ABSTRACT
Ischemic heart disease is the most common cause of death worldwide. In patients with ST-segment elevation myocardial infarction (STEMI), optimizing primary percutaneous coronary intervention is crucial to improve prognosis. Over the years, many studies have been published on the value of second-generation stents, strategies to reduce myocardial damage, how to achieve complete revascularization and also on percutaneous mechanical circulatory support devices, which all are attractive therapeutic options to treat patients with STEMI complicated by cardiogenic shock. In this review we will be discussing how primary percutaneous coronary intervention can be optimized with respect to stent selection and revascularization strategy to reduce myocardial damage and improve clinical outcomes. In addition, we review published data on the use of mechanical circulatory support devices in patients with STEMI complicated by cardiogenic shock.
Keywords: ST-segment elevation myocardial infarction. Percutaneous coronary intervention. Drug-eluting stent. Cardiogenic shock.
RESUMEN
La cardiopatía isquémica es la causa más común de mortalidad en todo el mundo. En pacientes con infarto agudo de miocardio con elevación del segmento ST (IAMCEST), la optimización de la intervención coronaria percutánea primaria es crucial para mejorar el pronóstico. Durante estos últimos años, se han publicado muchos estudios sobre el valor de los stents de segunda generación, sobre estrategias para reducir el daño miocárdico, sobre cómo conseguir la revascularización completa y finalmente también sobre dispositivos de apoyo circulatorio mecánico percutáneo que representan una opción terapéutica atractiva en pacientes con infarto agudo de miocardio con elevación del segmento ST (IAMCEST) complicado con shock cardiogénico. En esta revisión discutimos cómo se puede optimizar la intervención coronaria percutánea primaria con respecto a la selección de stents y estrategia de revascularización, con el fin de reducir el daño miocárdico y mejorar los resultados clínicos. Además, revisamos los datos publicados sobre el uso de dispositivos de apoyo circulatorio mecánico en pacientes con IAMCEST complicado por shock cardiogénico.
Palabras clave: Infarto de miocardio con elevación del segmento ST. Intervención coronaria percutánea. Stent farmacoactivo. Shock cardiogénico.
Abbreviations
CS: cardiogenic shock. DES: drug-eluting stents. IABP: intra-aortic balloon pump. LV: left ventricle. MVD: multivessel coronary artery disease. PCI: percutaneous coronary intervention. STEMI: ST-segment elevation myocardial infarction. VA-ECMO: venoarterial extracorporeal membrane oxygenation.
INTRODUCTION
Ischemic heart disease is the leading cause of death across the world. Over the last few decades, thanks to the improvements made in reperfusion and antithrombotic therapies, and primary prevention, the relative rates of ST-segment elevation myocardial infarction (STEMI) and long-term and acute mortality have decreased significantly.1 However, despite this reduction, the mortality rate of patients with STEMI is still substantial with in-hospital mortality rates ranging from 4% to 12%, and a 1-year follow-up mortality rate close to 10%.2-4
In STEMI patients, mortality depends on various factors like the Killip classification at presentation, old age, the presence of cardiovascular risk factors, left ventricular function, the spread of the disease in the coronary arteries, and the delayed administration of reperfusion therapy. An early diagnosis and restoration of myocardial blood flow from symptom onset are essential to optimize myocardial salvage and lower the mortality rate.5 Primary percutaneous coronary intervention (PCI) is the reperfusion strategy of choice in STEMI patients if timely performed.5 Optimizing the primary reperfusion strategy is essential to reduce myocardial damage and prevent further reperfusion lesions.
The objective of this review is to give a general overview on current and future percutaneous devices that can potentially improve the benefit of primary PCI including stents, revascularization strategies, and mechanical circulatory support devices for the management of STEMI complicated with cardiogenic shock (CS) like the intra-aortic balloon pump (IABP), the Impella device (Abiomed, Danvers, Massachusetts, United States), TandemHeart (Pittsburgh, Pensilvania, United States), and venoarterial extracorporeal membrane oxygenation (VA-ECMO).
THROMBUS ASPIRATION
Intracoronary thrombus can be found in most STEMI patients. Distal embolization has been reported in 5% to 10% of the cases and can cause obstruction what worsens the results.6 Some time ago, the thrombectomy technique was proposed as a coadjuvant therapy to help restore the coronary blood flow at epicardial level by reducing the chances of distal embolization, the no-reflow phenomenon, and the size of the infarction. Also, it could reduce the thrombotic load prior to stent implantation, thus reducing the rate of associated complications due to stent malapposition. Manual thrombus aspiration was systematically recommended in primary PCI following small randomized clinical trials and a meta-analysis that showed reperfusion improvement with lower cardiovascular mortality rates.7-9 However, after the publication of 2 large statistically powered randomized clinical trials to detect the superiority of routine manual aspiration vs PCI, only 1 change in the recommendation has occurred.5 Neither the TOTAL (N = 10 732 patients) nor the TASTE (N = 7244 patients) clinical trials showed any differences with the thrombectomy in the clinical outcomes compared to the PCI alone.10,11 Also, the TOTAL trial posed a safety issue associated with a higher risk of stroke in patients treated with thrombectomy compared to those treated with PCI alone.12
Based on these data, thrombus aspiration is now not recommended as a routine strategy in STEMI patients treated with a PCI primary PCI. However, it can be considered in patients with high thrombotic load after vessel recanalization. A subanalysis of the EXAMINATION trial (N = 1498) confirmed that the use of thrombectomy was associated with a higher rate of direct stenting, a lower rate of postdilatation, and a smaller number of stents implanted with a larger stent size.13 However, the optimized angiographic result did not impact the long-term outcomes since no differences were seen in the clinical endpoints reported between the arms at the 2-year follow-up.
SELECTING THE TYPE OF STENT
Coronary stent implantation is the recommended therapy during a primary PCI for the management of STEMI patients. Direct stenting without predilatation in STEMI culprit lesions can reduce the embolization of the plaque components, the rate of no-reflow phenomenon, and increase myocardial perfusion.14 This hypothesis was confirmed in the post-hoc analysis of the HORIZONS-AMI trial and the EUROTRANSFER registry; both showed a lower mortality rate at the 1-year follow-up associated with the use of direct stenting.15,16
Delayed stent implantation after restoring coronary flow has also been proposed through a minimalistic mechanical procedure to reduce the risk of no-reflow phenomenon.17 Several observational trials showed benefits in terms of an improved left ventricular ejection fraction and a lower rate of adverse events with the delayed compared to the immediate stent implantation strategy in STEMI patients.18,19 Also, a proof-of-concept randomized clinical trial (DEFER-STEMI, N = 411) reported a lower no-reflow phenomenon rate with the delayed stent implantation strategy in a population of patients with STEMI.20 However, the DANAMI 3-DEFER trial randomized 1215 STEMI patients to receive delayed vs immediate stent implantation. At the 2-year follow-up no differences were seen in the primary endpoint of all-cause mortality, hospital admission due to heart failure, recurrent infarction, and any unplanned revascularizations between the study groups (18% vs 17%; hazard ratio [HR], 0.99; 95% confidence interval [95%CI], 0.76–1.29; P = .92).21 Afterwards, the MIMI randomized clinical trial (N = 140), that excluded patients with a high thrombotic load, and the INNOVATION trial (N = 114) did not show any changes either in the infarction size or microvascular obstruction with the delayed compared to the immediate stent implantation strategy.22,23 Finally, a meta-analysis of randomized and observational clinical trials found no improvement either in the rates of no-reflow, death, myocardial infarction or repeat revascularizations with the delayed stent implantation strategy for the management of STEMI.24 Surprisingly, an improved left ventricular (LV) function was reported in the long-term. For all these reasons, to this date, the delayed stent implantation strategy is ill-advised in the primary PCI.
