 cases, sodium heparin in 4/10 cases, and no anticoagulation in 2. Fibrinolytic agents were not used in any patients. Access route was femoral in all the cases (24-Fr) with vascular closure in 4/10 (1 Perclose Proglide Abbott Vascular device, United States), figure-of-eight suture in 4/10, and manual compression in 2/10. No vascular access complications were reported. Hemodynamic parameters improved after the procedure as shown on table 1. The volume of blood drawn is significant, and 2/3 of the patients from the PE-HR group required transfusion during admission. Probably after the learning curve the volume should be smaller (in the FLASH cohort, the mean volume was 255 mL.4) Additionally, the FlowTriever system has a filter capable of returning most of the aspirated blood— thrombus-free—back to the patient. However, this version is still not available in Europe.
Table 1. Clinical and procedural data
| Preoperative clinical data (N = 10) | ||
| Age | 57.3 ± 16.1 | |
| Woman | 60% | |
| Past medical history of VTED | 30% | |
| Elevated troponin levels | 100% | |
| Venous lactate at admission (mmol/L) | 3.01 ± 1.9 | |
| Inotropic support | 60% | |
| Ultrasound and hemodynamic parameters | Pre-CDT | Post-CDT |
| RV/LV ratio > 0.9 | 100% | 20% |
| TAPSE | 12.6 ± 3.5 | 19 ± 34 |
| Systemic systolic pressure | 107 ± 24 | 138 ± 23 |
| Heart rate (bpm) | 114 ± 16 | 95 ± 11 |
| Pulmonary systolic pressure (mmHg) | 51 ± 8 | 37 ± 12 |
| Other procedural data | ||
| Contrast (mL) | 156 ± 77 | |
| Aspirated volume (mL) | 396 ± 167 | |
| Procedural time (min) | 126 ± 52 | |
|
CDT, catheter-directed therapy; LV, left ventricle; RV, right ventricle; TAPSE, tricuspid annular plane systolic excursion; VTED, venous thromboembolic disease. |
||
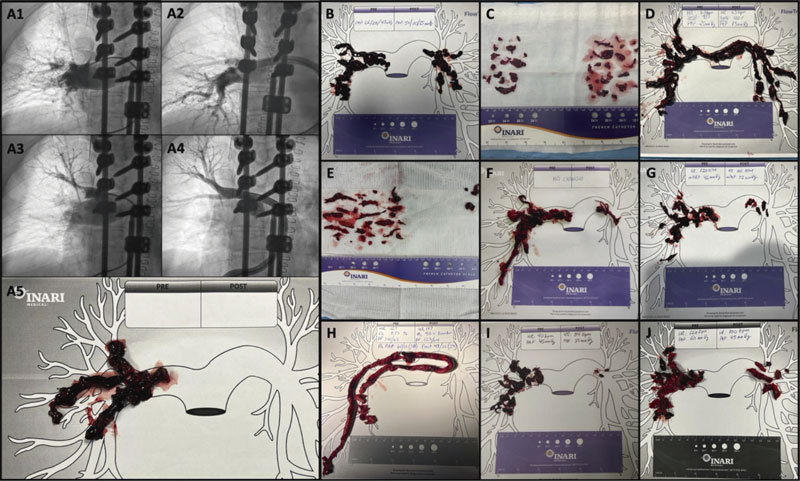
Figure 1. A: case #1 from Hospital General Universitario Gregorio Marañón. Sub-selective angiographies before (A1/A3) and after (A2/A3) the catheter-directed therapy, and material obtained (A5). B to D: material obtained in the cases from Hospital General Universitario Gregorio Marañón. E and F: cases from Hospital Clínico San Carlos. G and H: cases from Hospital La Paz. I and J: cases from Centro Hospitalar de Lisboa Ocidental.
The composite endpoint of major adverse events used in large registries consists of device-related death, major bleeding (Bleeding Academic Research Consortium type ≥ 3b) or procedure or device-related adverse events.4 In our case series 1 major adverse event in 1 procedure was reported: a stroke within the first 24 hours after the procedure due to paradoxical embolism. The presence of patent foramen ovale was confirmed during the procedure due to the crossing of the guidewire towards the left atrium. It is not possible to determine whether the embolism could have been triggered by the manipulation with the guidewire inside the atrium or whether it could have happened spontaneously. The patient recovered and was discharged 9 days after the procedure. Among the cases of PE-HR, 1 patient improved rapidly, and a second one remained in shock with distributive component in the immediate postoperative setting (< 24 h) and improved late with hospital discharge. The third one was admitted after an out-of-hospital cardiac arrest and died due to post-anoxic encephalopathy (the only case of in-hospital death considered unrelated to the procedure). Therefore, mortality 1/3 in the PE-HR and PE-IHR groups was 1/3 and 0/7, respectively. No additional adverse events or readmissions have been reported at 30-day follow-up in any of the patients.
Early diagnosis, risk stratification, and knowledge of these invasive therapies performed by interventional radiologists or cardiologists can contribute to improving the patients’ prognoses. Coordination from response teams to PE to optimize workflow and the decision-making process is of paramount importance.1 Reproducibility, efficacy and, safety are always sought at the dawn of interventional techniques. In a meta-analysis of ultrasound-facilitated local thrombolysis of 2135 patients, efficacy was described as reduced radiographic thrombus loads and ventricular diameters with rates of major bleeding > 5.4%.5 In contemporary studies on mechanical thrombectomy with aspiration with specific devices, a similar efficacy has been described. However, the rates of major bleeding are lower: 1.7% with the Indigo system (Penumbra, United States) in 119 patients,6 and 1.2% with the FlowTriever system (Inari, Estados United States) in 250 patients.4 Therefore, since comparative studies like the PEERLESS trial (NCT05111613) are lacking, mechanical thrombectomy with aspiration seems to offer similar results with a lower bleeding risk. Our early experience with the device confirms that the proper description of the technique and proctoring training during the case can shorten the learning curve significantly. In this early experience, we managed to replicate the results of efficacy and safety previously reported with this promising device for the interventional management of PE.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
P. Salinas: study idea, supervision, investigation, formal analysis, and drafting of the original manuscript. M.E. Vázquez-Álvarez: investigation, data management, review, and edition of the manuscript. A. Jurado-Román: investigation, data management, review, and edition of the manuscript. S. Leal: investigation, data management, review, and edition of the manuscript. M. Huanca: investigation, data management, review, and edition of the manuscript.
CONFLICTS OF INTEREST
None reported.
REFERENCES
1. Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Heart J. 2020;41:543-603.
2. Salinas P. A debate: Terapia farmacológica o invasiva en la tromboembolia pulmonar aguda. Perspectiva del intervencionista. REC Interv Cardiol. 2022;4:240-242.
3. Salinas P, Real C, Fernández-Ortiz A. Tromboaspiración con sistema FlowTriever en embolia pulmonar aguda. Med Clin. 2022;159:154.
4. Toma C, Bunte MC, Cho KH, et al. Percutaneous mechanical thrombectomy in a real-world pulmonary embolism population: Interim results of the FLASH registry. Catheter Cardiovasc Interv. 2022;99:1345-1355.
5. Pei DT, Liu J, Yaqoob M, et al. Meta-Analysis of Catheter Directed Ultrasound-Assisted Thrombolysis in Pulmonary Embolism. Am J Cardiol. 2019;124:1470-1477.
6. Sista AK, Horowitz JM, Tapson VF, et al. Indigo Aspiration System for Treatment of Pulmonary Embolism: Results of the EXTRACT-PE Trial. JACC Cardiovasc Interv. 2021;14:319-329.