To the Editor,
Surgery of aortic arch diseases like aneurysms and dissections require invasive procedures that include cardiopulmonary bypass, selective cerebral perfusion, aortic cross-clamping, and hypothermic circulatory arrest.1 Unfortunately, a large number of these patients are considered non-eligible for open surgical repair and receive medical therapy only.
The NEXUS Stent Graft System (Endospan Ltd., Israel) is indicated to treat high-risk surgical patients with ascending aorta and aortic arch diseases.2 This device received European CE mark approval back in March 2019 for the endovascular repair of aortic arch disease including both aneurysms and dissections requiring landing in the zone 0. The NEXUS Stent Graft System is a dual module device that is inserted through a 20-Fr delivery system. The integrated brachiocephalic artery branch avoids the possibility of branch separation and the interlocking latch mechanism avoids the migration or separation of the modules. This article describes the implantation of a NEXUS branched aortic stent graft system in Spain for the first time. Patient's written informed consent for publication was obtained.
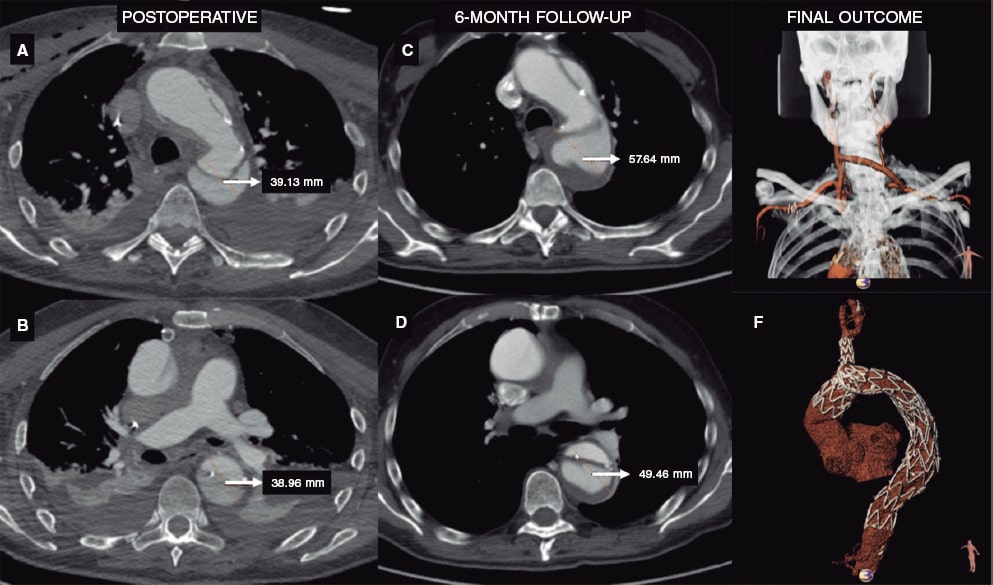
This is the case of a 75-years-old male patient with an enlarged downstream aorta 6 months after ascending aortic replacement with open distal anastomosis due to acute type A aortic dissection. The follow-up coronary computed tomography angiography performed revealed the presence of an enlarged aortic arch and descending aorta (from 4.1 mm to 5.2 mm). The patient was considered non-eligible for open surgical repair by the heart team and a decision was made to proceed with the endovascular management of the diseased segment of the aorta.
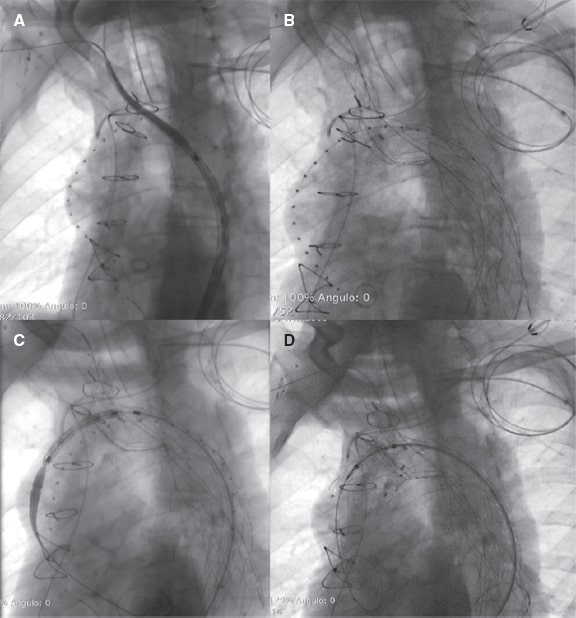
The patient underwent extra-anatomic reconstruction with right common carotid artery to left subclavian artery bypass (both end-to-side) and proximal ligation with an 8-mm polytetrafluoroethylene vascular graft and end-to-side reimplantation of the right common carotid artery on the graft. Six weeks later, the endovascular procedure was performed. The day prior to the procedure, cardiac pacing was used to induce hypotension during stent graft deployment. Under general anesthesia, the NEXUS Stent Graft System was inserted via right groin towards the ascending aorta. Intravenous heparin was infused (5000 IU) during the preparation prior to the insertion of the device and on the introduction of the NEXUS device main body module. More heparin was infused (300 IU/kg of the patient’s body weight) to reach an activated clotting time > 300 seconds to 400 seconds. Added to percutaneous groin access (20-Fr sheath), 2 smaller access sites are required, 1 at opposite groin level (12-Fr sheath with 2 5-Fr angiographic catheters) and the other one at right brachial artery level (7-Fr sheath). The main module was introduced via right brachio-femoral access using the through-and-through wire technique (0.035 in x 450 cm, Hydra Jagwire, Boston Scientific, United States). The side-branch landed on the brachiocephalic artery covering both the aortic arch and the descending aorta (figure 1). Afterwards, through a guidewire (0.035 in x 300 cm, double curved Lunderquist Extra-Stiff Wire, Cook Medical, United States) placed in the left ventricle and during rapid pacing, the ascending aorta module was implanted. After aortic remodelling with a kissing balloon angioplasty (Reliant, Medtronic and 12 mm x 40 mm Armada 35 PTA, Abbott Vascular, United States), a thoracic stent graft was implanted 3 cm above the celiac trunk (E-vita thoracic, Jotec). The patient was discharged 6 days after the endovascular procedure on 75 mg/day of clopidogrel. The coronary computed tomography angiography performed at the 3-month follow-up confirmed the absence of endoleaks and false lumen thrombosis until the end of the implanted stent grafts (figure 2).