Another aspect to be taken into consideration before performing a primary PCI is what device should be implanted. Several randomized clinical trials and meta-analyses assessing first-generation drug-eluting stents (DES), whether sirolimus or paclitaxel, showed lower in-stent restenosis and target lesion revascularization rates compared to conventional bare metal stents (BMS).25-32 However, safety concerns soon appeared given the high rate of late thrombosis associated with first-generation DES.33-35
To overcome this problem, second-generation DES with different drugs, thinner struts, and durable or bioresorbable polymers more biocompatible have been designed. The COMFORTABLE AMI clinical trial randomized 1161 STEMI patients on a 1:1 ratio to receive a BMS or a biodegradable polymer biolimus-eluting stent. At the 1-year follow-up a lower rate of major adverse cardiovascular events was reported in the biolimus-eluting group compared to the BMS group (4.3% vs 8.7%; HR, 0.49; 95%CI, 0.30–0.80; P = .044) mainly triggered by a lower risk of spontaneous myocardial infarction and target lesion revascularization.36 Similarly, at the 2-year follow-up a lower rate of major adverse cardiovascular events was reported in the biolimus-elutin group (5.8% vs 11.9%; HR, 0.48; 95%CI, 0.31–0.72]; P < .001).37 Both at the 1- and 2-year follow-ups, the rates of definitive or probable stent thrombosis were also numerically lower with the DES although not statistically significant.36,37
The EXAMINATION clinical trial38,39 randomized 1498 STEMI patients to receive a second-generation everolimus-eluting stent (EES) or a BMS. At the 1-year follow-up, the EES was superior to the BMS with a significantly lower rate of definitive thrombosis, and definitive or probable thrombosis (0.5% vs 1.9%, and 0.9% vs 2.5%, respectively; P = .019 for both]).38 Also, at the 5-year follow-up, the rate of all-cause mortality was significantly lower in the EES group compared to the BMS group (9% vs 12%; HR, 0.72; 95%CI, 0.52–0.10]; P = .047)39. Also, a meta-analysis of both the EXAMINATION and the CONFORTABLE-AMI clinical trials found a significant reduction of the risk of definitive thrombosis with the use of the DES (HR, 0.35; 95%CI, 0.16–0.75; P = .006) compared to the BMS.40 Given the conclusions of these clinical trials, the DES is currently the device of choice according to the recommendations established in the clinical practice guidelines published by the European Society of Cardiology on the management of STEMI.5
The researchers of the EXAMINATION trial investigators have recently reported that the 10-year follow-up results confirm the superiority of EES over BMS in terms of patient or device related cardiovascular adverse events. Between the 5- and the 10-year follow-up periods, a low rate of adverse cardiovascular events was associated with failed devices.41
Fully bioresorbable vascular scaffolds (BVS) were introduced to overcome the long-term limitation of the permanent presence of metal within the coronary artery. The data on their use for the management of STEMI is still limited. Although unavailable for clinical use, we believe the existing data should be discussed. The early experiences with the Absorb BVS (Abbott Vascular, Illinois, United States) for the management of STEMI showed positive and negative clinical results alike.42-44 The TROFI II clinical trial randomized 191 STEMI patients to receive a BVS or a EES and found no differences between the 2 regarding scarring of the infarct-related artery.45 However, other studies showed disturbing data following the high rate of thrombosis with the BVS device. In the BVS EXAMINATION clinical trial, the safety and efficacy profile of BVS vs EES was compared in STEMI patients. At the 1- and 2-year follow-up periods, no differences were found in the device-oriented composite endpoint between both groups.46,47 We should mention that at the 2-year follow-up, the rate of definitive thrombosis was often higher in the BVS group compared to the EES group (3.3% vs 1.0%; P = .081). At the 5-year follow-up, the risk of the device-oriented composite endpoint was higher in the BVS group, indicative that the chances of obtaining favorable outcomes at a very long-term follow-up is low.48
The BVS STEMI STRATEGY-IT clinical trial was designed to reduce the rate of adverse events. It proved that a prespecified BVS implantation strategy in STEMI patients treated with a primary PCI was feasible and yielded good clinical outcomes at the 30-day and 1-year follow-up periods (rate of device thrombosis between 0.2 and 0.4%, respectively).49,50
We should mention that the long-term results of randomized clinical trials that proved a significantly higher rate of BVS thrombosis were the reason of their withdrawal from the market.51-53
The Magmaris (Biotronik, Bülach, Switzerland) is a magnesium-based bioresorbable sirolimus-eluting stent. It has shown promising early results at the 1-year follow-up in stable patients with very limited data on STEMI.54 The MAGSTEMI trial is the only randomized clinical trial that compared the efficacy and safety profile of the Magmaris device in STEMI patients.55 This study randomized 150 patients to receive a primary PCI with Magmaris vs sirolimus-eluting stents using a prespecified implantation technique. Compared to the sirolimus-eluting stent, the Magmaris device showed a greater capacity of vasomotor response to drug agents (whether independent from the endothelium or endothelium-dependent) at the 1-year follow-up. However, the Magmaris device was associated with a lower angiographic efficacy and a higher rate of clinical restenosis, but no thrombotic issues.56 In the prespecified MAGSTEMI-optical coherence tomography substudy, both the Magmaris and the sirolimus-eluting stent showed a low degree of neointimal healing. However, lumen dimensions were smaller with the Magmaris at the 1-year follow-up. Although Magmaris advanced bioresorption state complicates the assessment of the scaffold, this seems to be the main mechanism of restenosis.57,58 Cases of significant delayed resorption of the Magmaris device have been reported, and intraluminal scaffold remnants have been found 2 years after implantation.59
MULTIVESSEL CORONARY ARTERY DISEASE
Around 50% of STEMI patients show multivessel coronary artery disease (MVD).60 Multiple clinical trials have studied the best revascularization strategy: treat the culprit lesion only vs complete revascularization. The PRAMI trial randomized 465 patients with STEMI and MVD to culprit lesion treatment only or revascularization of all obstructive lesions (angiographic stenosis > 50%) during the index procedure. Complete revascularization during the index procedure was associated with a 65% lower relative risk in the primary endpoint (cardiac death, infarction or refractory angina) compared to treating the culprit lesion only.61 Similarly, the CvLPRIT trial (N = 269) showed that complete revascularization (angiographic stenosis > 70%) during the index hospitalization was superior to the PCI of the infarct-related lesion only in the composite endpoint of death, reinfarction, heart failure, and repeat revascularization at the 12-month follow-up.62
Measuring the fractional flow reserve of coronary flow to guide the need for non-culprit lesion revascularization has been proposed. The DANAMI-3-PRIMULTI trial (N = 627) proved that fractional flow reserve-guided complete revascularization significantly reduced the risk of future cardiovascular adverse events compared to any other invasive procedure after the primary PCI. This effect is due to a significantly lower number of repeat revascularization procedures because the rates of all-cause mortality and non-fatal reinfarction did not vary between the groups.63 Also, the Compare-Acute trial (N = 885) proved that fractional flow reserve-guided complete revascularization during the index procedure significantly reduced the rate of cardiovascular adverse events.64
The COMPLETE clinical trial included 4041 patients randomized to complete revascularization vs culprit lesion therapy who were followed for up to 3 years. Complete revascularization was superior to the PCI only in the culprit lesion to reduce the risk of cardiovascular death or myocardial infarction, and the risk of cardiovascular death, myocardial infarction or ischemia-induced revascularization.65 Currently, the BioVasc trial (NCT03621501) is studying how to optimize the treatment algorithm for patients with acute coronary syndrome with MVD to find out what the best time is to perform complete revascularization, whether immediate or delayed.66
According to the current guidelines of the European Society of Cardiology, during hospitalization and before hospital discharge, the complete revascularization of the non-culprit lesions of patients with STEMI and MVD should be considered.5 However, this indication is likely to change after the publication of the upcoming COMPLETE trial clinical results.
