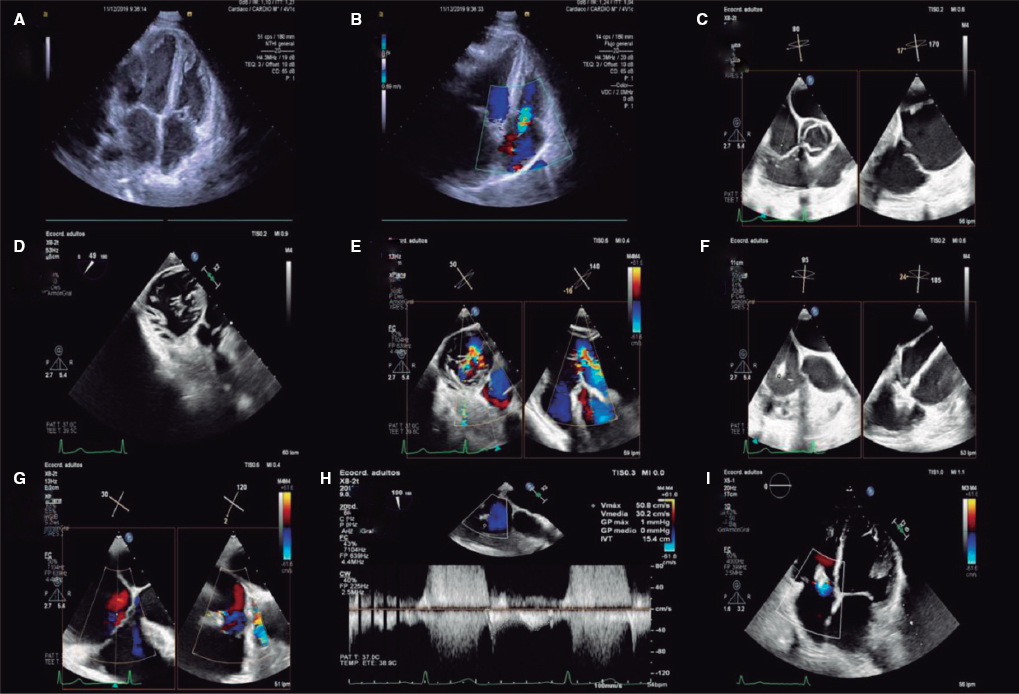
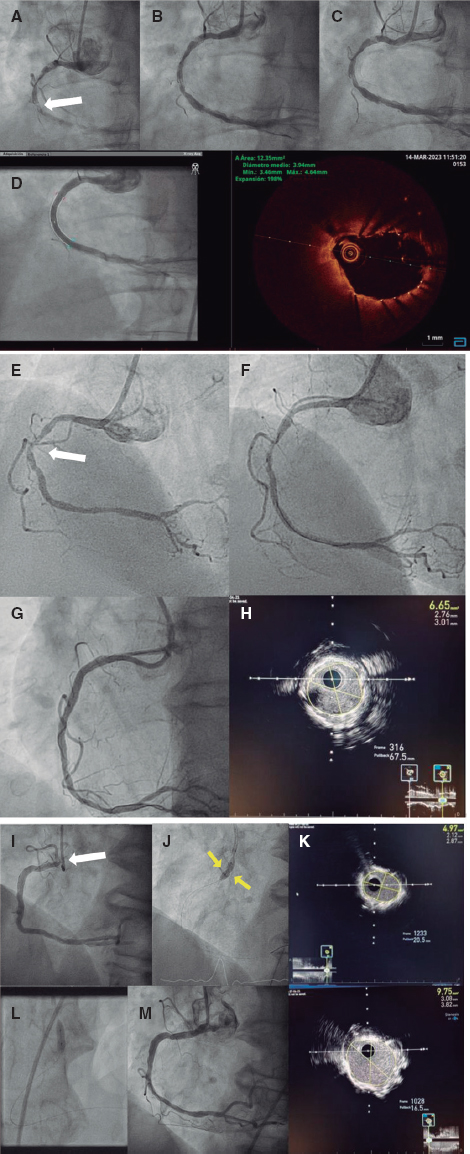
QUESTION: Briefly, what is the evidence behind the percutaneous left atrial appendage closure (LAAC) regarding oral anticoagulation (OA)? Is there any evidence on direct-acting oral anticoagulants (DAOAs)?