Figure 1. A, B, C, D: aortic enlargement after open ascending aorta replacement as seen on the coronary computed tomography angiogram performed at the 6-month follow-up. E: extra-anatomic reconstruction with right common carotid artery to left subclavian artery bypass (both end-to-side), proximal ligation, and end-to-side reimplantation of the right common carotid artery on the graft. F: ascending aorta open replacement, NEXUS Stent Graft System implantation into aortic arch and descending thoracic aorta with false lumen thrombosis.
Figure 2. A: NEXUS Stent Graft main module position. B: NEXUS main module deployment. C: NEXUS ascending module positioning. D: NEXUS complete deployment.
The NEXUS aortic arch stent graft system is indicated for the endovascular management of thoracic aortic diseases involving the aortic arch with proximal landing zone into the ascending aorta and the brachiocephalic artery.
Early experiences with the NEXUS arch graft have been successful. Lindsay et al.3 published a prospective cohort of 5 patients treated with the NEXUS Stent Graft System. No periprocedural strokes occurred. However, postoperative imaging revealed the presence of an ascending aortic hematoma in 1 patient who required ascending aortic replacement. After a 1.5-month-to-13-month follow-up, no endoleaks or other complications were seen.
During 2019, the 2-year results if a prospective, multicenter, premarket study including 25 patients (mean age, 73 years) treated with the NEXUS Stent Graft System were presented.4 Technical success was achieved in all the deployments (100%). At 30 days, 2 patients (8%) died of cardiac causes and another 2 (8%) experienced non-disabling stroke that resolved completely within 30 days. During a mean follow-up of 25-months, there was 1 additional procedure-related death due to stroke, and 1 patient crossed over to open surgery after a retrograde type A dissection.
The practical therapeutic implications include the management of aortic arch diseases like aneurysms and chronic dissections, especially in high-risk surgical patients. This device allows a minimally invasive procedure with no cardiopulmonary bypass, aortic cross-clamping, and circulatory arrest.
The NEXUS Stent Graft System represents the first branched endoprosthesis available off-the-shelf in Europe for the endovascular repair of the aortic arch, especially in high-risk patients with complex aortic arch diseases. It is a promising minimally invasive technique. Still, more experience on this regard and a longer follow-up are needed to confirm the promising mid-term results reported.
FUNDING
None.
AUTHORS' CONTRIBUTION
E.M. San Norberto, N. Cenizo and C. Vaquero performed the interventional procedure, C.M. Flota contributed to case preparation, E.M. San Norberto and C. Vaquero wrote the article in consultation with N. Cenizo and C.M. Flota. All authors provided critical feedback, and helped shape the research and analysis.
CONFLICTS OF INTEREST
None.
REFERENCES
1. Erbel R, Aboyans V, Boileau C, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases:Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014;35:2873-2926.
2. Lindsay T, Ouzounian M, Tan K Early Results of Arch Disease Treated with Nexus Arch Endograft. J Vasc Surg. 2017;66:E67.
3. Clair D. Nexus arch branch stent graft system-Mid-term results. Presented at Vascular Interventional Advances (VIVA) congress 2019. Available at:https://vivaphysicians.org/news-article?id=793.
4. D'Onofrio A, Antonello M, Lachat M, et al. Endovascular treatment of aortic arch aneurysm with a single-branched double-stage stent graft. J Thorac Cardiovasc Surg. 2017;154:e75-e77.