In the specific case of CS-complicated STEMI patients, the CULPRIT-SHOCK trial randomized 1075 patients with CS-complicated STEMI with MVD to be treated with a PCI on the infarct-related artery or a multivessel PCI of all lesions (angiographic stenosis > 70%). Both at the 30-day and 1-year follow-up, the PCI performed on the culprit lesion only significantly reduced the risk of death or renal replacement therapy.67,68 This difference was mainly triggered by a significantly lower mortality rate. In this sense, the European Society of Cardiology published an update of its guidelines on the management of STEMI where, in the presence of STEMI with CS and MVD only the culprit lesion of the acute event should be treated.69
CARDIOGENIC SHOCK
Around 5% to 8% of STEMI patients also show CS, which is defined as persistent hypotension (systolic pressure < 90 mmHg) with signs of peripheral hypoperfusion. CS is one of the leading causes of death with in-hospital mortality rates that can be over 50%.70 In patients with CS refractory to drug therapy, percutaneous mechanical circulatory support can help reduce the LV workload and oxygen demand, keep organs and coronary arteries perfused, and stand as a bridging therapy to a more definitive therapy.71,72 Currently, we have assist devices from the LV to the aorta (IABP and Impella), from the left atrium to systemic arterial circulation (TandemHeart), and from the right atrium to systemic arterial circulation (VA-ECMO). The technical characteristics of percutaneous mechanical circulatory support systems currently available are shown on table 1.
Table 1. Technical characteristics of percutaneous mechanical circulatory support devices currently available
| BIAC | Impella | TandemHeart | ECMO-VA | |
|---|---|---|---|---|
| Hemodynamic effect | Unloading of LV pressure and volume | Unloading of LV pressure and volume | Unloading of LV volume | Unloading of RV and LV pressure and volume |
| Mechanism | Aorta | LV to the aorta | LA to the aorta | RA to the aorta |
| Heart blood flow | 0.3 L/min to 0.5 L/min | 1 L/min to 5 L/min | 2.5 L/min to 5 L/min | 3.0 L/min to 7.0 L/min |
| Peripheral resistances | ↓ | ↓ | ↑ | ↑↑↑ |
| Size | 8 Fr | 13-Fr to 22-Fr | 21-Fr inflow cannula and 15-Fr to 17-Fr outflow cannula | 18-Fr to 21-Fr inflow cannula and 15-Fr to 22-Fr outflow cannula |
| Implantation complexity | Low | - Moderate with Impella 2.5 - High with Impella 5.0 | High | High |
| Recommended use duration | Weeks | 7 days | 14 days | 7 days |
| Contraindications | - Severe aortic regurgitation - Aortic dissection - Severe peripheral vascular disease | - Severe aortic valvular heart disease - Aortic mechanic valve - Thrombus in the LV - Severe peripheral vascular disease - Contraindication to anticoagulation | - Severe peripheral vascular disease - Thrombus in the LA - Contraindication to anticoagulation - Moderate-to-severe aortic regurgitation - Interventricular septal defect | - Moderate-to-severe aortic regurgitation - Severe peripheral vascular disease - Contraindication to anticoagulation |
| Complications* | - Thrombocytopenia - Thrombosis - Arterial flow obstruction due to incorrect positioning - Aortic dissection or rupture - Plaque or air embolism | - Hemolysis - Device migration - Lesion or aortic failure - LV perforation or tamponade - Ventricular arrhythmia | - Migration of the cannula - LV perforation or tamponade - Thromboembolism - Air embolism during the insertion of the cannula - Development of interatrial shunt | - Circuit thrombosis - Upper body hypoxia due to incomplete retrograde oxygenation - LV dilatation - Systemic gas embolism |
|
* Complications that are common to all devices: bleeding and infections associated or not with puncture site, vascular complication, and neurological damage. Fr, French sizing; IABP, intra-aortic balloon pump; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle; VA-ECMO, venoarterial extracorporeal membrane oxygenation. |
||||
Left ventricular assist device to the aorta
Intra-aortic balloon pump
Intra-aortic balloon pump (IABP) has been the most commonly used mechanical support device until 2010. After this year, its use dropped significantly after some clinical trial results questioned its efficacy.73 It requires an 8-Fr introducer sheath into the femoral or axillary arteries and consists of a balloon mounted over a catheter that is placed in the descending aorta. The balloon is inflated during diastole and deflated during systole. The IABP increases the diastolic aortic pressure, reduces the aortic systolic pressure, increases the mean systemic arterial pressure, reduces the LV volume and diastolic pressure, and increases the coronary perfusion pressure. However, the hemodynamic support provided by the IABP is strictly associated with the LV function since it is less effective when it shows severe dysfunction.
Observational trials and meta-analyses have traditionally supported the use of IABP in CS-complicated STEMI.74-76 However, prospective clinical trials have showed no benefit whatsoever from the IABP therapy in patients with STEMI with or without CS. The CRISP AMI trial (N = 337) showed that IABP implantation right before the PCI to treat an anterior STEMI without CS did not reduce the size of the infarction or improve the short-term survival rate.77 The TACTICS trial randomized 57 patients with acute myocardial infarction and 48 hours of fibrinolytic therapy to receive the IABP or optimal medical therapy. This trial found no differences in the mortality endpoint at the 6-month follow-up.78 Also, the IABP SHOCK trial6 randomized 45 patients with STEMI and CS for IABP implantation or standard medical therapy and found no significant hemodynamic improvements after additional therapy with the IABP.79
The IABP SHOCK II trial randomized 600 patients with STEMI and CS not associated with mechanical complications to compare IABP implantation the optimal medical therapy.80 It was expected that all patients underwent early revascularization (predominantly with PCI) and received the optimal medical attention available. At the 30-day follow-up, no differences were seen in the all-cause mortality rate between the IABP and the optimal medical therapy (39.7% vs 41.3%; relative risk, 0.96; 95%CI, 0.79-1.17]; P = .69) or in the length of the stay in the intensive care unit, renal function, major bleeding, peripheral ischemic complications, sepsis or stroke.80 At the 12-month follow-up, no differences were seen either in the mortality rate and secondary endpoints reported.81 A meta-analysis of 12 randomized clinical trials and 15 observational studies found no benefits from the IABP therapy in the management of STEMI or in the 30-day mortality rate regardless of the presence (odds ratio [OR], 0.94; 95%CI, 0.69–1.28) or absence (OR, 0.98; 95%CI, 0.57–1.69) of CS. Currently, based on the available evidence, the European Society of Cardiology clinical practice guidelines always contraindicate the IABP in patients with CS.5
The Impella system
The Impella system is a continuous axial flow pump that is inserted into the LV in a retrograde fashion through the aortic valve and provides active support by expelling suctioned blood from the LV into the ascending aorta, thus restoring blood flow to ischemic organs.82 The Impella device increases the mean arterial pressure, reduces the LV pressure and volume, and increases coronary flow. It comes in 3 different sizes: 2.5 (maximum output, 2.5 L/min), 3.7 (Impella CP, maximum output, 3.7 L/min), and 5.0 (maximum output, 5 L/min). The smallest devices can be placed percutaneously through a 12-Fr to 14-Fr introducer sheath and the 5.0 device through a 22-Fr introducer sheath.82
Two large registries confirmed the safety of the Impella 2.5 system in high-risk complex PCIs.83,84 The ISAR-SHOCK trial randomized 26 patients with STEMI and CS to receive the Impella 2.5 system or the IABP. The endpoint, a change in the cardiac index from baseline to 30 min after implantation, improved significantly in the Impella group. However, secondary endpoints like lactic acidosis, hemolysis, and mortality at the 30-day follow-up did not vary between the 2 arms.85 At the 30-day follow-up, the cohort overall mortality rate was 46%. The IMPRESS in Severe Shock clinical trial randomized 48 patients with mechanical ventilation associated with CS after STEMI to receive the Impella system or IABP implantation. We should mention that the device was implanted at the discretion of the treating physician. The trial proved that, compared to the IABP, the Impella system did not reduce the 30-day mortality rate, and the overall mortality rate at the 6-month follow-up was 50%.86 Both vascular complications and major bleeding were more common in the Impella group.