ANSWER: The main clinical evidence published on this regard comes from 3 randomized clinical trials (2 vs anti-vitamin K and 1 vs DAOAs), registries, and case series. The PROTECT AF clinical trial1 included 707 patients with atrial fibrillation (AF) and CHADS2 scores ≥ 1 who were randomized on a 2:1 ratio to receive the Watchman device (Boston Scientific, United States) or warfarin. The composite endpoint (stroke, cardiovascular death, and systemic embolism) was less prevalent in patients with the Watchman device (relative risk [RR], 0.62; 95% confidence interval [95%CI], 0.35-1.25); however, adverse events were more common in the device group (RR, 1.69; 95%CI, 1.01-3.19) mainly due to periprocedural complications. These increased adverse events triggered a second clinical trial, the PREVAIL,2 that included 407 patients with CHADS ≥ 2 randomized on a 2:1 ratio to receive the Watchman device or warfarin. The primary efficacy endpoint at the 18-month follow-up (stroke, systemic embolism or cardiovascular or inexplicable death) occurred less often than expected in the warfarin group and the non-inferiority target was not achieved. However, the secondary efficacy endpoint (stroke or systemic embolism 7 days after implantation) was achieved; also, adverse events were less common compared to those of the PROTECT AF trial.
Data from the PROTECT AF1 and PREVAIL2 clinical trials were combined in a meta-analys3 that concluded that the Watchman device was not inferior to anti-vitamin K therapy for the composite endpoint of stroke, cardiovascular death, and systemic embolism. We should mention that patients with the Watchman device had lower chances of bleeding including hemorrhagic strokes. Nonetheless, there was a statistically insignificant higher rate of ischemic stroke and systemic embolism in the Watchman group.
The controversial aspect of these 2 clinical trials is that they only included patients with an indication for OA. However, in the routine clinical practice, the LAAC is used in patients contraindicated to OA.
The third and last clinical trial is the PRAGUE-174 that compared the LAAC to DAOAs. It included 402 patients with AF (CHA2DS2-VASc: 4.7 ± 1.5) and a past medical history of major bleeding or cardioembolic event while on DAOAs or with o con CHA2DS2-VASc ≥ 3 and HAS-BLED > 2. Apixaban was the most commonly used DAOA (95.5%). The median follow-up was 19.9 months. No statistically significant differences were found in the annual rates of the primary endpoint (stroke, transient ischemic attack, systemic embolism, cardiovascular death, clinically relevant or major bleeding or procedural complications) that were 10.99% with the LAAC and 13.42% with DAOAs (sub-hazard ratio, 0.84; 95%CI, 0.53-1.31 for non-inferiority). A total of 9 patients (4.5%) experienced major complications due to device implantation.
Regarding real-world registries, the NCDR LAAO5 is a large registry designed to assess the utility, safety, and effectiveness of LAAC percutaneous devices in the clinical practice. It includes 38 158 procedures performed in the United States. The mean score in the CHADS-VASC scale was 4.6 ± 1.5, and in the HAS-BLED scale, 3.0 ± 1.1. In-hospital major adverse events occurred in 2.16% of the patients; the most common complications were pericardial effusion that required intervention (1.39%), and major bleeding (1.25%), while strokes (0.17%) and death (0.19%) were rare.
Q.: Which are the current indications for the LAAC? Should the clinical guidelines most recently published be changed somehow?
A.: The most common indications in the routine clinical practice are high bleeding risk and contraindications to DAOAs (eg, dialysis), but as I said, the LAAC has not been studied in clinical trials with the proper statistical power in these subgroups
The recent guidelines published by the European Society of Cardiology (ESC)6 give the LAAC a class IIb recommendation and a B level of evidence for the prevention of strokes in patients with AF and contraindications to long-term anticoagulant therapy (eg, intracranial hemorrhage without reversible cause). The clinical guidelines published by the American Heart Association, American College of Cardiology, and the Heart Rhythm Society (AHA/ACC/HRS)7 also give it a class IIb recommendations as a therapeutic option for patients with an indication for oral anticoagulation, but with a high risk of bleeding, poor compliance or tolerance to anticoagulant therapy. In both guidelines, recommendations are limited. The consensus document published by the European Heart Rhythm Association (EHRA)8 regarding the LAAC makes an extensive review including the largest possible number of indications:
1)Patients with contraindications to OA due to:
a) High risk of potentially life-threatening or incapacitating hemorrhage or due to untreatable causes like intracranial/intraspinal bleeding (eg, diffuse amyloid angiopathy or untreatable vascular malformation) or severe gastrointestinal (eg, diffuse angiodysplasia) pulmonary or urogenital bleeding that cannot be corrected.
b) Serious adverse events with anti-vitamin K therapy or contraindications to DAOAs.
2) Patients who don’t follow their anticoagulant therapy properly. This indication is controversial, and we should always inform the patient that his main treatment is anticoagulation while trying to solve the problem associated with poor compliance.
3)Some specific subgroups:
a) Ineffective OA: stroke in a patient properly anticoagulated.
b) After pulmonary vein ablation with left atrial appendage electrical isolation due to the high embolic risk involved after this procedure.
c) Combination of pulmonary vein ablation and LAAC within the same procedure.
d) «Primary» prevention: in patients with interatrial communication treated with percutaneous closure during the same procedure even before the patient develops AF.
Q.: Which is the common practice at your center?
A.: The most common indications are patients with AF on hemodialysis who, we already know, are poor candidates to anti-vitamin K therapy and have contraindications to DAOAs. Also, patients with very high risk of bleeding without treatable cause, especially digestive or due to other causes (eg, Rendu-Osler-Weber disease) and always after trying to solve it with a DAOA.
