To the Editor,
Despite the expansion of transcatheter aortic valve implantation (TAVI) to lower-risk patients, the length of stay after TAVI remains high for an average 8 days according to the Spanish TAVI registry.1 Recently published studies have demonstrated that early discharge following balloon-expandable transfemoral TAVI is feasible and safe.2,3 The unprecedented demand sustained by the healthcare services during the current COVID-19 pandemic has led to redirecting assets and restricting many cardiovascular procedures to protect the limited resources available like anesthesia support, ventilators, and critical care infrastructures. The present fast-track protocol was developed in response to the COVID-19 pandemic to assess the safety and feasibility of early discharge after minimalist TAVI with either balloon-expandable or self-expanding valves in our setting.
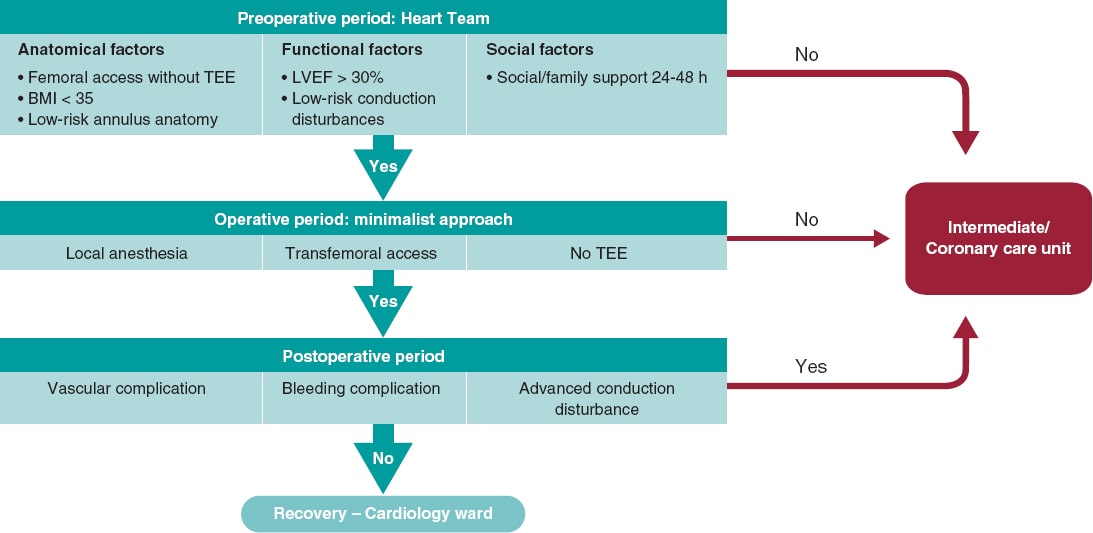
Patients undergoing transfemoral TAVI were prospectively recruited. The inclusion criteria in the fast-track pathway were based on the 3M TAVI study: femoral access eligible for percutaneous closure, body mass index < 35, low-risk aortic annulus anatomy (coronary height > 10 mm, tricuspid valve, non-severe left ventricular outflow tract calcification), ejection fraction ≥ 30%, low anticipated risk of advanced conduction disturbances (PR interval < 240 ms, absence of right bundle branch block), familial support within the first 24-48 hours. All procedures were performed under local anaesthesia and conscious sedation. Pre-closure of the access site was performed using the double ProGlide technique (Abbott Vascular, United States), and in general patients received unfractionated heparin (70 U/kg) without a reversal agent. The implantation technique included a coplanar view of the balloon-expandable valves and a cusp overlap view of the self-expanding valves. In the absence of procedural complications (major vascular complications, major bleeding or high-degree atrioventricular block), and after 2 hours at the cath lab recovery room, patients were transferred to the cardiology ward under cardiac telemetry monitoring (figure 1). Conduction disturbances were managed as described in the 2019 expert consensus document.4 The study was approved by Hospital de la Santa Creu i Sant Pau local ethics committee, and patients gave their informed consent to the procedures.