We should mention that, to date, the Impella device has not been compared to standard therapy in patients with CS in a proper statistically powered randomized clinical trial regarding relevant clinical events. In this sense, the DanGer Shock clinical trial (NCT01633502) will include 360 patients with STEMI and CS who will be randomized to receive circulatory support with the Impella system or standard medical therapy.87 The study is still recruiting patients and its primary endpoint is all-cause mortality at the 6-month follow-up.
Back in 2018 a groundbreaking idea was introduced: the use of the Impella system to unload the LV and, therefore, reduce the size of myocardial infarction in animal models with STEMI but without CS.88 These animal models led to the design and conduction of the DTU-STEMI pilot study that randomized 50 STEMI patients without CS to LV unloading with the Impella CP device or optimal medical therapy. This trial revealed that LV unloading therapy prior to STEMI reperfusion with the Impella device was feasible and not associated with a significant delay in STEMI reperfusion.89 However, the use of the unloading therapy was not associated with a reduced infarction size at 1-month follow-up. Currently recruiting patients, the DTU-STEMI clinical trial (NCT03947619) will be enrolling 668 patients to test the hypothesis of the use of the LV unloading therapy with the Impella CP device to reduce the infarction size as seen on the cardiovascular magnetic resonance imaging.
Left atrium-to-systemic circulation assist devices
TandemHeart
The TandemHeart is an extracorporeal ventricular assist device to aspirate oxygenated blood from the left atrium and pump it into the lower abdominal aorta or iliac arteries to avoid running through the LV. The 21-Fr inflow cannula is inserted via femoral vein access and advanced through the interatrial septum towards the left atrium. The 15-Fr to 17-Fr outflow arterial cannula and the system can provide up to 5 L/min of blood flow.90 The device basically reduces the LV preload and left atrial volume by removing blood from the left atrium, thus reducing the LV stress and workload. It also increases the systemic mean arterial pressure and myocardial perfusion.
There is little experience regarding registries and studies on this device. Thiele et al.91 informed on the use of this device in 18 patients with STEMI and SC. The device provided up to 4 L/min of assisted cardiac output. Patients improved their cardiac index and mean arterial pressure, and reduced their pulmonary artery pressure, pulmonary capillary wedge pressure, and central venous pressure with an average 4 days on ventilatory assistance. Kar et al.92 published a series of 117 patients with CS treated with the TandemHeart device that quickly reversed the end-stage hemodynamic compromise seen in patients with STEMI and CS refractory to the IABP and vasopressor support. A randomized clinical trial included 42 patients treated with the IABP (N = 14) or the TandemHeart device (N = 19). The TandemHeart device improved the patients’ hemodynamic parameters significantly even in IABP-refractory patients. However, the mortality rate was similar in both groups.93 To this date, we do not know of any other randomized clinical trials on this technology.
Right atrium-to-systemic arterial circulation assist devices
Extracorporeal membrane oxygenation
VA-ECMO is a cardiopulmonary support system that aspirates blood from the femoral vein or internal jugular vein through a 21-Fr cannula. Through an artificial membrane lung, carbon dioxide is eliminated and oxygen is added to venous blood to later return to the arterial system through a 15-Fr to 22-Fr outflow cannula via the femoral or axillary arteries.93 One of the greatest advantages of ECMO is that it can be implemented everywhere (emergency room, cath lab, etc.) since it is fully portable and does not require fluoroscopic or echocardiographic guidance for a successful implantation. This device provides circulatory support of up to 7 L/min in patients with circulatory and respiratory failure. Some of its limitations are that the VA-ECMO system cannot unload the LV, which can trigger an increased afterload, which is in turn associated with LV distension, worsening of LV function, LV thrombus, and swelling or untreatable alveolar hemorrhage.94 For these reasons it has been proposed that ECMO should be administered with other devices like the IABP and the Impella to reduce pulmonary artery pressures and the dimensions of the LV.95,96 A multicenter, international cohort study included 686 consecutive patients with CS (not due to STEMI exclusively) treated with ECMO. Those patients who underwent LV unloading with the Impella device had a better prognosis and a lower mortality rate, but also higher rates of implantation related bleeding and vascular complications.97 Other authors also recommend procedures like percutaneous balloon atrial septostomy, to allow left-to-right shunting, or the administration of dobutamine to improve contractility and reduce the afterload.94
Aortic regurgitation, aortic dissection, severe peripheral arterial disease, and some ethical considerations are absolute contraindications to ECMO implantation.90 Active bleeding is a relative contraindication because ECMO requires heparin for anticoagulation; however, it has been used in some high-risk patients without heparin since it was the only strategy to save the patient’s life.98 Complications are mainly vascular like lower limb ischemia, compartmental syndrome, major bleeding, stroke, air embolism, and serious infection.90
Yet despite ECMO is widely used in experienced centers, the data supporting its use in patients with acute myocardial infarction complicated with CS are mostly single-center small case-series. Sheu et al. conducted a single-center retrospective observational registry that compared the clinical outcomes of patients with STEMI treated with a primary PCI. The investigators studied 2 different timeframes:1993-2002 for the non-ECMO cohort and 2002-2009 for the ECMO cohort. The study proved that the ECMO assisted PCI improved results at the 30-day follow-up.99 However, interpreting these results is difficult because of the significant discrepancies seen in the treatment strategies used between the groups. In a different study, Muller et al. included 138 STEMI patients treated with ECMO. They developed a mortality risk score in the intensive care unit setting called the ENCOURAGE score. The variables associated with worse prognosis were age > 60 years, female sex, body mass index > 25, Glasgow score < 6, elevated creatinine and lactate serum levels, and prothrombin times < 50%. Survival rates at 6-month and 1-year follow-up were 41% and 38%, respectively.100
Currently, the effects of the use of VA-ECMO on the mortality of patients CS-complicated STEMI is being studied in 3 randomized clinical trials: the EUROSHOCK (NCT03813134), the ANCHOR (NCT04184635), and the ECLS-SHOCK (NCT03637205) clinical trials.101 On top of studying mortality, these clinical trials are an opportunity to analyze the indication, way, and effect of LV unloading.102
CONCLUSION
Despite the improvements made in reperfusion therapies, the mortality of STEMI patients is still high. Together with drug therapy, the rapid restoration of coronary flow and stent implantation are the strategies recommended (figure 1). The routine use of manual thrombus aspiration is discouraged given the lack of clinical benefit compared to the PCI alone. Regarding the type of device selected, second-generation DES are the standard of choice in STEMI patients treated with a primary PCI since the short and long term results are better compared to BMS and first generation DES. In patients with STEMI and MVD, the current evidence recommends complete revascularization, although the optimal time to perform it remains unknown. Exclusively in the case of patients with CS, only the revascularization of the infarct-related artery is advised. Patients with CS-complicated STEMI is undoubtedly the clinical setting with less significant advances. Their mortality rate is still somewhere around 40% to 50%. To this date, several clinical trials are being conducted to assess the impact of circulatory assist devices like the Impella and VA-ECMO on these patients’ mortality rate.
