To the Editor,
Despite recent improvements in the management of pediatric pulmonary hypertension (PH) and reduced overall mortality rates due to pathway-targeted therapy, there is currently no cure for most patients, and morbimortality remains high with lung transplant being the destination therapy for most.1,2
According to the clinical practice guidelines, the creation of a pre-tricuspid restrictive defect with atrial septostomy (AS) has no strong evidence but may be considered for patients in WHO/Ross III-IV functional class (FC) with syncope and/or severe right ventricular failure who remain unresponsive to maximal pharmacological treatment or as a bridging therapy to lung transplant.1,2 There is relatively little experience among pediatric patients. However, the AS/PH drug combo has improved short-term outcomes like FC, syncope or survival.3,4 When performed by experienced teams the rate of complications drops. However, it is still challenging in the context of high-risk patients. Main fatal events are related to a severe PH crisis or severe cyanosis due to unpredictable shunt size.
Different approaches have been described to create a restrictive AS. Balloon (or blade) septostomy shows early spontaneous closure, so implanting a device in the atrial septum would be a common way to prevent it. The use of a customized fenestrated closure device has been reported, but this shows a high rate of long-term occlusion and a less predictable diameter of the shunt. At our center, we often perform AS with a diabolo-shaped stent in the septum. This allows us to predict the diameter of the shunt, but it has a relatively higher risk of embolization and potential in-stent neointimal hyperplasia.4 An alternative has appeared with the availability of the atrial flow regulator (AFR, Occlutech, Turkey), a self-expandable nitinol wire mesh device made up of 2 symmetric discs connected by a central fenestrated waist. It comes in 8 mm and 10 mm fenestration diameters (4 mm and 6 mm for compassionate use) with disc diameters of 21 mm and 23-mm, respectively, and 2 models (M/L) depending of the height of the central waist (5 mm and 10 mm) associated with the thickness of the atrial septum. A welded ball connector is located on his proximal disc to secure it to the delivery system. The AFR has been used in different scenarios including PH in adults and older children. Although data on smaller ones are more limited it’s still promising.5,6 Larger data are needed to support this evidence.
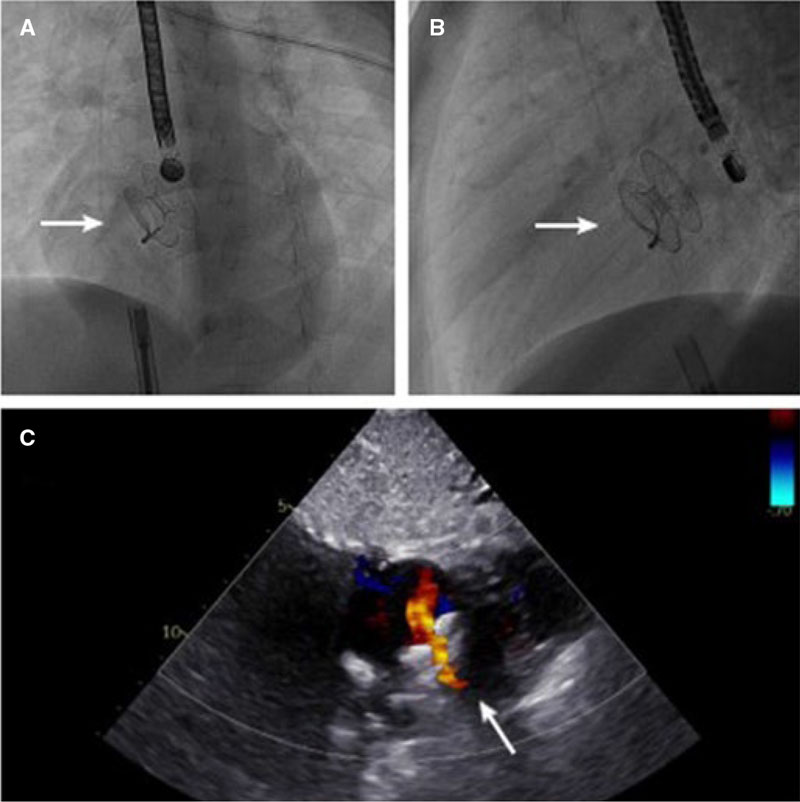
We report our first experience with 2 4-year-old twins (13 kg and 12.5 kg) with PH diagnosed due to recurrent syncope. Both showed severe PH with estimated supra-systemic systolic pulmonary artery pressure, right ventricular hypertrophy, and severe dilatation with preserved function without structural abnormalities. Pharmacological therapy (sildenafil, bosentan, and treprostinil) was started in both twins. Patient #1 had the worst evolution with the persistence of syncope, and up-titration of the medication was required while patient #2 had an impaired FC. The procedure (table 1) was performed on PH-targeted pharmacotherapy, mechanical ventilation on high doses of oxygen, and under fluoroscopy and transoesophageal echocardiography guidance with extracorporeal membrane oxygenation and cardiac surgery teams on standby. Invasive data confirmed the diagnosis. Puncture of the fossa ovalis was performed in patient #1 using a Brockenbrough needle (video 1 of the supplementary data). In patient #2, the left atrium was accessed through a patent foramen ovalis. In both cases, an AFR with an 8 mm fenestration size and model M was used, which was the smallest CE approved device. The atrial septum defect was dilated (video 2 of the supplementary data) with balloons that were smaller than recommended by the manufacturer (Advance balloon, Cook Medical, United States, 8 mm*2 cm in patient #1, and 7 mm*2 cm in patient #2) so that the central fenestration of AFR would be slightly constrained by the interatrial septum, thus allowing a shunt slightly under 8 mm. The remaining procedure is the same as an atrial septal defect closure with a standard device (figure 1, video 3 of the supplementary data). The shunt final sizes were 6.5 mm,and 6.3 mm in patients #1 and #2, respectively on the transoesophageal echocardiography. There were no further complications. Both patients were admitted to the intensive care unit for postoperative care where they remained < 24 hours. They were discharged on aspirin and PH-targeted treatment (sildenafil, bosentan, and treprostinil). Procedures were relatively straightforward, easier, and shorter compared to our diabolo-shaped stent series.4