Q.: How valuable are imaging modalities in the selection of patients and procedural guidance?
A.: The use of imaging modalities is essential if we wish to perform successful procedures. Before the intervention, the anatomy of the left atrial appendage should be studied to see if it is eligible for closure. Also, the right material should be chosen and the presence of thrombi in the left atrium and left atrial appendage should be discarded. In general, this preprocedural assessment is performed with a transesophageal echocardiography, yet the computed tomography scan has been gaining traction.
During the procedure, x-ray images and transesophageal echocardiography are the common imaging modalities to use. When available, intracardiac echocardiography can also be used.
Between 6 and 24 weeks after the intervention, a transesophageal echocardiography or a computed tomography scan should be performed to discard the presence of thrombi in the device (that can appear in up to 2% to 4% of the cases) and significant peridevice leaks (> 5 mm).8
Q.: Are there any significant differences among the different devices available?
A.: The need for antithrombotic therapy post-LAAC shows that there is room for improvement because this procedure maintains the bleeding risk for a longer period of time, and it is still unclear what the optimal protocol should be. The PROTECT AF and the PREVAIL clinical trials kept patients on warfarin and acetylsalicylic acid for 45 days followed by a 6-month course of dual antiplatelet therapy and then acetylsalicylic acid for life.
The guidelines published by the ESC6 recommend acetylsalicylic acid permanently, clopidogrel between 1 and 6 months after the procedure and, for the Watchman device, in cases of low-risk of bleeding, a 45-day course of OA after implantation.
The consensus document published by the EHRA8 includes general recommendations that are very similar to those from the guidelines published by the ESC.6 It also claims that in patients with very high-risk of bleeding the use of single antiplatelet therapy could be an option (acetylsalicylic acid or clopidogrel) for short periods of time. However, this should always be an informed decision and the patient should think about it thoroughly.
Q.: Please tell us about any relevant ongoing clinical trials. In your professional opinion, what type of study should be conducted?
A.: The main ongoing trials are the ASAP-TOO that is assessing the LAAC in 888 patients with AF considered ineligible for OA; the OPTION trial, that is studying 1600 patients with AF to see if the LAAC with the Watchman FLX device is a reasonable option to OA (including DAOAs) after pulmonary vein ablation; and the CATALYST and CHAMPION-AF clinical trials, that will be conducting a long-term comparison between the Amulet (Abbott Vascular, United States) and the Watchman FLX device with DAOAs in patients with an indication for anticoagulant therapy due to AF.
Future clinical trials should prioritize the study of new devices comparing them to DAOAs since they are currently the therapy of choice to prevent strokes in patients with AF, especially those with relative or absolute contraindications to anticoagulation therapy. The use of these devices in patients with ischemic stroke despite the proper anticoagulation therapy should be analyzed too by associating the LAAC with an DAOA and then comparing it to an isolated DAOA. Finally, it is essential to clarify antithrombotic therapy after implantation in order to minimize it, above all, in patients with high risk of bleeding, while still keeping its efficacy.
FUNDING
No funding was received for this work.
CONFLICTS OF INTEREST
None.
REFERENCES
1. Holmes DR, Reddy VY, Turi ZG, et al. PROTECT AF Investigators. Percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation:a randomised non-inferiority trial. Lancet. 2009;374:534-542.
2. Holmes DR Jr, Kar S, Price MJ, et al. Prospective randomized evaluation of the WATCHMAN Left Atrial Appendage Closure device in patients with atrial fibrillation versus long-term warfarin therapy:the PREVAIL trial. J Am Coll Cardiol. 2014;64:1-12.
3. Reddy VY, Doshi SK, Kar S, et al.;PREVAIL and PROTECT AF Investigators. 5-Year Outcomes After Left Atrial Appendage Closure. From the PREVAIL and PROTECT AF Trials. J Am Coll Cardiol. 2017;70:2964-2975.
4. Osmancik P, Herman D, Neuzil P, et al. Left atrial appendage closure versus direct oral anticoagulants in high-risk patients with atrial fibrillation. J Am Coll Cardiol. 2020;75:3122-3135.
5. Freeman JV, Varosy P, Price MJ, et al. The NCDR Left Atrial Appendage Occlusion Registry. J Am Coll Cardiol. 2020;75:1503-1518.
6. Hindricks G, Potpara T, Dagres N, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2020;42:373-498.
7. January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation. Circulation. 2019;140:e125-e151.
8. Glikson M, Wolff R, Hindricks G, et al. ESC Scientific Document Group, EHRA/EAPCI expert consensus statement on catheter-based left atrial appendage occlusion –an update. Europace. 2020;22:184.
* Corresponding author: Servicio de Cardiología, Hospital Arnau de Vilanova, San Clemente 12, 46015 Valencia, Spain.
E-mail address: jcosinsales@gmail.com (J. Cosín-Sales).