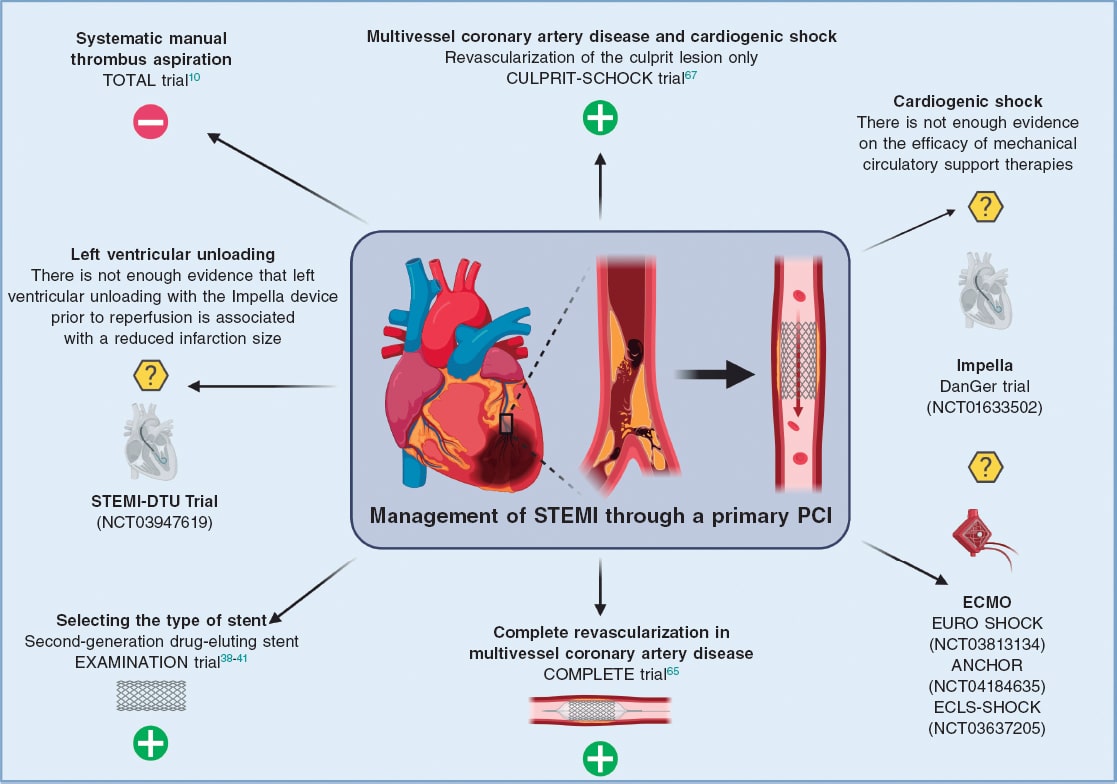
Figure 1. Current evidence and future perspectives of percutaneous coronary interventions for the management of ST-segment elevation myocardial infarction. The green + sign indicates that the procedure is recommended by the European Society of Cardiology clinical practice guidelines; the red minus sign (−) indicates that the procedure is not recommended; the yellow question mark (?) symbol indicates that there is not enough evidence (for or against) to recommend it. ECMO, extracorporeal membrane oxygenation; STEMI, ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention.
FUNDING
None.
AUTHORS’ CONTRIBUTION
L. Ortega-Paz wrote the review draft on the current state of the interventional management of myocardial infarction. S. Brugaletta, and M. Sabaté conducted the critical review of the manuscript with the corresponding changes of content and format.
CONFLICTS OF INTEREST
M. Sabaté is a consultor for Abbott Vascular, and IVascular with no links to this study whatsoever. S. Brugaletta is a consultor for Boston Scientific, and IVascular with no links to this study. L. Ortega-Paz declared no conflicts of interest whatsoever.
REFERENCES
1. Sugiyama T, Hasegawa K, Kobayashi Y, Takahashi O, Fukui T, Tsugawa Y. Differential time trends of outcomes and costs of care for acute myocardial infarction hospitalizations by ST elevation and type of intervention in the United States, 2001-2011. J Am Heart Assoc. 2015;4:e001445.
2. Kristensen SD, Laut KG, Fajadet J, et al. Reperfusion therapy for ST elevation acute myocardial infarction 2010/2011:current status in 37 ESC countries. Eur Heart J. 2014;35:1957-1970.
3. Pedersen F, Butrymovich V, Kelbaek H, et al. Short- and long-term cause of death in patients treated with primary PCI for STEMI. J Am Coll Cardiol. 2014;64:2101-2108.
4. Fokkema ML, James SK, Albertsson P, et al. Population trends in percutaneous coronary intervention:20-year results from the SCAAR (Swedish Coronary Angiography and Angioplasty Registry). J Am Coll Cardiol. 2013;61:1222-1230.
5. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation:The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39:119-177.
6. Montalescot G, Barragan P, Wittenberg O, et al. Platelet glycoprotein IIb/IIIa inhibition with coronary stenting for acute myocardial infarction. N Engl J Med. 2001;344:1895-1903.
7. Sardella G, Mancone M, Bucciarelli-Ducci C, et al. Thrombus aspiration during primary percutaneous coronary intervention improves myocardial reperfusion and reduces infarct size:the EXPIRA (thrombectomy with export catheter in infarct-related artery during primary percutaneous coronary intervention) prospective, randomized trial. J Am Coll Cardiol. 2009;53:309-315.
8. Vlaar PJ, Svilaas T, van der Horst IC, et al. Cardiac death and reinfarction after 1 year in the Thrombus Aspiration during Percutaneous coronary intervention in Acute myocardial infarction Study (TAPAS):a 1-year follow-up study. Lancet. 2008;371:1915-1920.
9. Burzotta F, De Vita M, Gu YL, et al. Clinical impact of thrombectomy in acute ST-elevation myocardial infarction:an individual patient-data pooled analysis of 11 trials. Eur Heart J. 2009;30:2193-2203.
10. Jolly SS, Cairns JA, Yusuf S, et al. Randomized trial of primary PCI with or without routine manual thrombectomy. N Engl J Med. 2015;372:1389-1398.
11. Frobert O, Lagerqvist B, Olivecrona GK, et al. Thrombus aspiration during ST-segment elevation myocardial infarction. N Engl J Med. 2013;369:1587-1597.
12. Jolly SS, Cairns JA, Yusuf S, et al. Stroke in the TOTAL trial:a randomized trial of routine thrombectomy vs. percutaneous coronary intervention alone in ST elevation myocardial infarction. Eur Heart J. 2015;36:2364-2372.
13. Fernandez-Rodriguez D, Regueiro A, Brugaletta S, et al. Optimization in stent implantation by manual thrombus aspiration in ST-segment-elevation myocardial infarction:findings from the EXAMINATION trial. Circ Cardiovasc Interv. 2014;7:294-300.
14. Loubeyre C, Morice MC, Lefevre T, Piechaud JF, Louvard Y, Dumas P. A randomized comparison of direct stenting with conventional stent implantation in selected patients with acute myocardial infarction. J Am Coll Cardiol. 2002;39:15-21.