Table 1. Procedural characteristics
| Patient #1 | Patient #2 | |
|---|---|---|
| Sex | Male | Male |
| Age (years) | 4 | 4 |
| Weight (kg) | 13 | 12.5 |
| RA (mmHg) | 4 | 6 |
| LA (mmHg) | 6 | 8 |
| PA (mmHg) | 63/42/51 | 69/24/46 |
| Ao (mmHg) | 72/40/56 | 80/39/56 |
| Previous O2 saturation (%) | 98 | 98 |
| Postoperative O2 saturation (%) | 94 | 96 |
| Fenestration diameter on the TEE (mm) | 6.5 | 6.3 |
| AFR device (mm) | ||
| Fenestration diameter | 8 | 8 |
| Waist height | 5 | 5 |
| Diameters of discs | 21 | 21 |
| Venous access | femoral | femoral |
| Maximum venous access size (Fr) | 12 | 12 |
| TS access | Brockenbrough needle | PFO |
| Balloon AS dilatation | Advance 35 LP 8 mm × 2 cm |
Advance 35 LP 7 mm × 2 cm |
| Procedural time (min) | 116 | 74 |
| Fluoroscopy time (min) | 25 | 12 |
| Radiation dose (Gy/cm2) | 14.8 | 5.9 |
|
AFR, atrial flow regulator; Ao, aortic systolic/diastolic/mean pressure; AS, atrial septum; LA, left atrial mean pressure; LP, low profile; PA, pulmonary artery systolic/diastolic/mean pressure; PFO, patent foramen ovalis; RA, right atrial mean pressure; TEE, transesophageal echocardiography; TS, transseptal. |
||
Figure 1. A and B: fluoroscopy frames at left anterior oblique (A) and lateral (B) projections showing the atrial flow regulator (AFR) device released in its target position in the interatrial septum (white arrow). C: transthoracic color Doppler echocardiography image at subcostal view of the AFR (white arrow).
At their last follow-up (after 9 and 8 months) both had good disease progression: the device was patent with a bidirectional but predominant left-to-right shunt (videos 4 to 6 of the supplementary data) without significant baseline desaturation (> 94%) or syncope and with normal FC. The N-terminal pro-B-type natriuretic peptide (pg/mL) levels were slightly lower in both patients (163 pg/mL to 159 pg/mL in patient #1 and 376 pg/mL to 157 pg/mL in #2). They are not currently listed for lung transplant and are still on triple therapy up-titrated according to weight gain.
Our data are consistent with those reported,5,6 making the AFR a valid alternative also in small children with severe high-risk PH. Its main advantage is the possibility of creating a shunt of predictable size combined with greater technical ease and low risk of device migration.
The twin’s parents gave their explicit informed written consent to publish the data and images, and to perform the procedure.
FUNDING
None whatsoever.
AUTHORS’ CONTRIBUTIONS
L. Bianco participated in clinical data collection, and draft manuscript preparation with inputs from all the authors. P. Betrián Blasco performed the procedure and supervised the manuscript final version. A. Torrent Vernetta, and A. Sabaté Rotés were involved in the patient’s healthcare plan and reviewed the manuscript. All authors approved the manuscript final version.
CONFLICTS OF INTEREST
None reported.
SUPPLEMENTARY DATA
Vídeo 1. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 2. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 3a. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 3b. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 4. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 5. Bianco L. DOI: 10.24875/RECICE.M23000391
Vídeo 6. Bianco L. DOI: 10.24875/RECICE.M23000391
REFERENCES
1. Hansmann G, Koestenberger M, Alastalo TP, et al. 2019 updated consensus statement on the diagnosis and treatment of pediatric pulmonary hypertension: The European Pediatric Pulmonary Vascular Disease Network (EPPVDN), endorsed by AEPC, ESPR and ISHLT. J Heart Lung Transplant. 2019;38:879–901.
2. Rosenzweig EB, Abman SH, Adatia I, et al. Paediatric pulmonary arterial hypertension: updates on definition, classification, diagnostics and management. Eur Respir J. 2019;53:1801916.
3. Grady RM. Beyond transplant: Roles of atrial septostomy and Potts shunt in pediatric pulmonary hypertension. Pediatr Pulmonol. 2021;56:656–660.
4. Degano Iglesias LA, Sabaté Rotés A, Betrian Blasco P, Torrent Vernetta A, Moreno-Galdó A, Albert Brotons DC. Septostomía auricular en niños con hipertensión pulmonar. Rev Esp Cardiol. 2019;72:688–691.
5. Sivakumar K, Rohitraj GR, Rajendran M, Thivianathan N. Study of the effect of Occlutech Atrial Flow Regulator on symptoms, hemodynamics, and echocardiographic parameters in advanced pulmonary arterial hypertension. Pulm Circ. 2021;11:1–10.
6. Vanhie E, VandeKerckhove K, Haas NA, De Wolf D. Atrial flow regulator for drug-resistant pulmonary hypertension in a young child. Catheter Cardiovasc Interv. 2021;97:E830-E834.