Figure 1. Fast-track TAVI pathway. BMI, body mass index; LVEF, left ventricular ejection fraction; TEE, transesophageal echocardiography.
A total of 38 out of the 93 patients (40.9%) treated with TAVI at our centre between October 2020 through July 2021 met the fast-track inclusion criteria. The mean age was 84 years (63.2%, women) with a mean STS-score of 3.7%. One third of the patients (29.0%) had atrial fibrillation, and chronic obstructive pulmonary disease while 16 patients (42.1%) had coronary artery disease. Further baseline characteristics are shown on table 1.
Table 1. Baseline, in-hospital, and 30-day outcomes
| Baseline characteristics | N = 38 |
|---|---|
| Age, years | 84.4 ± 3.9 |
| Female | 24 (63.2) |
| Diabetes mellitus | 9 (23.7) |
| Hypertension | 31 (81.6) |
| Atrial fibrillation | 11 (29.0) |
| COPD | 11 (29.0) |
| Previous coronary artery disease | 16 (42.1) |
| Previous stroke | 4 (10.5) |
| LVEF, % | 59.1 ± 11.9 |
| Aortic valve gradient, mean, mmHg | 48.1 ± 10.3 |
| Aortic valve area, cm2 | 0.7 ± 0.2 |
| STS-PROM, % | 3.7 ± 1.4 |
| EuroSCORE II, % | 3.7 ± 2.2 |
| Procedural characteristics | |
| Transfemoral approach | 38 (100) |
| Local anesthesia | 38 (100) |
| Transradial secondary access | 38 (100) |
| Crossover technique | 6 (15.8) |
| Pacing over guidewire | 37 (97.4) |
| Prosthesis type | |
| SAPIEN 3/Ultra | 23 (60.5) |
| Portico | 13 (34.2) |
| Evolut R/Pro | 2 (5.3) |
| Procedural success | 38 (100) |
| Valve embolization | 0 (0) |
| Coronary occlusion | 0 (0) |
| Conversion to surgery | 0 (0) |
| Procedural death | 0 (0) |
| In-hospital, and 30-day outcomes | |
| Length of stay | 2 (2-3) |
| < 24 h | 6 (15.8) |
| < 48 h | 23 (60.5) |
| < 72 h | 30 (78.9) |
| Intensive/intermediate care unit | 3 (7.9) |
| Mortality | 0 (0) |
| Stroke/TIA | 1 (2.6) |
| Major vascular complication | 1 (2.6) |
| Major bleeding | 1 (2.6) |
| Permanent pacemaker implantation | 5 (13.2) |
| Readmission at 30 days | 2 (5.3) |
| Pacemaker implantation after discharge | 0 (0) |
|
COPD, chronic obstructive pulmonary disease; LVEF, left ventricular ejection fraction; STS-PROM, Society of Thoracic Surgeons Predicted Risk of Mortality; TIA, transient ischemic attack. Data are expressed as no. (%), mean ± standard deviation or median [interquartile range]. |
|
The minimalist approach was suggested in most patients: ultrasound-guided percutaneous primary access (100%), transradial secondary access (100%) while the simplified crossover technique via radial artery was required in 6 patients (15.8%) only (upfront guidewire protection was used in 4 obese patients, bailout balloon inflation in 2 patients to achieve femoral hemostasis). Also, pacing over stiff guidewire was used in all but 1 patient (with incomplete right bundle branch block). A balloon-expandable valve was used in 23 patients (60.5%) and a self-expanding valve in 15 patients (39.5%). Procedural success was achieved in all patients. There were no cases of valve embolization, coronary occlusion, annulus rupture or procedural death. A total of 3 patients (7.9%) were transferred to the intermediate or coronary care unit following vascular complications (n = 1) or high-degree atrioventricular block (n = 2). Next-day discharge was achieved in 6 patients (15.8%), and in nearly 80% within 72 hours for a median length of stay of 2 days (2-3). One patient experienced 2 adverse events (major bleeding and vascular failure that required covered stent graft implantation). A permanent pacemaker (PPM) was implanted in 5 patients (13.2%, 3 after balloon-expandable valve implantation, 2 after self-expanding valve implantation). Thirty-day readmission occurred in 2 patients (5.3%) with heart failure (n = 1) and transient ischemic attack (n = 1), the latter patient being discharged from the emergency room on the same day. No cases of mortality or late heart block requiring PPM after hospital discharge were reported. With the experience gained, the rate of patients discharged within the first 24 hours increased from 9% within the first trimester to 33% during the third.