15. Dudek D, Siudak Z, Janzon M, et al. European registry on patients with ST-elevation myocardial infarction transferred for mechanical reperfusion with a special focus on early administration of abciximab —EUROTRANSFER Registry. Am Heart J. 2008;156:1147-1154.
16. Mockel M, Vollert J, Lansky AJ, et al. Comparison of direct stenting with conventional stent implantation in acute myocardial infarction. Am J Cardiol. 2011;108:1697-1703.
17. Isaaz K, Robin C, Cerisier A, et al. A new approach of primary angioplasty for ST-elevation acute myocardial infarction based on minimalist immediate mechanical intervention. Coron Artery Dis. 2006;17:261-269.
18. Meneveau N, Seronde MF, Descotes-Genon V, et al. Immediate versus delayed angioplasty in infarct-related arteries with TIMI III flow and ST segment recovery:a matched comparison in acute myocardial infarction patients. Clin Res Cardiol. 2009;98:257-264.
19. Pascal J, Veugeois A, Slama M, et al. Delayed Stenting for ST-Elevation Acute Myocardial Infarction in Daily Practice:A Single-Centre Experience. Can J Cardiol. 2016;32:988-995.
20. Carrick D, Oldroyd KG, McEntegart M, et al. A randomized trial of deferred stenting versus immediate stenting to prevent no- or slow-reflow in acute ST-segment elevation myocardial infarction (DEFER-STEMI). J Am Coll Cardiol. 2014;63:2088-2098.
21. Kelbaek H, Hofsten DE, Kober L, et al. Deferred versus conventional stent implantation in patients with ST-segment elevation myocardial infarction (DANAMI 3-DEFER):an open-label, randomised controlled trial. Lancet. 2016;387:2199-2206.
22. Belle L, Motreff P, Mangin L, et al. Comparison of Immediate With Delayed Stenting Using the Minimalist Immediate Mechanical Intervention Approach in Acute ST-Segment-Elevation Myocardial Infarction:The MIMI Study. Circ Cardiovasc Interv. 2016;9:e003388.
23. Kim JS, Lee HJ, Woong Yu C, et al. INNOVATION Study (Impact of Immediate Stent Implantation Versus Deferred Stent Implantation on Infarct Size and Microvascular Perfusion in Patients With ST-Segment-Elevation Myocardial Infarction). Circ Cardiovasc Interv. 2016;9:e004101.
24. Qiao J, Pan L, Zhang B, et al. Deferred Versus Immediate Stenting in Patients With ST-Segment Elevation Myocardial Infarction:A Systematic Review and Meta-Analysis. J Am Heart Assoc. 2017;6:e004838.
25. Spaulding C, Teiger E, Commeau P, et al. Four-year follow-up of TYPHOON (trial to assess the use of the CYPHer sirolimus-eluting coronary stent in acute myocardial infarction treated with BallOON angioplasty). JACC Cardiovasc Interv. 2011;4:14-23.
26. Vink MA, Dirksen MT, Suttorp MJ, et al. 5-year follow-up after primary percutaneous coronary intervention with a paclitaxel-eluting stent versus a bare-metal stent in acute ST-segment elevation myocardial infarction:a follow-up study of the PASSION (Paclitaxel-Eluting Versus Conventional Stent in Myocardial Infarction with ST-Segment Elevation) trial. JACC Cardiovasc Interv. 2011;4:24-29.
27. Violini R, Musto C, De Felice F, et al. Maintenance of long-term clinical benefit with sirolimus-eluting stents in patients with ST-segment elevation myocardial infarction 3-year results of the SESAMI (sirolimus-eluting stent versus bare-metal stent in acute myocardial infarction) trial. J Am Coll Cardiol. 2010;55:810-814.
28. Valgimigli M, Percoco G, Malagutti P, et al. Tirofiban and sirolimus-eluting stent vs abciximab and bare-metal stent for acute myocardial infarction:a randomized trial. JAMA. 2005;293:2109-2117.
29. Valgimigli M, Campo G, Percoco G, et al. Comparison of angioplasty with infusion of tirofiban or abciximab and with implantation of sirolimus-eluting or uncoated stents for acute myocardial infarction:the MULTISTRATEGY randomized trial. JAMA. 2008;299:1788-1799.
30. Di Lorenzo E, De Luca G, Sauro R, et al. The PASEO (PaclitAxel or Sirolimus-Eluting Stent Versus Bare Metal Stent in Primary Angioplasty) Randomized Trial. JACC Cardiovasc Interv. 2009;2:515-523.
31. Stone GW, Lansky AJ, Pocock SJ, et al. Paclitaxel-eluting stents versus bare-metal stents in acute myocardial infarction. N Engl J Med. 2009;360:1946-1959.
32. Kastrati A, Dibra A, Spaulding C, et al. Meta-analysis of randomized trials on drug-eluting stents vs. bare-metal stents in patients with acute myocardial infarction. Eur Heart J. 2007;28:2706-2713.
33. Holmvang L, Kelbaek H, Kaltoft A, et al. Long-term outcome after drug-eluting versus bare-metal stent implantation in patients with ST-segment elevation myocardial infarction:5 years follow-up from the randomized DEDICATION trial (Drug Elution and Distal Protection in Acute Myocardial Infarction). JACC Cardiovasc Interv. 2013;6:548-553.
34. Camenzind E, Steg PG, Wijns W. Stent thrombosis late after implantation of first-generation drug-eluting stents:a cause for concern. Circulation. 2007;115:1440-1455.
35. Joner M, Finn AV, Farb A, et al. Pathology of drug-eluting stents in humans:delayed healing and late thrombotic risk. J Am Coll Cardiol. 2006;48:193-202.
36. Raber L, Kelbaek H, Ostojic M, et al. Effect of biolimus-eluting stents with biodegradable polymer vs bare-metal stents on cardiovascular events among patients with acute myocardial infarction:the COMFORTABLE AMI randomized trial. JAMA. 2012;308:777-787.
37. Raber L, Kelbaek H, Taniwaki M, et al. Biolimus-eluting stents with biodegradable polymer versus bare-metal stents in acute myocardial infarction:two-year clinical results of the COMFORTABLE AMI trial. Circ Cardiovasc Interv. 2014;7:355-364.
38. Sabate M, Cequier A, Iniguez A, et al. Everolimus-eluting stent versus bare-metal stent in ST-segment elevation myocardial infarction (EXAMINATION):1 year results of a randomised controlled trial. Lancet. 2012;380:1482-1490.
39. Sabate M, Brugaletta S, Cequier A, et al. Clinical outcomes in patients with ST-segment elevation myocardial infarction treated with everolimus-eluting stents versus bare-metal stents (EXAMINATION):5-year results of a randomised trial. Lancet. 2016;387:357-366.
40. Sabate M, Raber L, Heg D, et al. Comparison of newer-generation drug-eluting with bare-metal stents in patients with acute ST-segment elevation myocardial infarction:a pooled analysis of the EXAMINATION (clinical Evaluation of the Xience-V stent in Acute Myocardial INfArcTION) and COMFORTABLE-AMI (Comparison of Biolimus Eluted From an Erodible Stent Coating With Bare Metal Stents in Acute ST-Elevation Myocardial Infarction) trials. JACC Cardiovasc Interv. 2014;7:55-63.
41. Brugaletta S, Lara JG, Ortega-Paz L, et al. TCT CONNECT-7 Everolimus-Eluting Stent Versus Bare-Metal Stent in ST-Segment Elevation Myocardial Infarction:10-Year Follow-Up of the Multicenter Randomized Controlled Examination Trial. J Am Coll Cardiol. 2020;76(17 Supplement S):B4-B4.