Our study main findings are: a) a pre-defined accelerated pathway after minimalist TAVI using either balloon-expandable or self-expanding valves was safe and feasible with a high rate of procedural success, and a low rate of adverse events; b) direct transfer to the cardiology ward was achieved in most patients (92%); c) it enabled early discharges within the first 72 hours in 80% of patients and next-day discharges in nearly a fifth of the patients.
To our knowledge this study is the first experience of an accelerated pathway after TAVI in our country. Its objective was to pursue the TAVI program during the pandemic without collapsing the limited resources available. In recent years, 2 studies have shown the safety of early discharge after TAVI in selected patients who received a balloon-expandable transcatheter aortic heart valve. Of note, nearly 40% of the patients received a self-expanding valve in our study. In the 3M TAVI study that included 55% of the screened patients, 89% of the patients were discharged within 48 hours with a good safety and efficacy profile (mortality, stroke, major vascular complication, PPM implantation, and readmission 30-day rates of 1.5%, 1.5%, 2.4%, 5.7%, and 9.2%, respectively).2 Similarly, in the FAST-TAVI trial, most patients (72%) were early discharged within 72 hours with low mortality (1.1%), stroke (1.7%), PPM implantation (7.3%), and rehospitalization (3.7%) 30-day rates.3 Despite the limited size of the sample, our results are consistent with both studies with a remarkable 0% mortality rate at 30 days, but with a higher rate of PPM implantation, which may be explained by the inclusion of self-expanding valves in the present study.
While the volume of TAVI will continue to grow, there is an urgent need to optimize the patient care pathway and reduce the length of stay. The widely used minimalist approach plus the challenges posed by the COVID-19 pandemic pave the way for more efficient clinical pathways that guarantee safe early discharges using minimal resources and critical care infrastructure.
FUNDING
This work was supported by an institutional research grant from Edwards, and Abbott.
AUTHORS’ CONTRIBUTIONS
All authors participated in the collection of data, analysis, drafting, and review of the manuscript.
CONFLICTS OF INTERESTS
D. Arzamendi has received proctoring, and speaker fees from Abbott and Edwards. L. Asmarats is a proctor for Abbott, and received speaking fees from Edwards. X. Millán received proctoring and speaking fees from Abbott. The remaining authors have no relationships to disclose relevant to the content of this paper.
REFERENCES
1. Jiménez-Quevedo P, Muñoz-García A, Trillo-Nouche R, et al. Time trend in transcatheter aortic valve implantation:an analysis of the Spanish TAVI registry. REC Interv Cardiol. 2020;2:98-105.
2. Wood DA, Lauck SB, Cairns JA, et al. The Vancouver 3M (Multidisciplinary, Multimodality, But Minimalist) Clinical Pathway Facilitates Safe Next-Day Discharge Home at Low-, Medium-, and High-Volume Transfemoral Transcatheter Aortic Valve Replacement Centers:The 3M TAVR Study. JACC Cardiovasc Interv. 2019;12:459-469.
3. Barbanti M, van Mourik MS, Spence MS, et al. Optimising patient discharge management after transfemoral transcatheter aortic valve implantation:the multicentre European FAST-TAVI trial. EuroIntervention. 2019;15:147-154.
4. Rodes-Cabau J, Ellenbogen KA, Krahn AD, et al. Management of Conduction Disturbances Associated With Transcatheter Aortic Valve Replacement:JACC Scientific Expert Panel. J Am Coll Cardiol. 2019;74:1086-1106.