42. Tousek P, Kocka V, Maly M, et al. Long-term follow-up after bioresorbable vascular scaffold implantation in STEMI patients:PRAGUE-19 study update. EuroIntervention. 2016;12:23-29.
43. Diletti R, Karanasos A, Muramatsu T, et al. Everolimus-eluting bioresorbable vascular scaffolds for treatment of patients presenting with ST-segment elevation myocardial infarction:BVS STEMI first study. Eur Heart J. 2014;35:777-786.
44. Cortese B, Ielasi A, Romagnoli E, et al. Clinical Comparison With Short-Term Follow-Up of Bioresorbable Vascular Scaffold Versus Everolimus-Eluting Stent in Primary Percutaneous Coronary Interventions. Am J Cardiol. 2015;116:705-710.
45. Sabate M, Windecker S, Iniguez A, et al. Everolimus-eluting bioresorbable stent vs. durable polymer everolimus-eluting metallic stent in patients with ST-segment elevation myocardial infarction:results of the randomized ABSORB ST-segment elevation myocardial infarction-TROFI II trial. Eur Heart J. 2016;37:229-240.
46. Brugaletta S, Gori T, Low AF, et al. Absorb bioresorbable vascular scaffold versus everolimus-eluting metallic stent in ST-segment elevation myocardial infarction:1-year results of a propensity score matching comparison:the BVS-EXAMINATION Study (bioresorbable vascular scaffold-a clinical evaluation of everolimus eluting coronary stents in the treatment of patients with ST-segment elevation myocardial infarction). JACC Cardiovasc Interv. 2015;8(1 Pt B):189-197.
47. Brugaletta S, Gori T, Low AF, et al. ABSORB bioresorbable vascular scaffold vs. everolimus-eluting metallic stent in ST-segment elevation myocardial infarction (BVS EXAMINATION study):2-year results from a propensity score matched comparison. Int J Cardiol. 2016;214:483-484.
48. Brugaletta S, Gori T, Tousek P, et al. Bioresorbable vascular scaffolds versus everolimus-eluting metallic stents in patients with ST-segment elevation myocardial infarction:5-year results of the BVS-EXAMINATION study. EuroIntervention. 2020;15:1436-1443.
49. Ielasi A, Campo G, Rapetto C, et al. A Prospective Evaluation of a Pre-Specified Absorb BVS Implantation Strategy in ST-Segment Elevation Myocardial Infarction:The BVS STEMI STRATEGY-IT Study. JACC Cardiovasc Interv. 2017;10:1855-1864.
50. Ielasi A, Campo G, Cortese B, et al. One-Year Results Following a Pre-Specified ABSORB Implantation Strategy in ST-Elevation Myocardial Infarction (BVS STEMI STRATEGY-IT Study). Cardiovasc Revasc Med. 2019;20:700-704.
51. Serruys PW, Chevalier B, Sotomi Y, et al. Comparison of an everolimus-eluting bioresorbable scaffold with an everolimus-eluting metallic stent for the treatment of coronary artery stenosis (ABSORB II):a 3 year, randomised, controlled, single-blind, multicentre clinical trial. Lancet. 2016;388:2479-2491.
52. Wykrzykowska JJ, Kraak RP, Hofma SH, et al. Bioresorbable Scaffolds versus Metallic Stents in Routine PCI. N Engl J Med. 2017;376:2319-2328.
53. Kereiakes DJ, Ellis SG, Metzger C, et al. 3-Year Clinical Outcomes With Everolimus-Eluting Bioresorbable Coronary Scaffolds:The ABSORB III Trial. J Am Coll Cardiol. 2017;70:2852-2862.
54. Haude M, Ince H, Abizaid A, et al. Sustained safety and performance of the second-generation drug-eluting absorbable metal scaffold in patients with de novo coronary lesions:12-month clinical results and angiographic findings of the BIOSOLVE-II first-in-man trial. Eur Heart J. 2016;37:2701-2709.
55. Brugaletta S, Cequier A, Alfonso F, et al. MAGnesium-based bioresorbable scaffold and vasomotor function in patients with acute ST segment elevation myocardial infarction:The MAGSTEMI trial:Rationale and design. Catheter Cardiovasc Interv. 2019;93:64-70.
56. Sabate M, Alfonso F, Cequier A, et al. Magnesium-Based Resorbable Scaffold Versus Permanent Metallic Sirolimus-Eluting Stent in Patients With ST-Segment Elevation Myocardial Infarction:The MAGSTEMI Randomized Clinical Trial. Circulation. 2019;140:1904-1916.
57. Ortega-Paz L, Brugaletta S, Gomez-Lara J, et al. Target lesion revascularisation of bioresorbable metal scaffolds:a case series study and literature review. EuroIntervention. 2021;16:1100-1103.
58. Gomez-Lara J, Ortega-Paz L, Brugaletta S, et al. Bioresorbable scaffolds versus permanent sirolimus-eluting stents in patients with ST-segment elevation myocardial infarction:vascular healing outcomes from the MAGSTEMI trial. EuroIntervention. 2020;16:e913-e921.
59. Ortega-Paz L, Brugaletta S, Sabate M. Very-late restenosis of a magnesium-based resorbable scaffold. Eur Heart J. 2020;41:2602.
60. Dziewierz A, Siudak Z, Rakowski T, Zasada W, Dubiel JS, Dudek D. Impact of multivessel coronary artery disease and noninfarct-related artery revascularization on outcome of patients with ST-elevation myocardial infarction transferred for primary percutaneous coronary intervention (from the EUROTRANSFER Registry). Am J Cardiol. 2010;106:342-347.
61. Wald DS, Morris JK, Wald NJ, et al. Randomized trial of preventive angioplasty in myocardial infarction. N Engl J Med. 2013;369:1115-1123.
62. Gershlick AH, Khan JN, Kelly DJ, et al. Randomized trial of complete versus lesion-only revascularization in patients undergoing primary percutaneous coronary intervention for STEMI and multivessel disease:the CvLPRIT trial. J Am Coll Cardiol. 2015;65:963-972.
63. Engstrom T, Kelbaek H, Helqvist S, et al. Complete revascularisation versus treatment of the culprit lesion only in patients with ST-segment elevation myocardial infarction and multivessel disease (DANAMI-3-PRIMULTI):an open-label, randomised controlled trial. Lancet. 2015;386:665-671.
64. Smits PC, Abdel-Wahab M, Neumann FJ, et al. Fractional Flow Reserve-Guided Multivessel Angioplasty in Myocardial Infarction. N Engl J Med. 2017;376:1234-1244.
65. Mehta SR, Wood DA, Storey RF, et al. Complete Revascularization with Multivessel PCI for Myocardial Infarction. N Engl J Med. 2019;381:1411-1421.
66. den Dekker WK, Van Mieghem NM, Bennett J, et al. Percutaneous complete revascularization strategies using sirolimus-eluting biodegradable polymer-coated stents in patients presenting with acute coronary syndrome and multivessel disease:Rationale and design of the BIOVASC trial. Am Heart J. 2020;227:111-117.
67. Thiele H, Akin I, Sandri M, et al. PCI Strategies in Patients with Acute Myocardial Infarction and Cardiogenic Shock. N Engl J Med. 2017;377:2419-2432.
68. Thiele H, Akin I, Sandri M, et al. One-Year Outcomes after PCI Strategies in Cardiogenic Shock. N Engl J Med. 2018;379:1699-1710.
69. Ibanez B, Halvorsen S, Roffi M, et al. Integrating the results of the CULPRIT-SHOCK trial in the 2017 ESC ST-elevation myocardial infarction guidelines:viewpoint of the task force. Eur Heart J. 2018;39:4239-4242.
70. Kolte D, Khera S, Aronow WS, et al. Trends in incidence, management, and outcomes of cardiogenic shock complicating ST-elevation myocardial infarction in the United States. J Am Heart Assoc. 2014;3:e000590.
71. Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure:The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129-2200.
72. Atkinson TM, Ohman EM, O'Neill WW, Rab T, Cigarroa JE, Interventional Scientific Council of the American College of Cardiology. A Practical Approach to Mechanical Circulatory Support in Patients Undergoing Percutaneous Coronary Intervention:An Interventional Perspective. JACC Cardiovasc Interv. 2016;9:871-883.
73. Helgestad OKL, Josiassen J, Hassager C, et al. Contemporary trends in use of mechanical circulatory support in patients with acute MI and cardiogenic shock. Open Heart. 2020;7:e001214.
74. Waksman R, Weiss AT, Gotsman MS, Hasin Y. Intra-aortic balloon counterpulsation improves survival in cardiogenic shock complicating acute myocardial infarction. Eur Heart J. 1993;14:71-74.
75. Bengtson JR, Kaplan AJ, Pieper KS, et al. Prognosis in cardiogenic shock after acute myocardial infarction in the interventional era. J Am Coll Cardiol. 1992;20:1482-1489.
76. Sjauw KD, Engstrom AE, Vis MM, et al. A systematic review and meta-analysis of intra-aortic balloon pump therapy in ST-elevation myocardial infarction:should we change the guidelines?Eur Heart J. 2009;30:459-468.
77. Patel MR, Smalling RW, Thiele H, et al. Intra-aortic balloon counterpulsation and infarct size in patients with acute anterior myocardial infarction without shock:the CRISP AMI randomized trial. JAMA. 2011;306:1329-1337.
78. Ohman EM, Nanas J, Stomel RJ, et al. Thrombolysis and counterpulsation to improve survival in myocardial infarction complicated by hypotension and suspected cardiogenic shock or heart failure:results of the TACTICS Trial. J Thromb Thrombolysis. 2005;19:33-39.
79. Prondzinsky R, Lemm H, Swyter M, et al. Intra-aortic balloon counterpulsation in patients with acute myocardial infarction complicated by cardiogenic shock:the prospective, randomized IABP SHOCK Trial for attenuation of multiorgan dysfunction syndrome. Crit Care Med. 2010;38:152-160.
80. Thiele H, Zeymer U, Neumann FJ, et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med. 2012;367:1287-1296.
81. Thiele H, Zeymer U, Neumann FJ, et al. Intra-aortic balloon counterpulsation in acute myocardial infarction complicated by cardiogenic shock (IABP-SHOCK II):final 12 month results of a randomised, open-label trial. Lancet. 2013;382:1638-1645.
82. Iannaccone M, Albani S, Giannini F, et al. Short term outcomes of Impella in cardiogenic shock:A review and meta-analysis of observational studies. Int J Cardiol.2021;324:44-51.
83. Sjauw KD, Konorza T, Erbel R, et al. Supported high-risk percutaneous coronary intervention with the Impella 2.5 device the Europella registry. J Am Coll Cardiol. 2009;54:2430-2434.
84. Maini B, Naidu SS, Mulukutla S, et al. Real-world use of the Impella 2.5 circulatory support system in complex high-risk percutaneous coronary intervention:the USpella Registry. Catheter Cardiovasc Interv. 2012;80:717-725.
85. Seyfarth M, Sibbing D, Bauer I, et al. A randomized clinical trial to evaluate the safety and efficacy of a percutaneous left ventricular assist device versus intra-aortic balloon pumping for treatment of cardiogenic shock caused by myocardial infarction. J Am Coll Cardiol. 2008;52:1584-1588.
86. Ouweneel DM, Eriksen E, Sjauw KD, et al. Percutaneous Mechanical Circulatory Support Versus Intra-Aortic Balloon Pump in Cardiogenic Shock After Acute Myocardial Infarction. J Am Coll Cardiol. 2017;69:278-287.
87. Udesen NJ, Moller JE, Lindholm MG, et al. Rationale and design of DanGer shock:Danish-German cardiogenic shock trial. Am Heart J. 2019;214:60-68.
88. Watanabe S, Fish K, Kovacic JC, et al. Left Ventricular Unloading Using an Impella CP Improves Coronary Flow and Infarct Zone Perfusion in Ischemic Heart Failure. J Am Heart Assoc. 2018;7:e006462.
89. Kapur NK, Alkhouli MA, DeMartini TJ, et al. Unloading the Left Ventricle Before Reperfusion in Patients With Anterior ST-Segment-Elevation Myocardial Infarction. Circulation. 2019;139:337-346.
90. Combes A, Price S, Slutsky AS, Brodie D. Temporary circulatory support for cardiogenic shock. Lancet. 2020;396:199-212.
91. Thiele H, Lauer B, Hambrecht R, Boudriot E, Cohen HA, Schuler G. Reversal of cardiogenic shock by percutaneous left atrial-to-femoral arterial bypass assistance. Circulation. 2001;104:2917-2922.
92. Kar B, Gregoric ID, Basra SS, Idelchik GM, Loyalka P. The percutaneous ventricular assist device in severe refractory cardiogenic shock. J Am Coll Cardiol. 2011;57:688-696.
93. Burkhoff D, Cohen H, Brunckhorst C, O'Neill WW, TandemHeart Investigators Group. A randomized multicenter clinical study to evaluate the safety and efficacy of the TandemHeart percutaneous ventricular assist device versus conventional therapy with intraaortic balloon pumping for treatment of cardiogenic shock. Am Heart J. 2006;152:469e1-8.
94. Burkhoff D, Sayer G, Doshi D, Uriel N. Hemodynamics of Mechanical Circulatory Support. J Am Coll Cardiol. 2015;66:2663-2674.
95. Petroni T, Harrois A, Amour J, et al. Intra-aortic balloon pump effects on macrocirculation and microcirculation in cardiogenic shock patients supported by venoarterial extracorporeal membrane oxygenation. Crit Care Med. 2014;42:2075-2082.
96. Cheng A, Swartz MF, Massey HT. Impella to unload the left ventricle during peripheral extracorporeal membrane oxygenation. ASAIO J. 2013;59:533-536.
97. Schrage B, Becher PM, Bernhardt A, et al. Left Ventricular Unloading Is Associated With Lower Mortality in Patients With Cardiogenic Shock Treated With Venoarterial Extracorporeal Membrane Oxygenation:Results From an International, Multicenter Cohort Study. Circulation. 2020;142:2095-2106.
98. Abrams D, Agerstrand CL, Biscotti M, Burkart KM, Bacchetta M, Brodie D. Extracorporeal membrane oxygenation in the management of diffuse alveolar hemorrhage. ASAIO J. 2015;61:216-218.
99. Sheu JJ, Tsai TH, Lee FY, et al. Early extracorporeal membrane oxygenator-assisted primary percutaneous coronary intervention improved 30-day clinical outcomes in patients with ST-segment elevation myocardial infarction complicated with profound cardiogenic shock. Crit Care Med. 2010;38:1810-1817.
100. Muller G, Flecher E, Lebreton G, et al. The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock. Intensive Care Med. 2016;42:370-378.
101. Banning AS, Adriaenssens T, Berry C, et al. The EURO SHOCK Trial:Design, Aims and Objectives. EuroIntervention. 2020;EIJ-D-20-01076.
102. Lusebrink E, Orban M, Kupka D, et al. Prevention and treatment of pulmonary congestion in patients undergoing venoarterial extracorporeal membrane oxygenation for cardiogenic shock. Eur Heart J. 2020;41:3753-3761.